Eating Jet Lag: A Marker of the Variability in Meal Timing and Its Association with Body Mass Index
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design, Settings, Participants, and Protocol
2.2. Eating Period
2.3. Eating Jet Lag
2.4. Anthropometric Parameters
2.5. Sleep and Circadian Parameters
2.6. Lifestyle Variables
2.7. Statistical Analyses
3. Results
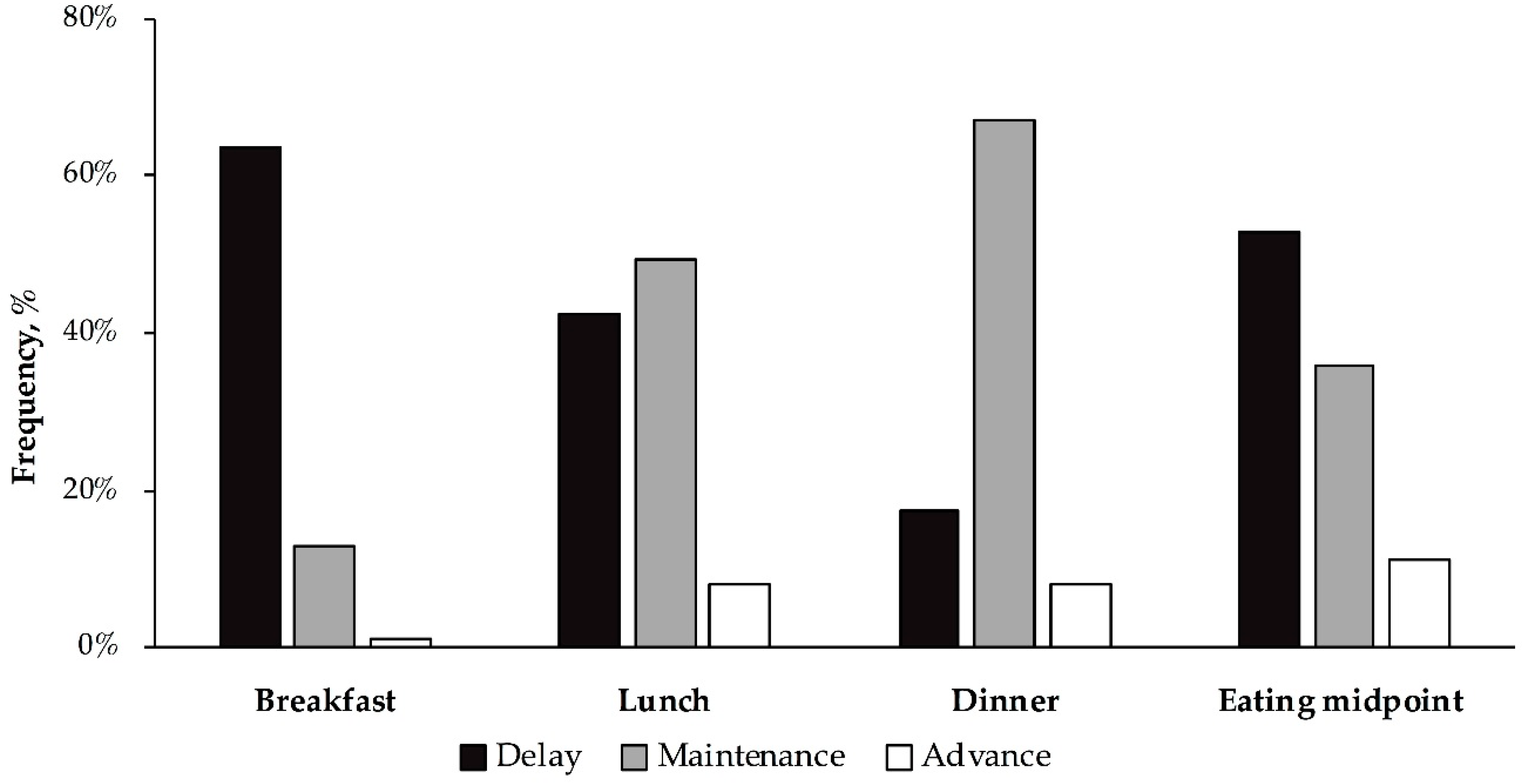
3.1. Differences in the Timing of Food Intake between Weekends and Weekdays among Young Adults
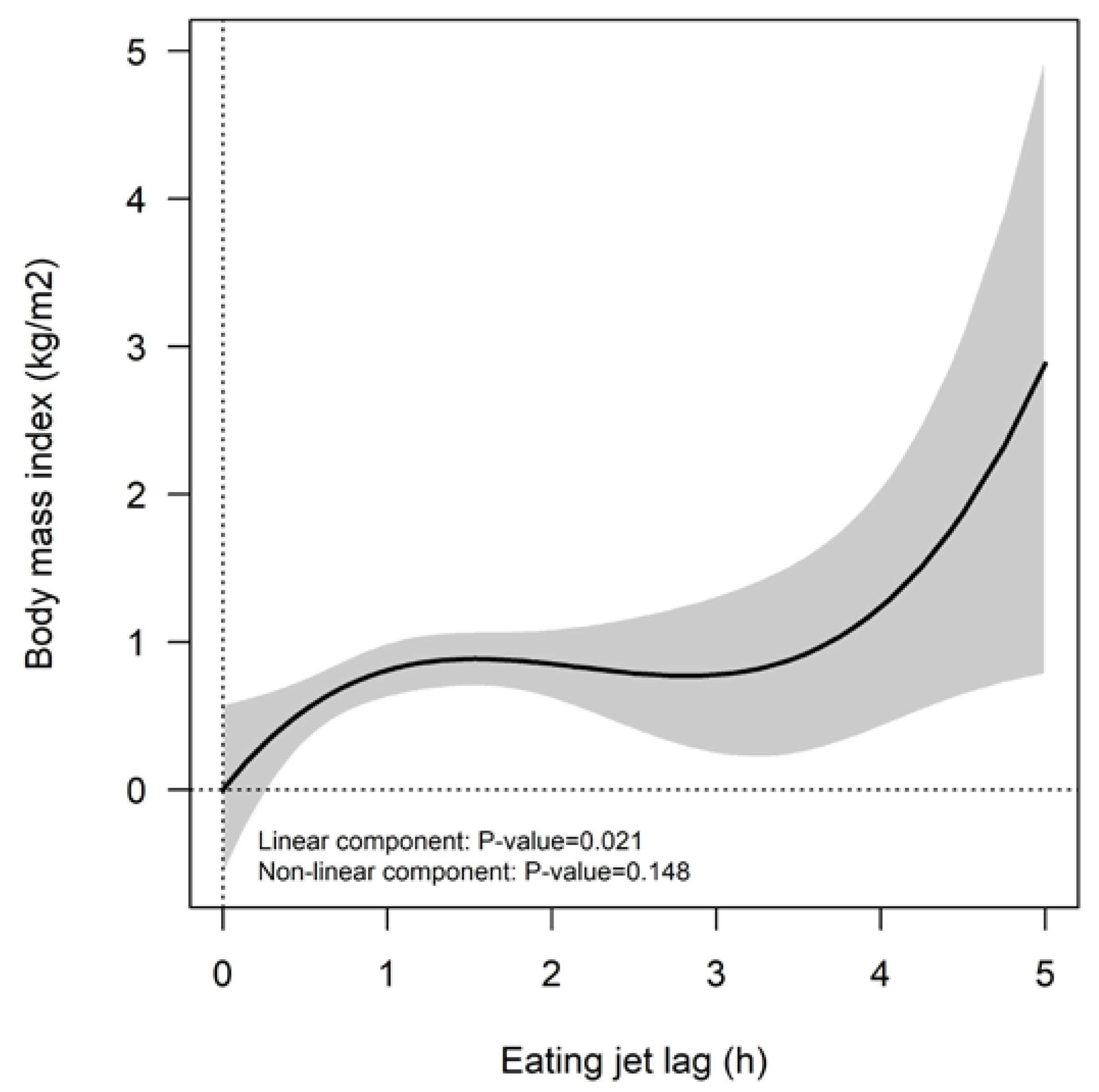
3.2. Increments in the Eating Jet Lag Are Associated with Higher BMI Values among the Population Studied
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Dashti, H.S.; Scheer, F.A.J.L.; Saxena, R.; Garaulet, M. Timing of Food Intake: Identifying Contributing Factors to Design Effective Interventions. Adv. Nutr. 2019, 10, 1–15. [Google Scholar] [CrossRef]
- LaCaille, L. Eating behavior. Encycl. Behav. Med. 2013. [Google Scholar]
- Leech, R.M.; Worsley, A.; Timperio, A.; McNaughton, S.A. Temporal eating patterns: A latent class analysis approach. Int. J. Behav. Nutr. Phys. Act. 2017, 14, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Garaulet, M.; Gomez-Abellán, P.; Alburquerque-Bejar, J.J.; Lee, Y.; Ordovas, J.M.; Scheer, F.A.J.L. Timing of food intake predicts weight loss effectiveness. Int. J. Obes. 2013, 37, 604–611. [Google Scholar] [CrossRef] [PubMed]
- Mchill, A.W.; Phillips, A.J.K.; Czeisler, C.A.; Keating, L.; Yee, K.; Barger, L.K.; Garaulet, M.; Scheer, F.A.J.L.; Klerman, E.B. Later circadian timing of food intake is associated with increased body fat. Am. J. Clin. Nutr. 2017, 106, 1213–1219. [Google Scholar] [CrossRef] [PubMed]
