Poor Dietary Polyphenol Intake in Childhood Cancer Patients
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Recruitment
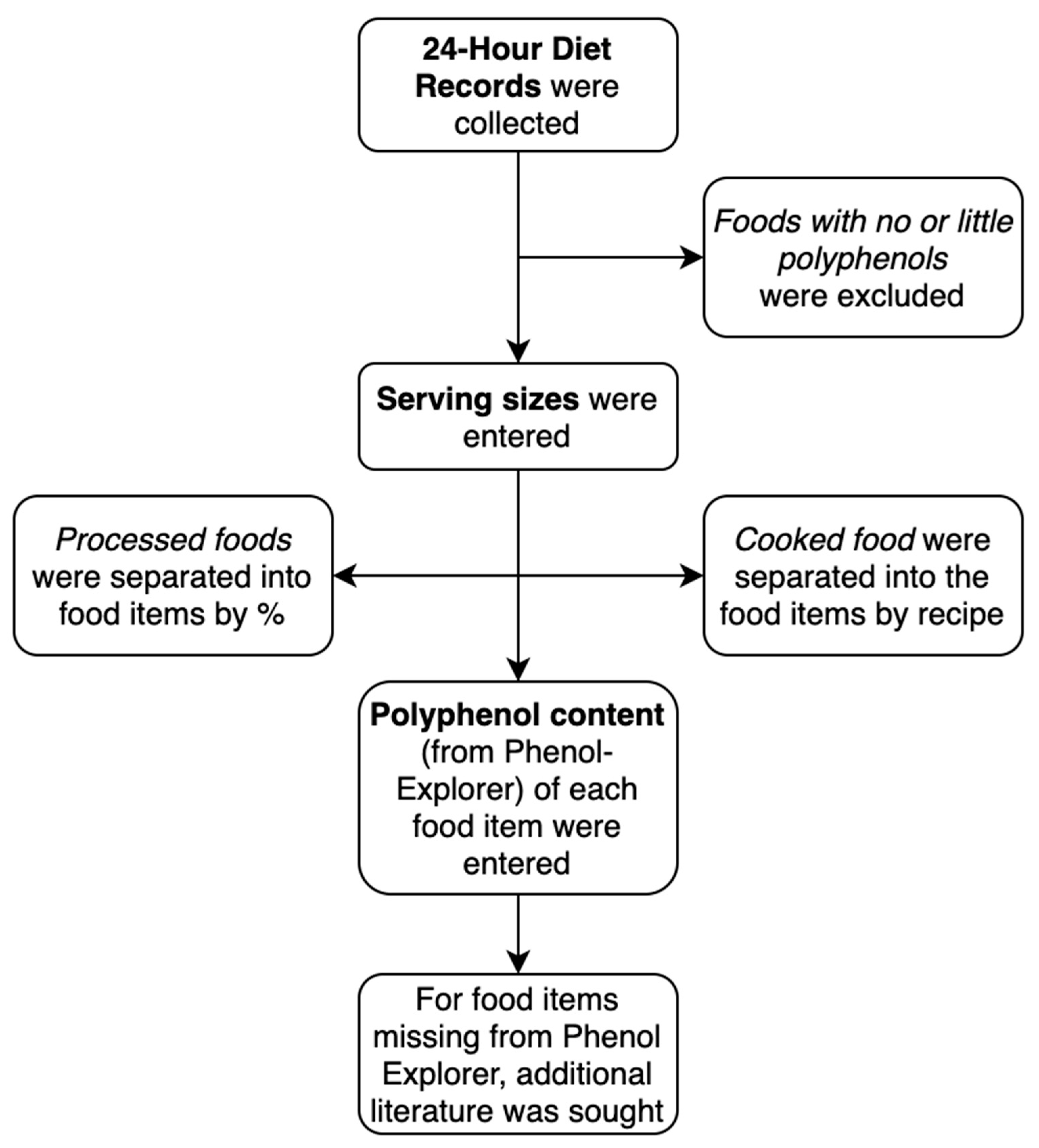
2.3. Data Collection
2.4. Data Analyses
3. Results
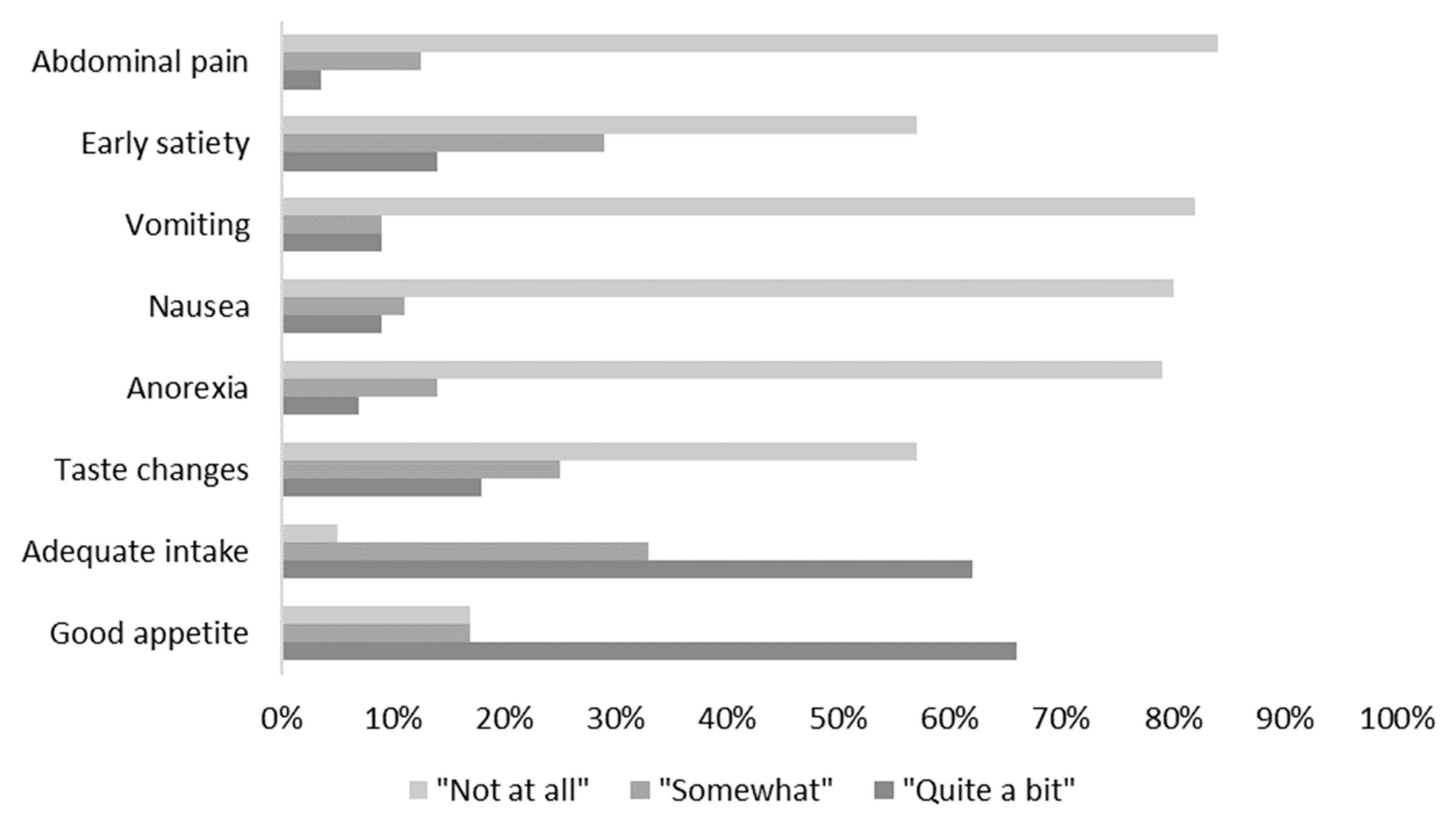
3.1. Patient Demographics
3.2. Polyphenol Intake
3.3. Socio-Demographic and Lifestyle Determinants
4. Discussion
Strengths and Limitations
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Brinksma, A.; Roodbol, P.F.; Sulkers, E.; Kamps, W.A.; De Bont, E.S.; Boot, A.M.; Burgerhof, J.G.; Tamminga, R.Y.; Tissing, W.J. Changes in nutritional status in childhood cancer patients: A prospective cohort study. Clin. Nutr. 2015, 34, 66–73. [Google Scholar] [CrossRef] [PubMed]
- Schadler, K.L.; Kleinerman, E.S.; Chandra, J. Diet and exercise interventions for pediatric cancer patients during therapy: Tipping the scales for better outcomes. Pediatric Res. 2018, 83, 50. [Google Scholar] [CrossRef] [PubMed]
- Wallace, T.C.; Bultman, S.; D’Adamo, C.; Daniel, C.R.; Debelius, J.; Ho, E.; Eliassen, H.; Lemanne, D.; Mukherjee, P.; Seyfried, T.N.; et al. Personalized Nutrition in Disrupting Cancer—Proceedings From the 2017 American College of Nutrition Annual Meeting. J. Am. Coll. Nutr. 2019, 38, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Bauer, J.; Jürgens, H.; Frühwald, M.C. Important Aspects of Nutrition in Children with Cancer1. Adv. Nutr. 2011, 2, 67–77. [Google Scholar] [CrossRef] [PubMed]
- Ladas, E.J. Nutrition therapy: Support for integration into cancer care. Pediatr. Blood Cancer 2013, 60, 895–896. [Google Scholar] [CrossRef] [PubMed]
- Ladas, E.J.; Arora, B.; Howard, S.C.; Rogers, P.C.; Mosby, T.T.; Barr, R.D. A Framework for Adapted Nutritional Therapy for Children with Cancer in Low- and Middle-Income Countries: A Report From the SIOP PODC Nutrition Working Group. Pediatr. Blood Cancer 2016, 63, 1339–1348. [Google Scholar] [CrossRef] [PubMed]
