Association of Strawberries and Anthocyanidin Intake with Alzheimer’s Dementia Risk
Abstract
1. Introduction:
2. Material and Methods
2.1. Study Population
2.2. Alzheimer’s Dementia
2.3. Diet Assessment
2.4. Covariates
2.5. Statistical Methods
3. Results
3.1. Strawberries and Alzheimer’s Dementia Risk
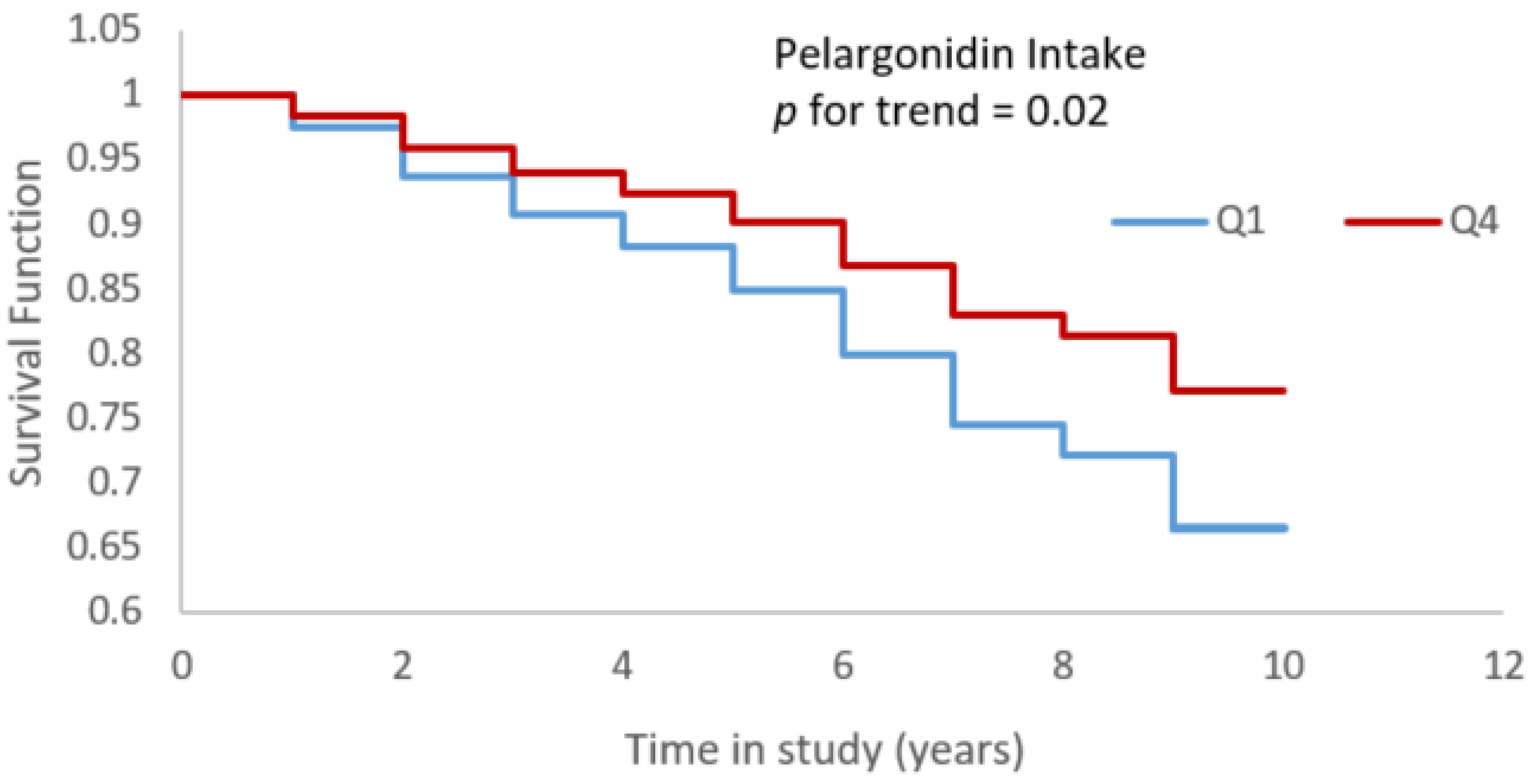
3.2. Bioactives and Alzheimer’s Dementia Risk
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wilson, R.S.; Boyle, P.A.; Segawa, E.; Yu, L.; Begeny, C.T.; Anagnos, S.E.; Bennett, D.A. The influence of cognitive decline on well-being in old age. Psychol. Aging 2013, 28, 304–313. [Google Scholar] [CrossRef] [PubMed]
- Wilson, R.S.; Schneider, J.A.; Bienias, J.L.; Evans, D.A.; Bennett, D.A. Parkinsonianlike signs and risk of incident Alzheimer disease in older persons. Arch. Neurol. 2003, 60, 539–544. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, N.T.; Wilson, R.S.; Beck, T.L.; Bienias, J.L.; Bennett, D.A. Motor dysfunction in mild cognitive impairment and the risk of incident Alzheimer disease. Arch. Neurol. 2006, 63, 1763–1769. [Google Scholar] [CrossRef] [PubMed]
- Sopher, B.L.; Fukuchi, K.; Kavanagh, T.J.; Furlong, C.E.; Martin, G.M. Neurodegenerative mechanisms in Alzheimer disease. A role for oxidative damage in amyloid beta protein precursor-mediated cell death. Mol. Chem. Neuropathol. 1996, 29, 153–168. [Google Scholar] [CrossRef] [PubMed]
- Shukitt-Hale, B. The effects of aging and oxidative stress on psychomotor and cognitive behavior. Age 1999, 22, 9–17. [Google Scholar] [CrossRef]
- Hauss-Wegrzyniak, B.; Vannucchi, M.G.; Wenk, G.L. Behavioral and ultrastructural changes induced by chronic neuroinflammation in young rats. Brain Res. 2000, 859, 157–166. [Google Scholar] [CrossRef]
