Prevention of Type 2 Diabetes by Lifestyle Changes: A Systematic Review and Meta-Analysis
Abstract
:1. Introduction
- (a) What is the evidence that T2D is preventable by lifestyle changes in adults with impaired glucose tolerance (IGT) and (b) what are the long-term results on the prevention of T2D?
- What is the evidence that the lifestyle changes aimed to prevent T2D also modify the risk of cardiovascular disease and microvascular complications in people with IGT?
- What is the optimal dietary composition for the prevention of T2D in people with IGT?
2. Evidence That T2D Is Preventable by Changing Lifestyles
3. Methods
3.1. Search Strategy and Study Selection
3.2. Data Extraction
3.3. Risk of Bias Assessment
3.4. Data Syntheses
3.5. Grading of the Evidence
4. Results
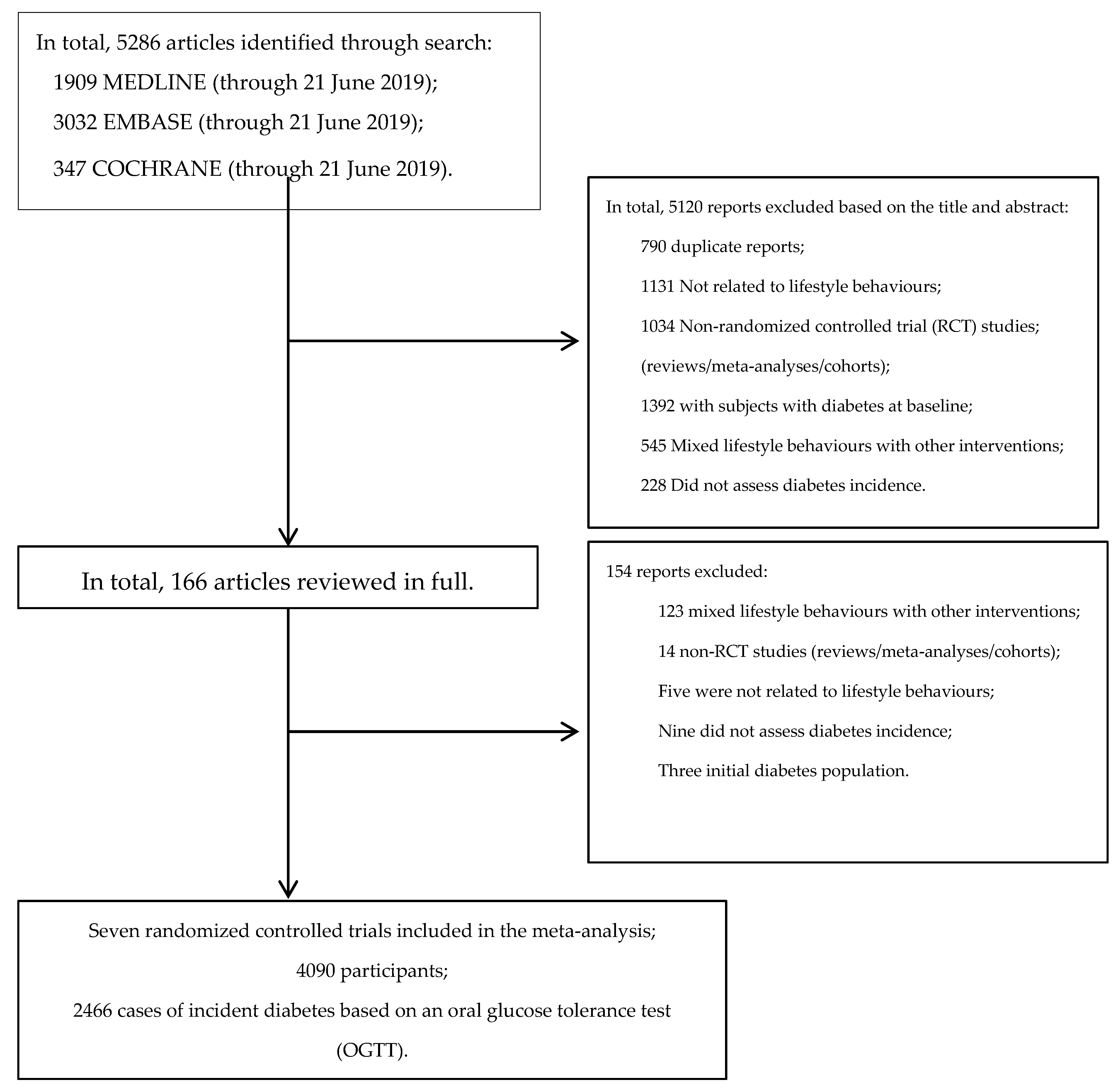
4.1. Search Results
4.2. Randomized Controlled Trials
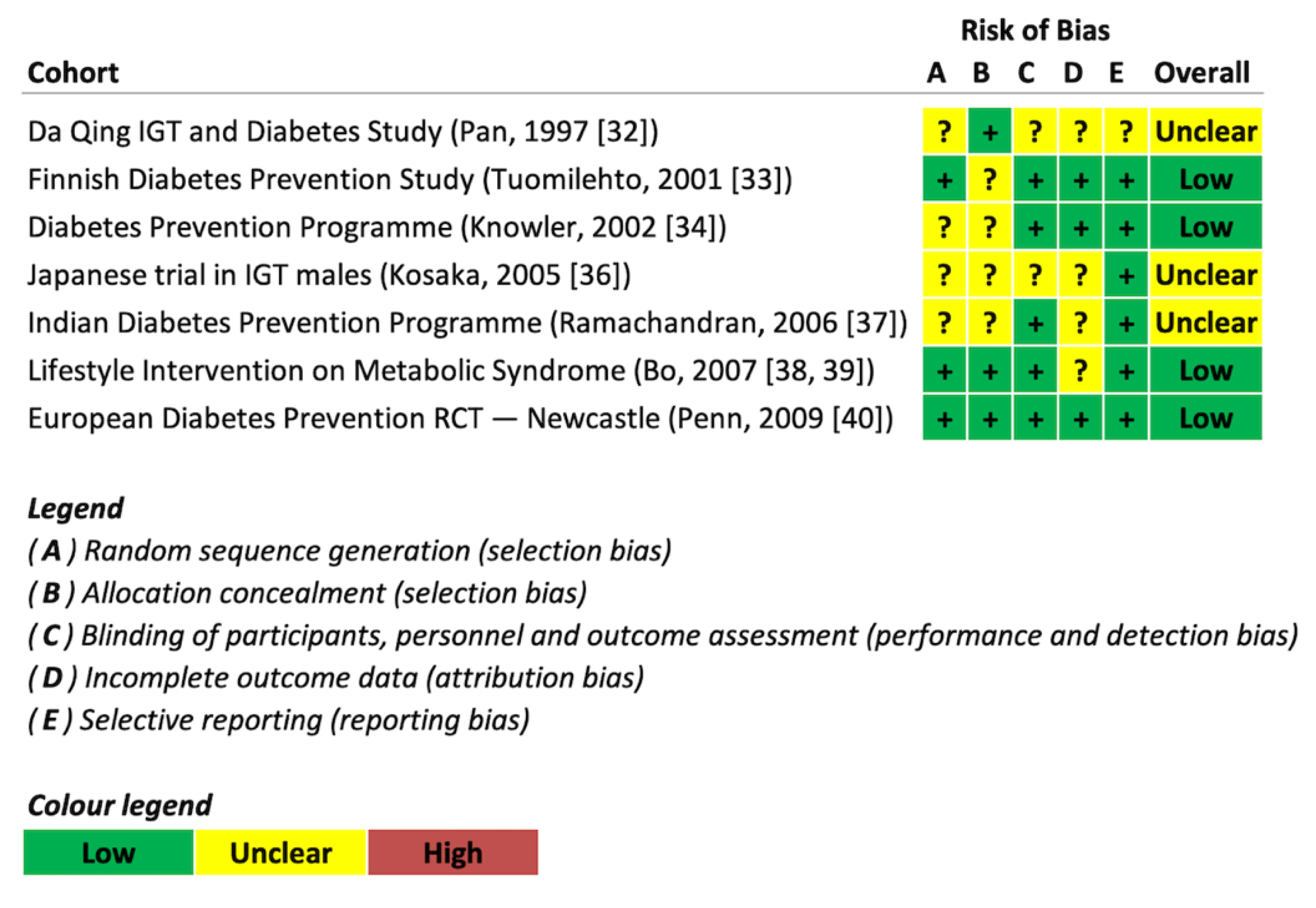
4.3. Risk of Bias
4.4. Effect of Lifestyle Changes on Type 2 Diabetes Risk
4.5. Sensitivity and Subgroup Analyses
4.6. Publication Bias
4.7. GRADE Assessment
5. Discussion on the Systematic Review and Meta-Analysis
5.1. Results in the Context of Existing Literature
5.2. Strengths and Limitations
6. Long-Term Results on the Prevention of Type 2 Diabetes
7. Evidence That the Prevention of T2D in High-Risk Individuals Results in a Lower Risk of Cardiovascular Disease (CVD) and Microvascular Complications
Discussion on Macro- and Microvascular Risk Reduction in the T2D Prevention Trials
8. Discussion on the Factors Explaining the Risk Reduction of T2D Including the Optimal Dietary Composition for the Prevention of T2D
8.1. What Are the Factors Explaining the Risk Reduction of T2D in Randomized Controlled Trials?
8.2. Weight Reduction
8.3. Optimal Diet
8.3.1. Individual Nutrients and Foods
8.3.2. Dietary Patterns
8.3.3. Diet and Weight Loss
8.3.4. Physical Activity
9. Conclusions
- We have a high certainty of evidence that T2D is preventable by changing lifestyle, i.e., weight reduction by diet change according to the current recommendations in terms of quality of fat, fiber intake, increased use of whole grain products, fruit, and vegetables, and increasing physical activity. The risk reduction of T2D is strongly related to the degree of long-term weight loss and adherence to lifestyle changes, and this preventive effect has been demonstrated to sustain for many years after active intervention.
- Additional well-controlled intervention studies are needed to identify the optimal diet to prevent T2D. Currently, a diet moderate in fat, low in saturated fat intake, rich in fiber, whole grains, and fruit and vegetables, as well as a Mediterranean-type diet, may be recommended for the prevention of T2D in prediabetes.
- There is still limited/insufficient evidence that the prevention of T2D by changing lifestyle may also prevent CVD or microvascular diseases.
Author Contributions
Funding
Conflicts of Interest
References
- International Diabetes Federation. IDF Atlas, 8th ed.; International Diabetes Federation: Brussels, Belgium, 2017. [Google Scholar]
- World Health Organization. Global Report on Diabetes 2016; WHO: Geneva, Switzerland, 2017. [Google Scholar]
- Davies, M.J.; D’Alessio, D.A.; Fradkin, J.; Kernan, W.N.; Mathieu, C.; Mingrone, G.; Rossing, P.; Tsapas, A.; Wexler, D.J.; Buse, J.B. Management of hyperglycaemia in type 2 diabetes, 2018. A consensus report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetologia 2018, 61, 2461–2498. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gloy, V.L.; Briel, M.; Bhatt, D.L.; Kashyap, S.R.; Schauer, P.R.; Mingrone, G.; Bucher, H.C.; Nordmann, A.J. Bariatric surgery versus non-surgical treatment for obesity: A systematic review and meta-analysis of randomised controlled trials. BMJ 2013, 347, 5934. [Google Scholar] [CrossRef] [PubMed]
- Douglas, I.J.; Bhaskaran, K.; Batterham, R.L.; Smeeth, L. Bariatric Surgery in the United Kingdom: A Cohort Study of Weight Loss and Clinical Outcomes in Routine Clinical Care. PLoS Med. 2015, 12, e1001925. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Diabetes Mellitus, Report of a WHO Study Group; WHO Technical Report; WHO: Geneva, Switzerland, 1985. [Google Scholar]
- Mann, J.I.; De Leeuw, I.; Hermansen, K.; Karamanos, B.; Karlstrom, B.; Katsilambros, N.; Riccardi, G.; Rivellese, A.A.; Rizkalla, S.; Slama, G.; et al. Evidence-based nutritional approaches to the treatment and prevention of diabetes mellitus. Nutr. Metab. Cardiovasc. Dis. 2004, 14, 373–394. [Google Scholar] [CrossRef]
- Higgins, J.P.T.; Green, S. Cochrane Handbook for Systematic Reviews of Interventions; Cochrane Collaboration: Oxford, UK, 2011. [Google Scholar]
