Effect of Fish Oil Supplementation on Hepatic and Visceral Fat in Overweight Men: A Randomized Controlled Trial
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Experimental Design
2.3. Measurements
2.3.1. Anthropometric Assessment
2.3.2. Biochemical Parameters
2.3.3. Habitual Dietary and Physical Activity Control
2.3.4. Magnetic Resonance Imaging (MRI) and Proton Magnetic Resonance Spectroscopy (1H-MRS)
2.4. Statistical Analysis
3. Results
3.1. Recruitment and Participation
3.2. Baseline Measures
3.3. Effects of 12 Weeks of Supplementation
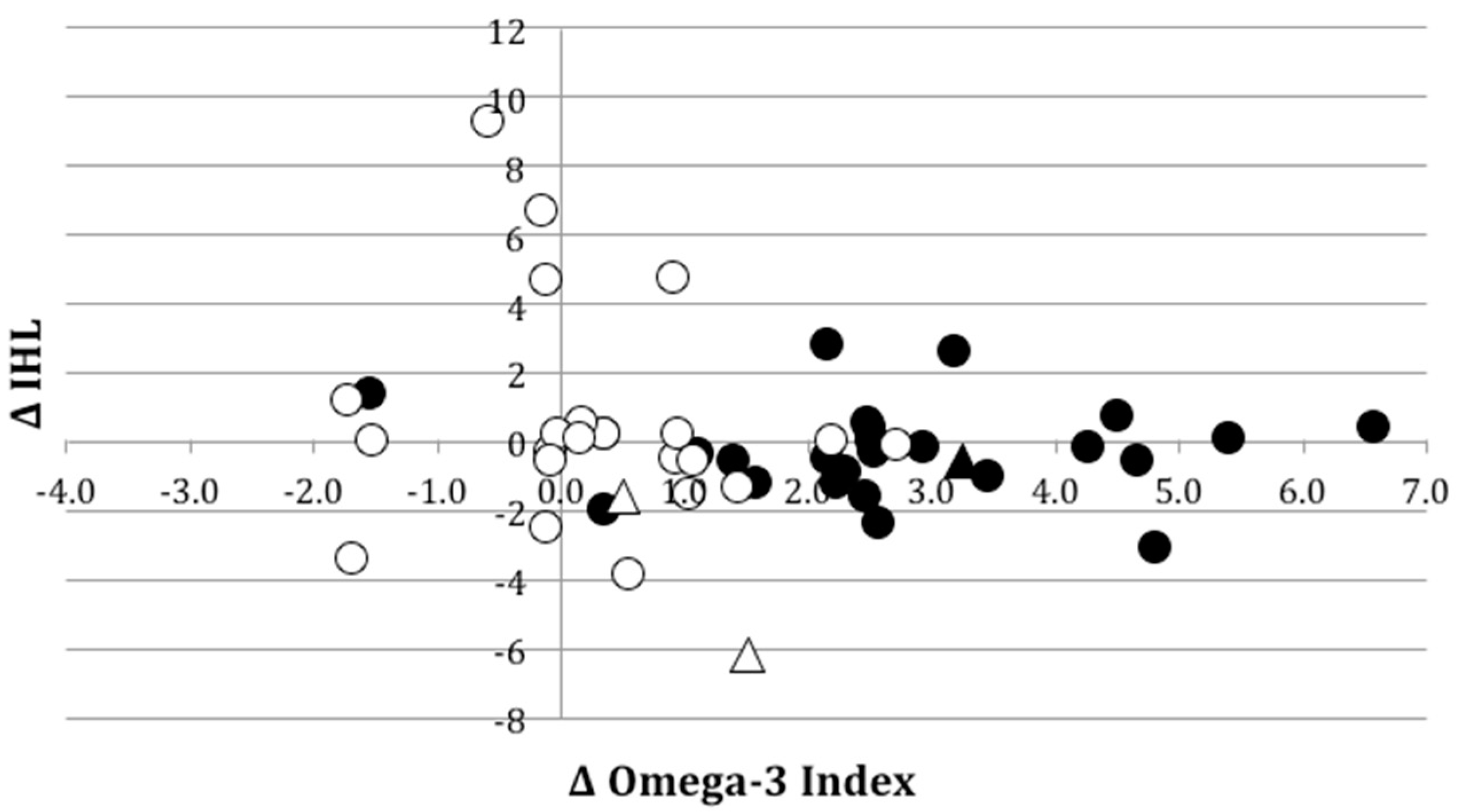
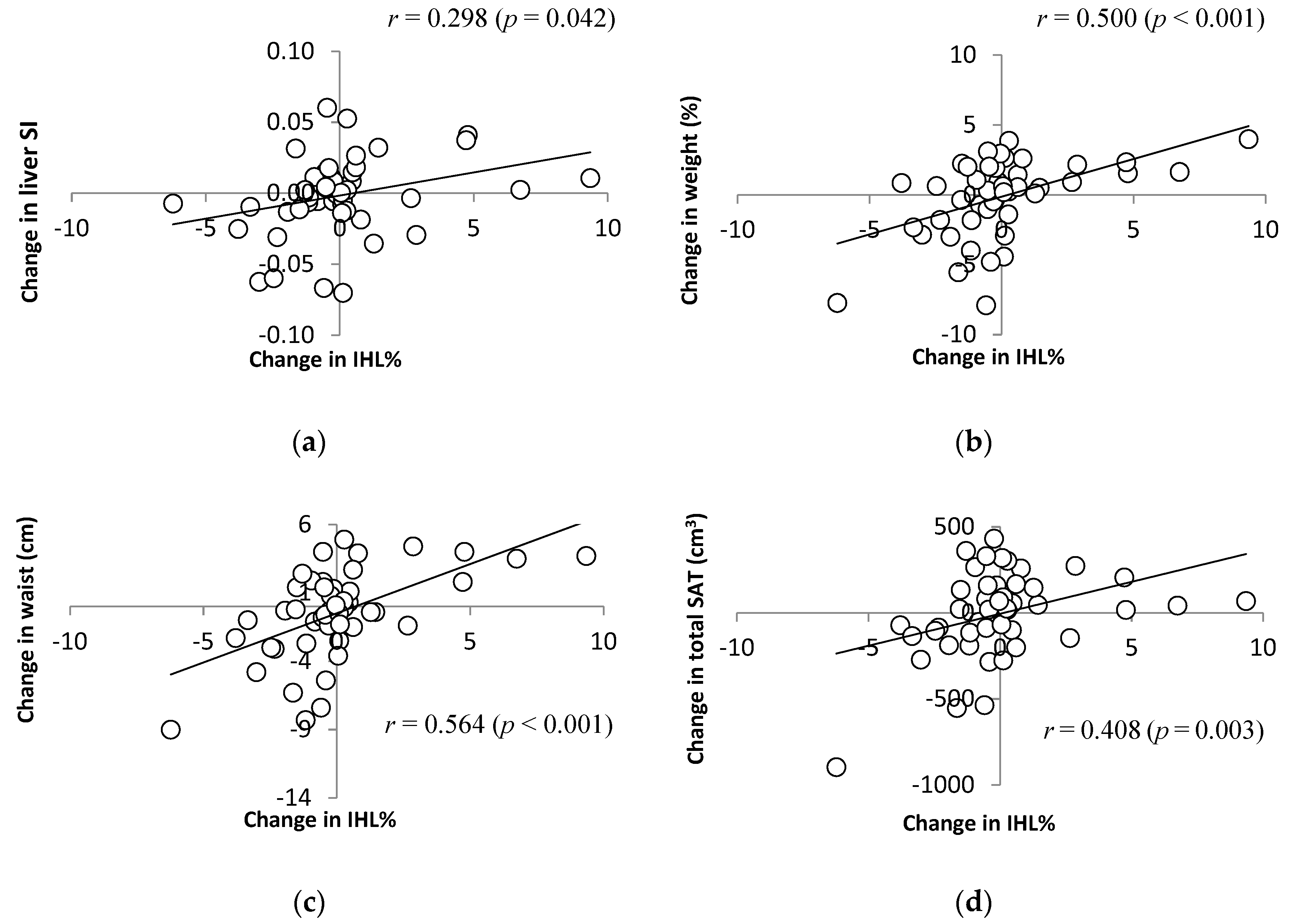
3.3.1. Liver Fat Concentration and Body Composition
3.3.2. Blood Biochemistry
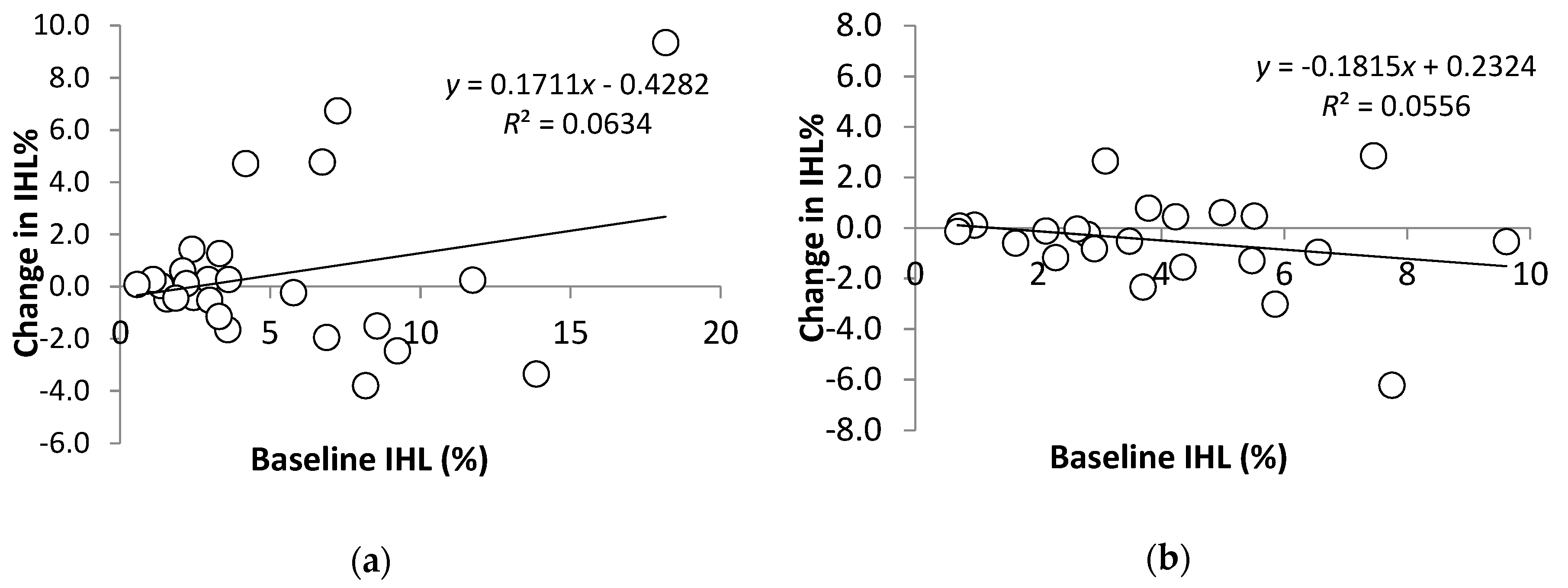
3.3.3. Sub-Analysis of Participants with NAFLD
3.3.4. Self-Reported Habitual Dietary Intake and Physical Activity
4. Discussion
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Ng, M.; Fleming, T.; Robinson, M.; Thomson, B.; Graetz, N.; Margono, C.; Mullany, E.C.; Biryukov, S.; Abbafati, C.; Abera, S.F. Global, regional, and national prevalence of overweight and obesity in children and adults during 1980–2013: A systematic analysis for the Global Burden of Disease Study 2013. Lancet 2014, 384, 766–781. [Google Scholar] [CrossRef]
