Reproducibility and Validity of a Short Food Frequency Questionnaire for Dietary Assessment in Children Aged 7–9 Years in Spain
Abstract
1. Introduction
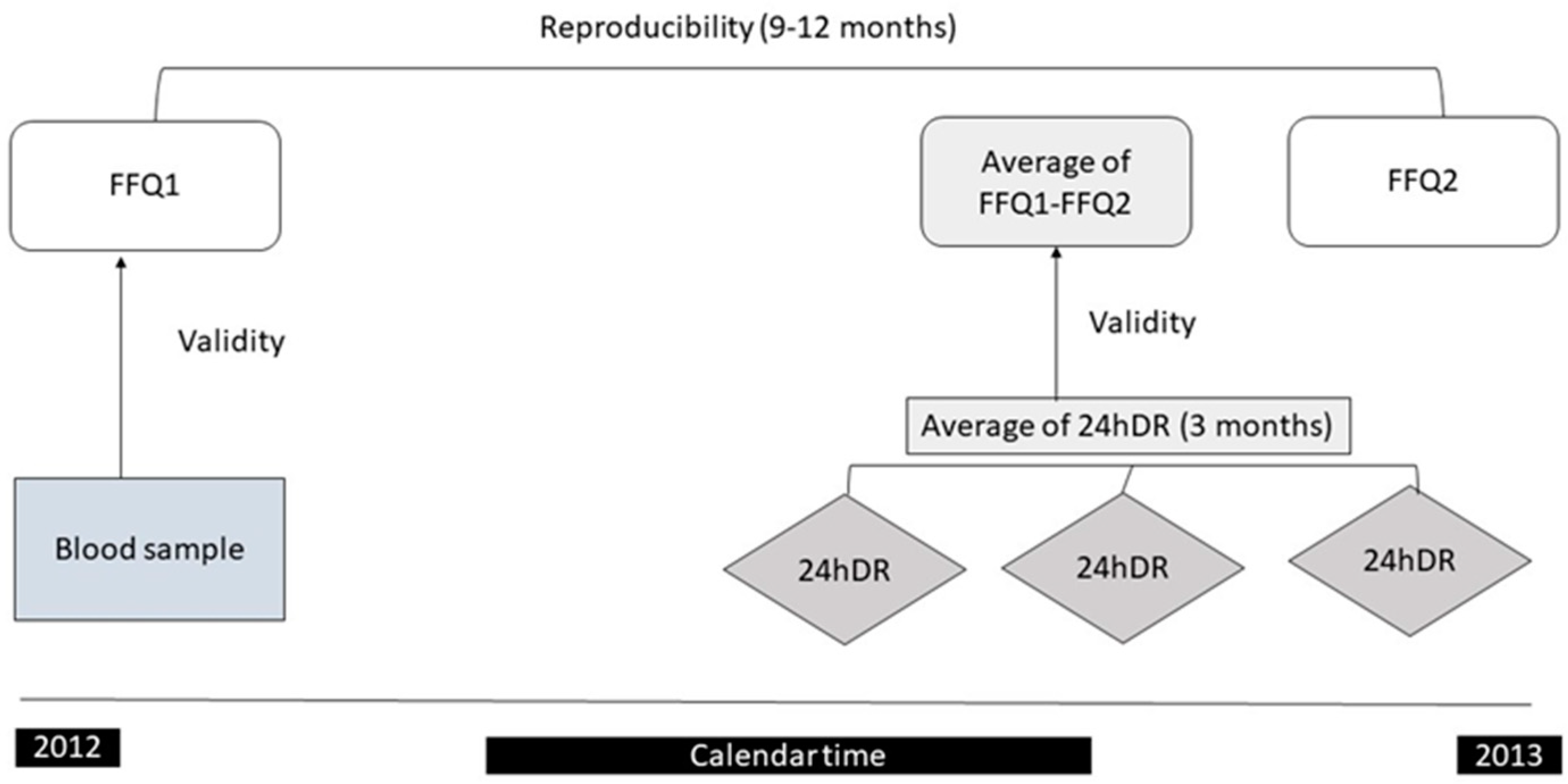
2. Materials and Methods
2.1. Study Population
2.2. Dietary Assessment: Semi-Quantitative Food Frequency Questionnaire (FFQ)
2.3. 24-Hour Dietary Recall (24hDR)
2.4. Nutritional Biomarkers
2.5. Statistical Analysis
3. Results
3.1. Reproducibility
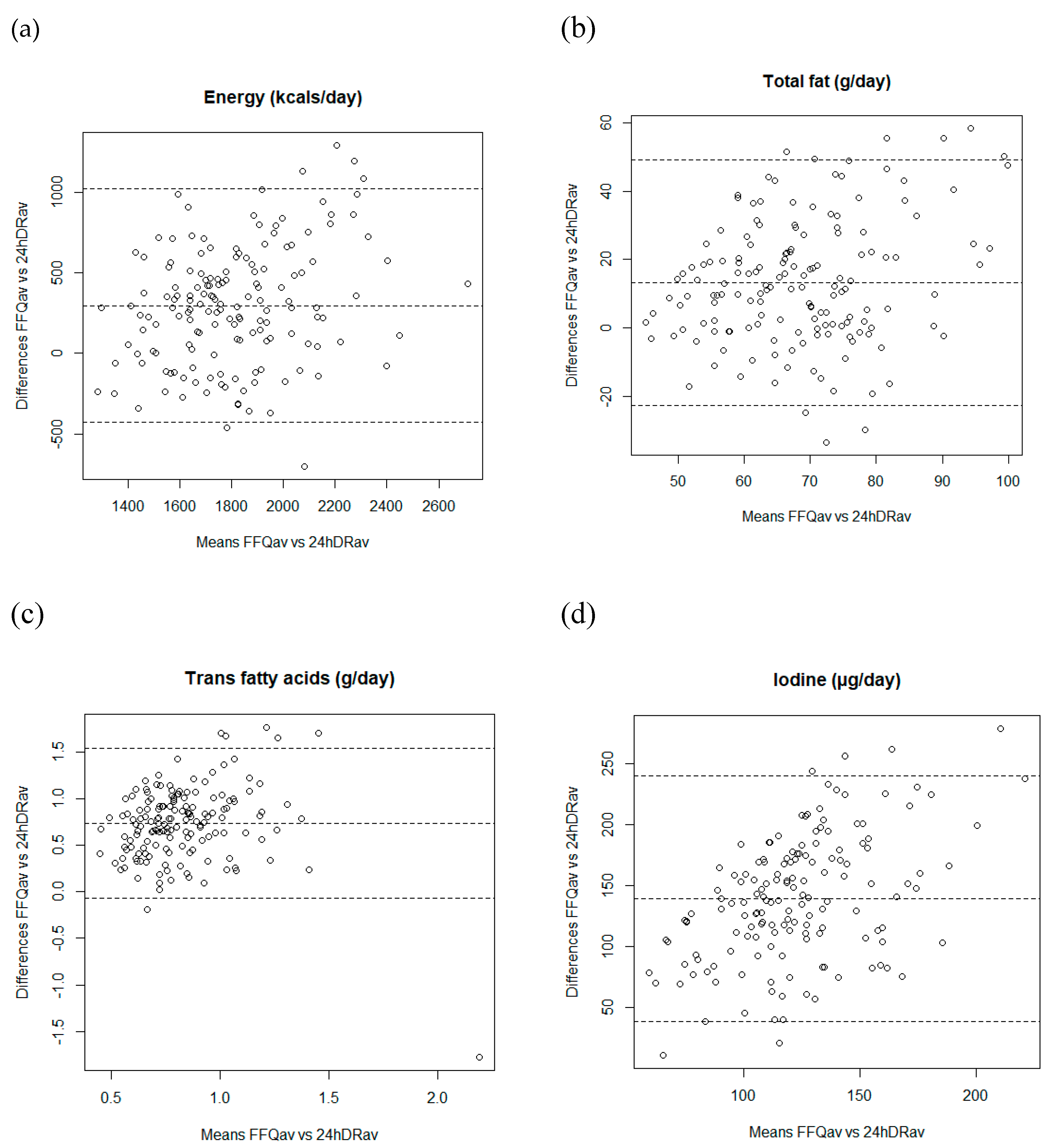
3.2. Validity
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Langley-Evans, S.C. Nutrition in early life and the programming of adult disease: A review. J. Hum. Nutr. Diet. Off. J. Br. Diet. Assoc. 2015, 28 (Suppl. 1), 1–14. [Google Scholar] [CrossRef]
- Koletzko, B.; Brands, B.; Grote, V.; Kirchberg, F.F.; Prell, C.; Rzehak, P.; Uhl, O.; Weber, M. Early Nutrition Programming Project Long-Term Health Impact of Early Nutrition: The Power of Programming. Ann. Nutr. Metab. 2017, 70, 161–169. [Google Scholar] [CrossRef]
- Willett, W.C. Nutritional Epidemiology, 3rd ed.; Oxford University Press: New York, NY, USA, 2012. [Google Scholar]
- Henríquez-Sánchez, P.; Sánchez-Villegas, A.; Doreste-Alonso, J.; Ortiz-Andrellucchi, A.; Pfrimer, K.; Serra-Majem, L. Dietary assessment methods for micronutrient intake: A systematic review on vitamins. Br. J. Nutr. 2009, 102 (Suppl. 1), S10–S37. [Google Scholar] [CrossRef] [PubMed]
- Øverby, N.C.; Serra-Majem, L.; Andersen, L.F. Dietary assessment methods on n-3 fatty acid intake: A systematic review. Br. J. Nutr. 2009, 102 (Suppl. 1), S56–S63. [Google Scholar] [CrossRef]
- Serra-Majem, L.; Pfrimer, K.; Doreste-Alonso, J.; Ribas-Barba, L.; Sánchez-Villegas, A.; Ortiz-Andrellucchi, A.; Henríquez-Sánchez, P. Dietary assessment methods for intakes of iron, calcium, selenium, zinc and iodine. Br. J. Nutr. 2009, 102 (Suppl. 1), S38–S55. [Google Scholar] [CrossRef] [PubMed]
- Ortiz-Andrellucchi, A.; Doreste-Alonso, J.; Henríquez-Sánchez, P.; Cetin, I.; Serra-Majem, L. Dietary assessment methods for micronutrient intake in pregnant women: A systematic review. Br. J. Nutr. 2009, 102 (Suppl. 1), S64–S86. [Google Scholar] [CrossRef] [PubMed]
- Lovell, A.; Bulloch, R.; Wall, C.R.; Grant, C.C. Quality of food-frequency questionnaire validation studies in the dietary assessment of children aged 12 to 36 months: A systematic literature review. J. Nutr. Sci. 2017, 6, e16. [Google Scholar] [CrossRef]
- Ortiz-Andrellucchi, A.; Henríquez-Sánchez, P.; Sánchez-Villegas, A.; Peña-Quintana, L.; Mendez, M.; Serra-Majem, L. Dietary assessment methods for micronutrient intake in infants, children and adolescents: A systematic review. Br. J. Nutr. 2009, 102 (Suppl. 1), S87–S117. [Google Scholar] [CrossRef]
- Tabacchi, G.; Filippi, A.R.; Amodio, E.; Jemni, M.; Bianco, A.; Firenze, A.; Mammina, C. A meta-analysis of the validity of FFQ targeted to adolescents. Public Health Nutr. 2016, 19, 1168–1183. [Google Scholar] [CrossRef]
