Mean Dietary Salt Intake in Vanuatu: A Population Survey of 755 Participants on Efate Island
Abstract
:1. Introduction
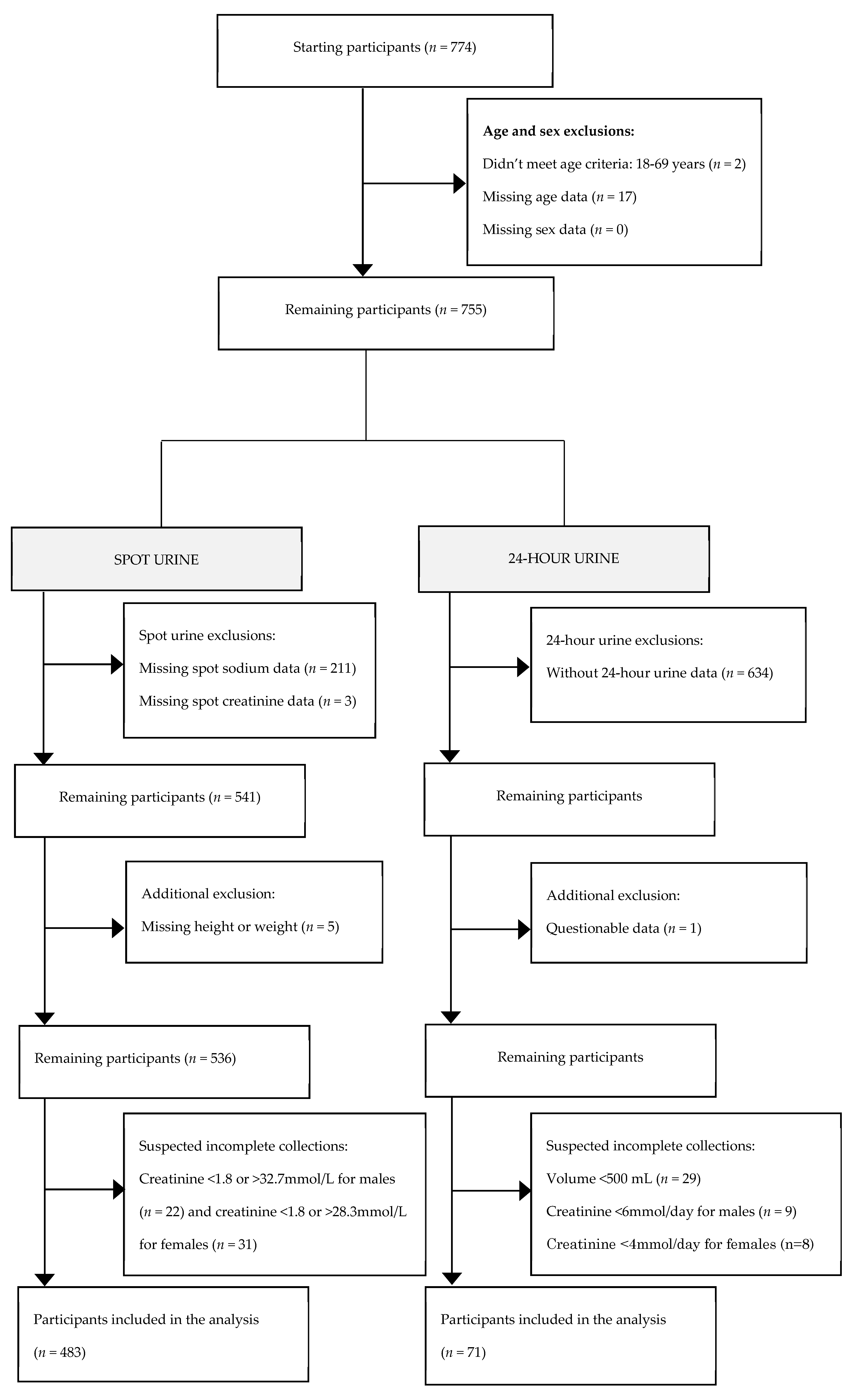
2. Materials and Methods
2.1. Survey Population and Sampling
2.2. Data Collection
2.3. Biochemical Assessment
2.4. Statistical Analyses
3. Results
Estimated Salt Intake from Urine Samples
4. Discussion
Recommendations
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Disclaimer
Appendix A
| Characteristics | Spot Urine | 24-h Urine | p-Value |
|---|---|---|---|
| (n = 483) | (n = 71) | ||
| Age in years, mean (SD) | 36.6 (12.6) | 36.4 (12.6) | 0.898 |
| Female, % | 49.2 | 49.2 | 1.000 |
| Area, % | |||
| Rural | 34.1 | 34.0 | 1.000 |
| Urban | 66.0 | 66.0 | |
| Completed Education, % | |||
| No formal schooling | 2.5 | 1.8 | |
| Primary level | 43.6 | 54.4 | 0.386 |
| Secondary level | 41.7 | 36.1 | |
| Tertiary level | 12.1 | 7.8 | |
| Employed, % | 52.8 | 48.4 | 0.540 |
| Height in cm, mean (SD) | 164.1 (7.9) | 165.2 (9.3) | 0.284 |
| Weight in kg, mean (SD) | 73.6 (15.6) | 76.4 (15.1) | 0.162 |
| Body mass index in kg/m2, mean (SD) | 27.3 (5.4) | 28.1 (5.8) | 0.271 |
