Antitumor Effects of Quercetin in Hepatocarcinoma In Vitro and In Vivo Models: A Systematic Review
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Selection Criteria
2.2. Search Strategy and Study Selection
2.3. Data Extraction
3. Results
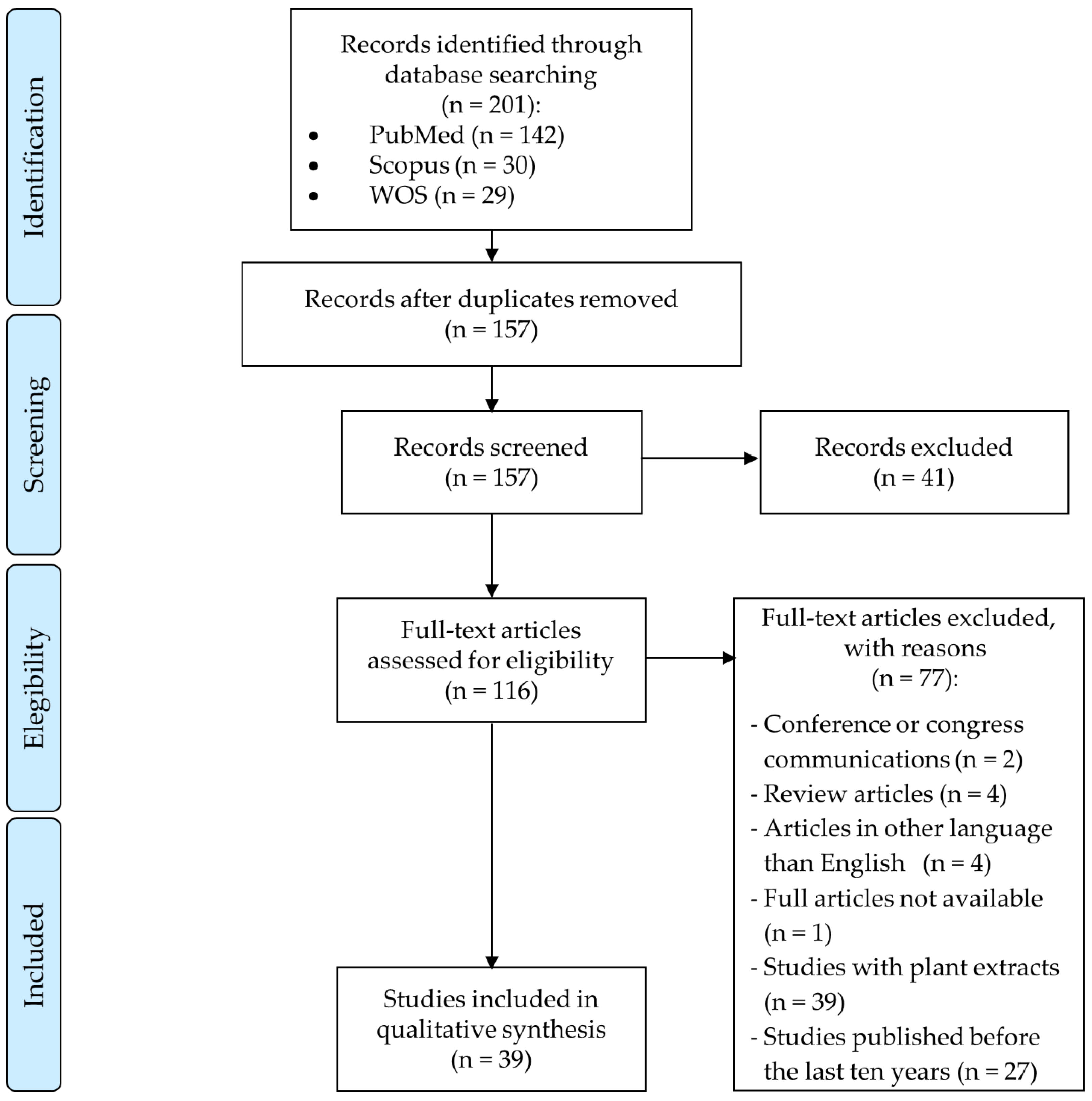
3.1. Study Selection
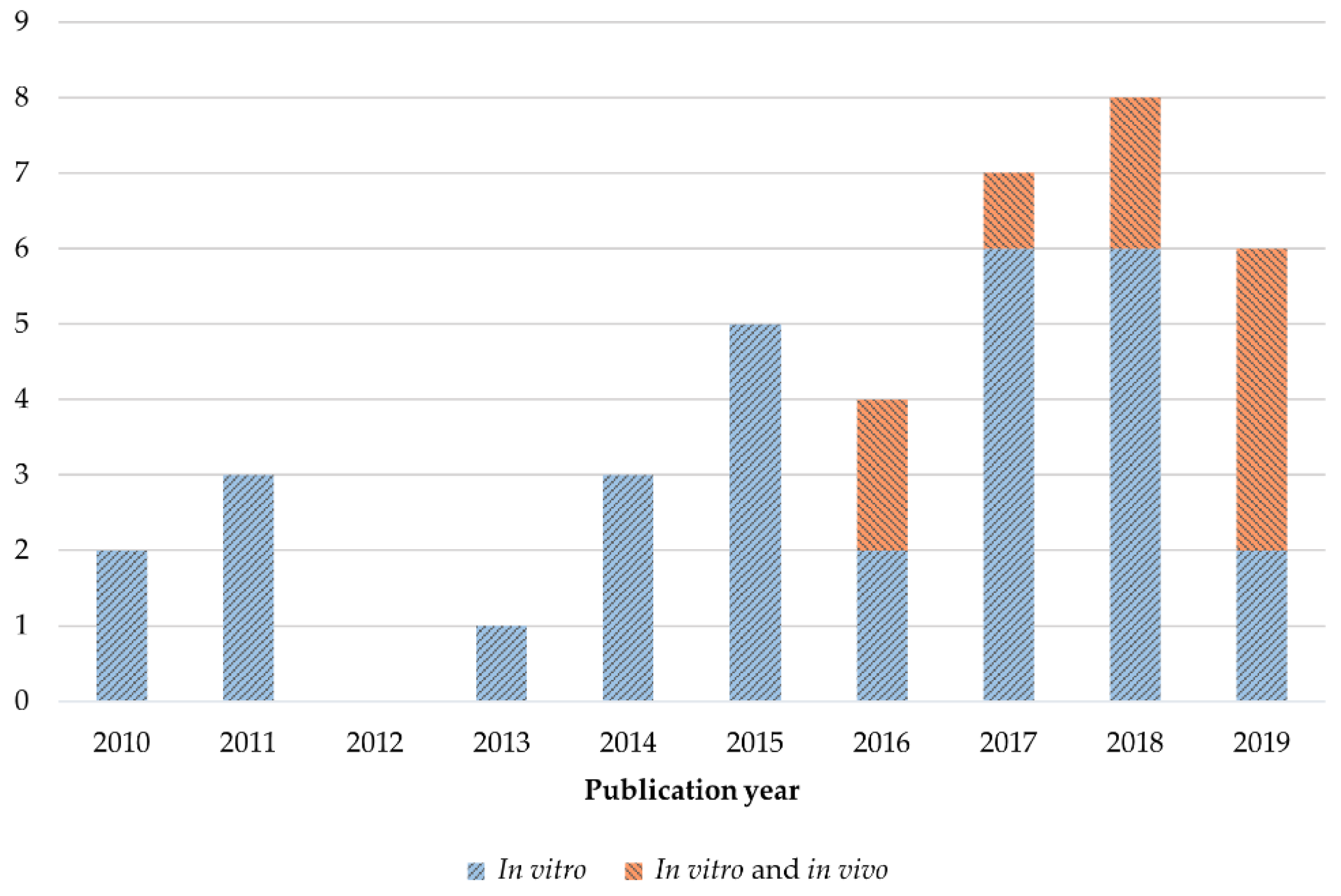
3.2. Study Characteristics
3.3. Anticancer Activities of Quercetin in HCC Preclinical Models
3.3.1. Antitumor Properties of Quercetin as Single Agent against HCC
3.3.2. Encapsulation for the Improvement of Quercetin Effects in HCC
3.3.3. Synergistic Effects through Quercetin Combination against HCC
3.3.4. Effects of Quercetin Derivatives Treatment in HCC
4. Discussion
Limitations
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Khan, F.; Niaz, K.; Maqbool, F.; Hassan, F.I.; Abdollahi, M.; Venkata, K.C.N.; Nabavi, S.M.; Bishayee, A. Molecular targets underlying the anticancer effects of quercetin: An update. Nutrients 2016, 8, 529. [Google Scholar] [CrossRef]
- Boots, A.W.; Haenen, G.R.M.M.; Bast, A. Health effects of quercetin: From antioxidant to nutraceutical. Eur. J. Pharmacol. 2008, 585, 325–337. [Google Scholar] [CrossRef]
- Oboh, G.; Ademosun, A.O.; Ogunsuyi, O.B. Quercetin and its role in chronic diseases. Adv. Exp. Med. Biol. 2016, 929, 377–387. [Google Scholar]
- Elumalai, P.; Lakshmi, S. Role of quercetin benefits in neurodegeneration. Adv. Neurobiol. 2016, 12, 229–245. [Google Scholar]
- Kawabata, K.; Mukai, R.; Ishisaka, A. Quercetin and related polyphenols: New insights and implications for their bioactivity and bioavailability. Food Funct. 2015, 6, 1399–1417. [Google Scholar] [CrossRef]
- Babaei, F.; Mirzababaei, M.; Nassiri-Asl, M. Quercetin in food: Possible mechanisms of its effect on memory. J. Food Sci. 2018, 83, 2280–2287. [Google Scholar] [CrossRef]
- Bae, M.; Park, Y.-K.; Lee, J.-Y. Food components with anti-fibrotic activity and implications in prevention of liver disease. J. Nutr. Biochem. 2018, 55, 1–11. [Google Scholar] [CrossRef]
- Ferramosca, A.; Di Giacomo, M.; Zara, V. Antioxidant dietary approach in treatment of fatty liver: New insights and updates. World J. Gastroenterol. 2017, 23, 4146–4157. [Google Scholar] [CrossRef]
- Miltonprabu, S.; Tomczyk, M.; Skalicka-Wozniak, K.; Rastrelli, L.; Daglia, M.; Nabavi, S.F.; Alavian, S.M.; Nabavi, S.M. Hepatoprotective effect of quercetin: From chemistry to medicine. Food Chem. Toxicol. 2017, 108, 365–374. [Google Scholar] [CrossRef]
- Li, X.; Jin, Q.; Yao, Q.; Xu, B.; Li, L.; Zhang, S.; Tu, C. The flavonoid quercetin ameliorates liver inflammation and fibrosis by regulating hepatic macrophages activation and polarization in mice. Front. Pharmacol. 2018, 9, 72. [Google Scholar] [CrossRef]
- Pisonero-Vaquero, S.; Martínez-Ferreras, Á.; García-Mediavilla, M.V.; Martínez-Flórez, S.; Fernández, A.; Benet, M.; Olcoz, J.L.; Jover, R.; González-Gallego, J.; Sánchez-Campos, S. Quercetin ameliorates dysregulation of lipid metabolism genes via the PI3K/AKT pathway in a diet-induced mouse model of nonalcoholic fatty liver disease. Mol. Nutr. Food Res. 2015, 59, 879–893. [Google Scholar] [CrossRef]
- Porras, D.; Nistal, E.; Martínez-Flórez, S.; Pisonero-Vaquero, S.; Olcoz, J.L.; Jover, R.; González-Gallego, J.; García-Mediavilla, M.V.; Sánchez-Campos, S. Protective effect of quercetin on high-fat diet-induced non-alcoholic fatty liver disease in mice is mediated by modulating intestinal microbiota imbalance and related gut-liver axis activation. Free Radic. Biol. Med. 2017, 102, 188–202. [Google Scholar] [CrossRef]
- Tieppo, J.; Cuevas, M.J.; Vercelino, R.; Tuñón, M.J.; Marroni, N.P.; González-Gallego, J. Quercetin administration ameliorates pulmonary complications of cirrhosis in rats. J. Nutr. 2009, 139, 1339–1346. [Google Scholar] [CrossRef]
- Casas-Grajales, S.; Vázquez-Flores, L.F.; Ramos-Tovar, E.; Hernández-Aquino, E.; Flores-Beltrán, R.E.; Cerda-García-Rojas, C.M.; Camacho, J.; Shibayama, M.; Muriel, P. Quercetin reverses experimental cirrhosis by immunomodulation of the proinflammatory and profibrotic processes. Fundam. Clin. Pharmacol. 2017, 31, 610–624. [Google Scholar] [CrossRef]
- Forner, A.; Reig, M.; Bruix, J. Hepatocellular carcinoma. Lancet 2018, 391, 1301–1314. [Google Scholar] [CrossRef]
- Fondevila, F.; Méndez-Blanco, C.; Fernández-Palanca, P.; González-Gallego, J.; Mauriz, J.L. Anti-tumoral activity of single and combined regorafenib treatments in preclinical models of liver and gastrointestinal cancers. Exp. Mol. Med. 2019, 51, 109. [Google Scholar] [CrossRef]
- Méndez-Blanco, C.; Fondevila, F.; García-Palomo, A.; González-Gallego, J.; Mauriz, J.L. Sorafenib resistance in hepatocarcinoma: Role of hypoxia-inducible factors. Exp. Mol. Med. 2018, 50, 134. [Google Scholar] [CrossRef]
- Rawat, D.; Shrivastava, S.; Naik, R.A.; Chhonker, S.K.; Mehrotra, A.; Koiri, R.K. An overview of natural plant products in the treatment of hepatocellular carcinoma. Anticancer. Agents Med. Chem. 2018, 18, 1838–1859. [Google Scholar] [CrossRef]
- Prieto-Domínguez, N.; Ordóñez, R.; Fernández, A.; Méndez-Blanco, C.; Baulies, A.; Garcia-Ruiz, C.; Fernández-Checa, J.C.; Mauriz, J.L.; González-Gallego, J. Melatonin-induced increase in sensitivity of human hepatocellular carcinoma cells to sorafenib is associated with reactive oxygen species production and mitophagy. J. Pineal Res. 2016, 61, 396–407. [Google Scholar] [CrossRef]