- Schellenberg, E.S.; Dryden, D.M.; Vandermeer, B.; Ha, C.; Korownyk, C. Lifestyle interventions for patients with and at risk for type 2 diabetes: A systematic review and meta-analysis. Ann. Intern. Med. 2013, 159, 543–551. [Google Scholar] [CrossRef] [PubMed]
- Barry, E.; Roberts, S.; Oke, J.; Vijayaraghavan, S.; Normansell, R.; Greenhalgh, T. Efficacy and effectiveness of screen and treat policies in prevention of type 2 diabetes: Systematic review and meta-analysis of screening tests and interventions. BMJ 2017, 356, 6538. [Google Scholar] [CrossRef]
- Haw, J.S.; Galaviz, K.I.; Straus, A.N.; Kowalski, A.J.; Magee, M.J.; Weber, M.B.; Wei, J.; Narayan, K.M.V.; Ali, M.K. Long-term Sustainability of Diabetes Prevention Approaches: A Systematic Review and Meta-analysis of Randomized Clinical Trials. JAMA Intern. Med. 2017, 177, 1808–1817. [Google Scholar] [CrossRef]
- Hemmingsen, B.; Gimenez-Perez, G.; Mauricio, D.; Roque, I.F.M.; Metzendorf, M.I.; Richter, B. Diet, physical activity or both for prevention or delay of type 2 diabetes mellitus and its associated complications in people at increased risk of developing type 2 diabetes mellitus. Cochrane Database Syst. Rev. 2017, 12, 003054. [Google Scholar] [CrossRef]
- Brunetti, M.; Shemilt, I.; Pregno, S.; Vale, L.; Oxman, A.D.; Lord, J.; Sisk, J.; Ruiz, F.; Hill, S.; Guyatt, G.H.; et al. GRADE guidelines: 10. Considering resource use and rating the quality of economic evidence. J. Clin. Epidemiol. 2013, 66, 140–150. [Google Scholar] [CrossRef]
- Balshem, H.; Helfand, M.; Schunemann, H.J.; Oxman, A.D.; Kunz, R.; Brozek, J.; Vist, G.E.; Falck-Ytter, Y.; Meerpohl, J.; Norris, S.; et al. GRADE guidelines: 3. Rating the quality of evidence. J. Clin. Epidemiol. 2011, 64, 401–406. [Google Scholar] [CrossRef]
- Higgins, J.P.T.; Green, S. Cochrane Handbook for Systematic Reviews of Interventions; Wiley: Hoboken, NJ, USA, 2008; p. 672. [Google Scholar]
- Thompson, S.G.; Higgins, J.P. How should meta-regression analyses be undertaken and interpreted? Stat. Med. 2002, 21, 1559–1573. [Google Scholar] [CrossRef] [PubMed]
- Sterne, J.A.; Gavaghan, D.; Egger, M. Publication and related bias in meta-analysis: Power of statistical tests and prevalence in the literature. J. Clin. Epidemiol. 2000, 53, 1119–1129. [Google Scholar] [CrossRef]
- Guyatt, G.; Oxman, A.D.; Akl, E.A.; Kunz, R.; Vist, G.; Brozek, J.; Norris, S.; Falck-Ytter, Y.; Glasziou, P.; DeBeer, H.; et al. GRADE guidelines: 1. Introduction-GRADE evidence profiles and summary of findings tables. J. Clin. Epidemiol. 2011, 64, 383–394. [Google Scholar] [CrossRef]
- Guyatt, G.; Oxman, A.D.; Sultan, S.; Brozek, J.; Glasziou, P.; Alonso-Coello, P.; Atkins, D.; Kunz, R.; Montori, V.; Jaeschke, R.; et al. GRADE guidelines: 11. Making an overall rating of confidence in effect estimates for a single outcome and for all outcomes. J. Clin. Epidemiol. 2013, 66, 151–157. [Google Scholar] [CrossRef] [PubMed]
- Guyatt, G.H.; Oxman, A.D.; Kunz, R.; Atkins, D.; Brozek, J.; Vist, G.; Alderson, P.; Glasziou, P.; Falck-Ytter, Y.; Schunemann, H.J. GRADE guidelines: 2. Framing the question and deciding on important outcomes. J. Clin. Epidemiol. 2011, 64, 395–400. [Google Scholar] [CrossRef] [PubMed]
- Guyatt, G.H.; Oxman, A.D.; Kunz, R.; Brozek, J.; Alonso-Coello, P.; Rind, D.; Devereaux, P.J.; Montori, V.M.; Freyschuss, B.; Vist, G.; et al. GRADE guidelines 6. Rating the quality of evidence—Imprecision. J. Clin. Epidemiol. 2011, 64, 1283–1293. [Google Scholar] [CrossRef]
- Guyatt, G.H.; Oxman, A.D.; Kunz, R.; Woodcock, J.; Brozek, J.; Helfand, M.; Alonso-Coello, P.; Falck-Ytter, Y.; Jaeschke, R.; Vist, G.; et al. GRADE guidelines: 8. Rating the quality of evidence—Indirectness. J. Clin. Epidemiol. 2011, 64, 1303–1310. [Google Scholar] [CrossRef] [PubMed]
- Guyatt, G.H.; Oxman, A.D.; Kunz, R.; Woodcock, J.; Brozek, J.; Helfand, M.; Alonso-Coello, P.; Glasziou, P.; Jaeschke, R.; Akl, E.A.; et al. GRADE guidelines: 7. Rating the quality of evidence—Inconsistency. J. Clin. Epidemiol. 2011, 64, 1294–1302. [Google Scholar] [CrossRef]
- Guyatt, G.H.; Oxman, A.D.; Montori, V.; Vist, G.; Kunz, R.; Brozek, J.; Alonso-Coello, P.; Djulbegovic, B.; Atkins, D.; Falck-Ytter, Y.; et al. GRADE guidelines: 5. Rating the quality of evidence—Publication bias. J. Clin. Epidemiol. 2011, 64, 1277–1282. [Google Scholar] [CrossRef]
- Guyatt, G.H.; Oxman, A.D.; Santesso, N.; Helfand, M.; Vist, G.; Kunz, R.; Brozek, J.; Norris, S.; Meerpohl, J.; Djulbegovic, B.; et al. GRADE guidelines: 12. Preparing summary of findings tables-binary outcomes. J. Clin. Epidemiol. 2013, 66, 158–172. [Google Scholar] [CrossRef]
