Association between Lower Intake of Minerals and Depressive Symptoms among Elderly Japanese Women but Not Men: Findings from Shika Study
Abstract
1. Introduction
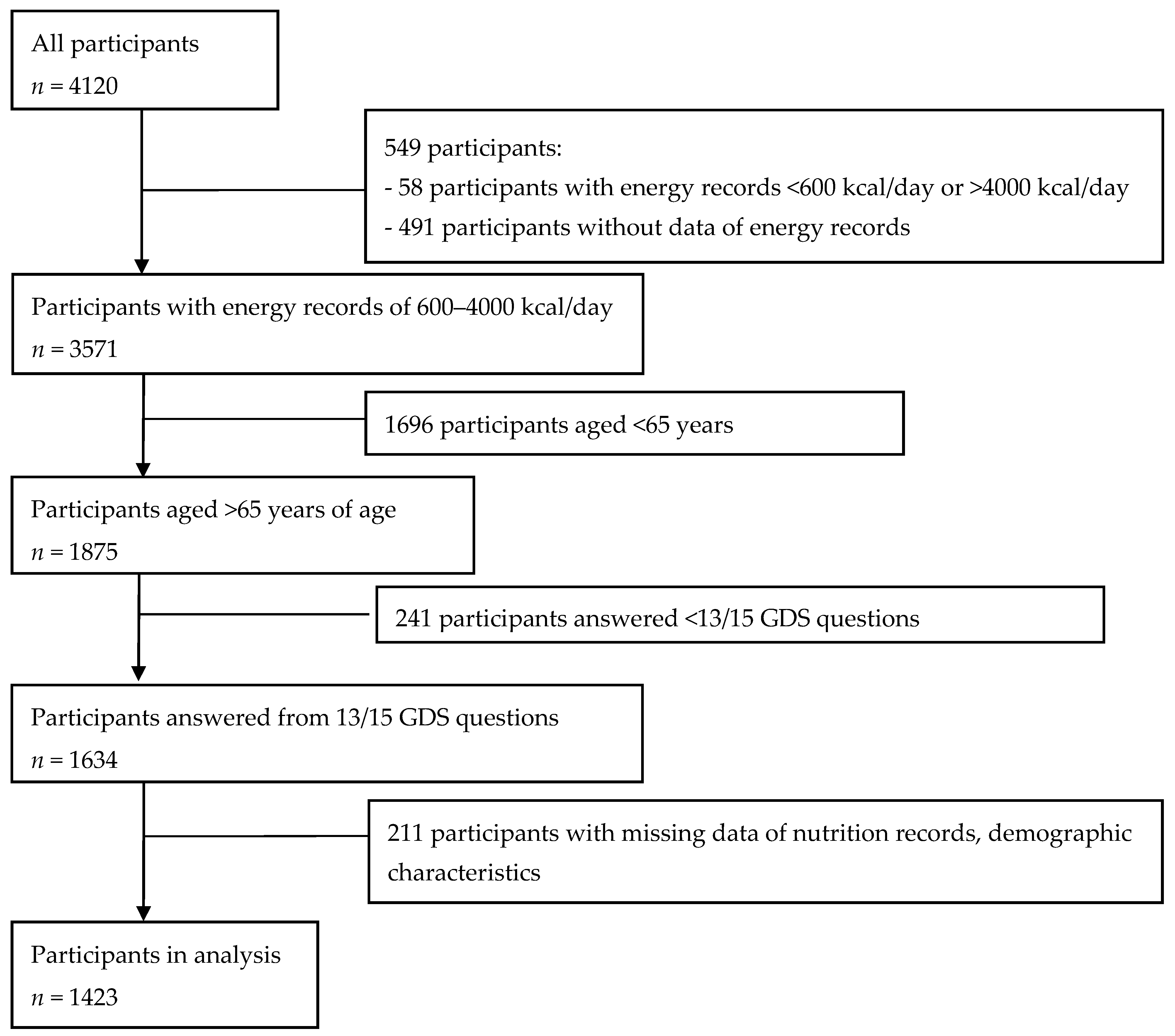
2. Materials and Methods
2.1. Study Population
2.2. Depressive Symptoms Assessment
2.3. Nutrients Assessment
2.4. Other Variables
2.5. Statistical Analysis
3. Results
3.1. Characteristics of Participants in Non-Depressive and Depressive Symptoms Groups.
3.2. Mineral Intake in Non-Depressive and Depressive Symptoms Groups in Each Gender
3.3. Relationship between Mineral Intake and Depressive Symptoms
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ferrari, A.J.; Charlson, F.J.; Norman, R.E.; Patten, S.B.; Freedman, G.; Murray, C.J.L.; Vos, T.; Whiteford, H.A. Burden of Depressive Disorders by Country, Sex, Age, and Year: Findings from the Global Burden of Disease Study 2010. PLoS Med. 2013, 10, e1001547. [Google Scholar] [CrossRef] [PubMed]
- Kessler, R.C.; Berglund, P.; Demler, O.; Jin, R.; Koretz, D.; Merikangas, K.R.; Rush, A.J.; Walters, E.E.; Wang, P.S. The Epidemiology of Major Depressive Disorder. JAMA 2003, 289, 3095. [Google Scholar] [CrossRef]
- Horackova, K.; Kopecek, M.; Machů, V.; Kagstrom, A.; Aarsland, D.; Motlova, L.B.; Cermakova, P. Prevalence of late-life depression and gap in mental health service use across European regions. Eur. Psychiatry 2019, 57, 19–25. [Google Scholar] [CrossRef]
- WHO. Depression and Other Common Mental Disorders. Global Health Estimates; WHO: Geneva, Switzerland, 2017; Available online: http://apps.who.int/iris/bitstream/handle/10665/254610/WHO-MSD-MER-2017.2-eng.pdf;jsessionid=7666696F2B865494E1DDFB4C5132EF81?sequence=1 (accessed on 7 September 2018).
- Hasin, D.S.; Goodwin, R.D.; Stinson, F.S.; Grant, B.F. Epidemiology of Major Depressive Disorder: Results From the National Epidemiologic Survey on Alcoholism and Related Conditions. Arch. Gen. Psychiatry 2005, 62, 1097. [Google Scholar] [CrossRef] [PubMed]
- Blazer, D.G. Depression in Late Life: Review and Commentary. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2003, 58, M249–M265. [Google Scholar] [CrossRef]
- Zis, P.; Daskalaki, A.; Bountouni, I.; Sykioti, P.; Varrassi, G.; Paladini, A. Depression and chronic pain in the elderly: Links and management challenges. Clin. Interv. Aging 2017, 12, 709–720. [Google Scholar] [CrossRef]
- Ministry of Health, Labour and Welfare. Summary of Patient Survey; Ministry of Health, Labour and Welfare: Tokoyo, Japan, 2014. Available online: https://www.mhlw.go.jp/english/database/db-hss/sps_2014.html (accessed on 12 September 2018).
- Li, Z.; Wang, W.; Xin, X.; Song, X.; Zhang, D. Association of total zinc, iron, copper and selenium intakes with depression in the US adults. J. Affect. Disord. 2018, 228, 68–74. [Google Scholar] [CrossRef] [PubMed]