- Morris, M.C.; Evans, D.A.; Bienias, J.L.; Tangney, C.C.; Bennett, D.A.; Aggarwal, N.; Wilson, R.S.; Scherr, P.A. Dietary intake of antioxidant nutrients and the risk of incident Alzheimer disease in a biracial community study. JAMA 2002, 287, 3230–3237. [Google Scholar] [CrossRef]
- Luchsinger, J.A.; Tang, M.X.; Shea, S.; Mayeux, R. Antioxidant vitamin intake and risk of Alzheimer disease. Arch. Neurol. 2003, 60, 203–208. [Google Scholar] [CrossRef]
- Commenges, D.; Scotet, V.; Renaud, S.; Jacqmin-Gadda, H.; Barberger-Gateau, P.; Dartigues, J.F. Intake of flavonoids and risk of dementia. Eur. J. Epidemiol. 2000, 16, 357–363. [Google Scholar] [CrossRef]
- Engelhart, M.J.; Geerlings, M.I.; Ruitenberg, A.; van Swieten, J.C.; Hofman, A.; Witteman, J.C.; Breteler, M.M. Dietary intake of antioxidants and risk of Alzheimer disease. JAMA 2002, 287, 3223–3229. [Google Scholar] [CrossRef]
- Stevenson, D.E.; Hurst, R.D. Polyphenolic phytochemicals--just antioxidants or much more? Cell. Mol. Life Sci. 2007, 64, 2900–2916. [Google Scholar] [CrossRef] [PubMed]
- Shukitt-Hale, B.; Bielinski, D.F.; Lau, F.C.; Willis, L.M.; Carey, A.N.; Joseph, J.A. The beneficial effects of berries on cognition, motor behaviour and neuronal function in ageing. Br. J. Nutr. 2015, 114, 1542–1549. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, A.; Salo, I.; Plaza, M.; Bjorck, I. Effects of a mixed berry beverage on cognitive functions and cardiometabolic risk markers; A randomized cross-over study in healthy older adults. PLoS ONE 2017, 12, e0188173. [Google Scholar] [CrossRef] [PubMed]
- Joseph, J.A.; Denisova, N.A.; Prior, R.L.; Cao, G.; Martin, A.; Taglialatela, G.; Bickford, P.C. Long-Term Dietary Strawberry, Spinach, or Vitamin E Supplementation Retards the Onset of Age-Related Neuronal Signal-Transduction and Cognitive Behavioral Deficit. J. Neurosci. 1998, 1, 8047–8055. [Google Scholar] [CrossRef]
- Shukitt-Hale, B.; Smith, D.E.; Meydani, M.; Joseph, J.A. The effects of dietary antioxidants on psychomotor performance in aged mice. Exp. Gerontol. 1999, 34, 797–808. [Google Scholar] [CrossRef]