- Bandín, C.; Scheer, F.; Luque, A.J.; Zamora, S.; Madrid, J.A.; Garaulet, M. Meal timing affects glucose tolerance, substrate oxidation and circadian-related variables: A randomized, crossover trial. Int. J. Obes. 2015, 39, 828–833. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Lozano, T.; Vidal, J.; de Hollanda, A.; Scheer, F.A.J.L.; Garaulet, M.; Izquierdo-Pulido, M. Timing of food intake is associated with weight loss evolution in severe obese patients after bariatric surgery. Clin. Nutr. 2016, 35, 1308–1314. [Google Scholar] [CrossRef]
- St-Onge, M.P.; Ard, J.; Baskin, M.L.; Chiuve, S.E.; Johnson, H.M.; Kris-Etherton, P.; Varady, K. Meal Timing and Frequency: Implications for Cardiovascular Disease Prevention: A Scientific Statement from the American Heart Association. Circulation 2017, 135, e96–e121. [Google Scholar] [CrossRef]
- Moreno, J.P.; Crowley, S.J.; Alfano, C.A.; Hannay, K.M.; Thompson, D.; Baranowski, T. Potential circadian and circannual rhythm contributions to the obesity epidemic in elementary school age children. Int. J. Behav. Nutr. Phys. Act. 2019, 16, 1–10. [Google Scholar] [CrossRef]
- Potter, G.D.M.; Cade, J.E.; Grant, P.J.; Hardie, L.J. Nutrition and the circadian system. Br. J. Nutr. 2016, 116, 434–442. [Google Scholar] [CrossRef]
- Potter, G.D.M.; Skene, D.J.; Arendt, J.; Cade, J.E.; Grant, P.J.; Hardie, L.J. Circadian rhythm and sleep disruption: causes, metabolic consequences, and countermeasures. Endocr. Rev. 2016, 37, 584–608. [Google Scholar] [CrossRef] [PubMed]
- Chaix, A.; Manoogian, E.N.C.; Melkani, G.C.; Panda, S. Time-Restricted Eating to Prevent and Manage Chronic Metabolic Diseases. Annu. Rev. Nutr. 2019, 39, 1–25. [Google Scholar] [CrossRef] [PubMed]
- Vetter, C. Circadian disruption: What do we actually mean? Eur. J. Neurosci. 2018, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Dashti, H.S.; Scheer, F.A.J.L.; Jacques, P.F.; Lamon-Fava, S.; Ordovas, J.M. Short sleep duration and dietary intake: Epidemiologic evidence, mechanisms and health implications. Adv. Nutr. 2015, 6, 648–659. [Google Scholar] [CrossRef] [PubMed]
