Effect of Structural Individual Low-FODMAP Dietary Advice vs. Brief Advice on a Commonly Recommended Diet on IBS Symptoms and Intestinal Gas Production
Abstract
:1. Introduction
2. Methods
2.1. Subjects
2.2. Study Protocol
2.2.1. Brief Advice on a Commonly Recommended Diet (BRD) Protocol
2.2.2. The Structural Individual Low-FODMAP Dietary Advice (SILFD) Protocol
2.3. Measurement and Analysis
2.4. Statistical Analysis
3. Results
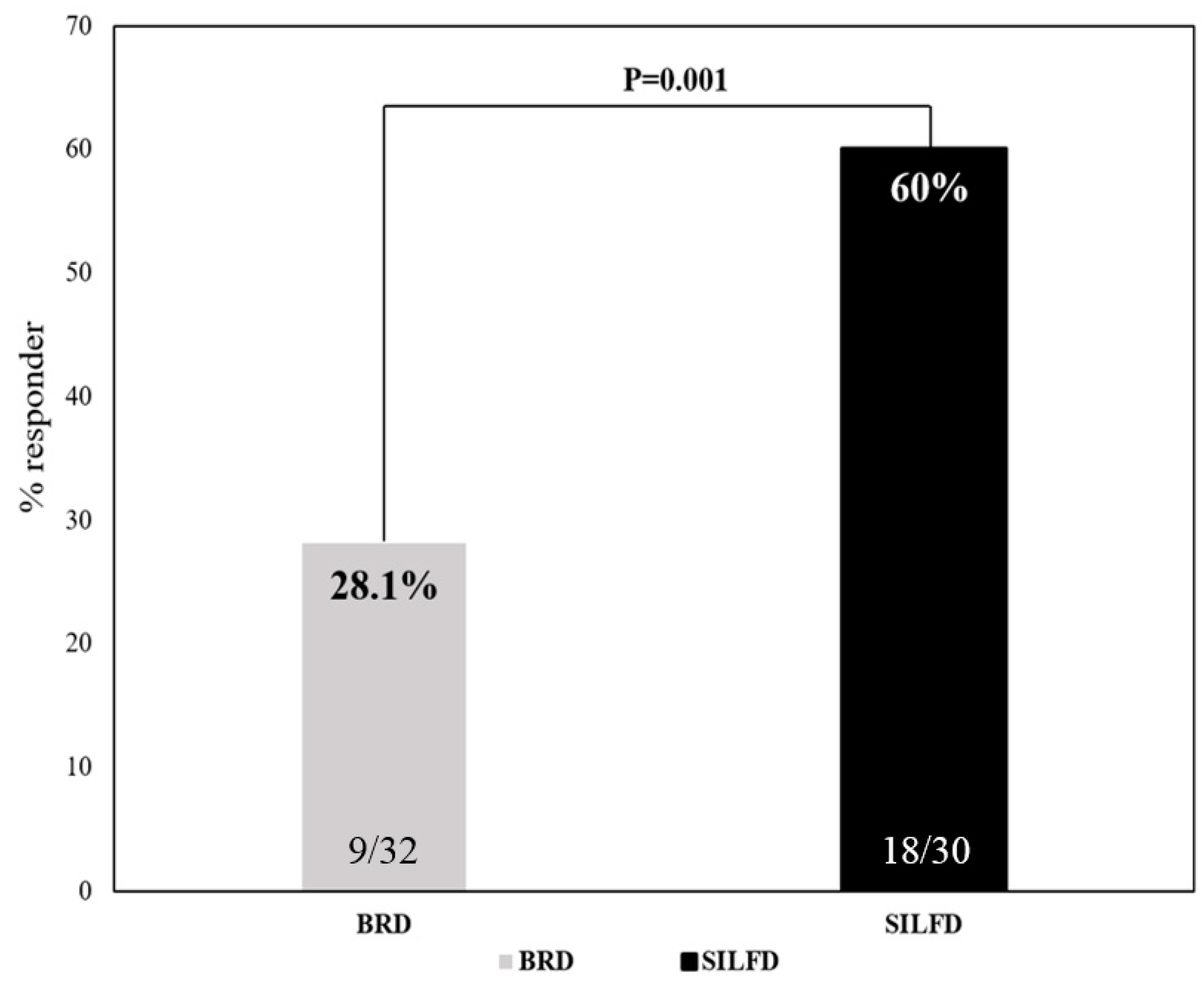
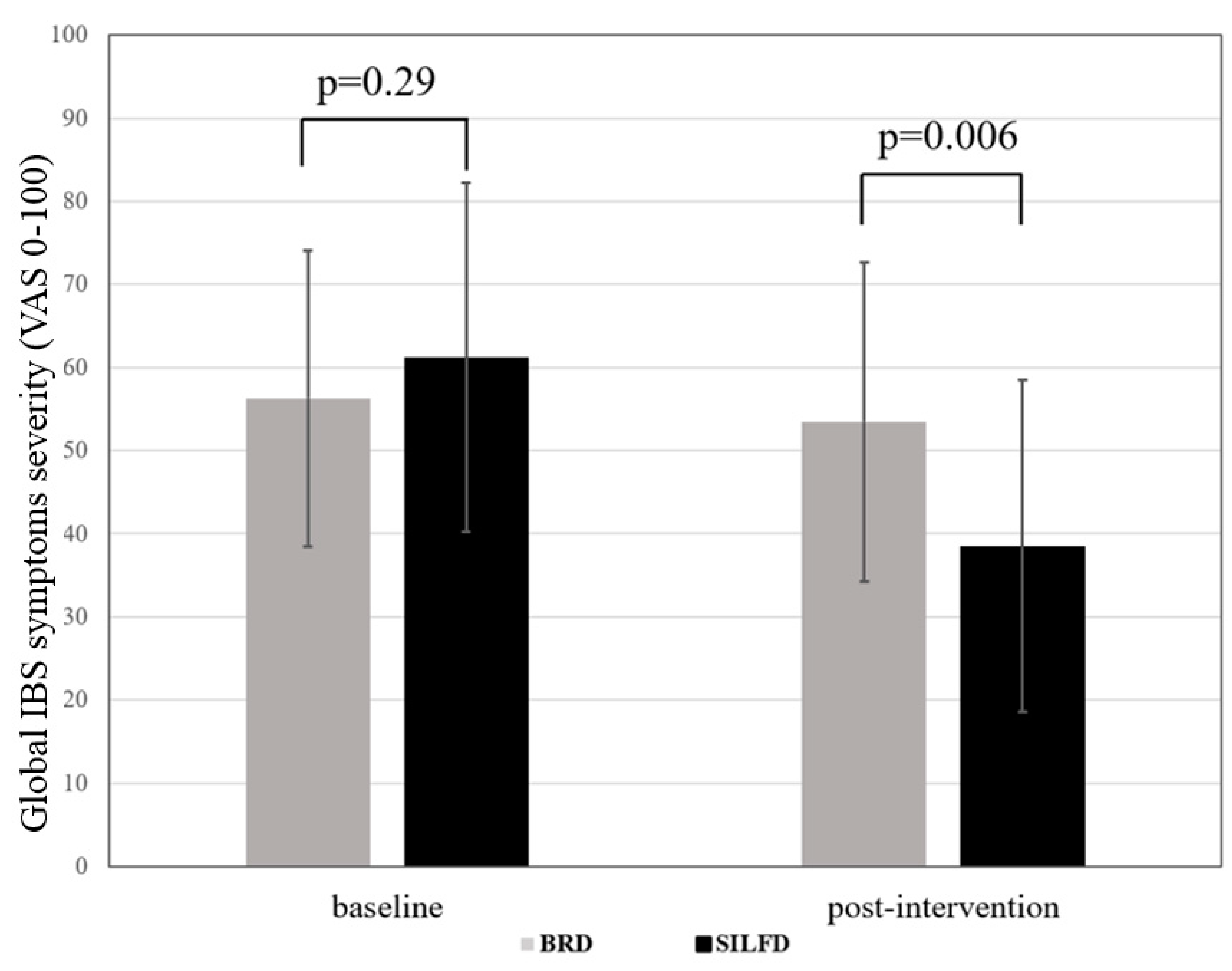
3.1. Effect of Dietary Advice on Gastrointestinal Symptoms
3.2. Effect of Dietary Advice on Intestinal Gas Production
3.3. Effect of Dietary Advice on Gastrointestinal Symptoms in Each IBS Subtype
3.4. Dietary Compliance
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Lovell, R.M.; Ford, A.C. Global prevalence of and risk factors for irritable bowel syndrome: A meta-analysis. Clin. Gastroenterol. Hepatol. 2012, 10, 712–721. [Google Scholar] [CrossRef] [PubMed]
- Mearin, F.; Lacy, B.E.; Chang, L.; Chey, W.D.; Lembo, A.J.; Simren, M.; Spiller, R. Bowel Disorders. Gastroenterology 2016, 10. [Google Scholar] [CrossRef]
- Simren, M.; Mansson, A.; Langkilde, A.M.; Svedlund, J.; Abrahamsson, H.; Bengtsson, U.; Björnsson, E.S. Food-related gastrointestinal symptoms in the irritable bowel syndrome. Digestion 2001, 63, 108–115. [Google Scholar] [CrossRef]
- Fritscher-Ravens, A.; Schuppan, D.; Ellrichmann, M.; Schoch, S.; Röcken, C.; Brasch, J.; Bethge, J.; Böttner, M.; Klose, J.; Milla, P.J. Confocal endomicroscopy shows food-associated changes in the intestinal mucosa of patients with irritable bowel syndrome. Gastroenterology 2014, 147, 1012–1020. [Google Scholar] [CrossRef] [PubMed]
- Barrett, J.S.; Gearry, R.B.; Muir, J.G.; Irving, P.M.; Rose, R.; Rosella, O.; Haines, M.L.; Shepherd, S.J.; Gibson, P.R. Dietary poorly absorbed, short-chain carbohydrates increase delivery of water and fermentable substrates to the proximal colon. Aliment. Pharmacol. Ther. 2010, 31, 874–882. [Google Scholar] [CrossRef] [PubMed]