- Bickford, P.C.; Gould, T.; Briederick, L.; Chadman, K.; Pollock, A.; Young, D.; Shukitt-Hale, B.; Joseph, J. Antioxidant-rich diets improve cerebellar physiology and motor learning in aged rats. Brain Res. 2000, 866, 211–217. [Google Scholar] [CrossRef]
- Devore, E.E.; Kang, J.H.; Breteler, M.M.; Grodstein, F. Dietary intakes of berries and flavonoids in relation to cognitive decline. Ann. Neurol. 2012, 72, 135–143. [Google Scholar] [CrossRef]
- Bennett, D.A.; Schneider, J.A.; Buchman, A.S.; Barnes, L.L.; Boyle, P.A.; Wilson, R.S. Overview and findings from the rush Memory and Aging Project. Curr. Alzheimer Res. 2012, 9, 646–663. [Google Scholar] [CrossRef]
- Bennett, D.A.; Schneider, J.A.; Arvanitakis, Z.; Kelly, J.F.; Aggarwal, N.T.; Shah, R.C.; Wilson, R.S. Neuropathology of older persons without cognitive impairment from two community-based studies. Neurology 2006, 66, 1837–1844. [Google Scholar] [CrossRef]
- Group TN-RW. Consensus recommendations for the postmortem diagnosis of Alzheimer’s disease. The National Institute on Aging, and Reagan Institute Working Group on Diagnostic Criteria for the Neuropathological Assessment of Alzheimer’s Disease. Neurobiol. Aging 1997, 18, S1–S2. [Google Scholar]
- Morris, M.C. Validity and Reproducibility of a Food Frequency Questionnaire by Cognition in an Older Biracial Sample. Am. J. Epidemiol. 2003, 158, 1213–1217. [Google Scholar] [CrossRef] [PubMed]
- Willett, W.C.; Sampson, L.; Stampfer, M.J.; Rosner, B.; Bain, C.; Witschi, J.; Hennekens, C.H.; Speizer, F.E. Reproducibility and validity of a semiquantitative food frequency questionnaire. Am. J. Epidemiol. 1985, 122, 51–65. [Google Scholar] [CrossRef] [PubMed]
- USDA. Nutrient Database for Standard Reference; Release 26; US Department Agriculture: Washington, DC, USA, 2013.