| Systolic blood pressure in mmHg, mean (SD) | 121.7 (19.2) | 123.9 (19.2) | 0.385 |
| Diastolic blood pressure in mmHg, mean (SD) | 77.9 (12.7) | 79.9 (13.2) | 0.224 |
| History of hypertension, % | 10.3 | 12.2 | 0.634 |
| History of high cholesterol in blood, % | 3.7 | 4.3 | 0.799 |
| History of heart attack, % | 1.8 | 2.5 | 0.766 |
| History of stroke, % | 1.1 | 0.0 | 0.413 |
| History of diabetes, % | 2.0 | 0.0 | 0.243 |
| History of chronic kidney disease, % | 1.8 | 2.6 | 0.741 |
Appendix B
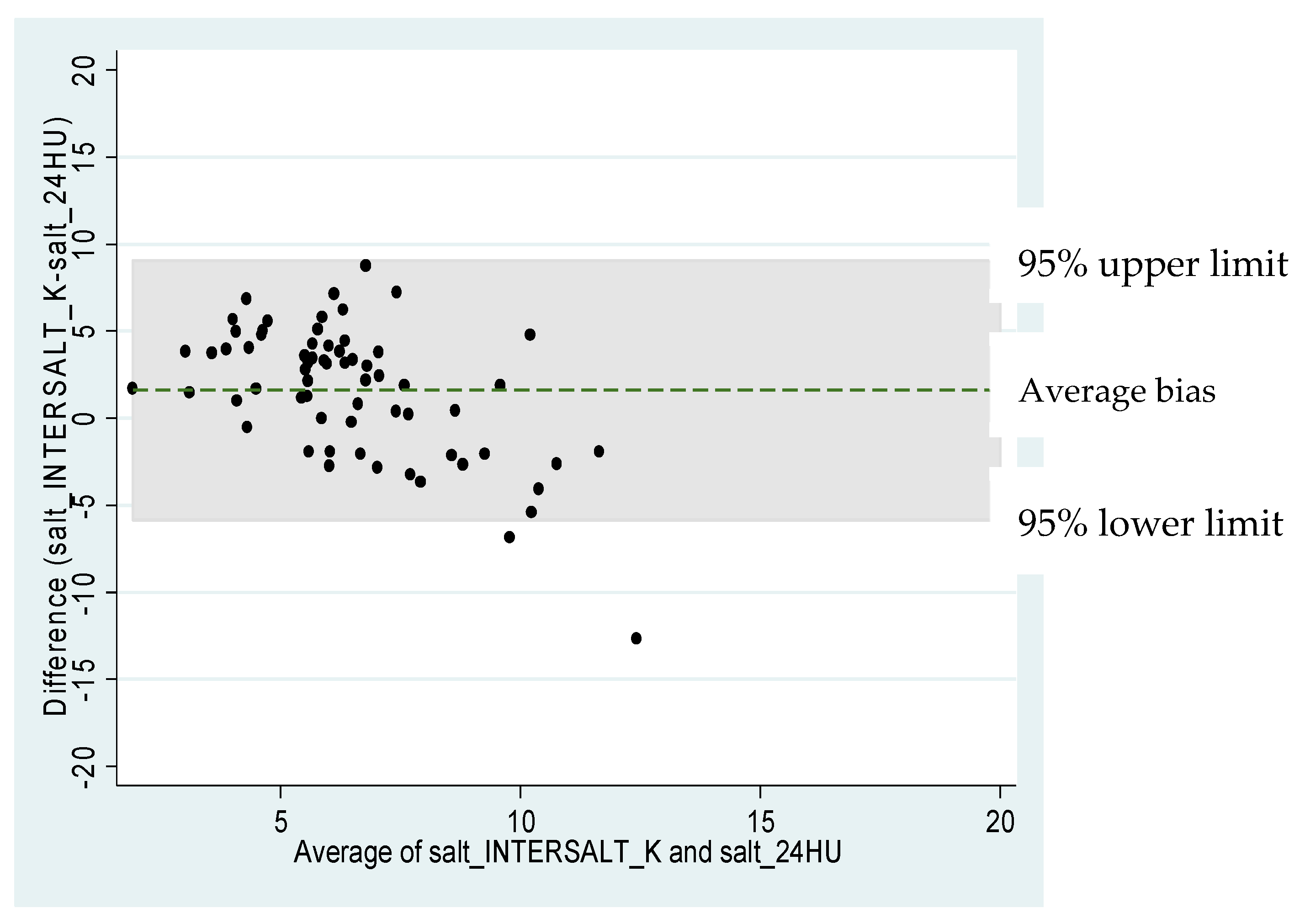
References
- Aburto, N.J.; Aburto, N.J.; Ziolkovska, A.; Hooper, L.; Elliott, P.; Cappuccio, F.P.; Meerpohl, J.J. Effect of lower sodium intake on health: Systematic review and meta-analyses. Br. Med. J. 2013, 346, f1326. [Google Scholar] [CrossRef]
- World Health Organization. Global Action Plan for the Prevention and Control of Noncommunicable Diseases 2013–2020; World Health Organization: Geneva, Switzerland, 2013. [Google Scholar]
- Roth, G.A.; Johnson, C.; Abajobir, A.; Abd-Allah, F.; Abera, S.F.; Abyu, G.; Ahmed, M.; Aksut, B.; Alam, T.; Alam, K. Global, regional, and national burden of cardiovascular diseases for 10 causes, 1990 to 2015. J. Am. Coll. Cardiol. 2017, 70, 1–25. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Global Status Report on Noncommunicable Diseases 2014; World Health Organization: Geneva, Switzerland, 2014. [Google Scholar]
- World Health Organization. Salt Reduction Fact Sheet. Available online: http://www.who.int/mediacentre/factsheets/fs393/en/ (accessed on 15 November 2017).
- World Health Organization. World Economic Forum. From Burden to “Best Buys”: Reducing the Economic Impact of Non-Communicable Diseases in Low-and Middle-Income Countries; World Health Organization: Geneva, Switzerland, 2011. [Google Scholar]
- World Health Organization. Prevention of Cardiovascular Disease: Guidelines for Assessment and Management of Cardiovascular Risk; World Health Organization: Geneva, Switzerland, 2007. [Google Scholar]
- Mozaffarian, D.; Fahimi, S.; Singh, G.M.; Micha, R.; Khatibzadeh, S.; Engell, R.E.; Lim, S.; Danaei, G.; Ezzati, M.; Powles, J.; et al. Global sodium consumption and death from cardiovascular causes. N. Engl. J. Med. 2014, 371, 624. [Google Scholar] [CrossRef] [PubMed]
- The World Bank Group. Vanuatu. Available online: https://data.worldbank.org/country/Vanuatu (accessed on 26 February 2018).
