Prostate Cancer Disparity, Chemoprevention, and Treatment by Specific Medicinal Plants
Abstract
1. Introduction
2. Approaches
3. Results and Discussions
4. Prostate Cancer Disparity in the United States
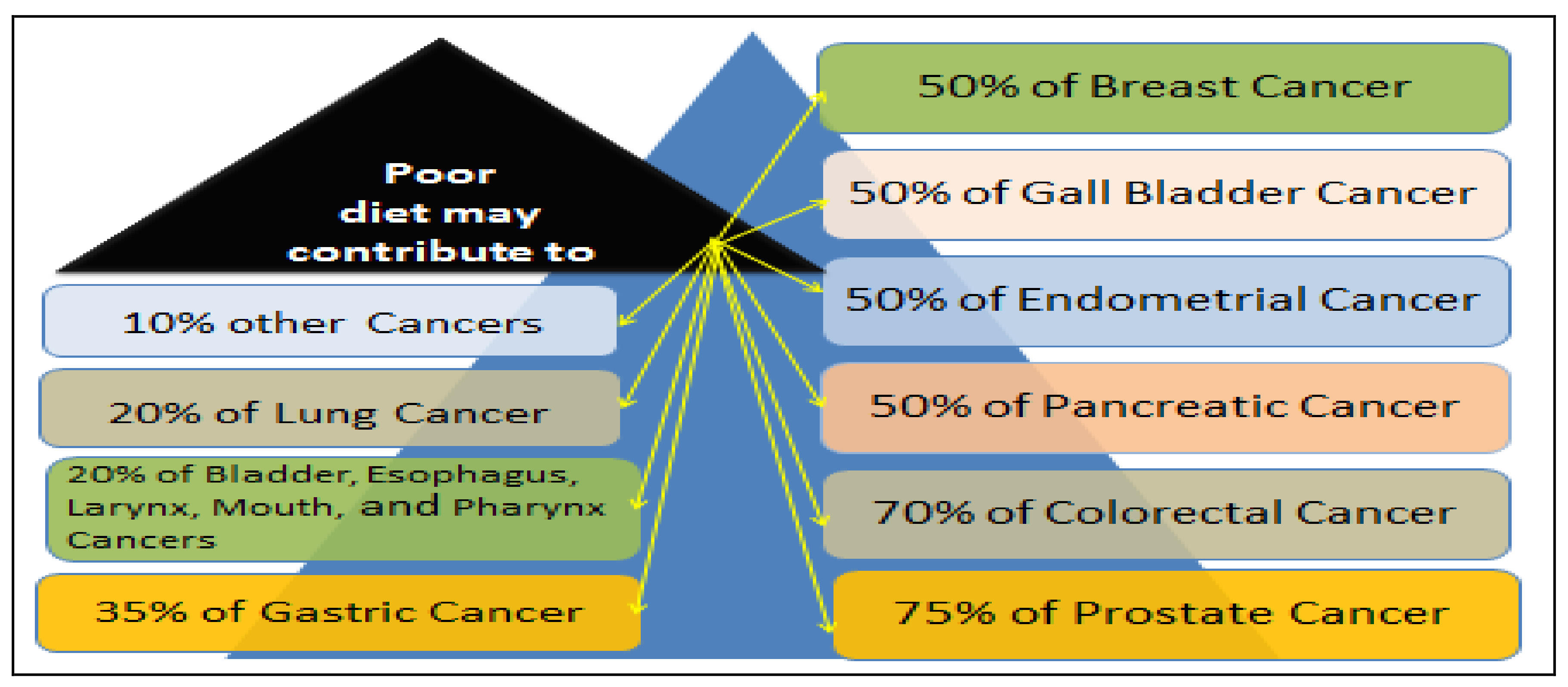
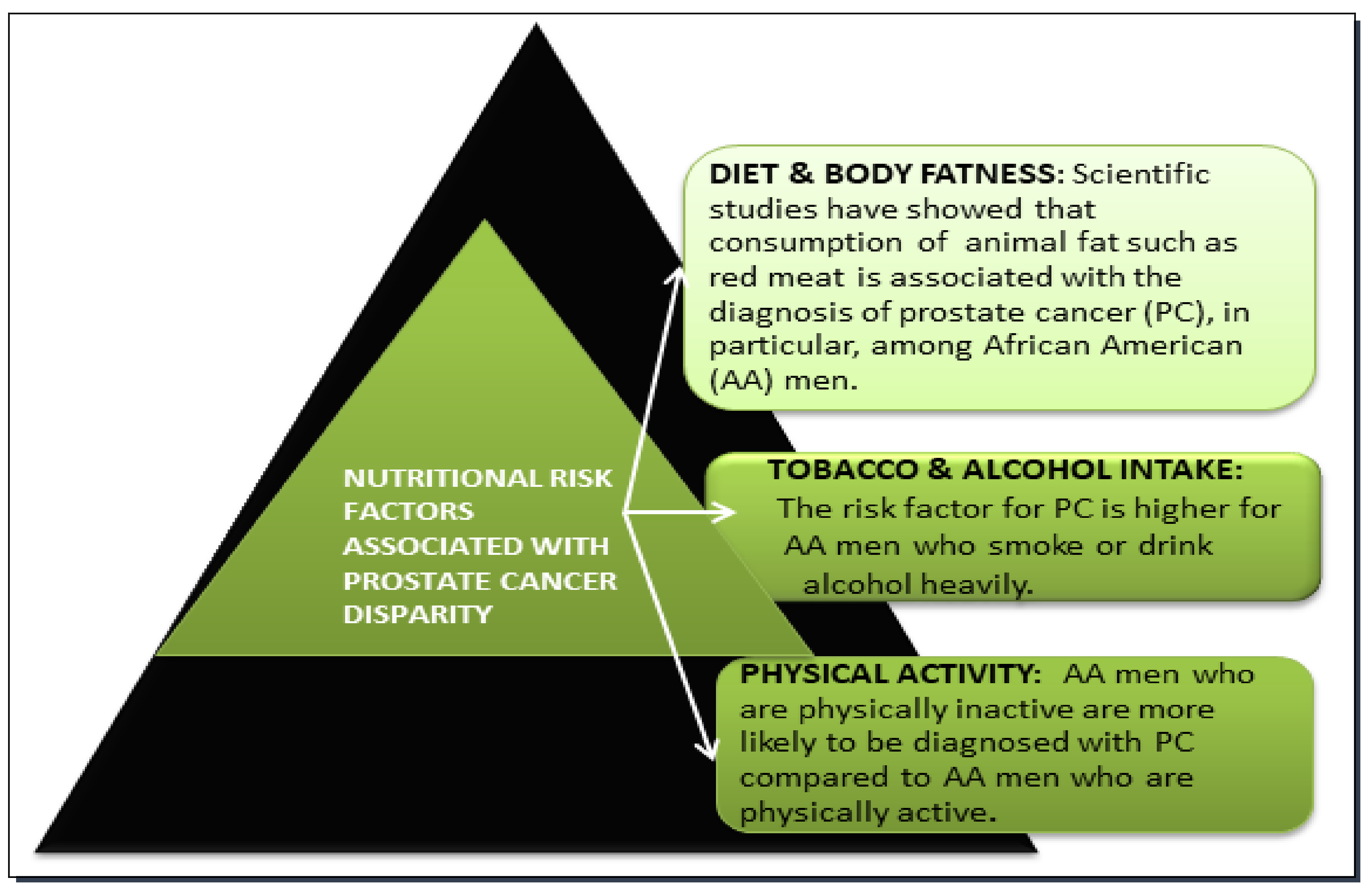
4.1. Dietary Factor in Prostate Cancer Disparity
4.2. Tobacco and Alcohol Factors in Prostate Cancer Disparity
4.3. Physical Activity Factor in Prostate Cancer Disparity
4.4. Geographic Factor in Prostate Cancer Disparity
4.5. Socioeconomic Status in Prostate Cancer Disparity
4.6. Oxidative Stress and Age Factors in Prostate Cancer Disparity
5. Chemo-Preventive and Therapeutic Effects of Curcumin on Prostate Cancer
6. Chemo-Preventive and Therapeutic Effects of Garlic on Prostate Cancer
7. Chemo-Preventive and Therapeutic Effects of Vernonia amygdalina on Prostate Cancer
8. Summary of Chemo-Preventive and Therapeutic Effects of Curcumin, Garlic, and Vernonia amygdalina on Prostate Cancer
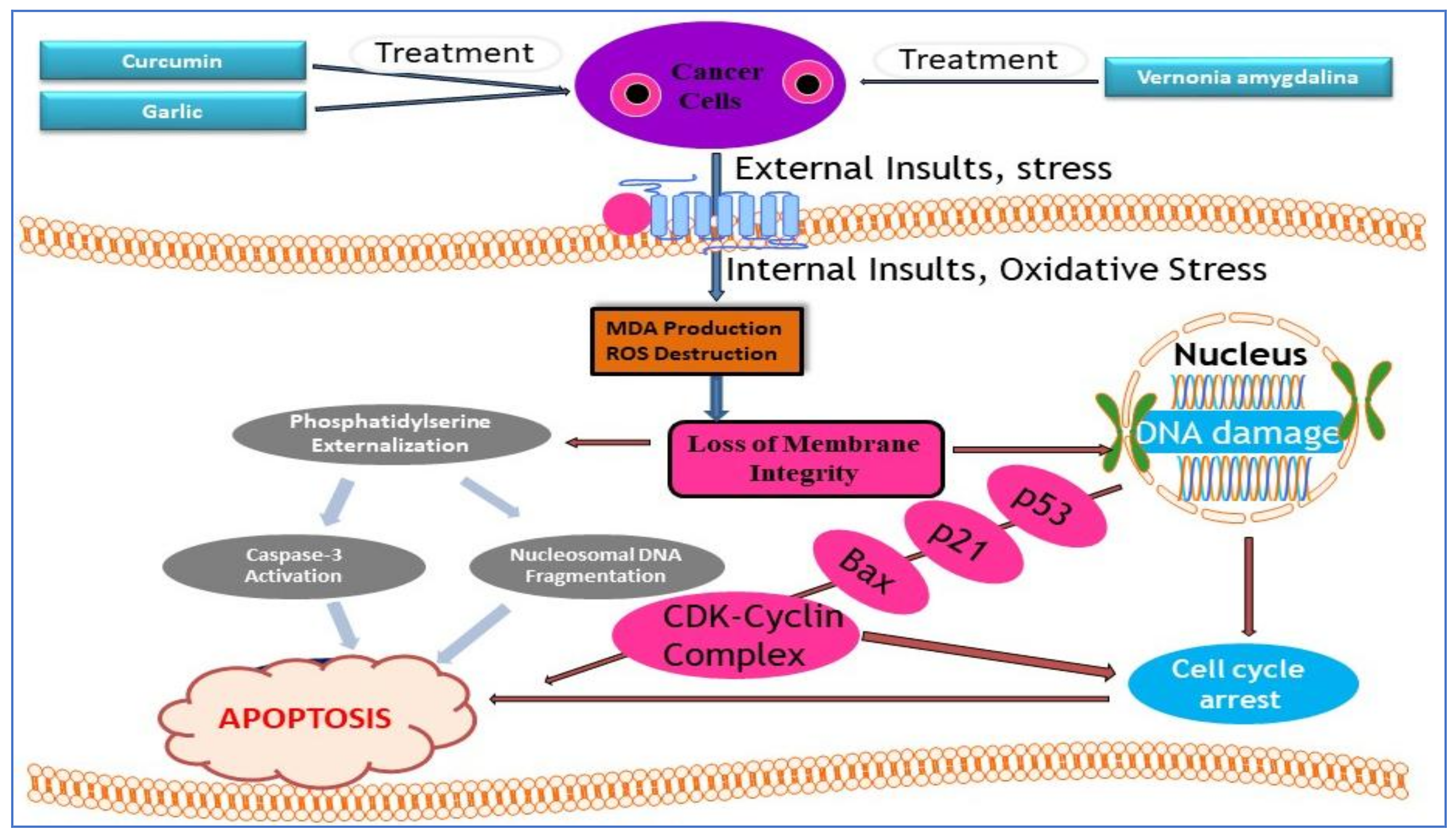
9. Molecular Mechanisms of Action of Medicinal Plants (Curcumin, Garlic, and Vernonia amygdalina)
10. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ferlay, J.; Parkin, D.M.; Steliarova-Foucher, E. Estimates of cancer incidence and mortality in Europe in 2008. Eur. J. Cancer 2010, 46, 765–781. [Google Scholar] [CrossRef] [PubMed]
- Siegel, R.; Miller, K.; Jemal, A. Cancer statistics, 2015. CA Cancer J. Clin. 2015, 65, 29. [Google Scholar] [CrossRef] [PubMed]
- Ferraldeschi, R.; Pezaro, C.; Karavasilis, V.; de Bono, J. Abiraterone and Novel Antiandrogens: Overcoming Castration Resistance in Prostate Cancer. Annu. Rev. Med. 2013, 64, 1–13. [Google Scholar] [CrossRef]
- Roudier, M.P.; True, L.D.; Higano, C.S.; Vesselle, H.; Ellis, W.; Lange, P.; Vessella, R.L. Phenotypic heterogeneity of end-stage prostate carcinoma metastatic to bone. Hum. Pathol. 2003, 34, 646–653. [Google Scholar] [CrossRef]
- Pagliarulo, V. Androgen Deprivation Therapy for Prostate Cancer. Adv. Exp. Med. Biol. 2018, in press. [Google Scholar]
- Watson, P.A.; Arora, V.K.; Sawyers, C.L. Emerging mechanisms of resistance to androgen receptor inhibitors in prostate cancer. Nat. Rev. Cancer 2015, 15, 701–711. [Google Scholar] [CrossRef] [PubMed]
- Pelekanou, V.; Castanas, E. Androgen Control in Prostate Cancer. J. Cell. Biochem. 2016, 117, 2224–2234. [Google Scholar] [CrossRef]
- Teply, B.A.; Antonarakis, E.S. Novel mechanism-based therapeutics for androgen axis blockade in castration-resistant prostate cancer. Curr. Opin. Endocrinol. Diabetes Obes. 2016, 23, 279–290. [Google Scholar] [CrossRef]
- Shahinian, V.B.; Kuo, Y.F.; Freeman, J.L.; Goodwin, J.S. Risk of the “androgen deprivation syndrome” in men receiving androgen deprivation for prostate cancer. Arch. Intern. Med. 2006, 166, 465–471. [Google Scholar]
- Nead, K.T.; Gaskin, G.; Chester, C.; Swisher-McClure, S.; Leeper, N.J.; Shah, N.H. Association between androgen deprivation therapy and risk of dementia. JAMA Oncol. 2017, 3, 49–55. [Google Scholar] [CrossRef]
- Viani, G.A.; Bernardes Da Silva, L.G.; Stefano, E.J. Prevention of gynecomastia and breast pain caused by androgen deprivation therapy in prostate cancer: Tamoxifen or radiotherapy? Int. J. Radiat. Oncol. Biol. Phys. 2012, 83, e519–e524. [Google Scholar] [CrossRef] [PubMed]
- Higano, C.S. Bone loss and the evolving role of bisphosphonate therapy in prostate cancer. Urol. Oncol. Semin. Orig. Investig. 2003, 21, 392–398. [Google Scholar] [CrossRef]
- Scher, H.I.; Fizazi, K.; Saad, F.; Taplin, M.-E.; Sternberg, C.N.; Miller, K.; de Wit, R.; Mulders, P.; Chi, K.N.; Shore, N.D.; et al. Increased Survival with Enzalutamide in Prostate Cancer after Chemotherapy - Supplementary Appendix. N. Engl. J. Med. 2012, 367, 1187–1197. [Google Scholar] [CrossRef] [PubMed]
- Fizazi, K.; Scher, H.I.; Molina, A.; Logothetis, C.J.; Chi, K.N.; Jones, R.J.; Staffurth, J.N.; North, S.; Vogelzang, N.J.; Saad, F.; et al. Abiraterone acetate for treatment of metastatic castration-resistant prostate cancer: Final overall survival analysis of the COU-AA-301 randomised, double-blind, placebo-controlled phase 3 study. Lancet Oncol. 2012, 13, 983–992. [Google Scholar] [CrossRef]