- Van Gaal, L.F.; Mertens, I.L.; De Block, C.E. Mechanisms linking obesity with cardiovascular disease. Nature 2006, 444, 875–880. [Google Scholar] [CrossRef] [PubMed]
- Targher, G.; Arcaro, G. Non-alcoholic fatty liver disease and increased risk of cardiovascular disease. Atherosclerosis 2007, 191, 235–240. [Google Scholar] [CrossRef] [PubMed]
- Angulo, P. GI Epidemiology: Nonalcoholic fatty liver disease. Aliment. Pharmacol. Ther. 2007, 25, 883–889. [Google Scholar] [CrossRef] [PubMed]
- Mahady, S.E.; Webster, A.C.; Walker, S.; Sanyal, A.; George, J. The role of thiazolidinediones in non-alcoholic steatohepatitis—A systematic review and meta analysis. J. Hepatol. 2011, 55, 1383–1390. [Google Scholar] [CrossRef] [PubMed]
- Musso, G.; Gambino, R.; Cassader, M.; Pagano, G. A Meta-Analysis of Randomized Trials for the Treatment of Nonalcoholic Fatty Liver Disease. Hepatology 2010, 52, 79–104. [Google Scholar] [CrossRef] [PubMed]
- Volynets, V.; Machann, J.; Kuper, M.A.; Maier, I.B.; Spruss, A.; Konigsrainer, A.; Bischoff, S.C.; Bergheim, I. A moderate weight reduction through dietary intervention decreases hepatic fat content in patients with non-alcoholic fatty liver disease (NAFLD): A pilot study. Eur. J. Nutr. 2013, 52, 527–535. [Google Scholar] [CrossRef] [PubMed]
- Brea, A.; Puzo, J. Non-alcoholic fatty liver disease and cardiovascular risk. Int. J. Cardiol. 2013, 167, 1109–1117. [Google Scholar] [CrossRef] [PubMed]
- Franz, M.J.; Vanwormer, J.J.; Crain, A.L.; Boucher, J.L.; Histon, T.; Caplan, W.; Bowman, J.D.; Pronk, N.P. Weight-loss outcomes: A systematic review and meta-analysis of weight-loss clinical trials with a minimum 1-year follow-up. J. Am. Diet. Assoc. 2007, 107, 1755–1767. [Google Scholar] [CrossRef] [PubMed]
- Flock, M.R.; Harris, W.S.; Kris-Etherton, P.M. Long-chain omega-3 fatty acids: Time to establish a dietary reference intake. Nutr. Rev. 2013, 71, 692–707. [Google Scholar] [CrossRef] [PubMed]
- Food Standards Australia and New Zealand. Australian Food, Supplement and Nutrient Database (AUSNUT) 2011–2013. Available online: www.foodstandards.gov.au/science/monitoringnutrients/ausnut/Pages/default.aspx (accessed on 15 July 2017).