- World Health Organization. Noncommunicable Diseases Country Profiles 2011; World Health Organization: Geneva, Switzerland, 2011. [Google Scholar]
- World Health Organization. Vanuatu NCD Risk Factors STEPS Report; World Health Organization: Geneva, Switzerland, 2013. [Google Scholar]
- The World Bank Group. The Economic Cost of Noncommunicable Diseases in the Pacific Islands; The World Bank: Washington, DC, USA, 2012. [Google Scholar]
- Anderson, I.; Sanburg, A.; Aru, H.; Tarivonda, L.; Ivatts, S.; Latu, R.; Kool, J. The costs and affordability of drug treatments for type 2 diabetes and hypertension in Vanuatu. Pac. Health Dialog 2013, 19, 1. [Google Scholar]
- Thow, A.M.; Heywood, P.; Schultz, J.; Quested, C.; Jan, S.; Colagiuri, S. Trade and the nutrition transition: Strengthening policy for health in the pacific. Ecol. Food Nutr. 2011, 50, 18–42. [Google Scholar] [CrossRef]
- World Health Organization. Salt Matters for Pacific Island Countries: Mobilizing for Effective Action to Reduce Population Salt Intake in the Pacific Island Countries; WHO Regional Office for the Western Pacific: Manila, Philippines, 2014; p. 74. [Google Scholar]
- Snowdon, W.; Thow, A.M. Trade policy and obesity prevention: Challenges and innovation in the pacific islands. Obes. Rev. 2013, 14, 150–158. [Google Scholar] [CrossRef]
- ’Ofa, S.V.; Gani, A. Trade policy and health implication for pacific island countries. Int. J. Soc. Econ. 2017, 44, 816–830. [Google Scholar]
- Christoforou, A.; Snowdon, W.; Laesango, N.; Vatucawaqa, S.; Lamar, D.; Alam, L.; Lippwe, K.; Havea, I.L.; Tairea, K.; Hoejskov, P.; et al. Progress on salt reduction in the pacific islands: From strategies to action. HeartLung Circ. 2015, 24, 503–509. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Strategies to Monitor and Evaluate Population Sodium Consumption and Sources of Sodium in the Diet: Report of a Joint Technical Meeting Convened by who and the Government of Canada; World Health Organization: Geneva, Switzerland, 2011. [Google Scholar]
- McLean, R.M. Measuring population sodium intake: A review of methods. Nutrients 2014, 6, 4651–4662. [Google Scholar] [CrossRef] [PubMed]
- Pillay, A.; Trieu, K.; Santos, J.A.; Sukhu, A.; Schultz, J.; Wate, J.; Bell, C.; Moodie, M.; Snowdon, W.; Ma, G.; et al. Assessment of a salt reduction intervention on adult population salt intake in Fiji. Nutrients 2017, 9, 1350. [Google Scholar] [CrossRef] [PubMed]
- Trieu, K.; Ieremia, M.; Santos, J.; Neal, B.; Woodward, M.; Moodie, M.; Bell, C.; Snowdon, W.; Faumuina, T.; Webster, J. Effects of a nationwide strategy to reduce salt intake in Samoa. J. Hypertens. 2018, 36, 188–198. [Google Scholar] [CrossRef] [PubMed]
- Vanuatu National Statistics Office. Mini—Census Report. Available online: https://vnso.gov.vu/index.php/2-uncategorised/153-mini-census (accessed on 16 July 2018).
- World Health Organization. Who STEPS Instrument (Core and Expanded). Available online: https://www.who.int/ncds/surveillance/steps/instrument/en/ (accessed on 16 July 2018).
- Singh, A.S.; Masuku, M.B. Sampling techniques & determination of sample size in applied statistics research: An overview. Int. J. Econ. Commer. Manag. 2014, 2, 1–22. [Google Scholar]
- Brown, I.J.; Dyer, A.R.; Chan, Q.; Cogswell, M.E.; Ueshima, H.; Stamler, J.; Elliott, P. Estimating 24-hour urinary sodium excretion from casual urinary sodium concentrations in western populations. Am. J. Epidemiol. 2013, 177, 1180. [Google Scholar] [CrossRef] [PubMed]