- Prieto-Domínguez, N.; Méndez-Blanco, C.; Carbajo-Pescador, S.; Fondevila, F.; García-Palomo, A.; González-Gallego, J.; Mauriz, J.L. Melatonin enhances sorafenib actions in human hepatocarcinoma cells by inhibiting mTORC1/p70S6K/HIF-1α and hypoxia-mediated mitophagy. Oncotarget 2017, 8, 91402–91414. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef]
- Wu, L.; Li, J.; Liu, T.; Li, S.; Feng, J.; Yu, Q.; Zhang, J.; Chen, J.; Zhou, Y.; Ji, J.; et al. Quercetin shows anti-tumor effect in hepatocellular carcinoma LM3 cells by abrogating JAK2/STAT3 signaling pathway. Cancer Med. 2019, 8, 4806–4820. [Google Scholar] [CrossRef]
- Wu, H.; Pan, L.; Gao, C.; Xu, H.; Li, Y.; Zhang, L.; Ma, L.; Meng, L.; Sun, X.; Qin, H. Quercetin inhibits the proliferation of glycolysis-addicted HCC cells by reducing hexokinase 2 and Akt-mTOR pathway. Molecules 2019, 24, 1993. [Google Scholar] [CrossRef]
- Ji, Y.; Li, L.; Ma, Y.-X.; Li, W.-T.; Li, L.; Zhu, H.-Z.; Wu, M.-H.; Zhou, J.-R. Quercetin inhibits growth of hepatocellular carcinoma by apoptosis induction in part via autophagy stimulation in mice. J. Nutr. Biochem. 2019, 69, 108–119. [Google Scholar] [CrossRef]
- Jeon, J.-S.; Kwon, S.; Ban, K.; Hong, Y.-K.; Ahn, C.; Sung, J.-S.; Choi, I. Regulation of the intracellular ROS level is critical for the antiproliferative effect of quercetin in the hepatocellular carcinoma cell line HepG2. Nutr. Cancer 2019, 71, 861–869. [Google Scholar] [CrossRef]
- Chen, Z.; Huang, C.; Ma, T.; Jiang, L.; Tang, L.; Shi, T.; Zhang, S.; Zhang, L.; Zhu, P.; Li, J.; et al. Reversal effect of quercetin on multidrug resistance via FZD7/β-catenin pathway in hepatocellular carcinoma cells. Phytomedicine 2018, 43, 37–45. [Google Scholar] [CrossRef]
- Ding, Y.; Chen, X.; Wang, B.; Yu, B.; Ge, J.; Shi, X. Quercetin suppresses the chymotrypsin-like activity of proteasome via inhibition of MEK1/ERK1/2 signaling pathway in hepatocellular carcinoma HepG2 cells. Can. J. Physiol. Pharmacol. 2018, 96, 521–526. [Google Scholar] [CrossRef]
- Kellett, M.E.; Greenspan, P.; Pegg, R.B. Modification of the cellular antioxidant activity (CAA) assay to study phenolic antioxidants in a Caco-2 cell line. Food Chem. 2018, 244, 359–363. [Google Scholar] [CrossRef]
- Shaalan, Y.M.; Handoussa, H.; Youness, R.A.; Assal, R.A.; El-Khatib, A.H.; Linscheid, M.W.; El Tayebi, H.M.; Abdelaziz, A.I. Destabilizing the interplay between miR-1275 and IGF2BPs by Tamarix articulata and quercetin in hepatocellular carcinoma. Nat. Prod. Res. 2018, 32, 2217–2220. [Google Scholar] [CrossRef]
- Pi, J.; Li, B.; Tu, L.; Zhu, H.; Jin, H.; Yang, F.; Bai, H.; Cai, H.; Cai, J. Investigation of quercetin-induced HepG2 cell apoptosis-associated cellular biophysical alterations by atomic force microscopy. Scanning 2016, 38, 100–112. [Google Scholar] [CrossRef]
- Maurya, A.K.; Vinayak, M. Anticarcinogenic action of quercetin by downregulation of phosphatidylinositol 3-kinase (PI3K) and protein kinase C (PKC) via induction of p53 in hepatocellular carcinoma (HepG2) cell line. Mol. Biol. Rep. 2015, 42, 1419–1429. [Google Scholar] [CrossRef]
- Zhang, Q.; Cheng, G.; Qiu, H.; Zhu, L.; Ren, Z.; Zhao, W.; Zhang, T.; Liu, L. The p53-inducible gene 3 involved in flavonoid-induced cytotoxicity through the reactive oxygen species-mediated mitochondrial apoptotic pathway in human hepatoma cells. Food Funct. 2015, 6, 1518–1525. [Google Scholar] [CrossRef]
- Lee, R.H.; Cho, J.H.; Jeon, Y.-J.; Bang, W.; Cho, J.-J.; Choi, N.-J.; Seo, K.S.; Shim, J.-H.; Chae, J. Quercetin induces antiproliferative activity against human hepatocellular carcinoma (HepG2) cells by suppressing specificity protein 1 (Sp1). Drug Dev. Res. 2015, 76, 9–16. [Google Scholar] [CrossRef]
- Dabbagh-Bazarbachi, H.; Clergeaud, G.; Quesada, I.M.; Ortiz, M.; O’Sullivan, C.K.; Fernández-Larrea, J.B. Zinc ionophore activity of quercetin and epigallocatechin-gallate: From Hepa 1-6 cells to a liposome model. J. Agric. Food Chem. 2014, 62, 8085–8093. [Google Scholar] [CrossRef]
- Kozics, K.; Valovicova, Z.; Slamenova, D. Structure of flavonoids influences the degree inhibition of Benzo(a)pyrene – induced DNA damage and micronuclei in HepG2 cells. Neoplasma 2011, 58, 516–524. [Google Scholar] [CrossRef]
- Oliva, J.; Bardag-Gorce, F.; Tillman, B.; French, S.W. Protective effect of quercetin, EGCG, catechin and betaine against oxidative stress induced by ethanol in vitro. Exp. Mol. Pathol. 2011, 90, 295–299. [Google Scholar] [CrossRef]
- Choi, K.-C.; Chung, W.-T.; Kwon, J.-K.; Yu, J.-Y.; Jang, Y.-S.; Park, S.-M.; Lee, S.-Y.; Lee, J.-C. Inhibitory effects of quercetin on aflatoxin B1-induced hepatic damage in mice. Food Chem. Toxicol. 2010, 48, 2747–2753. [Google Scholar] [CrossRef]
- Granado-Serrano, A.B.; Martín, M.A.; Bravo, L.; Goya, L.; Ramos, S. Quercetin modulates NF-κB and AP-1/JNK pathways to induce cell death in human hepatoma cells. Nutr. Cancer 2010, 62, 390–401. [Google Scholar] [CrossRef]
- Srisa-nga, K.; Mankhetkorn, S.; Okonogi, S.; Khonkarn, R. Delivery of superparamagnetic polymeric micelles loaded with quercetin to hepatocellular carcinoma cells. J. Pharm. Sci. 2019, 108, 996–1006. [Google Scholar] [CrossRef]
