Compliance and Adherence to Enteral Nutrition Treatment in Adults: A Systematic Review
Abstract
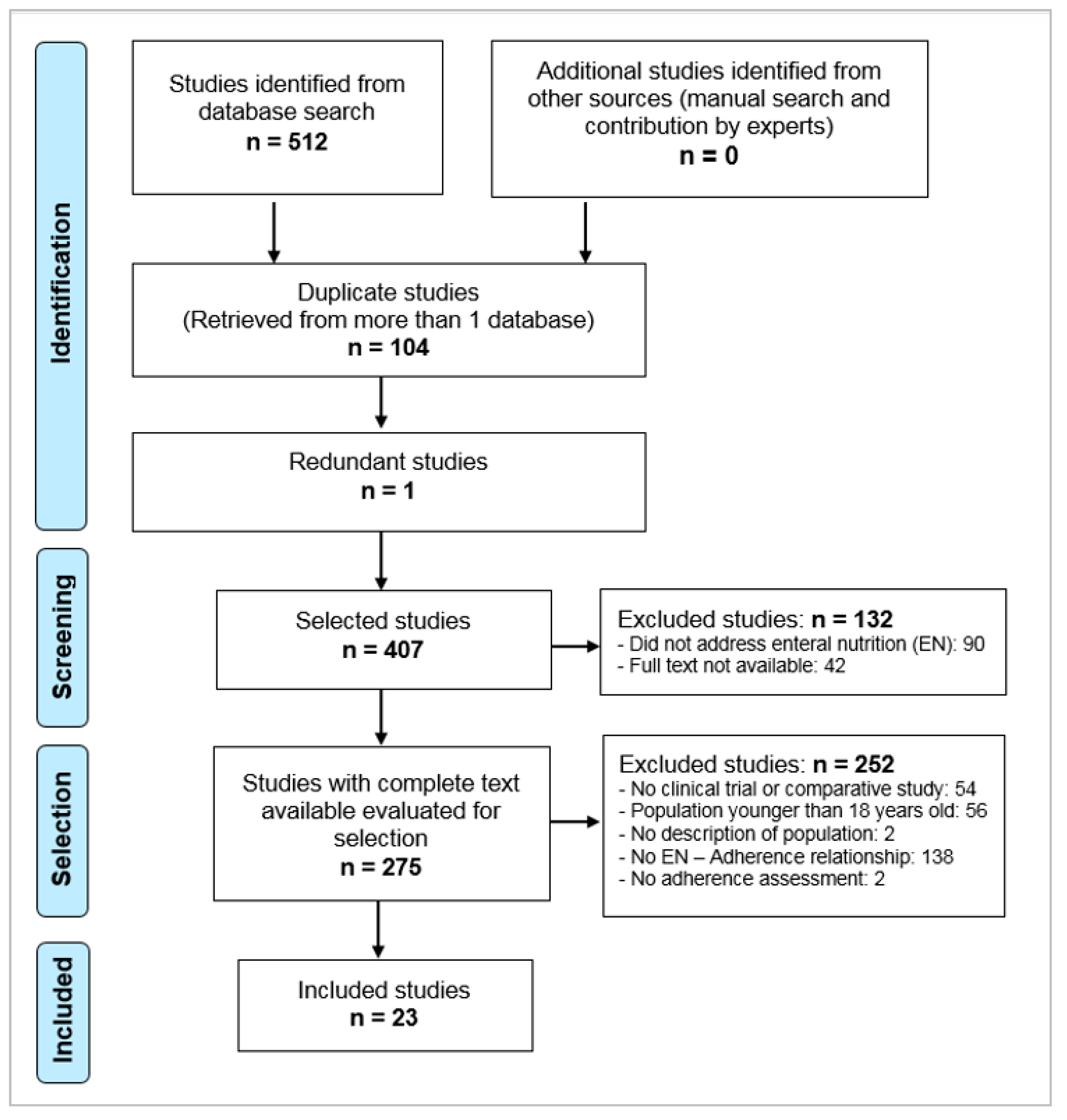
:1. Introduction
2. Materials and Methods
2.1. Design
2.2. Source of Data Collection
2.3. Information Search
- -
- Population: adults with an age equal to or greater than 19 years;
- -
- Intervention: EN;
- -
- Outcome: known compliance and adherence to treatment and the method used for its assessment.
2.4. Final Selection of Articles
2.5. Quality of Reporting of the Selected Documents
2.6. Obsolescence
2.7. Data Extraction
2.8. Study Variables
- The studies were grouped according to the variables studied to systematize and facilitate the interpretation of the results, considering the following data:
- Author: the first author of the article was selected;
- Year: year of publication of the article;
- Design: procedures, methods and techniques through which the article was accepted for review. In this case, only clinical trials or comparative studies were accepted;
- Population studied: Adults undergoing EN intervention;
- Country: location where the intervention took place;
- Pathology: disease of the population for which the intervention was performed;
- Type of nutrition: if total/exclusive enteral nutrition (EEN), when the only source of food is formula-based, or partial (PEN);
- Form and frequency of administration: route through which EN was administered and its periodicity;
- Technique for measuring adherence: procedure used to determine adherence to EN;
- Outcome observed: causal relationship derived from the intervention (administration of EN).
3. Results
3.1. Adherence Measurement Methods
3.2. Periodicity and Time of Measuring Adherence
3.3. Adherence Rates and/or Compliance with EN Protocols
4. Discussion
4.1. Adherence Measurement Methods
4.2. Periodicity and Time of Measuring Adherence
4.3. Adherence Rates and/or Compliance with EN Protocols
4.4. Limitations of the Review
4.5. Critical Review by the Authors
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Wanden-Berghe, C.; Luengo, L.M.; Álvarez, J.; Burgos, R.; Cuerda, C.; Matía, P.; Gómez Candela, C.; Martínez Olmos, M.Á.; Gonzalo, M.; Calleja, A.; et al. Spanish home enteral nutrition registry of the year 2014 and 2015 from the NADYA-SENPE Group. Nutr. Hosp. 2017, 34, 15. [Google Scholar] [CrossRef] [PubMed]
- Wanden-Berghe, C.; Cheikh Moussa, K.; Sanz-Valero, J. Adherence to home enteral nutrition. Hosp. Domic. 2018, 2, 11–18. [Google Scholar] [CrossRef]
- World Health Organization (WHO). The World Health Report 2002—Reducing Risks, Promoting Healthy Life; WHO: Geneva, Switzerland, 2002; Available online: http://bit.ly/2Y9twq2 (accessed on 23 June 2019).
