DASH Dietary Pattern and Cardiometabolic Outcomes: An Umbrella Review of Systematic Reviews and Meta-Analyses
Abstract
:1. Introduction
2. Materials and Methods
2.1. Design
2.2. Study selection
2.3. Data extraction
2.4. Risk of Bias Assessment
2.5. Outomes
2.6. Evidence Synthesis
2.7. Grading of the Evidence
3. Results
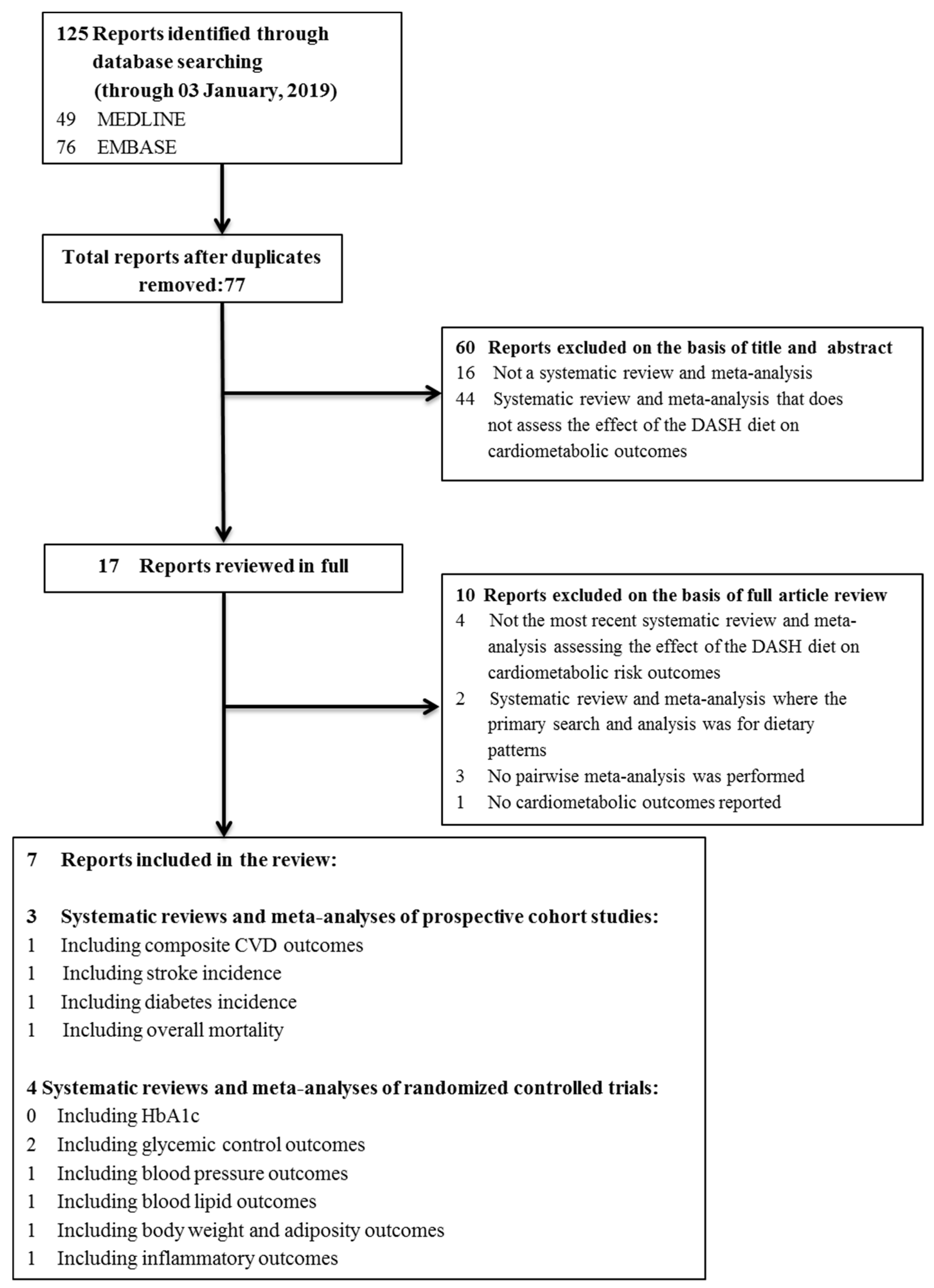
3.1. Search Results
3.2. Outcomes
3.2.1. Systematic Reviews and Meta-Analyses of Prospective Cohort Studies
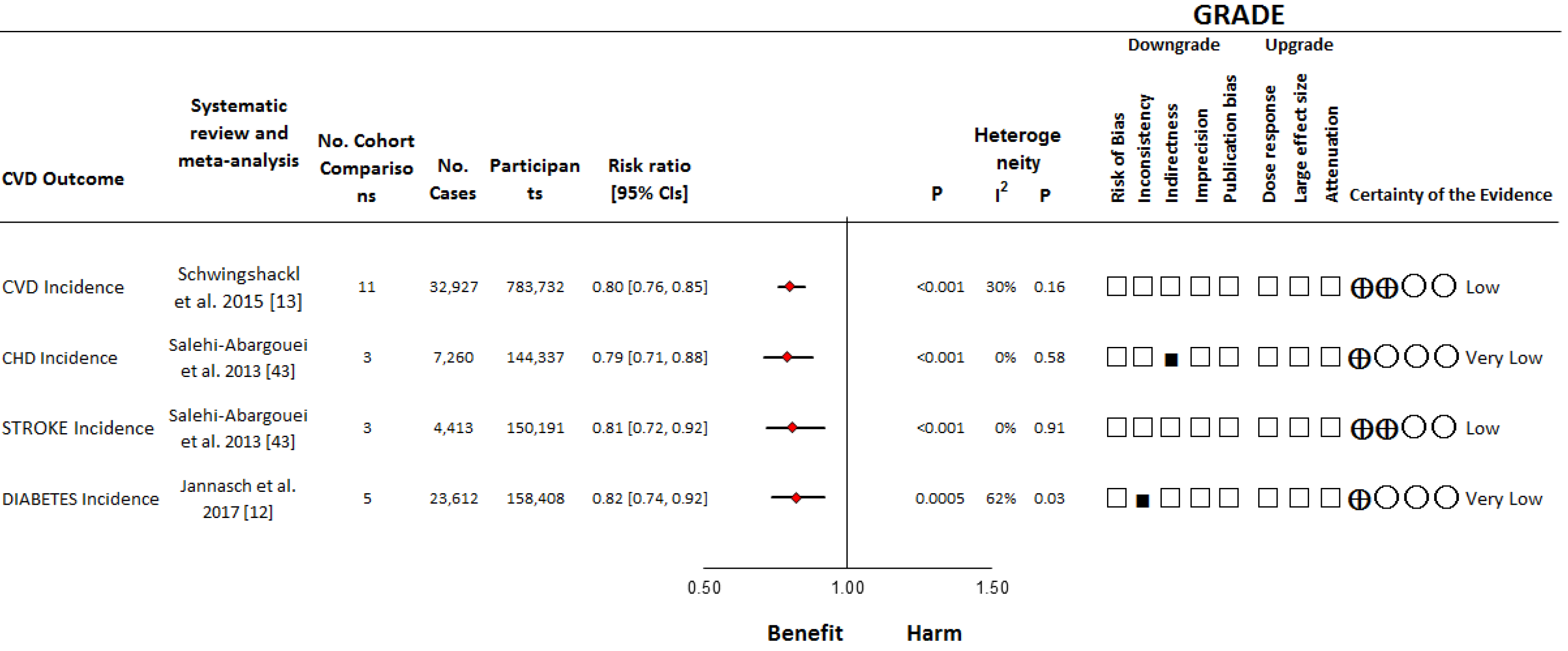
Cardiovascular Disease Incidence
Coronary Heart Disease Incidence
Stroke Incidence
Diabetes Incidence
3.2.2. Systematic Reviews and Meta-analyses of Controlled Trials
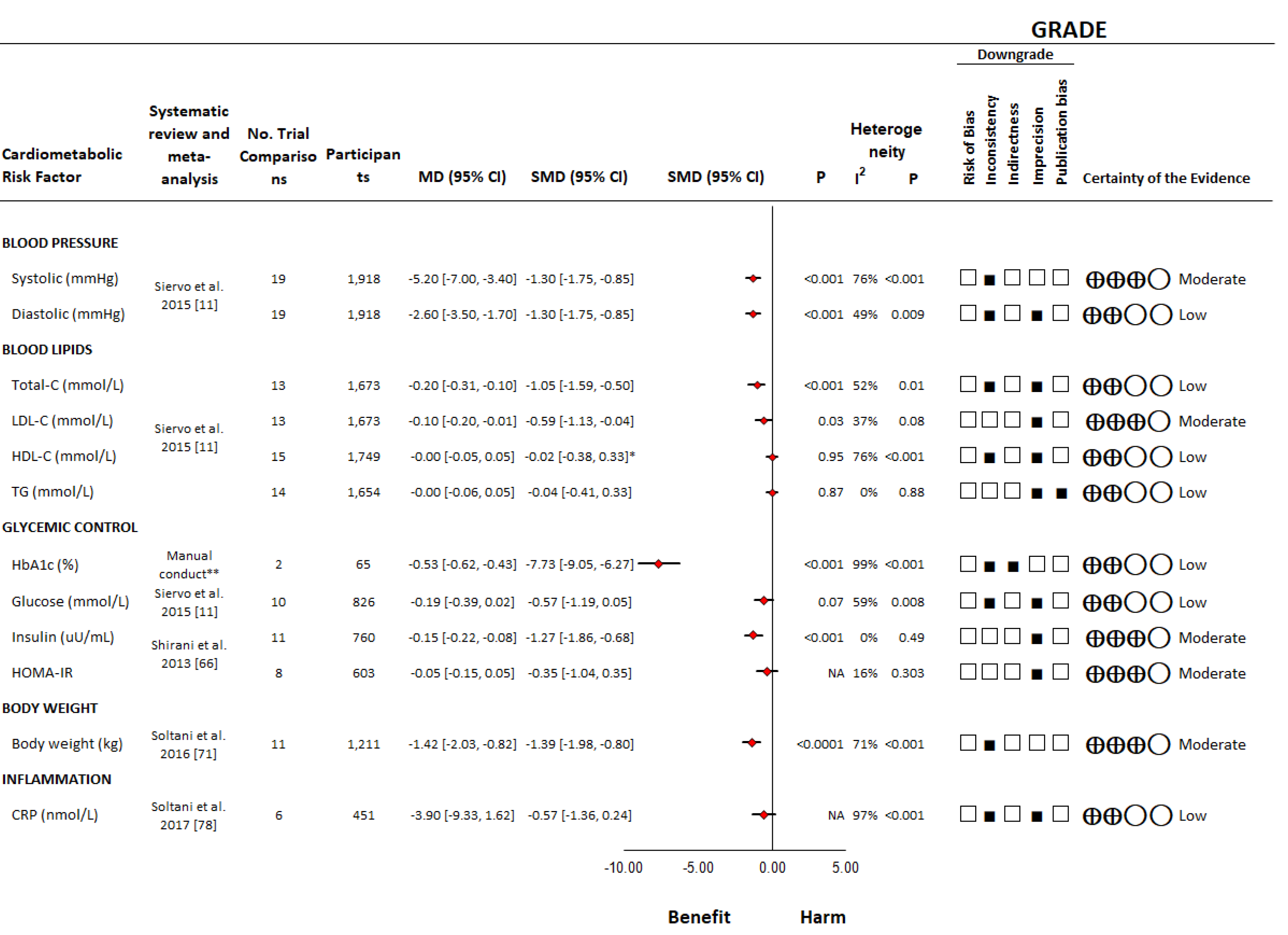
Blood Pressure
Blood Lipids
Glycemic Control
Body Weight
Inflammation
4. Discussion
4.1. Findings in the Context of the Literature
4.2. Strengths and limitations
4.3. Implications
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- International Diabetes Federation. IDF Diabetes Atlas, 7th ed. 2015. Available online: http://www.diabetesatlas.org/resources/2015-atlas.html (accessed on 30 January 2018).