- Harris, W.S.; Kris-Etherton, P.M.; Harris, K.A. Intakes of Long-Chain Omega-3 Fatty Acid Associated with Reduced Risk for Death from Coronary Heart Disease in Healthy Adults. Curr. Atherosclerol. Rep. 2008, 10, 503–509. [Google Scholar] [CrossRef]
- Zelber-Sagi, S.; Nitzan-Kaluski, D.; Goldsmith, R.; Webb, M.; Blendis, L.; Halpern, Z.; Oren, R. Long term nutritional intake and the risk for non-alcoholic fatty liver disease (NAFLD): A population based study. J. Hepatol. 2007, 47, 711–717. [Google Scholar] [CrossRef] [PubMed]
- Alwayn, I.P.J.; Gura, K.; Nose, V.; Zausche, B.; Javid, P.; Garza, J.; Verbesey, J.; Voss, S.; Ollero, M.; Andersson, C.; et al. Omega-3 fatty acid supplementation prevents hepatic steatosis in a murine model of nonalcoholic fatty liver disease. Pediatr. Res. 2005, 57, 445–452. [Google Scholar] [CrossRef] [PubMed]
- Buchman, A. Total parenteral nutrition-associated liver disease. JPEN J. Parenter. Enter. Nutr. 2002, 26, S43–S48. [Google Scholar] [CrossRef] [PubMed]
- Burns, D.L.; Gill, B.M. Reversal of parenteral nutrition-associated liver disease with a fish oil-based lipid emulsion (Omegaven) in an adult dependent on home parenteral nutrition. JPEN J. Parenter. Enter. Nutr. 2013, 37, 274–280. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Periz, A.; Horrillo, R.; Ferre, N.; Gronert, K.; Dong, B.; Moran-Salvador, E.; Titos, E.; Martinez-Clemente, M.; Lopez-Parra, M.; Arroyo, V.; et al. Obesity-induced insulin resistance and hepatic steatosis are alleviated by omega-3 fatty acids: A role for resolvins and protectins. FASEB J. 2009, 23, 1946–1957. [Google Scholar] [CrossRef] [PubMed]
- Parker, H.M.; Johnson, N.A.; Burdon, C.A.; Cohn, J.S.; O’Connor, H.T.; George, J. Omega-3 supplementation and non-alcoholic fatty liver disease: A systematic review and meta-analysis. J. Hepatol. 2012, 56, 944–951. [Google Scholar] [CrossRef] [PubMed]
- Munro, I.A.; Garg, M.L. Prior supplementation with long chain omega-3 polyunsaturated fatty acids promotes weight loss in obese adults: A double-blinded randomised controlled trial. Food Funct. 2013, 4, 650–658. [Google Scholar] [CrossRef] [PubMed]
- Masterton, G.S.; Plevris, J.N.; Hayes, P.C. Review article: Omega-3 fatty acids—A promising novel therapy for non-alcoholic fatty liver disease. Aliment. Pharmacol. Ther. 2010, 31, 679–692. [Google Scholar] [CrossRef] [PubMed]
- Sawangjit, R.; Chongmelaxme, B.; Phisalprapa, P.; Saokaew, S.; Thakkinstian, A.; Kowdley, K.V.; Chaiyakunapruk, N. Comparative efficacy of interventions on nonalcoholic fatty liver disease (NAFLD): A PRISMA-compliant systematic review and network meta-analysis. Medicine 2016, 95, e4529. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.-H.; Wang, Y.-F.; Xu, Q.-H.; Chen, S.-S. Omega-3 fatty acids as a treatment for non-alcoholic fatty liver disease in children: A systematic review and meta-analysis of randomized controlled trials. Clin. Nutr. 2016, 37, 516–521. [Google Scholar] [CrossRef] [PubMed]
- De Castro, G.S.; Calder, P.C. Non-alcoholic fatty liver disease and its treatment with n-3 polyunsaturated fatty acids. Clin. Nutr. 2018, 37, 37–55. [Google Scholar] [CrossRef] [PubMed]
- Zuta, P.; Simpson, B.; Zhao, X.; Leclerc, L. The effect of α-tocopherol on the oxidation of mackerel oil. Food Chem. 2007, 100, 800–807. [Google Scholar] [CrossRef]
- Madmani, M.E.; Yusuf Solaiman, A.; Tamr Agha, K.; Madmani, Y.; Shahrour, Y.; Essali, A.; Kadro, W. Coenzyme Q10 for heart failure. Cochrane Database Syst. Rev. 2014, 6. [Google Scholar] [CrossRef] [PubMed]
- Overvad, K.; Diamant, B.; Holm, L.; Hølmer, G.; Mortensen, S.; Stender, S. Coenzyme Q 10 in health and disease. Eur. J. Clin. Nutr. 1999, 53, 764–770. [Google Scholar] [CrossRef] [PubMed]
- Parker, H.M.; O’Connor, H.T.; Keating, S.E.; Cohn, J.S.; Garg, M.L.; Caterson, I.D.; George, J.; Johnson, N.A. Efficacy of the Omega-3 Index in predicting non-alcoholic fatty liver disease in overweight and obese adults: A pilot study. Br. J. Nutr. 2015, 114, 780–787. [Google Scholar] [CrossRef] [PubMed]
- Heart Foundation. Fish, Fish Oils, n-3 Polyunsaturated Fatty Acids and Cardiovascular Health. Available online: www.heartfoundation.org.au/images/uploads/main/For_professionals/Fish-FishOils-revie-of-evidence.pdf (accessed on 23 Februaty 2019).
