The Role of Probiotics and Prebiotics in the Prevention and Treatment of Obesity
Abstract
:1. Introduction
2. Pathophysiology of Obesity
3. The Role of Intestinal Microbiota in the Metabolic State
4. Experimental Studies with Probiotics and Prebiotics for Prevention and Management of Obesity
5. Review of Clinical Studies Using Probiotics and Prebiotics in Obesity
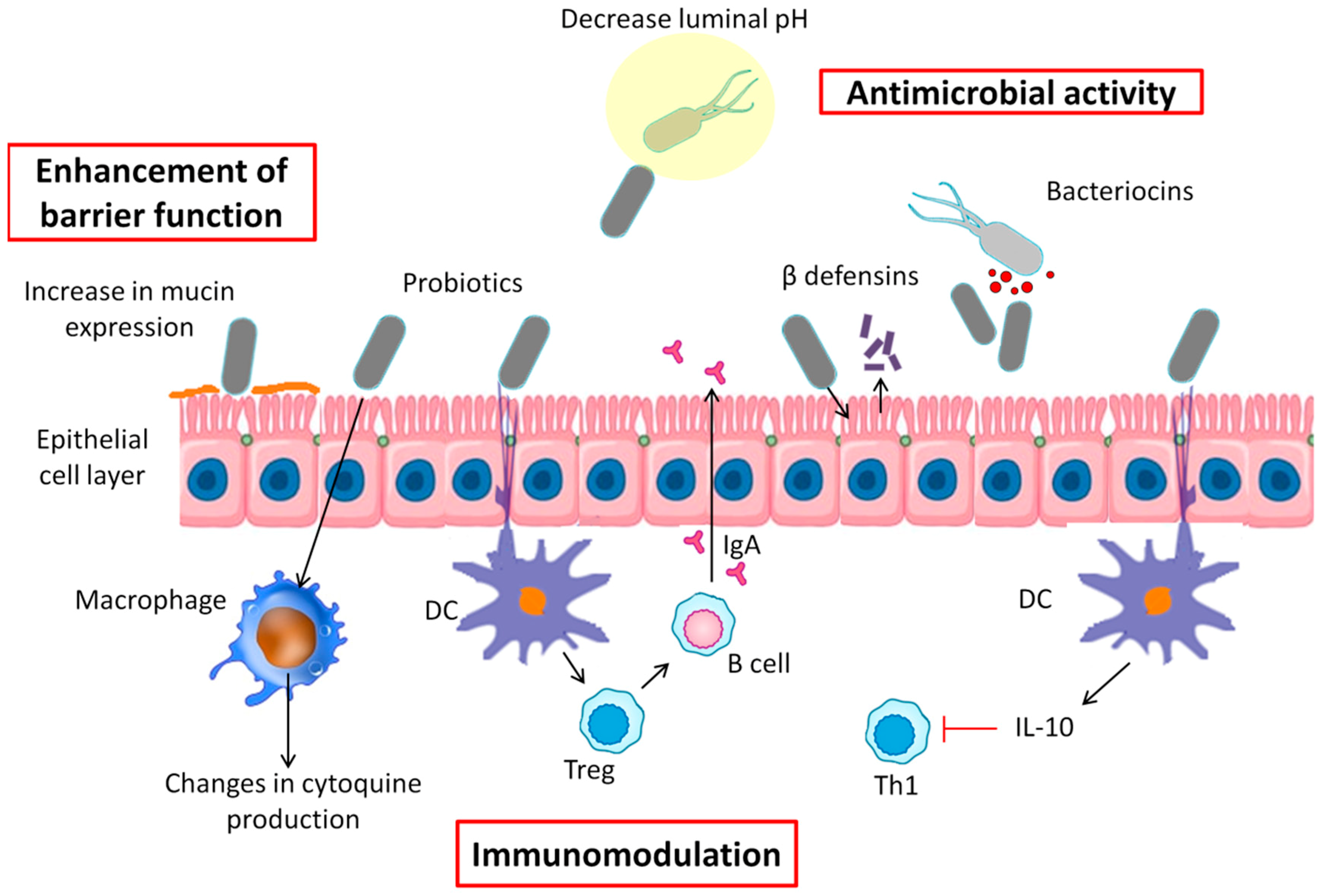
6. Probiotics Mechanisms of Action
6.1. Antimicrobial Activity
6.2. Enhancement of Barrier Function
6.3. Immunomodulation
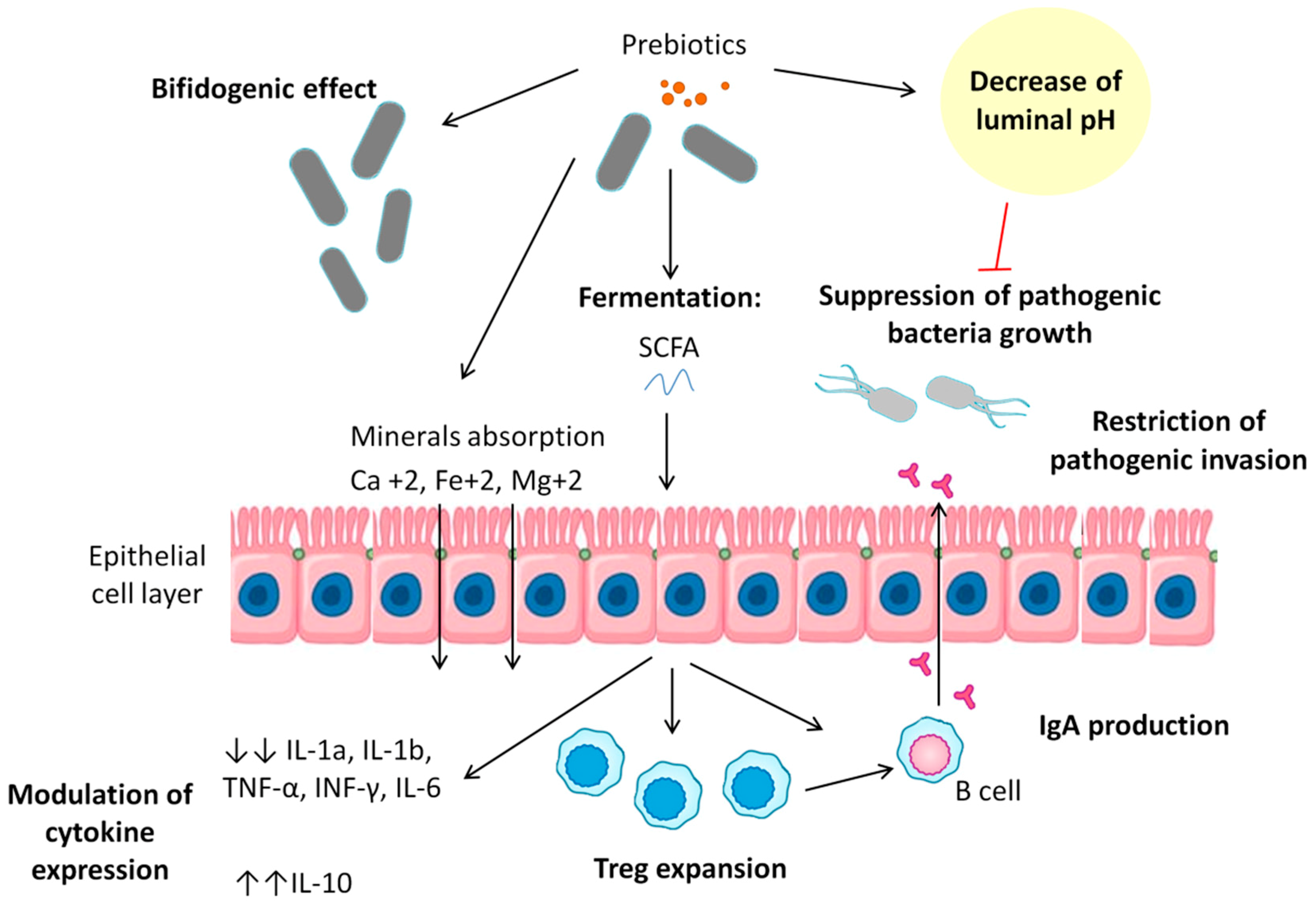
7. Prebiotics: Mechanism of Action
8. Conclusions and Perspectives
Funding
Conflicts of Interest
References
- World Health Organization. Obesity: Preventing and Managing the Global Epidemic; World Health Organization: Geneva, Switzerland, 2000. [Google Scholar]
- Khayatzadeh-Mahani, A.; Ruckert, A.; Labonté, R. Obesity prevention: Co-framing for intersectoral ‘buy-in’. Crit. Public Health 2018, 28, 4–11. [Google Scholar] [CrossRef]
- Bray, G.A.; Ryan, D.H. Clinical evaluation of the overweight patient. Endocrine 2000, 13, 167–186. [Google Scholar] [CrossRef]
- Monteiro, C.A.; Moubarac, J.C.; Cannon, G.; Ng, S.W.; Popkin, B. Ultra-processed products are becoming dominant in the global food system. Obes. Rev. 2013, 14, 21–28. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kasselman, L.J.; Vernice, N.A.; DeLeon, J.; Reiss, A.B. The gut microbiome and elevated cardiovascular risk in obesity and autoimmunity. Atherosclerosis 2018, 271, 203–213. [Google Scholar] [CrossRef] [PubMed]
- Cerdó, T.; Ruiz, A.; Campoy, C. Human Gut Microbiota and Obesity during Development, Adiposity—Omics and Molecular Understanding; Gordeladze, J., Ed.; InTech: Vienna, Austria, 2017; ISBN1 978-953-51-2997-4. ISBN2 978-953-51-2998-1. Available online: https://www.intechopen.com/books/adiposity-omics-and-molecular-understanding/human-gut-microbiota-and-obesity-during-development (accessed on 1 February 2017). [CrossRef]
- Cox, A.J.; West, N.P.; Cripps, A.W. Obesity, inflammation, and the gut microbiota. Lancet Diabetes Endocrinol. 2015, 3, 207–215. [Google Scholar] [CrossRef]
- Morelli, L.; Capurso, L. FAO/WHO guidelines on probiotics: 10 years later. J. Clin. Gastroenterol. 2012, 46, S1–S2. [Google Scholar] [CrossRef] [PubMed]
- Huaman, J.-W.; Mego, M.; Manichanh, C.; Cañellas, N.; Cañueto, D.; Segurola, H.; Jansana, M.; Malagelada, C.; Accarino, A.; Vulevic, J.; et al. Effects of prebiotics vs. a diet low in FODMAPs in patients with functional gut disorders. Gastroenterology 2018, 155, 1004–1007. [Google Scholar] [CrossRef] [PubMed]
- Delgado-Fernández, P.; Corzo, N.; Olano, A.; Hernández-Hernández, O.; Moreno, F.J. Effect of selected prebiotics on the growth of lactic acid bacteria and physicochemical properties of yoghurts. Int. Dairy J. 2019, 89, 77–85. [Google Scholar] [CrossRef]
- Parnell, J.A.; Raman, M.; Rioux, K.P.; Reimer, R.A. The potential role of prebiotic fibre for treatment and management of non-alcoholic fatty liver disease and associated obesity and insulin resistance. Liver Int. 2012, 32, 701–711. [Google Scholar] [CrossRef]
- Parekh, P.J.; Oldfield, E.C., IV; Lamba, A.; David, A. The Role of Gut Microflora in Obesity-Does the Data Provide an Option for Intervention? In Anti-Obesity Drug Discovery and Development; Bentham Science Publishers: Oak Park, IL, USA, 2017; Volume 3, pp. 204–227. [Google Scholar]
- Butel, M.J.; Waligora-Dupriet, A.J. Probiotics and prebiotics: What are they and what can they do for us? In The Human Microbiota and Chronic Disease: Dysbiosis as a Cause of Human Pathology; John Wiley & Sons: Hoboken, NJ, USA, 2016; pp. 467–478. [Google Scholar]
- Bray, G.; Kim, K.; Wilding, J.; Federation, W.O. Obesity: A chronic relapsing progressive disease process. A position statement of the World Obesity Federation. Obes. Rev. 2017, 18, 715–723. [Google Scholar] [CrossRef] [PubMed]
- Isolauri, E. Microbiota and Obesity. In Intestinal Microbiome: Functional Aspects in Health and Disease; Nestle Nutrition Institute Workshop Series; Karger Publishers: Basel, Switzerland, 2017; Volume 18, pp. 95–106. ISBN 978-3-318-06030-0. [Google Scholar]
- Duranti, S.; Ferrario, C.; van Sinderen, D.; Ventura, M.; Turroni, F. Obesity and microbiota: An example of an intricate relationship. Genes Nutr. 2017, 12, 18. [Google Scholar] [CrossRef] [PubMed]
- Vermeulen, E.; Stronks, K.; Snijder, M.B.; Schene, A.H.; Lok, A.; de Vries, J.H.; Visser, M.; Brouwer, IA.; Nicolaou, M. A combined high-sugar and high-saturated-fat dietary pattern is associated with more depressive symptoms in a multi-ethnic population: The helius (healthy life in an urban setting) study. Public Health Nutr. 2017, 20, 2374–2382. [Google Scholar] [CrossRef] [PubMed]
- Foster, J.A.; Rinaman, L.; Cryan, J.F. Stress & the gut-brain axis: Regulation by the microbiome. Neurobiol. Stress 2017, 7, 124–136. [Google Scholar] [PubMed] [Green Version]
- Al-Assal, K.; Martinez, A.C.; Torrinhas, R.S.; Cardinelli, C.; Waitzberg, D. Gut microbiota and obesity. Clin. Nutr. Exp. 2018, 20, 60–64. [Google Scholar] [CrossRef]
- Maruvada, P.; Leone, V.; Kaplan, L.M.; Chang, E.B. The human microbiome and obesity: Moving beyond associations. Cell Host Microbe 2017, 22, 589–599. [Google Scholar] [CrossRef] [PubMed]
- Kobyliak, N.; Conte, C.; Cammarota, G.; Haley, A.P.; Styriak, I.; Gaspar, L.; Fusek, J.; Rodrigo, L.; Kruzliak, P. Probiotics in prevention and treatment of obesity: A critical view. Nutr. Metab. 2016, 13, 14. [Google Scholar] [CrossRef] [PubMed]
- De Clercq, N.C.; Groen, A.K.; Romijn, J.A.; Nieuwdorp, M. Gut Microbiota in Obesity and Undernutrition. Adv. Nutr. 2016, 7, 1080–1089. [Google Scholar] [CrossRef] [PubMed]
- Duca, F.; Lam, T. Gut microbiota, nutrient sensing and energy balance. Diabetes Obes. Metab. 2014, 16, 68–76. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aron-Wisnewsky, J.; Doré, J.; Clement, K. The importance of the gut microbiota after bariatric surgery. Nat. Rev. Gastroenterol. Hepatol. 2012, 9, 590. [Google Scholar] [CrossRef] [PubMed]
- Schwiertz, A.; Taras, D.; Schäfer, K.; Beijer, S.; Bos, N.A.; Donus, C.; Hardt, P.D. Microbiota and SCFA in lean and overweight healthy subjects. Obesity 2010, 18, 190–195. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, J.; Su, W.; Rahat-Rozenbloom, S.; Wolever, T.; Comelli, E. Adiposity, gut microbiota and faecal short chain fatty acids are linked in adult humans. Nutr. Diabetes 2014, 4, e121. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hildebrandt, M.A.; Hoffmann, C.; Sherrill–Mix, S.A.; Keilbaugh, S.A.; Hamady, M.; Chen, Y.Y.; Knight, R.; Ahima, R.S.; Bushman, F.; Wu, G.D. High-fat diet determines the composition of the murine gut microbiome independently of obesity. Gastroenterology 2009, 137, 1716–1724.e2. [Google Scholar] [CrossRef] [PubMed]
- Harley, I.T.; Karp, C.L. Obesity and the gut microbiome: Striving for causality. Mol. Metab. 2012, 1, 21–31. [Google Scholar] [CrossRef] [PubMed]
- Virgin, H.W.; Todd, J.A. Metagenomics and personalized medicine. Cell 2011, 147, 44–56. [Google Scholar] [CrossRef] [PubMed]
- Turnbaugh, P.J.; Ley, R.E.; Mahowald, M.A.; Magrini, V.; Mardis, E.R.; Gordon, J.I. An obesity-associated gut microbiome with increased capacity for energy harvest. Nature 2006, 444, 1027. [Google Scholar] [CrossRef] [PubMed]