- Fletcher, R.H.; Fairfield, K.M. Vitamins for chronic disease prevention in adults: Clinical applications. JAMA 2002, 287, 3127–3129. [Google Scholar] [CrossRef] [PubMed]
- Reddy, A.; Kaelin, W.G., Jr. Using cancer genetics to guide the selection of anticancer drug targets. Curr. Opin. Pharmacol. 2002, 2, 366–373. [Google Scholar] [CrossRef]
- World Cancer Research Fund; American Institute for Cancer Research. Food, Nutrition, Physical Activity, and the Prevention of Cancer: A Global Perspective; World Cancer Research Fund: London, UK, 2007; ISBN 9780972252225. [Google Scholar]
- Yedjou, C.; Tchounwou, P.; Payton, M.; Miele, L.; Fonseca, D.; Lowe, L.; Alo, R. Assessing the Racial and Ethnic Disparities in Breast Cancer Mortality in the United States. Int. J. Environ. Res. Public Health 2017, 14, 486. [Google Scholar] [CrossRef]
- Syed, D.N.; Khan, N.; Afaq, F.; Mukhtar, H. Chemoprevention of prostate cancer through dietary agents: Progress and promise. Cancer Epidemiol. Biomarkers Prev. 2007, 16, 2193–2203. [Google Scholar] [CrossRef]
- Soerjomataram, I.; Oomen, D.; Lemmens, V.; Oenema, A.; Benetou, V.; Trichopoulou, A.; Coebergh, J.W.; Barendregt, J.; De Vries, E. Increased consumption of fruit and vegetables and future cancer incidence in selected European countries. Eur. J. Cancer 2010, 46, 2563–2580. [Google Scholar] [CrossRef]
- Turati, F.; Rossi, M.; Pelucchi, C.; Levi, F.; La Vecchia, C. Fruit and vegetables and cancer risk: A review of southern European studies. Br. J. Nutr 2015, 113, S102–S110. [Google Scholar] [CrossRef]
- Martínez, M.E.; Jacobs, E.T.; Baron, J.A.; Marshall, J.R.; Byers, T. Dietary supplements and cancer prevention: Balancing potential benefits against proven harms. J. Natl. Cancer Inst. 2012, 104, 732–739. [Google Scholar] [CrossRef] [PubMed]
- Bjelakovic, G.; Nikolova, D.; Gluud, L.L.; Simonetti, R.G.; Gluud, C. Antioxidant supplements for prevention of mortality in healthy participants and patients with various diseases. Sao Paulo Med. J. 2015, 133, 164. [Google Scholar] [CrossRef]
- Ochwang’i, D.O.; Kimwele, C.N.; Oduma, J.A.; Gathumbi, P.K.; Mbaria, J.M.; Kiama, S.G. Medicinal plants used in treatment and management of cancer in Kakamega County, Kenya. J. Ethnopharmacol. 2014, 151, 1040–1055. [Google Scholar] [CrossRef]
- Sivaraj, R.; Rahman, P.K.S.M.; Rajiv, P.; Narendhran, S.; Venckatesh, R. Biosynthesis and characterization of Acalypha indica mediated copper oxide nanoparticles and evaluation of its antimicrobial and anticancer activity. Spectrochim. Acta Part. A Mol. Biomol. Spectrosc. 2014, 129, 25–258. [Google Scholar] [CrossRef]
- Mothupi, M.C. Use of herbal medicine during pregnancy among women with access to public healthcare in Nairobi, Kenya: A cross-sectional survey. BMC Complement. Altern. Med. 2014, 14. [Google Scholar] [CrossRef] [PubMed]
- Rao, P.V.; Nallappan, D.; Madhavi, K.; Rahman, S.; Jun Wei, L.; Gan, S.H. Phytochemicals and Biogenic Metallic Nanoparticles as Anticancer Agents. Oxid. Med. Cell. Longev. 2016, 2016. [Google Scholar] [CrossRef] [PubMed]
- Tavakoli, J.; Miar, S.; Zadehzare, M.M.; Akbari, H. Evaluation of effectiveness of herbal medication in cancer care: A review study. Iran J. Cancer Prev. 2012, 5, 144–156. [Google Scholar]
- Wheat, J.; Currie, G. Herbal medicine for cancer patients: An evidence based review. Internet J. Altern. Med. 2008, 5, 5. [Google Scholar]
- Jemal, A.; Siegel, R.; Xu, J.; Ward, E. Cancer statistics, 2010. CA Cancer J. Clin. 2010, 60, 277–300. [Google Scholar] [CrossRef]
- Ward, E.; Jemal, A.; Cokkinides, V.; Singh, G.K.; Cardinez, C.; Ghafoor, A.; Thun, M. Cancer Disparities by Race/Ethnicity and Socioeconomic Status. CA. Cancer J. Clin. 2004, 54, 78–93. [Google Scholar] [CrossRef]
- Underwood, W.; DeMonner, S.; Ubel, P.; Fagerlin, A.; Sanda, M.G.; Wei, J.T. Racial/ethnic disparities in the treatment of localized/regional prostate cancer. J. Urol. 2004, 171, 1504–1507. [Google Scholar] [CrossRef] [PubMed]