- Harris, W.S.; von Schacky, C. The Omega-3 Index: A new risk factor for death from coronary heart disease? Prev. Med. 2004, 39, 212–220. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Physical Status: The Use of and Interpretation of Anthropometry; Report of a WHO Expert Committee; World Health Organization: Geneva, Switzerland, 1995. [Google Scholar]
- Lepage, G.; Roy, C.C. Direct transesterification of all classes of lipids in a one-step reaction. J. Lipid Res. 1986, 27, 114–120. [Google Scholar] [PubMed]
- Bouchard, C.; Tremblay, A.; Leblanc, C.; Lortie, G.; Savard, R.; Theriault, G. A method to assess energy-expenditure in children and adults. Am. J. Clin. Nutr. 1983, 37, 461–467. [Google Scholar] [CrossRef] [PubMed]
- Johnson, N.A.; Walton, D.W.; Sachinwalla, T.; Thompson, C.H.; Smith, K.; Ruell, P.A.; Stannard, S.R.; George, J. Noninvasive assessment of hepatic lipid composition: Advancing understanding and management of fatty liver disorders. Hepatology 2008, 47, 1513–1523. [Google Scholar] [CrossRef] [PubMed]
- Positano, V.; Gastaldelli, A.; Sironi, A.M.; Santarelli, M.F.; Lombardi, M.; Landini, L. An accurate and robust method for unsupervised assessment of abdominal fat by MRI. J. Magn. Reson. Imaging 2004, 20, 684–689. [Google Scholar] [CrossRef] [PubMed]
- Ryan, M.C.; Itsiopoulos, C.; Thodis, T.; Ward, G.; Trost, N.; Hofferberth, S.; O’Dea, K.; Desmond, P.V.; Johnson, N.A.; Wilson, A.M. The Mediterranean diet improves hepatic steatosis and insulin sensitivity in individuals with non-alcoholic fatty liver disease. J. Hepatol. 2013, 59, 138–143. [Google Scholar] [CrossRef] [PubMed]
- Szczepaniak, L.S.; Nurenberg, P.; Leonard, D.; Browning, J.D.; Reingold, J.S.; Grundy, S.; Hobbs, H.H.; Dobbins, R.L. Magnetic resonance spectroscopy to measure hepatic triglyceride content: Prevalence of hepatic steatosis in the general population. Am. J. Physiol. Endocrinol. MeTable 2005, 288, E462–E468. [Google Scholar] [CrossRef] [PubMed]
- Armjo-Olivo, S.; Warren, S.; Magee, D. Intention to treat analysis, compliance, drop-outs and how to deal with missing data in clinical researcl: A review. Phys. Ther. Rev. 2009, 14, 36–49. [Google Scholar] [CrossRef]
- Scorletti, E.; Bhatia, L.; McCormick, K.G.; Clough, G.F.; Nash, K.; Hodson, L.; Moyses, H.E.; Calder, P.C.; Byrne, C.D.; Investigators, W.S. Effects of purified eicosapentaenoic and docosahexaenoic acids in nonalcoholic fatty liver disease: Results from the Welcome* study. Hepatology 2014, 60, 1211–1221. [Google Scholar] [CrossRef] [PubMed]
- Iser, D.; Ryan, M. Fatty liver disease A practical guide for GPs. Aust. Fam. Phys. 2013, 42, 444–447. [Google Scholar]
- Vega, G.L.; Chandalia, M.; Szczepaniak, L.S.; Grundy, S.M. Effects of N-3 fatty acids on hepatic triglyceride content in humans. J. Investig. Med. 2008, 56, 780–785. [Google Scholar] [CrossRef] [PubMed]
- Argo, C.K.; Patrie, J.T.; Lackner, C.; Henry, T.D.; de Lange, E.E.; Weltman, A.L.; Shah, N.L.; Al-Osaimi, A.M.; Pramoonjago, P.; Jayakumar, S. Effects of n-3 fish oil on metabolic and histological parameters in NASH: A double-blind, randomized, placebo-controlled trial. J. Hepatol. 2015, 62, 190–197. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.-H.; Yang, L.-H.; Sha, K.-H.; Liu, T.-G.; Zhang, L.-G.; Liu, X.-X. Efficacy of poly-unsaturated fatty acid therapy on patients with nonalcoholic steatohepatitis. World J. Gastroenterol. 2015, 21, 7008. [Google Scholar] [CrossRef] [PubMed]