- AbouAitah, K.; Swiderska-Sroda, A.; Farghali, A.A.; Wojnarowicz, J.; Stefanek, A.; Gierlotka, S.; Opalinska, A.; Allayeh, A.K.; Ciach, T.; Lojkowski, W. Folic acid–conjugated mesoporous silica particles as nanocarriers of natural prodrugs for cancer targeting and antioxidant action. Oncotarget 2018, 9, 26466–26490. [Google Scholar] [CrossRef]
- Abd-Rabou, A.A.; Ahmed, H.H. CS-PEG decorated PLGA nano-prototype for delivery of bioactive compounds: A novel approach for induction of apoptosis in HepG2 cell line. Adv. Med. Sci. 2017, 62, 357–367. [Google Scholar] [CrossRef]
- Ren, K.-W.; Li, Y.-H.; Wu, G.; Ren, J.-Z.; Lu, H.-B.; Li, Z.-M.; Han, X.-W. Quercetin nanoparticles display antitumor activity via proliferation inhibition and apoptosis induction in liver cancer cells. Int. J. Oncol. 2017, 50, 1299–1311. [Google Scholar] [CrossRef]
- Bishayee, K.; Khuda-Bukhsh, A.R.; Huh, S.-O. PLGA-loaded gold-nanoparticles precipitated with quercetin downregulate HDAC-Akt activities controlling proliferation and activate p53-ROS crosstalk to induce apoptosis in hepatocarcinoma cells. Mol. Cells 2015, 38, 518–527. [Google Scholar] [CrossRef]
- Varshosaz, J.; Jafarian, A.; Salehi, G.; Zolfaghari, B. Comparing different sterol containing solid lipid nanoparticles for targeted delivery of quercetin in hepatocellular carcinoma. J. Liposome Res. 2013, 24, 191–203. [Google Scholar] [CrossRef]
- Kovacovicova, K.; Skolnaja, M.; Heinmaa, M.; Mistrik, M.; Pata, P.; Pata, I.; Bartek, J.; Vinciguerra, M. Senolytic cocktail dasatinib+quercetin (D+Q) does not enhance the efficacy of senescence-inducing chemotherapy in liver cancer. Front. Oncol. 2018, 8, 459. [Google Scholar] [CrossRef]
- Bahman, A.A.; Abaza, M.S.I.; Khoushiash, S.I.; Al-Attiyah, R.J. Sequence-dependent effect of sorafenib in combination with natural phenolic compounds on hepatic cancer cells and the possible mechanism of action. Int. J. Mol. Med. 2018, 42, 1695–1715. [Google Scholar] [CrossRef]
- Zou, H.; Zheng, Y.; Ge, W.; Wang, S.; Mou, X. Synergistic anti-tumour effects of quercetin and oncolytic adenovirus expressing TRAIL in human hepatocellular carcinoma. Sci. Rep. 2018, 8, 2182. [Google Scholar] [CrossRef]
- Igbe, I.; Shen, X.-F.; Jiao, W.; Qiang, Z.; Deng, T.; Li, S.; Liu, W.-L.; Liu, H.-W.; Zhang, G.-L.; Wang, F. Dietary quercetin potentiates the antiproliferative effect of interferon-α in hepatocellular carcinoma cells through activation of JAK/STAT pathway signaling by inhibition of SHP2 phosphatase. Oncotarget 2017, 8, 113734–113748. [Google Scholar] [CrossRef]
- Carrasco-Torres, G.; Baltiérrez-Hoyos, R.; Andrade-Jorge, E.; Villa-Treviño, S.; Trujillo-Ferrara, J.G.; Vásquez-Garzón, V.R. Cytotoxicity, oxidative stress, cell cycle arrest, and mitochondrial apoptosis after combined treatment of hepatocarcinoma cells with maleic anhydride derivatives and quercetin. Oxid. Med. Cell. Longev. 2017, 2017, 2734976. [Google Scholar] [CrossRef]
- Yu, C.-P.; Qiu, R.-G.; Shi, L.; Liang, J. Celecoxib and quercetin induce apoptosis in human hepatocarcinoma. Biomed. Res. 2017, 28, 3465–3470. [Google Scholar]
- Brito, A.F.; Ribeiro, M.; Abrantes, A.M.; Mamede, A.C.; Laranjo, M.; Casalta-Lopes, J.E.; Gonçalves, A.C.; Sarmento-Ribeiro, A.B.; Tralhão, J.G.; Botelho, M.F. New approach for treatment of primary liver tumors: The role of quercetin. Nutr. Cancer 2016, 68, 250–266. [Google Scholar] [CrossRef]
- Dai, W.; Gao, Q.; Qiu, J.; Yuan, J.; Wu, G.; Shen, G. Quercetin induces apoptosis and enhances 5-FU therapeutic efficacy in hepatocellular carcinoma. Tumor Biol. 2016, 37, 6307–6313. [Google Scholar] [CrossRef]
- Zhao, J.; Zhao, J.; Jiao, H. Synergistic growth-suppressive effects of quercetin and cisplatin on HepG2 human hepatocellular carcinoma cells. Appl. Biochem. Biotechnol. 2014, 172, 784–791. [Google Scholar] [CrossRef]
- Sharma, A.; Bhat, M.K. Enhancement of carboplatin- and quercetin-induced cell death by roscovitine is Akt dependent and p53 independent in hepatoma cells. Integr. Cancer Ther. 2011, 10, 4–14. [Google Scholar] [CrossRef]
- Abdelmoneem, M.A.; Elnaggar, M.A.; Hammady, R.S.; Kamel, S.M.; Helmy, M.W.; Abdulkader, M.A.; Zaky, A.; Fang, J.-Y.; Elkhodairy, K.A.; Elzoghby, A.O. Dual-targeted lactoferrin shell-oily core nanocapsules for synergistic targeted/herbal therapy of hepatocellular carcinoma. Appl. Mater. Interfaces 2019, 11, 26731–26744. [Google Scholar] [CrossRef]
- Wang, C.; Su, L.; Wu, C.; Wu, J.; Zhu, C.; Yuan, G. RGD peptide targeted lipid coated nanoparticles for combinatorial delivery of sorafenib and quercetin against hepatocellular carcinoma. Drug Dev. Ind. Pharm. 2016, 42, 1938–1944. [Google Scholar] [CrossRef]
- Lee, Y.-J.; Beak, S.-Y.; Choi, I.; Sung, J.-S. Quercetin and its metabolites protect hepatocytes against ethanol-induced oxidative stress by activation of Nrf2 and AP-1. Food Sci. Biotechnol. 2017, 27, 809–817. [Google Scholar] [CrossRef]
- Liu, Y.; Kurita, A.; Nakashima, S.; Zhu, B.; Munemasa, S.; Nakamura, T.; Murata, Y.; Nakamura, Y. 3,4-Dihydroxyphenylacetic acid is a potential aldehyde dehydrogenase inducer in murine hepatoma Hepa1c1c7 cells. Biosci. Biotechnol. Biochem. 2017, 81, 1978–1983. [Google Scholar] [CrossRef]