- Boffetta, P.; Couto, E.; Wichmann, J.; Ferrari, P.; Trichopoulos, D.; Bueno-De-Mesquita, H.B.; Van Duijnhoven, F.J.B.; Büchner, F.L.; Key, T.; Boeing, H.; et al. Fruit and Vegetable Intake and Overall Cancer Risk in the European Prospective Investigation into Cancer and Nutrition (EPIC). J. Natl. Cancer Inst. 2010, 102, 529–537. [Google Scholar] [CrossRef]
- Davis, C.; Bryan, J.; Hodgson, J.; Murphy, K. Definition of the Mediterranean Diet: A Literature Review. Nutrients 2015, 7, 9139–9153. [Google Scholar] [CrossRef]
- Schwingshackl, L.; Schwedhelm, C.; Galbete, C.; Hoffmann, G. Adherence to Mediterranean Diet and Risk of Cancer: An Updated Systematic Review and Meta-Analysis. Nutrients 2017, 9, 1063. [Google Scholar] [CrossRef]
- Jochems, S.H.; Van Osch, F.H.; Bryan, R.T.; Wesselius, A.; van Schooten, F.J.; Cheng, K.K.; Zeegers, M.P. Impact of dietary patterns and the main food groups on mortality and recurrence in cancer survivors: A systematic review of current epidemiological literature. BMJ Open 2018, 8, e014530. [Google Scholar] [CrossRef]
- Maruca, A.; Catalano, R.; Bagetta, D.; Mesiti, F.; Ambrosio, F.A.; Romeo, I.; Moraca, F.; Rocca, R.; Ortuso, F.; Artese, A.; et al. The Mediterranean Diet as source of bioactive compounds with multi-targeting anti-cancer profile. Eur. J. Med. Chem. 2019, 181, 111579. [Google Scholar] [CrossRef] [PubMed]
- Erdrich, S.; Bishop, K.S.; Karunasinghe, N.; Han, D.Y.; Ferguson, L.R. A pilot study to investigate if New Zealand men with prostate cancer benefit from a Mediterranean-style diet. PeerJ 2015, 3, e1080. [Google Scholar] [CrossRef] [PubMed]
- Richman, E.L.; Carroll, P.R.; Chan, J.M. Vegetable and fruit intake after diagnosis and risk of prostate cancer progression. Int. J. Cancer 2012, 131, 201–210. [Google Scholar] [CrossRef] [PubMed]