- Shepherd, S.J.; Lomer, M.C.; Gibson, P.R. Short-chain carbohydrates and functional gastrointestinal disorders. Am. J. Gastroenterol. 2013, 108, 707–717. [Google Scholar] [CrossRef]
- Varju, P.; Farkas, N.; Hegyi, P.; Garami, A.; Szabó, I.; Illés, A.; Solymár, M.; Vincze, A.; Balaskó, M.; Pár, M. Low fermentable oligosaccharides, disaccharides, monosaccharides and polyols (FODMAP) diet improves symptoms in adults suffering from irritable bowel syndrome (IBS) compared to standard IBS diet: A meta-analysis of clinical studies. PLoS ONE 2017, 12, e0182942. [Google Scholar] [CrossRef]
- Hookway, C.; Buckner, S.; Crosland, P.; Longson, D. Irritable bowel syndrome in adults in primary care: Summary of updated NICE guidance. BMJ 2015, 35, h701. [Google Scholar] [CrossRef]
- McKenzie, Y.A.; Bowyer, R.K.; Leach, H.; Gulia, P.; Horobin, J.; O’Sullivan, N.A.; Pettitt, C.; Reeves, L.B.; Seamark, L.; Williams, M.; et al. British Dietetic Association systematic review and evidence-based practice guidelines for the dietary management of irritable bowel syndrome in adults (2016 update). J. Hum. Nutr. Diet. 2016, 29, 549–575. [Google Scholar] [CrossRef]
- Gwee, K.A.; Gonlachanvit, S.; Ghoshal, U.C.; Chua, A.S.B.; Miwa, H.; Wu, J.; Bak, Y.T.; Lee, O.Y.; Lu, C.L.; Park, H. Second Asian Consensus on Irritable Bowel Syndrome. J. Neurogastrol. Motil. 2019, 25, 343–362. [Google Scholar] [CrossRef]
- Zigmond, A.S.; Snaith, R.P. The hospital anxiety and depression scale. Acta Psychiatr. Scand. 1983, 67, 361–370. [Google Scholar] [CrossRef] [PubMed]
- Staudacher, H.M.; Whelan, K.; Irving, P.M.; Lomer, M.C. Comparison of symptom response following advice for a diet low in fermentable carbohydrates (FODMAPs) versus standard dietary advice in patients with irritable bowel syndrome. J. Hum. Nutr. Diet. 2011, 24, 487–495. [Google Scholar] [CrossRef] [PubMed]