- World Health Organization (WHO). Media Centre. Failure to Take Prescribed Medicine for Chronic Diseases Is a Massive, World-Wide Problema; WHO: Geneva, Switzerland, 2003; Available online: http://bit.ly/2ZEyKdx (accessed on 23 June 2019).
- Sabaté, E. (Ed.) Adherence to Long-Term Therapies: Evidence to Action; World Health Organization: Geneva, Switzerland, 2003; Available online: http://bit.ly/2RwTUrl (accessed on 23 June 2019).
- ASPEN Board of Directors and the Clinical Guidelines Task Force. Guidelines for the use of parenteral and enteral nutrition in adult and pediatric patients. JPEN J. Parenter Enter. Nutr. 2002, 26, S1–S138. [Google Scholar] [CrossRef]
- Howard, P.; Jonkers-Schuitema, C.; Furniss, L.; Kyle, U.; Muehlebach, S.; Ödlund-Olin, A.; Page, M.; Wheatley, C. Managing the Patient Journey through Enteral Nutritional Care. Clin. Nutr. 2006, 25, 187–195. [Google Scholar] [CrossRef]
- van den Broek, P.W.J.H.; Rasmussen-Conrad, E.L.; Naber, A.H.J.; Wanten, G.J.A. What you think is not what they get: Significant discrepancies between prescribed and administered doses of tube feeding. Br. J. Nutr. 2009, 101, 68–71. [Google Scholar] [CrossRef]
- Wanden-Berghe, C.; Sanz-Valero, J. Systematic reviews in nutrition: Standardized methodology. Br. J. Nutr. 2012, 107, S3–S7. [Google Scholar] [CrossRef]
- Pandis, N.; Chung, B.; Scherer, R.W.; Elbourne, D.; Altman, D.G. CONSORT 2010 statement: Extension checklist for reporting within person randomised trials. BMJ 2017, 357, 2835. [Google Scholar] [CrossRef]
- Hirai, F.; Ishida, T.; Takeshima, F.; Yamamoto, S.; Yoshikawa, I.; Ashizuka, S.; Inatsu, H.; Mitsuyama, K.; Sou, S.; Iwakiri, R.; et al. Effect of a concomitant elemental diet with maintenance anti-tumor necrosis factor-α antibody therapy in patients with Crohn’s disease: A multicenter, prospective cohort study. J. Gastroenterol. Hepatol. 2019, 34, 132–139. [Google Scholar] [CrossRef]
- Wall, C.L.; Gearry, R.B.; Day, A.S. Treatment of Active Crohn’s Disease with Exclusive and Partial Enteral Nutrition: A Pilot Study in Adults. Inflamm. Intest. Dis. 2018, 2, 219–227. [Google Scholar] [CrossRef]
- Benton, K.; Thomson, I.; Isenring, E.; Smithers, B.M.; Agarwal, E. An investigation into the nutritional status of patients receiving an Enhanced Recovery After Surgery (ERAS) protocol versus standard care following Oesophagectomy. Support. Care Cancer 2018, 26, 2057–2062. [Google Scholar] [CrossRef] [Green Version]
- Deane, A.M.; Lamontagne, F.; Dukes, G.E.; Neil, D.; Vasist, L.; Barton, M.E.; Hacquoil, K.; Ou, X.; Richards, D.; Stelfox, H.T.; et al. Nutrition adequacy therapeutic enhancement in the critically ill: A randomized double-blind, placebo-controlled trial of the motilin receptor agonist camicinal (GSK962040): The NUTRIATE Study. JPEN J. Parenter Enter. Nutr. 2018, 42, 949–959. [Google Scholar] [CrossRef] [PubMed]
- Brown, T.; Banks, M.; Hughes, B.G.; Lin, C.; Kenny, L.; Bauer, J. Tube feeding during treatment for head and neck cancer—Adherence and patient reported barriers. Oral Oncol. 2017, 72, 140–149. [Google Scholar] [CrossRef] [PubMed]
- Healy, L.A.; Ryan, A.; Doyle, S.L.; Ní Bhuachalla, É.B.; Cushen, S.; Segurado, R.; Murphy, T.; Ravi, N.; Donohoe, C.L.; Reynolds, J.V. Does Prolonged Enteral Feeding With Supplemental Omega-3 Fatty Acids Impact on Recovery Post-esophagectomy: Results of a Randomized Double-Blind Trial. Ann. Surg. 2017, 266, 720–728. [Google Scholar] [CrossRef] [PubMed]
- Harvey, S.E.; Parrott, F.; Harrison, D.A.; Sadique, M.Z.; Grieve, R.D.; Canter, R.R.; McLennan, B.K.; Tan, J.C.; Bear, D.E.; Segaran, E.; et al. A multicentre, randomised controlled trial comparing the clinical effectiveness and cost-effectiveness of early nutritional support via the parenteral versus the enteral route in critically ill patients (CALORIES). Heal. Technol. Assess. 2016, 20, 1–144. [Google Scholar] [CrossRef]
- Zhao, L. Effect of nutritional diet improvement on the digestive system of intensive care unit patients. World Chin. J. Dig. 2015, 23, 5728–5733. [Google Scholar] [CrossRef]
- Stow, R.; Ives, N.; Smith, C.; Rick, C.; Rushton, A. A cluster randomised feasibility trial evaluating nutritional interventions in the treatment of malnutrition in care home adult residents. Trials 2015, 16, 433. [Google Scholar] [CrossRef]
- Hamza, N.; Darwish, A.; O’Reilly, D.A.; Denton, J.; Sheen, A.J.; Chang, D.; Sherlock, D.J.; Ammori, B.J. Perioperative Enteral Immunonutrition Modulates Systemic and Mucosal Immunity and the Inflammatory Response in Patients With Periampullary Cancer Scheduled for Pancreaticoduodenectomy. Pancreas 2015, 44, 41–52. [Google Scholar] [CrossRef]
- Bowrey, D.J.; Baker, M.; Halliday, V.; Thomas, A.L.; Pulikottil-Jacob, R.; Smith, K.; Morris, T.; Ring, A. A randomised controlled trial of six weeks of home enteral nutrition versus standard care after oesophagectomy or total gastrectomy for cancer: Report on a pilot and feasibility study. Trials 2015, 16, 531. [Google Scholar] [CrossRef]
- Sukkar, S.G.; Signori, A.; Borrini, C.; Barisione, G.; Ivaldi, C.; Romeo, C.; Gradaschi, R.; Machello, N.; Nanetti, E.; Vaccaro, A.L. Feasibility of protein-sparing modified fast by tube (ProMoFasT) in obesity treatment: A phase II pilot trial on clinical safety and efficacy (appetite control, body composition, muscular strength, metabolic pattern, pulmonary function test). Mediterr. J. Nutr. Metab. 2013, 6, 165–176. [Google Scholar] [CrossRef]