- Guyatt, G.H.; Oxman, A.D.; Sultan, S.; Glasziou, P.; Akl, E.A.; Alonso-Coello, P.; Atkins, D.; Kunz, R.; Brozek, J.; Montori, V.; et al. GRADE guidelines: 9. Rating up the quality of evidence. J. Clin. Epidemiol. 2011, 64, 1311–1316. [Google Scholar] [CrossRef] [PubMed]
- Guyatt, G.H.; Oxman, A.D.; Vist, G.; Kunz, R.; Brozek, J.; Alonso-Coello, P.; Montori, V.; Akl, E.A.; Djulbegovic, B.; Falck-Ytter, Y.; et al. GRADE guidelines: 4. Rating the quality of evidence—Study limitations (risk of bias). J. Clin. Epidemiol. 2011, 64, 407–415. [Google Scholar] [CrossRef] [PubMed]
- Guyatt, G.H.; Thorlund, K.; Oxman, A.D.; Walter, S.D.; Patrick, D.; Furukawa, T.A.; Johnston, B.C.; Karanicolas, P.; Akl, E.A.; Vist, G.; et al. GRADE guidelines: 13. Preparing summary of findings tables and evidence profiles-continuous outcomes. J. Clin. Epidemiol. 2013, 66, 173–183. [Google Scholar] [CrossRef] [PubMed]
- Langan, D.; Higgins, J.P.T.; Jackson, D.; Bowden, J.; Veroniki, A.A.; Kontopantelis, E.; Viechtbauer, W.; Simmonds, M. A comparison of heterogeneity variance estimators in simulated random-effects meta-analyses. Res. Synth. Methods 2019, 10, 83–98. [Google Scholar] [CrossRef] [PubMed]
- Riley, R.D.; Higgins, J.P.; Deeks, J.J. Interpretation of random effects meta-analyses. BMJ 2011, 342, 549. [Google Scholar] [CrossRef] [PubMed]
- Booth, G.L.; Kapral, M.K.; Fung, K.; Tu, J.V. Relation between age and cardiovascular disease in men and women with diabetes compared with non-diabetic people: A population-based retrospective cohort study. Lancet 2006, 368, 29–36. [Google Scholar] [CrossRef]
- Pan, X.R.; Li, G.W.; Hu, Y.H.; Wang, J.X.; Yang, W.Y.; An, Z.X.; Hu, Z.X.; Lin, J.; Xiao, J.Z.; Cao, H.B.; et al. Effects of diet and exercise in preventing NIDDM in people with impaired glucose tolerance. The Da Qing IGT and Diabetes Study. Diabetes Care 1997, 20, 537–544. [Google Scholar] [CrossRef]
- Tuomilehto, J.; Lindstrom, J.; Eriksson, J.G.; Valle, T.T.; Hamalainen, H.; Ilanne-Parikka, P.; Keinanen-Kiukaanniemi, S.; Laakso, M.; Louheranta, A.; Rastas, M.; et al. Prevention of type 2 diabetes mellitus by changes in lifestyle among subjects with impaired glucose tolerance. N. Engl. J. Med. 2001, 344, 1343–1350. [Google Scholar] [CrossRef]
- Knowler, W.C.; Barrett-Connor, E.; Fowler, S.E.; Hamman, R.F.; Lachin, J.M.; Walker, E.A.; Nathan, D.M. Diabetes Prevention Program Research, G. Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N. Engl. J. Med. 2002, 346, 393–403. [Google Scholar] [CrossRef]
- Mensink, M.; Blaak, E.E.; Corpeleijn, E.; Saris, W.H.; de Bruin, T.W.; Feskens, E.J. Lifestyle intervention according to general recommendations improves glucose tolerance. Obes. Res. 2003, 11, 1588–1596. [Google Scholar] [CrossRef]
- Kosaka, K.; Noda, M.; Kuzuya, T. Prevention of type 2 diabetes by lifestyle intervention: A Japanese trial in IGT males. Diabetes Res. Clin. Pract. 2005, 67, 152–162. [Google Scholar] [CrossRef] [PubMed]
- Ramachandran, A.; Snehalatha, C.; Mary, S.; Mukesh, B.; Bhaskar, A.D.; Vijay, V. Indian Diabetes Prevention, P. The Indian Diabetes Prevention Programme shows that lifestyle modification and metformin prevent type 2 diabetes in Asian Indian subjects with impaired glucose tolerance (IDPP-1). Diabetologia 2006, 49, 289–297. [Google Scholar] [CrossRef] [PubMed]
- Bo, S.; Ciccone, G.; Baldi, C.; Benini, L.; Dusio, F.; Forastiere, G.; Lucia, C.; Nuti, C.; Durazzo, M.; Cassader, M.; et al. Effectiveness of a lifestyle intervention on metabolic syndrome. A randomized controlled trial. J. Gen. Intern. Med. 2007, 22, 1695–1703. [Google Scholar] [CrossRef] [PubMed]
- Bo, S.; Gambino, R.; Ciccone, G.; Rosato, R.; Milanesio, N.; Villois, P.; Pagano, G.; Cassader, M.; Gentile, L.; Durazzo, M.; et al. Effects of TCF7L2 polymorphisms on glucose values after a lifestyle intervention. Am. J. Clin. Nutr. 2009, 90, 1502–1508. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Penn, L.; White, M.; Oldroyd, J.; Walker, M.; Alberti, K.G.; Mathers, J.C. Prevention of type 2 diabetes in adults with impaired glucose tolerance: The European Diabetes Prevention RCT in Newcastle upon Tyne, UK. BMC Public Health 2009, 9, 342. [Google Scholar] [CrossRef]
- Eriksson, K.F.; Lindgarde, F. Prevention of type 2 (non-insulin-dependent) diabetes mellitus by diet and physical exercise. The 6-year Malmo feasibility study. Diabetologia 1991, 34, 891–898. [Google Scholar] [CrossRef]
- Penn, L.; White, M.; Lindstrom, J.; den Boer, A.T.; Blaak, E.; Eriksson, J.G.; Feskens, E.; Ilanne-Parikka, P.; Keinanen-Kiukaanniemi, S.M.; Walker, M.; et al. Importance of weight loss maintenance and risk prediction in the prevention of type 2 diabetes: Analysis of European Diabetes Prevention Study RCT. PLoS ONE 2013, 8, e57143. [Google Scholar] [CrossRef]