- Ridaura, V.K.; Faith, J.J.; Rey, F.E.; Cheng, J.; Duncan, A.E.; Kau, A.L.; Griffin, N.W.; Lombard, V.; Henrissa, B.; Bain, J.R.; et al. Gut microbiota from twins discordant for obesity modulate metabolism in mice. Science 2013, 341, 1241214. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.J.; Gerasimidis, K.; Edwards, C.A.; Shaikh, M.G. Role of gut microbiota in the aetiology of obesity: Proposed mechanisms and review of the literature. J. Obes. 2016, 2016, 7353642. [Google Scholar] [CrossRef] [PubMed]
- Patterson, E.; Ryan, P.M.; Cryan, J.F.; Dinan, T.G.; Ross, R.P.; Fitzgerald, G.F.; Stanton, C. Gut microbiota, obesity and diabetes. Postgrad. Med. J. 2016, 92, 286–300. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dinan, T.G.; Cryan, J.F. Mood by microbe: Towards clinical translation. Genome Med. 2016, 8, 36. [Google Scholar] [CrossRef] [PubMed]
- Neufeld, K.-A.M.; Kang, N.; Bienenstock, J.; Foster, J.A. Effects of intestinal microbiota on anxiety-like behavior. Commun. Integr. Biol. 2011, 4, 492–494. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Torres-Fuentes, C.; Schellekens, H.; Dinan, T.G.; Cryan, J.F. The microbiota–gut–brain axis in obesity. Lancet Gastroenterol. Hepatol. 2017, 2, 747–756. [Google Scholar] [CrossRef]
- Krajmalnik-Brown, R.; Ilhan, Z.E.; Kang, D.W.; DiBaise, J.K. Effects of gut microbes on nutrient absorption and energy regulation. Nutr. Clin. Pract. 2012, 27, 201–214. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.S.; Davies, S.S. Microbial metabolism of dietary components to bioactive metabolites: Opportunities for new therapeutic interventions. Genome Med. 2016, 8, 46. [Google Scholar] [CrossRef] [PubMed]
- Ridlon, J.M.; Kang, D.J.; Hylemon, P.B.; Bajaj, J.S. Bile acids and the gut microbiome. Curr. Opin. Gastroenterol. 2014, 30, 332. [Google Scholar] [CrossRef] [PubMed]
- Trabelsi, M.-S.; Lestavel, S.; Staels, B.; Collet, X. Intestinal bile acid receptors are key regulators of glucose homeostasis. Proc. Nutr. Soc. 2017, 76, 192–202. [Google Scholar] [CrossRef] [PubMed]
- Pindjakova, J.; Sartini, C.; Lo Re, O.; Rappa, F.; Coupe, B.; Lelouvier, B.; Pazienza, V.; Vinciguerra, M. Gut dysbiosis and adaptive immune response in diet-induced obesity vs. systemic inflammation. Front. Microbiol. 2017, 8, 1157. [Google Scholar] [CrossRef] [PubMed]
- Lau, K.; Srivatsav, V.; Rizwan, A.; Nashed, A.; Liu, R.; Shen, R.; Akhtar, M. Bridging the Gap between Gut Microbial Dysbiosis and Cardiovascular Diseases. Nutrients 2017, 9, 859. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, L.A.; Finlay, B.B. Early life factors that affect allergy development. Nat. Rev. Immunol. 2017, 17, 518–528. [Google Scholar] [CrossRef] [PubMed]
- Tamburini, S.; Shen, N.; Wu, H.C.; Clemente, J.C. The microbiome in early life: Implications for health outcomes. Nat. Med. 2016, 22, 713–722. [Google Scholar] [CrossRef] [PubMed]
- Kumari, M.; Kozyrskyj, A.L. Gut microbial metabolism defines host metabolism: An emerging perspective in obesity and allergic inflammation. Obes. Rev. 2017, 18, 18–31. [Google Scholar] [CrossRef] [PubMed]
- Koleva, P.T.; Kim, J.S.; Scott, J.A.; Kozyrskyj, A.L. Microbial programming of health and disease starts during fetal life. Birth Defects Res. Part C 2015, 105, 265–277. [Google Scholar] [CrossRef] [PubMed]
- Shao, X.; Ding, X.; Wang, B.; Li, L.; An, X.; Yao, Q.; Zhang, J.A. Antibiotic exposure in early life increases risk of childhood obesity: A systematic review and meta-analysis. Front. Endocrinol. 2017, 8, 170. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, J.L.; Walker, W.A. Early gut colonization and subsequent obesity risk. Curr. Opin. Clin. Nutr. Metab. Care 2012, 15, 278–284. [Google Scholar] [CrossRef] [PubMed]
- Aziz, Q.; Doré, J.; Emmanuel, A.; Guarner, F.; Quigley, E. Gut microbiota and gastrointestinal health: Current concepts and future directions. Neurogastroenterol. Motil. 2013, 25, 4–15. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, K.; Sugi, Y.; Nakano, K.; Tsuda, M.; Kurihara, K.; Hosono, A.; Kaminogawa, S. Epigenetic control of host gene by commensal bacteria in large intestinal epithelial cells. J. Biol. Chem. 2011, 286, 35755–35762. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Kelly, A.S. Review of childhood obesity: From epidemiology, aetiology, and comorbidities to clinical assessment and treatment. Mayo Clin. Proc. 2017, 92, 251–265. [Google Scholar] [CrossRef] [PubMed]
- Colquitt, J.L.; Pickett, K.; Loveman, E.; Frampton, G.K. Surgery for weight loss in adults. Cochrane Libr. 2014, 8, CD003641. [Google Scholar] [CrossRef] [PubMed]
- Durkin, N.; Desai, A.P. What Is the Evidence for Paediatric/Adolescent Bariatric Surgery? Curr. Obes. Rep. 2017, 6, 278–285. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ickovics, J.R.; Duffany, K.O.C.; Shebl, F.M.; Peters, S.M.; Read, M.A.; Gilstad-Hayden, K.R.; Schwartz, M.B. Implementing School-Based Policies to Prevent Obesity: Cluster Randomized Trial. Am. J. Prev. Med. 2019, 56, e1–e11. [Google Scholar] [CrossRef] [PubMed]
- Ganjayi, M.S.; Balaji, M.; Sreenivasulu, D.; Balaji, H.; Karunakaran, R.S. Recent Developments in the Prevention of Obesity by Using Microorganisms. Recent Dev. Appl. Microbiol. Biochem. 2019, 47–60. [Google Scholar] [CrossRef]
- Markowiak, P.; Śliżewska, K. Effects of Probiotics, Prebiotics, and Synbiotics on Human Health. Nutrients 2017, 9, 1021. [Google Scholar] [CrossRef] [PubMed]
- Sonnenburg, J.L.; Bäckhed, F. Diet–microbiota interactions as moderators of human metabolism. Nature 2016, 535, 56. [Google Scholar] [CrossRef] [PubMed]
- Food and Agriculture Organization. Probiotics in Food: Health and Nutritional Properties and Guidelines for Evaluation; FAO: Rome, Italy, 2006. [Google Scholar]
- Ejtahed, H.-S.; Angoorani, P.; Soroush, A.-R.; Atlasi, R.; Hasani-Ranjbar, S.; Mortazavian, A.M.; Larijanid, B. Probiotics supplementation for the obesity management; A systematic review of animal studies and clinical trials. J. Funct. Foods. 2019, 52, 228–242. [Google Scholar] [CrossRef]
- Ali, A.A.; Velasquez, M.T.; Hansen, C.T.; Mohamed, A.I.; Bhathena, S.J. Effects of soybean isoflavones, probiotics, and their interactions on lipid metabolism and endocrine system in an animal model of obesity and diabetes. J. Nutr. Biochem. 2004, 15, 583–590. [Google Scholar] [CrossRef] [PubMed]
- Bubnov, R.V.; Babenko, L.P.; Lazarenko, L.M.; Mokrozub, V.V.; Demchenko, O.A.; Nechypurenko, O.V.; Spivak, M.Y. Comparative study of probiotic effects of Lactobacillus and Bifidobacteria strains on cholesterol levels, liver morphology and the gut microbiota in obese mice. EPMA J. 2017, 8, 357–376. [Google Scholar] [CrossRef] [PubMed]
- Andersson, U.; Bränning, C.; Ahrné, S.; Molin, G.; Alenfall, J.; Önning, G.; Nyman, M.; Holm, C. Probiotics lower plasma glucose in the high-fat fed C57BL/6J mouse. Benef. Microbes 2010, 1, 189–196. [Google Scholar] [CrossRef] [PubMed]
- Bomhof, M.R.; Saha, D.C.; Reid, D.T.; Paul, H.A.; Reimer, R.A. Combined effects of oligofructose and Bifidobacterium animalis on gut microbiota and glycemia in obese rats. Obesity 2014, 22, 763–771. [Google Scholar] [CrossRef] [PubMed]
- Yin, Y.-N.; Yu, Q.-F.; Fu, N.; Liu, X.-W.; Lu, F.-G. Effects of four Bifidobacteria on obesity in high-fat diet induced rats. World J. Gastroenterol. 2010, 16, 3394. [Google Scholar] [CrossRef] [PubMed]
- Roselli, M.; Finamore, A.; Brasili, E.; Rami, R.; Nobili, F.; Orsi, C.; Zambrinib, A.V.; Mengheri, E. Beneficial effects of a selected probiotic mixture administered to high fat-fed mice before and after the development of obesity. J. Funct. Foods. 2018, 45, 321–329. [Google Scholar] [CrossRef]