- Tabacchi, G.; Amodio, E.; Di Pasquale, M.; Bianco, A.; Jemni, M.; Mammina, C. Validation and reproducibility of dietary assessment methods in adolescents: A systematic literature review. Public Health Nutr. 2014, 17, 2700–2714. [Google Scholar] [CrossRef]
- Ortiz-Andrellucchi, A.; Sánchez-Villegas, A.; Doreste-Alonso, J.; de Vries, J.; de Groot, L.; Serra-Majem, L. Dietary assessment methods for micronutrient intake in elderly people: A systematic review. Br. J. Nutr. 2009, 102 (Suppl. 1), S118–S149. [Google Scholar] [CrossRef] [PubMed]
- Del Pino, D.L.; Friedman, R. Adaptation and validation of an FFQ for 6-10-year-old children. Public Health Nutr. 2011, 14, 826–834. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, T.; Kamimura, M.; Imai, S.; Toji, C.; Okamoto, N.; Fukui, M.; Date, C. Reproducibility and validity of the food frequency questionnaire for estimating habitual dietary intake in children and adolescents. Nutr. J. 2011, 10, 27. [Google Scholar] [CrossRef] [PubMed]
- Matos, S.M.A.; Prado, M.S.; Santos, C.A.; D’Innocenzo, S.; Assis, A.M.O.; Dourado, L.S.; Oliveira, N.S.; Rodrigues, L.C.; Barreto, M.L. Validation of a food frequency questionnaire for children and adolescents aged 4 to 11 years living in Salvador, Bahia. Nutr. Hosp. 2012, 27, 1114–1119. [Google Scholar] [CrossRef] [PubMed]
- Moghames, P.; Hammami, N.; Hwalla, N.; Yazbeck, N.; Shoaib, H.; Nasreddine, L.; Naja, F. Validity and reliability of a food frequency questionnaire to estimate dietary intake among Lebanese children. Nutr. J. 2016, 15, 4. [Google Scholar] [CrossRef] [PubMed]
- Preston, A.M.; Palacios, C.; Rodríguez, C.A.; Vélez-Rodríguez, R.M. Validation and reproducibility of a semi-quantitative food frequency questionnaire for use in Puerto Rican children. P. R. Health Sci. J. 2011, 30, 58–64. [Google Scholar] [PubMed]
- Rahmawaty, S.; Charlton, K.; Lyons-Wall, P.; Meyer, B.J. Development and validation of a food frequency questionnaire to assess omega-3 long chain polyunsaturated fatty acid intake in Australian children aged 9-13 years. J. Hum. Nutr. Diet. Off. J. Br. Diet. Assoc. 2017, 30, 429–438. [Google Scholar] [CrossRef]
- Rodriguez, C.A.; Smith, E.R.; Villamor, E.; Zavaleta, N.; Respicio-Torres, G.; Contreras, C.; Perea, S.; Jimenez, J.; Tintaya, K.; Lecca, L.; et al. Development and Validation of a Food Frequency Questionnaire to Estimate Intake among Children and Adolescents in Urban Peru. Nutrients 2017, 9, 1121. [Google Scholar] [CrossRef]
- Saeedi, P.; Skeaff, S.A.; Wong, J.E.; Skidmore, P.M.L. Reproducibility and Relative Validity of a Short Food Frequency Questionnaire in 9–10 Year-Old Children. Nutrients 2016, 8, 271. [Google Scholar] [CrossRef] [PubMed]
- Sahashi, Y.; Tsuji, M.; Wada, K.; Tamai, Y.; Nakamura, K.; Nagata, C. Validity and reproducibility of food frequency questionnaire in Japanese children aged 6 years. J. Nutr. Sci. Vitaminol. 2011, 57, 372–376. [Google Scholar] [CrossRef]
- Scagliusi, F.B.; Garcia, M.T.; Indiani, A.L.C.; Cardoso, M.A. Relative validity of a food-frequency questionnaire developed to assess food intake of schoolchildren living in the Brazilian Western Amazon. Cad. Saude Publica 2011, 27, 2197–2206. [Google Scholar] [CrossRef]