- Salas-Salvado, J.; Bullo, M.; Babio, N.; Martinez-Gonzalez, M.A.; Ibarrola-Jurado, N.; Basora, J.; Estruch, R.; Covas, M.I.; Corella, D.; Aros, F.; et al. Reduction in the incidence of type 2 diabetes with the Mediterranean diet: Results of the PREDIMED-Reus nutrition intervention randomized trial. Diabetes Care 2011, 34, 14–19. [Google Scholar] [CrossRef]
- Salas-Salvado, J.; Bullo, M.; Estruch, R.; Ros, E.; Covas, M.I.; Ibarrola-Jurado, N.; Corella, D.; Aros, F.; Gomez-Gracia, E.; Ruiz-Gutierrez, V.; et al. Prevention of diabetes with Mediterranean diets: A subgroup analysis of a randomized trial. Ann. Intern. Med. 2014, 160, 1–10. [Google Scholar] [CrossRef]
- Rücker, G.; Schwarzer, G.; Carpenter, J.R.; Schumacher, M. Undue reliance on I(2) in assessing heterogeneity may mislead. BMC Med. Res. Methodol. 2008, 8, 79. [Google Scholar] [CrossRef]
- Li, G.; Zhang, P.; Wang, J.; Gregg, E.W.; Yang, W.; Gong, Q.; Li, H.; Li, H.; Jiang, Y.; An, Y.; et al. The long-term effect of lifestyle interventions to prevent diabetes in the China Da Qing Diabetes Prevention Study: A 20-year follow-up study. Lancet 2008, 371, 1783–1789. [Google Scholar] [CrossRef]
- Lindstrom, J.; Peltonen, M.; Eriksson, J.G.; Ilanne-Parikka, P.; Aunola, S.; Keinanen-Kiukaanniemi, S.; Uusitupa, M.; Tuomilehto, J. Improved lifestyle and decreased diabetes risk over 13 years: Long-term follow-up of the randomised Finnish Diabetes Prevention Study (DPS). Diabetologia 2013, 56, 284–293. [Google Scholar] [CrossRef] [PubMed]
- Lindstrom, J.; Ilanne-Parikka, P.; Peltonen, M.; Aunola, S.; Eriksson, J.G.; Hemio, K.; Hamalainen, H.; Harkonen, P.; Keinanen-Kiukaanniemi, S.; Laakso, M.; et al. Sustained reduction in the incidence of type 2 diabetes by lifestyle intervention: Follow-up of the Finnish Diabetes Prevention Study. Lancet 2006, 368, 1673–1679. [Google Scholar] [CrossRef]
- Diabetes Prevention Program Research Group. 10-year follow-up of diabetes incidence and weight loss in the Diabetes Prevention Program Outcomes Study. Lancet 2009, 374, 1677–1686. [Google Scholar] [CrossRef] [Green Version]
- Nathan, D.M. Long-term complications of diabetes mellitus. N. Engl. J. Med. 1993, 328, 1676–1685. [Google Scholar] [CrossRef]
- de Mello, V.D.; Lindström, J.; Eriksson, J.; Ilanne-Parikka, P.; Keinänen-Kiukaanniemi, S.; Sundvall, J.; Laakso, M.; Tuomilehto, J.; Uusitupa, M. Insulin secretion and its determinants in the progression of impaired glucose tolerance to type 2 diabetes in impaired glucose-tolerant individuals: The Finnish Diabetes Prevention Study. Diabetes Care 2012, 35, 211–217. [Google Scholar] [CrossRef]
- Salas-Salvado, J.; Diaz-Lopez, A.; Ruiz-Canela, M.; Basora, J.; Fito, M.; Corella, D.; Serra-Majem, L.; Warnberg, J.; Romaguera, D.; Estruch, R.; et al. Effect of a Lifestyle Intervention Program with Energy-Restricted Mediterranean Diet and Exercise on Weight Loss and Cardiovascular Risk Factors: One-Year Results of the PREDIMED-Plus Trial. Diabetes Care 2019, 42, 777–788. [Google Scholar] [CrossRef]
- Uusitupa, M.; Lindstrom, J.; Tuomilehto, J. Prevention of type 2 diabetes-success story that is waiting for next steps. Eur. J. Clin. Nutr. 2018, 72, 1260–1266. [Google Scholar] [CrossRef]
- Gong, Q.; Gregg, E.W.; Wang, J.; An, Y.; Zhang, P.; Yang, W.; Li, H.; Li, H.; Jiang, Y.; Shuai, Y.; et al. Long-term effects of a randomised trial of a 6-year lifestyle intervention in impaired glucose tolerance on diabetes-related microvascular complications: The China Da Qing Diabetes Prevention Outcome Study. Diabetologia 2011, 54, 300–307. [Google Scholar] [CrossRef]
- Diabetes Prevention Program Research Group. Long-term effects of lifestyle intervention or metformin on diabetes development and microvascular complications over 15-year follow-up: The Diabetes Prevention Program Outcomes Study. Lancet Diabetes Endocrinol. 2015, 3, 866–875. [Google Scholar] [CrossRef]
- Uusitupa, M.; Peltonen, M.; Lindstrom, J.; Aunola, S.; Ilanne-Parikka, P.; Keinanen-Kiukaanniemi, S.; Valle, T.T.; Eriksson, J.G.; Tuomilehto, J. Ten-year mortality and cardiovascular morbidity in the Finnish Diabetes Prevention Study—Secondary analysis of the randomized trial. PLoS ONE 2009, 4, e5656. [Google Scholar] [CrossRef] [PubMed]
- Aro, A.; Kauppinen, A.; Kivinen, N.; Selander, T.; Kinnunen, K.; Tuomilehto, J.; Keinanen-Kiukaanniemi, S.; Lindstrom, J.; Uusitupa, M.; Kaarniranta, K. Life Style Intervention Improves Retinopathy Status-The Finnish Diabetes Prevention Study. Nutrients 2019, 11, 1691. [Google Scholar] [CrossRef] [PubMed]
