Vitamin B12 Deficiency Is Prevalent Among Czech Vegans Who Do Not Use Vitamin B12 Supplements
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Laboratory Methods
2.3. Statistical Evaluation
3. Results
3.1. Characteristics of the Study Population
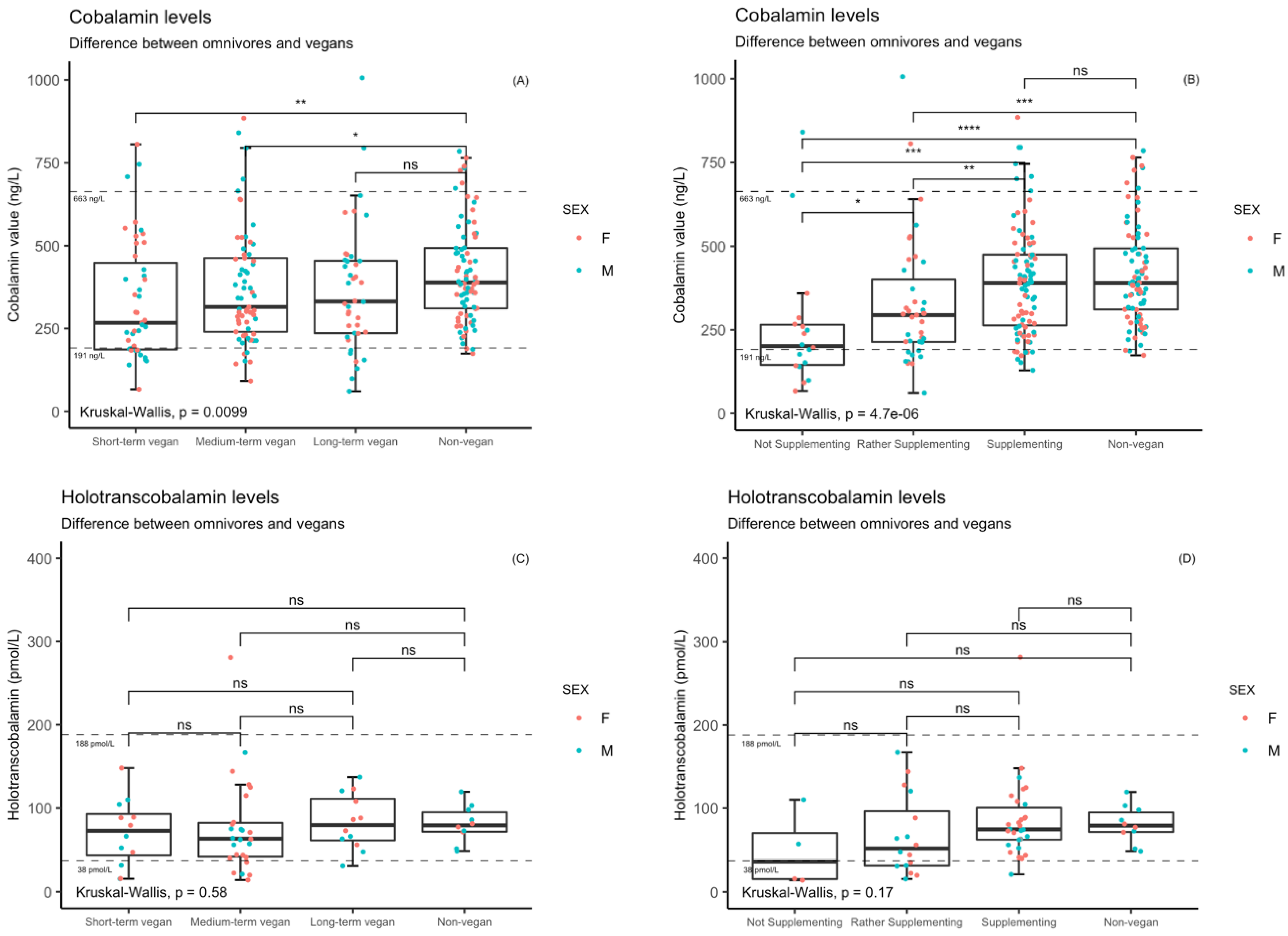
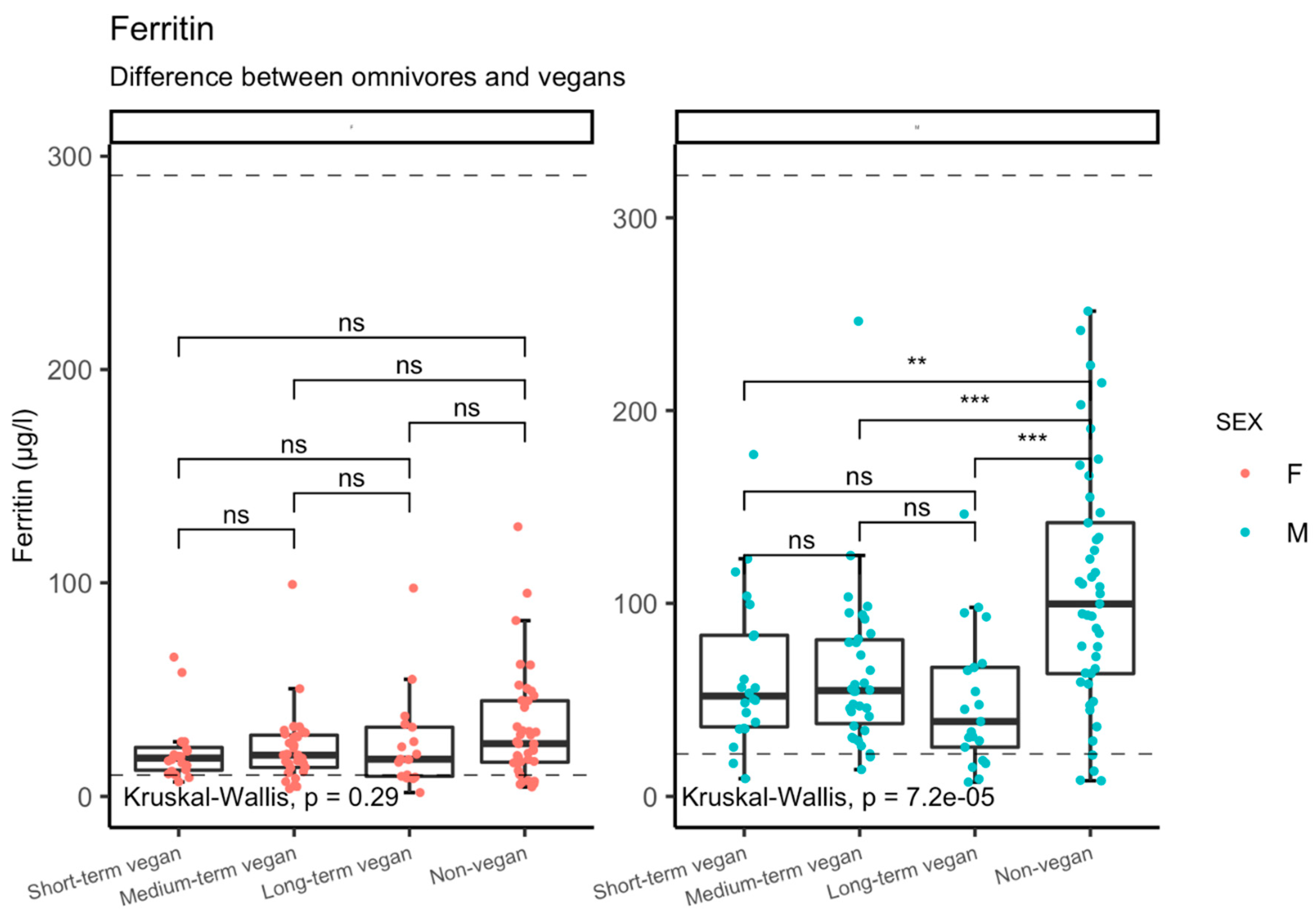
3.2. Differences among Vegans and Non-Vegans in Selected Blood Markers
3.3. Cobalamin Status in Relation to Supplement Use and Duration of Vegan Diet
Differences in Markers Stratified by Supplementation Habit or Duration of Veganism
3.4. Risk of Deficiency
3.4.1. Cobalamin Metabolism
3.4.2. Markers of Iron Deficiency
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Definition of Veganism. The Vegan Society. Available online: https://www.vegansociety.com/go-vegan/definition-veganism (accessed on 30 August 2018).
- Research and Markets. Top Trends in Prepared Foods 2017: Exploring trends in meat, fish and seafood; pasta, noodles and rice; prepared meals; savory deli food; soup; and meat substitutes. Am. East. Daylight Time 2017, 11, 59. [Google Scholar]
- Vegetarian.cz—Novinky. Available online: https://www.vegetarian.cz/ostatni/stemmark.html (accessed on 30 August 2018).