- University of Minnesota. University of Minnesota Nutrition Coordinating Center Flavonoid and Proanthocynidin Provisional Table; University of Minnesota: Williamson Hall, MN, USA, 2018. [Google Scholar]
- Yu, L.; Lutz, M.W.; Wilson, R.S.; Burns, D.K.; Roses, A.D.; Saunders, A.M.; Gaiteri, C.; Ger, P.L.D.; Barnes, L.L.; Bennett, D.A. TOMM40’523 variant and cognitive decline in older persons with APOE epsilon3/3 genotype. Neurology 2017, 88, 661–668. [Google Scholar] [CrossRef] [PubMed]
- Wilson, R.S.; Segawa, E.; Boyle, P.A.; Bennett, D.A. Influence of late-life cognitive activity on cognitive health. Neurology 2012, 78, 1123–1129. [Google Scholar] [CrossRef] [PubMed]
- Buchman, A.S.; Boyle, P.A.; Wilson, R.S.; Bienias, J.L.; Bennett, D.A. Physical activity and motor decline in older persons. Muscle Nerve 2007, 35, 354–362. [Google Scholar] [CrossRef] [PubMed]
- Bennett, D.A.; Buchman, A.S.; Boyle, P.A.; Barnes, L.L.; Wilson, R.S.; Schneider, J.A. Religious Orders Study and Rush Memory and Aging Project. J. Alzheimer’s Dis. 2018, 64, S161–S189. [Google Scholar] [CrossRef] [PubMed]
- Kelly, A.; Calamia, M.; Koval, A.; Terrera, G.M.; Piccinin, A.M.; Clouston, S.; Hassing, L.B.; Bennett, D.A.; Johansson, B.; Hofer, S.M. Independent and interactive impacts of hypertension and diabetes mellitus on verbal memory: A coordinated analysis of longitudinal data from England, Sweden, and the United States. Psychol. Aging 2016, 31, 262–273. [Google Scholar] [CrossRef]
- Bennett, D.A. Secular trends in stroke incidence and survival, and the occurrence of dementia. Stroke 2006, 37, 1144–1145. [Google Scholar] [CrossRef]
- Willett, W.; Stampfer, M.J. Total energy intake: Implications for epidemiologic analyses. Am. J. Epidemiol. 1986, 124, 17–27. [Google Scholar] [CrossRef]
- Letenneur, L.; Proust-Lima, C.; Le Gouge, A.; Dartigues, J.F.; Barberger-Gateau, P. Flavonoid intake and cognitive decline over a 10-year period. Am. J. Epidemiol. 2007, 165, 1364–1371. [Google Scholar] [CrossRef]
- Basambombo, L.L.; Carmichael, P.H.; Cote, S.; Laurin, D. Use of Vitamin E and C Supplements for the Prevention of Cognitive Decline. Ann. Pharmacother. 2017, 51, 118–124. [Google Scholar] [CrossRef] [PubMed]