- Shirakawa, H.; Kinoshita, T.; Gotohda, N.; Takahashi, S.; Nakagohri, T.; Konishi, M. Compliance with and effects of preoperative immunonutrition in patients undergoing pancreaticoduodenectomy. J. Hepatobiliary Pancreat Sci. 2012, 19, 249–258. [Google Scholar] [CrossRef]
- Sadasivan, A.; Faizal, B.; Kumar, M. Nasogastric and Percutaneous Endoscopic Gastrostomy Tube Use in Advanced Head and Neck Cancer Patients: A Comparative Study. J. Pain Palliat. Care Pharmacother. 2012, 26, 226–232. [Google Scholar] [CrossRef] [PubMed]
- Kraft, M.; Berg, N.V.D.; Kraft, K.; Schmekel, S.; Gärtner, S.; Krüger, J.; Meyer, J.; Lerch, M.M.; Hoffmann, W. Development of a telemedical monitoring concept for the care of malnourished geriatric home-dwelling patients: A pilot study. Maturitas 2012, 72, 126–131. [Google Scholar] [CrossRef] [PubMed]
- Miyata, H.; Yano, M.; Yasuda, T.; Hamano, R.; Yamasaki, M.; Hou, E.; Motoori, M.; Shiraishi, O.; Tanaka, K.; Mori, M.; et al. Randomized study of clinical effect of enteral nutrition support during neoadjuvant chemotherapy on chemotherapy-related toxicity in patients with esophageal cancer. Clin. Nutr. 2012, 31, 330–336. [Google Scholar] [CrossRef] [PubMed]
- Pohl, M.; Mayr, P.; Mertl-Roetzer, M.; Lauster, F.; Haslbeck, M.; Hipper, B.; Steube, D.; Tietjen, M.; Eriksen, J.; Rahlfs, V.W. Glycemic control in patients with type 2 diabetes mellitus with a disease-specific enteral formula: Stage II of a randomized, controlled multicenter trial. JPEN J. Parenter Enter. Nutr. 2009, 33, 37–49. [Google Scholar] [CrossRef]
- Takagi, S.; Utsunomiya, K.; Kuriyama, S.; Yokoyama, H.; Iwabuchi, M.; Takahashi, H.; Takahashi, S.; Kinouchi, Y.; Hiwatashi, N.; Funayama, Y.; et al. Effectiveness of an ’half elemental diet’ as maintenance therapy for Crohn’s disease: A randomized-controlled trial. Aliment. Pharmacol. Ther. 2006, 24, 1333–1340. [Google Scholar] [CrossRef]
- McGough, C.; Baldwin, C.; Norman, A.; Frost, G.; Blake, P.; Tait, D.; Khoo, V.; Harrington, K.; Andreyev, H.J.N. Is supplementation with elemental diet feasible in patients undergoing pelvic radiotherapy? Clin. Nutr. 2006, 25, 109–116. [Google Scholar] [CrossRef]
- Piquet, M.-A.; Ozsahin, M.; Larpin, I.; Zouhair, A.; Coti, P.; Monney, M.; Monnier, P.; Mirimanoff, R.-O.; Roulet, M. Early nutritional intervention in oropharyngeal cancer patients undergoing radiotherapy. Support. Care Cancer 2002, 10, 502–504. [Google Scholar] [CrossRef]
- Tsujikawa, T.; Satoh, J.; Uda, K.; Ihara, T.; Okamoto, T.; Araki, Y.; Sasaki, M.; Fujiyama, Y.; Bamba, T. Clinical importance of n-3 fatty acid-rich diet and nutritional education for the maintenance of remission in Crohn’s disease. J. Gastroenterol. 2000, 35, 99–104. [Google Scholar] [CrossRef]
- Lawson, R.; Doshi, M.; Ingoe, L.; Colligan, J.; Barton, J.; Cobden, I. Compliance of orthopaedic patients with postoperative oral nutritional supplementation. Clin. Nutr. 2000, 19, 171–175. [Google Scholar] [CrossRef]
- Park, R.H.; Allison, M.C.; Lang, J.; Spence, E.; Morris, A.J.; Danesh, B.J.; Russell, R.I.; Mills, P.R. Randomised comparison of percutaneous endoscopic gastrostomy and nasogastric tube feeding in patients with persisting neurological dysphagia. BMJ 1992, 304, 1406–1409. [Google Scholar] [CrossRef]
- Brown, T.E.; Banks, M.D.; Hughes, B.G.M.; Lin, C.Y.; Kenny, L.M.; Bauer, J.D. Randomised controlled trial of early prophylactic feeding vs standard care in patients with head and neck cancer. Br. J. Cancer 2017, 117, 15–24. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Goh, K.-H.; Acharyya, S.; Ng, S.Y.-E.; Boo, J.P.-L.; Kooi, A.H.-J.; Ng, H.-L.; Li, W.; Tay, K.-Y.; Au, W.-L.; Tan, L.C.-S. Risk and prognostic factors for pneumonia and choking amongst Parkinson’s disease patients with dysphagia. Park. Relat. Disord. 2016, 29, 30–34. [Google Scholar] [CrossRef] [PubMed]
- Peerawong, T.; Phungrassami, T.; Pruegsanusak, K.; Sangthong, R. Comparison of treatment compliance and nutritional outcomes among patients with nasopharyngeal carcinoma with and without percutaneous endoscopic gastrostomy during chemoradiation. Asian Pac. J. Cancer Prev. 2012, 13, 5805–5809. [Google Scholar] [CrossRef] [PubMed]
- Aracil-Lavado, E.; Wanden-Berghe, C.; Sanz-Valero, J. Evaluation of quality of life according to the nutritional status of the adult palliative patient: Systematic review. Hosp. Domic. 2017, 1, 199–210. [Google Scholar] [CrossRef]
- Tomás-Casterá, V.; Sanz-Valero, J.; Juan-Quilis, V. Bibliometric analysis of the scientific production and consumption on nutrition journals indexed in scielo network. Nutr. Hosp. 2013, 28, 969–970. [Google Scholar] [PubMed]
- Franco-López, A.; Sanz-Valero, J.; Culebras, J.M. To publish in Spanish or in any non English language, negative for impact factor and citations. J. Negat. Posit. Results 2016, 1, 65–70. [Google Scholar] [CrossRef]
- Ravasco, P.; Grillo, I.M.; Camilo, M. Cancer wasting and quality of life react to early individualized nutritional counselling! Clin. Nutr. 2007, 26, 7–15. [Google Scholar] [CrossRef]
- Planas Vilá, M.; Wanden-Berghe, C.; de la Cuerda, C.; Grupo NADYA-SENPE. Guía de Nutrición Enteral Domiciliaria en el Sistema Nacional de Salud, 2nd ed.; Ministerio de Sanidad y Consumo: Madrid, Spain, 2008. Available online: http://bit.ly/2xYkSPH (accessed on 19 July 2019).