- Casas, I.A.; Dobrogosz, W.J. Validation of the probiotic concept: Lactobacillus reuteri confers broad-spectrum protection against disease in humans and animals. Microb. Ecol. Health Dis. 2000, 12, 247–285. [Google Scholar] [CrossRef]
- Zhao, X.; Higashikawa, F.; Noda, M.; Kawamura, Y.; Matoba, Y.; Kumagai, T.; Sugiyama, M. The obesity and fatty liver are reduced by plant-derived Pediococcus pentosaceus LP28 in high fat diet-induced obese mice. PLoS ONE 2012, 7, e30696. [Google Scholar] [CrossRef] [PubMed]
- Cano, P.G.; Santacruz, A.; Moya, Á.; Sanz, Y. Bacteroides uniformis CECT 7771 ameliorates metabolic and immunological dysfunction in mice with high-fat-diet induced obesity. PLoS ONE 2012, 7, e41079. [Google Scholar]
- Everard, A.; Belzer, C.; Geurts, L.; Ouwerkerk, J.P.; Druart, C.; Bindels, L.B.; Guiot, Y.; Derrien, M.; Muccioli, G.; Delzenne, N.M.; et al. Cross-talk between Akkermansia muciniphila and intestinal epithelium controls diet-induced obesity. Proc. Natl. Acad. Sci. USA 2013, 110, 9066–9071. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Everard, A.; Matamoros, S.; Geurts, L.; Delzenne, N.M.; Cani, P.D. Saccharomyces boulardii administration changes gut microbiota and reduces hepatic steatosis, low-grade inflammation, and fat mass in obese and type 2 diabetic DB/DB mice. MBio 2014, 5, e01011–e01014. [Google Scholar] [CrossRef] [PubMed]
- Brusaferro, A.; Cozzali, R.; Orabona, C.; Biscarini, A.; Farinelli, E.; Cavalli, E.; Grohmann, U.; Principi, N.; Esposito, S. Is It Time to Use Probiotics to Prevent or Treat Obesity? Nutrients 2018, 10, 1613. [Google Scholar] [CrossRef] [PubMed]
- Pineiro, M.; Asp, N.-G.; Reid, G.; Macfarlane, S.; Morelli, L.; Brunser, O.; Tuohy, K. FAO Technical meeting on prebiotics. J. Clin. Gastroenterol. 2008, 42, S156–S159. [Google Scholar] [CrossRef] [PubMed]
- Younis, K.; Ahmad, S.; Jahan, K. Health benefits and application of prebiotics in foods. J. Food Process. Technol. 2015, 6, 1. [Google Scholar]
- Connolly, M.L.; Lovegrove, J.A.; Tuohy, K.M. In vitro fermentation characteristics of whole grain wheat flakes and the effect of toasting on prebiotic potential. J. Med. Food 2012, 15, 33–43. [Google Scholar] [CrossRef] [PubMed]
- Parnell, J.A.; Reimer, R.A. Effect of prebiotic fibre supplementation on hepatic gene expression and serum lipids: A dose–response study in JCR: LA-cp rats. Br. J. Nutr. 2010, 103, 1577–1584. [Google Scholar] [CrossRef] [PubMed]
- Everard, A.; Lazarevic, V.; Derrien, M.; Girard, M.; Muccioli, G.M.; Neyrinck, A.M.; Possemiers, S.; Van Holle, A.; François, P.; de Vos, W.M.; et al. Responses of gut microbiota and glucose and lipid metabolism to prebiotics in genetic obese and diet-induced leptin-resistant mice. Diabetes 2011, 60, 2775–2786. [Google Scholar] [CrossRef] [PubMed]
- Cani, P.D.; Possemiers, S.; Van de Wiele, T.; Guiot, Y.; Everard, A.; Rottier, O.; Geurts, L.; Naslain, D.; Neyrinck, A.; Lambert, D.M.; et al. Changes in gut microbiota control inflammation in obese mice through a mechanism involving GLP-2-driven improvement of gut permeability. Gut 2009, 58, 1091–1103. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Respondek, F.; Gerard, P.; Bossis, M.; Boschat, L.; Bruneau, A.; Rabot, S.; Wagner, A.; Martin, J.C. Short-chain fructo-oligosaccharides modulate intestinal microbiota and metabolic parameters of humanized gnotobiotic diet induced obesity mice. PLoS ONE 2013, 8, e71026. [Google Scholar] [CrossRef] [PubMed]
- Nihei, N.; Okamoto, H.; Furune, T.; Ikuta, N.; Sasaki, K.; Rimbach, G.; Yoshikawa, Y.; Terao, K. Dietary α-cyclodextrin modifies gut microbiota and reduces fat accumulation in high-fat-diet-fed obese mice. BioFactors 2018, 44, 336–347. [Google Scholar] [CrossRef] [PubMed]
- Posovszky, C.; Wabitsch, M. Regulation of appetite, satiation, and body weight by enteroendocrine cells. Part 2: Therapeutic potential of enteroendocrine cells in the treatment of obesity. Horm. Res. Paediatr. 2015, 83, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Reimer, R.A.; Russell, J.C. Glucose tolerance, lipids, and GLP-1 secretion in JCR: LA-cp rats fed a high protein fiber diet. Obesity 2008, 16, 40–46. [Google Scholar] [CrossRef] [PubMed]
- Liong, M.-T.; Dunshea, F.R.; Shah, N.P. Effects of a synbiotic containing Lactobacillus acidophilus ATCC 4962 on plasma lipid profiles and morphology of erythrocytes in hypercholesterolaemic pigs on high-and low-fat diets. Br. J. Nutr. 2007, 98, 736–744. [Google Scholar] [CrossRef] [PubMed]
- Mischke, M.; Arora, T.; Tims, S.; Engels, E.; Sommer, N.; van Limpt, K.; Baars, A.; Oozeer, R.; Oosting, A.; Bäckhed, F.; et al. Specific synbiotics in early life protect against diet-induced obesity in adult mice. Diabetes Obes. Metab. 2018, 20, 1408–1418. [Google Scholar] [CrossRef] [PubMed]
- Luoto, R.; Kalliomäki, M.; Laitinen, K.; Isolauri, E. The impact of perinatal probiotic intervention on the development of overweight and obesity: Follow-up study from birth to 10 years. Int. J. Obes. 2010, 34, 1531. [Google Scholar] [CrossRef] [PubMed]
- Vajro, P.; Mandato, C.; Licenziati, M.R.; Franzese, A.; Vitale, D.F.; Lenta, S.; Caropreso, M.; Vallone, G.; Meli, R. Effects of Lactobacillus rhamnosus strain GG in pediatric obesity-related liver disease. J. Pediatr. Gastroenterol. Nutr. 2011, 52, 740–743. [Google Scholar] [CrossRef] [PubMed]
- Alisi, A.; Bedogni, G.; Baviera, G.; Giorgio, V.; Porro, E.; Paris, C.; Giammaria, P.; Reali, L.; Anania, F.; Nobili, V. Randomised clinical trial: The beneficial effects of VSL#3 in obese children with non-alcoholic steatohepatitis. Aliment. Pharmacol. Ther. 2014, 39, 1276–1285. [Google Scholar] [PubMed]
- Famouri, F.; Shariat, Z.; Hashemipour, M.; Keikha, M.; Kelishadi, R. Effects of probiotics on nonalcoholic fatty liver disease in obese children and adolescents. J. Pediatr. Gastroenterol. Nutr. 2017, 64, 413–417. [Google Scholar] [CrossRef] [PubMed]
- Sanchis-Chordà, J.; del Pulgar, E.M.G.; Carrasco-Luna, J.; Benítez-Páez, A.; Sanz, Y.; Codoñer-Franch, P. Bifidobacterium pseudocatenulatum CECT 7765 supplementation improves inflammatory status in insulin-resistant obese children. Eur. J. Nutr. 2018. [Google Scholar] [CrossRef] [PubMed]
- Gøbel, R.J.; Larsen, N.; Jakobsen, M.; Mølgaard, C.; Michaelsen, K.F. Probiotics to adolescents with obesity: Effects on inflammation and metabolic syndrome. J. Pediatr. Gastroenterol. Nutr. 2012, 55, 673–678. [Google Scholar] [CrossRef] [PubMed]
- Jones, R.B.; Alderete, T.L.; Martin, A.A.; Geary, B.A.; Hwang, D.H.; Palmer, S.L.; Goran, M.I. Probiotic supplementation increases obesity with no detectable effects on liver fat or gut microbiota in obese Hispanic adolescents: A 16-week, randomized, placebo-controlled trial. Pediatr. Obes. 2018, 13, 705–714. [Google Scholar] [CrossRef] [PubMed]
- Jung, S.; Lee, Y.J.; Kim, M.; Kim, M.; Kwak, J.H.; Lee, J.-W.; Ahn, Y.T.; Hun, J.; HoLee, S. Supplementation with two probiotic strains, Lactobacillus curvatus HY7601 and Lactobacillus plantarum KY1032, reduced body adiposity and Lp-PLA2 activity in overweight subjects. J. Funct. Foods 2015, 19, 744–752. [Google Scholar] [CrossRef]
- Gomes, A.C.; de Sousa, R.G.M.; Botelho, P.B.; Gomes, T.L.N.; Prada, P.O.; Mota, J.F. The additional effects of a probiotic mix on abdominal adiposity and antioxidant Status: A double-blind, randomized trial. Obesity 2017, 25, 30–38. [Google Scholar] [CrossRef] [PubMed]