- Miki, T.; Kochi, T.; Eguchi, M.; Kuwahara, K.; Tsuruoka, H.; Kurotani, K.; Ito, R.; Akter, S.; Kashino, I.; Pham, N.M.; et al. Dietary intake of minerals in relation to depressive symptoms in Japanese employees: The Furukawa Nutrition and Health Study. Nutrition 2015, 31, 686–690. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Li, B.; Song, X.; Zhang, D. Dietary zinc and iron intake and risk of depression: A meta-analysis. Psychiatry Res. 2017, 251, 41–47. [Google Scholar] [CrossRef]
- Miyake, Y.; Tanaka, K.; Okubo, H.; Sasaki, S.; Furukawa, S.; Arakawa, M. Manganese intake is inversely associated with depressive symptoms during pregnancy in Japan: Baseline data from the Kyushu Okinawa Maternal and Child Health Study. J. Affect. Disord. 2017, 211, 124–129. [Google Scholar] [CrossRef]
- Dama, M.; Van Lieshout, R.J.; Mattina, G.; Steiner, M. Iron Deficiency and Risk of Maternal Depression in Pregnancy: An Observational Study. J. Obstet. Gynaecol. Can. 2018, 40, 698–703. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Trumpff, C.; Genkinger, J.; Davis, A.; Spann, M.; Werner, E.; Monk, C.; Singh, A.; Trumpff, C.; Genkinger, J.; et al. Micronutrient Dietary Intake in Latina Pregnant Adolescents and Its Association with Level of Depression, Stress, and Social Support. Nutrients 2017, 9, 1212. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.T.T.; Tsujiguchi, H.; Kambayashi, Y.; Hara, A.; Miyagi, S.; Yamada, Y.; Nakamura, H.; Shimizu, Y.; Hori, D.; Suzuki, F.; et al. Relationship between Vitamin Intake and Depressive Symptoms in Elderly Japanese Individuals: Differences with Gender and Body Mass Index. Nutrients 2017, 9, 1319. [Google Scholar] [CrossRef] [PubMed]
- Burke, W.J.; Roccaforte, W.H.; Wengel, S.P. The Short Form of the Geriatric Depression Scale: A Comparison With the 30-Item Form. Top. Geriatr. 1991, 4, 173–178. [Google Scholar] [CrossRef]
- Sugishita, K.; Sugishita, M.; Hemmi, I.; Asada, T.; Tanigawa, T. A Validity and Reliability Study of the Japanese Version of the Geriatric Depression Scale 15 (GDS-15-J). Clin. Gerontol. 2017, 40, 233–240. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, S.; Honda, S.; Murakami, K.; Sasaki, S.; Okubo, H.; Hirota, N.; Notsu, A.; Fukui, M.; Date, C. Both comprehensive and brief self-administered diet history questionnaires satisfactorily rank nutrient intakes in Japanese adults. J. Epidemiol. 2012, 22, 151–159. [Google Scholar] [CrossRef] [PubMed]
- Okubo, H.; Sasaki, S.; Rafamantanantsoa, H.H.; Ishikawa-Takata, K.; Okazaki, H.; Tabata, I. Validation of self-reported energy intake by a self-administered diet history questionnaire using the doubly labeled water method in 140 Japanese adults. Eur. J. Clin. Nutr. 2008, 62, 1343–1350. [Google Scholar] [CrossRef]
- Sasaki, S.; Yanagibori, R.; Amano, K. Self-Administered Diet History Questionnaire Developed for Health Education: A Relative Validation of The Test-Version by Comparison with 3-Day Diet Record in Women. J. Epidemiol. 1998, 8, 203–215. [Google Scholar] [CrossRef]
- Ministry of Health, Labour and Welfare. The National Health and Nutrition Survey in Japan; Ministry of Health, Labour and Welfare: Tokoyo, Japan, 2012. Available online: http://www.mhlw.go.jp/bunya/kenkou/eiyou/h24-houkoku.html (accessed on 25 August 2018).