- Bankhead, R.; Boullata, J.; Brantley, S.; Corkins, M.; Guenter, P.; Krenitsky, J.; Lyman, B.; Metheny, N.A.; Mueller, C.; Robbins, S.; et al. Enteral nutrition practice recommendations. JPEN J. Parenter Enter. Nutr. 2009, 33, 122–167. [Google Scholar] [CrossRef]
- Lehmann, A.; Aslani, P.; Ahmed, R.; Celio, J.; Gauchet, A.; Bedouch, P.; Bugnon, O.; Allenet, B.; Schneider, M.P. Assessing medication adherence: Options to consider. Int. J. Clin. Pharm. 2014, 36, 55–69. [Google Scholar] [CrossRef]
- Wanden-Berghe, C.; Nolasco, A.; Planas, M.; Sanz-Valero, J.; Rodríguez, T.; Cuerda, C.; Guardiola, R.; Castelló-Botia, I. Health-related quality of life according to the main caregiver in patients with home nutritional support. Med. Clín. 2008, 131, 281–284. [Google Scholar] [CrossRef]
- Vrijens, B.; De Geest, S.; Hughes, D.A.; Przemyslaw, K.; Demonceau, J.; Ruppar, T.; Dobbels, F.; Fargher, E.; Morrison, V.; Lewek, P.; et al. A new taxonomy for describing and defining adherence to medications. Br. J. Clin. Pharmacol. 2012, 73, 691–705. [Google Scholar] [CrossRef] [PubMed]
- Wanden-Berghe Lozano, C.; Campos, C.; Burgos Peláez, R.; Álvarez, J.; Frias Soriano, L.; Matia Martín, M.P.; Lobo-Támer, G.; de Luis Román, D.A.; Gonzalo Marín, M.; Gómez Candela, C.; et al. NADYA-SENPE Group. Spanish home enteral nutrition registry of the year 2016 and 2017 from the NADYA-SENPE Group. Nutr. Hosp. 2019, 36, 233–237. [Google Scholar] [CrossRef] [PubMed]
- Kozeniecki, M.; Fritzshall, R. Enteral Nutrition for Adults in the Hospital Setting. Nutr. Clin. Pract. 2015, 30, 634–651. [Google Scholar] [CrossRef] [PubMed]
- Sanchis, J.; Gich, I.; Pedersen, S.; Aerosol Drug Management Improvement Team (ADMIT). Systematic review of errors in inhaler use: Has patient technique improved over time? Chest 2016, 150, 394–406. [Google Scholar] [CrossRef]
- Náfrádi, L.; Nakamoto, K.; Schulz, P.J. Is patient empowerment the key to promote adherence? A systematic review of the relationship between self-efficacy, health locus of control and medication adherence. PLoS ONE 2017, 12, e0186458. [Google Scholar] [CrossRef] [PubMed]
- Gearing, R.E.; Townsend, L.; MacKenzie, M.; Charach, A. Reconceptualizing Medication Adherence: Six Phases of Dynamic Adherence. Harv. Rev. Psychiatry 2011, 19, 177–189. [Google Scholar] [CrossRef] [PubMed]
- Eggerth, A.; Hayn, D.; Schreier, G. Medication management needs information and communications technology-based approaches, including telehealth and artificial intelligence. Br. J. Clin. Pharmacol. 2019. [Google Scholar] [CrossRef]
- Consejo General de Colegios Oficiales de Farmacéuticos (CGCOF). Guía Práctica Para los Servicios de Atención Farmacéutica en Farmacia Comunitaria; CGCOF: Madrid, Spain, 2010; Available online: http://bit.ly/2YxQhDv (accessed on 1 August 2019).
- Bubalo, J.; Clark, R.K.; Jiing, S.S.; Johnson, N.B.; Miller, K.A.; Clemens-Shipman, C.J.; Sweet, A.L. Medication adherence: Pharmacist perspective. J. Am. Pharm. Assoc. 2010, 50, 394–406. [Google Scholar] [CrossRef]
- Conn, V.S.; Ruppar, T.M. Medication adherence outcomes of 771 intervention trials: Systematic review and meta-analysis. Prev. Med. 2017, 99, 269–276. [Google Scholar] [CrossRef]
- Milosavljevic, A.; Aspden, T.; Harrison, J. Community pharmacist-led interventions and their impact on patients’ medication adherence and other health outcomes: A systematic review. Int. J. Pharm. Pract. 2018, 26, 387–397. [Google Scholar] [CrossRef]
- Haynes, R.B.; McDonald, H.P.; Garg, A.X. Helping patients follow prescribed treatment: Clinical applications. JAMA 2002, 288, 2880–2883. [Google Scholar] [CrossRef] [PubMed]
- Lumbreras, B.; López-Pintor, E. Impact of changes in pill appearance in the adherence to angiotensin receptor blockers and in the blood pressure levels: A retrospective cohort study. BMJ Open 2017, 7, 012586. [Google Scholar] [CrossRef] [PubMed]
- Domingo-Pueyo, A.; Sanz-Valero, J.; Wanden-Berghe, C. Effects of occupational exposure to chromium and its compounds: A systematic review. Arch. Prev. Riesgos Laborales 2014, 17, 142–153. [Google Scholar] [CrossRef] [PubMed]
- Bernabeu-Martínez, M.A.; Merino, M.R.; Gago, J.M.S.; Sabucedo, L.M.A.; Wanden-Berghe, C.; Sanz-Valero, J. Guidelines for safe handling of hazardous drugs: A systematic review. PLoS ONE 2018, 13, e0197172. [Google Scholar] [CrossRef] [PubMed]
- Apolo Carvajal, F. Guia farmacogeriátrica: Nutrición Enteral; Comunidad de Madrid: Madrid, Spain, 2018; Available online: http://bit.ly/2mzUGJ5 (accessed on 27 September 2019).