- Estruch, R.; Ros, E.; Salas-Salvado, J.; Covas, M.I.; Corella, D.; Aros, F.; Gomez-Gracia, E.; Ruiz-Gutierrez, V.; Fiol, M.; Lapetra, J.; et al. Primary Prevention of Cardiovascular Disease with a Mediterranean Diet Supplemented with Extra-Virgin Olive Oil or Nuts. N. Engl. J. Med. 2018, 378, e34. [Google Scholar] [CrossRef] [PubMed]
- Diaz-Lopez, A.; Babio, N.; Martinez-Gonzalez, M.A.; Corella, D.; Amor, A.J.; Fito, M.; Estruch, R.; Aros, F.; Gomez-Gracia, E.; Fiol, M.; et al. Mediterranean Diet, Retinopathy, Nephropathy, and Microvascular Diabetes Complications: A Post Hoc Analysis of a Randomized Trial. Diabetes Care 2015, 38, 2134–2141. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Look Ahead Research Group; Gregg, E.W.; Jakicic, J.M.; Blackburn, G.; Bloomquist, P.; Bray, G.A.; Clark, J.M.; Coday, M.; Curtis, J.M.; Egan, C.; et al. Association of the magnitude of weight loss and changes in physical fitness with long-term cardiovascular disease outcomes in overweight or obese people with type 2 diabetes: A post-hoc analysis of the Look AHEAD randomised clinical trial. Lancet Diabetes Endocrinol. 2016, 4, 913–921. [Google Scholar] [CrossRef] [PubMed]
- Becerra-Tomas, N.; Blanco Mejia, S.; Viguiliouk, E.; Khan, T.; Kendall, C.W.C.; Kahleova, H.; Rahelic, D.; Sievenpiper, J.L.; Salas-Salvado, J. Mediterranean diet, cardiovascular disease and mortality in diabetes: A systematic review and meta-analysis of prospective cohort studies and randomized clinical trials. Crit. Rev. Food Sci. Nutr. 2019. [Google Scholar] [CrossRef] [PubMed]
- Nathan, D.M.; Bennett, P.H.; Crandall, J.P.; Edelstein, S.L.; Goldberg, R.B.; Kahn, S.E.; Knowler, W.C.; Mather, K.J.; Mudaliar, S.; Orchard, T.J.; et al. Does diabetes prevention translate into reduced long-term vascular complications of diabetes? Diabetologia 2019, 62, 1319–1328. [Google Scholar] [CrossRef] [Green Version]
- Aschner, P. New IDF clinical practice recommendations for managing type 2 diabetes in primary care. Diabetes Res. Clin. Pract. 2017, 132, 169–170. [Google Scholar] [CrossRef]
- Uusitupa, M.; Lindi, V.; Louheranta, A.; Salopuro, T.; Lindstrom, J.; Tuomilehto, J. Long-term improvement in insulin sensitivity by changing lifestyles of people with impaired glucose tolerance: 4-year results from the Finnish Diabetes Prevention Study. Diabetes 2003, 52, 2532–2538. [Google Scholar] [CrossRef]
- Kitabchi, A.E.; Temprosa, M.; Knowler, W.C.; Kahn, S.E.; Fowler, S.E.; Haffner, S.M.; Andres, R.; Saudek, C.; Edelstein, S.L.; Arakaki, R.; et al. Role of insulin secretion and sensitivity in the evolution of type 2 diabetes in the diabetes prevention program: effects of lifestyle intervention and metformin. Diabetes 2005, 54, 2404–2414. [Google Scholar] [CrossRef]
- Hamman, R.F.; Horton, E.; Barrett-Connor, E.; Bray, G.A.; Christophi, C.A.; Crandall, J.; Florez, J.C.; Fowler, S.; Goldberg, R.; Kahn, S.E.; et al. Factors affecting the decline in incidence of diabetes in the Diabetes Prevention Program Outcomes Study (DPPOS). Diabetes 2015, 64, 989–998. [Google Scholar] [CrossRef] [PubMed]
- Lean, M.E.; Leslie, W.S.; Barnes, A.C.; Brosnahan, N.; Thom, G.; McCombie, L.; Peters, C.; Zhyzhneuskaya, S.; Al-Mrabeh, A.; Hollingsworth, K.G.; et al. Primary care-led weight management for remission of type 2 diabetes (DiRECT): An open-label, cluster-randomised trial. Lancet 2018, 391, 541–551. [Google Scholar] [CrossRef]
- Ley, S.H.; Hamdy, O.; Mohan, V.; Hu, F.B. Prevention and management of type 2 diabetes: Dietary components and nutritional strategies. Lancet 2014, 383, 1999–2007. [Google Scholar] [CrossRef]
- Muraki, I.; Imamura, F.; Manson, J.E.; Hu, F.B.; Willett, W.C.; van Dam, R.M.; Sun, Q. Fruit consumption and risk of type 2 diabetes: Results from three prospective longitudinal cohort studies. BMJ 2013, 347, f5001. [Google Scholar] [CrossRef]
- Salas-Salvado, J.; Guasch-Ferre, M.; Diaz-Lopez, A.; Babio, N. Yogurt and Diabetes: Overview of Recent Observational Studies. J. Nutr. 2017, 147, 1452–1461. [Google Scholar] [CrossRef]
- Meyer, K.A.; Kushi, L.H.; Jacobs, D.R.; Slavin, J.; Sellers, T.A.; Folsom, A.R. Carbohydrates, dietary fiber, and incident type 2 diabetes in older women. Am. J. Clin. Nutr. 2000, 71, 921–930. [Google Scholar] [CrossRef] [Green Version]
- Hodge, A.M.; English, D.R.; O’Dea, K.; Giles, G.G. Glycemic index and dietary fiber and the risk of type 2 diabetes. Diabetes Care 2004, 27, 2701–2706. [Google Scholar] [CrossRef]
- Bazzano, L.A.; Li, T.Y.; Joshipura, K.J.; Hu, F.B. Intake of fruit, vegetables, and fruit juices and risk of diabetes in women. Diabetes care 2008, 31, 1311–1317. [Google Scholar] [CrossRef]
- Villegas, R.; Gao, Y.T.; Yang, G.; Li, H.L.; Elasy, T.A.; Zheng, W.; Shu, X.O. Legume and soy food intake and the incidence of type 2 diabetes in the Shanghai Women’s Health Study. Am. J. Clin. Nutr. 2008, 87, 162–167. [Google Scholar] [CrossRef]