- Sudan, S.; Rupasinghe, V. Antiproliferative activity of long chain acylated esters of quercetin-3-O-glucoside in hepatocellular carcinoma HepG2 cells. Exp. Biol. Med. 2015, 240, 1452–1464. [Google Scholar] [CrossRef]
- Sudan, S.; Rupasinghe, H.P.V. Quercetin-3-O-glucoside induces human DNA topoisomerase II inhibition, cell cycle arrest and apoptosis in hepatocellular carcinoma cells. Anticancer Res. 2014, 34, 1691–1700. [Google Scholar]
- Houghton, M.J.; Kerimi, A.; Tumova, S.; Boyle, J.P.; Williamson, G. Quercetin preserves redox status and stimulates mitochondrial function in metabolically-stressed HepG2 cells. Free Radic. Biol. Med. 2018, 129, 296–309. [Google Scholar] [CrossRef]
- San-Miguel, B.; Crespo, I.; Sánchez, D.I.; González-Fernández, B.; Ortiz De Urbina, J.J.; Tuñón, M.J.; González-Gallego, J. Melatonin inhibits autophagy and endoplasmic reticulum stress in mice with carbon tetrachloride-induced fibrosis. J. Pineal Res. 2015, 59, 151–162. [Google Scholar] [CrossRef]
- Kim, D.H.; Khan, H.; Ullah, H.; Hassan, S.T.S.; Šmejkal, K.; Efferth, T.; Mahomoodally, M.F.; Xu, S.; Habtemariam, S.; Filosa, R.; et al. MicroRNA targeting by quercetin in cancer treatment and chemoprotection. Pharmacol. Res. 2019, 147, 104346. [Google Scholar] [CrossRef]
- Klimaszewska-Wisniewska, A.; Hałas-Wisniewska, M.; Izdebska, M.; Gagat, M.; Grzanka, A.; Grzanka, D. Antiproliferative and antimetastatic action of quercetin on A549 non-small cell lung cancer cells through its effect on the cytoskeleton. Acta Histochem. 2017, 119, 99–112. [Google Scholar] [CrossRef]
- Ranganathan, S.; Halagowder, D.; Devaraj Sivasithambaram, N. Quercetin suppresses Twist to induce apoptosis in MCF-7 breast cancer cells. PLoS ONE 2015, 10, e0141370. [Google Scholar] [CrossRef]
- Nguyen, L.T.; Lee, Y.-H.; Sharma, A.R.; Park, J.-B.; Jagga, S.; Sharma, G.; Lee, S.-S.; Nam, J.-S. Quercetin induces apoptosis and cell cycle arrest in triple-negative breast cancer cells through modulation of Foxo3a activity. Korean J. Physiol. Pharmacol. 2017, 21, 205–213. [Google Scholar] [CrossRef]
- Gao, B.; Wang, H.; Lafdil, F.; Feng, D. STAT proteins–key regulators of anti-viral responses, inflammation, and tumorigenesis in the liver. J. Hepatol. 2012, 57, 430–441. [Google Scholar] [CrossRef]
- Tang, Q.; Ji, F.; Wang, J.; Guo, L.; Li, Y.; Bao, Y. Quercetin exerts synergetic anti-cancer activity with 10-hydroxy camptothecin. Eur. J. Pharm. Sci. 2017, 109, 223–232. [Google Scholar] [CrossRef]
- Sarkar, A.; Ghosh, S.; Chowdhury, S.; Pandey, B.; Sil, P.C. Targeted delivery of quercetin loaded mesoporous silica nanoparticles to the breast cancer cells. Biochim. Biophys. Acta 2016, 1860, 2065–2075. [Google Scholar] [CrossRef]
- Martino, R.; Arcos, M.L.B.; Alonso, R.; Sülsen, V.; Cremaschi, G.; Anesini, C. Polyphenol-rich fraction from Larrea divaricata and its main flavonoid quercetin-3-methyl ether induce apoptosis in lymphoma cells through nitrosative stress. Phyther. Res. 2016, 30, 1128–1136. [Google Scholar] [CrossRef]
- Cao, L.; Yang, Y.; Ye, Z.; Lin, B.; Zeng, J.; Li, C.; Liang, T.; Zhou, K.; Li, J. Quercetin-3-methyl ether suppresses human breast cancer stem cell formation by inhibiting the Notch1 and PI3K/Akt signaling pathways. Int. J. Mol. Med. 2018, 42, 1625–1636. [Google Scholar] [CrossRef]
- Mostafavi-Pour, Z.; Ramezani, F.; Keshavarzi, F.; Samadi, N. The role of quercetin and vitamin C in Nrf2-dependent oxidative stress production in breast cancer cells. Oncol. Lett. 2017, 13, 1965–1973. [Google Scholar] [CrossRef]
- Sharmila, G.; Bhat, F.A.; Arunkumar, R.; Elumalai, P.; Singh, P.R.; Senthilkumar, K.; Arunakaran, J. Chemopreventive effect of quercetin, a natural dietary flavonoid on prostate cancer in in vivo model. Clin. Nutr. 2014, 33, 718–726. [Google Scholar] [CrossRef]
- Lan, H.; Hong, W.; Fan, P.; Qian, D.; Zhu, J.; Bai, B. Quercetin inhibits cell migration and invasion in human osteosarcoma cells. Cell. Physiol. Biochem. 2017, 43, 553–567. [Google Scholar] [CrossRef]
- Niazvand, F.; Orazizadeh, M.; Khorsandi, L.; Abbaspour, M.; Mansouri, E.; Khodadadi, A. Effects of quercetin-loaded nanoparticles on MCF-7 human breast cancer cells. Medicina 2019, 55, 114. [Google Scholar] [CrossRef]
- Xu, G.; Shi, H.; Ren, L.; Gou, H.; Gong, D.; Gao, X.; Huang, N. Enhancing the anti-colon cancer activity of quercetin by self-assembled micelles. Int. J. Nanomed. 2015, 10, 2051–2063. [Google Scholar]
- Thao, L.Q.; Lee, C.; Kim, B.; Lee, S.; Kim, T.H.; Kim, J.O.; Lee, E.S.; Oh, K.T.; Choi, H.-G.; Yoo, S.D.; et al. Doxorubicin and paclitaxel co-bound lactosylated albumin nanoparticles having targetability to hepatocellular carcinoma. Colloids Surf. B Biointerfaces 2017, 152, 183–191. [Google Scholar] [CrossRef]
- Yang, F.; Song, L.; Wang, H.; Wang, J.; Xu, Z.; Xing, N. Combination of quercetin and 2-methoxyestradiol enhances inhibition of human prostate cancer LNCaP and PC-3 cells xenograft tumor growth. PLoS ONE 2015, 10, e0128277. [Google Scholar] [CrossRef]
- Chen, Q.; Li, P.; Li, P.; Xu, Y.; Li, Y.; Tang, B. Isoquercitrin inhibits the progression of pancreatic cancer in vivo and in vitro by regulating opioid receptors and the mitogen-activated protein kinase signalling pathway. Oncol. Rep. 2015, 33, 840–848. [Google Scholar] [CrossRef]