- O’Rourke, K.; VanderZanden, A.; Shepard, D.; Leach-Kemon, K. Cardiovascular disease worldwide, 1990–2013. JAMA 2015, 314, 1905. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Obesity. Available online: http://www.who.int/gho/ncd/riskfactors/obesity_text/en/ (accessed on 30 January 2018).
- Chobanian, A.V.; Bakris, G.L.; Black, H.R.; Cushman, W.C.; Green, L.A.; Izzo, J.L., Jr.; Jones, D.W.; Materson, B.J.; Oparil, S.; Wright, J.T., Jr.; et al. The Seventh Report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure: The JNC 7 report. JAMA 2003, 289, 2560–2572. [Google Scholar] [CrossRef]
- American Heart Association Nutrition Committee; Lichtenstein, A.H.; Appel, L.J.; Brands, M.; Carnethon, M.; Daniels, S.; Franch, H.A.; Franklin, B.; Kris-Etherton, P.; Harris, W.S.; et al. Diet and lifestyle recommendations revision 2006: A scientific statement from the American Heart Association Nutrition Committee. Circulation 2006, 114, 82–96. [Google Scholar] [CrossRef] [PubMed]
- Anderson, T.J.; Gregoire, J.; Pearson, G.J.; Barry, A.R.; Couture, P.; Dawes, M.; Francis, G.A.; Genest, J., Jr.; Grover, S.; Gupta, M.; et al. 2016 Canadian Cardiovascular Society Guidelines for the Management of Dyslipidemia for the Prevention of Cardiovascular Disease in the Adult. Can. J. Cardiol. 2016, 32, 1263–1282. [Google Scholar] [CrossRef]
- Evert, A.B.; Boucher, J.L.; Cypress, M.; Dunbar, S.A.; Franz, M.J.; Mayer-Davis, E.J.; Neumiller, J.J.; Nwankwo, R.; Verdi, C.L.; Urbanski, P.; et al. Nutrition therapy recommendations for the management of adults with diabetes. Diabetes Care 2014, 37 (Suppl. 1), S120–S143. [Google Scholar] [CrossRef]
- Canadian Diabetes Association Clinical Practice Guidelines Expert Committee; Dworatzek, P.D.; Arcudi, K.; Gougeon, R.; Husein, N.; Sievenpiper, J.L.; Williams, S.L. Nutrition therapy. Can. J. Diabetes 2013, 37 (Suppl. 1), S45–S55. [Google Scholar] [CrossRef] [PubMed]
- Nerenberg, K.A.; Zarnke, K.B.; Leung, A.A.; Dasgupta, K.; Butalia, S.; McBrien, K.; Harris, K.C.; Nakhla, M.; Cloutier, L.; Gelfer, M.; et al. Hypertension Canada’s 2018 Guidelines for Diagnosis, Risk Assessment, Prevention, and Treatment of Hypertension in Adults and Children. Can. J. Cardiol. 2018, 34, 506–525. [Google Scholar] [CrossRef]
- Sacks, F.M.; Svetkey, L.P.; Vollmer, W.M.; Appel, L.J.; Bray, G.A.; Harsha, D.; Obarzanek, E.; Conlin, P.R.; Miller, E.R., 3rd; Simons-Morton, D.G.; et al. Effects on blood pressure of reduced dietary sodium and the Dietary Approaches to Stop Hypertension (DASH) diet. DASH-Sodium Collaborative Research Group. N. Engl. J. Med. 2001, 344, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Siervo, M.; Lara, J.; Chowdhury, S.; Ashor, A.; Oggioni, C.; Mathers, J.C. Effects of the Dietary Approach to Stop Hypertension (DASH) diet on cardiovascular risk factors: A systematic review and meta-analysis. Br. J. Nutr. 2015, 113, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Jannasch, F.; Kroger, J.; Schulze, M.B. Dietary Patterns and Type 2 Diabetes: A Systematic Literature Review and Meta-Analysis of Prospective Studies. J. Nutr. 2017, 147, 1174–1182. [Google Scholar] [CrossRef]
- Schwingshackl, L.; Hoffmann, G. Diet quality as assessed by the Healthy Eating Index, the Alternate Healthy Eating Index, the Dietary Approaches to Stop Hypertension score, and health outcomes: A systematic review and meta-analysis of cohort studies. J. Acad. Nutr. Diet. 2015, 115, 780–800 e785. [Google Scholar] [CrossRef] [PubMed]
- U.S. Department of Health and Human Services and U.S. Department of Agriculture. 2015–2020 Dietary Guidelines for Americans, 8th ed.; December 2015. Available online: http://health.gov/dietaryguidelines/2015/guidelines/ (accessed on 27 November 2018).
- Diabetes Canada Clinical Practice Guidelines Expert Committee; Sievenpiper, J.L.; Chan, C.B.; Dworatzek, P.D.; Freeze, C.; Williams, S.L. Nutrition Therapy. Can. J. Diabetes 2018, 42 (Suppl. 1), S64–S79. [Google Scholar] [CrossRef] [PubMed]
- Piepoli, M.F.; Hoes, A.W.; Agewall, S.; Albus, C.; Brotons, C.; Catapano, A.L.; Cooney, M.T.; Corra, U.; Cosyns, B.; Deaton, C.; et al. 2016 European Guidelines on cardiovascular disease prevention in clinical practice: The Sixth Joint Task Force of the European Society of Cardiology and Other Societies on Cardiovascular Disease Prevention in Clinical Practice (constituted by representatives of 10 societies and by invited experts)Developed with the special contribution of the European Association for Cardiovascular Prevention & Rehabilitation (EACPR). Eur. Heart J. 2016, 37, 2315–2381. [Google Scholar] [PubMed]
- Eckel, R.H.; Jakicic, J.M.; Ard, J.D.; de Jesus, J.M.; Houston Miller, N.; Hubbard, V.S.; Lee, I.M.; Lichtenstein, A.H.; Loria, C.M.; Millen, B.E.; et al. 2013 AHA/ACC guideline on lifestyle management to reduce cardiovascular risk: A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. J. Am. Coll. Cardiol. 2014, 63, 2960–2984. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.T.; Green, S. Cochrane Handbook for Systematic Reviews of Interventions version 5.1.0 [updated March 2011]. Cochrane Collaboration. Available online: https://handbook-5-1.cochrane.org/ (accessed on 27 November 2018).
- Guyatt, G.; Oxman, A.D.; Akl, E.A.; Kunz, R.; Vist, G.; Brozek, J.; Norris, S.; Falck-Ytter, Y.; Glasziou, P.; DeBeer, H.; et al. GRADE guidelines: 1. Introduction-GRADE evidence profiles and summary of findings tables. J. Clin. Epidemiol. 2011, 64, 383–394. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; Group, P. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. Int. J. Surg. 2010, 8, 336–341. [Google Scholar] [CrossRef] [PubMed]
- Wells, G.A.; Shea, B.; O’Connell, D.; Peterson, J.; Welch, V.; Losos, M.; Tugwell, P. The Newcastle–Ottawa Scale (NOS) for Assessing the Quality of Nonrandomised Studies in Meta-Analyses; Ottawa Hospital Research Institute: Ottawa, Canada, 2014; Available online: www.ohri.ca/programs/clinical_epidemiology/oxford.asp (accessed on 30 January 2018).