- Böhn, L.; Störsrud, S.; Liljebo, T.; Collin, L.; Lindfors, P.; Törnblom, H.; Simrén, M. Diet low in FODMAPs reduces symptoms of irritable bowel syndrome as well as traditional dietary advice: A randomized controlled trial. Gastroenterology 2015, 149, 1399–1407. [Google Scholar] [CrossRef] [PubMed]
- Eswaran, S.L.; Chey, W.D.; Han-Markey, T.; Ball, S.; Jackson, K. A Randomized Controlled Trial Comparing the Low FODMAP Diet vs. Modified NICE Guidelines in US Adults with IBS-D. Am. J. Gastroenterol. 2016, 111, 1824–1832. [Google Scholar] [CrossRef] [PubMed]
- McIntosh, K.; Reed, D.E.; Schneider, T.; Dang, F.; Keshteli, A.H.; De Palma, G.; Madsen, K.; Bercik, P.; Vanner, S. FODMAPs alter symptoms and the metabolome of patients with IBS: A randomised controlled trial. Gut 2017, 66, 1241–1251. [Google Scholar] [CrossRef] [PubMed]
- Spiegel, B.; Bolus, R.; Harris, L.A.; Lucak, S.; Naliboff, B.; Esrailian, E.; Chey, W.D.; Lembo, A.; Karsan, H.; Tillisch, K.; et al. Measuring irritable bowel syndrome patient-reported outcomes with an abdominal pain numeric rating scale. Aliment. Pharmacol. Ther. 2009, 30, 1159–1170. [Google Scholar] [CrossRef]
- Gonlachanvit, S. Are rice and spicy diet good for functional gastrointestinal disorders? J. Neurogastrol. Motil. 2010, 16, 131–138. [Google Scholar] [CrossRef]
- Guo, Y.B.; Zhuang, K.M.; Kuang, L.; Zhan, Q.; Wang, X.F.; Liu, S.D. Association between Diet and Lifestyle Habits and Irritable Bowel Syndrome: A Case-Control Study. Gut Liver 2015, 9, 649–656. [Google Scholar] [CrossRef]
- Singh, N.; Makharia, G.K.; Joshi, Y.K. Dietary survey and total dietary fiber intake in patients with irritable bowel syndrome attending a tertiary referral hospital. Indian J. Gastroenterol. 2008, 27, 66–70. [Google Scholar]