- Gross, C.P.; Smith, B.D.; Wolf, E.; Andersen, M. Racial disparities in cancer therapy: Did the gap narrow between 1992 and 2002? Cancer 2008, 112, 900–908. [Google Scholar] [CrossRef] [PubMed]
- Chu, K.C.; Tarone, R.E.; Freeman, H.P. Trends in prostate cancer mortality among black men and white men in the United States. Cancer 2003, 97, 1507–1516. [Google Scholar] [CrossRef] [PubMed]
- Ghafoor, A.; Jemal, A.; Cokkinides, V.; Cardinez, C.; Murray, T.; Samuels, A.; Thun, M.J. Cancer statistics for African Americans. CA Cancer J. Clin. 2002, 52, 326–341. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, C.; McCullough, M.L.; Mondul, A.M.; Jacobs, E.J.; Chao, A.; Patel, A.V.; Thun, M.J.; Calle, E.E. Meat consumption among Black and White men and risk of prostate cancer in the Cancer Prevention Study II Nutrition Cohort. Cancer Epidemiol. Biomarkers Prev. 2006. [Google Scholar] [CrossRef] [PubMed]
- Amin, M.; Jeyaganth, S.; Fahmy, N.; Bégin, L.R.; Aronson, S.; Jacobson, S.; Tanguay, S.; Kassouf, W.; Aprikian, A. Dietary habits and prostate cancer detection: A case-control study. Can. Urol. Assoc. J. 2008, 2, 510–515. [Google Scholar] [CrossRef] [PubMed]
- American Cancer Society Cancer Facts & Figures 2013. Am. Cancer Soc. 2013. [CrossRef]
- Kurahashi, N.; Iwasaki, M.; Inoue, M.; Sasazuki, S.; Tsugane, S. Plasma isoflavones and subsequent risk of prostate cancer in a nested case-control study: The Japan Public Health Center. J. Clin. Oncol. 2008. [Google Scholar] [CrossRef]
- Kumar, N.B.; Kang, L.; Pow-Sang, J.; Xu, P.; Allen, K.; Riccardi, D.; Besterman-Dahan, K.; Krischer, J.P. Results of a randomized phase I dose-finding trial of several doses of isoflavones in men with localized prostate cancer: Administration prior to radical prostatectomy. J. Soc. Integr. Oncol. 2010. [Google Scholar] [CrossRef]
- Amling, C.L.; Riffenburgh, R.H.; Sun, L.; Moul, J.W.; Lance, R.S.; Kusuda, L.; Sexton, W.J.; Soderdahl, D.W.; Donahue, T.F.; Foley, J.P.; et al. Pathologic variables and recurrence rates as related to obesity and race in men with prostate cancer undergoing radical prostatectomy. J. Clin. Oncol. 2004. [Google Scholar] [CrossRef]
- Beebe-Dimmer, J.L.; Nock, N.L.; Neslund-Dudas, C.; Rundle, A.; Bock, C.H.; Tang, D.; Jankowski, M.; Rybicki, B.A. Racial Differences in Risk of Prostate Cancer Associated With Metabolic Syndrome. Urology 2009. [Google Scholar] [CrossRef] [PubMed]
- Gong, Z.; Neuhouser, M.L.; Goodman, P.J.; Albanes, D.; Chi, C.; Hsing, A.W.; Lippman, S.M.; Platz, E.A.; Pollak, M.N.; Thompson, I.M.; et al. Obesity, diabetes, and risk of prostate cancer: Results from the prostate cancer prevention trial. Cancer Epidemiol. Biomarkers Prev. 2006. [Google Scholar] [CrossRef] [PubMed]
- Spangler, E.; Zeigler-Johnson, C.M.; Coomes, M.; Malkowicz, S.B.; Wein, A.; Rebbeck, T.R. Association of obesity with tumor characteristics and treatment failure of prostate cancer in African-American and European American men. J. Urol. 2007. [Google Scholar] [CrossRef] [PubMed]
- Fesinmeyer, M.D.; Gulati, R.; Zeliadt, S.; Weiss, N.; Kristal, A.R.; Etzioni, R. Effect of population trends in body mass index on prostate cancer incidence and mortality in the United States. Cancer Epidemiol. Biomarkers Prev. 2009. [Google Scholar] [CrossRef] [PubMed]
- Putnam, S.D.; Cerhan, J.R.; Parker, A.S.; Bianchi, G.D.; Wallace, R.B.; Cantor, K.P.; Lynch, C.F. Lifestyle and anthropometric risk factors for prostate cancer in a cohort of Iowa men. Ann. Epidemiol. 2000. [Google Scholar] [CrossRef]
- Sung, J.F.C.; Lin, R.S.; Pu, Y.S.; Chen, Y.C.; Chang, H.C.; Lai, M.K. Risk factors for prostate carcinoma in Taiwan: A case-control study in a chinese population. Cancer 1999. [Google Scholar] [CrossRef]
- Schuurman, A.G.; Goldbohm, R.A.; Dorant, E.; Van Den Brandt, P.A. Anthropometry in relation to prostate cancer risk in the Netherlands Cohort Study. Am. J. Epidemiol. 2000. [Google Scholar] [CrossRef]
- Hsing, A.W.; Deng, J.; Sesterhenn, I.A.; Mostofi, F.K.; Stanczyk, F.Z.; Benichou, J.; Xie, T.; Gao, Y.T. Body size and prostate cancer: A population-based case-control study in China. Cancer Epidemiol. Biomarkers Prev. 2000, 9, 12. [Google Scholar]