- Nogueira, M.A.; Oliveira, C.P.; Alves, V.A.F.; Stefano, J.T.; dos Reis Rodrigues, L.S.; Torrinhas, R.S.; Cogliati, B.; Barbeiro, H.; Carrilho, F.J.; Waitzberg, D.L. Omega-3 polyunsaturated fatty acids in treating non-alcoholic steatohepatitis: A randomized, double-blind, placebo-controlled trial. Clin. Nutr. 2016, 35, 578–586. [Google Scholar] [CrossRef] [PubMed]
- Sanyal, A.J.; Abdelmalek, M.F.; Suzuki, A.; Cummings, O.W.; Chojkier, M.; EPE-A Study Group. No significant effects of ethyl-eicosapentanoic acid on histologic features of nonalcoholic steatohepatitis in a phase 2 trial. Gastroenterology 2014, 147, 377–384. [Google Scholar] [CrossRef] [PubMed]
- Scorletti, E.; Byrne, C.D. Omega-3 fatty acids and non-alcoholic fatty liver disease: Evidence of efficacy and mechanism of action. Mol. Asp. Med. 2018, 64, 135–146. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.; Minihane, A.M.; Talmud, P.J.; Wright, J.W.; Murphy, M.C.; Williams, C.M.; Griffin, B.A. Dietary long-chain n-3 PUFAs increase LPL gene expression in adipose tissue of subjects with an atherogenic lipoprotein phenotype. J. Lipid Res. 2002, 43, 979–985. [Google Scholar] [PubMed]
- Park, Y.; Harris, W.S. Dose-response of n-3 polyunsaturated fatty acids on lipid profile and tolerability in mildly hypertriglyceridemic subjects. J. Med. Food 2009, 12, 803–808. [Google Scholar] [CrossRef] [PubMed]
- Dasarathy, S.; Dasarathy, J.; Khiyami, A.; Yerian, L.; Hawkins, C.; Sargent, R.; McCullough, A.J. Double blind randomized placebo controlled clinical trial of omega 3 fatty acids for the treatment of diabetic patients with nonalcoholic steatohepatitis. J. Clin. Gastroenterol. 2015, 49, 137. [Google Scholar] [CrossRef] [PubMed]
- Capanni, M.; Calella, F.; Biagini, M.R.; Genise, S.; Raimondi, L.; Bedogni, G.; Svegliati-Baroni, G.; Sofi, F.; Milani, S.; Abbate, R.; et al. Prolonged n-3 polyunsaturated fatty acid supplementation ameliorates hepatic steatosis in patients with non-alcoholic fatty liver disease: A pilot study. Aliment. Pharmacol. Ther. 2006, 23, 1143–1151. [Google Scholar] [CrossRef] [PubMed]
- Sofi, F.; Giangrandi, I.; Cesari, F.; Corsani, I.; Abbate, R.; Gensini, G.F.; Casini, A. Effects of a 1-year dietary intervention with n-3 polyunsaturated fatty acid-enriched olive oil on non-alcoholic fatty liver disease patients: A preliminary study. Int. J. Food Sci. Nutr. 2010, 61, 792–802. [Google Scholar] [CrossRef] [PubMed]
- Cussons, A.J.; Watts, G.F.; Mori, T.A.; Stuckey, B.G.A. Omega-3 fatty acid supplementation decreases liver fat content in polycystic ovary syndrome: A randomized controlled trial employing proton magnetic resonance spectroscopy. J. Clin. Endocrinol. MeTable 2009, 94, 3842–3848. [Google Scholar] [CrossRef] [PubMed]
- Hatzitolios, A.; Savopoulos, C.; Lazaraki, G.; Sidiropoulos, I.; Haritanti, P.; Lefkopoulos, A.; Karagiannopoulou, G.; Tzioufa, V.; Dimitrios, K. Efficacy of omega-3 fatty acids, atorvastatin and orlistat in non-alcoholic fatty liver disease with dyslipidemia. Indian J. Gastroenterol. 2004, 23, 131–134. [Google Scholar] [PubMed]