- Guxens, M.; Ballester, F.; Espada, M.; Fernández, M.F.; Grimalt, J.O.; Ibarluzea, J.; Olea, N.; Rebagliato, M.; Tardón, A.; Torrent, M.; et al. INMA Project Cohort Profile: The INMA–INfancia y Medio Ambiente--(Environment and Childhood) Project. Int. J. Epidemiol. 2012, 41, 930–940. [Google Scholar] [CrossRef]
- Vioque, J.; Gimenez-Monzo, D.; Navarrete-Muñoz, E.M.; Garcia-de-la-Hera, M.; Gonzalez-Palacios, S.; Rebagliato, M.; Ballester, F.; Murcia, M.; Iñiguez, C.; Granado, F. INMA-Valencia Cohort Study Reproducibility and Validity of a Food Frequency Questionnaire Designed to Assess Diet in Children Aged 4–5 Years. PLoS ONE 2016, 11, e0167338. [Google Scholar] [CrossRef]
- Willett, W.C.; Sampson, L.; Stampfer, M.J.; Rosner, B.; Bain, C.; Witschi, J.; Hennekens, C.H.; Speizer, F.E. Reproducibility and validity of a semiquantitative food frequency questionnaire. Am. J. Epidemiol. 1985, 122, 51–65. [Google Scholar] [CrossRef]
- Watson, J.F.; Collins, C.E.; Sibbritt, D.W.; Dibley, M.J.; Garg, M.L. Reproducibility and comparative validity of a food frequency questionnaire for Australian children and adolescents. Int. J. Behav. Nutr. Phys. Act. 2009, 6, 62. [Google Scholar] [CrossRef]
- Palma, I.; Farran, A.; Cantos, D. Tablas de Composición de Alimentos por Medidas Caseras de Consumo Habitual en España; McGraw-Hill Interamerican: Madrid, Spain, 2008. [Google Scholar]
- US Department of Agriculture, Agricultural Research Service. USDA National Nutrient Database for Standard Reference, Release 28. Available online: https://www.ars.usda.gov/northeast-area/beltsville-md-bhnrc/beltsville-human-nutrition-research-center/nutrient-data-laboratory/docs/sr28-download-files/ (accessed on 17 January 2018).
- Moshfegh, A.J.; Rhodes, D.G.; Baer, D.J.; Murayi, T.; Clemens, J.C.; Rumpler, W.V.; Paul, D.R.; Sebastian, R.S.; Kuczynski, K.J.; Ingwersen, L.A.; et al. The US Department of Agriculture Automated Multiple-Pass Method reduces bias in the collection of energy intakes. Am. J. Clin. Nutr. 2008, 88, 324–332. [Google Scholar] [CrossRef]
- Vioque, J.; Weinbrenner, T.; Asensio, L.; Castelló, A.; Young, I.S.; Fletcher, A. Plasma concentrations of carotenoids and vitamin C are better correlated with dietary intake in normal weight than overweight and obese elderly subjects. Br. J. Nutr. 2007, 97, 977–986. [Google Scholar] [CrossRef]
- Vioque, J.; Navarrete-Muñoz, E.-M.; Gimenez-Monzó, D.; García-de-la-Hera, M.; Granado, F.; Young, I.S.; Ramón, R.; Ballester, F.; Murcia, M.; Rebagliato, M.; et al. INMA-Valencia Cohort Study Reproducibility and validity of a food frequency questionnaire among pregnant women in a Mediterranean area. Nutr. J. 2013, 12, 26. [Google Scholar] [CrossRef]
- Craft, N.E. Carotenoid reversed-phase high-performance liquid chromatography methods: Reference compendium. Methods Enzymol. 1992, 213, 185–205. [Google Scholar]