- Higashikawa, F.; Noda, M.; Awaya, T.; Danshiitsoodol, N.; Matoba, Y.; Kumagai, T.; Sugiyama, M. Antiobesity effect of Pediococcus pentosaceus LP28 on overweight subjects: A randomized, double-blind, placebo-controlled clinical trial. Eur. J. Clin. Nutr. 2016, 70, 582. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Yun, J.M.; Kim, M.K.; Kwon, O.; Cho, B. Lactobacillus gasseri BNR17 Supplementation Reduces the Visceral Fat Accumulation and Waist Circumference in Obese Adults: A Randomized, Double-Blind, Placebo-Controlled Trial. J. Med. Food 2018, 21, 454–461. [Google Scholar] [CrossRef] [PubMed]
- Minami, J.; Iwabuchi, N.; Tanaka, M.; Yamauchi, K.; Xiao, J.-Z.; Abe, F.; Sakane, N. Effects of Bifidobacterium breve B-3 on body fat reductions in pre-obese adults: A randomized, double-blind, placebo-controlled trial. Biosci. Microbiota Food Health 2018, 37, 67–75. [Google Scholar] [CrossRef] [PubMed]
- Pedret, A.; Valls, R.M.; Calderón-Pérez, L.; Llauradó, E.; Companys, J.; Pla-Pagà, L.; Moragas, A.; Martín-Luján, F.; Ortega, Y.; Giralt, M.; et al. Effects of daily consumption of the probiotic Bifidobacterium animalis subsp. lactis CECT 8145 on anthropometric adiposity biomarkers in abdominally obese subjects: A randomized controlled trial. Int. J. Obes. 2018. [Google Scholar] [CrossRef] [PubMed]
- Aller, R.; De Luis, D.; Izaola, O.; Conde, R.; Gonzalez Sagrado, M.; Primo, D.; De La Fuente, B.; Gonzalez, J. Effect of a probiotic on liver aminotransferases in nonalcoholic fatty liver disease patients: A double blind randomized clinical trial. Eur. Rev. Med. Pharm. Sci. 2011, 15, 1090–1095. [Google Scholar]
- Kadooka, Y.; Sato, M.; Imaizumi, K.; Ogawa, A.; Ikuyama, K.; Akai, Y.; Okano, M.; Kagoshima, M.; Tsuchida, T. Regulation of abdominal adiposity by probiotics (Lactobacillus gasseri SBT2055) in adults with obese tendencies in a randomized controlled trial. Eur. J. Clin. Nutr. 2010, 64, 636. [Google Scholar] [CrossRef] [PubMed]
- Mykhal’chyshyn, H.; Bodnar, P.; Kobyliak, N. Effect of probiotics on pro-inflammatory cytokines level in patients with type 2 diabetes and non-alcoholic fatty liver disease. Likars’ Ka Sprav. 2013, 2, 56–62. [Google Scholar]
- Osterberg, K.L.; Boutagy, N.E.; McMillan, R.P.; Stevens, J.R.; Frisard, M.I.; Kavanaugh, J.W.; Davy, B.M.; Davy, K.P.; Hulver, M.W. Probiotic supplementation attenuates increases in body mass and fat mass during high-fat diet in healthy young adults. Obesity 2015, 23, 2364–2370. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sanchez, M.; Darimont, C.; Panahi, S.; Drapeau, V.; Marette, A.; Taylor, V.H.; Doré, J.; Tremblay, A. Effects of a diet-based weight-reducing program with probiotic supplementation on satiety efficiency, eating behaviour traits, and psychosocial behaviours in obese individuals. Nutrients 2017, 9, 284. [Google Scholar] [CrossRef] [PubMed]
- Szulińska, M.; Łoniewski, I.; van Hemert, S.; Sobieska, M.; Bogdański, P. Dose-dependent effects of multispecies probiotic supplementation on the lipopolysaccharide (LPS) level and cardiometabolic profile in obese postmenopausal women: A 12-week randomized clinical trial. Nutrients 2018, 10, 773. [Google Scholar] [CrossRef] [PubMed]
- Cani, P.D.; Joly, E.; Horsmans, Y.; Delzenne, N.M. Oligofructose promotes satiety in healthy human: A pilot study. Eur. J. Clin. Nutr. 2006, 60, 567. [Google Scholar] [CrossRef] [PubMed]
- Cani, P.D.; Lecourt, E.; Dewulf, E.M.; Sohet, F.M.; Pachikian, B.D.; Naslain, D.; De Backer, F.; Neyrinck, A.M.; Delzenne, N.M. Gut microbiota fermentation of prebiotics increases satietogenic and incretin gut peptide production with consequences for appetite sensation and glucose response after a meal. Am. J. Clin. Nutr. 2009, 90, 1236–1243. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dehghan, P.; Gargari, B.P.; Jafar-Abadi, M.A.; Aliasgharzadeh, A. Inulin controls inflammation and metabolic endotoxemia in women with type 2 diabetes mellitus: A randomized-controlled clinical trial. Int. J. Food Sci. Nutr. 2014, 65, 117–123. [Google Scholar] [CrossRef] [PubMed]
- Edrisi, F.; Salehi, M.; Ahmadi, A.; Fararoei, M.; Rusta, F.; Mahmoodianfard, S. Effects of supplementation with rice husk powder and rice bran on inflammatory factors in overweight and obese adults following an energy-restricted diet: A randomized controlled trial. Eur. J. Nutr. 2018, 57, 833–843. [Google Scholar] [CrossRef] [PubMed]
- Genta, S.; Cabrera, W.; Habib, N.; Pons, J.; Carillo, I.M.; Grau, A.; Sánchez, S. Yacon syrup: Beneficial effects on obesity and insulin resistance in humans. Clin. Nutr. 2009, 28, 182–187. [Google Scholar] [CrossRef] [PubMed]
- Hume, M.P.; Nicolucci, A.C.; Reimer, R.A. Prebiotic supplementation improves appetite control in children with overweight and obesity: A randomized controlled trial. Am. J. Clin. Nutr. 2017, 105, 790–799. [Google Scholar] [CrossRef] [PubMed]
- Nicolucci, A.C.; Hume, M.P.; Martínez, I.; Mayengbam, S.; Walter, J.; Reimer, R.A. Prebiotics reduce body fat and alter intestinal microbiota in children who are overweight or with obesity. Gastroenterology 2017, 153, 711–722. [Google Scholar] [CrossRef] [PubMed]
- Parnell, J.A.; Reimer, R.A. Weight loss during oligofructose supplementation is associated with decreased ghrelin and increased peptide YY in overweight and obese adults. Am. J. Clin. Nutr. 2009, 89, 1751–1759. [Google Scholar] [CrossRef] [PubMed]
- Reimer, R.A.; Willis, H.J.; Tunnicliffe, J.M.; Park, H.; Madsen, K.L.; Soto-Vaca, A. Inulin-type fructans and whey protein both modulate appetite but only fructans alter gut microbiota in adults with overweight/obesity: A randomized controlled trial. Mol. Nutr. Food Res. 2017, 61, 1700484. [Google Scholar] [CrossRef] [PubMed]
- Russo, F.; Linsalata, M.; Clemente, C.; Chiloiro, M.; Orlando, A.; Marconi, E.; Chimienti, G.; Riezzo, G. Inulin-enriched pasta improves intestinal permeability and modifies the circulating levels of zonulin and glucagon-like peptide 2 in healthy young volunteers. Nutr. Res. 2012, 32, 940–946. [Google Scholar] [CrossRef] [PubMed]
- Stenman, L.K.; Lehtinen, M.J.; Meland, N.; Christensen, J.E.; Yeung, N.; Saarinen, M.T.; Courtneyc, M.; Burcelinc, R.; Lähdeahod, M.L.; Linros, J.; et al. Probiotic with or without fiber controls body fat mass, associated with serum zonulin, in overweight and obese adults—Randomized controlled trial. EBioMedicine 2016, 13, 190–200. [Google Scholar] [CrossRef] [PubMed]
- Verhoef, S.P.; Meyer, D.; Westerterp, K.R. Effects of oligofructose on appetite profile, glucagon-like peptide 1 and peptide YY3-36 concentrations and energy intake. Br. J. Nutr. 2011, 106, 1757–1762. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Whelan, K.; Efthymiou, L.; Judd, P.A.; Preedy, V.R.; Taylor, M.A. Appetite during consumption of enteral formula as a sole source of nutrition: The effect of supplementing pea-fibre and fructo-oligosaccharides. Br. J. Nutr. 2006, 96, 350–356. [Google Scholar] [CrossRef] [PubMed]
- Loguercio, C.; Federico, A.; Tuccillo, C.; Terracciano, F.; D’auria, M.V.; De Simone, C.; Del Vecchio Blanco, C. Beneficial effects of a probiotic VSL# 3 on parameters of liver dysfunction in chronic liver diseases. J. Clin. Gastroenterol. 2005, 39, 540–543. [Google Scholar] [PubMed]