- Jonssoni, F.; Wolk, A.; Pedersen, N.L.; Lichtenstein, P.; Terry, P.; Ahlbom, A.; Feychting, M. Obesity and hormone-dependent tumors: Cohort and co-twin control studies based on the Swedish Twin Registry. Int. J. Cancer 2003. [Google Scholar] [CrossRef]
- Lee, I.M.; Sesso, H.D.; Paffenbarger, R.S. A prospective cohort study of physical activity and body size in relation to prostate cancer risk (United States). Cancer Causes Control. 2001. [Google Scholar] [CrossRef]
- Rodriguez, C.; Freedland, S.J.; Deka, A.; Jacobs, E.J.; McCullough, M.L.; Patel, A.V.; Thun, M.J.; Calle, E.E. Body mass index, weight change, and risk of prostate cancer in the Cancer Prevention Study II Nutrition Cohort. Cancer Epidemiol. Biomarkers Prev. 2007. [Google Scholar] [CrossRef] [PubMed]
- Giovannucci, E.; Rimm, E.B.; Liu, Y.; Leitzmann, M.; Wu, K.; Stampfer, M.J.; Willett, W.C. Body mass index and risk of prostate cancer in U.S. health professionals. J. Natl. Cancer Inst. 2003. [Google Scholar] [CrossRef]
- Watters, J.L.; Park, Y.; Hollenbeck, A.; Schatzkin, A.; Albanes, D. Cigarette smoking and prostate cancer in a prospective US cohort study. Cancer Epidemiol. Biomarkers Prev. 2009. [Google Scholar] [CrossRef] [PubMed]
- Huncharek, M.; Sue Haddock, K.; Reid, R.; Kupelnick, B. Smoking as a risk factor for prostate cancer: A meta-analysis of 24 prospective cohort studies. Am. J. Public Health 2010. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention (CDC). Vital Signs: Current Cigarette Smoking among Adults Aged ≥18 Years with Mental Illness—United States, 2009–2011; MMWR, Morb. Mortal. Wkly. Rep.; CDC: Atlanta, GA, USA, 2013. [Google Scholar]
- Fu, S.S.; Burgess, D.; van Ryn, M.; Hatsukami, D.K.; Solomon, J.; Joseph, A.M. Views on smoking cessation methods in ethnic minority communities: A qualitative investigation. Prev. Med. (Baltim). 2007. [Google Scholar] [CrossRef] [PubMed]
- Fu, S.S.; Kodl, M.M.; Joseph, A.M.; Hatsukami, D.K.; Johnson, E.O.; Breslau, N.; Wu, B.; Bierut, L. Racial/ethnic disparities in the use of nicotine replacement therapy and quit ratios in lifetime smokers ages 25to 44 years. Cancer Epidemiol. Biomarkers Prev. 2008. [Google Scholar] [CrossRef]
- Trinidad, D.R.; Pérez-Stable, E.J.; White, M.M.; Emery, S.L.; Messer, K. A nationwide analysis of US racial/ethnic disparities in smoking behaviors, smoking cessation, and cessation-related factors. Am. J. Public Health 2011. [Google Scholar] [CrossRef]
- Levinson, A.H.; Borrayo, E.A.; Espinoza, P.; Flores, E.T.; Pérez-Stable, E.J. An Exploration of Latino Smokers and the Use of Pharmaceutical Aids. Am. J. Prev. Med. 2006. [Google Scholar] [CrossRef]
- Murphy, A.B.; Akereyeni, F.; Nyame, Y.A.; Guy, M.C.; Martin, I.K.; Hollowell, C.M.P.; Walker, K.; Kittles, R.A.; Ahaghotu, C. Smoking and prostate cancer in a multi-ethnic cohort. Prostate 2013. [Google Scholar] [CrossRef]
- Scoccianti, C.; Cecchini, M.; Anderson, A.S.; Berrino, F.; Boutron-Ruault, M.C.; Espina, C.; Key, T.J.; Leitzmann, M.; Norat, T.; Powers, H.; et al. European Code against Cancer 4th Edition: Alcohol drinking and cancer. Cancer Epidemiol. 2015. [Google Scholar] [CrossRef]
- World Cancer Research Fund. For C.R. Food, Nutrition, Physical Activity, and the Prevention of Cancer: A Global Perspective; AICR: Washington, DC, USA, 2007. [Google Scholar]
- Clarke, G.; Whittemore, A.S. Prostate cancer risk in relation to anthropometry and physical activity: The National Health and Nutrition Examination Survey I Epidemiological Follow-Up Study. Cancer Epidemiol. Biomarkers Prev. 2000, 9, 875–881. [Google Scholar] [PubMed]
- Moore, S.C.; Peters, T.M.; Ahn, J.; Park, Y.; Schatzkin, A.; Albanes, D.; Hollenbeck, A.; Leitzmann, M.F. Age-specific physical activity and prostate cancer risk among white men and black men. Cancer 2009. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. GLOBOCAN 2012: Estimated Cancer Incidence, Mortality and Prevalence Worldwide in 2012. Globocan 2012, 1–6. [Google Scholar] [CrossRef]