- Standard Tables of Food Composition in Japan-2015-(Seventh Revised Version); Ministry of Education, Culture, Sports, Science and Technology: Tokoyo, Japan, 2015. Available online: http://www.mext.go.jp/en/policy/science_technology/policy/title01/detail01/1374030.htm (accessed on 8 November 2017).
- Kobayashi, S.; Murakami, K.; Sasaki, S.; Okubo, H.; Hirota, N.; Notsu, A.; Fukui, M.; Date, C. Comparison of relative validity of food group intakes estimated by comprehensive and brief-type self-administered diet history questionnaires against 16 d dietary records in Japanese adults. Public Health Nutr. 2011, 14, 1200–1211. [Google Scholar] [CrossRef]
- Kaner, G.; Soylu, M.; Yüksel, N.; Inanç, N.; Ongan, D.; Başmısırlı, E. Evaluation of Nutritional Status of Patients with Depression. Biomed Res. Int. 2015, 2015, 521481. [Google Scholar] [CrossRef]
- Anjom-Shoae, J.; Sadeghi, O.; Keshteli, A.H.; Afshar, H.; Esmaillzadeh, A.; Adibi, P. The association between dietary intake of magnesium and psychiatric disorders among Iranian adults: A cross-sectional study. Br. J. Nutr. 2018, 120, 693–702. [Google Scholar] [CrossRef] [PubMed]
- Yary, T.; Aazami, S.; Soleimannejad, K. Dietary Intake of Magnesium May Modulate Depression. Biol. Trace Elem. Res. 2013, 151, 324–329. [Google Scholar] [CrossRef] [PubMed]
- Jacka, F.N.; Overland, S.; Stewart, R.; Tell, G.S.; Bjelland, I.; Mykletun, A. Association Between Magnesium Intake and Depression and Anxiety in Community-Dwelling Adults: The Hordaland Health Study. Aust. N. Z. J. Psychiatry 2009, 43, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Ángel Martínez-González, M.; Sánchez-Villegas, A.; Martínez-González, M.A. Magnesium intake and depression: The SUN cohort *. Magnes. Res. 2016, 29, 102–113. [Google Scholar]
- Derom, M.-L.; Martínez-González, M.A.; Sayón-Orea Mdel, C.; Bes-Rastrollo, M.; Beunza, J.J.; Sánchez-Villegas, A. Magnesium Intake Is Not Related to Depression Risk in Spanish University Graduates. J. Nutr. 2012, 142, 1053–1059. [Google Scholar] [CrossRef] [PubMed]
- Yary, T.; Aazami, S. Dietary Intake of Zinc was Inversely Associated with Depression. Biol. Trace Elem. Res. 2012, 145, 286–290. [Google Scholar] [CrossRef] [PubMed]
- Vashum, K.P.; McEvoy, M.; Milton, A.H.; McElduff, P.; Hure, A.; Byles, J.; Attia, J. Dietary zinc is associated with a lower incidence of depression: Findings from two Australian cohorts. J. Affect. Disord. 2014, 166, 249–257. [Google Scholar] [CrossRef] [PubMed]
- Fulkerson, J.A.; Sherwood, N.E.; Perry, C.L.; Neumark-Sztainer, D.; Story, M. Depressive symptoms and adolescent eating and health behaviors: A multifaceted view in a population-based sample. Prev. Med. 2004, 38, 865–875. [Google Scholar] [CrossRef]
- Kim, T.-H.; Choi, J.; Lee, H.-H.; Park, Y. Associations between Dietary Pattern and Depression in Korean Adolescent Girls. J. Pediatr. Adolesc. Gynecol. 2015, 28, 533–537. [Google Scholar] [CrossRef]
- Bae, Y.-J.; Kim, S.-K. Low dietary calcium is associated with self-rated depression in middle-aged Korean women. Nutr. Res. Pract. 2012, 6, 527. [Google Scholar] [CrossRef]
- Miyake, Y.; Tanaka, K.; Okubo, H.; Sasaki, S.; Arakawa, M. Intake of dairy products and calcium and prevalence of depressive symptoms during pregnancy in Japan: A cross-sectional study. BJOG 2015, 122, 336–343. [Google Scholar] [CrossRef] [PubMed]