- Yamauchi, K.; Afroze, S.H.; Mitsunaga, T.; McCormick, T.; Kuehl, T.; Zawieja, D.C.; Uddin, M.N. 3,4’,7-O-trimethylquercetin inhibits invasion and migration of ovarian cancer cells. Anticancer Res. 2017, 37, 2823–2829. [Google Scholar]
| First Author, Year of Publication | Quercetin Administration Strategy | Cell Line | General Effects | Molecular Alterations | Altered Signaling Pathways |
|---|---|---|---|---|---|
| Wu, 2019 [22] | Quercetin | LM3 cells | Cell viability reduction Apoptosis induction Cell cycle arrest at S and G2/M phases Autophagy induction Cell migration and invasion suppression Morphological changes | ↑ Early stage apoptotic cells ↓ PCNA mRNA and protein levels ↑ Bax mRNA and protein levels ↓ Colony formation ↑ Fluorescence in TUNEL staining ↓ G0/G1 phase cells and ↑ S and G2/M phase cells ↓ Cyclin B1 protein expression ↑ E-cadherin and ↓ vimentin and MMP-9 mRNA and protein levels ↓ N-cadherin protein expression ↓ Invasiveness (Transwell invasion assay) ↓ Migrating cells (Wound-healing assay) ↑ LC3 mRNA and protein levels ↑ Beclin1 protein expression ↓ p62 mRNA and protein levels ↓ p-STAT3 protein expression ↑ LC3 protein levels decreased by IL-6 ↓ PCNA and MMP-9 protein levels enhanced by IL-6 ↓ Wound healing speed | JAK2/STAT3 inhibition |
| Wu, 2019 [23] | Quercetin | SMMC-7721, BEL-7402 HCC cells LO2 normal liver cells | Proliferation suppression of HCC cell lines No cytotoxic for normal hepatic cells Glycolysis inhibition | ↓ Glucose uptake and lactate production ↑ 2-DG-derived cell growth inhibition ↓ HK2 mRNA and protein expression ↓ p-Akt/Akt and p-mTOR/mTOR rates | Akt/mTOR inhibition |
| Ji, 2019 [24] | Quercetin | SMMC-7721 and HepG2 HCC cells LO2 normal hepatic cells | Cell growth inhibition in HCC cell lines Absence of antiproliferation effect in LO2 cells Induction of autophagy Apoptosis increase | In both tumor cell lines: ↑ LC3A/LC3B-II and Beclin1 protein levels ↓ p62 protein expression In SMMC-7721: ↑ Autophagosomes and autolysosomes In all cell lines: ↓ p-Akt, p-mTOR, p-p70S6K and p-4EBP1 protein levels ↑ p-JNK, p-ERK1/2 and p-p38 MAPK protein expression ↑ Apoptotic cells percentage ↑ Bax and cleaved caspase-3 protein levels ↓ Bcl-2 protein expression | Akt/mTOR inhibition MAPK activation |
| Jeon, 2019 [25] | Quercetin | HepG2, HuH7, PLC/PRF-5 and Hep3B cells | Proliferation inhibition (in HepG2, PLC/PRF-5 and Hep3B cells) ROS levels reduction (in HepG2 cells) Morphological alterations | Only in HepG2 cell line: ↑ p53 and HO-1 protein expression ↓ Cyclin A and CHK1 protein levels No variation in cyclin E and SOD1 protein expression | - |
| Chen, 2018 [26] | Quercetin | BEL-7402 HCC cells Multidrug resistant cell line BEL/5-FU | Increase of 5-FU, MMC and ADR chemosensitivity in BEL/5-FU cells | Only in BEL/5-FU cell line: ↓ IC50 of 5-FU, MMC and ADR ↓ ABCB1, ABCC1 and ABCC2 mRNA levels ↑ Rh123 accumulation Inhibition of ABCC2 function ↑ ADR accumulation In both cell lines: ↓ FZD7, β-catenin (nuclear and cytoplasmic), ABCB1, ABCC1 and ABCC2 mRNA and protein expression | FZD7/β-catenin inhibition |
| Ding, 2018 [27] | Quercetin | HepG2 HCC cell line | Decrease of cell viability Apoptosis induction Inhibition of chymotrypsin-like activity | ↑ TUNEL-positive cells ↑ Cleaved caspase-3, cleaved PARP and Bax protein expression ↓ Bcl-2 protein levels ↓ Chymotrypsin-like activity No changes in trypsin-like and caspase-like activities ↑ p-p38 MAPK and JNK protein expression ↓ p-ERK1/2 protein levels ↓ Protein expression of β1, β2 and β5 proteasomal subunits | MEK1/ERK1/2 inhibition |
| Kellet, 2018 [28] | Quercetin | HepG2 cells | Antioxidant activity | ↑ CAA unit dose dependent | - |
| Shaalan, 2018 [29] | Quercetin | HuH7 cell line | - | ↑ miR-1275 mRNA levels ↓ IGF2BP1 and IGF2BP3 mRNA expression | - |
| Pi, 2016 [30] | Quercetin | HepG2 cells | Suppression of cell proliferation Cell cycle arrest at G2/M phase Apoptosis increase Disruption of mitochondrial membrane potential Morphological alterations Changes in surface ultrastructure | ↑ G2/M cell population ↑ Early apoptotic, late apoptotic and necrotic cells ↓ Fluorescence signal of Rh123 ↑ F-actin filaments aggregation in apoptotic cells ↑ Particle size on HepG2 membrane ↑ Surface root-mean-squared and surface average roughness ↑ Cell stiffness and Young’s modulus | - |
| Maurya, 2015 [31] | Quercetin | HepG2 cells | Antiproliferative activity Morphological changes | ↓ ROS generation and PKC activity ↓ p-p85α, p-PKC, PKCα, COX-2 protein levels ↑ p53 protein expression and Bax mRNA levels | PI3K/p53/COX-2 and PKC/p53/COX-2 inhibition |
| Zhang, 2015 [32] | Quercetin | HepG2 cells | Cell viability inhibition Induction of apoptosis | Chromatin condensation and nuclei fragmentation into oligonucleosomes ↑ PIG3 mRNA and protein expression ↑ Early apoptotic cells ↑ ROS accumulation ↓ Mitochondrial membrane potential ↓ Mitochondrial cytochrome c and Bcl-2 protein expression ↑ Cytosolic cytochrome c, Bax and activated caspases -9 and -3 | - |
| Lee, 2015 [33] | Quercetin | HepG2 cells | Decrease in cell viability Apoptosis induction | ↑ Nuclear condensation and fragmentation ↑ Early and late apoptotic cells ↓ Sp1 mRNA and protein levels ↑ p21, p27, Bax, cleaved caspase-3 and cleaved PARP protein expression ↓ Cyclin D1, Mcl-1, survivin and Bcl-xL protein levels | - |