- Challet, E. The circadian regulation of food intake. Nat. Rev. Endocrinol. 2019, 15, 393–405. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Minguez, J.; Saxena, R.; Bandín, C.; Scheer, F.A.; Garaulet, M. Late dinner impairs glucose tolerance in MTNR1B risk allele carriers: A randomized, cross-over study. Clin. Nutr. 2018, 37, 1133–1140. [Google Scholar] [CrossRef]
- Zarrinpar, A.; Chaix, A.; Panda, S. Daily Eating Patterns and Their Impact on Health and Disease. Trends Endocrinol. Metab. 2016, 27, 69–83. [Google Scholar] [CrossRef]
- Vollmers, C.; Gill, S.; DiTacchio, L.; Pulivarthy, S.R.; Le, H.D.; Panda, S. Time of feeding and the intrinsic circadian clock drive rhythms in hepatic gene expression. Proc. Natl. Acad. Sci. USA 2009, 106, 21453–21458. [Google Scholar] [CrossRef]
- Roenneberg, T.; Allebrandt, K.V.; Merrow, M.; Vetter, C. Social Jetlag and Obesity. Curr. Biol. 2012, 23, 737. [Google Scholar] [CrossRef]
- Malone, S.K.; Zemel, B.; Compher, C.; Souders, M.; Chittams, J.; Thompson, A.L.; Pack, A.; Lipman, T.H.; Kohl, S.; Zemel, B.; et al. Social jet lag, chronotype and body mass index in 14–17-year-old adolescents. Chronobiol. Int. 2016, 33, 1255–1266. [Google Scholar] [CrossRef]
- Roenneberg, T.; Pilz, L.K.; Zerbini, G.; Winnebeck, E.C. Chronotype and Social Jetlag-A (self-) critical review. Preprints 2019, 8, 54. [Google Scholar] [CrossRef] [PubMed]
- Parsons, M.J.; Moffitt, T.E.; Gregory, A.M.; Goldman-Mellor, S.; Nolan, P.M.; Poulton, R.; Caspi, A. Social jetlag, obesity and metabolic disorder: Investigation in a cohort study. Int. J. Obes. 2015, 39, 842–848. [Google Scholar] [CrossRef] [PubMed]
- Zerón-Rugerio, M.F.; Cambras, T.; Izquierdo-Pulido, M. Social Jet Lag Associates Negatively with the Adherence to the Mediterranean Diet and Body Mass Index among Young Adults. Nutrients 2019, 11, 1756. [Google Scholar] [CrossRef] [PubMed]
- Guinter, M.A.; Park, Y.-M.; Steck, S.E.; Sandler, D.P. Day-to-day regularity in breakfast consumption is associated with weight status in a prospective cohort of women. Int. J. Obes. 2019. [Google Scholar] [CrossRef]