- De Luis, D.; De la Fuente, B.; Izaola, O.; Conde, R.; Gutiérrez, S.; Morillo, M.; Teba Torres, C. Double blind randomized clinical trial controlled by placebo with an alpha linoleic acid and prebiotic enriched cookie on risk cardiovascular factor in obese patients. Nutr. Hosp. 2011, 26, 827–833. [Google Scholar] [PubMed]
- Seidel, C.; Boehm, V.; Vogelsang, H.; Wagner, A.; Persin, C.; Glei, M.; Pool-Zobel, B.L.; Jahreis, G. Influence of prebiotics and antioxidants in bread on the immune system, antioxidative status and antioxidative capacity in male smokers and non-smokers. Br. J. Nutr. 2007, 97, 349–356. [Google Scholar] [CrossRef] [PubMed]
- Liber, A.; Szajewska, H. Effect of oligofructose supplementation on body weight in overweight and obese children: A randomised, double-blind, placebo-controlled trial. Br. J. Nutr. 2014, 112, 2068–2074. [Google Scholar] [CrossRef] [PubMed]
- Canfora, E.E.; van der Beek, C.M.; Hermes, G.D.; Goossens, G.H.; Jocken, J.W.; Holst, J.J.; van Eijk, H.M.; Venema, K.; Smidt, H.; Zoetendal, E.G.; et al. Supplementation of diet with galacto-oligosaccharides increases bifidobacteria, but not insulin sensitivity, in obese prediabetic individuals. Gastroenterology 2017, 153, 87–97.e3. [Google Scholar] [CrossRef] [PubMed]
- Smiljanec, K.; Mitchell, C.M.; Privitera, O.F.; Neilson, A.P.; Davy, K.P.; Davy, B.M. Pre-meal inulin consumption does not affect acute energy intake in overweight and obese middle-aged and older adults: A randomized controlled crossover pilot trial. Nutr. Health 2017, 23, 75–81. [Google Scholar] [CrossRef] [PubMed]
- Peters, H.P.; Boers, H.M.; Haddeman, E.; Melnikov, S.M.; Qvyjt, F. No effect of added β-glucan or of fructooligosaccharide on appetite or energy intake. Am. J. Clin. Nutr. 2008, 89, 58–63. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kellow, N.J.; Coughlan, M.T.; Reid, C.M. Metabolic benefits of dietary prebiotics in human subjects: A systematic review of randomised controlled trials. Br. J. Nutr. 2014, 111, 1147–1161. [Google Scholar] [CrossRef] [PubMed]
- Tarantino, G.; Finelli, C. Systematic review on intervention with prebiotics/probiotics in patients with obesity-related non-alcoholic fatty liver disease. Future Microbiol. 2015, 10, 889–902. [Google Scholar] [CrossRef] [PubMed]
- Yadav, A.N.; Verma, P.; Kumar, R.; Kumar, S.; Kumar, V.; Kumar, K. (Eds.) Probiotic microbes: Biodiversity, mechanisms of action and potential role in human health. In Proceedings of the National Conference on Advances in Food Science and Technology, Paris, France, 23–25 October 2017; p. 33. [Google Scholar]
- Ng, S.; Hart, A.; Kamm, M.; Stagg, A.; Knight, S.C. Mechanisms of action of probiotics: Recent advances. Inflamm. Bowel Dis. 2008, 15, 300–310. [Google Scholar] [CrossRef] [PubMed]
- Duncan, S.H.; Louis, P.; Thomson, J.M.; Flint, H.J. The role of pH in determining the species composition of the human colonic microbiota. Environ. Microbiol. 2009, 11, 2112–2122. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Qian, K.; Wang, C.; Wu, Y. Roles of Probiotic Lactobacilli Inclusion in Helping Piglets Establish Healthy Intestinal Inter-environment for Pathogen Defence. Probiotics Antimicrob. Proteins 2018, 10, 243–250. [Google Scholar] [CrossRef] [PubMed]
- Roy, C.C.; Kien, C.L.; Bouthillier, L.; Levy, E. Short-chain fatty acids: Ready for prime time? Nutr. Clin. Pract. 2006, 21, 351–366. [Google Scholar] [CrossRef] [PubMed]
- Duncan, S.H.; Belenguer, A.; Holtrop, G.; Johnstone, A.M.; Flint, H.J.; Lobley, G.E. Reduced dietary intake of carbohydrates by obese subjects results in decreased concentrations of butyrate and butyrate-producing bacteria in feces. Appl. Environ. Microbiol. 2007, 73, 1073–1078. [Google Scholar] [CrossRef] [PubMed]
- Chikindas, M.L.; Weeks, R.; Drider, D.; Chistyakov, V.A.; Dicks, L.M. Functions and emerging applications of bacteriocins. Curr. Opin. Biotechnol. 2018, 49, 23–28. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.-S.; Wang, Y.-C.; Chow, Y.-S.; Yanagida, F.; Liao, C.-C.; Chiu, C.-M. Purification and characterization of plantaricin, Y.; a novel bacteriocin produced by Lactobacillus plantarum 510. Arch. Microbiol. 2014, 196, 193–199. [Google Scholar] [CrossRef] [PubMed]
- Sand, S.L.; Nissen-Meyer, J.; Sand, O.; Haug, T.M.; Plantaricin, A. A cationic peptide produced by Lactobacillus plantarum, permeabilizes eukaryotic cell membranes by a mechanism dependent on negative surface charge linked to glycosylated membrane proteins. Biochim. Biophys. Acta 2013, 1828, 249–259. [Google Scholar] [CrossRef] [PubMed]
- Spinler, J.K.; Taweechotipatr, M.; Rognerud, C.L.; Ou, C.N.; Tumwasorn, S.; Versalovic, J. Human-derived probiotic Lactobacillus reuteri demonstrate antimicrobial activities targeting diverse enteric bacterial pathogens. Anaerobe 2008, 14, 166–171. [Google Scholar] [CrossRef] [PubMed]
- Drissi, F.; Merhej, V.; Angelakis, E.; El Kaoutari, A.; Carrière, F.; Henrissat, B.; Raoult, D. Comparative genomics analysis of Lactobacillus species associated with weight gain or weight protection. Nutr. Diabetes 2014, 4, e109. [Google Scholar] [CrossRef] [PubMed]
- Cleusix, V.; Lacroix, C.; Vollenweider, S.; Le Blay, G. Glycerol induces reuterin production and decreases Escherichia coli population in an in vitro model of colonic fermentation with immobilized human feces. FEMS Microbiol. Ecol. 2008, 63, 56–64. [Google Scholar] [CrossRef] [PubMed]
- Boirivant, M.; Strober, W. The mechanism of action of probiotics. Curr. Opin. Gastroenterol. 2007, 23, 679–692. [Google Scholar] [CrossRef] [PubMed]
- Wehkamp, J.; Harder, J.; Wehkamp, K.; Wehkamp-von Meissner, B.; Schlee, M.; Enders, C.; Sonnenborn, U.; Nuding, S.; Bengmark, S.; Fellermann, K.; et al. NF-κB-and AP-1-mediated induction of human beta defensin-2 in intestinal epithelial cells by Escherichia coli Nissle 1917: A novel effect of a probiotic bacterium. Infect. Immun. 2004, 72, 5750–5758. [Google Scholar] [CrossRef] [PubMed]
- Schlee, M.; Harder, J.; Köten, B.; Stange, E.; Wehkamp, J.; Fellermann, K. Probiotic lactobacilli and VSL# 3 induce enterocyte β-defensin 2. Clin. Exp. Immunol. 2008, 151, 528–535. [Google Scholar] [PubMed]
- Martens, E.C.; Neumann, M.; Desai, M.S. Interactions of commensal and pathogenic microorganisms with the intestinal mucosal barrier. Nat. Rev. Microbiol. 2018, 16, 457–470. [Google Scholar] [CrossRef] [PubMed]
- Lamacchia, C.; Musaico, D.; Henderson, M.E.; Bergillos-Meca, T.; Roul, M.; Landriscina, L.; Decinaa, I.; Coronab, G.; Costabile, A. Temperature-treated gluten proteins in Gluten-Friendly™ bread increase mucus production and gut-barrier function in human intestinal goblet cells. J. Funct. Foods 2018, 48, 507–514. [Google Scholar] [CrossRef]
- Xu, P.; Becker, H.; Elizalde, M.; Masclee, A.; Jonkers, D. Intestinal organoid culture model is a valuable system to study epithelial barrier function in IBD. Gut 2018, 67, 1905–1906. [Google Scholar] [CrossRef] [PubMed]
- Gavin, P.G.; Mullaney, J.A.; Loo, D.; Lê Cao, K.-A.; Gottlieb, P.A.; Hill, M.M.; Zipris, D.; Hamilton-Williams, E.E. Intestinal metaproteomics reveals host-microbiota interactions in subjects at risk for type 1 diabetes. Diabetes Care 2018, 41, 2178–2186. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Chatterjee, I.; Anbazhagan, A.N.; Jayawardena, D.; Priyamvada, S.; Alrefai, W.A.; Sun, J.; Borthakur, A.; Dudeja, P.K. Cryptosporidium parvum disrupts intestinal epithelial barrier function via altering expression of key tight junction and adherens junction proteins. Cell. Microbiol. 2018, 20, e12830. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.; Gillingham, T.; Guo, Y.; Meng, D.; Zhu, W.; Walker, W.A.; Ganguli, K. Secretions of Bifidobacterium infantis and Lactobacillus acidophilus protect intestinal epithelial barrier function. J. Pediatr. Gastroenterol. Nutr. 2017, 64, 404–412. [Google Scholar] [CrossRef] [PubMed]