- Do, H.T.P.; Santos, J.A.; Trieu, K.; Petersen, K.; Le, M.B.; Lai, D.T.; Bauman, A.; Webster, J. Effectiveness of a communication for behavioral impact (COMBI) intervention to reduce salt intake in a vietnamese province based on estimations from spot urine samples. J. Clin. Hypertens. 2016, 18, 1135–1142. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T.; Okamura, T.; Miura, K.; Kadowaki, T.; Ueshima, H.; Nakagawa, H.; Hashimoto, T. A simple method to estimate populational 24-h urinary sodium and potassium excretion using a casual urine specimen. J. Hum. Hypertens. 2002, 16, 97. [Google Scholar] [CrossRef]
- Petersen, K.S.; Wu, J.H.Y.; Webster, J.; Grimes, C.; Woodward, M.; Nowson, C.A.; Neal, B. Estimating mean change in population salt intake using spot urine samples. Int. J. Epidemiol. 2016, 46, 1542–1550. [Google Scholar] [CrossRef]
- Huang, L.; Crino, M.; Wu, J.H.Y.; Woodward, M.; Barzi, F.; Land, M.-A.; McLean, R.; Webster, J.; Enkhtungalag, B.; Neal, B. Mean population salt intake estimated from 24-h urine samples and spot urine samples: A systematic review and meta-analysis. Int. J. Epidemiol. 2016, 45, 239–250. [Google Scholar] [CrossRef]
- Powles, J.; Fahimi, S.; Micha, R.; Khatibzadeh, S.; Shi, P.; Ezzati, M.; Engell, R.E.; Lim, S.S.; Danaei, G.; Mozaffarian, D. Global, regional and national sodium intakes in 1990 and 2010: A systematic analysis of 24 h urinary sodium excretion and dietary surveys worldwide. BMJ Open 2013, 3, e003733. [Google Scholar] [CrossRef]
- Martyn, T.; Yi, D.; Fiti, L. Identifying the Household Factors, and Food Items, Most Important to Nutrition in Vanuatu; Food and Agricultural Organisation of the United Nations: Port Vila, Vanuatu, 2015; p. 108. [Google Scholar]
- Friedman, J.; Beegle, K.; De Weerdt, J.; Gibson, J. Decomposing Response Errors in Food Consumption Measurement: Implications for Survey Design from a Survey Experiment in Tanzania; World Bank: Washington, DC, USA, 2016. [Google Scholar]
- Da Costa Louzada, M.L.; Levy, R.B.; Martins, A.P.B.; Claro, R.M.; Steele, E.M.; Verly, E., Jr.; Cafiero, C.; Monteiro, C.A. Validating the usage of household food acquisition surveys to assess the consumption of ultra-processed foods: Evidence from brazil. Food Policy 2017, 72, 112–120. [Google Scholar] [CrossRef]
- Webster, J.; Su’a, S.A.F.; Ieremia, M.; Bompoint, S.; Johnson, C.; Faeamani, G.; Vaiaso, M.; Snowdon, W.; Land, M.A.; Trieu, K.; et al. Salt intakes, knowledge, and behavior in Samoa: Monitoring salt-consumption patterns through the world health organization’s surveillance of noncommunicable disease risk factors (STEPS). J. Clin. Hypertens. 2016, 18, 884–891. [Google Scholar] [CrossRef]
- The World Bank Group. Samoa. Available online: https://data.worldbank.org/country/Samoa (accessed on 26 February 2018).
- Spurrier, C.T. Cassava, Coconut and Curry: Food and National Identity in Post-Colonial Fiji; ProQuest Dissertations Publishing: Ann Arbor, MI, USA, 2010. [Google Scholar]
- Land, M.-A.; Webster, J.; Christoforou, A.; Praveen, D.; Jeffery, P.; Chalmers, J.; Smith, W.; Woodward, M.; Barzi, F.; Nowson, C.; et al. Salt intake assessed by 24 h urinary sodium excretion in a random and opportunistic sample in australia. BMJ Open 2014, 4, e003720. [Google Scholar] [CrossRef]
- McLaren, L.; Sumar, N.; Lorenzetti, D.L.; Campbell, N.R.; McIntyre, L.; Tarasuk, V. Population-level interventions in government jurisdictions for dietary sodium reduction. Cochrane Database Syst. Rev. 2016. [Google Scholar] [CrossRef]
- Trieu, K.; Neal, B.; Hawkes, C.; Dunford, E.; Campbell, N.; Rodriguez-Fernandez, R.; Legetic, B.; McLaren, L.; Barberio, A.; Webster, J. Salt reduction initiatives around the world—A systematic review of progress towards the global target. PLoS ONE 2015, 10, e0130247. [Google Scholar] [CrossRef]