- Ericson, U.; Sonestedt, E.; Gullberg, B.; Hellstrand, S.; Hindy, G.; Wirfalt, E.; Orho-Melander, M. High intakes of protein and processed meat associate with increased incidence of type 2 diabetes. Br. J. Nutr. 2013, 109, 1143–1153. [Google Scholar] [CrossRef]
- von Ruesten, A.; Feller, S.; Bergmann, M.M.; Boeing, H. Diet and risk of chronic diseases: Results from the first 8 years of follow-up in the EPIC-Potsdam study. Eur. J. Clin. Nutr. 2013, 67, 412–419. [Google Scholar] [CrossRef] [PubMed]
- Becerra-Tomas, N.; Diaz-Lopez, A.; Rosique-Esteban, N.; Ros, E.; Buil-Cosiales, P.; Corella, D.; Estruch, R.; Fito, M.; Serra-Majem, L.; Aros, F.; et al. Legume consumption is inversely associated with type 2 diabetes incidence in adults: A prospective assessment from the PREDIMED study. Clin. Nutr. 2018, 37, 906–913. [Google Scholar] [CrossRef] [PubMed]
- Karamanos, B.; Thanopoulou, A.; Anastasiou, E.; Assaad-Khalil, S.; Albache, N.; Bachaoui, M.; Slama, C.B.; El Ghomari, H.; Jotic, A.; Lalic, N.; et al. Relation of the Mediterranean diet with the incidence of gestational diabetes. Eur. J. Clin. Nutr. 2014, 68, 8–13. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, A.; Mann, J.; Cummings, J.; Winter, N.; Mete, E.; Te Morenga, L. Carbohydrate quality and human health: A series of systematic reviews and meta-analyses. Lancet 2019, 393, 434–445. [Google Scholar] [CrossRef]
- Fung, T.T.; Schulze, M.; Manson, J.E.; Willett, W.C.; Hu, F.B. Dietary patterns, meat intake, and the risk of type 2 diabetes in women. Arch. Intern. Med. 2004, 164, 2235–2240. [Google Scholar] [CrossRef]
- Salas-Salvado, J.; Guasch-Ferre, M.; Lee, C.H.; Estruch, R.; Clish, C.B.; Ros, E. Protective Effects of the Mediterranean Diet on Type 2 Diabetes and Metabolic Syndrome. J. Nutr. 2016, 146, 920–927. [Google Scholar] [CrossRef]
- Jannasch, F.; Kroger, J.; Schulze, M.B. Dietary Patterns and Type 2 Diabetes: A Systematic Literature Review and Meta-Analysis of Prospective Studies. J. Nutr. 2017, 147, 1174–1182. [Google Scholar] [CrossRef] [Green Version]
- Tonstad, S.; Stewart, K.; Oda, K.; Batech, M.; Herring, R.P.; Fraser, G.E. Vegetarian diets and incidence of diabetes in the Adventist Health Study-2. Nutr. Metab. Cardiovasc. Dis. 2013, 23, 292–299. [Google Scholar] [CrossRef]
- Snowdon, D.A.; Phillips, R.L. Does a vegetarian diet reduce the occurrence of diabetes? Am. J. Public Health 1985, 75, 507–512. [Google Scholar] [CrossRef] [Green Version]
- Chiu, T.H.T.; Pan, W.H.; Lin, M.N.; Lin, C.L. Vegetarian diet, change in dietary patterns, and diabetes risk: A prospective study. Nutr. Diabetes 2018, 8, 12. [Google Scholar] [CrossRef]
- Zhang, C.; Tobias, D.K.; Chavarro, J.E.; Bao, W.; Wang, D.; Ley, S.H.; Hu, F.B. Adherence to healthy lifestyle and risk of gestational diabetes mellitus: Prospective cohort study. BMJ 2014, 349, g5450. [Google Scholar] [CrossRef] [PubMed]
- Mekary, R.A.; Giovannucci, E.; Willett, W.C.; van Dam, R.M.; Hu, F.B. Eating patterns and type 2 diabetes risk in men: Breakfast omission, eating frequency, and snacking. Am. J. Clin. Nutr. 2012, 95, 1182–1189. [Google Scholar] [CrossRef] [PubMed]
- Mekary, R.A.; Giovannucci, E.; Cahill, L.; Willett, W.C.; van Dam, R.M.; Hu, F.B. Eating patterns and type 2 diabetes risk in older women: Breakfast consumption and eating frequency. Am. J. Clin. Nutr. 2013, 98, 436–443. [Google Scholar] [CrossRef]
- Ballon, A.; Neuenschwander, M.; Schlesinger, S. Breakfast Skipping Is Associated with Increased Risk of Type 2 Diabetes among Adults: A Systematic Review and Meta-Analysis of Prospective Cohort Studies. J. Nutr. 2019, 149, 106–113. [Google Scholar] [CrossRef] [PubMed]
- Lindstrom, J.; Peltonen, M.; Eriksson, J.G.; Louheranta, A.; Fogelholm, M.; Uusitupa, M.; Tuomilehto, J. High-fibre, low-fat diet predicts long-term weight loss and decreased type 2 diabetes risk: The Finnish Diabetes Prevention Study. Diabetologia 2006, 49, 912–920. [Google Scholar] [CrossRef]
- Sylvetsky, A.C.; Edelstein, S.L.; Walford, G.; Boyko, E.J.; Horton, E.S.; Ibebuogu, U.N.; Knowler, W.C.; Montez, M.G.; Temprosa, M.; Hoskin, M.; et al. A High-Carbohydrate, High-Fiber, Low-Fat Diet Results in Weight Loss among Adults at High Risk of Type 2 Diabetes. J. Nutr. 2017, 147, 2060–2066. [Google Scholar] [CrossRef]
- Wu, J.H.Y.; Marklund, M.; Imamura, F.; Tintle, N.; Ardisson Korat, A.V.; de Goede, J.; Zhou, X.; Yang, W.S.; de Oliveira Otto, M.C.; Kröger, J.; et al. Omega-6 fatty acid biomarkers and incident type 2 diabetes: Pooled analysis of individual-level data for 39 740 adults from 20 prospective cohort studies. Lancet Diabetes Endocrinol. 2017, 5, 965–974. [Google Scholar] [CrossRef]