| Dabbagh-Bazarbachi, 2014 [34] | Quercetin | Mouse hepatoma Hepa 1-6 cell line | Augmented cytoplasmic labile zinc High ionophore activity | ↑ FluoZin-3-detectable zinc ↑ Fluorescence signal of FluoZin-3 | - |
| Kozics, 2011 [35] | Quercetin | HepG2 cells | Reduction of cell proliferation | ↓ B(a)P-induced micronuclei formation and DNA damage | - |
| Oliva, 2011 [36] | Quercetin | Cederbaum’s CYP2E1 overexpressing HepG2 cell line | Decrease of ethanol-derived oxidative stress | ↓ MDA, 4-HNE and carbonyl protein levels augmented by ethanol ↓ Ethanol-induced glutathione peroxidase 4 and SOD2 mRNA expression ↓ Gadd45b mRNA levels ↑ Nrf2 protein levels reduced by ethanol | - |
| Choi, 2010 [37] | Quercetin | HepG2 cells | Reduction of the AFB1 antiproliferative effect | ↓ ROS accumulation generated by AFB1 ↑ AFB1-reduced GSH levels | - |
| Granado-Serrano, 2010 [38] | Quercetin | HepG2 cells | Cell proliferation suppression | ↓ NF-κB and p65 nuclear translocation, NF-κB DNA-binding activity ↓ p-IκBα and IKKα protein expression ↓ Chymotrypsin-like activity No changes in trypsin-like activity ↑ DNA-binding activity of AP-1 ↑ Nuclear c-Jun levels | NF-κB inhibition AP-1/JNK activation |
| Srisa-nga, 2019 [39] | Quercetin encapsulation (Quercetin-SPION-loaded micelles) | HepG2.2.15 cell line | Suppression of cell growth Morphological alterations | ↑ G0/G1 and ↓G2/M phase cells | - |
| AbouAitah, 2018 [40] | Quercetin encapsulation (FA-conjugated MSNs) | HepG2 cells | Increased antiproliferative activity | ↑ Antioxidant effect ↑ Inhibition of ABTS.+ radical formation | - |
| Abd-Rabou, 2017 [41] | Quercetin encapsulation (CS and PEG-decorated PLGA nano-prototypes) | HepG2 cells | Cell viability reduction Apoptosis induction | ↓ Quercetin IC50 ↑ Late apoptotic and necrotic cells | - |
| Ren, 2017 [42] | Quercetin encapsulation (PLGA-loaded gold-quercetin nanoparticles) | MHCC97H, Hep3B, HCCLM3 and BEL-7402 HCC cell lines | Decreased cell proliferation Only in MHCC97H cell line: Morphological alterations Reduction of cell migration ability Apoptosis increase | Only in MHCC97H line: ↓ Colony formation ↑ Cell-to-cell adhesions and ↓ filopodia generation and cell spreading ↓ Migrating cells ↑ P-27 protein levels ↓ c-Myc, cyclin D1, CDK1, MMP-7 and β-catenin protein expression ↑ Apoptotic cell number ↑ Cleaved caspases -9 and -3 protein levels ↑ Cytochrome c release to cytoplasm ↓ hTERT and AP-2β mRNA and protein expression ↓ hTERT promoter-binding activity of AP-2β ↓ COX-2 protein expression ↓ Binding activity of p50 on COX-2 promoter ↓ p-IKKα and p-IκBα protein levels ↑ NF-κB and p50 cytoplasm translocation from nuclei ↓ p-Akt and p-ERK1/2 protein levels | AP-2β/hTERT inhibition p50/NF-κB/COX-2 inhibition Akt/ERK1/2 inhibition |
| Bishayee, 2015 [43] | Quercetin encapsulation (PLGA-loaded gold-quercetin nanoparticles) | HepG2 cells | Inhibition of cell proliferation Growth rate reduction Apoptosis stimulation Morphological changes | Alteration of B-conformation of DNA ↓ p-Akt protein expression ↑ sub G-phase cells and ↓ S-phase cells ↑ p21 protein levels ↓ CDK1 and cyclin D1 protein expression ↓ HDAC activity and HDAC1/2 protein levels ↑ ROS formation ↑ rac-1 activity and later returned to basal levels Depolarization of mitochondrial membrane Bax translocation to the mitochondrial outer membrane ↑ Cytochrome c release to cytosol Generation of DNA damage ↓ Mcl-1, Bcl-2 and Bcl-xL protein levels ↑ Apaf1, caspases -9 and -3, and cleaved PARP protein expression | - |
| Varshosaz, 2013 [44] | Quercetin encapsulation (SLNs containing cholesterol, stigmastanol or stigmasterol) | HepG2 cells | Cell growth inhibition (the highest with cholesterol) | - | - |
| Kovacovicova, 2018 [45] | Quercetin combined with dasatinib | HepG2 and HuH7 cell lines | No senolytic activity exhibited | No effects in β-galactosidase activity No protein expression alteration of the senescence markers p16 and γH2A.X | - |
| Bahman, 2018 [46] | Quercetin | HepG2 and Hep3B cells | Antiproliferative effect | - | - |
| Quercetin combined with sorafenib | Suppression of cell proliferation | - | - | ||
| Zou, 2018 [47] | Quercetin combined with ZD55-TRAIL | SMMC-7721, HepG2 and HuH7 cell lines | Decrease of cell proliferation Apoptosis induction | ↑ Apoptotic bodies, nuclear fragmentation and chromatin condensation ↑ Cleaved caspases -9 and -3, cleaved PARP, Bid and Bax protein expression ↓ Bcl-2 and FLIP protein levels ↓ IκBα, p65 and p50 protein expression | NF-κB inhibition |
| Igbe, 2017 [48] | Quercetin | HepG2 and HuH7 HCC cell lines | Inhibition of cell viability | ↓ SHP-1 and SHP-2 protein expression in HepG2 cells | - |
| Quercetin combined with IFN-α | Increased cell growth inhibition in both HCC cell lines | Only in HepG2 cell line: ↓ SHP-2 protein expression ↑ p-STAT1, p-Jak1 and p-Tyk2 protein levels ↑ ISRE reporter expression ↑ 2′,5′-OAS and PKR mRNA levels ↓ Colony formation ↓ Cyclin D1 protein expression | JAK/STAT activation via SHP2 inhibition | ||