- Rhodes, D.G.; Murayi, T.; Clemens, J.C.; Baer, D.J.; Sebastian, R.S.; Moshfegh, A.J. The usda automated multiple-pass method accurately assesses population sodium intakes. Am. Clin. Nutr. 2013, 97, 958–964. [Google Scholar] [CrossRef]
- Sparks, E.; Paterson, K.; Santos, J.A.; Trieu, K.; Hinge, N.; Tarivonda, L.; Snowdon, W.; Johnson, C.; Websterm, J. Salt-related knowledge, attitudes, and behaviors on Efate Island, Vanuatu. Int. J. Environ. Res. Public Health 2019, 16, 1027. [Google Scholar] [CrossRef]
- World Health Organization. Shake the Salt Habit: The Shake Technical Package for Salt Reduction; World Health Organization: Geneva, Switzerland, 2016. [Google Scholar]
| Characteristics | Overall | Female | Male |
|---|---|---|---|
| Age in years, mean (SD) | 36.6 (12.6) | 35.6 (12.3) | 37.5 (12.8) |
| Female, % | 49.2 | - | - |
| Area, % | |||
| Rural | 34.1 | 34.7 | 33.4 |
| Urban | 66.0 | 65.3 | 66.6 |
| Completed education, % | |||
| No formal schooling | 2.5 | 2.8 | 2.2 |
| Primary level | 43.6 | 47.9 | 39.6 |
| Secondary level | 41.7 | 38.7 | 44.7 |
| Tertiary level | 12.1 | 10.7 | 13.5 |
| Employed, % | 52.8 | 43.7 | 61.6 |
| Height in cm, mean (SD) | 164.1 (7.9) | 158.9 (5.7) | 169.1 (6.3) |
| Weight in kg, mean (SD) | 73.6 (15.6) | 71.1 (16.3) | 76.0 (14.6) |
| Body mass index in kg/m2, mean (SD) | 27.3 (5.4) | 28.1 (5.9) | 26.6 (4.8) |
| Overweight or obese, % | 60.3 | 54.8 | 65.9 |
| Systolic blood pressure in mmHg, mean (SD) | 121.7 (19.2) | 116.5 (18.1) | 126.8 (19.0) |
| Diastolic blood pressure in mmHg, mean (SD) | 77.9 (12.7) | 76.4 (11.4) | 79.3 (13.8) |
| History of hypertension, % | 10.3 | 10.9 | 9.8 |
| Measured hypertension, % | 21.6 | 16.1 | 26.9 |
| Pre-existing hypertension (those who had measured high blood pressure OR were taking Western hypertension medication prior to the survey), % | 24.0 | 18.8 | 29.0 |
| History of high cholesterol in blood, % | 3.7 | 5.1 | 2.4 |
| History of heart attack, % | 1.8 | 0.8 | 2.9 |
| History of stroke, % | 1.1 | 0.4 | 1.7 |
| History of diabetes, % | 2.0 | 1.0 | 3.1 |
| History of chronic kidney disease, % | 1.8 | 1.0 | 2.6 |
| n | Overall | Female | Male | p-Value | |
|---|---|---|---|---|---|
| 24-h urine in g/day, mean (SD) | 71 | 5.9 (3.6) | 5.6 (3.7) | 6.2 (3.5) | 0.496 |
| Spot urine using “INTERSALT with potassium” equation in g/day, mean (SD) | 483 | 7.2 (2.3) | 6.5 (1.7) | 7.8 (2.6) | <0.001 |
| Salt intake above the 5 g WHO target, % | 483 | 84.4 | 81.1 | 87.6 |
© 2019 by the WHO. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Paterson, K.; Hinge, N.; Sparks, E.; Trieu, K.; Santos, J.A.; Tarivonda, L.; Snowdon, W.; Webster, J.; Johnson, C. Mean Dietary Salt Intake in Vanuatu: A Population Survey of 755 Participants on Efate Island. Nutrients 2019, 11, 916. https://doi.org/10.3390/nu11040916
Paterson K, Hinge N, Sparks E, Trieu K, Santos JA, Tarivonda L, Snowdon W, Webster J, Johnson C. Mean Dietary Salt Intake in Vanuatu: A Population Survey of 755 Participants on Efate Island. Nutrients. 2019; 11(4):916. https://doi.org/10.3390/nu11040916
Chicago/Turabian StylePaterson, Katherine, Nerida Hinge, Emalie Sparks, Kathy Trieu, Joseph Alvin Santos, Len Tarivonda, Wendy Snowdon, Jacqui Webster, and Claire Johnson. 2019. "Mean Dietary Salt Intake in Vanuatu: A Population Survey of 755 Participants on Efate Island" Nutrients 11, no. 4: 916. https://doi.org/10.3390/nu11040916
APA StylePaterson, K., Hinge, N., Sparks, E., Trieu, K., Santos, J. A., Tarivonda, L., Snowdon, W., Webster, J., & Johnson, C. (2019). Mean Dietary Salt Intake in Vanuatu: A Population Survey of 755 Participants on Efate Island. Nutrients, 11(4), 916. https://doi.org/10.3390/nu11040916