- Gill, S.; Panda, S. A Smartphone App Reveals Erratic Diurnal Eating Patterns in Humans that Can Be Modulated for Health Benefits. Cell Metab. 2015, 22, 789–798. [Google Scholar] [CrossRef]
- Wittmann, M.; Dinich, J.; Merrow, M.; Roenneberg, T. Social jetlag: Misalignment of biological and social time. Chronobiol. Int. 2006, 23, 497–509. [Google Scholar] [CrossRef]
- Mota, M.C.; Silva, C.; Cristina, L.; Balieiro, T.; Gonçalves, B.; Fahmy, W.; Crispim, C. Association between social jetlag food consumption and meal times in patients with obesity-related chronic diseases. PLoS ONE 2019, 14, 1–14. [Google Scholar] [CrossRef]
- Roenneberg, T.; Wirz-Justice, A.; Merrow, M. Life between clocks: daily temporal patterns of human chronotypes. J. Biol. Rhythms 2003, 18, 80–90. [Google Scholar] [CrossRef]
- Ruiz-Lozano, T.; Vidal, J.; Hollanda, A.; Canteras, M.; Garaulet, M.; Izquierdo-Pulido, M. Evening chronotype associates with obesity in severely obese subjects: Interaction with CLOCK 3111T/C. Int. J. Obes. 2016, 40, 1550–1557. [Google Scholar] [CrossRef]
- Serra-Majem, L.; Ribas, L.; Ngo, J.; Ortega, R.M.; Garcia, A.; Perez-Rodrigo, C.; Aranceta, J. Food, youth and the Mediterranean diet in Spain. Development of KIDMED, Mediterranean Diet Quality Index in children and adolescents. Public Health Nutr. 2004, 7, 931–935. [Google Scholar] [CrossRef]
- Ramos, E.; Ramírez, E.; Salas, R.; Núñez, G.; Villarreal, J. Calidad del patron de consumo alimentario del noreste de Mexico. Respyn 2016, 15, 8–15. [Google Scholar]
- Román-Viñas, B.; Ribas-Barba, L.; Ngoa, J.; Serra-Majem, L. Validación en población catalana del cuestionario internacional de actividad física. Gac. Sanit. 2013, 27, 254–257. [Google Scholar] [CrossRef] [PubMed]
- R Core Team and Contributors World Wide GLM: Fitting Generalized Linear Models. Available online: https://stat.ethz.ch/R-manual/R-devel/library/stats/html/glm.html (accessed on 31 July 2019).