| Author, Year | Design | Population Studied | Pathology | Scope | Country | Period of Study | Type of Nutrition | Form and Frequency of Administration | Technique for “Measuring” Adherence | Observed Outcome |
|---|---|---|---|---|---|---|---|---|---|---|
| Hirai et al. 2019 [11] | Comparative: Prospective multicenter cohorts | N = 72 M/F: 52/20 Age: IG (18–59; median 27); CG (18–70; median 28 years) | Crohn’s disease in maintenance treatment with anti-TNF antibodies | PNH | Japan | Recruitment: July 2011–March 2014. Monitoring period: 2 years. | PEN | Oral or tube. Formula Standard ≥900 kcal/day | Doctor-patient interviews every 8 weeks. Measurement of compliance with the prescribed amount | Low adherence rate in IG. Only 11/37 patients were adherent to EN. |
| Wall et al. 2018 [12] | Prospective nonrandomized trial | N = 38 Age: EEN Group (15.8–38.4; median 23.3); PEN Group: (16.5–38.2; median 19.2) | Chron’s disease (active) | PNH | New Zealand | Recruitment: May 2013–December 2015. Monitoring period: 8 weeks. | IG: EEN + EEN CG: EEN + PEN | Oral. 2 weeks. EEN + 6 weeks EEN or PEN. | (1) Monitoring by dietitian at weeks 0, 2, 4, 6 and 8. (2) Record of nutritional and anthropometric markers of nutritional status (3) Assessment of ingestion from the electronic self-report of intake by patient (dietary intake). | Significant improvement of symptoms, nutritional and inflammatory markers at 2 weeks. No effect at 8 weeks |
| Benton et al. 2018 [13] | Comparative study of historical cohort | N: 30 Age: IG (48–77, median = 62); CG (50–78, median = 61) | Esophagectomy | PNH | Australia | IG: Recruitment: October 2014–November 2016; CG: historical cohort of patients undergoing surgery between January 2011 and December 2012. Monitoring period: 42 days | PEN | Form: JS. (1) IG: surgery, days 1–3: clear fluids; days 4-12 (discharge): soft diet + EN. Days 12–42: oral diet + oral EN (2) CG: surgery, days 1–3 EN; days 4–7 clear fluids + EN; days 8–12 (discharge): soft diet + EN on demand. Days 12–42 total cessation EN. | Measures at the beginning and at the end of the study; (1) Nutritional status; (2) IG: Telephone interview with dietician 1 week after discharge; (3) Dietary intake (3 days of patient reporting) | The calorie and protein requirements were not met at day 42 postsurgery in any group. |
| Deane et al. 2018 [14] | Randomized clinical trial | N: 84 M/F: 48/32 Age: CG (mean = 55 (SD 16)); IG (mean 60 (SD 17)) | Critical patients | PH | USA | Monitoring period: 7 days | EEN | Form: Gastric tube. Frequency: Individualized determination of the requirements in each patient. | Adequacy of delivered daily amount (delivered/prescribed × 100) | No differences between groups in the increase in calories and proteins received by the critical patient. |
| Brown et al. 2017 [15] | Randomized clinical trial | N: 125 M/F: 112/13 Age: median 60 years | Neoplasia of the head and neck | PNH | Australia | Recruitment: September 2012 to June 2015. Monitoring period: NC | PEN | Form: GS. Frequency: IG: (Prophylactic phase: early intubation prior to surgery): EN+ oral intake. Clinical nutrition phase: IG: increment EN; CG: EN according to nutritional requirements | (1) Daily self-support of EN intake; (2) record of symptoms from nutritional impact; (3) Reasons for noncompliance. Weekly collection by the dietitian | Overall adherence to tube feeding was significantly greater in IG (58% versus 38% CG). Early tube feeding may improve patient adherence to clinically indicated feeding. |
| Healy et al. 2017 [16] | Randomized clinical trial | N = 191 (IG = 97; CG = 94). M/F = NC Age: Not available | Esophagus neoplasia | PNH | Ireland | Recruitment period: January 2011 to December 2014. Monitoring period: 6 months | PEN | G1: omega 3 EN, oral or NG G2 = standard EN. Frequency: G1 and G2 = 600 kcal/day 3 days before and 7 days after two 17-day cycles of chemotherapy + oral intake. | (1) Self-reporting of compliance by the patient and analysis of product dispensed and returned (recount). (2) Assessment of quality of life, (oncology generic questionnaire) | Adherence in patients at admission: 98%; at home: 96%. Mean days of EN lost: 1.1 (range 0 to 13 days). Differences in quality of life between the two groups. |
| Harvey et al. 2016 [17] | Randomized clinical trial | N: 1197 (EN group) M/F: 725/472 Age: 62.9 ± 15.4 | Critical patients | PH | United Kingdom | Recruitment: June 2011–October 2013. Monitoring period: 5 days | NP vs EEN | Form: NG/ND EN. Frequency: 1365–2540 kcal/day. | Adherence to nutritional protocol, with sporadic monitoring visits. during the first 120 h. | Adherence rate = 97%. Patients in the EEN group were more likely to have complete days without nutritional support. |
| Zhao 2015 [18] | Randomized clinical trial | N: 126 M/F: IG = 39/24; CG = 37/26 Age: Mean = 52.8 ± 9.5 years | Patients admitted to the Intensive Care Unit | PH | China | Recruitment: January–December 2014. Monitoring period: 7 days. | EEN | Form: NG tube. Frequency: CG = 430 g vegan formula IG = 500 g improved EN formula: 50 mL/4 h | Questionnaire of patient satisfaction at hospital discharge. | Satisfaction greater in the IG. |