- Granado-Lorencio, F.; Herrero-Barbudo, C.; Blanco-Navarro, I.; Pérez-Sacristán, B. Suitability of ultra-high performance liquid chromatography for the determination of fat-soluble nutritional status (vitamins A, E, D, and individual carotenoids). Anal. Bioanal. Chem. 2010, 397, 1389–1393. [Google Scholar] [CrossRef]
- Willett, W.C.; Howe, G.R.; Kushi, L.H. Adjustment for total energy intake in epidemiologic studies. Am. J. Clin. Nutr. 1997, 65, 1220S–1231S; discussion 1229S–1231S. [Google Scholar] [CrossRef] [PubMed]
- Bland, J.; Altman, D. Statistical methods for assessing agreement between two methods of clinical measurement. Lancet 1986, 1, 307–310. [Google Scholar] [CrossRef]
- Arnold, J.E.; Rohan, T.; Howe, G.; Leblanc, M. Reproducibility and validity of a food-frequency questionnaire designed for use in girls age 7 to 12 years. Ann. Epidemiol. 1995, 5, 369–377. [Google Scholar] [CrossRef]
- Field, A.E.; Peterson, K.E.; Gortmaker, S.L.; Cheung, L.; Rockett, H.; Fox, M.K.; Colditz, G.A. Reproducibility and validity of a food frequency questionnaire among fourth to seventh grade inner-city school children: Implications of age and day-to-day variation in dietary intake. Public Health Nutr. 1999, 2, 293–300. [Google Scholar] [CrossRef] [PubMed]
- Grosso, G.; Mistretta, A.; Turconi, G.; Cena, H.; Roggi, C.; Galvano, F. Nutrition knowledge and other determinants of food intake and lifestyle habits in children and young adolescents living in a rural area of Sicily, South Italy. Public Health Nutr. 2013, 16, 1827–1836. [Google Scholar] [CrossRef] [PubMed]
- Buch-Andersen, T.; Pérez-Cueto, F.J.A.; Toft, U. Relative validity and reproducibility of a parent-administered semi-quantitative FFQ for assessing food intake in Danish children aged 3–9 years. Public Health Nutr. 2016, 19, 1184–1194. [Google Scholar] [CrossRef] [PubMed]
- Cade, J.; Thompson, R.; Burley, V.; Warm, D. Development, validation and utilisation of food-frequency questionnaires—A review. Public Health Nutr. 2002, 5, 567–587. [Google Scholar] [CrossRef] [PubMed]
- Livingstone, M.B.; Robson, P.J. Measurement of dietary intake in children. Proc. Nutr. Soc. 2000, 59, 279–293. [Google Scholar] [CrossRef]
- Byers, T.; Trieber, F.; Gunter, E.; Coates, R.; Sowell, A.; Leonard, S.; Mokdad, A.; Jewell, S.; Miller, D.; Serdula, M. The accuracy of parental reports of their children’s intake of fruits and vegetables: Validation of a food frequency questionnaire with serum levels of carotenoids and vitamins C, A, and E. Epidemiology 1993, 4, 350–355. [Google Scholar] [CrossRef]
- Burrows, T.L.; Warren, J.M.; Colyvas, K.; Garg, M.L.; Collins, C.E. Validation of overweight children’s fruit and vegetable intake using plasma carotenoids. Obesity 2009, 17, 162–168. [Google Scholar] [CrossRef]
- Neuhouser, M.L.; Rock, C.L.; Eldridge, A.L.; Kristal, A.R.; Patterson, R.E.; Cooper, D.A.; Neumark-Sztainer, D.; Cheskin, L.J.; Thornquist, M.D. Serum concentrations of retinol, alpha-tocopherol and the carotenoids are influenced by diet, race and obesity in a sample of healthy adolescents. J. Nutr. 2001, 131, 2184–2191. [Google Scholar] [CrossRef]