- Wehrens, S.M.T.; Christou, S.; Isherwood, C.; Archer, S.N.; Skene, D.J.; Johnston, J.D.; Wehrens, S.M.T.; Christou, S.; Isherwood, C.; Middleton, B.; et al. Meal Timing Regulates the Human Circadian System Report Meal Timing Regulates the Human Circadian System. Curr. Biol. 2017, 27, 1768–1775. [Google Scholar] [CrossRef] [PubMed]
- Scheer, F.A.J.L.; Hilton, M.F.; Mantzoros, C.S.; Shea, S.A. Adverse metabolic and cardiovascular consequences of circadian misalignment. Proc. Natl. Acad. Sci. USA 2009, 106, 4453–4458. [Google Scholar] [CrossRef] [PubMed]
- Sun, M.; Feng, W.; Wang, F.; Li, P.; Li, Z.; Li, M.; Tse, G.; Vlaanderen, J.; Vermeulen, R.; Tse, L.A. Meta-analysis on shift work and risks of specific obesity types. Obes. Rev. 2018, 19, 28–40. [Google Scholar] [CrossRef] [PubMed]
- Bonham, M.P.; Bonnell, E.K.; Huggins, C.E. Energy intake of shift workers compared to fixed day workers: A systematic review and meta-analysis. Chronobiol. Int. 2016, 33, 1086–1100. [Google Scholar] [CrossRef]
- Panda, S. Circadian physiology of metabolism. Science 2016, 354, 317–322. [Google Scholar] [CrossRef]
- Xiao, Q.; Garaulet, M.; Scheer, F.A.J.L. Meal timing and obesity: Interactions with macronutrient intake and chronotype. Int. J. Obes. 2019, 1701–1711. [Google Scholar] [CrossRef]
- Roßbach, S.; Diederichs, T.; Nöthlings, U.; Buyken, A.E.; Alexy, U. Relevance of chronotype for eating patterns in adolescents. Chronobiol. Int. 2018, 35, 336–347. [Google Scholar] [CrossRef]
- Silva, C.M.; Mota, M.C.; Miranda, M.T.; Paim, S.L.; Waterhouse, J.; Crispim, C.A.; Mendes, C.; Mota, M.C.; Miranda, M.T.; Paim, S.L.; et al. Chronotype, social jetlag and sleep debt are associated with dietary intake among Brazilian undergraduate students. Chronobiol. Int. 2016, 33, 740–748. [Google Scholar] [CrossRef]
- Fong, M.; Caterson, I.D.; Madigan, C.D. Are large dinners associated with excess weight, and does eating a smaller dinner achieve greater weight loss? A systematic review and meta-analysis. Br. J. Nutr. 2017, 118, 616–628. [Google Scholar] [CrossRef] [PubMed]
| Meal Timing Variables | Mean (SD) |
|---|---|
| Breakfast | |
| Weekdays, hh:mm | 08:21 (01:15) |
| Weekends, hh:mm | 10:24 (01:08) |
| p-value a | <0.001 |
| Breakfast jet lag, h | 2.0 (1.2) |
| Lunch | |
| Weekdays, hh:mm | 14:15 (01:09) |
| Weekends, hh:mm | 14:53 (00:58) |
| p-value a | <0.001 |
| Lunch jet lag, h | 0.9 (0.8) |
| Dinner | |
| Weekdays, hh:mm | 21:17 (00:47) |
| Weekends, hh:mm | 21:32 (00:56) |
| p-value a | <0.001 |
| Dinner jet lag, h | 0.5 (0.7) |
| Eating midpoint | |
| Weekdays, h | 15:09 (01:28) |
| Weekends, h | 16:01 (01:18) |