- Zhang, J.; Dhakal, I.B.; Zhao, Z.; Li, L. Trends in mortality from cancers of the breast, colon, prostate, esophagus, and stomach in East Asia: Role of nutrition transition. Eur. J. Cancer Prev. 2012. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, R.M.; Harlan, L.C.; Klabunde, C.N.; Gilliland, F.D.; Stephenson, R.A.; Hunt, W.C.; Potosky, A.L. Racial Differences in Initial Treatment for Clinically Localized Prostate Cancer: Results from the Prostate Cancer Outcomes Study. J. Gen. Intern. Med. 2003. [Google Scholar] [CrossRef]
- Barocas, D.A.; Penson, D.F. Racial variation in the pattern and quality of care for prostate cancer in the USA: Mind the gap. BJU Int. 2010. [Google Scholar] [CrossRef] [PubMed]
- American Cancer Society Cancer Facts & Figures 2018. Am. Cancer Soc. 2018. [CrossRef]
- Brawley, O.W.; Jani, A.B.; Master, V. Prostate Cancer and Race. Curr. Probl. Cancer 2007. [Google Scholar] [CrossRef]
- Robbins, A.S.; Koppie, T.M.; Gomez, S.L.; Parikh-Patel, A.; Mills, P.K. Differences in prognostic factors and survival among white and Asian men with prostate cancer, California, 1995–2004. Cancer 2007. [Google Scholar] [CrossRef]
- Chhatre, S.; Bruce Malkowicz, S.; Sanford Schwartz, J.; Jayadevappa, R. Understanding the Racial and Ethnic Differences in Cost and Mortality Among Advanced Stage Prostate Cancer Patients (STROBE). Medicine (Baltimore) 2015. [Google Scholar] [CrossRef]
- Willis, M.S.; Wians, F.H. The role of nutrition in preventing prostate cancer: A review of the proposed mechanism of action of various dietary substances. Clin. Chim. Acta 2003. [Google Scholar] [CrossRef]
- Cheng, I.; Witte, J.S.; McClure, L.A.; Shema, S.J.; Cockburn, M.G.; John, E.M.; Clarke, C.A. Socioeconomic status and prostate cancer incidence and mortality rates among the diverse population of California. Cancer Causes Control. 2009. [Google Scholar] [CrossRef] [PubMed]
- Woods, L.M.; Rachet, B.; Coleman, M.P. Origins of socio-economic inequalities in cancer survival: A review. Ann. Oncol. 2006. [Google Scholar] [CrossRef] [PubMed]
- Weinrich, S.P.; Reynolds, W.A.J.; Tingen, M.S.; Starr, C.R. Barriers to prostate cancer screening. Cancer Nurs. 2000, 23, 117–121. [Google Scholar] [CrossRef] [PubMed]
- Gilligan, T. Social disparities and prostate cancer: Mapping the gaps in our knowledge. Cancer Causes Control 2005, 16, 45–53. [Google Scholar] [CrossRef] [PubMed]
- Penson, D.F.; Stoddard, M.L.; Pasta, D.J.; Lubeck, D.P.; Flanders, S.C.; Litwin, M.S. The Association Between Socioeconomic Status, Health Insurance Coverage, and Quality of Life in Men with Prostate Cancer. J. Clin. Epidemiol. 2001, 80, 350–358. [Google Scholar] [CrossRef]
- White, A.; Coker, A.L.; Du, X.L.; Eggleston, K.S.; Williams, M. Racial/ethnic disparities in survival among men diagnosed with prostate cancer in Texas. Cancer 2011. [Google Scholar] [CrossRef] [PubMed]
- Ripple, M.O.; Henry, W.F.; Rago, R.P.; Wilding, G. Prooxidant-antioxidant shift induced by androgen treatment of human prostate carcinoma cells. J. Natl. Cancer Inst. 1997. [Google Scholar] [CrossRef]
- Jones, R.A.; Underwood, S.M.; Rivers, B.M. Reducing prostate cancer morbidity and mortality in African American men: Issues and challenges. Clin. J. Oncol. Nurs. 2007. [Google Scholar] [CrossRef]
- Society, A.C. American Cancer Society. Cancer facts and figures 2007. Am. Cancer Soc. 2007. [Google Scholar] [CrossRef]
- Morris, M.J.; Basch, E.M.; Wilding, G.; Hussain, M.; Carducci, M.A.; Higano, C.; Kantoff, P.; Oh, W.K.; Small, E.J.; George, D.; et al. Department of Defense prostate cancer clinical trials consortium: A new instrument for prostate cancer clinical research. Clin. Genitourin. Cancer 2009. [Google Scholar] [CrossRef] [PubMed]
- Facts, C. Cancer Facts & Figures 2013. Atlanta: American Cancer Society; 2013. Am. Cancer Soc. 2013. [Google Scholar] [CrossRef]
- Arias, E. United States life Tables, 2007. National Vital Statistics Reports; Vol 59 No 9; Natl. Vital Stat. Reports; National Center for Health Statistics: Hyattsville, MD, USA, 2011.