- Spadaro, L.; Magliocco, O.; Spampinato, D.; Piro, S.; Oliveri, C.; Alagona, C.; Papa, G.; Rabuazzo, A.M.; Purrello, F. Effects of n-3 polyunsaturated fatty acids in subjects with nonalcoholic fatty liver disease. Dig. Liver Dis. 2008, 40, 194–199. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, N.; Sano, K.; Horiuchi, A.; Tanaka, E.; Kiyosawa, K.; Aoyama, T. Highly purified eicosapentaenoic acid treatment improves nonalcoholic steatohepatitis. J. Clin. Gastroenterol. 2008, 42, 413–418. [Google Scholar] [CrossRef] [PubMed]
- Zhu, F.S.; Liu, S.; Chen, X.M.; Huang, Z.G.; Zhang, D.W. Effects of n-3 polyunsaturated fatty acids from seal oils on nonalcoholic fatty liver disease associated with hyperlipidemia. World J. Gastroenterol. 2008, 14, 6395–6400. [Google Scholar] [CrossRef] [PubMed]
- Di Minno, M.N.D.; Russolillo, A.; Lupoli, R.; Ambrosino, P.; Di Minno, A.; Tarantino, G. Omega-3 fatty acids for the treatment of non-alcoholic fatty liver disease. World J. Gastroenterol. 2012, 18, 5839–5847. [Google Scholar] [CrossRef] [PubMed]
- Von Schacky, C. Omega-3 index and cardiovascular health. Nutrients 2014, 6, 799–814. [Google Scholar] [CrossRef] [PubMed]
- Howe, P.R.; Buckley, J.D.; Murphy, K.J.; Pettman, T.; Milte, C.; Coates, A.M. Relationship between erythrocyte omega-3 content and obesity is gender dependent. Nutrients 2014, 6, 1850–1860. [Google Scholar] [CrossRef] [PubMed]
- Jump, D.B.; Depner, C.M.; Tripathy, S. Omega-3 fatty acid supplementation and cardiovascular disease. J. Lipid Res. 2012, 53, 2525–2545. [Google Scholar] [CrossRef] [PubMed]
- Balk, E.M.; Lichtenstein, A.H.; Chung, M.; Kupelnick, B.; Chew, P.; Lau, J. Effects of omega-3 fatty acids on serum markers of cardiovascular disease risk: A systematic review. Atherosclerosis 2006, 189, 19–30. [Google Scholar] [CrossRef] [PubMed]
- Eslick, G.D.; Howe, P.R.C.; Smith, C.; Priest, R.; Bensoussan, A. Benefits of fish oil supplementation in hyperlipidemia: A systematic review and meta-analysis. Int. J. Cardiol. 2009, 136, 4–16. [Google Scholar] [CrossRef] [PubMed]
- Hartweg, J.; Farmer, A.J.; Perera, R.; Holman, R.R.; Neil, H.A.W. Meta-analysis of the effects of n-3 polyunsaturated fatty acids on lipoproteins and other emerging lipid cardiovascular risk markers in patients with type 2 diabetes. Diabetologia 2007, 50, 1593–1602. [Google Scholar] [CrossRef] [PubMed]
- Kris-Etherton, P.M.; Harris, W.S.; Appel, L.J.; Nutrition, C. Fish consumption, fish oil, omega-3 fatty acids, and cardiovascular disease. Circulation 2002, 106, 2747–2757. [Google Scholar] [CrossRef] [PubMed]
| Characteristic | Total (n = 50) | Placebo (n = 25) | Fish Oil (n = 25) | p-Value a |
|---|---|---|---|---|
| Age (years) | 34.2 (1.5) | 34.7 (2.3) | 33.6 (2.0) | 0.723 |
| Caucasian ethnicity (n, %) | 44 (86) | 23 (92) | 21 (84) | 0.384 |
| Anthropometry | ||||
| Weight (kg) | 90.9 (1.3) | 92.4 (2.0) | 89.3 (1.6) | 0.221 |
| Waist circumference (cm) | 100.5 (0.7) | 101.0 (0.9) | 99.9 (1.1) | 0.435 |
| BMI (kg·m−2) | 27.9 (0.2) | 28.0 (0.3) | 27.8 (0.3) | 0.646 |
| Liver fat and adiposity | ||||
| IHL (%) | 4.6 (0.5) | 5.5 (0.9) | 3.7 (0.4) | 0.078 |
| Hepatic SI | 0.949 (0.006) | 0.950 (0.006) | 0.948 (0.010) | 0.830 |
| SAT at umbilicus (cm3) | 279.7 (9.1) | 285.7 (12.4) | 273.6 (13.4) | 0.510 |
| VAT at umbilicus (cm3) | 103.5 (8.0) | 104.1 (12.4) | 102.9 (10.5) | 0.942 |
| Body Fat by BIA (%) | 23.0 (0.4) | 22.8 (0.6) | 23.1 (0.5) | 0.660 |
| NAFLD (Y/N) | 16/34 | 10/15 | 6/19 | 0.225 |
| Diet and exercise | ||||