- Elkhadragy, M.F.; Kassab, R.B.; Metwally, D.; Almeer, R.S.; Abdel-Gaber, R.; Al-Olayan, E.M.; Essawy, E.A.; Amin, H.K.; Abdel Moneim, A.E. Protective effects of Fragaria ananassa methanolic extract in a rat model of cadmium chloride-induced neurotoxicity. Biosci. Rep. 2018, 38. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Suarez, J.M.; Giampieri, F.; Tulipani, S.; Casoli, T.; Di Stefano, G.; González-Paramás, A.M.; Santos-Buelga, C.; Busco, F.; Quiles, J.L.; Cordero, M.D.; et al. One-month strawberry-rich anthocyanin supplementation ameliorates cardiovascular risk, oxidative stress markers and platelet activation in humans. J. Nutr. Biochem. 2014, 25, 289–294. [Google Scholar] [CrossRef] [PubMed]
- Ellis, C.L.; Edirisinghe, I.; Kappagoda, T.; Burton-Freeman, B. Attenuation of meal-induced inflammatory and thrombotic responses in overweight men and women after 6-week daily strawberry (Fragaria) intake. A randomized placebo-controlled trial. J. Atheroscler. Thromb. 2011, 18, 318–327. [Google Scholar] [CrossRef]
- Giampieri, F.; Alvarez-Suarez, J.M.; Mazzoni, L.; Forbes-Hernandez, T.Y.; Gasparrini, M.; Gonzàlez-Paramàs, A.M.; Santos-Buelga, C.; Quiles, J.L.; Bompadre, S.; Mezzetti, B.; et al. Polyphenol-rich strawberry extract protects human dermal fibroblasts against hydrogen peroxide oxidative damage and improves mitochondrial functionality. Molecules 2014, 19, 7798–7816. [Google Scholar] [CrossRef]
- Youdim, K.A.; Dobbie, M.S.; Kuhnle, G.; Proteggente, A.R.; Abbott, N.J.; Rice-Evans, C. Interaction between flavonoids and the blood-brain barrier: In vitro studies. J. Neurochem. 2003, 85, 180–192. [Google Scholar] [CrossRef]
- Youdim, K.A.; Qaiser, M.Z.; Begley, D.J.; Rice-Evans, C.A.; Abbott, N.J. Flavonoid permeability across an in situ model of the blood-brain barrier. Free Radic. Biol. Med. 2004, 36, 592–604. [Google Scholar] [CrossRef]
- Jaeger, B.N.; Parylak, S.L.; Gage, F.H. Mechanisms of dietary flavonoid action in neuronal function and neuroinflammation. Mol. Asp. Med. 2018, 61, 50–62. [Google Scholar] [CrossRef]
- Ali, T.; Kim, T.; Rehman, S.U.; Khan, M.S.; Amin, F.U.; Khan, M.; Ikram, M.; Kim, M.O. Natural Dietary Supplementation of Anthocyanins via PI3K/Akt/Nrf2/HO-1 Pathways Mitigate Oxidative Stress, Neurodegeneration, and Memory Impairment in a Mouse Model of Alzheimer’s Disease. Mol. Neurobiol. 2018, 55, 6076–6093. [Google Scholar] [CrossRef]