- Szewczyk, B.; Kubera, M.; Nowak, G. The role of zinc in neurodegenerative inflammatory pathways in depression. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2011, 35, 693–701. [Google Scholar] [CrossRef] [PubMed]
- Bao, B.; Prasad, A.S.; Beck, F.W.; Fitzgerald, J.T.; Snell, D.; Bao, G.W.; Singh, T.; Cardozo, L.J. Zinc decreases C-reactive protein, lipid peroxidation, and inflammatory cytokines in elderly subjects: A potential implication of zinc as an atheroprotective agent. Am. J. Clin. Nutr. 2010, 91, 1634–1641. [Google Scholar] [CrossRef] [PubMed]
- Swardfager, W.; Herrmann, N.; Mazereeuw, G.; Goldberger, K.; Harimoto, T.; Lanctôt, K.L. Zinc in Depression: A Meta-Analysis. Biol. Psychiatry 2013, 74, 872–878. [Google Scholar] [CrossRef] [PubMed]
- Beard, J.L.; Connor, J.R.; Jones, B.C. Iron in the Brain. Nutr. Rev. 2009, 51, 157–170. [Google Scholar] [CrossRef]
- Erikson, K.M.; Jones, B.C.; Hess, E.J.; Zhang, Q.; Beard, J.L. Iron deficiency decreases dopamine D1 and D2 receptors in rat brain. Pharmacol. Biochem. Behav. 2001, 69, 409–418. [Google Scholar] [CrossRef]
- Bourre, J.M. Effects of nutrients (in food) on the structure and function of the nervous system: Update on dietary requirements for brain. Part 1: Micronutrients. J. Nutr. Health Aging 2006, 10, 377–385. [Google Scholar] [PubMed]
- Cubała, W.J.; Landowski, J.; Dziadziuszko, M.; Chrzanowska, A.; Wielgomas, B. Magnesium, C-reactive protein, and cortisol in drug-naïve patients with short illness-duration, first episode major depressive disorder: Possible immunomodulatory role for magnesium. Magnes. Res. 2016, 29, 169–174. [Google Scholar] [PubMed]
- Murck, H. Magnesium and Affective Disorders. Nutr. Neurosci. 2002, 5, 375–389. [Google Scholar] [CrossRef] [PubMed]
- Takeda, A.; Tamano, H. Insight into zinc signaling from dietary zinc deficiency. Brain Res. Rev. 2009, 62, 33–44. [Google Scholar] [CrossRef] [PubMed]
- Morris, M.E. Brain and CSF magnesium concentrations during magnesium deficit in animals and humans: Neurological symptoms. Magnes. Res. 1992, 5, 303–313. [Google Scholar] [PubMed]
- Yu, S.; Feng, Y.; Shen, Z.; Li, M. Diet supplementation with iron augments brain oxidative stress status in a rat model of psychological stress. Nutrition 2011, 27, 1048–1052. [Google Scholar] [CrossRef] [PubMed]
- Knapp, S.; Mandell, A.J.; Bullard, W.P. Calcium activation of brain tryptophan hydroxylase. Life Sci. 1975, 16, 1583–1593. [Google Scholar] [CrossRef]
- Song, M.-F.; Dong, J.-Z.; Wang, Y.-W.; He, J.; Ju, X.; Zhang, L.; Zhang, Y.-H.; Shi, J.-F.; Lv, Y.-Y. CSF miR-16 is decreased in major depression patients and its neutralization in rats induces depression-like behaviors via a serotonin transmitter system. J. Affect. Disord. 2015, 178, 25–31. [Google Scholar] [CrossRef]
- Carman, J.S.; Wyatt, R.J. Calcium: Bivalent cation in the bivalent psychoses. Biol. Psychiatry 1979, 14, 295–336. [Google Scholar] [PubMed]
- Jones, C.E.; Underwood, C.K.; Coulson, E.J.; Taylor, P.J. Copper induced oxidation of serotonin: Analysis of products and toxicity. J. Neurochem. 2007, 102, 1035–1043. [Google Scholar] [CrossRef] [PubMed]