- Mattar, A.; Teitelbaum, D.H.; Drongowski, R.; Yongyi, F.; Harmon, C.; Coran, A. Probiotics up-regulate MUC-2 mucin gene expression in a Caco-2 cell-culture model. Pediatr. Surg. Int. 2002, 18, 586–590. [Google Scholar] [PubMed]
- Mack, D.; Ahrné, S.; Hyde, L.; Wei, S.; Hollingsworth, M. Extracellular MUC3 mucin secretion follows adherence of Lactobacillus strains to intestinal epithelial cells in vitro. Gut 2003, 52, 827–833. [Google Scholar] [CrossRef] [PubMed]
- Urdaci, M.C.; Lefevre, M.; Lafforgue, G.; Cartier, C.; Rodriguez, B.; Fioramonti, J. Antidiarrheal Action of Bacillus subtilis CU1 CNCM I-2745 and Lactobacillus plantarum CNCM I-4547 in Mice Through Different Cellular Pathways. Front. Microbiol. 2018, 9, 1537. [Google Scholar] [CrossRef] [PubMed]
- Resta-Lenert, S.; Barrett, K. Live probiotics protect intestinal epithelial cells from the effects of infection with entero-invasive Escherichia coli (EIEC). Gut 2003, 52, 988–997. [Google Scholar] [CrossRef] [PubMed]
- Otte, J.-M.; Podolsky, D.K. Functional modulation of enterocytes by gram-positive and gram-negative microorganisms. Am. J. Physiol. Gastrointest. Liver Physiol. 2004, 286, G613–G626. [Google Scholar] [CrossRef] [PubMed]
- Donato, K.A.; Gareau, M.G.; Wang, Y.J.J.; Sherman, P.M. Lactobacillus rhamnosus GG attenuates interferon-γ and tumour necrosis factor-α-induced barrier dysfunction and pro-inflammatory signalling. Microbiology 2010, 156, 3288–3297. [Google Scholar] [CrossRef] [PubMed]
- Ueno, N.; Fujiya, M.; Segawa, S.; Nata, T.; Moriichi, K.; Tanabe, H.; Mizukami, Y.; Kobayashi, N.; Ito, K.; Kohgo, Y. Heat-killed body of Lactobacillus brevis SBC8803 ameliorates intestinal injury in a murine model of colitis by enhancing the intestinal barrier function. Inflamm. Bowel Dis. 2011, 17, 2235–2250. [Google Scholar] [CrossRef] [PubMed]
- Osman, N.; Adawi, D.; Ahrné, S.; Jeppsson, B.; Molin, G. Probiotics and blueberry attenuate the severity of DSS-induced colitis. Dig. Dis. Sci. 2008, 53, 2464–2473. [Google Scholar] [CrossRef] [PubMed]
- Zakostelska, Z.; Kverka, M.; Klimesova, K.; Rossmann, P.; Mrazek, J.; Kopecny, J.; Hornova, M.; Srutkova, D.; Hudcovic, T.; Ridl, J.; et al. Lysate of probiotic Lactobacillus casei DN-114 001 ameliorates colitis by strengthening the gut barrier function and changing the gut microenvironment. PLoS ONE 2011, 6, e27961. [Google Scholar] [CrossRef] [PubMed]
- Iwasaki, A.; Kelsall, B.L. Freshly isolated Peyer’s patch, but not spleen, dendritic cells produce interleukin 10 and induce the differentiation of T helper type 2 cells. J. Exp. Med. 1999, 190, 229–240. [Google Scholar] [CrossRef] [PubMed]
- Rescigno, M.; Urbano, M.; Valzasina, B.; Francolini, M.; Rotta, G.; Bonasio, R.; Granucci, F.; Kraehenbuhl, J.P.; Ricciardi-Castagnoli, P. Dendritic cells express tight junction proteins and penetrate gut epithelial monolayers to sample bacteria. Nat. Immunol. 2001, 2, 361. [Google Scholar] [CrossRef] [PubMed]
- Stagg, A.; Hart, A.; Knight, S.; Kamm, M. The dendritic cell: Its role in intestinal inflammation and relationship with gut bacteria. Gut 2003, 52, 1522–1529. [Google Scholar] [CrossRef] [PubMed]
- Hart, A.; Lammers, K.; Brigidi, P.; Vitali, B.; Rizzello, F.; Gionchetti, P.; Campieri, M.; Kamm, M.A.; Knight, S.C.; Stagg, A.J.; et al. Modulation of human dendritic cell phenotype and function by probiotic bacteria. Gut 2004, 53, 1602–1609. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kalinina, O.; Knight, K.L. Reduction of autoantibody in SLE by probiotic exopolysaccharide-induced inhibitory dendritic cells. Am. Assoc. Immnol. 2018, 200, 162. [Google Scholar]
- Hume, D.A. Macrophages as APC and the dendritic cell myth. J. Immunol. 2008, 181, 5829–5835. [Google Scholar] [CrossRef] [PubMed]
- Kaji, R.; Kiyoshima-Shibata, J.; Tsujibe, S.; Nanno, M.; Shida, K. Probiotic induction of interleukin-10 and interleukin-12 production by macrophages is modulated by co-stimulation with microbial components. J. Dairy Sci. 2018, 101, 2838–2841. [Google Scholar] [CrossRef] [PubMed]
- Savino, F.; Garro, M.; Montanari, P.; Galliano, I.; Bergallo, M. Crying Time and RORγ/FOXP3 Expression in Lactobacillus reuteri DSM17938-Treated Infants with Colic: A Randomized Trial. J. Pediatr. 2018, 192, 171–177.e1. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.J.; Lee, A.; Yoo, H.J.; Kim, M.; Noh, G.M.; Lee, J.H. Supplementation with the probiotic strain Weissella cibaria JW15 enhances natural killer cell activity in nondiabetic subjects. J. Funct. Foods. 2018, 48, 153–158. [Google Scholar] [CrossRef]
- Cummings, J.; Macfarlane, G. Gastrointestinal effects of prebiotics. Br. J. Nutr. 2002, 87, S145–S151. [Google Scholar] [CrossRef] [PubMed]
- Gibson, G.R.; Hutkins, R.; Sanders, M.E.; Prescott, S.L.; Reimer, R.A.; Salminen, S.J.; Scott, K.; Stanton, C.; Swanson, K.S.; Cani, P.D.; et al. Expert consensus document: The International Scientific Association for Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of prebiotics. Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 491. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Quigley, E.M.; Fried, M.; Gwee, K.-A.; Khalif, I.; Hungin, A.; Lindberg, G.; Abbas, Z.; Fernandez, LB.; Bhatia, S.J.; Schmulson, M.; et al. World Gastroenterology Organisation global guidelines irritable bowel syndrome: A global perspective update September 2015. J. Clin. Gastroenterol. 2016, 50, 704–713. [Google Scholar] [CrossRef] [PubMed]
- Bindels, L.B.; Delzenne, N.M.; Cani, P.D.; Walter, J. Towards a more comprehensive concept for prebiotics. Nat. Rev. Gastroenterol. Hepatol. 2015, 12, 303. [Google Scholar] [CrossRef] [PubMed]
- Oozeer, R.; van Limpt, K.; Ludwig, T.; Ben Amor, K.; Martin, R.; Wind, R.D.; Boehm, G.; Knol, J. Intestinal microbiology in early life: Specific prebiotics can have similar functionalities as human-milk oligosaccharides. Am. J. Clin. Nutr. 2013, 98, 561S–571S. [Google Scholar] [CrossRef] [PubMed]
- Douëllou, T.; Montel, M.-C.; Sergentet, D.T. Invited review: Anti-adhesive properties of bovine oligosaccharides and bovine milk fat globule membrane-associated glycoconjugates against bacterial food enteropathogens. J. Dairy Sci. 2017, 100, 3348–3359. [Google Scholar] [CrossRef] [PubMed]
- Weaver, C.M.; Martin, B.R.; Nakatsu, C.H.; Armstrong, A.P.; Clavijo, A.; McCabe, L.D.; Duignan, S.; Schoterman, M.H.; van den Heuvel, E.G. Galactooligosaccharides improve mineral absorption and bone properties in growing rats through gut fermentation. J. Agric. Food Chem. 2011, 59, 6501–6510. [Google Scholar] [CrossRef] [PubMed]
- Sazawal, S.; Dhingra, U.; Sarkar, A.; Dhingra, P.; Deb, S.; Marwah, D.; Menon, V.P.; Kumar, J.; Black, R.E. Efficacy of milk fortified with a probiotic Bifidobacterium lactis (DR-10) and prebiotic galacto-oligosaccharides in prevention of morbidity and on nutritional status. Asia Pac. J. Clin. Nutr. 2004, 13, S28. [Google Scholar]
- Yeung, C.K.; Glahn, R.E.; Welch, R.M.; Miller, D.D. Prebiotics and iron bioavailability—Is there a connection? J. Food Sci. 2005, 70, R88–R92. [Google Scholar] [CrossRef]