- Higgins, J.P.; Altman, D.G.; Gotzsche, P.C.; Juni, P.; Moher, D.; Oxman, A.D.; Savovic, J.; Schulz, K.F.; Weeks, L.; Sterne, J.A.; et al. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 2011, 343, d5928. [Google Scholar] [CrossRef]
- Balshem, H.; Helfand, M.; Schunemann, H.J.; Oxman, A.D.; Kunz, R.; Brozek, J.; Vist, G.E.; Falck-Ytter, Y.; Meerpohl, J.; Norris, S.; et al. GRADE guidelines: 3. Rating the quality of evidence. J. Clin. Epidemiol. 2011, 64, 401–406. [Google Scholar] [CrossRef]
- Brunetti, M.; Shemilt, I.; Pregno, S.; Vale, L.; Oxman, A.D.; Lord, J.; Sisk, J.; Ruiz, F.; Hill, S.; Guyatt, G.H.; et al. GRADE guidelines: 10. Considering resource use and rating the quality of economic evidence. J. Clin. Epidemiol. 2013, 66, 140–150. [Google Scholar] [CrossRef]
- Guyatt, G.; Oxman, A.D.; Sultan, S.; Brozek, J.; Glasziou, P.; Alonso-Coello, P.; Atkins, D.; Kunz, R.; Montori, V.; Jaeschke, R.; et al. GRADE guidelines: 11. Making an overall rating of confidence in effect estimates for a single outcome and for all outcomes. J. Clin. Epidemiol. 2013, 66, 151–157. [Google Scholar] [CrossRef]
- Guyatt, G.H.; Oxman, A.D.; Kunz, R.; Atkins, D.; Brozek, J.; Vist, G.; Alderson, P.; Glasziou, P.; Falck-Ytter, Y.; Schunemann, H.J. GRADE guidelines: 2. Framing the question and deciding on important outcomes. J. Clin. Epidemiol. 2011, 64, 395–400. [Google Scholar] [CrossRef] [PubMed]
- Guyatt, G.H.; Oxman, A.D.; Kunz, R.; Brozek, J.; Alonso-Coello, P.; Rind, D.; Devereaux, P.J.; Montori, V.M.; Freyschuss, B.; Vist, G.; et al. GRADE guidelines 6. Rating the quality of evidence--imprecision. J. Clin. Epidemiol. 2011, 64, 1283–1293. [Google Scholar] [CrossRef] [PubMed]
- Guyatt, G.H.; Oxman, A.D.; Kunz, R.; Woodcock, J.; Brozek, J.; Helfand, M.; Alonso-Coello, P.; Falck-Ytter, Y.; Jaeschke, R.; Vist, G.; et al. GRADE guidelines: 8. Rating the quality of evidence--indirectness. J. Clin. Epidemiol. 2011, 64, 1303–1310. [Google Scholar] [CrossRef] [PubMed]
- Guyatt, G.H.; Oxman, A.D.; Kunz, R.; Woodcock, J.; Brozek, J.; Helfand, M.; Alonso-Coello, P.; Glasziou, P.; Jaeschke, R.; Akl, E.A.; et al. GRADE guidelines: 7. Rating the quality of evidence--inconsistency. J. Clin. Epidemiol. 2011, 64, 1294–1302. [Google Scholar] [CrossRef] [PubMed]
- Guyatt, G.H.; Oxman, A.D.; Montori, V.; Vist, G.; Kunz, R.; Brozek, J.; Alonso-Coello, P.; Djulbegovic, B.; Atkins, D.; Falck-Ytter, Y.; et al. GRADE guidelines: 5. Rating the quality of evidence—Publication bias. J. Clin. Epidemiol. 2011, 64, 1277–1282. [Google Scholar] [CrossRef] [PubMed]
- Guyatt, G.H.; Oxman, A.D.; Santesso, N.; Helfand, M.; Vist, G.; Kunz, R.; Brozek, J.; Norris, S.; Meerpohl, J.; Djulbegovic, B.; et al. GRADE guidelines: 12. Preparing summary of findings tables-binary outcomes. J. Clin. Epidemiol. 2013, 66, 158–172. [Google Scholar] [CrossRef] [PubMed]
- Guyatt, G.H.; Oxman, A.D.; Sultan, S.; Glasziou, P.; Akl, E.A.; Alonso-Coello, P.; Atkins, D.; Kunz, R.; Brozek, J.; Montori, V.; et al. GRADE guidelines: 9. Rating up the quality of evidence. J. Clin. Epidemiol. 2011, 64, 1311–1316. [Google Scholar] [CrossRef]
- Guyatt, G.H.; Oxman, A.D.; Vist, G.; Kunz, R.; Brozek, J.; Alonso-Coello, P.; Montori, V.; Akl, E.A.; Djulbegovic, B.; Falck-Ytter, Y.; et al. GRADE guidelines: 4. Rating the quality of evidence—Study limitations (risk of bias). J. Clin. Epidemiol. 2011, 64, 407–415. [Google Scholar] [CrossRef]
- Guyatt, G.H.; Thorlund, K.; Oxman, A.D.; Walter, S.D.; Patrick, D.; Furukawa, T.A.; Johnston, B.C.; Karanicolas, P.; Akl, E.A.; Vist, G.; et al. GRADE guidelines: 13. Preparing summary of findings tables and evidence profiles-continuous outcomes. J. Clin. Epidemiol. 2013, 66, 173–183. [Google Scholar] [CrossRef]
- Agnoli, C.; Krogh, V.; Grioni, S.; Sieri, S.; Palli, D.; Masala, G.; Sacerdote, C.; Vineis, P.; Tumino, R.; Frasca, G.; et al. A priori-defined dietary patterns are associated with reduced risk of stroke in a large Italian cohort. J. Nutr. 2011, 141, 1552–1558. [Google Scholar] [CrossRef]
- Bertoia, M.L.; Triche, E.W.; Michaud, D.S.; Baylin, A.; Hogan, J.W.; Neuhouser, M.L.; Tinker, L.F.; Van Horn, L.; Waring, M.E.; Li, W.; et al. Mediterranean and Dietary Approaches to Stop Hypertension dietary patterns and risk of sudden cardiac death in postmenopausal women. Am. J. Clin. Nutr. 2014, 99, 344–351. [Google Scholar] [CrossRef] [PubMed]
- Fitzgerald, K.C.; Chiuve, S.E.; Buring, J.E.; Ridker, P.M.; Glynn, R.J. Comparison of associations of adherence to a Dietary Approaches to Stop Hypertension (DASH)-style diet with risks of cardiovascular disease and venous thromboembolism. J. Thromb. Haemost. 2012, 10, 189–198. [Google Scholar] [CrossRef] [PubMed]
- Folsom, A.R.; Parker, E.D.; Harnack, L.J. Degree of concordance with DASH diet guidelines and incidence of hypertension and fatal cardiovascular disease. Am. J. Hypertens. 2007, 20, 225–232. [Google Scholar] [CrossRef] [PubMed]
- Fung, T.T.; Chiuve, S.E.; McCullough, M.L.; Rexrode, K.M.; Logroscino, G.; Hu, F.B. Adherence to a DASH-style diet and risk of coronary heart disease and stroke in women. Arch. Intern. Med. 2008, 168, 713–720. [Google Scholar] [CrossRef] [PubMed]
- Levitan, E.B.; Wolk, A.; Mittleman, M.A. Consistency with the DASH diet and incidence of heart failure. Arch. Intern. Med. 2009, 169, 851–857. [Google Scholar] [CrossRef] [PubMed]
- Lin, P.H.; Yeh, W.T.; Svetkey, L.P.; Chuang, S.Y.; Chang, Y.C.; Wang, C.; Pan, W.H. Dietary intakes consistent with the DASH dietary pattern reduce blood pressure increase with age and risk for stroke in a Chinese population. Asia Pac. J. Clin. Nutr. 2013, 22, 482–491. [Google Scholar] [PubMed]
- Reedy, J.; Krebs-Smith, S.M.; Miller, P.E.; Liese, A.D.; Kahle, L.L.; Park, Y.; Subar, A.F. Higher diet quality is associated with decreased risk of all-cause, cardiovascular disease, and cancer mortality among older adults. J. Nutr. 2014, 144, 881–889. [Google Scholar] [CrossRef]
- Salehi-Abargouei, A.; Maghsoudi, Z.; Shirani, F.; Azadbakht, L. Effects of Dietary Approaches to Stop Hypertension (DASH)-style diet on fatal or nonfatal cardiovascular diseases—incidence: A systematic review and meta-analysis on observational prospective studies. Nutrition 2013, 29, 611–618. [Google Scholar] [CrossRef]
- de Koning, L.; Chiuve, S.E.; Fung, T.T.; Willett, W.C.; Rimm, E.B.; Hu, F.B. Diet-quality scores and the risk of type 2 diabetes in men. Diabetes Care 2011, 34, 1150–1156. [Google Scholar] [CrossRef]
- Otto, M.C.; Padhye, N.S.; Bertoni, A.G.; Jacobs, D.R., Jr.; Mozaffarian, D. Everything in Moderation—Dietary Diversity and Quality, Central Obesity and Risk of Diabetes. PLoS ONE 2015, 10, e0141341. [Google Scholar]
- Jacobs, S.; Harmon, B.E.; Boushey, C.J.; Morimoto, Y.; Wilkens, L.R.; Le Marchand, L.; Kroger, J.; Schulze, M.B.; Kolonel, L.N.; Maskarinec, G. A priori-defined diet quality indexes and risk of type 2 diabetes: The Multiethnic Cohort. Diabetologia 2015, 58, 98–112. [Google Scholar] [CrossRef] [PubMed]