- Rodríguez-Dehli, A.C.; Riaño-Galán, I.; Fernández-Somoano, A.; Navarrete-Muñoz, E.M.; Espada, M.; Vioque, J.; Tardón, A. [Hypovitaminosis D and associated factors in 4-year old children in northern Spain]. An. Pediatr. 2017, 86, 188–196. [Google Scholar] [CrossRef]
- Willett, W.C.; Stampfer, M.J.; Underwood, B.A.; Speizer, F.E.; Rosner, B.; Hennekens, C.H. Validation of a dietary questionnaire with plasma carotenoid and alpha-tocopherol levels. Am. J. Clin. Nutr. 1983, 38, 631–639. [Google Scholar] [CrossRef]
| Food Groups (Number of Foods Items) | Foods |
|---|---|
| Dairy Products (4) | whole dairy products; semi-skimmed, skimmed and fortified dairy products; Petit Suisse; cheese. |
| Eggs (1) | Eggs |
| White meat (1) | chicken or turkey |
| Red meat (1) | beef, pork or lamb |
| Processed meat (3) | sausages; ham, salami and others; serrano ham |
| Fatty fish (3) | swordfish, bonito, and fresh tuna; small oily fish (mackerel, sardine; anchovy); canned sardine or mackerel |
| Lean fish (1) | A assorted or mixed fried fish (hake; gilthead sea bream and sole) |
| Seafood (2) | clams, mussels, squid, octopus, shellfish (cramps, shrimps, lobster); surimi and other fish-based food products |
| Fruits (2) | oranges; other fruit (apples; banana; pears; watermelon; melon; peach; kiwi; cherries, strawberries) |
| Vegetables (2) | raw vegetables (tomatoes; onions; lettuces; peppers and carrots) and cooked vegetables (spinach; cabbage, cauliflower or broccoli; carrots or squash; eggplant, zucchini, or cucumber; green, red, or yellow peppers) |
| Nuts (1) | almonds, walnuts, peanuts and other types of nuts |
| Legumes (1) | lentils, chickpeas, beans, peas, and green beans |
| Cereals and Pasta (2) | breakfast cereals; corn, rice and pasta |
| Bread (2) | white and whole breads |
| Potatoes (2) | frozen French fry; homemade boiled/stew |
| Sweets and sugar (5) | biscuits and baked goods; biscuits and baked goods with chocolate; peanut butter (e.g., Nutella/Nocilla); chocolate/cocoa powder; candies, marmalade, and honey |
| Sweetened beverages (3) | packages juices; sugar soft drinks and artificially soft drinks |
| Participation in the Validation Study | p-Value 1 | ||
|---|---|---|---|
| Yes (n = 156) | No (n = 305) | ||
| Maternal Characteristics | |||
| Age (years), mean (standard deviation (SD)) | 30.0 (4.0) | 30.8 (4.2) | 0.058 |
| Education (University studies), % | 31.4 | 29.5 | 0.645 |
| Social class (I + II; high), % | 22.4 | 19.7 | 0.469 |
| Country of birth (Spain), % | 88.5 | 86.2 | 0.560 |
| Child Characteristics | |||
| Age (years), mean (SD) | 7.6 (0.1) | 7.6 (0.2) | 0.666 |
| Sex (female), % | 45.5 | 51.8 | 0.237 |
| Body Mass Index (Kg/m2), mean (SD) | 17.1 (2.7) | 18.0 (3.0) | 0.001 |
| School lunchroom (≥1 time/week), % | 62.8 | 64.8 | 0.682 |
| Energy (Kcals/day), mean (SD) | 1960 (459) | 1902 (435) | 0.188 |
| Proteins (g/day), mean (SD) | 87 (18) | 85 (18) | 0.391 |
| Carbohydrates (g/day), mean (SD) | 245 (65) | 233 (62) | 0.054 |
| Fats (g/day), mean (SD) | 75 (20) | 74 (19) | 0.725 |