- Schwab, U.; Lauritzen, L.; Tholstrup, T.; Haldorssoni, T.; Riserus, U.; Uusitupa, M.; Becker, W. Effect of the amount and type of dietary fat on cardiometabolic risk factors and risk of developing type 2 diabetes, cardiovascular diseases, and cancer: A systematic review. Food Nutr. Res. 2014, 58, 25145. [Google Scholar] [CrossRef]
- Laaksonen, D.E.; Lindstrom, J.; Lakka, T.A.; Eriksson, J.G.; Niskanen, L.; Wikstrom, K.; Aunola, S.; Keinanen-Kiukaanniemi, S.; Laakso, M.; Valle, T.T.; et al. Physical activity in the prevention of type 2 diabetes: The Finnish diabetes prevention study. Diabetes 2005, 54, 158–165. [Google Scholar] [CrossRef]
- Aune, D.; Norat, T.; Leitzmann, M.; Tonstad, S.; Vatten, L.J. Physical activity and the risk of type 2 diabetes: A systematic review and dose-response meta-analysis. Eur. J. Epidemiol. 2015, 30, 529–542. [Google Scholar] [CrossRef]
- Burr, J.F.; Rowan, C.P.; Jamnik, V.K.; Riddell, M.C. The role of physical activity in type 2 diabetes prevention: Physiological and practical perspectives. Phys. Sportsmed. 2010, 38, 72–82. [Google Scholar] [CrossRef] [PubMed]

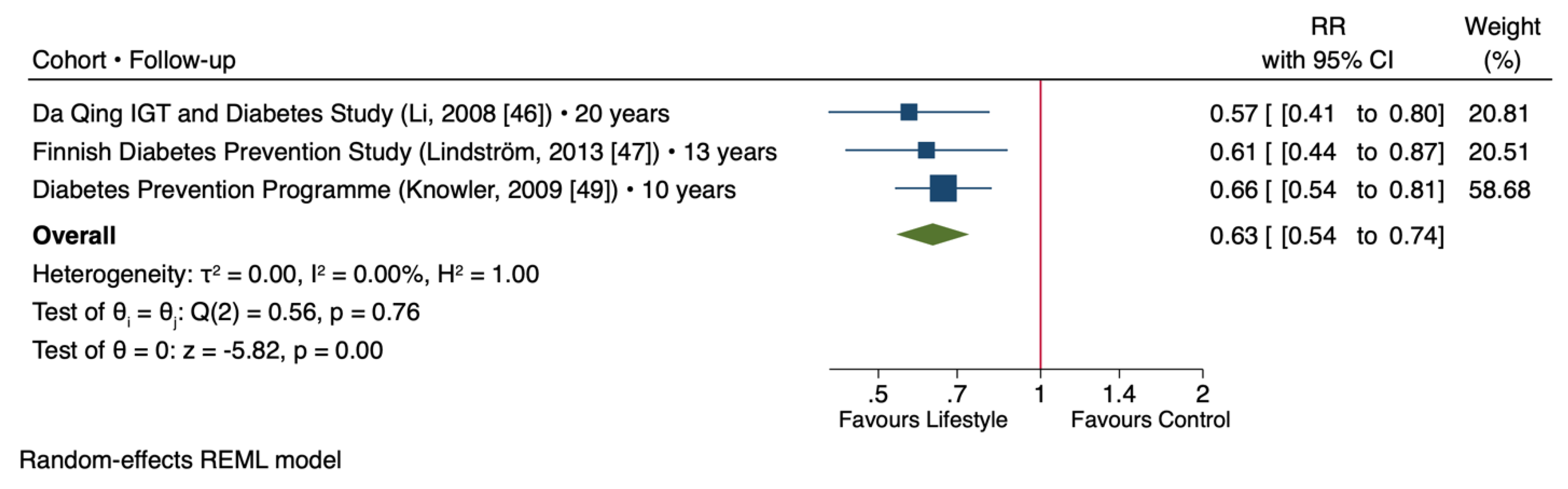
| Study | Country | N, Characteristics | Study Duration | Risk Reduction of T2D with Lifestyle versus Control | Dietary Goals | Changes in Diet When Available | Physical Activity, Goals/Changes | Comment |
|---|---|---|---|---|---|---|---|---|
| Da Qing IGT and Diabetes Study, Pan XR et al. Diabetes Care 1997 [32] | China | In total, 577; all had IGT; 33 health care clinics | 6 yrs | Diet 33%; exercise 47%; diet + exercise 38% | Weight reduction in overweight; calorie restriction | CHO 58–60 E%; protein 11 E%; fat 25–27 E%; total calories decrease 100–240 kcal | Increase, e.g., walking | Randomization by clinic; follow-up data available |
| FDPS, Tuomilehto J et al. N Engl J Med 2001 [33] | Finland | In total, 522; IGT; five centers | 3.2 yrs; median 4 yrs | In total, 58%, weight loss; difference 3.5 and 2.6 kg after 1 and 3 yrs, respectively. | Weight reduction >5%; reduce total and SFA; increase dietary fiber | 3 yr results: energy reduction 204 kcal; CHO increase 3 E%; fat reduction 5 E%; SFA reduction 3 E%; fiber increase 2 g/1000 kcal | 4 h/wk, sedentary people at yr 3: 17% vs. 29% for intervention and control groups, respectively | Individual dietary data and long-term follow-up data available |
| DPP, Knowler WC et al. New Engl J Med 2002 [34] | USA | In total, 3234; IGT; 27 centers | 2.8 yrs | Lifestyle 58%; Metformin 31%; weight loss at yr 1: −5.6 vs. −0.1 kg for intervention vs. control, respectively. | NCEP Step 1; weight loss goal 7% | Energy intake reduction 450 vs. 249 kcal and fat intake reduction 6.6 vs. 0.8 E% for intervention and control, respectively. | 150 min/wk | Follow-up data available |
| Japanese trial in IGT males, Kosaka K et al. Diabetes Res Clin Pract 2005 [36] | Japan | In total, 458 IGT; 356 in control, 102 in intervention, OGTT (100 g glucose dose) | 4 yrs | Incidence of T2D 3.0% vs. 9.3%; risk reduction 67.4%; weight loss −2.18 kg | BMI goal 22 kg/m2; increase vegetables; reduce food intake by 10%; fat < 50 g/d; alcohol restriction | Not reported | 30–40 min walking/d | Normal and overweight men |
| IDPP-1, Ramachandran A et al. Diabetologia 2006 [37] | India | In total, 531; IGT; lifestyle 133; metformin 133; lifestyle-plus-metformin 129; control 136 | 30 months | Lifestyle 28.5%; Metformin 26.4%; lifestyle-plus-Metformin 28.2%; no change in body weight | Reduce total calories, refined CHO, fat and sugar; increase high fiber-rich foods | Dietary adherence increased in Intervention groups | Walking 30 min a day | |
| Lifestyle intervention on metabolic syndrome. Bo S, J Gen Intern Med 2007 [38], Bo S et al. Am J Clin Nutr 2009 [39] | Italy | In total, 375 with dysmetabolism; 169 intervention; 166 control; focus on metabolic syndrome | 1 yr, | Risk reduction for T2D 77%, (OR 0.23; 95% CI 0.06–0.85) at year 1. | General recommendations for lose weight and decrease SFA and increase PUFA and fiber | Body weight minus 0.75 vs. plus 1.63 kg; total calories minus 74.6 vs. 43.7 kcal; fat minus 2.64 E%; SFA minus 1.97 E%; CHO 2.14 E%; prot 1.7 E%; NS for control | Increase | 4 yrs diabetes incidence 5.4% vs. 10.2% in intervention and control groups, respectively |
| EDIPS-Newcastle, Penn L. BMC Public Health 2009 [40] | UK | In total, 102; IGT; 51 in intervention and control, respectively | 3 yrs | Diabetes incidence 5% vs. 11, 1% yr. body weight change −2.5 kg | Like in FDPS, decrease fat and SFA; increase fiber; body weight reduction | Not reported | Like in FDPS | Sustained beneficial changes in lifestyles predicted better outcome |
| Author (Removed) | Risk Ratio (RR) with 95% CI | P-Effect | I2 (%) | P-Heterogeneity |