| Carrasco-Torres, 2017 [49] | Quercetin | HuH7 and HepG2 HCC cells | Antiproliferative effect Cell cycle arrest at G0/G1 phase | ↑ G0/G1 cell population ↓ ROS levels and oxidized glutathione levels ↑ Reduced glutathione and GSH/GSSG index ↑ Nuclear condensation ↑ Pro-caspase-9 and cleaved caspases -9 and -3 protein expression | - |
| Quercetin combined with 3′5′-dimaleamylbenzoic acid or 3′5′-dimaleimylbenzoic acid | Cell viability reduction Cell cycle arrest at S phase Antioxidant activity Apoptosis induction | In both cell lines: ↓ G2/M-phase and ↑ S-phase populations ↓ Reduced and oxidized glutathione levels and GSH/GSSG index in both cell lines (maleic anhydride derivative + quercetin) ↑ Nuclear condensation, degradation of actin and DNA ↑ Pyknotic nuclei number and TUNEL-positive cells ↑ Pro-caspase-9 and cleaved caspases -9 and -3 protein expression In HuH7 line: ↓ ROS levels In HepG2 line: ↓ ROS levels (quercetin + maleic anhydride derivative) ↑ ROS levels (maleic anhydride derivative + quercetin) ↑ Reduced glutathione levels and de novo glutathione synthesis (quercetin + maleic anhydride derivative) | - | ||
| Yu, 2017 [50] | Quercetin combined with celecoxib | HepG2 and HuH7 cell lines | Antiproliferative effect Apoptosis induction | ↑ DNA fragmentation ↑ Bax protein expression ↓ Bcl-2 protein levels | - |
| Brito, 2016 [51] | Quercetin | HepG2, HuH7 and Hep3B2.1-7 HCC cell lines | Inhibition of cell growth and survival Apoptosis increase Cell cycle arrest | ↑ Apoptotic and necrotic cells ↑ Bax/Bcl-2 ratio ↑ G0/G1 and G2/M cell population in HepG2 and HuH7 ↓ S phase cells in all cell lines ↓ p53 protein expression in HepG2 and HuH7 cells ↑ DNA damage ↑ Membrane expression of GLUT-1 ↓ Cytoplasmic expression of GLUT-1 in HepG2 and HuH7 cells ↓ 18F-FDG uptake | - |
| Quercetin combined with sorafenib | Decrease in sorafenib IC50 | - | |||
| Dai, 2016 [52] | Quercetin | HepG2 and SMMC-7721 HCC cells | Suppression of cell proliferation Cell cycle arrest at G0/G1 phase Apoptosis increase | ↑ G0/G1 phase and ↓S phase cell population ↑ Bax and Bad protein expression ↓ Bcl-2 and surviving protein levels | - |
| Quercetin combined with 5-FU | Rise of 5-FU antiproliferative effects Higher apoptotic activity | - | - | ||
| Zhao, 2014 [53] | Quercetin | HepG2 cells | Inhibition of cell survival Apoptosis induction G1-phase arrest of cell cycle | ↑ Cleaved caspase-3 and cleaved PARP protein levels ↑ p21, p53 and p16 protein expression ↑ G1-phase cells and ↓ S-phase cells ↑ sub-G1 cell population | - |
| Quercetin combined with cisplatin | Increased growth inhibitory action Greater apoptotic effects | ↑ Cleaved caspase-3 and cleaved PARP protein levels ↑ p21 and p53 protein levels | |||
| Sharma, 2011 [54] | Quercetin | HepG2 and Hep3B cell lines | Reduced cell survival Morphological changes Apoptosis induction | ↑ Apoptotic bodies ↑ p53 protein expression in HepG2 cells ↓ Pro-caspase-9 and ↑ caspase-9 protein levels in HepG2 cells | - |
| Quercetin combined with roscovitine | Augmented cell proliferation inhibition Morphological alterations Apoptosis stimulation | ↓ Cell density ↑ Floating cells number and apoptotic bodies ↓ p-Akt, Bcl-2 and pro-caspases -9 and -3 protein expression ↓ Bcl-2/Bax ratio and ↑ Caspases -9 and -3 protein levels | - | ||
| Abdelmoneem, 2019 [55] | Co-encapsulated quercetin and sorafenib (LF-coated, LA/LF-coated or GA/LF-coated nanocapsules) | HepG2 cells | Higher antitumoral efficacy of quercetin and sorafenib Cell viability suppression | ↓ IC50 of quercetin and sorafenib ↓ Combination index ↑ Dose reduction index of quercetin and sorafenib ↑ Cellular uptake of both drugs | - |
| Wang, 2016 [56] | Co-encapsulated quercetin and sorafenib (RGD-modified lipid-coated nanoparticles) | HepG2 cells | Reduced cell proliferation | ↓ IC50 of quercetin and sorafenib | - |
| Lee, 2017 [57] | Quercetin | HepG2 cells | Reduced antiproliferative action of ethanol Antioxidant activity | Reversal of ethanol effects: ↓ ROS formation ↓ MDA levels ↑ GSH, SOD and CAT levels ↑ HO-1 and nuclear Nrf2 protein expression ↑ AP-1 activity | Nrf2/HO-1 activation AP-1/HO-1 activation |
| 3′MQ | Lower ethanol-induced cell viability inhibition Antioxidant activity | Reversal of ethanol effects: ↓ ROS formation ↑ SOD and CAT levels ↑ HO-1 and nuclear Nrf2 protein expression ↑ AP-1 activity | Nrf2/HO-1 activation AP-1/HO-1 activation | ||
| Q3GA | Reversion of proliferation suppression induced by ethanol Antioxidant activity | Reversal of ethanol effects: ↓ ROS formation and ↑ GSH, SOD and CAT levels ↑ HO-1 and nuclear Nrf2 protein expression ↑ AP-1 activity | Nrf2/HO-1 activation AP-1/HO-1 activation | ||
| Liu, 2017 [58] | DOPAC | Mouse hepatoma Hepa1c1c7 cell line | Reduced acetaldehyde-derived cell growth inhibition | ↑ ALDH activity ↑ ALDH1A1, ALDH2 and ALDH3A1 mRNA and protein levels ↑ Nrf2 and AhR total and nuclear protein expression ↓ NF-κB nuclear expression | Nrf2 activation AhR activation NF-κB inhibition |
| Sudan, 2015 [59] | Six Q3G esters: Stearic acid ester Oleic acid ester Linoleic acid ester Alpha-linoleic acid ester Eicosapentaenoic acid ester Docosahexanoic acid ester | HepG2 HCC cells and normal hepatocytes | Higher cell viability of normal hepatocytes In HepG2 cells: Cell proliferation decrease Morphology changes Apoptosis induction Activity as catalytic inhibitor by DNA relaxation activity blockade | In HepG2 cells: ↓ HepG2 cell number ↑ DNA fragmentation ↑ Caspase-3 activity ↑ S and G2/M cell population ↓ G0/G1-phase cells No stabilization of topoisomerase II cleavage complexes and no formation of single linear DNA ↑ Supercoiled DNA intensity | - |