- Abbaszadeh, H.; Keikhaei, B.; Mottaghi, S. A review of molecular mechanisms involved in anticancer and antiangiogenic effects of natural polyphenolic compounds. Phytotherapy Res. 2019, 33, 2002–2014. [Google Scholar] [CrossRef]
- Mileo, A.M.; Miccadei, S. Polyphenols as Modulator of Oxidative Stress in Cancer Disease: New Therapeutic Strategies. Oxidative Med. Cell. Longev. 2016, 229, 54–72. [Google Scholar] [CrossRef]
- Oyenihi, A.; Smith, C. Are polyphenol antioxidants at the root of medicinal plant anti-cancer success? J. Ethnopharmacol. 2019, 229, 54–72. [Google Scholar] [CrossRef]
- Khan, H.Y.; Zubair, H.; Faisal, M.; Ullah, M.F.; Farhan, M.; Sarkar, F.H.; Hadi, S.M. Plant polyphenol induced cell death in human cancer cells involves mobilization of intracellular copper ions and reactive oxygen species generation: A mechanism for cancer chemopreventive action. Mol. Nutr. Food Res. 2014, 58, 437–446. [Google Scholar] [CrossRef]
- Khan, H.Y.; Zubair, H.; Ullah, M.F.; Ahmad, A.; Hadi, S.M. A prooxidant mechanism for the anticancer and chemopreventive properties of plant polyphenols. Curr. Drug Targets 2012, 13, 1738–1749. [Google Scholar] [CrossRef]
- Fernando, W.; Rupasinghe, H.V.; Hoskin, D.W. Dietary phytochemicals with anti-oxidant and pro-oxidant activities: A double-edged sword in relation to adjuvant chemotherapy and radiotherapy? Cancer Lett. 2019, 452, 168–177. [Google Scholar] [CrossRef]
- Parmar, A.; Pascali, G.; Voli, F.; Lerra, L.; Yee, E.; Ahmed-Cox, A.; Kimpton, K.; Cirillo, G.; Arthur, A.; Zahra, D.; et al. In vivo [64Cu]CuCl2 PET imaging reveals activity of Dextran-Catechin on tumor copper homeostasis. Theranostics 2018, 8, 5645–5659. [Google Scholar] [CrossRef]