|---|---|---|---|---|
| Overall | 0.53 [0.41, 0.67] | <0.001 | 63 | 0.01 |
| Da Qing IGT And Diabetes Study (Pan, 1997 [32]) | 0.53 [0.41, 0.67] | <0.001 | 55 | 0.052 |
| Diabetes Prevention Programme (Knowler, 2002 [34]) | 0.49 [0.37, 0.64] | <0.001 | 43 | 0.163 |
| European Diabetes Prevention RCT—Newcastle (Penn, 2009 [40]) | 0.57 [0.44, 0.74] | <0.001 | 69 | 0.005 |
| Finnish Diabetes Prevention Study (Tuomilehto, 2001 [33]) | 0.53 [0.41, 0.68] | <0.001 | 67 | 0.006 |
| Indian Diabetes Prevention Programme (Ramachandran, 2006 [37]) | 0.54 [0.41, 0.72] | <0.001 | 57 | 0.038 |
| Japanese Trial in IGT Males (Kosaka, 2005 [36]) | 0.48 [0.37, 0.63] | <0.001 | 67 | 0.006 |
| Lifestyle Intervention on Metabolic Syndrome (Bo, 2007 [38,39]) | 0.54 [0.42, 0.69] | <0.001 | 66 | 0.008 |
| Outcome | No. of Studies | Study Design | Certainty Assessment | RR [95% CI] | Certainty | ||||
|---|---|---|---|---|---|---|---|---|---|
| Risk of Bias | Inconsistency | Indirectness | Imprecision | Other Considerations | |||||
| T2D risk reduction | Seven | randomized trials | not serious | not serious a | not serious | not serious | none | 0.53 [0.41, 0.67] | ⨁⨁⨁⨁ HIGH |
| Original Study | Risk Reduction | Comment |
|---|---|---|
| FDPS, Lindström J et al. Diabetologia 2013 [47] | Hazard Ratio 0.61, adjusted to 0.59 as compared to control group | Follow-up 13 years; follow-up data on the diet available |
| China Da Qing Diabetes Prevention Study, Li G et al. Lancet 2008 [46] | In total, 43% reduction in the combined intervention clinics as compared to control clinic | Follow-up 20 years; no detailed dietary data |
| Diabetes Prevention Program Group, Knowler WC et al. Lancet 2009 [49] | In total, 34% reduction in lifestyle intervention group and 18% reduction in metformin group as compared to placebo control group | Follow-up 10 year; no dietary data from the follow-up reported; long-term metformin use may modify the results |
| Original Study | Mortality | Cardiovascular Mortality | Reported Microvascular Complications |
|---|---|---|---|
| China Da Qing Diabetes Prevention Follow-up Study, Lancet Diabetes and Endocrinol, Gong Q et al., 2019 [54] | In total, 26% reduction in combined intervention clinics compared to original control group | In total, 33% reduction in combined intervention clinics compared to original control group | In total, 35% reduction in composite microvascular diseases and 40% reduction in any retinopathy in combined intervention clinics compared to original control group [54] |
| Diabetes Prevention Program Group, Lancet Diabetes and Endocrinol, Nathan DM et al., 2015 [55] | NA | NA | No group differences. Less microvascular complications in individuals who remained non-diabetic (RR 0.72, p < 0.001), less microvascular complications in intervention women (8.7% vs. control 11.0% or metformin groups, 11.2%, p = 0.03) |
| The Finnish Diabetes Prevention Follow-up Study PLoS One, Uusitupa M et al., 2009 [56] Nutrients, Aro A et al., 2019 [57] | NS between the original intervention and control groups | NS between the original intervention and control groups | Less early retinopathic changes in intervention (24% vs. 38%, adjusted odds ratio 0.52; 0.28–0.97, 95% CI, p = 0.039) than in control group; a subgroup analysis based on retinal photographs. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Uusitupa, M.; Khan, T.A.; Viguiliouk, E.; Kahleova, H.; Rivellese, A.A.; Hermansen, K.; Pfeiffer, A.; Thanopoulou, A.; Salas-Salvadó, J.; Schwab, U.; et al. Prevention of Type 2 Diabetes by Lifestyle Changes: A Systematic Review and Meta-Analysis. Nutrients 2019, 11, 2611. https://doi.org/10.3390/nu11112611
Uusitupa M, Khan TA, Viguiliouk E, Kahleova H, Rivellese AA, Hermansen K, Pfeiffer A, Thanopoulou A, Salas-Salvadó J, Schwab U, et al. Prevention of Type 2 Diabetes by Lifestyle Changes: A Systematic Review and Meta-Analysis. Nutrients. 2019; 11(11):2611. https://doi.org/10.3390/nu11112611
Chicago/Turabian StyleUusitupa, Matti, Tauseef A. Khan, Effie Viguiliouk, Hana Kahleova, Angela A Rivellese, Kjeld Hermansen, Andreas Pfeiffer, Anastasia Thanopoulou, Jordi Salas-Salvadó, Ursula Schwab, and et al. 2019. "Prevention of Type 2 Diabetes by Lifestyle Changes: A Systematic Review and Meta-Analysis" Nutrients 11, no. 11: 2611. https://doi.org/10.3390/nu11112611
APA StyleUusitupa, M., Khan, T. A., Viguiliouk, E., Kahleova, H., Rivellese, A. A., Hermansen, K., Pfeiffer, A., Thanopoulou, A., Salas-Salvadó, J., Schwab, U., & Sievenpiper, J. L. (2019). Prevention of Type 2 Diabetes by Lifestyle Changes: A Systematic Review and Meta-Analysis. Nutrients, 11(11), 2611. https://doi.org/10.3390/nu11112611