- Sohanaki, H.; Baluchnejadmojarad, T.; Nikbakht, F.; Roghani, M. Pelargonidin improves memory deficit in amyloid beta25-35 rat model of Alzheimer’s disease by inhibition of glial activation, cholinesterase, and oxidative stress. Biomed. Pharmacother. 2016, 83, 85–91. [Google Scholar] [CrossRef]
- Sandhu, A.K.; Miller, M.G.; Thangthaeng, N.; Scott, T.M.; Shukitt-Hale, B.; Edirisinghe, I.; Burton-Freeman, B. Metabolic fate of strawberry polyphenols after chronic intake in healthy older adults. Food Funct. 2018, 9, 96–106. [Google Scholar] [CrossRef] [PubMed]
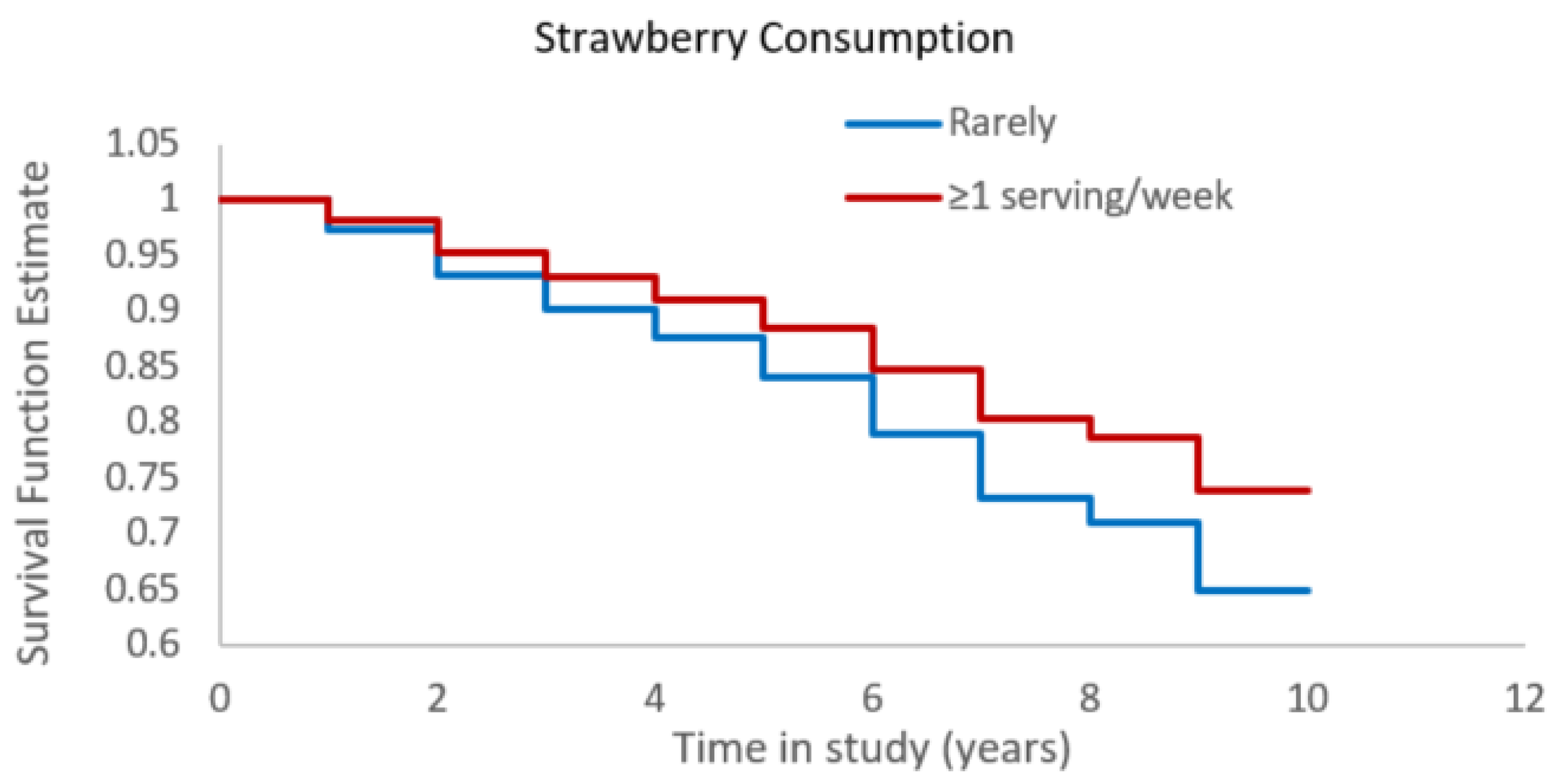
| Total (n = 925) | Strawberry Consumption | |||
|---|---|---|---|---|
| Rarely n = 243 | 1–3 Times/Month n = 428 | >= Once/Week n = 254 | ||
| Age, y, mean | 81.16 ± 7.2 | 81.9 ± 7.1 | 81.2 ± 7.03 | 80.4 ± 7.4 |
| Female, % | 75% | 71% | 77% | 75% |
| Education, y, mean | 14.9 ± 2.9 | 14.6 ± 3.0 | 15.1 ± 3.1 | 15.0 ± 2.6 |
| APOE-ɛ4 status, % | 21.5% | 23% | 21% | 21% |
| Cognitive activities, mean frequency | 3.2 ± 0.6 | 3.1 ± 0.6 | 3.2 ± 0.7 | 3.3 ± 0.6 |
| Physical activities, mean hours/week | 3.4 ± 3.6 | 2.9 ± 3.6 | 3.3 ± 3.6 | 3.9 ± 3.8 |
| Other Fruits Intake, servings/week | 14.3 ± 7.3 | 12.9 ± 7.0 | 13.8 ± 7.0 | 16.4 ± 7.8 |
| Total calories, kcal/day | 1733 ± 539 | 1628 ± 555 | 1729 ± 518 | 1843 ± 538 |
| Total flavonoid intake (mg/day) | 223.2 ± 142.2 | 204.3 ± 150.0 | 218.0 ± 140.0 | 250.0 ± 135.0 |
| Total vitamin E intake (mg/day) | 71.3 ± 108.0 | 64.5 ± 99.0 | 76.6 ± 122.0 | 68.7 ± 89.4 |
| Cardiovascular conditions, % present | ||||
| Hypertension | 69% | 69% | 70% | 68% |
| Myocardial Infarction | 16% | 18% | 15% | 15% |
| Diabetes | 13% | 12% | 13.5% | 13% |
| Stroke | 11.5% | 13% | 11% | 11% |
| Strawberry and Nutrients | Model 1: Basic (n = 925) | Model 2: Basic + Total vitamin E intake’ (n = 925) | |
|---|---|---|---|
| Strawberry Intake | |||
| Mean/Median Intake | HR (95% CI) | ||
| Continuous variable * | 0.64 servings/week | 0.76 (0.60, 0.96) | 0.76 (0.61, 0.96) |
| Quartile Intake of Vitamin C and Flavonoids from Food Sources ** | |||
| Vitamin C Intake | |||
| 1 | 56.0 mg/day | Ref | Ref |
| 2 | 98.4 mg/day | 0.84 (0.59, 1.20) | 0.85 (0.60, 1.22) |
| 3 | 135.8 mg/day | 0.77 (0.53, 1.10) | 0.79 (0.55, 1.14) |
| 4 | 199.5 mg/day | 0.64 (0.45, 0.92) | 0.67 (0.47, 0.97) |
| p for trend | 0.01 | 0.03 | |
| Pelargonidin Intake | |||