- Bayer, T.A.; Schäfer, S.; Simons, A.; Kemmling, A.; Kamer, T.; Tepest, R.; Eckert, A.; Schüssel, K.; Eikenberg, O.; Sturchler-Pierrat, C.; et al. Dietary Cu stabilizes brain superoxide dismutase 1 activity and reduces amyloid Abeta production in APP23 transgenic mice. Proc. Natl. Acad. Sci. USA 2003, 100, 14187–14192. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, D.; Chu, C.; Mu, J.-J.; Wang, M.; Liu, F.-Q.; Xie, B.-Q.; Yang, F.; Dong, Z.-Z.; Yuan, Z.-Y. Effect of salt intake and potassium supplementation on urinary renalase and serum dopamine levels in Chinese adults. Cardiology 2015, 130, 242–248. [Google Scholar] [CrossRef]
- Dailly, E.; Chenu, F.; Renard, C.E.; Bourin, M. Dopamine, depression and antidepressants. Fundam. Clin. Pharmacol. 2004, 18, 601–607. [Google Scholar] [CrossRef]
- Hori, H.; Kunugi, H. Dopamine agonist-responsive depression. Psychogeriatrics 2013, 13, 189–195. [Google Scholar] [CrossRef]
- Maserejian, N.N.; Hall, S.A.; McKinlay, J.B. Low dietary or supplemental zinc is associated with depression symptoms among women, but not men, in a population-based epidemiological survey. J. Affect. Disord. 2012, 136, 781–788. [Google Scholar] [CrossRef] [PubMed]
- Blazer, D.G.; Kessler, R.C.; McGonagle, K.A.; Swartz, M.S. The prevalence and distribution of major depression in a national community sample: The National Comorbidity Survey. Am. J. Psychiatry 1994, 151, 979–986. [Google Scholar] [PubMed]
- Van de Velde, S.; Bracke, P.; Levecque, K. Gender differences in depression in 23 European countries. Cross-national variation in the gender gap in depression. Soc. Sci. Med. 2010, 71, 305–313. [Google Scholar] [CrossRef] [PubMed]
- Schuch, J.J.J.; Roest, A.M.; Nolen, W.A.; Penninx, B.W.J.H.; de Jonge, P. Gender differences in major depressive disorder: Results from the Netherlands study of depression and anxiety. J. Affect. Disord. 2014, 156, 156–163. [Google Scholar] [CrossRef] [PubMed]
- Parker, G.; Fletcher, K.; Paterson, A.; Anderson, J.; Hong, M. Gender differences in depression severity and symptoms across depressive sub-types. J. Affect. Disord. 2014, 167, 351–357. [Google Scholar] [CrossRef] [PubMed]
- Shim, R.S.; Baltrus, P.; Ye, J.; Rust, G. Prevalence, treatment, and control of depressive symptoms in the United States: Results from the National Health and Nutrition Examination Survey (NHANES), 2005–2008. J. Am. Board Fam. Med. 2011, 24, 33–38. [Google Scholar] [CrossRef] [PubMed]
- Kornstein, S.G.; Schatzberg, A.F.; Thase, M.E.; Yonkers, K.A.; McCullough, J.P.; Keitner, G.I.; Gelenberg, A.J.; Davis, S.M.; Harrison, W.M.; Keller, M.B. Gender Differences in Treatment Response to Sertraline Versus Imipramine in Chronic Depression. Am. J. Psychiatry 2000, 157, 1445–1452. [Google Scholar] [CrossRef] [PubMed]
- Halbreich, U.; Lumley, L.A. The multiple interactional biological processes that might lead to depression and gender differences in its appearance. J. Affect. Disord. 1993, 29, 159–173. [Google Scholar] [CrossRef]
- Chang, C.-C.; Chang, H.-A.; Fang, W.-H.; Chang, T.-C.; Huang, S.-Y. Gender-specific association between serotonin transporter polymorphisms (5-HTTLPR and rs25531) and neuroticism, anxiety and depression in well-defined healthy Han Chinese. J. Affect. Disord. 2017, 207, 422–428. [Google Scholar] [CrossRef] [PubMed]
- Wurtman, J.J. Depression and weight gain: The serotonin connection. J. Affect. Disord. 1993, 29, 183–192. [Google Scholar] [CrossRef]