| Nutrient Intakes (units/day) | FFQ1 1 | FFQ2 1 | p-Value 2 | Pearson Coefficient Correlations between FFQ1 and FFQ2 | % of Agreement 5 | |
|---|---|---|---|---|---|---|
| Mean (SD) | Mean (SD) | r 3 | r adj.4 | |||
| Energy (kcals/day) | 1953 (458) | 1962 (410) | 0.852 | 0.34 | 65.4 | |
| Protein (g/day) | 87 (18) | 85 (17) | 0.344 | 0.29 | 0.48 | 62.8 |
| Total carbohydrates (g/day) | 245 (65) | 243 (60) | 0.698 | 0.36 | 0.40 | 68.6 |
| Dietary fiber (g/day) | 20.3 (6.1) | 21.1 (6.1) | 0.226 | 0.43 | 0.51 | 66.7 |
| Cholesterol | 286 (69) | 284 (69) | 0.849 | 0.43 | 0.47 | 71.8 |
| Total fat (g/day) | 73 (20) | 77 (20) | 0.125 | 0.35 | 0.32 | 67.3 |
| SFA (g/ day) | 25.7 (7.9) | 25.6 (7.6) | 0.903 | 0.34 | 0.42 | 62.2 |
| MUFA (g/day) | 28.2 (8.7) | 31.2 (9) | 0.004 | 0.33 | 0.24 | 59.6 |
| PUFA (g/day) | 12.6 (3.7) | 13.4 (4) | 0.101 | 0.42 | 0.49 | 67.9 |
| Omega 3 (g/day) | 1.3 (0.4) | 1.4 (0.4) | 0.411 | 0.43 | 0.57 | 67.9 |
| Omega 6 (g/day) | 11.1 (3.3) | 11.8 (3.7) | 0.083 | 0.42 | 0.47 | 67.9 |
| Trans fatty acid (g/day) | 1.2 (0.4) | 1.2 (0.4) | 0.386 | 0.32 | 0.42 | 75.6 |
| Retinol (μg/day) | 452 (216) | 440 (174) | 0.594 | 0.44 | 0.47 | 75.6 |
| α-carotene (μg/day) | 816 (647) | 908 (717) | 0.236 | 0.64 | 0.63 | 80.8 |
| β-carotene (μg/day) | 2721 (2038) | 3083(2259) | 0.138 | 0.65 | 0.65 | 81.4 |
| β-Cryptoxanthin (μg/day) | 159 (102) | 203 (145) | 0.002 | 0.54 | 0.49 | 71.8 |
| Lutein + Zeaxanthin (μg/day) | 1451 (780) | 1481 (939) | 0.758 | 0.54 | 0.55 | 73.7 |
| Lycopene (μg/day) | 2800 (1355) | 2942 (1682) | 0.412 | 0.44 | 0.45 | 70.5 |
| Vitamin B6 (mg/day) | 1.7 (0.7) | 1.7 (0.6) | 0.962 | 0.47 | 0.49 | 71.2 |
| Folate (μg/day) | 213 (50) | 222 (61) | 0.164 | 0.41 | 0.39 | 70.5 |
| Vitamin B12 (μg/day) | 6.4 (2.3) | 6.3 (1.9) | 0.497 | 0.39 | 0.42 | 69.2 |
| Vitamin C (mg/day) | 78 (41) | 95 (58) | 0.002 | 0.51 | 0.46 | 72.4 |
| Vitamin D (μg/day) | 4.4 (1.6) | 4.4 (1.5) | 0.789 | 0.47 | 0.54 | 69.9 |
| Vitamin E (mg/day) | 7.6 (2.3) | 8.4 (2.6) | 0.002 | 0.39 | 0.44 | 63.5 |
| Calcium (mg/day) | 1135 (344) | 1105 (310) | 0.429 | 0.25 | 0.35 | 65.4 |
| Iron (mg/day) | 13 (5.2) | 13 (4.5) | 0.942 | 0.45 | 0.46 | 72.4 |
| Magnesium (mg/day) | 284 (64) | 282 (64) | 0.752 | 0.31 | 0.43 | 64.7 |
| Potassium (mg/day) | 2730 (607) | 2731 (645) | 0.989 | 0.33 | 0.41 | 69.2 |
| Sodium (mg/day) | 2812 (703) | 2746 (552) | 0.355 | 0.30 | 0.43 | 64.7 |
| Zinc (mg/day) | 10.1 (2.6) | 9.8 (2.2) | 0.326 | 0.32 | 0.40 | 66.0 |
| Iodine (μg/day) | 196 (57) | 186 (55) | 0.112 | 0.48 | 0.59 | 73.1 |
| Average of correlation coefficients | 0.41 | 0.46 | 69.2 | |||
| Food Groups (g/day) | FFQ1 1 | FFQ2 1 | p-Value 2 | Pearson Coefficient Correlations between FFQ1 and FFQ2 | |
|---|---|---|---|---|---|
| Mean (SD) | Mean (SD) | r 3 | r adj. 4 | ||
| Dairy products | 561 (244) | 505 (211) | 0.031 | 0.25 | 0.32 |
| Eggs | 22 (9) | 24 (9) | 0.046 | 0.59 | 0.57 |
| White meat | 40 (15) | 38 (14) | 0.497 | 0.42 | 0.42 |
| Red meat | 27 (15) | 25 (15) | 0.282 | 0.40 | 0.40 |
| Processed meat | 35 (22) | 32 (14) | 0.138 | 0.37 | 0.39 |