- Desai, N.; Sabanegh, E.; Kim, T.; Agarwal, A. Free Radical Theory of Aging: Implications in Male Infertility. Urology 2010. [Google Scholar] [CrossRef] [PubMed]
- Paschos, A.; Pandya, R.; Duivenvoorden, W.C.M.; Pinthus, J.H. Oxidative stress in prostate cancer: Changing research concepts towards a novel paradigm for prevention and therapeutics. Prostate Cancer Prostatic Dis. 2013. [Google Scholar] [CrossRef]
- Szewczyk-Golec, K.; Tyloch, J.; Czuczejko, J. Antioxidant defense system in prostate adenocarcinoma and benign prostate hyperplasia of elderly patients. Neoplasma 2015. [Google Scholar] [CrossRef]
- Aggarwal, B.B.; Harikumar, K.B. Potential therapeutic effects of curcumin, the anti-inflammatory agent, against neurodegenerative, cardiovascular, pulmonary, metabolic, autoimmune and neoplastic diseases. Int. J. Biochem. Cell Biol. 2009, 41, 40–59. [Google Scholar] [CrossRef] [PubMed]
- Teiten, M.H.; Gaascht, F.; Eifes, S.; Dicato, M.; Diederich, M. Chemopreventive potential of curcumin in prostate cancer. Genes Nutr. 2010, 5, 61–74. [Google Scholar] [CrossRef]
- Shanmugam, M.K.; Rane, G.; Kanchi, M.M.; Arfuso, F.; Chinnathambi, A.; Zayed, M.E.; Alharbi, S.A.; Tan, B.K.H.; Kumar, A.P.; Sethi, G. The multifaceted role of curcumin in cancer prevention and treatment. Molecules 2015, 20, 2728–2769. [Google Scholar] [CrossRef]
- Dorai, T.; Gehani, N.; Katz, A. Therapeutic potential of curcumin in human prostate cancer—I. curcumin induces apoptosis in both androgen-dependent and androgen-independent prostate cancer cells. Prostate Cancer Prostatic Dis. 2000, 3, 84–93. [Google Scholar] [CrossRef]
- Deng, G.; Yu, J.H.; Ye, Z.Q.; Hu, Z.Q. Curcumin inhibits the expression of vascular endothelial growth factor and androgen-independent prostate cancer cell line PC-3 in vitro. Zhonghua Nan Ke Xue 2008, 14, 116–121. [Google Scholar]
- Shankar, S.; Ganapathy, S.; Chen, Q.; Srivastava, R.K. Curcumin sensitizes TRAIL-resistant xenografts: Molecular mechanisms of apoptosis, metastasis and angiogenesis. Mol. Cancer 2008, 7. [Google Scholar] [CrossRef] [PubMed]
- Dorai, T.; Cao, Y.C.; Dorai, B.; Buttyan, R.; Katz, A.E. Therapeutic potential of curcumin in human prostate cancer. III. Curcumin inhibits proliferation, induces apoptosis, and inhibits angiogenesis of LNCaP prostate cancer cells in vivo. Prostate 2001, 47, 293–303. [Google Scholar] [CrossRef] [PubMed]
- Deeb, D.; Jiang, H.; Gao, X.; Hafner, M.S.; Wong, H.; Divine, G.; Chapman, R.A.; Dulchavsky, S.A.; Gautam, S.C. Curcumin sensitizes prostate cancer cells to tumor necrosis factor-related apoptosis-inducing ligand/Apo2L by inhibiting nuclear factor-kappaB through suppression of IkappaBalpha phosphorylation. Mol. Cancer Ther. 2004, 3, 803–812. [Google Scholar] [PubMed]
- Dorai, T.; Dutcher, J.P.; Dempster, D.W.; Wiernik, P.H. Therapeutic potential of curcumin in prostate cancer - IV: Interference with the osteomimetic properties of hormone refractory C4-2B prostate cancer cells. Prostate 2004, 60, 1–17. [Google Scholar] [CrossRef]
- Janane, D.; Orla, O.; Stephen B, D. Curcumin Inhibits Protate Cancer Bone Metastasis by Up-Regulating Bone Morphogenic Protein-7 in Vivo. J. Cancer Ther 2014, 5, 369–386. [Google Scholar]
- Yang, J.; Ning, J.; Peng, L.; He, D. Effect of curcumin on Bcl-2 and Bax expression in nude mice prostate cancer. Int. J. Clin. Exp. Pathol. 2015, 8, 9272–9278. [Google Scholar]
- Yang, J.; Wang, C.; Zhang, Z.; Chen, X.; Jia, Y.; Wang, B.; Kong, T. Curcumin inhibits the survival and metastasis of prostate cancer cells via the Notch-1 signaling pathway. APMIS 2017, 125, 134–140. [Google Scholar] [CrossRef]
- Du, Y.; Long, Q.; Zhang, L.; Shi, Y.; Liu, X.; Li, X.; Guan, B.; Tian, Y.; Wang, X.; Li, L.; et al. Curcumin inhibits cancer-associated fibroblast-driven prostate cancer invasion through MAOA/mTOR/HIF-1alpha signaling. Int. J. Oncol. 2015, 47, 2064–2072. [Google Scholar] [CrossRef]
- Hong, J.H.; Ahn, K.S.; Bae, E.; Jeon, S.S.; Choi, H.Y. The effects of curcumin on the invasiveness of prostate cancer in vitro and in vivo. Prostate Cancer Prostatic Dis. 2006, 9, 147–152. [Google Scholar] [CrossRef]
- Jordan, B.C.; Mock, C.D.; Thilagavathi, R.; Selvam, C. Molecular mechanisms of curcumin and its semisynthetic analogues in prostate cancer prevention and treatment. Life Sci. 2016. [Google Scholar] [CrossRef]
- Zhou, D.Y.; Zhao, S.Q.; Du, Z.Y.; Zheng, X.; Zhang, K. Pyridine analogues of curcumin exhibit high activity for inhibiting CWR-22Rv1 human prostate cancer cell growth and androgen receptor activation. Oncol. Lett. 2016. [Google Scholar] [CrossRef] [PubMed]
- Wei, X.; Du, Z.Y.; Cui, X.X.; Verano, M.; Mo, R.Q.; Tang, Z.K.; Conney, A.H.; Zheng, X.; Zhang, K. Effects of cyclohexanone analogues of curcumin on growth, apoptosis and NF-κB activity in PC-3 human prostate cancer cells. Oncol. Lett. 2012. [Google Scholar] [CrossRef] [PubMed]
- Muniyan, S.; Chou, Y.W.; Ingersoll, M.A.; Devine, A.; Morris, M.; Odero-Marah, V.A.; Khan, S.A.; Chaney, W.G.; Bu, X.R.; Lin, M.F. Antiproliferative activity of novel imidazopyridine derivatives on castration-resistant human prostate cancer cells. Cancer Lett. 2014. [Google Scholar] [CrossRef] [PubMed]
- Joseph, J.D.; Lu, N.; Qian, J.; Sensintaffar, J.; Shao, G.; Brigham, D.; Moon, M.; Maneval, E.C.; Chen, I.; Darimont, B.; et al. A clinically relevant androgen receptor mutation confers resistance to second-generation antiandrogens enzalutamide and ARN-509. Cancer Discov. 2013. [Google Scholar] [CrossRef] [PubMed]