- Li, A.-N.; Li, S.; Zhang, Y.-J.; Xu, X.-R.; Chen, Y.-M.; Li, H.-B. Resources and Biological Activities of Natural Polyphenols. Nutrients 2014, 6, 6020–6047. [Google Scholar] [CrossRef] [PubMed]
- Ostan, R.; Lanzarini, C.; Pini, E.; Scurti, M.; Vianello, D.; Bertarelli, C.; Fabbri, C.; Izzi, M.; Palmas, G.; Biondi, F.; et al. Inflammaging and Cancer: A Challenge for the Mediterranean Diet. Nutrients 2015, 7, 2589–2621. [Google Scholar] [CrossRef] [PubMed]
- Niedzwiecki, A.; Roomi, M.W.; Kalinovsky, T.; Rath, M. Anticancer Efficacy of Polyphenols and Their Combinations. Nutrients 2016, 8, 552. [Google Scholar] [CrossRef] [PubMed]
- Sak, K. Chemotherapy and Dietary Phytochemical Agents. Chemother. Res. Pr. 2012, 2012, 1–11. [Google Scholar] [CrossRef]
- Lall, R.K.; Syed, D.N.; Adhami, V.M.; Khan, M.I.; Mukhtar, H. Dietary Polyphenols in Prevention and Treatment of Prostate Cancer. Int. J. Mol. Sci. 2015, 16, 3350–3376. [Google Scholar] [CrossRef]
- Alam, M.N.; Almoyad, M.; Huq, F. Polyphenols in Colorectal Cancer: Current State of Knowledge including Clinical Trials and Molecular Mechanism of Action. BioMed Res. Int. 2018, 2018, 1–29. [Google Scholar] [CrossRef]
- Russo, G.L.; Tedesco, I.; Spagnuolo, C.; Russo, M. Antioxidant polyphenols in cancer treatment: Friend, foe or foil? Semin. Cancer Boil. 2017, 46, 1–13. [Google Scholar] [CrossRef]
- Kotecha, R.; Takami, A.; Espinoza, J.L. Dietary phytochemicals and cancer chemoprevention: A review of the clinical evidence. Oncotarget 2016, 7, 52517–52529. [Google Scholar] [CrossRef]
- Liu, R.H. Potential synergy of phytochemicals in cancer prevention: Mechanism of action. J. Nutr. 2004, 134, 3479S–3485S. [Google Scholar] [CrossRef]
- Australian Bureau of Statistics. Socio-Economic Indexes for Areas. 2018. Available online: https://www.abs.gov.au/websitedbs/censushome.nsf/home/seifa (accessed on 1 September 2019).