| Stow et al. 2015 [19] | Randomized clinical trial | N: 93 M/F: 82% female. Age: 65 years on average | Assisted living home care patients at risk of protein-energy malnutrition (MEP) | PNH | United Kingdom | Recruitment: From 2013–2014. Monitoring period: 6 months | PEN | Form: oral. Three groups: (1) standard care; (2) food-based intervention (increase in calories and protein) and (3) EN supplements 600 kcal and 24 g protein/day. | Assessment at initiation, 3 and 6 months. (1) Record (dietitian) of daily food intake or supplements. Adherence calculation: mean ingested intake, at 3 and 6 months. (2) Anthropometric parameters; (3) Satisfaction and quality of life (no results are shown) | Adherence to interventions = 74% (3 months) and 67% (6 months). 86% of the patients met less than 50% of the requirements of the interventions. Better compliance rates were obtained in food-based intervention versus supplements in all measurements. |
| Hamza et al. 2015 [20] | Randomized clinical trial | N: 37 M/F: 20/17 Age: IG 63 (58–69) CG 67 (63–70) | Pancreatic neoplasia | PH | United Kingdom | Recruitment: 28 months; Monitoring period: 21 days | Preoperative: PEN; Postoperative: EEN | Preoperative period. Form: oral EN. Frequency: 3 EN formula cartons + Normal intake. Postoperative period (7 days). Form: NJ catheter. Frequency: 100 kcal/100 mL up to a target of 25 kcal/kg. | Measurements: 14 days before surgery; on the day of surgery and on days 3 and 7 postsurgery. (1) Journal of compliance: record of quantities ingested 14 days before surgery. (2) Assessment of compliance: daily comparison with the count of the number of containers not consumed | No significant differences in the calories consumed between the two groups. No data on the assessment of compliance. |
| Bowrey et al. 2015 [21] | Randomized clinical trial | N = 41 M/F = 36/5 Age: IG (mean = 64.6 (SD = 8)); CG (mean = 63.1 (SD = 8.7) | Neoplasms of the esophagus or stomach | PNH | United Kingdom | Recruitment: October 2007–June 2009. Monitoring period: 6 months | PEN | Form: JS; Frequency: IG: nocturnal EN administration of 50% energy + protein requirements; CG (standard care): EN only if needed. | Assessment at discharge, at 6 weeks post discharge and at 3 and 6 months postsurgery. (1) Written information on dietary advice and nutritional supplements; (2) Nutritional intake: total energy, proteins, oral nutritional supplements and food by JS; (3) Quality of life. General and specific questionnaires | The results of quality of life were similar between the two groups. Monitoring of adherence to EN was not performed. |
| Sukkar et al. 2013 [22] | Comparative study | N: 22 M/F: 8/14 Age mean: 48.64 (SD = 10.10) | Obese patients | PNH | Italy | Recruitment period: March 2011–October 2011; Monitoring period: 30 days | EEN (10 days); Hypocaloric diet (20 days) | EEN phase: Form: NG: frequency: 10 days EN formula, individual calculation of requirements. Hypocaloric phase: form = oral; frequency: 20 days, caloric deficit of 10% on patient needs | (1) Lifestyle changes, diet and treatments explained by the dietitian at the beginning (2) Technical explanation at the beginning regarding the handling of EN pump; (3) EEN phase: recording by the patient of complications with the feeding tube. Monitoring: at the beginning and at the end of the study. | Three patients (9%) abandoned the study due to low compliance. Monitoring of diet compliance was not performed, only complications of NG tube management |
| Shirakawa et al. 2012 [23] | Comparative study | N = 25 M/F: 19/6 Age, median = 64 years | Pancreatic neoplasia | PH | Japan | Recruitment period: February 2005–November 2006. Monitoring period 5 days | PEN | IG. Form: oral. EN + usual diet 5 days before surgery CG = patients operated without preoperative intake of EN | Assessment of compliance with EN formula: monitoring by the physician during the study period | 82.6% (19 patients) fully complied with the protocol of ingestion of EN. Four patients were not compliant. |
| Sadasivan et al. 2012 [24] | Randomized clinical trial | N: 100 M/F: 67/33 Age: Not available | Neoplasia of the head and neck | PNH | India | Recruitment period: 2009–2011. Monitoring period: 6 months | EEN | Form: GS (G1) and NG tube (G2). | Assessment at weeks 1 and 6 and 6 months. (1) Assessment of nutritional status, complications of nutrition and patient satisfaction (pain, feeding tube management, comfort) using the questionnaire [EORTC QLQH&N35]. | No documentation or monitoring of intake in terms of calories. Higher satisfaction and lower rate of GS versus NG tube complications at 6 weeks. No comparative data at 6 months. |
| Kraft et al. 2012 [25] | Prospective, randomized controlled study | N = 26 M/F: 10/16 Age: median = 79.8, SD = 7.3 | Geriatric patients at risk of malnutrition | PNH | Ireland | Recruitment period: 1 March–31 August 2010. Monitoring period: Not available | PEN | Form: oral route. Frequency: nutritional supplements of 600 kcal. | CG: monitoring at 6 months, measure of nutritional status; IG: telemedicine monitoring. Daily assessment of body weight, number of supplements ingested and health status using telematic questionnaire. | There were no data on the rate of adherence in the IG, nor comparative data between the two groups. Monitoring nutrition and contact with HCP can improve adherence to EN. |