- InterAct, C. Adherence to predefined dietary patterns and incident type 2 diabetes in European populations: EPIC-InterAct Study. Diabetologia 2014, 57, 321–333. [Google Scholar]
- Liese, A.D.; Nichols, M.; Sun, X.; D’Agostino, R.B., Jr.; Haffner, S.M. Adherence to the DASH Diet is inversely associated with incidence of type 2 diabetes: The insulin resistance atherosclerosis study. Diabetes Care 2009, 32, 1434–1436. [Google Scholar] [CrossRef] [PubMed]
- Appel, L.J.; Moore, T.J.; Obarzanek, E.; Vollmer, W.M.; Svetkey, L.P.; Sacks, F.M.; Bray, G.A.; Vogt, T.M.; Cutler, J.A.; Windhauser, M.M.; et al. A clinical trial of the effects of dietary patterns on blood pressure. DASH Collaborative Research Group. N. Engl. J. Med. 1997, 336, 1117–1124. [Google Scholar] [CrossRef] [PubMed]
- Appel, L.J.; Champagne, C.M.; Harsha, D.W.; Cooper, L.S.; Obarzanek, E.; Elmer, P.J.; Stevens, V.J.; Vollmer, W.M.; Lin, P.H.; Svetkey, L.P.; et al. Effects of comprehensive lifestyle modification on blood pressure control: Main results of the PREMIER clinical trial. JAMA 2003, 289, 2083–2093. [Google Scholar] [PubMed]
- Conlin, P.R.; Erlinger, T.P.; Bohannon, A.; Miller, E.R., 3rd; Appel, L.J.; Svetkey, L.P.; Moore, T.J. The DASH diet enhances the blood pressure response to losartan in hypertensive patients. Am. J. Hypertens. 2003, 16, 337–342. [Google Scholar] [CrossRef]
- Lopes, H.F.; Martin, K.L.; Nashar, K.; Morrow, J.D.; Goodfriend, T.L.; Egan, B.M. DASH diet lowers blood pressure and lipid-induced oxidative stress in obesity. Hypertension 2003, 41, 422–430. [Google Scholar] [CrossRef] [PubMed]
- Nowson, C.A.; Wattanapenpaiboon, N.; Pachett, A. Low-sodium Dietary Approaches to Stop Hypertension-type diet including lean red meat lowers blood pressure in postmenopausal women. Nutr. Res. 2009, 29, 8–18. [Google Scholar] [CrossRef]
- Nowson, C.A.; Worsley, A.; Margerison, C.; Jorna, M.K.; Frame, A.G.; Torres, S.J.; Godfrey, S.J. Blood pressure response to dietary modifications in free-living individuals. J. Nutr. 2004, 134, 2322–2329. [Google Scholar] [CrossRef]
- Nowson, C.A.; Worsley, A.; Margerison, C.; Jorna, M.K.; Godfrey, S.J.; Booth, A. Blood pressure change with weight loss is affected by diet type in men. Am. J. Clin. Nutr. 2005, 81, 983–989. [Google Scholar] [CrossRef]
- Azadbakht, L.; Mirmiran, P.; Esmaillzadeh, A.; Azizi, T.; Azizi, F. Beneficial effects of a Dietary Approaches to Stop Hypertension eating plan on features of the metabolic syndrome. Diabetes Care 2005, 28, 2823–2831. [Google Scholar] [CrossRef] [PubMed]
- Azadbakht, L.; Surkan, P.J.; Esmaillzadeh, A.; Willett, W.C. The Dietary Approaches to Stop Hypertension eating plan affects C-reactive protein, coagulation abnormalities, and hepatic function tests among type 2 diabetic patients. J. Nutr. 2011, 141, 1083–1088. [Google Scholar] [CrossRef] [PubMed]
- Al-Solaiman, Y.; Jesri, A.; Mountford, W.K.; Lackland, D.T.; Zhao, Y.; Egan, B.M. DASH lowers blood pressure in obese hypertensives beyond potassium, magnesium and fibre. J. Hum. Hypertens. 2010, 24, 237–246. [Google Scholar] [CrossRef] [PubMed]
- Blumenthal, J.A.; Babyak, M.A.; Hinderliter, A.; Watkins, L.L.; Craighead, L.; Lin, P.H.; Caccia, C.; Johnson, J.; Waugh, R.; Sherwood, A. Effects of the DASH diet alone and in combination with exercise and weight loss on blood pressure and cardiovascular biomarkers in men and women with high blood pressure: The ENCORE study. Arch. Intern. Med. 2010, 170, 126–135. [Google Scholar] [CrossRef] [PubMed]
- Malloy-McFall, J.; Barkley, J.E.; Gordon, K.L.; Burzminski, N.; Glickman, E.L. Effect of the DASH Diet on Pre- and Stage 1 Hypertensive Individuals in a Free-Living Environment. Nutr. Metab. Insights 2010, 3, 15–23. [Google Scholar] [CrossRef] [PubMed]
- Edwards, K.M.; Wilson, K.L.; Sadja, J.; Ziegler, M.G.; Mills, P.J. Effects on blood pressure and autonomic nervous system function of a 12-week exercise or exercise plus DASH-diet intervention in individuals with elevated blood pressure. Acta Physiol. (Oxf.) 2011, 203, 343–350. [Google Scholar] [CrossRef] [PubMed]
- Lin, P.H.; Allen, J.D.; Li, Y.J.; Yu, M.; Lien, L.F.; Svetkey, L.P. Blood Pressure-Lowering Mechanisms of the DASH Dietary Pattern. J. Nutr. Metab. 2012, 2012, 472396. [Google Scholar] [CrossRef]
- Asemi, Z.; Tabassi, Z.; Samimi, M.; Fahiminejad, T.; Esmaillzadeh, A. Favourable effects of the Dietary Approaches to Stop Hypertension diet on glucose tolerance and lipid profiles in gestational diabetes: A randomised clinical trial. Br. J. Nutr. 2013, 109, 2024–2030. [Google Scholar] [CrossRef]
- Shirani, F.; Salehi-Abargouei, A.; Azadbakht, L. Effects of Dietary Approaches to Stop Hypertension (DASH) diet on some risk for developing type 2 diabetes: A systematic review and meta-analysis on controlled clinical trials. Nutrition 2013, 29, 939–947. [Google Scholar] [CrossRef]
- Soltani, S.; Shirani, F.; Chitsazi, M.J.; Salehi-Abargouei, A. The effect of dietary approaches to stop hypertension (DASH) diet on weight and body composition in adults: A systematic review and meta-analysis of randomized controlled clinical trials. Obes. Rev. 2016, 17, 442–454. [Google Scholar] [CrossRef]
- Soltani, S.; Chitsazi, M.J.; Salehi-Abargouei, A. The effect of dietary approaches to stop hypertension (DASH) on serum inflammatory markers: A systematic review and meta-analysis of randomized trials. Clin. Nutr. 2018, 37, 542–550. [Google Scholar] [CrossRef] [PubMed]
- Harsha, D.W.; Sacks, F.M.; Obarzanek, E.; Svetkey, L.P.; Lin, P.H.; Bray, G.A.; Aickin, M.; Conlin, P.R.; Miller, E.R., 3rd; Appel, L.J. Effect of dietary sodium intake on blood lipids: Results from the DASH-sodium trial. Hypertension 2004, 43, 393–398. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.T.; Maruthur, N.M.; Appel, L.J. The effect of dietary patterns on estimated coronary heart disease risk: Results from the Dietary Approaches to Stop Hypertension (DASH) trial. Circ. Cardiovasc. Qual. Outcomes 2010, 3, 484–489. [Google Scholar] [CrossRef] [PubMed]
- Ard, J.D.; Grambow, S.C.; Liu, D.; Slentz, C.A.; Kraus, W.E.; Svetkey, L.P.; study, P. The effect of the PREMIER interventions on insulin sensitivity. Diabetes Care 2004, 27, 340–347. [Google Scholar] [CrossRef] [PubMed]
- Lien, L.F.; Brown, A.J.; Ard, J.D.; Loria, C.; Erlinger, T.P.; Feldstein, A.C.; Lin, P.H.; Champagne, C.M.; King, A.C.; McGuire, H.L.; et al. Effects of PREMIER lifestyle modifications on participants with and without the metabolic syndrome. Hypertension 2007, 50, 609–616. [Google Scholar] [CrossRef] [PubMed]
- Al-Solaiman, Y.; Jesri, A.; Zhao, Y.; Morrow, J.D.; Egan, B.M. Low-Sodium DASH reduces oxidative stress and improves vascular function in salt-sensitive humans. J. Hum. Hypertens. 2009, 23, 826–835. [Google Scholar] [CrossRef] [PubMed]