- De Mel, D.; Suphioglu, C. Fishy business: Effect of omega-3 fatty acids on zinc transporters and free zinc availability in human neuronal cells. Nutrients 2014, 6, 3245–3258. [Google Scholar] [CrossRef] [PubMed]
- Opie, R.S.; Itsiopoulos, C.; Parletta, N.; Sanchez-Villegas, A.; Akbaraly, T.N.; Ruusunen, A.; Jacka, F.N. Dietary recommendations for the prevention of depression. Nutr. Neurosci. 2017, 20, 161–171. [Google Scholar] [CrossRef] [PubMed]
| Characteristics | Non-Depressive Symptoms (1143, 80%) | Depressive Symptoms (280, 20%) | p-Value | |
|---|---|---|---|---|
| Age (mean ± SD) | 73.5 ± 7.0 | 77.2 ± 8.3 | <0.001 | |
| Gender (n, %) | Male | 522 (45.7) | 131 (46.8) | 0.737 |
| Female | 621 (54.3) | 149 (53.2) | ||
| Drinking alcohol (n, %) | Yes | 697 (61.0) | 144 (51.4) | 0.004 |
| No | 446 (39.0) | 136 (48.6) | ||
| Smoking status (n, %) | Current-smoker | 116 (10.2) | 26 (9.3) | 0.282 |
| Ex-smoker | 309 (27.0) | 89 (31.8) | ||
| Non-smoker | 718 (62.8) | 165 (58.9) | ||
| Living status (n, %) | Alone | 139 (12.2) | 41 (14.6) | 0.263 |
| With other | 1004 (87.8) | 239 (85.4) | ||
| Married status (n, %) | Single | 25 (2.2) | 41 (14.6) | 0.001 |
| Married | 843 (73.8) | 239 (85.4) | ||
| Divorce/Die | 275 (24.1) | 6 (2.1) | ||
| Having a job (n, %) | Yes | 280 (24.5) | 35 (12.5) | <0.001 |
| No | 863 (75.5) | 245 (87.5) | ||
| Hypertension (n, %) | Yes | 463 (40.5) | 114 (40.7) | 0.950 |
| No | 680 (59.5) | 166 (59.3) | ||
| Diabetes (n, %) | Yes | 148 (12.9) | 38 (13.6) | 0.782 |
| No | 995 (87.1) | 242 (86.4) | ||
| Hyperlipidemia (n, %) | Yes | 227 (19.9) | 50 (17.9) | 0.448 |
| No | 916 (80.1) | 230 (81.2) | ||
| BMI (mean ± SD) | 23.0 ± 3.0 | 22.6 ± 3.6 | 0.057 | |
| Alcohol consumption (mean ± SD) | 4.48 ± 8.21 | 3.54 ± 8.03 | 0.082 | |
| Total energy (kcal/day) (mean ± SD) | 1862.96 ± 615.78 | 1735.79 ± 560.58 | 0.002 | |
| Nutrient intake (g/day) (mean ± SD) | ||||
| Protein | 73.97 ± 30.99 | 66.96 ± 27.38 | 0.001 | |
| Animal protein | 43.22 ± 24.06 | 38.38 ± 21.56 | 0.001 | |
| Vegetable protein | 30.75 ± 10.67 | 28.58 ± 9.81 | 0.001 | |
| Lipid | 51.69 ± 21.25 | 47.24 ± 20.01 | 0.002 | |
| Animal lipid | 24.72 ± 12.61 | 22.00 ± 11.40 | <0.001 | |
| Vegetable lipid | 26.96 ± 11.27 | 25.24 ± 11.38 | 0.022 | |
| Carbohydrates | 253.44 ± 85.60 | 243.18 ± 79.94 | 0.058 | |
| Ash | 19.87 ± 7.22 | 17.67 ± 6.06 | <0.001 | |
| Mineral intake (mean ± SD) | ||||
| Sodium (g/1000 kcal) | 2.50 ± 0.55 | 2.46 ± 0.55 | 0.231 | |
| Potassium (g/1000 kcal) | 1.47 ± 0.44 | 1.35 ± 0.40 | 0.001 | |
| Calcium (mg/1000 kcal) | 327.56 ± 116.17 | 308.60 ± 117.27 | 0.016 | |
| Magnesium (mg/1000 kcal) | 146.34 ± 34.50 | 136.85 ± 31.96 | 0.001 | |
| Phosphorus (mg/1000 kcal) | 603.40 ± 136.54 | 582.05 ± 134.34 | 0.018 | |
| Iron (mg/1000 kcal) | 4.46 ± 1.20 | 4.19 ± 1.04 | 0.001 | |
| Zinc (mg/1000 kcal) | 4.54 ± 0.67 | 4.43 ± 0.64 | 0.010 | |
| Copper (mg/1000 kcal) | 0.66 ± 0.11 | 0.65 ± 0.11 | 0.013 | |
| Manganese (mg/1000 kcal) | 1.68 ± 0.49 | 1.66 ± 0.50 | 0.417 | |
| Minerals Intakes (Mean ± SD) | Male | Female | p * | ||||
|---|---|---|---|---|---|---|---|
| Non-Depressive Symptoms (n = 522, 79.9%) | Depressive Symptoms (n = 131, 29.1%) | p-Value | Non-Depressive Symptoms (n = 621, 80.6%) | Depressive Symptoms (n = 149, 19.4%) | p-Value | ||