- Bezmasou Stravu Preferuje Desetina Mladých. Available online: https://www.ipsos.com/cs-cz/bezmasou-stravu-preferuje-desetina-mladych (accessed on 14 October 2019).
- American Dietetic Association. Dieticians of Canada Position of the American Dietetic Vegetarian Diets. J. Am. Diet. Assoc. 2009, 109, 1266–1282. [Google Scholar] [CrossRef] [PubMed]
- Weder, S.; Schaefer, C.; Keller, M. Die Gießener Vegane Lebensmittelpyramide. Ernährungs Umschau 2018, 65, 134–143. [Google Scholar]
- Pawlak, R.; Lester, S.E.; Babatunde, T. The prevalence of cobalamin deficiency among vegetarians assessed by serum vitamin B12: A review of literature. Eur. J. Clin. Nutr. 2014, 68, 541–548. [Google Scholar] [CrossRef] [PubMed]
- Krajčovičová-Kudláčková, M.; Blažíček, P.; Kopčová, J.; Béderová, A.; Babinská, K. Homocysteine levels in vegetarians versus omnivores. Ann. Nutr. Metab. 2000, 44, 135–138. [Google Scholar] [CrossRef] [PubMed]
- Gojda, J.; Rossmeislová, L.; Straková, R.; Tůmová, J.; Elkalaf, M.; Jaček, M.; Tůma, P.; Potočková, J.; Krauzová, E.; Waldauf, P.; et al. Chronic dietary exposure to branched chain amino acids impairs glucose disposal in vegans but not in omnivores. Eur. J. Clin. Nutr. 2017, 71, 594. [Google Scholar] [CrossRef] [PubMed]
- Gojda, J.; Patková, J.; Jaček, M.; Potočková, J.; Trnka, J.; Kraml, P.; Anděl, M. Higher insulin sensitivity in vegans is not associated with higher mitochondrial density. Eur. J. Clin. Nutr. 2013, 67, 1310–1315. [Google Scholar] [CrossRef] [PubMed]
- ZIMA, T. Laboratorní Diagnostika 2. Vyd; Galén: Praha, Czechia, 2007; p. 728. ISBN 978-80-7262-372-3. [Google Scholar]
- Martí-Carvajal, A.J.; Solà, I.; Lathyris, D. Homocysteine-lowering interventions for preventing cardiovascular events. Cochrane Database Syst. Rev. 2017, 8, CD006612. [Google Scholar] [CrossRef] [PubMed]
- Baggott, J.E.; Tamura, T. Homocysteine, iron and cardiovascular disease: A hypothesis. Nutrients 2015, 7, 1108–1118. [Google Scholar] [CrossRef] [PubMed]
- Sabaté, G.S.J. Health and sustainability outcomes of vegetarian dietary patterns: A revisit of the EPIC-Oxford and the Adventist Health Study-2 cohorts. Eur. J. Clin. Nutr. 2019, 72, 60–70. [Google Scholar]
- Toumpanakis, A.; Turnbull, T.; Alba-barba, I. Effectiveness of plant-based diets in promoting well-being in the management of type 2 diabetes: A systematic review. Drug Ther. Bull. 2018. [Google Scholar] [CrossRef] [PubMed]
- Ma, H.; Lin, H.; Hu, Y.; Li, X.; He, W.; Jin, X.; Gao, J.; Zhao, N.; Song, B.; Pan, B.; et al. Serum ferritin levels are associated with carotid atherosclerosis in Chinese postmenopausal women: The Shanghai Changfeng Study. Br. J. Nutr. 2015, 114, 1064–1071. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Alkhateeb, A.A.; Connor, J.R. The significance of ferritin in cancer: Anti-oxidation, inflammation and tumorigenesis. Biochim. Biophys. Acta Rev. Cancer 2013, 1836, 245–254. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.-H.; Bae, Y.J. Postmenopausal Vegetarians’ Low Serum Ferritin Level May Reduce the Risk for Metabolic Syndrome. Biol. Trace Elem. Res. 2012, 149, 34–41. [Google Scholar] [CrossRef] [PubMed]