| Sudan, 2014 [60] | Q3G | HepG2 cell line | Cell growth suppression S-phase arrest of cell cycle Morphology alterations Apoptosis induction Catalytic inhibitor action by DNA relaxation activity inhibition | ↑ S-phase and ↓ G0/G1 cell percentage ↑ DNA fragmentation ↑ Caspase-3 activity ↑ Apoptotic and necrotic cells No stabilization of topoisomerase II cleavage complexes and no formation of single linear DNA ↑ Supercoiled DNA intensity | - |
| First Author, Year of Publication | Quercetin Administration Strategy, Dose and Administration Route | Animal Model | General Effects | Molecular Alterations | Altered Signaling Pathways |
|---|---|---|---|---|---|
| Wu, 2019 [22] | Quercetin 100 mg/kg Oral gavage | Nude mice subcutaneously injected with LM3 HCC cells | Tumor growth inhibition | ↓ Tumor volume (70% vs. control) ↓ Mouse weight and tumor volume ↑ Necrosis ↑ TUNEL-positive cells ↓ PCNA protein levels ↑ Bax and Beclin1 protein levels | - |
| Wu, 2019 [23] | Quercetin 50 mg/kg Intraperitoneal injection | SMMC-7721 xenograft mouse model | Tumor growth inhibition | ↓ Tumor size ↓ HK2 and Ki67 protein expression ↓ p-Akt and p-mTOR protein levels | Akt/mTOR inhibition |
| Ji, 2019 [24] | Quercetin 60 mg/kg Oral gavage | Nude mice subcutaneously injected with SMMC-7221 HCC cells | Suppression of tumor growth Apoptosis and autophagy induction | ↓ Tumor weight and volume ↑ LC3A/LC3B and ↓ p62 protein levels ↑ Necrosis and TUNEL staining ↑ Bax and cleaved caspase-3 protein levels ↓ Bcl-2 protein expression | - |
| Ren, 2017 [42] | Quercetin encapsulation (PLGA-loaded gold-quercetin nanoparticles) 30, 40 and 50 mg/kg Intraperitoneal injection | MHCC97H xenograft mouse model | Suppression of tumor growth and progression Apoptosis increase | ↓ Tumor weight and volume ↓ AP-2β and COX-2 protein levels ↑ TUNEL-positive cells ↓ Cleaved caspases -9 and -3, cytoplasmic cytochrome c, p-IKKα, p-IκBα, p-NF-κB, p50, hTERT, p-Akt, Raf, and p-ERK1/2 protein expression | AP-2β/hTERT inhibition p50/NF-κB/COX-2 inhibition Akt/ERK1/2 inhibition |
| Kovacovicova, 2018 [45] | Quercetin combined with dasatinib 50 mg/kg of quercetin with 5 mg/kg of dasatinib Oral gavage | Mice subcutaneously injected with HuH7 cells | Absence of tumor growth inhibition | - | - |
| Zou, 2018 [47] | Quercetin combined with ZD55-TRAIL 150 mg/kg of quercetin with 1 × 109 plaque-forming units of ZD55-TRAIL Intragastrical injection of quercetin and intratumor injection of ZD55-TRAIL | HuH7 xenograft mouse model | Tumor growth inhibition | ↓ Tumor volume | - |
| Dai, 2016 [52] | Quercetin 40 mg/kg of quercetin with 30 mg/kg of 5-FU Intraperitoneal injection | Nude mice subcutaneously injected with HepG2 HCC cells | Decreased tumor progression | ↓ Tumor volume | - |
| Quercetin combined with 5-FU 40 mg/kg of quercetin with 30 mg/kg of 5-FU Intraperitoneal injection | Higher tumor growth inhibition | ↓ Tumor volume | - | ||
| Abdelmoneem, 2019 [55] | Co-encapsulated quercetin and sorafenib (LF-coated, LA/LF-coated or GA/LF-coated nanocapsules) 10 mg/kg of quercetin and sorafenib Intravenous injection | DEN-induced HCC in a rat model | Antiangiogenic activity Apoptosis induction Liver weight reduction | ↓ NF-κB and TNF-α mRNA expression ↓ VEGF and Ki67 protein expression ↑ Caspase-3 protein expression ↓ ALT levels by LF-coated nanocapsules ↓ ALT, AST and RLW levels by LA/LF-coated and GA/LF-coated nanoparticles Improved histological features | NF-κB inhibition |
| Wang, 2016 [56] | Co-encapsulated quercetin and sorafenib (RGD-modified lipid-coated nanoparticles) 40 mg/kg of quercetin alone In combination: 20 mg/kg of quercetin with 10 mg/kg of sorafenib Intravenous injection | HepG2 xenograft mouse model | Tumor progression suppression | ↓ Tumor volume | - |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fernández-Palanca, P.; Fondevila, F.; Méndez-Blanco, C.; Tuñón, M.J.; González-Gallego, J.; Mauriz, J.L. Antitumor Effects of Quercetin in Hepatocarcinoma In Vitro and In Vivo Models: A Systematic Review. Nutrients 2019, 11, 2875. https://doi.org/10.3390/nu11122875
Fernández-Palanca P, Fondevila F, Méndez-Blanco C, Tuñón MJ, González-Gallego J, Mauriz JL. Antitumor Effects of Quercetin in Hepatocarcinoma In Vitro and In Vivo Models: A Systematic Review. Nutrients. 2019; 11(12):2875. https://doi.org/10.3390/nu11122875
Chicago/Turabian StyleFernández-Palanca, Paula, Flavia Fondevila, Carolina Méndez-Blanco, María J. Tuñón, Javier González-Gallego, and José L. Mauriz. 2019. "Antitumor Effects of Quercetin in Hepatocarcinoma In Vitro and In Vivo Models: A Systematic Review" Nutrients 11, no. 12: 2875. https://doi.org/10.3390/nu11122875
APA StyleFernández-Palanca, P., Fondevila, F., Méndez-Blanco, C., Tuñón, M. J., González-Gallego, J., & Mauriz, J. L. (2019). Antitumor Effects of Quercetin in Hepatocarcinoma In Vitro and In Vivo Models: A Systematic Review. Nutrients, 11(12), 2875. https://doi.org/10.3390/nu11122875