- Hussain, M.; Corn, P.G.; Michaelson, M.D.; Hammers, H.J.; Alumkal, J.J.; Ryan, C.J.; Bruce, J.Y.; Moran, S.; Lee, S.Y.; Lin, H.M.; et al. Phase II study of single-agent orteronel (TAK-700) in patients with nonmetastatic castration-resistant prostate cancer and rising prostate-specific antigen. Clin. Cancer Res. 2014. [Google Scholar] [CrossRef] [PubMed]
- Köhler, A.; Demir, Ü.; Kickstein, E.; Krauss, S.; Aigner, J.; Aranda-Orgillés, B.; Karagiannidis, A.I.; Achmüller, C.; Bu, H.; Wunderlich, A.; et al. A hormone-dependent feedback-loop controls androgen receptor levels by limiting MID1, a novel translation enhancer and promoter of oncogenic signaling. Mol. Cancer 2014. [Google Scholar] [CrossRef] [PubMed]
- Dorai, T.; Gehani, N.; Katz, A. Therapeutic potential of curcumin in human prostate cancer. II. Curcumin inhibits tyrosine kinase activity of epidermal growth factor receptor and depletes the protein. Mol. Urol. 2000, 4, 1–6. [Google Scholar] [PubMed]
- Nakamura, K.; Yasunaga, Y.; Segawa, T.; Ko, D.; Moul, J.W.; Srivastava, S.; Rhim, J.S. Curcumin down-regulates AR gene expression and activation in prostate cancer cell lines. Int. J. Oncol. 2002, 21, 825–830. [Google Scholar] [CrossRef]
- Tsui, K.-H.; Feng, T.-H.; Lin, C.-M.; Chang, P.-L.; Juang, H.-H. Curcumin blocks the activation of androgen and interlukin-6 on prostate-specific antigen expression in human prostatic carcinoma cells. J. Androl. 2008, 29, 661–668. [Google Scholar] [CrossRef]
- Mukhopadhyay, A.; Bueso-Ramos, C.; Chatterjee, D.; Pantazis, P.; Aggarwal, B.B. Curcumin downregulates cell survival mechanisms in human prostate cancer cell lines. Oncogene 2001, 20, 7597–7609. [Google Scholar] [CrossRef]
- Mukhopadhyay, A.; Banerjee, S.; Stafford, L.J.; Xia, C.; Liu, M.; Aggarwal, B.B. Curcumin-induced suppression of cell proliferation correlates with down-regulation of cyclin D1 expression and CDK4-mediated retinoblastoma protein phosphorylation. Oncogene 2002, 21, 8852–8861. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, R.K.; Chen, Q.; Siddiqui, I.; Sarva, K.; Shankar, S. Linkage of curcumin-induced cell cycle arrest and apoptosis by cyclin-dependent kinase inhibitor p21/WAF1/CIP1. Cell Cycle 2007, 6, 2953–2961. [Google Scholar] [CrossRef] [PubMed]
- Killian, P.H.; Kronski, E.; Michalik, K.M.; Barbieri, O.; Astigiano, S.; Sommerhoff, C.P.; Pfeffer, U.; Nerlich, A.G.; Bachmeier, B.E. Curcumin inhibits prostate cancer metastasis in vivo by targeting the inflammatory cytokines CXCL1 and -2. Carcinogenesis 2012, 33, 2507–2519. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.C.; Patchva, S.; Aggarwal, B.B. Therapeutic Roles of Curcumin: Lessons Learned from Clinical Trials. AAPS J. 2013, 15, 195–218. [Google Scholar] [CrossRef] [PubMed]
- Ide, H.; Tokiwa, S.; Sakamaki, K.; Nishio, K.; Isotani, S.; Muto, S.; Hama, T.; Masuda, H.; Horie, S. Combined inhibitory effects of soy isoflavones and curcumin on the production of prostate-specific antigen. Prostate 2010, 70, 1127–1133. [Google Scholar] [CrossRef] [PubMed]
- Hsing, A.W.; Chokkalingam, A.P.; Gao, Y.-T.; Madigan, M.P.; Deng, J.; Gridley, G.; Fraumeni, J.F. Allium vegetables and risk of prostate cancer: A population-based study. J. Natl. Cancer Inst. 2002, 94, 1648–1651. [Google Scholar] [CrossRef] [PubMed]
- Xiao, D.; Lew, K.L.; Kim, Y.A.; Zeng, Y.; Hahm, E.R.; Dhir, R.; Singh, S.V. Diallyl trisulfide suppresses growth of PC-3 human prostate cancer xenograft in vivo in association with Bax and Bak induction. Clin. Cancer Res. 2006, 12, 6836–6843. [Google Scholar] [CrossRef] [PubMed]
- Petropoulos, S.A.; Fernandes, Â.; Ntatsi, G.; Petrotos, K.; Barros, L.; Ferreira, I.C.F.R. Nutritional value, chemical characterization and bulb morphology of Greek Garlic landraces. Molecules 2018, 23, 319. [Google Scholar] [CrossRef]
- Rasul Suleria, H.A.; Sadiq Butt, M.; Muhammad Anjum, F.; Saeed, F.; Batool, R.; Nisar Ahmad, A. Aqueous garlic extract and its phytochemical profile; Special reference to antioxidant status. Int. J. Food Sci. Nutr. 2012, 63, 431–439. [Google Scholar] [CrossRef]
- Thomson, M.; Ali, M. Garlic [Allium sativum]: A review of its potential use as an anti-cancer agent. Curr. Cancer Drug Targets 2003, 3, 67–81. [Google Scholar] [CrossRef]
- Herman-Antosiewicz, A.; Kim, Y.-A.; Kim, S.-H.; Xiao, D.; Singh, S. V Diallyl trisulfide-induced G2/M phase cell cycle arrest in DU145 cells is associated with delayed nuclear translocation of cyclin-dependent kinase 1. Pharm. Res. 2010, 27, 1072–1079. [Google Scholar] [CrossRef] [PubMed]
- Xiao, D.; Singh, S.V. Diallyl trisulfide, a constituent of processed garlic, inactivates Akt to trigger mitochondrial translocation of BAD and caspase-mediated apoptosis in human prostate cancer cells. Carcinogenesis 2006, 27, 533–540. [Google Scholar] [CrossRef] [PubMed]
- Chandra-Kuntal, K.; Singh, S.V. Diallyl trisulfide inhibits activation of signal transducer and activator of transcription 3 in prostate cancer cells in culture and In vivo. Cancer Prev. Res. 2010, 3, 1473–1483. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.H.; Bommareddy, A.; Singh, S.V. Garlic constituent diallyl trisulfide suppresses X-linked inhibitor of apoptosis protein in prostate cancer cells in culture and in vivo. Cancer Prev. Res. 2011, 4, 897–906. [Google Scholar] [CrossRef] [PubMed]