| White fish | 24 (13) | 23 (13) | 0.560 | 0.58 | 0.57 |
| Blue fish | 16 (14) | 18 (14) | 0.288 | 0.61 | 0.63 |
| Seafood | 9 (15) | 7 (9) | 0.131 | 0.45 | 0.45 |
| Vegetables | 95 (73) | 103 (82) | 0.392 | 0.60 | 0.60 |
| Fruit | 160 (107) | 218 (145) | <0.001 | 0.67 | 0.65 |
| Nuts | 4 (4) | 4 (4) | 0.785 | 0.28 | 0.30 |
| Pulse | 33 (21) | 31 (19) | 0.458 | 0.57 | 0.56 |
| Cereals | 82 (50) | 81 (38) | 0.846 | 0.18 | 0.15 |
| Potatoes | 40 (20) | 39 (19) | 0.628 | 0.34 | 0.32 |
| Bread | 98 (51) | 99 (48) | 0.872 | 0.35 | 0.39 |
| Sweet and sugar | 75 (52) | 68 (47) | 0.200 | 0.34 | 0.35 |
| Sweetened beverages | 141 (153) | 137 (142) | 0.807 | 0.68 | 0.68 |
| Average of correlation coefficients | 0.45 | 0.46 | |||
| Nutrients (units/day) | FFQav 1 | 24hDRav 2 | p-Value 3 | Pearson Coefficient Correlations between FFQav and 24hDRav | % of Agreement 7 | ||
|---|---|---|---|---|---|---|---|
| Mean (SD) | Mean (SD) | r 4 | r adj. 5 | r de-att. 6 | |||
| Energy (kcals/day) | 1957 (354) | 1662 (271) | <0.001 | 0.31 | 62.2 | ||
| Protein (g/day) | 86 (14) | 70 (13) | <0.001 | 0.25 | 0.36 | 0.42 | 51.9 |
| Total carbohydrates (g/day) | 244 (52) | 211 (43) | <0.001 | 0.40 | 0.34 | 0.39 | 65.4 |
| Dietary fiber (g/day) | 20.7 (5.2) | 12.9 (3.6) | <0.001 | 0.32 | 0.29 | 0.30 | 57.1 |
| Cholesterol (g/day) | 285 (58) | 229 (71) | <0.001 | 0.23 | 0.28 | 0.31 | 58.3 |
| Total fat (g/ day) | 75 (16) | 62 (13) | <0.001 | 0.24 | 0.32 | 0.34 | 57.7 |
| SFA (g/ day) | 25.6 (6.3) | 21.9 (5.2) | <0.001 | 0.35 | 0.32 | 0.35 | 60.3 |
| MUFA (g/day) | 29.7 (7.2) | 24.1 (5.8) | <0.001 | 0.18 | 0.26 | 0.28 | 58.3 |
| PUFA (g/ day) | 13 (3.2) | 9.8 (3.1) | <0.001 | 0.24 | 0.38 | 0.50 | 58.3 |
| Omega 3 (g/day) | 1.3 (0.4) | 1 (0.3) | <0.001 | 0.24 | 0.31 | 0.37 | 57.7 |
| Omega 6 (g/day) | 11.5 (2.9) | 8.7 (2.8) | <0.001 | 0.25 | 0.38 | 0.54 | 63.5 |
| Trans fatty acids (g/day) | 1.2 (0.3) | 0.5 (0.3) | <0.001 | 0.44 | 0.48 | 0.60 | 67.3 |
| Retinol (μg/day) | 446 (165) | 374 (205) | 0.001 | 0.46 | 0.45 | 0.57 | 71.2 |
| α-Carotene (μg/day) | 862 (601) | 200 (301) | <0.001 | 0.32 | 0.31 | 0.33 | 61.5 |
| β-Carotene (μg/day) | 2902 (1899) | 955 (944) | <0.001 | 0.43 | 0.43 | 0.45 | 64.1 |
| β- Cryptoxanthin (μg/day) | 181 (102) | 94 (95) | <0.001 | 0.48 | 0.48 | 0.54 | 64.7 |
| Lutein + Zeaxanthin (μg/day) | 1466 (726) | 655 (924) | <0.001 | 0.24 | 0.24 | 0.26 | 57.1 |
| Lycopene (μg/day) | 2871 (1300) | 2679 (2028) | 0.319 | 0.22 | 0.20 | 0.20 | 60.9 |
| Vitamin B6 (mg/day) | 1.7 (0.5) | 1.4 (0.5) | <0.001 | 0.46 | 0.49 | 0.50 | 68.6 |
| Folate (μg/day) | 218 (47) | 151 (50) | <0.001 | 0.25 | 0.28 | 0.29 | 60.3 |
| Vitamin B12 (μg/day) | 6.4 (1.8) | 5.8 (5.7) | 0.223 | 0.28 | 0.30 | 0.39 | 64.1 |
| Vitamin C (mg /day) | 87 (41) | 53 (35) | <0.001 | 0.43 | 0.44 | 0.56 | 64.1 |
| Vitamin D (μg/day) | 4.4 (1.3) | 2.7 (1.8) | <0.001 | 0.22 | 0.24 | 0.28 | 59.6 |
| Vitamin E (mg/day) | 8 (2) | 5.1 (1.7) | <0.001 | 0.19 | 0.23 | 0.26 | 57.7 |
| Calcium (mg/day) | 1120 (264) | 898 (240) | <0.001 | 0.38 | 0.45 | 0.50 | 59.6 |