- Hodson, L.; Harnden, K.E.; Roberts, R.; Dennis, A.L.; Frayn, K.N. Does the DASH diet lower blood pressure by altering peripheral vascular function? J. Hum. Hypertens. 2010, 24, 312–319. [Google Scholar] [CrossRef]
- Ard, J.D.; Coffman, C.J.; Lin, P.H.; Svetkey, L.P. One-year follow-up study of blood pressure and dietary patterns in dietary approaches to stop hypertension (DASH)-sodium participants. Am. J. Hypertens. 2004, 17, 1156–1162. [Google Scholar] [CrossRef] [PubMed]
- Elmer, P.J.; Obarzanek, E.; Vollmer, W.M.; Simons-Morton, D.; Stevens, V.J.; Young, D.R.; Lin, P.H.; Champagne, C.; Harsha, D.W.; Svetkey, L.P.; et al. Effects of comprehensive lifestyle modification on diet, weight, physical fitness, and blood pressure control: 18-month results of a randomized trial. Ann. Intern. Med. 2006, 144, 485–495. [Google Scholar] [CrossRef] [PubMed]
- Lima, S.T.; da Silva Nalin de Souza, B.; Franca, A.K.; Salgado Filho, N.; Sichieri, R. Dietary approach to hypertension based on low glycaemic index and principles of DASH (Dietary Approaches to Stop Hypertension): A randomised trial in a primary care service. Br. J. Nutr. 2013, 110, 1472–1479. [Google Scholar] [CrossRef]
- Rifai, L.; Pisano, C.; Hayden, J.; Sulo, S.; Silver, M.A. Impact of the DASH diet on endothelial function, exercise capacity, and quality of life in patients with heart failure. Bayl. Univ. Med. Cent. Proc. 2015, 28, 151–156. [Google Scholar] [CrossRef]
- Asemi, Z.; Samimi, M.; Tabassi, Z.; Esmaillzadeh, A. The effect of DASH diet on pregnancy outcomes in gestational diabetes: A randomized controlled clinical trial. Eur. J. Clin. Nutr. 2014, 68, 490–495. [Google Scholar] [CrossRef] [PubMed]
- Razavi Zade, M.; Telkabadi, M.H.; Bahmani, F.; Salehi, B.; Farshbaf, S.; Asemi, Z. The effects of DASH diet on weight loss and metabolic status in adults with non-alcoholic fatty liver disease: A randomized clinical trial. Liver Int. 2016, 36, 563–571. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, D.J.; Jones, P.J.; Frohlich, J.; Lamarche, B.; Ireland, C.; Nishi, S.K.; Srichaikul, K.; Galange, P.; Pellini, C.; Faulkner, D.; et al. The effect of a dietary portfolio compared to a DASH-type diet on blood pressure. Nutr. Metab. Cardiovasc. Dis. 2015, 25, 1132–1139. [Google Scholar] [CrossRef] [PubMed]
- King, D.E.; Egan, B.M.; Woolson, R.F.; Mainous, A.G., 3rd; Al-Solaiman, Y.; Jesri, A. Effect of a high-fiber diet vs a fiber-supplemented diet on C-reactive protein level. Arch. Intern. Med. 2007, 167, 502–506. [Google Scholar] [CrossRef] [PubMed]
- Roussell, M.A.; Hill, A.M.; Gaugler, T.L.; West, S.G.; Heuvel, J.P.; Alaupovic, P.; Gillies, P.J.; Kris-Etherton, P.M. Beef in an Optimal Lean Diet study: Effects on lipids, lipoproteins, and apolipoproteins. Am. J. Clin. Nutr. 2012, 95, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Ouyang, Y.; Liu, J.; Zhu, M.; Zhao, G.; Bao, W.; Hu, F.B. Fruit and vegetable consumption and mortality from all causes, cardiovascular disease, and cancer: Systematic review and dose-response meta-analysis of prospective cohort studies. BMJ 2014, 349, g4490. [Google Scholar] [CrossRef]
- Tang, G.; Wang, D.; Long, J.; Yang, F.; Si, L. Meta-analysis of the association between whole grain intake and coronary heart disease risk. Am. J. Cardiol. 2015, 115, 625–629. [Google Scholar] [CrossRef]
- Reynolds, A.N. Associations of fats and carbohydrates with cardiovascular disease and mortality-PURE and simple? Lancet 2018, 391, 1676. [Google Scholar] [CrossRef]
- Viguiliouk, E.; Blanco Mejia, S.; Kendall, C.W.; Sievenpiper, J.L. Can pulses play a role in improving cardiometabolic health? Evidence from systematic reviews and meta-analyses. Ann. N. Y. Acad. Sci. 2017, 1392, 43–57. [Google Scholar] [CrossRef]
- Aune, D.; Keum, N.; Giovannucci, E.; Fadnes, L.T.; Boffetta, P.; Greenwood, D.C.; Tonstad, S.; Vatten, L.J.; Riboli, E.; Norat, T. Nut consumption and risk of cardiovascular disease, total cancer, all-cause and cause-specific mortality: A systematic review and dose-response meta-analysis of prospective studies. BMC Med. 2016, 14, 207. [Google Scholar] [CrossRef] [PubMed]
- Abete, I.; Romaguera, D.; Vieira, A.R.; Lopez de Munain, A.; Norat, T. Association between total, processed, red and white meat consumption and all-cause, CVD and IHD mortality: A meta-analysis of cohort studies. Br. J. Nutr. 2014, 112, 762–775. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Lin, X.; Ouyang, Y.Y.; Liu, J.; Zhao, G.; Pan, A.; Hu, F.B. Red and processed meat consumption and mortality: Dose-response meta-analysis of prospective cohort studies. Public Health Nutr. 2016, 19, 893–905. [Google Scholar] [CrossRef] [PubMed]
- Threapleton, D.E.; Greenwood, D.C.; Evans, C.E.; Cleghorn, C.L.; Nykjaer, C.; Woodhead, C.; Cade, J.E.; Gale, C.P.; Burley, V.J. Dietary fibre intake and risk of cardiovascular disease: Systematic review and meta-analysis. BMJ 2013, 347, f6879. [Google Scholar] [CrossRef] [PubMed]
- Arts, I.C.; Hollman, P.C. Polyphenols and disease risk in epidemiologic studies. Am. J. Clin. Nutr. 2005, 81, 317S–325S. [Google Scholar] [CrossRef] [PubMed]
- Perez-Cano, F.J.; Castell, M. Flavonoids, Inflammation and Immune System. Nutrients 2016, 8, 659. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.S.; Vos, T.; Flaxman, A.D.; Danaei, G.; Shibuya, K.; Adair-Rohani, H.; Amann, M.; Anderson, H.R.; Andrews, K.G.; Aryee, M.; et al. A comparative risk assessment of burden of disease and injury attributable to 67 risk factors and risk factor clusters in 21 regions, 1990–2010: A systematic analysis for the Global Burden of Disease Study 2010. Lancet 2012, 380, 2224–2260. [Google Scholar] [CrossRef]
- Lewington, S.; Clarke, R.; Qizilbash, N.; Peto, R.; Collins, R.; Prospective Studies, Collaboration. Age-specific relevance of usual blood pressure to vascular mortality: A meta-analysis of individual data for one million adults in 61 prospective studies. Lancet 2002, 360, 1903–1913. [Google Scholar]
- Cholesterol Treatment Trialists Collaboration; Baigent, C.; Blackwell, L.; Emberson, J.; Holland, L.E.; Reith, C.; Bhala, N.; Peto, R.; Barnes, E.H.; Keech, A.; et al. Efficacy and safety of more intensive lowering of LDL cholesterol: A meta-analysis of data from 170,000 participants in 26 randomised trials. Lancet 2010, 376, 1670–1681. [Google Scholar]
- Cholesterol Treatment Trialists Collaboration; Fulcher, J.; O’Connell, R.; Voysey, M.; Emberson, J.; Blackwell, L.; Mihaylova, B.; Simes, J.; Collins, R.; Kirby, A.; et al. Efficacy and safety of LDL-lowering therapy among men and women: Meta-analysis of individual data from 174,000 participants in 27 randomised trials. Lancet 2015, 385, 1397–1405. [Google Scholar]
- Cannon, C.P.; Blazing, M.A.; Giugliano, R.P.; McCagg, A.; White, J.A.; Theroux, P.; Darius, H.; Lewis, B.S.; Ophuis, T.O.; Jukema, J.W.; et al. Ezetimibe Added to Statin Therapy after Acute Coronary Syndromes. N. Engl. J. Med. 2015, 372, 2387–2397. [Google Scholar] [CrossRef] [PubMed]