- Rizzo, G.; Laganà, A.S.; Rapisarda, A.M.C.; La Ferrera, G.M.G.; Buscema, M.; Rossetti, P.; Nigro, A.; Muscia, V.; Valenti, G.; Sapia, F.; et al. Vitamin B12 among vegetarians: Status, assessment and supplementation. Nutrients 2016, 8, 767. [Google Scholar] [CrossRef] [PubMed]
- Snow, C.F. Laboratory Diagnosis of Vitamin B 12 and Folate Deficiency. Arch. Intern. Med. 1999, 159, 1289–1298. [Google Scholar] [CrossRef] [PubMed]
- Scolamiero, E.; Villani, G.R.D.; Ingenito, L.; Pecce, R.; Albano, L.; Caterino, M.; Girolamo, M.G.D.; Stefano, C.D.; Franzese, I.; Gallo, G.; et al. Maternal vitamin B12 deficiency detected in expanded newborn screening. Clin. Biochem. 2014, 47, 312–317. [Google Scholar] [CrossRef] [PubMed]
| Group | Age | BMI | Sex | Waist Circumference | |||
|---|---|---|---|---|---|---|---|
| n | Mean Age | n | Mean BMI | Males | Females | Mean Waist Circumference | |
| Vegans | 151 | 32.8 ± 7.4 | 105 | 22.6 ± 2.7 | 78 | 73 | 81.5 ± 9.11 |
| Omnivores | 84 | 32.0 ± 7.0 | 84 | 23.7 ± 3.1 | 45 | 39 | 77.4 ± 9.98 |
| p-value | 0.394 | 0.005 | 0.779 | 0.002 | |||
| Group | n | Mean | Min | Max | p-Value | ANCOVA * | |
|---|---|---|---|---|---|---|---|
| Cobalamin (ng/L) | Vegans | 148 | 359 | 61 | 1006 | 0.002 | 0.014 |
| Non-vegans | 84 | 416 | 174 | 785 | |||
| HCY (µmol/L) | Vegans | 147 | 14.7 | 5.8 | 65 | 0.607 | 0.440 |
| Non-vegans | 84 | 14.1 | 7.2 | 29 | |||
| Folate (µg/L) | Vegans | 146 | 13.0 | 3.63 | 40.5 | <0.001 | <0.001 |
| Non-vegans | 84 | 8.74 | 2.53 | 21.0 | |||
| Holotranscobalamin (pmol/L) | Vegans | 99 | 89.7 | 14.0 | 682 | 0.121 | 0.909 |
| Non-vegans | 34 | 87.9 | 44.0 | 150 |
| Group | n | Mean | Min | Max | p-Value | ANCOVA | |
|---|---|---|---|---|---|---|---|
| Ferritin (µg/L) | Vegans | 145 | 43.6 | 1.8 | 246 | 0.001 | <0.001 |
| Non-vegans | 81 | 72.2 | 4.4 | 252 | |||
| Iron (µmol/L) | Vegans | 150 | 19.0 | 3.3 | 65.6 | 0.392 | 0.925 |
| Non-vegans | 84 | 19.2 | 5.2 | 41.5 | |||
| Iron Binding Capacity (µmol/L) | Vegans | 150 | 56.8 | 38 | 88 | 0.327 | 0.326 |
| Non-vegans | 84 | 56.3 | 37 | 92 | |||
| Transferrin (g/L) | Vegans | 139 | 2.74 | 1.74 | 4.18 | 0.396 | 0.993 |
| Non-vegans | 84 | 2.74 | 1.92 | 4.52 | |||
| Saturated Transferrin (%) | Vegans | 135 | 28.7 | 3.4 | 79.1 | 0.614 | 0.930 |
| Non-vegans | 68 | 28.8 | 7.5 | 70.9 | |||
| Hemoglobin (g/L) | Vegans | 150 | 138 | 97 | 169 | 0.022 | <0.001 |
| Non-vegans | 84 | 142 | 111 | 169 | |||
| MCV (fL) | Vegans | 149 | 88.5 | 87.2 | 103 | 0.009 | 0.022 |
| Non-vegans | 84 | 87.2 | 74.3 | 96.4 |
| Supplement Use | n | Mean | Min | Max | p-Value | ANCOVA | |
|---|---|---|---|---|---|---|---|
| Cobalamin (ng/L) | Regular | 90 | 398 | 129 | 885 | <0.001 | 0.002 |
| Irregular | 39 | 329 | 61 | 1006 | |||
| None | 17 | 256 | 67 | 841 | |||
| Non-vegan | 84 | 389 | 174 | 785 | |||