- Linlawan, S.; Patcharatrakul, T.; Somlaw, N.; Gonlachanvit, S. Effect of Rice, Wheat, and Mung Bean Ingestion on Intestinal Gas Production and Postprandial Gastrointestinal Symptoms in Non-constipation Irritable Bowel Syndrome Patients. Nutrients 2019, 11, 2061. [Google Scholar] [CrossRef]
- Halmos, E.P.; Christophersen, C.T.; Bird, A.R.; Shepherd, S.J.; Gibson, P.R.; Muir, J.G. Diets that differ in their FODMAP content alter the colonic luminal microenvironment. Gut 2015, 64, 93–100. [Google Scholar] [CrossRef] [PubMed]


| SILFD (n = 30) | BRD (n = 32) | p-Value | |
|---|---|---|---|
| Females, n (%) | 23 (76.7%) | 24 (75.0%) | 0.88 |
| Age (years) | 50.0 ± 13.7 | 52.0 ± 14.0 | 0.58 |
| BMI (kg/m2) | 22.5 ± 3.2 | 23.0 ± 4.3 | 0.62 |
| College education, n (%) | 14 (46.7%) | 18 (56.3%) | 0.45 |
| IBS-C, n (%) | 15 (50.0%) | 18 (56.3%) | 0.62 |
| Global IBS symptom score (0–100) | 61.2 ± 21.0 | 56.3 ± 17.8 | 0.33 |
| Symptoms severity score (0–10) - Abdominal pain - Abdominal discomfort - Bloating - Belching - Stool urgency | 4.8 (0–6.9) 5.5 (4.5–7.1) 5.1 (2.5–7.5) 1.4 (0–5.5) 0 (0–8.1) | 4.4 (0.5–6.4) 5.6 (4.1–7.1) 6.2 (2.1–7.8) 2.7 (0–5.6) 2.3 (0–6.6) | 0.95 0.93 0.98 0.96 0.99 |
| HAD score (0–8), anxiety | 7.4 ± 2.7 | 7.1 ± 3.8 | 0.75 |
| HAD score (0–8), depression | 3.7 ± 2.3 | 5.0 ± 3.5 | 0.11 |
| Total high-FODMAPs items per week | 16.0 ± 5.9 | 16.2 ± 5.7 | 0.90 |
| SILFD (n = 30) | BRD (n = 32) | Post-SILFD vs. Post-BRD | |||||
|---|---|---|---|---|---|---|---|
| Baseline | Post SILFD | p-Value | Baseline | Post-BRD | p-Value | p-Value | |
| Global IBS symptoms score (0–100) | 61.2 ± 21.0 | 38.5 ± 20.0 | <0.001 | 56.3 ± 17.8 | 53.5 ± 19.2 | 0.30 | 0.006 |