- Johnston, B.C.; Kanters, S.; Bandayrel, K.; Wu, P.; Naji, F.; Siemieniuk, R.A.; Ball, G.D.; Busse, J.W.; Thorlund, K.; Guyatt, G.; et al. Comparison of weight loss among named diet programs in overweight and obese adults: A meta-analysis. JAMA 2014, 312, 923–933. [Google Scholar] [CrossRef] [PubMed]
- Haslam, D.W.; James, W.P. Obesity. Lancet 2005, 366, 1197–1209. [Google Scholar] [CrossRef]
- Sotos-Prieto, M.; Bhupathiraju, S.N.; Mattei, J.; Fung, T.T.; Li, Y.; Pan, A.; Willett, W.C.; Rimm, E.B.; Hu, F.B. Association of Changes in Diet Quality with Total and Cause-Specific Mortality. N. Engl. J. Med. 2017, 377, 143–153. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Fan, Y.; Zhang, X.; Hou, W.; Tang, Z. Fruit and vegetable intake and risk of type 2 diabetes mellitus: Meta-analysis of prospective cohort studies. BMJ Open 2014, 4, e005497. [Google Scholar] [CrossRef] [PubMed]
- Gijsbers, L.; Ding, E.L.; Malik, V.S.; de Goede, J.; Geleijnse, J.M.; Soedamah-Muthu, S.S. Consumption of dairy foods and diabetes incidence: A dose-response meta-analysis of observational studies. Am. J. Clin. Nutr. 2016, 103, 1111–1124. [Google Scholar] [CrossRef] [PubMed]
- Wei, G.S.; Coady, S.A.; Goff, D.C., Jr.; Brancati, F.L.; Levy, D.; Selvin, E.; Vasan, R.S.; Fox, C.S. Blood pressure and the risk of developing diabetes in african americans and whites: ARIC, CARDIA, and the framingham heart study. Diabetes Care 2011, 34, 873–879. [Google Scholar] [CrossRef]
- Emdin, C.A.; Anderson, S.G.; Woodward, M.; Rahimi, K. Usual Blood Pressure and Risk of New-Onset Diabetes: Evidence From 4.1 Million Adults and a Meta-Analysis of Prospective Studies. J. Am. Coll. Cardiol. 2015, 66, 1552–1562. [Google Scholar] [CrossRef]
- Marott, S.C.; Nordestgaard, B.G.; Tybjaerg-Hansen, A.; Benn, M. Components of the Metabolic Syndrome and Risk of Type 2 Diabetes. J. Clin. Endocrinol. Metab. 2016, 101, 3212–3221. [Google Scholar] [CrossRef]
- Post, R.E.; Mainous, A.G., 3rd; King, D.E.; Simpson, K.N. Dietary fiber for the treatment of type 2 diabetes mellitus: A meta-analysis. J. Am. Board Fam. Med. 2012, 25, 16–23. [Google Scholar] [CrossRef]
- Evans, C.E.; Greenwood, D.C.; Threapleton, D.E.; Cleghorn, C.L.; Nykjaer, C.; Woodhead, C.E.; Gale, C.P.; Burley, V.J. Effects of dietary fibre type on blood pressure: A systematic review and meta-analysis of randomized controlled trials of healthy individuals. J. Hypertens. 2015, 33, 897–911. [Google Scholar] [CrossRef] [PubMed]
- Hou, Q.; Li, Y.; Li, L.; Cheng, G.; Sun, X.; Li, S.; Tian, H. The Metabolic Effects of Oats Intake in Patients with Type 2 Diabetes: A Systematic Review and Meta-Analysis. Nutrients 2015, 7, 10369–10387. [Google Scholar] [CrossRef] [PubMed]
- Sievenpiper, J.L.; Kendall, C.W.; Esfahani, A.; Wong, J.M.; Carleton, A.J.; Jiang, H.Y.; Bazinet, R.P.; Vidgen, E.; Jenkins, D.J. Effect of non-oil-seed pulses on glycaemic control: A systematic review and meta-analysis of randomised controlled experimental trials in people with and without diabetes. Diabetologia 2009, 52, 1479–1495. [Google Scholar] [CrossRef] [PubMed]
- Choo, V.L.; Viguiliouk, E.; Blanco Mejia, S.; Cozma, A.I.; Khan, T.A.; Ha, V.; Wolever, T.M.S.; Leiter, L.A.; Vuksan, V.; Kendall, C.W.C.; et al. Food sources of fructose-containing sugars and glycaemic control: Systematic review and meta-analysis of controlled intervention studies. BMJ 2018, 363, k4644. [Google Scholar] [CrossRef] [PubMed]
- InterAct Consortium. Dietary fibre and incidence of type 2 diabetes in eight European countries: The EPIC-InterAct Study and a meta-analysis of prospective studies. Diabetologia 2015, 58, 1394–1408. [Google Scholar] [CrossRef] [PubMed]
- Aune, D.; Norat, T.; Romundstad, P.; Vatten, L.J. Whole grain and refined grain consumption and the risk of type 2 diabetes: A systematic review and dose-response meta-analysis of cohort studies. Eur. J. Epidemiol. 2013, 28, 845–858. [Google Scholar] [CrossRef] [PubMed]
- Kotchen, T.A.; Kotchen, J.M. Dietary sodium and blood pressure: Interactions with other nutrients. Am. J. Clin. Nutr. 1997, 65, 708S–711S. [Google Scholar] [CrossRef]
- Whelton, P.K.; He, J.; Cutler, J.A.; Brancati, F.L.; Appel, L.J.; Follmann, D.; Klag, M.J. Effects of oral potassium on blood pressure. Meta-analysis of randomized controlled clinical trials. JAMA 1997, 277, 1624–1632. [Google Scholar] [CrossRef]
- Petrov, V.; Lijnen, P. Modification of intracellular calcium and plasma renin by dietary calcium in men. Am. J. Hypertens. 1999, 12, 1217–1224. [Google Scholar] [CrossRef]
- Kanbay, M.; Bayram, Y.; Solak, Y.; Sanders, P.W. Dietary potassium: A key mediator of the cardiovascular response to dietary sodium chloride. J. Am. Soc. Hypertens. 2013, 7, 395–400. [Google Scholar] [CrossRef]
- Haddy, F.J.; Vanhoutte, P.M.; Feletou, M. Role of potassium in regulating blood flow and blood pressure. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2006, 290, R546–R552. [Google Scholar] [CrossRef] [PubMed]
- Zacchia, M.; Abategiovanni, M.L.; Stratigis, S.; Capasso, G. Potassium: From Physiology to Clinical Implications. Kidney Dis. (Basel) 2016, 2, 72–79. [Google Scholar] [CrossRef] [PubMed]
- Siervo, M.; Lara, J.; Ogbonmwan, I.; Mathers, J.C. Inorganic nitrate and beetroot juice supplementation reduces blood pressure in adults: A systematic review and meta-analysis. J. Nutr. 2013, 143, 818–826. [Google Scholar] [CrossRef] [PubMed]
- Jayalath, V.H.; de Souza, R.J.; Sievenpiper, J.L.; Ha, V.; Chiavaroli, L.; Mirrahimi, A.; Di Buono, M.; Bernstein, A.M.; Leiter, L.A.; Kris-Etherton, P.M.; et al. Effect of dietary pulses on blood pressure: A systematic review and meta-analysis of controlled feeding trials. Am. J. Hypertens. 2014, 27, 56–64. [Google Scholar] [PubMed]
- Huang, H.; Chen, G.; Liao, D.; Zhu, Y.; Xue, X. Effects of Berries Consumption on Cardiovascular Risk Factors: A Meta-analysis with Trial Sequential Analysis of Randomized Controlled Trials. Sci. Rep. 2016, 6, 23625. [Google Scholar] [CrossRef] [PubMed]
- Khan, K.; Jovanovski, E.; Ho, H.V.T.; Marques, A.C.R.; Zurbau, A.; Mejia, S.B.; Sievenpiper, J.L.; Vuksan, V. The effect of viscous soluble fiber on blood pressure: A systematic review and meta-analysis of randomized controlled trials. Nutr. Metab. Cardiovasc. Dis. 2018, 28, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Ho, H.V.; Sievenpiper, J.L.; Zurbau, A.; Blanco Mejia, S.; Jovanovski, E.; Au-Yeung, F.; Jenkins, A.L.; Vuksan, V. The effect of oat beta-glucan on LDL-cholesterol, non-HDL-cholesterol and apoB for CVD risk reduction: A systematic review and meta-analysis of randomised-controlled trials. Br. J. Nutr. 2016, 116, 1369–1382. [Google Scholar] [CrossRef] [PubMed]
- Hollaender, P.L.; Ross, A.B.; Kristensen, M. Whole-grain and blood lipid changes in apparently healthy adults: A systematic review and meta-analysis of randomized controlled studies. Am. J. Clin. Nutr. 2015, 102, 556–572. [Google Scholar] [CrossRef]