| Holotranscobalamin (pmol/L) | Regular | 63 | 104 | 21.1 | 682 | <0.001 | 0.06 |
| Irregular | 25 | 72.2 | 15.5 | 167 | |||
| None | 11 | 46.1 | 14 | 110 | |||
| Non-vegans | 34 | 87.9 | 44.0 | 150 | |||
| Homocysteine (µmol/L) | Regular | 90 | 13.5 | 5.8 | 33.8 | 0.001 | <0.001 |
| Irregular | 37 | 15.2 | 7.9 | 65 | |||
| None | 17 | 20.3 | 9.8 | 46.4 | |||
| Non-vegans | 84 | 14.1 | 7.2 | 29 | |||
| MCV (fL) | Regular | 91 | 88.4 | 79 | 98.3 | 0.04 | 0.63 |
| Irregular | 38 | 88.6 | 63.9 | 103 | |||
| None | 17 | 89.5 | 84.6 | 94.8 | |||
| Non-vegans | 84 | 87.2 | 74.3 | 96.4 |
| Duration of Veganism | n | Mean | Min | Max | p-Value | * ANCOVA | |
|---|---|---|---|---|---|---|---|
| Cobalamin (ng/L) | Longterm vegan | 41 | 341 | 61 | 795 | 0.02 | 0.05 |
| Medium-term vegan | 66 | 388 | 92 | 1006 | |||
| Short-term vegan | 39 | 344 | 67 | 806 | |||
| Non-vegan | 84 | 389 | 174 | 785 | |||
| Holotranscobalamin (pmol/L) | Long-term vegan | 27 | 103 | 31.1 | 521 | 0.15 | 0.58 |
| Medium-term vegan | 53 | 88.2 | 14.0 | 682 | |||
| Short-term vegan | 19 | 74.5 | 15.5 | 148 | |||
| Non-vegan | 34 | 87.9 | 44.0 | 150 | |||
| Homocysteine (µmol/L) | Long-term vegan | 39 | 16.9 | 6.2 | 65 | 0.332 | 0.028 |
| Medium-term vegan | 65 | 13.3 | 6.8 | 28.9 | |||
| Short-term vegan | 40 | 14.9 | 5.8 | 46.4 | |||
| Non-vegans | 84 | 14.1 | 7.2 | 29 | |||
| Folate (µg/L) | Long-term vegan | 40 | 12.9 | 4.98 | 25.0 | <0.001 | <0.001 |
| Medium-term vegan | 65 | 13.0 | 3.63 | 40.5 | |||
| Short-term vegan | 37 | 13.0 | 4.5 | 21.9 | |||
| Non-vegan | 24 | 9.75 | 3.64 | 20.0 | |||
| MCV (fL) | Long-term vegan | 40 | 87.8 | 63.9 | 103 | 0.03 | 0.06 |
| Medium-term vegan | 66 | 88.8 | 79 | 96.1 | |||
| Short-term vegan | 40 | 88.8 | 80.4 | 98.3 | |||
| Non-vegans | 84 | 87.2 | 74.3 | 96.4 | |||
| Ferritin (µg/L) | Long-term vegan | 38 | 39.4 | 1.8 | 146 | 0.005 | <0.001 |
| Medium-term vegan | 65 | 41.6 | 3.6 | 246 | |||
| Short-term vegan | 39 | 51.1 | 6.8 | 177 | |||
| Non-vegans | 81 | 72.2 | 4.4 | 252 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Selinger, E.; Kühn, T.; Procházková, M.; Anděl, M.; Gojda, J. Vitamin B12 Deficiency Is Prevalent Among Czech Vegans Who Do Not Use Vitamin B12 Supplements. Nutrients 2019, 11, 3019. https://doi.org/10.3390/nu11123019
Selinger E, Kühn T, Procházková M, Anděl M, Gojda J. Vitamin B12 Deficiency Is Prevalent Among Czech Vegans Who Do Not Use Vitamin B12 Supplements. Nutrients. 2019; 11(12):3019. https://doi.org/10.3390/nu11123019
Chicago/Turabian StyleSelinger, Eliška, Tilman Kühn, Magdalena Procházková, Michal Anděl, and Jan Gojda. 2019. "Vitamin B12 Deficiency Is Prevalent Among Czech Vegans Who Do Not Use Vitamin B12 Supplements" Nutrients 11, no. 12: 3019. https://doi.org/10.3390/nu11123019
APA StyleSelinger, E., Kühn, T., Procházková, M., Anděl, M., & Gojda, J. (2019). Vitamin B12 Deficiency Is Prevalent Among Czech Vegans Who Do Not Use Vitamin B12 Supplements. Nutrients, 11(12), 3019. https://doi.org/10.3390/nu11123019