| 1 | 0.00 mg/day | Ref | Ref |
| 2 | 0.89 mg/day | 0.84 (0.60, 1.19) | 0.83 (0.59, 1.17) |
| 3 | 1.73 mg/day | 0.78 (0.52, 1.04) | 0.75 (0.53, 1.06) |
| 4 | 3.90 mg/day | 0.63 (0.43, 0.92) | 0.65 (0.44, 0.94) |
| p for trend | 0.02 | 0.02 | |
| Cyanidin Intake | |||
| 1 | 0.30 mg/day | Ref | Ref |
| 2 | 0.74 mg/day | 0.72 (0.51, 1.02) | 0.74 (0.52, 1.04) |
| 3 | 1.22 mg/day | 0.84 (0.60, 1.19) | 0.87 (0.61 1.22) |
| 4 | 2.20 mg/day | 0.71 (0.49, 1.01) | 0.74 (0.52, 1.07) |
| p for trend | 0.13 | 0.22 | |
| Total Anthocyanidin Intake | |||
| 1 | 4.41 mg/day | Ref | Ref |
| 2 | 8.74 mg/day | 0.60 (0.42, 0.85) | 0.61 (0.43, 0.87) |
| 3 | 14.0 mg/day | 0.67 (0.48, 0.95) | 0.68 (0.49, 0.97) |
| 4 | 32.94 mg/day | 0.69 (0.48, 0.99) | 0.70 (0.49, 1.00) |
| p for trend | 0.23 | 0.26 | |
| Total Proanthocyanidins Intake | |||
| 1 | 29.83 mg/day | Ref | Ref |
| 2 | 57.82 mg/day | 0.72 (0.50, 1.04) | 0.75 (0.52, 1.07) |
| 3 | 84.28 mg/day | 0.83 (0.59, 1.18) | 0.86 (0.61, 1.22) |
| 4 | 126.63 mg/day | 0.73 (0.51, 1.06) | 0.78 (0.56, 1.13) |
| p for trend | 0.19 | 0.32 | |
| Total Flavonoid Intake | |||
| 1 | 86.10 mg/ day | Ref | Ref |
| 2 | 151.80 mg/day | 0.94 (0.67, 1.32) | 0.97 (0.69, 1.37) |
| 3 | 227.05 mg/day | 0.98 (0.69, 1.39) | 0.99 (0.70, 1.41) |
| 4 | 395.50 mg/day | 0.67 (0.46, 0.98) | 0.69 (0.47, 1.02) |
| p for trend | 0.04 | 0.05 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Agarwal, P.; Holland, T.M.; Wang, Y.; Bennett, D.A.; Morris, M.C. Association of Strawberries and Anthocyanidin Intake with Alzheimer’s Dementia Risk. Nutrients 2019, 11, 3060. https://doi.org/10.3390/nu11123060
Agarwal P, Holland TM, Wang Y, Bennett DA, Morris MC. Association of Strawberries and Anthocyanidin Intake with Alzheimer’s Dementia Risk. Nutrients. 2019; 11(12):3060. https://doi.org/10.3390/nu11123060
Chicago/Turabian StyleAgarwal, Puja, Thomas M Holland, Yamin Wang, David A Bennett, and Martha Clare Morris. 2019. "Association of Strawberries and Anthocyanidin Intake with Alzheimer’s Dementia Risk" Nutrients 11, no. 12: 3060. https://doi.org/10.3390/nu11123060
APA StyleAgarwal, P., Holland, T. M., Wang, Y., Bennett, D. A., & Morris, M. C. (2019). Association of Strawberries and Anthocyanidin Intake with Alzheimer’s Dementia Risk. Nutrients, 11(12), 3060. https://doi.org/10.3390/nu11123060