- Ha, V.; Sievenpiper, J.L.; de Souza, R.J.; Jayalath, V.H.; Mirrahimi, A.; Agarwal, A.; Chiavaroli, L.; Mejia, S.B.; Sacks, F.M.; Di Buono, M.; et al. Effect of dietary pulse intake on established therapeutic lipid targets for cardiovascular risk reduction: A systematic review and meta-analysis of randomized controlled trials. CMAJ 2014, 186, E252–E262. [Google Scholar] [CrossRef]
- Hooper, L.; Martin, N.; Abdelhamid, A.; Davey Smith, G. Reduction in saturated fat intake for cardiovascular disease. Cochrane Database Syst. Rev. 2015. [Google Scholar] [CrossRef]
- Chiavaroli, L.; Nishi, S.K.; Khan, T.A.; Braunstein, C.R.; Glenn, A.J.; Mejia, S.B.; Rahelic, D.; Kahleova, H.; Salas-Salvado, J.; Jenkins, D.J.A.; et al. Portfolio Dietary Pattern and Cardiovascular Disease: A Systematic Review and Meta-analysis of Controlled Trials. Prog. Cardiovasc. Dis. 2018, 61, 43–53. [Google Scholar] [CrossRef] [PubMed]
- Mytton, O.T.; Nnoaham, K.; Eyles, H.; Scarborough, P.; Ni Mhurchu, C. Systematic review and meta-analysis of the effect of increased vegetable and fruit consumption on body weight and energy intake. BMC Public Health 2014, 14, 886. [Google Scholar] [CrossRef] [PubMed]
- Howarth, N.C.; Saltzman, E.; Roberts, S.B. Dietary fiber and weight regulation. Nutr. Rev. 2001, 59, 129–139. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.J.; de Souza, R.J.; Choo, V.L.; Ha, V.; Cozma, A.I.; Chiavaroli, L.; Mirrahimi, A.; Blanco Mejia, S.; Di Buono, M.; Bernstein, A.M.; et al. Effects of dietary pulse consumption on body weight: A systematic review and meta-analysis of randomized controlled trials. Am. J. Clin. Nutr. 2016, 103, 1213–1223. [Google Scholar] [CrossRef] [PubMed]
- Li, S.S.; Kendall, C.W.; de Souza, R.J.; Jayalath, V.H.; Cozma, A.I.; Ha, V.; Mirrahimi, A.; Chiavaroli, L.; Augustin, L.S.; Blanco Mejia, S.; et al. Dietary pulses, satiety and food intake: A systematic review and meta-analysis of acute feeding trials. Obesity (Silver Spring) 2014, 22, 1773–1780. [Google Scholar] [CrossRef] [PubMed]
- Oh, S.W.; Han, K.H.; Han, S.Y.; Koo, H.S.; Kim, S.; Chin, H.J. Association of Sodium Excretion With Metabolic Syndrome, Insulin Resistance, and Body Fat. Medicine (Baltimore) 2015, 94, e1650. [Google Scholar] [CrossRef] [PubMed]
- Jiao, J.; Xu, J.Y.; Zhang, W.; Han, S.; Qin, L.Q. Effect of dietary fiber on circulating C-reactive protein in overweight and obese adults: A meta-analysis of randomized controlled trials. Int. J. Food Sci. Nutr. 2015, 66, 114–119. [Google Scholar] [CrossRef]
- Estruch, R.; Martinez-Gonzalez, M.A.; Corella, D.; Basora-Gallisa, J.; Ruiz-Gutierrez, V.; Covas, M.I.; Fiol, M.; Gomez-Gracia, E.; Lopez-Sabater, M.C.; Escoda, R.; et al. Effects of dietary fibre intake on risk factors for cardiovascular disease in subjects at high risk. J. Epidemiol. Community Health 2009, 63, 582–588. [Google Scholar] [CrossRef]
- Kanauchi, O.; Mitsuyama, K.; Araki, Y.; Andoh, A. Modification of intestinal flora in the treatment of inflammatory bowel disease. Curr. Pharm Des. 2003, 9, 333–346. [Google Scholar] [CrossRef]
- Scheppach, W.; Wiggins, H.S.; Halliday, D.; Self, R.; Howard, J.; Branch, W.J.; Schrezenmeir, J.; Cummings, J.H. Effect of gut-derived acetate on glucose turnover in man. Clin. Sci. (Lond.) 1988, 75, 363–370. [Google Scholar] [CrossRef]
- Lee, Y.; Kang, D.; Lee, S.A. Effect of dietary patterns on serum C-reactive protein level. Nutr. Metab. Cardiovasc. Dis. 2014, 24, 1004–1011. [Google Scholar] [CrossRef] [PubMed]
- Watzl, B.; Kulling, S.E.; Moseneder, J.; Barth, S.W.; Bub, A. A 4-wk intervention with high intake of carotenoid-rich vegetables and fruit reduces plasma C-reactive protein in healthy, nonsmoking men. Am. J. Clin. Nutr. 2005, 82, 1052–1058. [Google Scholar] [CrossRef] [PubMed]
- Dibaba, D.T.; Xun, P.; He, K. Dietary magnesium intake is inversely associated with serum C-reactive protein levels: Meta-analysis and systematic review. Eur. J. Clin. Nutr. 2014, 68, 971. [Google Scholar] [CrossRef] [PubMed]
- Almoznino-Sarafian, D.; Berman, S.; Mor, A.; Shteinshnaider, M.; Gorelik, O.; Tzur, I.; Alon, I.; Modai, D.; Cohen, N. Magnesium and C-reactive protein in heart failure: An anti-inflammatory effect of magnesium administration? Eur. J. Nutr. 2007, 46, 230–237. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Touyz, R.M.; Park, J.B.; Schiffrin, E.L. Antioxidant effects of vitamins C and E are associated with altered activation of vascular NADPH oxidase and superoxide dismutase in stroke-prone SHR. Hypertension 2001, 38, 606–611. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Gao, M.; Nie, W.; Yuan, J.; Zhang, B.; Wang, Z.; Wu, Z. Dietary magnesium sulfate supplementation protects heat stress-induced oxidative damage by restoring the activities of anti-oxidative enzymes in broilers. Biol. Trace Elem. Res. 2012, 146, 53–58. [Google Scholar] [CrossRef] [PubMed]
- Blanco Mejia, S.; Kendall, C.W.; Viguiliouk, E.; Augustin, L.S.; Ha, V.; Cozma, A.I.; Mirrahimi, A.; Maroleanu, A.; Chiavaroli, L.; Leiter, L.A.; et al. Effect of tree nuts on metabolic syndrome criteria: A systematic review and meta-analysis of randomised controlled trials. BMJ Open 2014, 4, e004660. [Google Scholar] [CrossRef]
- Ho, H.V.; Sievenpiper, J.L.; Zurbau, A.; Blanco Mejia, S.; Jovanovski, E.; Au-Yeung, F.; Jenkins, A.L.; Vuksan, V. A systematic review and meta-analysis of randomized controlled trials of the effect of barley beta-glucan on LDL-C, non-HDL-C and apoB for cardiovascular disease risk reduction(i-iv). Eur. J. Clin. Nutr. 2016, 70, 1340. [Google Scholar] [CrossRef]
- Health Promotion and Disease Prevention: Fruit and Vegetables. European Commission. Available online: https://ec.europa.eu/jrc/en/health-knowledge-gateway/promotion-prevention/nutrition/fruit-vegetables#_jrc2018 (accessed on 28 November 2018).
- Health Promotion and Disease Prevention: Dietary Fats. European Commission. Available online: https://ec.europa.eu/jrc/en/health-knowledge-gateway/promotion-prevention/nutrition/fats#_Toctb5b (accessed on 28 November 2018).
- Hjartaker, A.; Lagiou, A.; Slimani, N.; Lund, E.; Chirlaque, M.D.; Vasilopoulou, E.; Zavitsanos, X.; Berrino, F.; Sacerdote, C.; Ocke, M.C.; et al. Consumption of dairy products in the European Prospective Investigation into Cancer and Nutrition (EPIC) cohort: Data from 35,955 24-hour dietary recalls in 10 European countries. Public Health Nutr. 2002, 5, 1259–1271. [Google Scholar] [CrossRef]
- Lee-Kwan, S.H.; Moore, L.V.; Blanck, H.M.; Harris, D.M.; Galuska, D. Disparities in State-Specific Adult Fruit and Vegetable Consumption—United States, 2015. MMWR Morb. Mortal Wkly. Rep. 2017, 66, 1241–1247. [Google Scholar] [CrossRef]
- U. S. Department of Agricultural Service. Food Patterns Equivalents Intakes from Food: Mean Amounts Consumed per Individual, by Gender and Age, What We Eat In America, NHANES 2009–2010. 2013. Available online: www.ars.usda.gov/ba/bhnrc/fsrg (accessed on 27 November 2018).