- World Health Organization. BMI-for-age (5-19 years). 2019. Available online: https://www.who.int/growthref/who2007_bmi_for_age/en/ (accessed on 1 September 2019).
- Daniels, S.R. The Use of BMI in the Clinical Setting. Pediatrics 2009, 124, S35–S41. [Google Scholar] [CrossRef]
- Kazak, A.E.; Hocking, M.C.; Ittenbach, R.F.; Meadows, A.T.; Hobbie, W.; DeRosa, B.W.; Reilly, A. A revision of the intensity of treatment rating scale: Classifying the intensity of pediatric cancer treatment. Pediatric Blood Cancer 2012, 59, 96–99. [Google Scholar] [CrossRef] [PubMed]
- Ribaudo, J.; Cella, D.; Hahn, E.; Lloyd, S.; Tchekmedyian, N.; Von Roenn, J.; Leslie, W. Re-validation and Shortening of the Functional Assessmentof Anorexia/Cachexia Therapy (FAACT) Questionnaire. Qual. Life Res. 2000, 9, 1137–1146. [Google Scholar] [CrossRef] [PubMed]
- McPherson, R.; Hoelscher, D.M.; Alexander, M.; Scanlon, K.S.; Serdula, M.K. Dietary Assessment Methods among School-Aged Children: Validity and Reliability. Prev. Med. 2000, 31, S11–S33. [Google Scholar] [CrossRef]
- Del Bo, C.; Bernardi, S.; Marino, M.; Porrini, M.; Tucci, M.; Guglielmetti, S.; Cherubini, A.; Carrieri, B.; Kirkup, B.; Kroon, P.; et al. Systematic Review on Polyphenol Intake and Health Outcomes: Is there Sufficient Evidence to Define a Health-Promoting Polyphenol-Rich Dietary Pattern? Nutrients 2019, 11, 1355. [Google Scholar]
- Crispim, S.P.; Nicolas, G.; Casagrande, C.; Knaze, V.; Illner, A.K.; Huybrechts, I.; Slimani, N. Quality assurance of the international computerised 24 h dietary recall method (EPIC-Soft). Br. J. Nutr. 2014, 111, 506–515. [Google Scholar] [CrossRef] [PubMed]
- Thompson, F.E.; Subar, A.F. Dietary assessment methodology. In Nutrition in the Prevention and Treatment of Disease; Elsevier: Amsterdam, The Netherlands, 2017; pp. 5–48. [Google Scholar]
- Bhagwat, S.; Haytowitz, D.B.; Prior, R.L.; Gu, L.; Hammerstone, J.; Gebhardt, S.E.; Holden, J.M. USDA Database for Proanthocyanidin Content of Selected Foods; US Department of Agriculture: Washington, DC, USA, 2004.
- Bhagwat, S.; Haytowitz, D.B.; Holden, J.M. USDA Database for the Flavonoid Content of Selected Foods, Release 3.1; US Department of Agriculture: Beltsville, MD, USA, 2014.
- Bhagwat, S.; Haytowitz, D.B.; Holden, J.M. USDA Database for the Isoflavone Content of Selected Foods, Release 2.0; US Department of Agriculture: Beltsville, MD, USA, 2008.