- Yedjou, C.G.; Tchounwou, P.B. In vitro assessment of oxidative stress and apoptotic mechanisms of garlic extract in the treatment of acute promyelocytic leukemia. J. Cancer Sci. Ther. 2012, 2012, 6. [Google Scholar] [CrossRef] [PubMed]
- Yedjou, C.; Izevbigie, E.; Tchounwou, P. Preclinical assessment of Vernonia amygdalina leaf extracts as DNA damaging anti-cancer agent in the management of breast cancer. Int. J. Environ. Res. Public Health 2008, 5, 337–341. [Google Scholar] [CrossRef]
- Erasto, P.; Grierson, D.S.; Afolayan, A.J. Evaluation of antioxidant activity and the fatty acid profile of the leaves of Vernonia amygdalina growing in South Africa. Food Chem. 2007, 104, 636–642. [Google Scholar] [CrossRef]
- Nwanjo, H.U. Efficacy of aqueous leaf extract of vernonia amygdalina on plasma lipoprotein and oxidative status in diabetic rat models. Niger. J. Physiol. Sci. 2005, 20, 39–42. [Google Scholar]
- Adaramoye, O.A.; Akintayo, O.; Achem, J.; Fafunso, M.A. Lipid-lowering effects of methanolic extract of Vernonia amygdalina leaves in rats fed on high cholesterol diet. Vasc. Health Risk Manag. 2008, 4, 235–241. [Google Scholar] [CrossRef]
- Erasto, P.; Grierson, D.S.; Afolayan, A.J. Bioactive sesquiterpene lactones from the leaves of Vernonia amygdalina. J. Ethnopharmacol. 2006, 106, 117–120. [Google Scholar] [CrossRef]
- Tona, L.; Cimanga, R.K.; Mesia, K.; Musuamba, C.T.; De Bruyne, T.; Apers, S.; Hernans, N.; Van Miert, S.; Pieters, L.; Totté, J.; et al. In vitro antiplasmodial activity of extracts and fractions from seven medicinal plants used in the Democratic Republic of Congo. J. Ethnopharmacol. 2004, 93. [Google Scholar] [CrossRef] [PubMed]
- Khlebnikov, A.I.; Schepetkin, I.A.; Domina, N.G.; Kirpotina, L.N.; Quinn, M.T. Improved quantitative structure-activity relationship models to predict antioxidant activity of flavonoids in chemical, enzymatic, and cellular systems. Bioorg. Med. Chem. 2007, 15, 1749–1770. [Google Scholar] [CrossRef] [PubMed]
- Johnson, W.; Tchounwou, P.B.; Yedjou, C.G. Therapeutic mechanisms of vernonia amygdalina delile in the treatment of prostate cancer. Molecules 2017, 22, 1594. [Google Scholar] [CrossRef]
- Lowe, H.I.C.; Daley-Beckford, D.; Toyang, N.J.; Watson, C.; Hartley, S.; Bryant, J. The anti-cancer activity of Vernonia divaricata Sw against leukaemia, breast and prostate cancers in vitro. West. Indian Med. J. 2014. [Google Scholar] [CrossRef] [PubMed]
- Izevbigie, E.B. Discovery of water-soluble anticancer agents (edotides) from a vegetable found in Benin City, Nigeria. Exp. Biol. Med. (Maywood). 2003, 228, 293–298. [Google Scholar] [CrossRef] [PubMed]
| Edible Medicinal Plants | Medicinal Uses | Pre-Clinical Studies | Clinical Studies | Summary & References |
|---|---|---|---|---|
| Curcumin | People use curcumin to prevent and/or treat cancer, Alzheimer, erectile dysfunction, baldness, hirsutism, and fertility | Inhibition of proinflammatory NF-B, reduction of prostate cancer growth, induction of apoptosis | Clinical trials needed | Curcumin has demonstrated the potential to slow growth and kill prostate tumor cells References [98,99,100,101,103,115]. |
| Garlic (Allium sativum) | People use garlic to prevent and/or treat many conditions including prostate cancer, breast cancer, rectal cancer, stomach cancer, colon cancer, yeast infection, high blood pressure, hepatitis, and diabetes. | Cell growth arrest, cell cycle arrest, DNA damage, protein expression disruption, and induction of apoptosis | Few clinical trials have been performed, but they are still inconsistent. Recognition of garlic as one of the vegetables with potential anti-cancer properties | High consumption of garlic lowers the risk of prostate cancer. Sulfur present in garlic neutralizes cancer cells and shrinks the tumor References [120,121,126,128]. |
| Vernonia amygdalina | People use Vernonia amygdalina to prevent and/or treat fever, hiccups, kidney problem, gastritis, enteritis, malaria, and rheumatism | Cell growth arrest, DNA damage, and induction of apoptosis as evidenced of phosphatidylserine externalization, activation of caspase-3, and cellular morphological changes. | Clinical trials needed | Limited scientific evidence. May act both as antioxidant and pro-oxidant depending on the dose. Further research needed. References [137,139]. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yedjou, C.G.; Mbemi, A.T.; Noubissi, F.; Tchounwou, S.S.; Tsabang, N.; Payton, M.; Miele, L.; Tchounwou, P.B. Prostate Cancer Disparity, Chemoprevention, and Treatment by Specific Medicinal Plants. Nutrients 2019, 11, 336. https://doi.org/10.3390/nu11020336
Yedjou CG, Mbemi AT, Noubissi F, Tchounwou SS, Tsabang N, Payton M, Miele L, Tchounwou PB. Prostate Cancer Disparity, Chemoprevention, and Treatment by Specific Medicinal Plants. Nutrients. 2019; 11(2):336. https://doi.org/10.3390/nu11020336
Chicago/Turabian StyleYedjou, Clement G., Ariane T. Mbemi, Felicite Noubissi, Solange S. Tchounwou, Nole Tsabang, Marinelle Payton, Lucio Miele, and Paul B. Tchounwou. 2019. "Prostate Cancer Disparity, Chemoprevention, and Treatment by Specific Medicinal Plants" Nutrients 11, no. 2: 336. https://doi.org/10.3390/nu11020336
APA StyleYedjou, C. G., Mbemi, A. T., Noubissi, F., Tchounwou, S. S., Tsabang, N., Payton, M., Miele, L., & Tchounwou, P. B. (2019). Prostate Cancer Disparity, Chemoprevention, and Treatment by Specific Medicinal Plants. Nutrients, 11(2), 336. https://doi.org/10.3390/nu11020336