| Sodium (g/1000 kcal) | 2.49 ± 0.57 | 2.47 ± 0.56 | 0.745 | 2.51 ± 0.53 | 2.45 ± 0.55 | 0.173 | 0.511 |
| Potassium (g/1000 kcal) | 1.35 ± 0.38 | 1.32 ± 0.39 | 0.443 | 1.57 ± 0.46 | 1.38 ± 0.41 | <0.001 | 0.003 |
| Calcium (mg/1000 kcal) | 299.77 ± 99.10 | 308.61 ± 121.44 | 0.442 | 350.93 ± 124.13 | 308.59 ± 113.88 | <0.001 | 0.001 |
| Magnesium (mg/1000 kcal) | 139.13 ± 30.01 | 136.94 ± 31.66 | 0.461 | 152.40 ± 36.80 | 136.76 ± 32.32 | <0.001 | 0.003 |
| Phosphorus (mg/1000 kcal) | 574.38 ± 122.65 | 584.48 ± 137.45 | 0.411 | 627.80 ± 142.81 | 579.93 ± 131.97 | <0.001 | 0.001 |
| Iron (mg/1000 kcal) | 4.24 ± 1.11 | 4.14 ± 1.04 | 0.342 | 4.64 ± 1.24 | 4.22 ± 1.04 | <0.001 | 0.045 |
| Zinc (mg/1000 kcal) | 4.39 ± 0.65 | 4.39 ± 0.63 | 0.931 | 4.67 ± 0.66 | 4.46 ± 0.66 | 0.001 | 0.014 |
| Copper (mg/1000 kcal) | 0.64 ± 0.11 | 0.64 ± 0.11 | 0.889 | 0.69 ± 0.11 | 0.65 ± 0.11 | <0.001 | 0.012 |
| Manganese (mg/1000 kcal) | 1.66 ± 0.51 | 1.66 ± 0.48 | 0.993 | 1.70 ± 0.47 | 1.65 ± 0.51 | 0.259 | 0.455 |
| Minerals | Male | Female | ||||
|---|---|---|---|---|---|---|
| B | OR (95% CI) | p-Value | B | OR (95% CI) | p-Value | |
| Sodium | −0.316 | 0.729 (0.502–1.058) | 0.096 | −0.187 | 1.030 (0.969–1.195) | 0.315 |
| Potassium | −0.343 | 0.710 (0.408–1.234) | 0.225 | −0.750 | 0.473 (0.297–0.753) | 0.002 |
| Calcium | <0.001 | 1.000 (0.098–1.002) | 0.743 | −0.002 | 0.998 (0.996–1000) | 0.016 |
| Magnesium | −0.003 | 0.997 (0.990–1.003) | 0.326 | −0.010 | 0.990 (0.985–0.996) | 0.001 |
| Phosphorus | <0.001 | 1.000 (0.999–1.002) | 0.657 | −0.002 | 0.998 (0.997–1.000) | 0.022 |
| Iron | −0.163 | 0.850 (0.702–1.029) | 0.095 | −0.220 | 0.802 (0.676–0.952) | 0.012 |
| Zinc | −0.221 | 0.802 (0.562–1.144) | 0.223 | −0.313 | 0.731 (0.541–0.987) | 0.041 |
| Copper | −1.266 | 0.282 (0.037–2.165) | 0.224 | −2.813 | 0.060 (0.009–0.386) | 0.003 |
| Manganese | −0.309 | 0.734 (0.484–1.114) | 0.146 | −0.225 | 0.799 (0.530–1.205) | 0.284 |
| Minerals | Male | Female | |||
|---|---|---|---|---|---|
| B | OR (95%CI) | B | OR (95%CI) | ||
| Sodium | Quartile 1 | 1.000 (reference | 1.000 (reference) | ||
| Quartile 2 | −0.421 | 0.656 (0.364–1.184) | −0.631 | 0.532 (0.309–0.916) * | |
| Quartile 3 | −0.133 | 0.876 (0.501–1.529) | −0.334 | 0.716 (0.426–1.204) | |
| Quartile 4 | −0.475 | 0.622 (0.342–1.131) | −0.165 | 0.848 (0.506–1.423) | |
| p-value | 0.154 | 0.118 | |||
| Potassium | Quartile 1 | 1.000 (reference) | 1.000 (reference) | ||
| Quartile 2 | 0.161 | 1.175 (0.671–2.058) | −0.053 | 0.948 (0.580–1.552) | |
| Quartile 3 | −0.519 | 0.595 (0.323–1.094) | −0.324 | 0.724 (0.428–1.222) | |
| Quartile 4 | −0.188 | 0.829 (0.465–1.477) | −0.911 | 0.402 (0.220–0.737) * | |
| p-value | 0.132 | 0.004 | |||
| Calcium | Quartile 1 | 1.000 (reference) | 1.000 (reference) | ||
| Quartile 2 | −0.033 | 0.967 (0.551–1.698) | −0.382 | 0.683 (0.413–1.129) | |
| Quartile 3 | −0.713 | 0.490 (0.261–0.921) * | −0.306 | 0.736 (0.443–1.224) | |
| Quartile 4 | 0.106 | 1.111 (0.643–1.920) | −1.007 | 0.365 (0.203–0.658) * | |
| p-value | 0.082 | 0.006 | |||
| Magnesium | Quartile 1 | 1.000 (reference) | 1.000 (reference) | ||
| Quartile 2 | 0.037 | 1.038 (0.594–1.811) | −0.082 | 0.921 (0.564–1.506) | |
| Quartile 3 | −0.256 | 0.774 (0.434–1.377) | −0.368 | 0.692 (0.409–1.170) | |