- Rothwell, J.A.; Perez-Jimenez, J.; Neveu, V.; Medina-Remón, A.; M’Hiri, N.; García-Lobato, P.; Manach, C.; Knox, C.; Eisner, R.; Wishart, D.S.; et al. Phenol-Explorer 3.0: A major update of the Phenol-Explorer database to incorporate data on the effects of food processing on polyphenol content. Database 2013, 2013, bat070. [Google Scholar] [CrossRef] [PubMed]
- Zamora-Ros, R.; Knaze, V.; Rothwell, J.A.; Hémon, B.; Moskal, A.; Overvad, K.; Touillaud, M. Dietary polyphenol intake in Europe: The European Prospective Investigation into Cancer and Nutrition (EPIC) study. Eur. J. Nutr. 2016, 55, 1359–1375. [Google Scholar] [CrossRef]
- Ziauddeen, N.; Rosi, A.; Del Rio, D.; Amoutzopoulos, B.; Nicholson, S.; Page, P.; Scazzina, F.; Brighenti, F.; Ray, S.; Mena, P. Dietary intake of (poly)phenols in children and adults: Cross-sectional analysis of UK National Diet and Nutrition Survey Rolling Programme (2008–2014). Eur. J. Nutr. 2018, 58, 3183–3198. [Google Scholar] [CrossRef]
- Pérez-Jiménez, J.; Fezeu, L.; Touvier, M.; Arnault, N.; Manach, C.; Hercberg, S.; Galan, P.; Scalbert, A. Dietary intake of 337 polyphenols in French adults. Am. J. Clin. Nutr. 2011, 93, 1220–1228. [Google Scholar] [CrossRef]
- Tresserra-Rimbau, A.; Medina-Remón, A.; Pérez-Jiménez, J.; Martinez-Gonzalez, M.A.; Covas, M.; Corella, D.; Salas-Salvadó, J.; Gomez-Gracia, E.; Lapetra, J.; Arós, F.; et al. Dietary intake and major food sources of polyphenols in a Spanish population at high cardiovascular risk: The PREDIMED study. Nutr. Metab. Cardiovasc. Dis. 2013, 23, 953–959. [Google Scholar] [CrossRef]
- Nutrition Australia. Australian Dietary Guidelines: Recommended daily intakes. 2013. Available online: http://www.nutritionaustralia.org/national/resource/australian-dietary-guidelines-recommended-daily-intakes (accessed on 1 September 2019).
- National Health and Medical Research Council. Australian Dietary Guidelines; National Health and Medical Research Council: Canberra, Australia, 2013.
- Rossi, M.; Bassett, M.; Sammán, N. Dietary nutritional profile and phenolic compounds consumption in school children of highlands of Argentine Northwest. Food Chem. 2018, 238, 111–116. [Google Scholar] [CrossRef] [PubMed]
- Kopeć, A.; Cieślik, E.; Leszczyńska, T.; Filipiak-Florkiewicz, A.; Wielgos, B.; Piątkowska, E.; Bodzich, A.; Grzych-Tuleja, E. Assessment of Polyphenols, Beta-Carotene, and Vitamin C Intake with Daily Diets by Primary School Children. Ecol. Food Nutr. 2013, 52, 21–33. [Google Scholar] [CrossRef] [PubMed]
- Wisnuwardani, R.W.; De Henauw, S.; Androutsos, O.; Forsner, M.; Gottrand, F.; Huybrechts, I.; Molnár, D. Estimated dietary intake of polyphenols in European adolescents: The HELENA study. Eur. J. Nutr. 2019, 58, 2345–2363. [Google Scholar] [CrossRef] [PubMed]
- Wisnuwardani, R.W.; De Henauw, S.; Forsner, M.; Gottrand, F.; Huybrechts, I.; Knaze, V.; Kersting, M.; Le Donne, C.; Manios, Y.; Marcos, A.; et al. Polyphenol intake and metabolic syndrome risk in European adolescents: The HELENA study. Eur. J. Nutr. Available online: https://link.springer.com/article/10.1007/s00394-019-01946-1 (accessed on 2 September 2019).
- Co-Reyes, E.; Li, R.; Huh, W.; Chandra, J. Malnutrition and obesity in pediatric oncology patients: Causes, consequences, and interventions. Pediatr. Blood Cancer 2012, 59, 1160–1167. [Google Scholar] [CrossRef] [PubMed]