- Rumessen, J.J.; Bodé, S.; Hamberg, O.; Gudmand-Høyer, E. Fructans of Jerusalem artichokes: Intestinal transport, absorption, fermentation, and influence on blood glucose, insulin, and C-peptide responses in healthy subjects. Am. J. Clin. Nutr. 1990, 52, 675–681. [Google Scholar] [CrossRef] [PubMed]
- Hamer, H.M.; Jonkers, D.; Venema, K.; Vanhoutvin, S.; Troost, F.; Brummer, R.J. The role of butyrate on colonic function. Aliment. Pharmacol. Ther. 2008, 27, 104–119. [Google Scholar] [CrossRef] [PubMed]
- Kim, C.H. B cell-helping functions of gut microbial metabolites. Microb. Cell 2016, 3, 529. [Google Scholar] [CrossRef] [PubMed]
- Meng, X.; Li, S.; Li, Y.; Gan, R.-Y.; Li, H.-B. Gut Microbiota’s Relationship with Liver Disease and Role in Hepatoprotection by Dietary Natural Products and Probiotics. Nutrients 2018, 10, 1457. [Google Scholar] [CrossRef] [PubMed]
- Vulevic, J.; Drakoularakou, A.; Yaqoob, P.; Tzortzis, G.; Gibson, G.R. Modulation of the fecal microflora profile and immune function by a novel trans-galactooligosaccharide mixture (B-GOS) in healthy elderly volunteers. Am. J. Clin. Nutr. 2008, 88, 1438–1446. [Google Scholar] [PubMed]
- Dehghan, P.; Gargari, B.P.; Jafar-Abadi, M.A. Oligofructose-enriched inulin improves some inflammatory markers and metabolic endotoxemia in women with type 2 diabetes mellitus: A randomized controlled clinical trial. Nutrition 2014, 30, 418–423. [Google Scholar] [CrossRef] [PubMed]
| Author/Year | Study Design | Population Characteristics | Intervention | Control/Placebo Group | Duration | Clinical Findings (vs. Control/Placebo Group) |
|---|---|---|---|---|---|---|
| Alisi et al. (2014) [86] | Parallel-arm, double-blind RCT | Children aged 11 years with NAFLD (n = 44); intervention (n = 22); placebo (n = 22) | VSL#3 (450 billion bacteria per sachet, one sachet/day) + low calorie diet + moderate physical activity | 1 cap/day + healthy habits (low calorie diet + moderate physical activity) | 4 months | <BMI, fatty liver, insulin resistance; >GLP-1 |
| Aller et al. (2011) [97] | Randomized, double-blind, parallel, placebo-controlled trial | Patients with NAFLD (n = 28) | 500 million of Lactobacillus bulgaricus and Streptococcus thermophilus (1 tablet/day) | 1 cap/day of starch | 3 months | Improved liver function, glucose metabolism and pro-inflammatory markers; no changes in anthropometric measures |
| Famouri et al. (2017) [87] | Triple-blind randomized placebo-controlled clinical trial | Obese children and adolescents (12.7 years) with NAFLD (n = 64); intervention (n = 32); placebo (n = 32) | L. acidophilus ATCC B3208 (3 × 109 CFU), L. rhamnosus DSMZ21690 (6 × 109 CFU), B. lactis DSMZ 32,296 (2 × 109 CFU) and B. bifidum ATCC SD6576 (2 × 109 CFU) cap/day + healthy lifestyle habits | 1 cap/day of placebo + healthy lifestyle habits | 12 weeks | =BMI, weight; <WC |
| Gomes et al. (2017) [92] | Randomized, double-blind, placebo-controlled, two arm, parallel-group clinical trial | Obese women aged 20–59 years (n = 43); intervention (n = 21); placebo (n = 22) | L. acidophilus LA-14, L. casei LC-11, Lactococcuslactis LL-23, B. bifidum BB-06, B. lactis BL-4 (2 × 1010 CFU/day) + dietary intervention | 1 cap/day placebo + dietary prescription | 8 weeks | =BMI and weight; <WC |
| Higashikawa et al. (2016) [93] | Randomized, double-blind, placebo-controlled clinical trial | Overweight adults aged 20–70 years (n = 62); Intervention I (n = 21); Intervention II (n = 21); placebo (n = 20) | Intervention I: Living LP28; Intervention II Heat-killed LP28 (Pediococcus pentosaceus) (1011 CFU/day) | 1 cap/day placebo | 12 weeks | <BMI, WC after Intervention II |
| Jung et al. (2015) [91] | Double-blind, placebo-controlled, randomized clinical trial | Obese adults aged 20–65 years (n = 120); intervention (n = 60); placebo (n = 60) | L.curvatus HY7601 + L. plantarum KY1032 (2.5 × 109 CFU of probiotics/2 cap/day) + healthy lifestyle habits | 2 cap/day placebo + healthy lifestyle habits | 12 weeks | <Body weight, WC and fat |
| Kadooka et al. (2010) [98] | Multicenter, double-blind, randomized, placebo-controlled intervention trial | Adults aged 33-63 years with obese tendencies (n = 87); intervention (n = 43); control group (n = 44) | Fermented milk containing Lactobacillus gasseri SBT2055 (5 × 1010 CFU/100 g fermented milk). Intake of 200 g/day | Intake of 200 g/day of fermented milk without probiotic | 12 weeks | <Abdominal visceral, subcutaneous fat areas, body weight and BMI |
| Kim et al. (2018) [94] | Randomized, double-blind, placebo-controlled trial | Obese adults aged 20–75 years (n = 90); low-dose intervention (n = 30); high-dose intervention (n = 30); placebo (n = 30) | Low (109 CFU/day) and high (1010 CFU/2 cap/twice a day) dose of Lactobacillus gasseri BNR17 + lifestyle changes | 2 cap/twice a day of placebo + lifestyle changes | 12 weeks | <Visceral adipose tissue; WC in high-dose group; <WC in low-dose group |
| Luoto et al. (2010) [84] | Randomized, double-blind, prospective follow-up study | Mother–child pairs (n = 113); intervention (n = 54); placebo (n = 59) | Lactobacillus rhamnosus GG (1 × 1010 CFU/day) | 1 cap/day of placebo (microcrystalline cellulose) | Mothers 4 weeks before expected delivery; in infants up to 6 month old | <Weight gain at 1 year of life and 4 years; no changes in later stages of development |
| Minami et al. (2018) [95] | Randomized, double-blind, placebo-controlled trial | Healthy pre-obese adults aged 20–64 years (n = 80); intervention (n = 40); placebo (n = 40) | Bifidobacterium breve B-3 (1010 CFU/2 cap/day) | 2 cap/day of placebo | 12 weeks | <Body fat mass |
| Mykhal´chyshyn et al. (2013) [99] | Open label study | Adult patients with T2D and NAFLD (n = 72); intervention (n = 45); control group (n = 27) | “Symbiter” containing concentrated biomass of 14 alive probiotic bacteria + oral antidiabetic therapy | Only hypoglycemic drugs | 4 weeks | <Pro-inflammatory markers; no changes in anthropometric measures |
| Osterberg et al. (2015) [100] | Randomized, double-blind placebo-controlled clinical trial | Healthy non-obese young male adults (18–30 years) (n = 20); intervention (n = 9); placebo (n = 11) | Two sachets of VSL#3 (450 billion bacteria per sachet in milk shake/once a day) + high fat diet (HFD) | Two sachets of placebo in milk shake/once a day + HFD | 4 weeks | <Weight and fat |
| Pedret et al. (2018) [96] | Randomized, parallel, double-blind, placebo-controlled trial | Abdominally obese adults (n = 126); Intervention I (n = 42); Intervention II (n = 44); placebo (n = 40) | Bifidobacterium animalis subsp. Lactis CECT 8145 (Intervention I) or its heat-killed form (Intervention II) (1010 CFU/cap/day) | 1 cap/day of placebo | 3 months | <BMI, WC and waist circumference/height ratio; no differences between live and heat-killed form |
| Sánchez et al. (2017) [101] | Double-blind, randomized, placebo-controlled trial | Obese adults aged 18–55 years (n = 125); intervention (n = 62); placebo (n = 63) | L. rhamnosus CGMCC1.3724 (1.62 × 108 CFU/2 cap/day) + healthy eating behavior | 250 mg of maltodextrin + 3 mg magnesium stearate + healthy eating behavior | 12 weeks | <Weight |
| Sanchis-Chordá et al. (2018) [88] | Double-blind, randomized, placebo-controlled trial | Obese children (aged 10–15 years) with insulin resistance (n = 48); intervention (n = 23); placebo (n = 25) | B. pseudocatenulatum CECT 7765 (109−10 CFU/day) + dietary recommendations | Placebo + dietary recommendations | 13 weeks | <Weight body |