| p-value a | <0.001 |
| Eating jet lag, h | 1.3 (0.9) |
| Chronotype (MSF) | Social Jet Lag | |||||
|---|---|---|---|---|---|---|
| β | 95% CI | p-Value a | β | 95% CI | p-Value a | |
| Breakfast jet lag | 0.320 | 0.250, 0.0390 | <0.00001 | 0.720 | 0.640, 0.800 | <0.00001 |
| Lunch jet lag | 0.100 | 0.061, 0.141 | <0.00001 | 0.049 | 0.002, 0.096 | 0.042 |
| Dinner jet lag | 0.073 | 0.061, 0.140 | <0.00001 | 0.072 | 0.038, 0.110 | <0.001 |
| Eating jet lag | 0.110 | 0.063, 0.160 | <0.00001 | 0.270 | 0.210, 0.330 | <0.00001 |
| Variables | Model 1 | Model 2 | Model 3 |
|---|---|---|---|
| β (95% CI) | β (95% CI) | β (95% CI) | |
| Eating jet lag, h | 0.336 (0.132, 0.540) ** | 0.280 (0.080, 0.479) ** | 0.283 (0.073, 0.494) ** |
| Nationality (1, Spanish; 2, Mexicans) | 1.671 (1.232, 2.111) ** | 1.621 (1.129, 2.113) *** | |
| age, years | 0.239 (0.162, 0.317) *** | 0.237 (0.159, 0.316) *** | |
| Gender (1, male; 2, females) | −1.076 (−1.550, −0.603) *** | −1.067 (−1.546, −0.588) *** | |
| Physical activity, METs | 0.000 (0.000, 0.000) | 0.000 (0.000, 0.000) | |
| Diet quality, z-score | −0.012 (−0.206, 0.182) | 0.013 (−0.192, 0.218) | |
| Sleep duration, h | −0.081 (−0.281, 0.120) | −0.094 (−0.300, 0.111) | |
| Average eating duration, h | −0.033 (−0.120, 0.054) | ||
| Chronotype, MSF | 0.031 (−0.207, 0.268) | ||
| Social jet lag, h | −0.034 (−0.320, 0.253) | ||
| R2 | 0.009 ** | 0.089 *** | 0.087 *** |
| R2 change | 0.009 ** | 0.085 *** | 0.001 |
| F change | 10.479 | 17.111 | 0.242 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zerón-Rugerio, M.F.; Hernáez, Á.; Porras-Loaiza, A.P.; Cambras, T.; Izquierdo-Pulido, M. Eating Jet Lag: A Marker of the Variability in Meal Timing and Its Association with Body Mass Index. Nutrients 2019, 11, 2980. https://doi.org/10.3390/nu11122980
Zerón-Rugerio MF, Hernáez Á, Porras-Loaiza AP, Cambras T, Izquierdo-Pulido M. Eating Jet Lag: A Marker of the Variability in Meal Timing and Its Association with Body Mass Index. Nutrients. 2019; 11(12):2980. https://doi.org/10.3390/nu11122980
Chicago/Turabian StyleZerón-Rugerio, María Fernanda, Álvaro Hernáez, Armida Patricia Porras-Loaiza, Trinitat Cambras, and Maria Izquierdo-Pulido. 2019. "Eating Jet Lag: A Marker of the Variability in Meal Timing and Its Association with Body Mass Index" Nutrients 11, no. 12: 2980. https://doi.org/10.3390/nu11122980
APA StyleZerón-Rugerio, M. F., Hernáez, Á., Porras-Loaiza, A. P., Cambras, T., & Izquierdo-Pulido, M. (2019). Eating Jet Lag: A Marker of the Variability in Meal Timing and Its Association with Body Mass Index. Nutrients, 11(12), 2980. https://doi.org/10.3390/nu11122980