- Owens, J.L.; Hanson, S.J.; McArthur, J.A.; Mikhailov, T.A. The Need for Evidence Based Nutritional Guidelines for Pediatric Acute Lymphoblastic Leukemia Patients: Acute and Long-Term Following Treatment. Nutrients 2013, 5, 4333–4346. [Google Scholar] [CrossRef] [PubMed]
- Slegtenhorst, S.; Visser, J.; Burke, A.; Meyer, R. Antioxidant intake in paediatric oncology patients. Clin. Nutr. 2015, 34, 1210–1214. [Google Scholar] [CrossRef][Green Version]
- Manach, C.; Scalbert, A.; Morand, C.; Rémésy, C.; Jiménez, L. Polyphenols: Food sources and bioavailability. Am. J. Clin. Nutr. 2004, 79, 727–747. [Google Scholar] [CrossRef]
- Koss-Mikołajczyk, I.; Baranowska, M.; Todorovic, V.; Albini, A.; Sansone, C.; Andreoletti, P.; Cherkaoui-Malki, M.; Lizard, G.; Noonan, D.; Sobajic, S.; et al. Prophylaxis of Non-communicable Diseases: Why Fruits and Vegetables may be Better Chemopreventive Agents than Dietary Supplements Based on Isolated Phytochemicals? Curr. Pharm. Des. 2019, 25, 1847–1860. [Google Scholar] [CrossRef]
- Liu, R.H. Health-Promoting Components of Fruits and Vegetables in the Diet12. Adv. Nutr. 2013, 4, 384S–392S. [Google Scholar] [CrossRef]
- Zhou, Y.; Zheng, J.; Li, Y.; Xu, D.-P.; Li, S.; Chen, Y.-M.; Li, H.-B. Natural Polyphenols for Prevention and Treatment of Cancer. Nutrients 2016, 8, 515. [Google Scholar] [CrossRef]
- El Gharras, H. Polyphenols: Food sources, properties and applications—A review. Int. J. Food Sci. Technol. 2009, 44, 2512–2518. [Google Scholar] [CrossRef]
- Urquiza-Salvat, N.; Pascual-Geler, M.; Lopez-Guarnido, O.; Rodrigo, L.; Martinez-Burgos, A.; Cozar, J.M.; Rivas, A. Adherence to Mediterranean diet and risk of prostate cancer. Aging Male 2019, 22, 102–108. [Google Scholar] [CrossRef] [PubMed]
- Castro-Quezada, I.; Román-Viñas, B.; Serra-Majem, L. The Mediterranean Diet and Nutritional Adequacy: A Review. Nutrients 2014, 6, 231–248. [Google Scholar] [CrossRef] [PubMed]
- Goddard, E.; Cohen, J.; Bramley, L.; Wakefield, C.E.; Beck, E.J. Dietary intake and diet quality in children receiving treatment for cancer. Nutr. Rev. 2019, 77, 267–277. [Google Scholar] [CrossRef] [PubMed]
- Cohen, J.; Wakefield, C.E.; Tapsell, L.C.; Walton, K.; Fleming, C.A.; Cohn, R.J. Exploring the views of parents regarding dietary habits of their young cancer-surviving children. Supportive Care Cancer 2015, 23, 463–471. [Google Scholar] [CrossRef]
- Giacosa, A.; Barale, R.; Bavaresco, L.; Gatenby, P.; Gerbi, V.; Janssens, J.; Morazzoni, P. Cancer prevention in Europe: The Mediterranean diet as a protective choice. Eur. J. Cancer Prev. 2013, 22, 90–95. [Google Scholar] [CrossRef]
- Leenders, M.; Sluijs, I.; Ros, M.M.; Boshuizen, H.C.; Siersema, P.D.; Ferrari, P.; Weikert, C.; Tjønneland, A.; Olsen, A.; Boutron-Ruault, M.-C.; et al. Fruit and Vegetable Consumption and Mortality. Am. J. Epidemiol. 2013, 178, 590–602. [Google Scholar] [CrossRef]
- Ortega, R. Importance of functional foods in the Mediterranean diet. Public Heal. Nutr. 2006, 9, 1136–1140. [Google Scholar] [CrossRef]
- Brinksma, A.; Huizinga, G.; Sulkers, E.; Kamps, W.; Roodbol, P.; Tissing, W. Malnutrition in childhood cancer patients: A review on its prevalence and possible causes. Crit. Rev. Oncol. 2012, 83, 249–275. [Google Scholar] [CrossRef]
- Cohen, J.; Wakefield, C.E.; Fleming, C.A.; Gawthorne, R.; Tapsell, L.C.; Cohn, R.J. Dietary intake after treatment in child cancer survivors. Pediatric Blood Cancer 2012, 58, 752–757. [Google Scholar] [CrossRef]