| Miyata et al. 2012 [26] | Randomized clinical trial | N = 91 G1 = 47 M/F = 34/13 Age: mean 62.4 G2 = 44 M/F = 35/9 Age: mean 63.2 | Esophagus neoplasia | PNH | Japan | Recruitment period: Not available Monitoring period: 2 17-day cycles of chemotherapy spaced 4 weeks apart | PEN: G1 NPP: G2 | G1: omega 3 EN, oral or NG tube. Frequency: G1 and G2 = 600 kcal/day 3 days before and 7 days after 2 17-day cycles of chemotherapy+ oral diet. | Daily consumption of calories (not indicated how it was measured) | Similar calorie intake between the two groups. Similar nutritional indicators between both groups at the end of the study. Six patients from G1 abandoned EN due to its adverse effects |
| Pohl et al. 2009 [27] | Randomized clinical trial | N: 105 M/F: 50/47 Age: 44–91 | Type 2 diabetes | PNH | Germany | Recruitment period: June 2004–June 2005. Monitoring period: 84 days | EEN | Form: NG tube or GS Frequency: continuous administration 30 mL (27 kcal)/kg/day to a maximum of 2025 kcal/day. IG: disease-specific EN formula; CG: standard isoenergetic formula | No monitoring of adherence. The presence of adverse effects (tolerability) on days 1, 28, 56 and 84 were measured. | No differences in EN tolerability. |
| Takagi et al. 2006 [28] | Randomized clinical trial | N: 51 M/F: 37/14 Age (mean, SD: IG = 30.8 (11.1) and CG = 28.9 (8.1) | Crohn’s disease | PNH | Japan | Recruitment period; 2002–2005. Monitoring period = 11, 9 months (mean monitoring: 1–28 months, SD = 1.7). | PEN | IG: (1/2 EN + 1/2 normal diet without restriction). Form = EN: oral/intubation according to patient preference Formula = IG: 900–1200 kcal/day elemental diet. CG = normal diet without restriction | Patients were advised on feeding and calculating the daily food intake. (1) At least one visit with clinicians every 3 months: record of adverse effects and anthropometric parameters. Ingestion of EN was not assessed. | Similar adherence in both groups, no concrete data nor specification regarding how it was measured |
| McGough et al. 2006 [29] | Randomized clinical trial | N: 50 M/F: 6/44 Age: (mean, SD) (58, 55–61) | Neoplasia of the pelvis | PNH | United Kingdom | Recruitment period: May 2003–May 2004. Monitoring period: 5 weeks | PEN | Form: Oral route, EN + normal diet. Frequency: different types of formulas and differences in% of daily caloric intake substituted by EN; G1 = 20%; G2 and G4 = 50%; G3 = 75% and G5 = 50%. | (1) Daily filled out by the patient with daily intake, flavor chosen and relevant comments. Collection at the end of treatment. (2) Weekly meeting with dietitian for support and advice on diet. Adherence was determined by evaluating the container counts and weekly diaries of patients | Only three patients complied with the entire treatment (3/50). The number of patients who consumed EN decreased over time: 92% week 1 to 46% week 5. The type of formulation did not affect compliance. In lower prescribed volumes, lower intake was observed. |
| Piquet et al. 2002 [30] | Comparative study | N: 90 M/F = 43/2 (IG), 42/3 (CG). Age = IG: 61 years (SD = 1.5); CG: 59 years (SD = 1.5) | Oropharyngeal neoplasia | PNH | Japan | Recruitment period: September 1998 to September 1999. Monitoring period: 6–7 months | IG: PEN; CG: EN (historical cohort of patients, does not specify EN/PEN) | IG. Form: GS, NG tube or oral route. Frequency: 30 kcal/kg/day, polymeric formula | IG: Initial nutritional assessment before the initiation of radiation therapy: recording of dietary intake, current weight, usual weight, height and Body Mass Index (BMI). Monitoring by dietitian at least three times during radiotherapy treatment. CG: Not specified. | IG: Compliance in 80% of patients, even in alcoholic patients because nutritional support was proposed by medical staff at an early stage as an important component of treatment. |
| Tsujikawa et al. 2000 [31] | Comparative study | N: 20 M/F = 13/7 Age: 28.2 (17–49) | Crohn’s disease | PNH | Japan | Recruitment period: January 1994–1997 | Diet rich in omega-3 fatty acids and EN at home; nutritional education | Form: oral route. G1 (omega 3-rich diet) + nutrition education; G2: elemental diet | (1) Dietitian nutrition education prior to discharge and monitoring compliance every 2–4 weeks in the first month. (2) Satisfaction questionnaire for patients with EN delivered when visiting the hospital (periodicity is not specified). | 80% of patients did not want to continue taking the elemental diet because it decreased their quality of life. Omega-3 diet + nutritional education improved patient satisfaction, and therefore compliance. |
| Lawson et al. 2000 [32] | Randomized clinical trial | N: 187 (84 IG, 97 CG) M/F: 27/57 Age: (mean, SD) 72 years (40–88) | Patients undergoing orthopedic intervention | PH | United Kingdom | Recruitment period: 18 months. Average monitoring period: 14.4 days | PEN | IG: Form, oral route. Frequency: two oral supplements/day during hospital stay + usual meals. CG: standard diet | (1) Signature by the patient when the supplement was given. Patients, nurses and home health staff recorded the amount ingested for each supplement. (2) Review by dietitian weekly: discussion with the patient regarding progress and recording of total ingested and/or rejected supplements. (3) Record by the patient of food and beverages consumed apart from hospital catering. Adherence: difference between the amount consumed and the amount prescribed. | No patient was 100% adherent. Mean compliance = 14.9%; mean = 0.9%. An average of 6.1 supplements were taken daily, for an average of 6.7 days. Patients took half of the total prescribed volume for half the indicated time. Readjustments of EN: 76% of patients changed the type of supplement, and eight patients decided not to take any. Lower level of compliance in patients with BMI less than 25 kg/m2. |