| Quartile 4 | −0.247 | 0.781 (0.443–1.378) | −0.891 | 0.410 (0.226–0.744) * | |
| p-value | 0.517 | 0.010 | |||
| Phosphorus | Quartile 1 | 1.000 (reference) | 1.000 (reference) | ||
| Quartile 2 | 0.053 | 1.055 (0.594–1.872) | −0.118 | 0.889 (0.540–1.463) | |
| Quartile 3 | −0.346 | 0.707 (0.385–1.299) | −0.254 | 0.776 (0.465–1.296) | |
| Quartile 4 | 0.208 | 1.231 (0.701–2.165) | −0.818 | 0.441 (0.243–0.803) * | |
| p-value | 0.426 | 0.034 | |||
| Iron | Quartile 1 | 1.000 (reference) | 1.000 (reference) | ||
| Quartile 2 | −0.152 | 0.859 (0.485–1.523) | −0.115 | 0.891 (0.534–1.487) | |
| Quartile 3 | −0.372 | 0.689 (0.381–1.246) | −0.120 | 0.887 (0.555–1.418) | |
| Quartile 4 | −0.212 | 0.809 (0.458–1.428) | −1.192 | 0.304 (0.101–0.917) * | |
| p-value | 0.366 | 0.050 | |||
| Zinc | Quartile 1 | 1.000 (reference) | 1.000 (reference) | ||
| Quartile 2 | 0.109 | 1.115 (0.613–2.030) | −0.082 | 0.922 (0.558–1.522) | |
| Quartile 3 | −0.054 | 0.948 (0.509–1.765) | −0.126 | 0.882 (0.532–1.461) | |
| Quartile 4 | −0.187 | 0.830 (0.444–1.550) | −0.771 | 0.463 (0.255–0.839) * | |
| p-value | 0.584 | 0.023 | |||
| Copper | Quartile 1 | 1.000 (reference) | 1.000 (reference) | ||
| Quartile 2 | −0.255 | 0.775 (0.422–1.424) | −0.209 | 0.811 (0.493–1.335) | |
| Quartile 3 | −0.253 | 0.776 (0.426–1.414) | −0.497 | 0.608 (0.361–1.024) | |
| Quartile 4 | −0.255 | 0.775 (0.421–1.424) | −0.837 | 0.433 (0.245–0.766) * | |
| p-value | 0.541 | 0.005 | |||
| Manganese | Quartile 1 | 1.000 (reference) | 1.000 (reference) | ||
| Quartile 2 | 0.188 | 1.207 (0.670–2.175) | 0.080 | 1.084 (0.645–1.820) | |
| Quartile 3 | 0.413 | 1.511 (0.843–2.707) | −0.224 | 0.800 (0.467–1.370) | |
| Quartile 4 | −0.190 | 0.827 (0.448–1.528) | −0.291 | 0.747 (0.430–1.299) | |
| p-value | 0.460 | 0.361 | |||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Thi Thu Nguyen, T.; Miyagi, S.; Tsujiguchi, H.; Kambayashi, Y.; Hara, A.; Nakamura, H.; Suzuki, K.; Yamada, Y.; Shimizu, Y.; Nakamura, H. Association between Lower Intake of Minerals and Depressive Symptoms among Elderly Japanese Women but Not Men: Findings from Shika Study. Nutrients 2019, 11, 389. https://doi.org/10.3390/nu11020389
Thi Thu Nguyen T, Miyagi S, Tsujiguchi H, Kambayashi Y, Hara A, Nakamura H, Suzuki K, Yamada Y, Shimizu Y, Nakamura H. Association between Lower Intake of Minerals and Depressive Symptoms among Elderly Japanese Women but Not Men: Findings from Shika Study. Nutrients. 2019; 11(2):389. https://doi.org/10.3390/nu11020389
Chicago/Turabian StyleThi Thu Nguyen, Thao, Sakae Miyagi, Hiromasa Tsujiguchi, Yasuhiro Kambayashi, Akinori Hara, Haruki Nakamura, Keita Suzuki, Yohei Yamada, Yukari Shimizu, and Hiroyuki Nakamura. 2019. "Association between Lower Intake of Minerals and Depressive Symptoms among Elderly Japanese Women but Not Men: Findings from Shika Study" Nutrients 11, no. 2: 389. https://doi.org/10.3390/nu11020389
APA StyleThi Thu Nguyen, T., Miyagi, S., Tsujiguchi, H., Kambayashi, Y., Hara, A., Nakamura, H., Suzuki, K., Yamada, Y., Shimizu, Y., & Nakamura, H. (2019). Association between Lower Intake of Minerals and Depressive Symptoms among Elderly Japanese Women but Not Men: Findings from Shika Study. Nutrients, 11(2), 389. https://doi.org/10.3390/nu11020389