| Szulinska et al. (2018) [102] | Randomized-double-blind, placebo-controlled clinical trial | Obese postmenopausal women aged 45–70 years (n = 81); low-dose intervention (n = 27); high-dose intervention (n = 27); placebo (n = 27) | Low (2.5 × 109 CFU/day) and high dose (1010 CFU/day/two sachets per day) of probiotic mixture including nine different strains of Lactobacillus and Bifidobacterium | 1 cap/day of placebo | 12 weeks | <Body weight, BMI and fat mass in low and high-dose group; improved lipid metabolism in the high-dose group |
| Vajro et al. (2011) [85] | Double-blind, placebo-controlled pilot study | Obese children (aged 10–13 years) with hypertransaminasemia and ultrasonographic bright liver (n = 20); intervention (n = 10); placebo (n = 10) | Lactobacillus rhamnosus GG (12 billion CFU/day) | 1 cap/day of placebo | 8 weeks | <Hypertransaminasemia Effects on BMI and visceral fat in combination with lifestyle interventions |
| Author/Year | Study Design | Population Characteristics | Intervention | Control/Placebo Group | Duration | Clinical Findings (vs. Control/Placebo Group) |
|---|---|---|---|---|---|---|
| Cani et al. (2006) [103] | Single-blinded, cross-over, placebo-controlled design, pilot study | Healthy non-obese adults aged 21–35 years (n = 10); intervention (n = 5); placebo (n = 5) | Prebiotic-supplemented diet (16 g oligofructose/day) divided into breakfast (8 g) and dinner (8 g) | Placebo (dextrin maltose) (16 g/day) divided into breakfast (8 g) and dinner (8 g) | 2 weeks | >Satiety; <hunger, energy intake after dinner and total energy intake |
| Cani et al. (2009) [104] | Randomized, double-blind, parallel, placebo-controlled trial | Healthy non-obese adults aged 21–38 years (n = 10); intervention (n = 5); placebo (n = 5) | Prebiotic-supplemented diet (16 g chicory-derived fructan/day) divided into breakfast (8 g) and dinner (8 g) | Placebo (dextrin maltose) (16 g/day) divided into breakfast (8 g) and dinner (8 g) | 2 weeks | >GLP-1, PYY <Hunger No effects on satiety |
| Dehghan et al. (2014) [105] | Triple-blind randomized controlled study | Adult women with T2D aged 20–65 years (n = 49); intervention (n = 24); placebo (n = 25) | Prebiotic-supplemented diet (10 g inulin/day) | 10 g maltodextrin/day | 8 weeks | <Fasting glucose, energy intake and pro-inflammatory and oxidative markers |
| Edrisi et al. (2018) [106] | RCT | Overweight and obese adults (n = 105) aged 20–50 years; Intervention I (n = 35); Intervention II (n = 35); control (n = 35) | Energy-restricted diet containing rice bran (Intervention I) or rice husk powder (Intervention II) (according to DRIs) | Low-calorie diet | 12 weeks | <Weight, BMI, WC and pro-inflammatory markers |
| Genta et al. (2009) [107] | Double-blind, placebo-controlled study | Obese women aged 31–49 years (n = 35) | Yacon syrup (approximately 12.5 g FOS/day) + healthy hypocaloric diet | Placebo syrup (tartaric acid 2.5%, carboxymethylcellulose 1.8%, saccharine 2.5% and glycerine 10%) + healthy hypocaloric diet | 17 weeks | <Body weight, BMI, WC, fasting serum insulin, HOMA; >satiety; no changes in total cholesterol and triglycerides |
| Hume et al. (2017) [108] | Randomized, double-blind, placebo-controlled trial | Overweight and obese children aged 7–12 years (n = 42); intervention (n = 22); control (n = 20) | 8 g oligofructose-enriched inulin/day | Equicaloric dose of a 3.3 g maltodextrin placebo/day | 16 weeks | >Satiety, prospective food consumption and ghrelin. <Energy intake |
| Nicolucci et al. (2017) [109] | Single center, double-blind, placebo-controlled trial | Overweight or obesity children aged 7–12 years (n = 42); intervention (n = 22); control (n = 20) | 8 g/day (13.2 kcal/day) of oligofructose-enriched inulin | Equicaloric dose of a 3.3 g maltodextrin placebo/day | 16 weeks | <Body weight z-score, percent body fat and trunk fat. >Bifidobacterium <Bacteroides |
| Parnell et al. (2009) [110] | Randomized, double-blind, placebo-controlled trial | Overweight and obese adults aged 20–70 years (n = 39); intervention (n = 21); control (n = 18) | Prebiotic-enriched diet (21 g oligofructose/day) | Equicaloric amount of maltodextrin placebo | 12 weeks | < Body weight, fat mass, energy intake, postprandial ghrelin and insulin; no effects on postprandial glucose, PYY and GLP-1 |
| Reimer et al. (2017) [111] | Single-centre, placebo-controlled, double-blind RCT | Adults with overweight/obesity aged 18–75 years (n = 96); control (n = 27); prebiotic (n = 26); protein bar (n = 21); combination (n = 22) | (1) control bar; (2) prebiotic bar (inulin-type fructans with 6 g oligofructose + 2 g inulin from chicory root); (3) protein bar (5 g whey protein); (4) combination bar (8 g inulin-type fructans + 5 g whey protein). | Control isocaloric bar (100 kcal/bar) | 12 weeks | <Body fat in (3) <Hunger, desire to eat and prospective food consumption in (2), (3) and (4) >Bifidobacterium in (2) and (4) |
| Russo et al. (2012) [112] | Cross-over RCT, double-blind | Healthy males adults aged 18–20 years (n = 20); intervention (n = 10); control (n = 10) | Prebiotic-supplemented diet (11% inulin-enriched pasta) | Control pasta diet (100% durum wheat semolina) | 5 weeks | >Neurotensin, somatostatin, GLP-2 |
| Stenman et al. (2016) [113] | Double-blind, randomized, parallel, placebo-controlled clinical trial | Healthy adults aged 18–65 years (n = 225); placebo (n = 56); LU (n = 53); B420 (n = 48); mix (n = 52) | Prebiotic treatment: dietary fiber Litesse® Ultra polydextrose (LU) (12 g/day); probiotic treatment: B420 (1010 CFU/day); mix treatment: LU + B420 | Microcrystalline cellulose placebo (12 g/day) | 6 months | Probiotic and Mix treatment: <body fat, WC and food intake; no effects of prebiotic treatment. |
| Verhoel et al. (2011) [114] | Randomized, placebo-controlled, cross-over, double-blind clinical trial | Normal weight and overweight adults aged 20–60 years (n = 29) | Prebiotic-supplemented diet containing (1) 10 g FOS/day or (2) 16 g FOS/day | Placebo based on maltodextrin 16 g/day | 13 days | >PYY in treatment (2); no effects on appetite, satiety, GLP-1 and energy intake |
| Whelan et al. (2006) [115] | Prospective, randomized, double-blind, cross-over trial | Healthy adults aged 28–30 years (n = 11) | Prebiotic-supplemented liquid enteral formula (18 g pea fiber + 10 g FOS/day) | Standard enteral formula (Nutren 1.0, Nestlé) | 2 weeks | >Fullness and satiety |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cerdó, T.; García-Santos, J.A.; G. Bermúdez, M.; Campoy, C. The Role of Probiotics and Prebiotics in the Prevention and Treatment of Obesity. Nutrients 2019, 11, 635. https://doi.org/10.3390/nu11030635
Cerdó T, García-Santos JA, G. Bermúdez M, Campoy C. The Role of Probiotics and Prebiotics in the Prevention and Treatment of Obesity. Nutrients. 2019; 11(3):635. https://doi.org/10.3390/nu11030635
Chicago/Turabian StyleCerdó, Tomás, José Antonio García-Santos, Mercedes G. Bermúdez, and Cristina Campoy. 2019. "The Role of Probiotics and Prebiotics in the Prevention and Treatment of Obesity" Nutrients 11, no. 3: 635. https://doi.org/10.3390/nu11030635
APA StyleCerdó, T., García-Santos, J. A., G. Bermúdez, M., & Campoy, C. (2019). The Role of Probiotics and Prebiotics in the Prevention and Treatment of Obesity. Nutrients, 11(3), 635. https://doi.org/10.3390/nu11030635