| Symptoms severity score (0–100) - Abdominal pain - Abdominal discomfort - Bloating - Belching - Stool urgency | 4.8 (0–6.9) 5.5 (4.5–7.1) 5.1 (2.5–7.5) 1.4 (0–5.5) 0 (0–8.1) | 1.7 (0–4.1) 3.2 (1.7–5.5) 3.1 (1.8–5.7) 0.7 (0–4.7) 0 (0–5.4) | 0.001 <0.001 0.02 0.21 0.13 | 4.4 (0.5–6.4) 5.6 (4.1–7.1) 6.2 (2.1–7.8) 2.7 (0–5.6) 2.3 (0–6.6) | 3.9 (0–5.2) 4.5 (2.6–6.6) 4.0 (0–6.2) 1.0 (0–5.4) 0 (0–4.2) | 0.11 0.09 0.25 0.78 0.26 | 0.16 0.09 0.61 0.79 0.66 |
| Stool frequency, times/week - IBS—constipation patients - IBS—non-constipation patients | 5.0 (2.0–7.0) 10.0 (4.0–14.0) | 7.0 (5.0–8.0) 7.0 (4.0–10.0) | 0.02 0.07 | 4.0 (2.0–5.3) 7.0 (5.3–14) | 6.5 (5.0–7.0) 7.0 (3.8–10.8) | 0.001 0.29 | 0.45 0.93 |
| Total high-FODMAPs items per week | 16.0 ± 5.9 | 9.6 ± 4.0 | <0.001 | 16.2 ± 5.7 | 15.4 ± 5.9 | 0.30 | <0.001 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Patcharatrakul, T.; Juntrapirat, A.; Lakananurak, N.; Gonlachanvit, S. Effect of Structural Individual Low-FODMAP Dietary Advice vs. Brief Advice on a Commonly Recommended Diet on IBS Symptoms and Intestinal Gas Production. Nutrients 2019, 11, 2856. https://doi.org/10.3390/nu11122856
Patcharatrakul T, Juntrapirat A, Lakananurak N, Gonlachanvit S. Effect of Structural Individual Low-FODMAP Dietary Advice vs. Brief Advice on a Commonly Recommended Diet on IBS Symptoms and Intestinal Gas Production. Nutrients. 2019; 11(12):2856. https://doi.org/10.3390/nu11122856
Chicago/Turabian StylePatcharatrakul, Tanisa, Akarawut Juntrapirat, Narisorn Lakananurak, and Sutep Gonlachanvit. 2019. "Effect of Structural Individual Low-FODMAP Dietary Advice vs. Brief Advice on a Commonly Recommended Diet on IBS Symptoms and Intestinal Gas Production" Nutrients 11, no. 12: 2856. https://doi.org/10.3390/nu11122856
APA StylePatcharatrakul, T., Juntrapirat, A., Lakananurak, N., & Gonlachanvit, S. (2019). Effect of Structural Individual Low-FODMAP Dietary Advice vs. Brief Advice on a Commonly Recommended Diet on IBS Symptoms and Intestinal Gas Production. Nutrients, 11(12), 2856. https://doi.org/10.3390/nu11122856