| Reported habitual energy expenditure (KJ/day) b | 16,590 (561) | 17,569 (998) | 15,630 (489) | 0.084 |
| Reported energy intake (kJ/day) | 10,529 (416) | 10,619 (652) | 10,436 (531) | 0.823 |
| Liver function tests | ||||
| ALT (U/L) | 34.4 (1.7) | 34.5 (2.4) | 34.3 (2.4) | 0.962 |
| AST (U/L) | 28.6 (1.3) | 26.5 (0.9) | 30.7 (2.4) | 0.106 |
| GGT (U/L) | 30.3 (2.4) | 30.5 (3.2) | 30.2 (3.6) | 0.947 |
| Other biochemistry | ||||
| Triglycerides (mmol/L) | 1.3 (0.1) | 1.5 (0.1) | 1.0 (0.1) | 0.001 |
| Omega-3 Index c | 7.9 (0.2) | 8.0 (0.3) | 7.9 (0.3) | 0.821 |
| Placebo | Fish Oil | P (t) | P (g × t) 1 | |||
|---|---|---|---|---|---|---|
| Week 0 to 6 | Week 0 to 12 | Week 0 to 6 | Week 0 to 12 | |||
| Anthropometry | ||||||
| Weight (kg) | 0.2 (0.4) | 0.3 (0.5) | −0.2 (0.2) | −0.4 (0.4) | 0.856 | 0.517 |
| Waist (cm) | 0.0 (0.6) | −0.1 (0.7) | −0.3 (0.3) | −0.9 (0.6) | 0.385 | 0.439 |
| BMI (kg·m-2) | 0.0 (0.1) | 0.1 (0.2) | −0.1 (0.1) | −0.1 (0.1) | 0.869 | 0.475 |
| Liver fat and adiposity | ||||||
| IHL (%) | 0.7 (0.7) | 0.3 (0.7) | 0.0 (0.3) | −0.2 (0.3) | 0.157 | 0.801 |
| Hepatic SI | 0.000 (0.008) | 0.002 (0.006) | −0.009 (0.007) | −0.006 (0.005) | 0.514 | 0.884 |
| SAT, umbilicus (cm3) | 1.6 (4.7) | 0.1 (5.4) | −3.0 (3.2) | −2.7 (2.8) | 0.823 | 0.746 |
| VAT, umbilicus (cm3) | −1.4 (5.5) | −3.8 (4.6) | −6.1 (3.6) | −1.3 (4.7) | 0.683 | 0.197 |
| SAT, total (L) | 0.00 (0.09) | 0.00 (0.12) | −0.07 (0.05) | −0.07 (0.07) | 0.909 | 0.970 |
| VAT, total (L) | 0.00 (0.07) | −0.07 (0.07) | −0.12 (0.07) | −0.07 (0.09) | 0.768 | 0.224 |
| Body fat, BIA (%) | 0.1 (0.2) | 0.0 (0.3) | −0.5 (0.3) | −0.2 (0.4) | 0.525 | 0.273 |
| Liver function tests | ||||||
| ALT (U/L) | −2.0 (1.5) | −3.6 (1.1) | 2.0 (2.5) | 4.1 (3.6) | 0.894 | 0.387 |
| AST (U/L) | −0.8 (0.7) | −2.3 (0.6) | 0.6 (3.2) | −0.8 (3.1) | 0.464 | 0.983 |
| GGT (U/L) | −0.1 (1.4) | −1.3 (1.4) | 0.0 (2.1) | 2.0 (1.8) | 0.596 | 0.052 |
| Other biochemistry | ||||||
| Triglycerides (mmol/L) | 0.0 (0.12) | 0.0 (0.13) | 0.1 (0.08) | 0.1 (0.08) | 0.772 | 0.694 |
| Omega-3 Index | 0.4 (0.26) | 0.3 (0.21) | 1.8 (0.27) | 2.8 (0.34) | 0.018 | 0.006 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Parker, H.M.; Cohn, J.S.; O’Connor, H.T.; Garg, M.L.; Caterson, I.D.; George, J.; Johnson, N.A. Effect of Fish Oil Supplementation on Hepatic and Visceral Fat in Overweight Men: A Randomized Controlled Trial. Nutrients 2019, 11, 475. https://doi.org/10.3390/nu11020475
Parker HM, Cohn JS, O’Connor HT, Garg ML, Caterson ID, George J, Johnson NA. Effect of Fish Oil Supplementation on Hepatic and Visceral Fat in Overweight Men: A Randomized Controlled Trial. Nutrients. 2019; 11(2):475. https://doi.org/10.3390/nu11020475
Chicago/Turabian StyleParker, Helen M., Jeffrey S. Cohn, Helen T. O’Connor, Manohar L. Garg, Ian D. Caterson, Jacob George, and Nathan A. Johnson. 2019. "Effect of Fish Oil Supplementation on Hepatic and Visceral Fat in Overweight Men: A Randomized Controlled Trial" Nutrients 11, no. 2: 475. https://doi.org/10.3390/nu11020475
APA StyleParker, H. M., Cohn, J. S., O’Connor, H. T., Garg, M. L., Caterson, I. D., George, J., & Johnson, N. A. (2019). Effect of Fish Oil Supplementation on Hepatic and Visceral Fat in Overweight Men: A Randomized Controlled Trial. Nutrients, 11(2), 475. https://doi.org/10.3390/nu11020475