| Iron (mg/day) | 13 (4.1) | 10.7 (4.3) | <0.001 | 0.41 | 0.37 | 0.43 | 66.7 |
| Magnesium (mg/day) | 283 (52) | 211 (39) | <0.001 | 0.36 | 0.39 | 0.41 | 61.5 |
| Potassium (mg/day) | 2731 (509) | 2124 (462) | <0.001 | 0.32 | 0.48 | 0.51 | 63.5 |
| Sodium (mg/day) | 2779 (504) | 2210 (511) | <0.001 | 0.25 | 0.29 | 0.32 | 57.7 |
| Zinc (mg/day) | 9.9 (2) | 8.1 (1.8) | <0.001 | 0.39 | 0.29 | 0.31 | 62.8 |
| Iodine (μg/day) | 191 (48) | 80 (44) | <0.001 | 0.14 | 0.19 | 0.20 | 57.8 |
| Average of correlation coefficients | 0.31 | 0.34 | 0.39 | 61.4 | |||
| Nutrients | FFQ1 1 | Vitamin Biomarkers | Pearson Coefficient Correlations between FFQ1 and Plasma Concentrations | Agreement (%) 4 | |
|---|---|---|---|---|---|
| Mean (SD) | Mean (SD) | r 2 | r adj 3 | ||
| Vitamin E | 7.6 (2.3) | 961 (173) | 0.05 | 0.10 | 56.4 |
| Vitamin D | 4.4 (1.6) | 66 (20) | 0.06 | 0.11 | 53.2 |
| Retinol | 452 (216) | 25 (6) | −0.09 | 0.05 | 51.9 |
| Carotenoids | |||||
| Lutein + Zeaxanthin | 1451 (780) | 13.6 (6.3) | 0.06 | 0.04 | 50.0 |
| β- Cryptoxanthin | 159 (102) | 14 (12) | 0.38 | 0.38 | 65.4 |
| Lycopene | 2800 (1355) | 28 (11) | 0.24 | 0.26 | 62.8 |
| α-Carotene | 816 (647) | 7.6 (4.7) | 0.23 | 0.23 | 55.1 |
| β-Carotene | 2721 (2038) | 24 (16) | 0.13 | 0.15 | 56.4 |
| Average of correlation coefficients | 0.21 | 0.21 | 59.0 | ||
| Fruits and vegetables vs. total carotenoids | 140 (153) | 88 (34) | 0.19 | 0.19 | 60.9 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vioque, J.; Garcia-de-la-Hera, M.; Gonzalez-Palacios, S.; Torres-Collado, L.; Notario-Barandiaran, L.; Oncina-Canovas, A.; Soler-Blasco, R.; Lozano, M.; Beneito, A.; Navarrete-Muñoz, E.-M. Reproducibility and Validity of a Short Food Frequency Questionnaire for Dietary Assessment in Children Aged 7–9 Years in Spain. Nutrients 2019, 11, 933. https://doi.org/10.3390/nu11040933
Vioque J, Garcia-de-la-Hera M, Gonzalez-Palacios S, Torres-Collado L, Notario-Barandiaran L, Oncina-Canovas A, Soler-Blasco R, Lozano M, Beneito A, Navarrete-Muñoz E-M. Reproducibility and Validity of a Short Food Frequency Questionnaire for Dietary Assessment in Children Aged 7–9 Years in Spain. Nutrients. 2019; 11(4):933. https://doi.org/10.3390/nu11040933
Chicago/Turabian StyleVioque, Jesus, Manuela Garcia-de-la-Hera, Sandra Gonzalez-Palacios, Laura Torres-Collado, Leyre Notario-Barandiaran, Alejandro Oncina-Canovas, Raquel Soler-Blasco, Manuel Lozano, Andrea Beneito, and Eva-Maria Navarrete-Muñoz. 2019. "Reproducibility and Validity of a Short Food Frequency Questionnaire for Dietary Assessment in Children Aged 7–9 Years in Spain" Nutrients 11, no. 4: 933. https://doi.org/10.3390/nu11040933
APA StyleVioque, J., Garcia-de-la-Hera, M., Gonzalez-Palacios, S., Torres-Collado, L., Notario-Barandiaran, L., Oncina-Canovas, A., Soler-Blasco, R., Lozano, M., Beneito, A., & Navarrete-Muñoz, E.-M. (2019). Reproducibility and Validity of a Short Food Frequency Questionnaire for Dietary Assessment in Children Aged 7–9 Years in Spain. Nutrients, 11(4), 933. https://doi.org/10.3390/nu11040933