- Nielsen, S.J.; Kit, B.K.; Ogden, C.L. Nut Consumption Among U.S. Adults, 2009–2010. NCHS Data Brief. 2014, 176, 1–8. [Google Scholar]
| Chronic Disease | Systematic Review and Meta-Analysis | Total no. of obs Studies | Total no. of Participants | Median no. Participants (Range) | Countries | Outcomes Assessed | Total no. of Incident Cases (Range) | Median Age, yr (Range) | Median Duration of Study, yr (Range) | Dietary Intake Assessments (at Baseline) | DASH Exposure Assessments | Method of Outcome Assessment | Funding Source ‡ | Risk of Bias Assessment ** |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CVD | Schwingshackl et al., 2015 [13] | 11 | 783,732 | 44,544 (2061–242,321) | 7 USA: 2 Sweden: 1 Italy: 1 China | 3 CHD incidence: 3 CVD mortality: 3 stroke incidence: 1 CVD incidence: 1 sudden cardiac death | 32,927 (123–15,497) | 60.5 (20–83) | 14.6 (7.9-24) | FFQ | 7 quintiles: 2 quartiles: 2 tertiles of DASH score | Record linkage | 8 Agency: 2 not reported: 1 Agency, Industry | 10 H: 1 Lo |
| CHD | Salehi-Abargouei et al., 2013 [43] | 3 | 144, 337 | 348,827 (20,993–88,517) | 3 USA | 1 CHD morbidity and mortality: 1 CHD mortality: 1 fatal and nonfatal CHD | 7260 (430–6210) | 52 (30–69) | 16 (14.6-24) | FFQ | 3 quintiles | Record linkage | 3 Agency | 2 H: 1 Lo |
| Stroke | Salehi-Abargouei et al., 2013 [43] | 3 | 150,191 | 40,681 (20,993–88,517) | 2 USA: 1 Italy | 1 stroke incidence: 1 stroke mortality: 1 fatal and nonfatal stroke | 4,413 (178–3999) | 52 (30–74) | 16 (7.9-24) | FFQ | 2 quintiles: 1 tertiles | Record linkage | 2 Agency: 1 Agency, Industry | 2 H: 1 Lo |
| Diabetes | Jannasch et al., 2017 [12] | 5 | 158,408 | 21, 616 (822–89,195) | 4 US: 1 Europe (8 countries) | diabetes incidence | 23,612 (129–11, 217) | 58 (25–84) | 11.5 (5-20) | FFQ | 4 quintiles: 1 tertiles | 3 self-reported + record linkage: 1 independent assessment: 1 OGTT or DM med use | 4 Agency: 1 Agency, Industry | 4 H: 1 Lo |
| Cardiometabolic Risk Factor | Systematic Review and Meta-Analysis | Total no. of Trials | Total n | Median Sample Size (Range) | Metabolic Phenotypes: no. of Trials | Median Age, yr (Rangea) | Median Follow-up, wks (range) | Trial Design: no. of Trials | Countries: no. of Trials | Randomized: no. of Trials | Intervention: no. of Trials | Comparator: no. of Trials | Feeding/Compliance: no. of Trials | Rob: no. of Trials b |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Blood Pressure (SBP + DBP) | Siervo et al., 2015 [11] | 19 | 1, 918 | 37 (12–537) | 9 HTN: 3 PreHTN + HTN: 2 MetS: 1 GDM: 1 Norm: 1 OH, lean: 1 OB: 1 T2DM | 44.3 (30.1–59.2) | 8 (2–24) | 6 CO: 13 P | 12 USA: 4 Iran: 3 Australia | 16 Y: 3 N | 15 DASH alone: 2 weight loss DASH: 1 weight loss DASH + exercise: 1 behavioural intervention plus DASH | 10 usual diet: 2 low fat diet: 2 low antioxidant diet: 2 weight loss diet:1 behavioural intervention alone: 1 exercise alone: 1 usual GDM practice | 14 dietary advice: 4 metabolic: 1 dietary advice plus some supplemented foods | 11 U: 8 Lo |
| Total-C + LDL-C | Siervo et al., 2015 [11] | 13 | 1, 673 | 54 (12–537) | 6 HTN: 2 PreHTN + HTN: 1 GDM: 1 Norm: 1 OH, Lean: 1 OB: 1 T2DM | 48.3 (30.1–59.2) | 4 (3–24) | 6 CO: 7 P | 8 USA: 3 Australia: 2 Iran | 10 Y: 3 N | 12 DASH alone: 1 behavioural intervention plus DASH | 7 usual diet: 2 low fat diet: 2 low antioxidant diet:1 behavioural intervention alone: 1 usual GDM practice | 10 dietary advice: 2 metabolic: 1 dietary advice plus some supplemented foods | 9 U: 4 Lo |
| HDL-C | Siervo et al., 2015 [11] | 15 | 1, 749 | 54 (12–537) | 6 HTN: 2 PreHTN + HTN: 2 MetS: 1 GDM: 1 Norm: 1 OH, lean: 1 OB: 1 T2DM | 44.0 (30.1–59.2) | 8 (3–24) | 6 CO: 9 P | 8 USA: 4 Iran: 3 Australia | 12 Y: 3 N | 12 DASH alone: 2 weight loss DASH: 1 behavioural intervention plus DASH | 7 usual diet: 2 low fat diet: 2 low antioxidant diet: 2 weight loss diet:1 behavioural intervention alone: 1 usual GDM practice | 12 dietary advice: 2 metabolic: 1 dietary advice plus some supplemented foods | 11 U: 4 Lo |
| Triglycerides | Siervo et al., 2015 [11] | 14 | 1, 654 | 44 (12–537) | 5 HTN: 2 PreHTN + HTN: 2 MetS: 1 GDM: 1 Norm: 1 OH, Lean: 1 OB: 1 T2DM | 42.6 (30.1–55.6) | 6 (3–24) | 6 CO: 8 P | 8 USA: 4 Iran: 2 Australia | 11 Y: 3 N | 11 DASH alone: 2 weight loss DASH: 1 behavioural intervention plus DASH | 7 usual diet: 1 low fat diet: 2 low antioxidant diet: 2 weight loss diet:1 behavioural intervention alone: 1 usual GDM practice | 12 dietary advice: 2 metabolic | 10 U: 4 Lo |
| HbA1c | Manual conductc | 2 | 65 | 33 (31–34) | 1 GDM: 1 T2DM | 42.6 (30.1–55.0) | 6 (4–8) | 1 CO: 1P | 2 Iran | 2 Y | 2 DASH alone | 2 usual diet | 2 dietary advice | 1 U: 1 Lo |
| Blood glucose | Siervo et al., 2015 [11] | 10 | 826 | 27 (12–537) | 1 HTN: 2 PreHTN + HTN: 2 MetS: 1 GDM: 1 Norm: 1 OH, Lean: 1 OB: 1 T2DM | 40.8 (30.1–55.0) | 6 (3–24) | 5 CO: 5 P | 6 USA: 4 Iran | 8 Y: 2 N | 7 DASH alone: 2 weight loss DASH: 1 behavioural intervention plus DASH | 4 usual diet: 2 low antioxidant diet: 2 weight loss diet: 1 behavioural intervention alone: 1 usual GDM practice | 10 dietary advice | 7 U: 3 Lo |
| Fasting insulin | Shirani et al., 2013 [64] | 11 | 760 | 15 (9–266) | 2 HTN: 4 PreHTN + HTN: 1 Norm: 1 OH: 1 OH, Lean: 2 OW/OB | 44.1 (34.3–51.8) | 4 (3–24) | 6 CO: 5 P | 10 USA: 1 UK | 6 Y: 5 N | 8 DASH alone: 3 behavioural intervention plus DASH | 6 usual diet: 2 low antioxidant diet: 3 advice only | 10 dietary advice: 1 dietary advice plus some supplemented foods | 7 U: 4 Lo |
| HOMA-IR | Shirani et al., 2013 [64] | 8 | 603 | 14 (9–266) | 1 HTN: 3 PreHTN + HTN: 1 Norm: 1 OH: 1 OH, Lean: 1 OB | 39.7 (34.3–49.8) | 3.5 (3–24) | 6 CO: 2 P | 8 USA | 4 Y: 4 N | 6 DASH alone: 2 behavioural intervention plus DASH | 4 usual diet: 2 low antioxidant diet: 2 advice only | 8 dietary advice | 6 U: 2 Lo |
| Body weight | Soltani et al., 2016 [65] | 11 | 1,211 | 54 (22–476) | 5 HTN: 2 MetS: 1 PreHTN + HTN: 1 HF patients: 1 OW/OB, NAFLD: 1 OW/OB, PCOS | 48.5 (30.1–62.0) | 16 (8–52) | 0 CO: 11 P | 4 USA: 4 Iran: 2 Australia: 1 Brazil | 11 Y: 0 N | 5 DASH alone: 4 weight loss DASH: 1 behavioural intervention Plus DASH: 1 DASH + LGI | 4 weight loss: 2 low fat: 2 usual diet: 1 behavioural intervention: 1 general HF recommendations: 1 standard low sodium HTN advice | 10 dietary advice: 1 dietary advice plus some supplemented foods | 6 U: 5 Lo |
| CRP | Soltani et al., 2017 [66] | 6 | 451 | 42 (31–241) | 2 Hyperlipidemic: 1 Lean Norm + OB HTN: 1 OW/OB, NAFLD: 1 OW/OB, PCOS: 1 T2DM | 45.7 (30.1–55.0) | 8 (3–24) | 3 CO: 3 P | 3 Iran: 2 USA: 1 Canada | 16 Y: 0 N | 3 DASH alone: 2 weight loss DASH: 1 lacto-ovo vegetarian DASH | 2 weight loss: 1 usual diet: 1 usual plus fibre, potassium, magnesium: 1 healthy American: 1 Portfolio diet (plant-based with soy protein, viscous fibres and nuts) | 15 dietary advice: 1 metabolic | 3 U: 3 Lo |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chiavaroli, L.; Viguiliouk, E.; Nishi, S.K.; Blanco Mejia, S.; Rahelić, D.; Kahleová, H.; Salas-Salvadó, J.; Kendall, C.W.; Sievenpiper, J.L. DASH Dietary Pattern and Cardiometabolic Outcomes: An Umbrella Review of Systematic Reviews and Meta-Analyses. Nutrients 2019, 11, 338. https://doi.org/10.3390/nu11020338
Chiavaroli L, Viguiliouk E, Nishi SK, Blanco Mejia S, Rahelić D, Kahleová H, Salas-Salvadó J, Kendall CW, Sievenpiper JL. DASH Dietary Pattern and Cardiometabolic Outcomes: An Umbrella Review of Systematic Reviews and Meta-Analyses. Nutrients. 2019; 11(2):338. https://doi.org/10.3390/nu11020338
Chicago/Turabian StyleChiavaroli, Laura, Effie Viguiliouk, Stephanie K Nishi, Sonia Blanco Mejia, Dario Rahelić, Hana Kahleová, Jordi Salas-Salvadó, Cyril WC Kendall, and John L Sievenpiper. 2019. "DASH Dietary Pattern and Cardiometabolic Outcomes: An Umbrella Review of Systematic Reviews and Meta-Analyses" Nutrients 11, no. 2: 338. https://doi.org/10.3390/nu11020338
APA StyleChiavaroli, L., Viguiliouk, E., Nishi, S. K., Blanco Mejia, S., Rahelić, D., Kahleová, H., Salas-Salvadó, J., Kendall, C. W., & Sievenpiper, J. L. (2019). DASH Dietary Pattern and Cardiometabolic Outcomes: An Umbrella Review of Systematic Reviews and Meta-Analyses. Nutrients, 11(2), 338. https://doi.org/10.3390/nu11020338