- Cleverdon, R.; Elhalaby, Y.; McAlpine, M.D.; Gittings, W.; Ward, W.E. Total Polyphenol Content and Antioxidant Capacity of Tea Bags: Comparison of Black, Green, Red Rooibos, Chamomile and Peppermint over Different Steep Times. Beverages 2018, 4, 15. [Google Scholar] [CrossRef]
| Characteristics | N (%) | Mean (Range, SD) |
|---|---|---|
| Age at time of survey (nearest year) | 7.8 (2–16, 4.4) | |
| Years since diagnosis | 5.9 | |
| Age at diagnosis | ||
| Gender | ||
| Female | 24 (40.7%) | |
| Male | 35 (59.3%) | |
| Body mass index (BMI) z-score 1 | 17.4 (12.7–23.2) | |
| Obese | 9 (15.8%) | |
| Overweight | 4 (7%) | |
| Healthy | 42 (73.7%) | |
| Underweight | 2 (3.5%) | |
| Socioeconomic status | ||
| Low | 9 (15.2%) | |
| Medium | 8 (13.6%) | |
| High | 42 (71.2%) | |
| Diagnosis | ||
| Acute lymphoblastic leukaemia | 33 (55.9%) | |
| Acute myeloid leukaemia | 1 (1.7%) | |
| Brain cancer (e.g., Medulloblastoma, Glioma) | 4 (6.8%) | |
| Hodgkin’s lymphoma | 4 (6.8%) | |
| Neuroblastoma | 2 (3.4%) | |
| Non-Hodgkin’s lymphoma (including Burkitt’s lymphoma) | 4 (6.8%) | |
| Wilms’ tumour | 4 (6.8%) | |
| Sarcoma of the bone (e.g., Osteosarcoma) | 2 (3.4%) | |
| Soft tissue sarcoma | 1 (1.7%) | |
| Other | 4 (6.8%) | |
| Relapse 2 | ||
| Yes | 7 (12.1%) | |
| No | 51 (87.9%) | |
| Treatment received | ||
| Surgery | 13 (22%) | |
| Chemotherapy | 54 (91.5%) | |
| Radiotherapy | 10 (16.9%) | |
| Bone marrow transplant/stem cell transplant | 5 (8.5%) | |
| Other | 5 (8.5%) | |
| Treatment Intensity | ||
| 1 | 2 (3.4%) | |
| 2 | 28 (47.5%) | |
| 3 | 20 (33.9%) | |
| 4 | 9 (15.3%) |
| Polyphenol Classes | Mean ± SD (Mg/Day) | Median (Mg/Day) | Range (Mg/Day) |
|---|---|---|---|
| Flavonoids | 99.85 ± 116.42 | 51.87 | 0.00–541.72 |
| Phenolic acids | 43.70 ± 36.94 | 31.49 | 1.64–150.45 |
| Lignans | 13.33 ± 21.51 | 4.24 | 0.00–120.69 |
| Stilbenes | 0.25 ± 0.75 | 0.00 | 0.00–3.53 |
| Other | 15.09 ± 20.56 | 8.02 | 0.00–90.31 |
| Total polyphenol intake 1 | 173.31 ± 141.02 | 114.29 | 3.42–665.54 |
| Food Groups | Average Polyphenol Intake (Mg/Day) | Relative Contribution (%) | Top 3 Common Food Sources |
|---|---|---|---|
| Fruit | 44.74 | 26.63 | Apple, banana, strawberry |
| Vegetable | 38.35 | 9.34 | Tomato, potatoes, cucumber |
| Cereal | 22.39 | 16.24 | Whole meal bread, rice, bread (other) |
| Oils | 3.97 | 4.64 | Olive oil (virgin, extra-virgin), vegetable oil |
| Beverages | 50.49 | 17.98 | Orange juice, apple juice, lemon juice |
| Nuts and seeds | 3.96 | 1.56 | Primarily found in muesli and seeded bread |
| Seasoning | 3.04 | 1.38 | Garlic, soy sauce, basil |
| Cocoa products | 5.13 | 2.23 | Cocoa, hot chocolate |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, A.; Cohen, J.; Vittorio, O. Poor Dietary Polyphenol Intake in Childhood Cancer Patients. Nutrients 2019, 11, 2835. https://doi.org/10.3390/nu11112835
Liu A, Cohen J, Vittorio O. Poor Dietary Polyphenol Intake in Childhood Cancer Patients. Nutrients. 2019; 11(11):2835. https://doi.org/10.3390/nu11112835
Chicago/Turabian StyleLiu, Ashly, Jennifer Cohen, and Orazio Vittorio. 2019. "Poor Dietary Polyphenol Intake in Childhood Cancer Patients" Nutrients 11, no. 11: 2835. https://doi.org/10.3390/nu11112835
APA StyleLiu, A., Cohen, J., & Vittorio, O. (2019). Poor Dietary Polyphenol Intake in Childhood Cancer Patients. Nutrients, 11(11), 2835. https://doi.org/10.3390/nu11112835