| Park et al. 1992 [33] | Randomized clinical trial | N: 40 M/F: 22/18 Age: 56 years (SD = 4.8) G1 and 65 years (SD = 2.6) G2 | Neurological patient with persistent dysphagia | PH | United Kingdom | Monitoring period: 28 days | EEN | Form: G1: NG tube; G2: GS. Frequency: infusion 24 h. Volumes adjusted to the needs of each patient. | (1) Measured daily record of the remnant in the feeding tube. (2) Adherence = amount received, % above the prescribed. (3) EN Acceptance Measurement Questionnaire completed by patient or family. (4) Treatment failure | 93% (SD = 2%) adherence G2 versus G1 (55% (SD = 4%)). Increased weight gain in the first week of treatment, G2 versus G1. 95% G1 patients had treatment administration failures, and none in G2. |
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | 19 | 20 | 21 | 22 | 23 | 24 | 25 | Total | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Hirai et al. 2019 [11] | 0.5 | 1 | 0.5 | 1 | 0 | 0.5 | 0.5 | 0 | 0 | 0 | 0 | 0.5 | 0.5 | 0.5 | 1 | 1 | 0.5 | 0 | 1 | 1 | 0 | 1 | 0 | 0 | 0 | 7 28% |
| Wall et al. 2018 [12] | 0.5 | 1 | 0 | 1 | 1 | 0.5 | 0 | 0 | 0 | 0 | 0 | 0.5 | 1 | 0 | 1 | 1 | 0.5 | 0 | 1 | 1 | 1 | 1 | 0 | 0 | 1 | 11 44% |
| Benton et al. 2018 [13] | 0.5 | 1 | 1 | 1 | 1 | 0.5 | 0.5 | 0.5 | 0 | 0 | 0 | 0.5 | 1 | 0 | 1 | 1 | 0.5 | 1 | 0 | 1 | 0 | 1 | 0 | 0 | 1 | 11 44% |
| Deane et al. 2018 [14] | 1 | 1 | 0.5 | 0.5 | 1 | 0.5 | 0 | 1 | 0 | 0 | 0.5 | 0.5 | 1 | 0.5 | 1 | 1 | 0.5 | 0 | 1 | 1 | 0 | 1 | 0 | 1 | 0 | 11 44% |
| Brown et al. 2017 [15] | 0.5 | 1 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0.5 | 1 | 0.5 | 1 | 1 | 0.5 | 0 | 1 | 1 | 0 | 1 | 0 | 0 | 1 | 9 36% |
| Healy et al. 2017 [16] | 0 | 0.5 | 0 | 1 | 1 | 0.5 | 0.5 | 1 | 0 | 0 | 0.5 | 0.5 | 1 | 0.5 | 1 | 1 | 0.5 | 0 | 1 | 0 | 0 | 1 | 0 | 0 | 0 | 11.5 46% |
| Harvey et al. 2016 [17] | 1 | 1 | 0.5 | 1 | 1 | 0.5 | 1 | 1 | 1 | 1 | 0.5 | 1 | 1 | 0.5 | 1 | 1 | 0.5 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 20 80% |
| Zhao 2015 [18] | 0.5 | 0.5 | 0 | 0.5 | 1 | 0.5 | 0 | 0 | 0 | 0 | 0 | 0.5 | 0 | 0 | 0 | 0 | 0.5 | 0 | 1 | 0 | 1 | 1 | 0 | 0 | 0 | 7 28% |
| Stow et al. 2015 [19] | 1 | 1 | 0.5 | 1 | 1 | 0.5 | 0.5 | 1 | 1 | 1 | 0.5 | 0.5 | 1 | 1 | 1 | 1 | 0.5 | 0 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 21 84% |
| Hamza et al. 2015 [20] | 1 | 1 | 0.5 | 0.5 | 1 | 0.5 | 0.5 | 1 | 0 | 0 | 0 | 1 | 1 | 0 | 1 | 1 | 0.5 | 0 | 0 | 1 | 0 | 1 | 0 | 0 | 0 | 12.5 50% |
| Bowrey et al. 2015 [21] | 1 | 1 | 0.5 | 0.5 | 0 | 1 | 0.5 | 1 | 0 | 0.5 | 0 | 0.5 | 1 | 0.5 | 1 | 1 | 0 | 0 | 1 | 0 | 1 | 1 | 0 | 0 | 1 | 14 56% |
| Sukkar et al. 2013 [22] | 0 | 1 | 0 | 0.5 | 1 | 0.5 | 0 | 0 | 0 | 0 | 0.5 | 0.5 | 1 | 0.5 | 1 | 0 | 0.5 | 0 | 1 | 0 | 1 | 1 | 0 | 1 | 1 | 12 48% |
| Shirakawa et al. 2012 [23] | 0.5 | 1 | 0 | 0.5 | 1 | 0.5 | 0 | 0 | 0 | 0 | 0 | 0.5 | 0.5 | 0.5 | 1 | 1 | 0.5 | 0 | 1 | 0 | 1 | 1 | 0 | 0 | 0 | 11.5 46% |
| Sadasivan et al. 2012 [24] | 0 | 1 | 1 | 0.5 | 1 | 0.5 | 0.5 | 1 | 0 | 0 | 0 | 0.5 | 1 | 0.5 | 1 | 1 | 0.5 | 0 | 1 | 1 | 0 | 1 | 0 | 0 | 0 | 13 52% |
| Kraft et al. 2012 [25] | 0 | 1 | 0.5 | 0.5 | 1 | 0.5 | 0.5 | 0.5 | 0 | 0 | 0 | 0 | 1 | 0.5 | 1 | 1 | 0 | 0 | 0 | 0 | 1 | 1 | 0 | 0 | 1 | 11 44% |
| Miyata et al. 2012 [26] | 1 | 1 | 0 | 0.5 | 1 | 0.5 | 0 | 0.5 | 0 | 1 | 0 | 0.5 | 1 | 0.5 | 1 | 1 | 0.5 | 0 | 1 | 1 | 0 | 1 | 0 | 0 | 1 | 14 56% |
| Pohl et al. 2009 [27] | 1 | 0.5 | 0.5 | 1 | 1 | 0.5 | 0.5 | 0.5 | 1 | 1 | 1 | 0.5 | 1 | 1 | 1 | 1 | 1 | 0 | 1 | 1 | 1 | 1 | 0 | 0 | 0 | 18 72% |
| Takagi et al. 2006 [28] | 1 | 1 | 0 | 1 | 1 | 0.5 | 0.5 | 1 | 1 | 1 | 0.5 | 0.5 | 1 | 1 | 1 | 1 | 0.5 | 0 | 1 | 1 | 1 | 1 | 0 | 0 | 0 | 17.5 70% |
| McGough et al. 2006 [29] | 0.5 | 1 | 0.5 | 1 | 1 | 0.5 | 0.5 | 1 | 1 | 1 | 0 | 1 | 0.5 | 0 | 1 | 1 | 0 | 1 | 1 | 0 | 1 | 1 | 0 | 0 | 1 | 16.5 66% |
| Piquet et al. 2002 [30] | 0 | 1 | 0 | 0 | 1 | 0.5 | 0 | 0 | 0 | 0 | 0 | 0.5 | 0.5 | 0.5 | 1 | 1 | 0.5 | 0 | 1 | 0 | 1 | 1 | 0 | 0 | 0 | 9.5 38% |
| Tsujikawa et al. 2000 [31] | 0 | 1 | 0 | 1 | 1 | 0.5 | 0 | 0 | 0 | 0 | 0 | 0.5 | 0 | 0 | 0 | 0 | 0.5 | 1 | 0 | 1 | 0 | 1 | 0 | 0 | 0 | 7.5 30% |
| Lawson et al. 2000 [32] | 0.5 | 1 | 0 | 1 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0.5 | 0.5 | 0 | 0 | 0 | 0.5 | 0 | 1 | 0 | 0 | 1 | 0 | 0 | 0 | 7 28% |
| Park et al. 1992 [33] | 1 | 0.5 | 0.5 | 1 | 1 | 0.5 | 0.5 | 0.5 | 1 | 0 | 0 | 1 | 1 | 1 | 1 | 1 | 0.5 | 1 | 1 | 0 | 1 | 1 | 0 | 0 | 1 | 17 68% |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gea Cabrera, A.; Sanz-Lorente, M.; Sanz-Valero, J.; López-Pintor, E. Compliance and Adherence to Enteral Nutrition Treatment in Adults: A Systematic Review. Nutrients 2019, 11, 2627. https://doi.org/10.3390/nu11112627
Gea Cabrera A, Sanz-Lorente M, Sanz-Valero J, López-Pintor E. Compliance and Adherence to Enteral Nutrition Treatment in Adults: A Systematic Review. Nutrients. 2019; 11(11):2627. https://doi.org/10.3390/nu11112627
Chicago/Turabian StyleGea Cabrera, Alicia, María Sanz-Lorente, Javier Sanz-Valero, and Elsa López-Pintor. 2019. "Compliance and Adherence to Enteral Nutrition Treatment in Adults: A Systematic Review" Nutrients 11, no. 11: 2627. https://doi.org/10.3390/nu11112627
APA StyleGea Cabrera, A., Sanz-Lorente, M., Sanz-Valero, J., & López-Pintor, E. (2019). Compliance and Adherence to Enteral Nutrition Treatment in Adults: A Systematic Review. Nutrients, 11(11), 2627. https://doi.org/10.3390/nu11112627





