Fatty Acid Profile and Antioxidant Status Fingerprint in Sarcopenic Elderly Patients: Role of Diet and Exercise
Abstract
:1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Dietetic Program
2.3. Physical Activity
- -
- Chair exercise: repetitions of toe raises, heel raises, knee lifts, knee extensions, and others were done while seated on a chair. Hip flexions, lateral leg raises, and repetitions of other exercises were done while standing upright behind the chair, holding the back of the chair for stability.
- -
- Ankle-weight exercise: to strengthen the legs, a fixed weight was placed on the ankle while participants did strengthening exercises. Weights of 0.50, 0.75, 1.00, and 1.50 kg were prepared and used in accordance with each participant’s strength as the resistance progressively increased. The exercises using these ankle weights included seated knee flexion and extension as well as standing knee flexion and extensions.
- -
- Exercises with a resistance band: resistance bands were used to strengthen the upper and lower body. Lower-body exercises included leg extension and hip flexion. Upper-body exercises included double-arm pull downs and bicep curls.
- -
- Balance and gait training: exercises included standing on one leg, multidirectional weight shifts, tandem stand, and tandem walk. Participants practiced proper gait mechanics focused on maintaining stability during walking and increasing stride length, toe elevation of the forward limb, heel elevation of the rear limb, frequency of stepping, and a heel-floor angle. Exercises included raising the toes (dorsiflexion) during the forward swing of the leg, kicking off the floor with the ball of the foot, walking with directional changes, and gait pattern variations.
2.4. Dietary Supplementation
2.5. Blood Sampling
2.6. Lipid Analysis
2.7. Plasma Malondialdehyde (MDA) Analysis and Erythrocyte Antioxidant Enzymes
2.8. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Cruz-Jentoft, A.J.; Baeyens, J.P.; Bauer, J.M.; Boirie, Y.; Cederholm, T.; Landi, F.; Martin, F.C.; Michel, J.P.; Rolland, Y.; Schneider, S.M.; et al. Sarcopenia: European consensus on definition and diagnosis: Report of the European Working Group on Sarcopenia in Older People. Age Ageing 2010, 39, 412–423. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cooper, C.; Dere, W.; Evans, W.; Kanis, J.A.; Rizzoli, R.; Sayer, A.A.; Sieber, C.C.; Kaufman, J.M.; Abellan van Kan, G.; Boonen, S.; et al. Frailty and sarcopenia: Definitions and outcome parameters. Osteoporos Int. 2012, 23, 1839–1848. [Google Scholar] [CrossRef] [PubMed]
- Harraz, M.M.; Marden, J.J.; Zhou, W.; Zhang, Y.; Williams, A.; Sharov, V.S.; Nelson, K.; Luo, M.; Paulson, H.; Schöneich, C.; et al. SOD1 mutations disrupt redox-sensitive Rac regulation of NADPH oxidase in a familial ALS model. J. Clin. Investig. 2008, 118, 659–670. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jang, Y.C.; Liu, Y.; Hayworth, C.R.; Bhattacharya, A.; Lustgarten, M.S.; Muller, F.L.; Chaudhuri, A.; Qi, W.; Li, Y.; Huang, J.Y.; et al. Dietary restriction attenuates age-associated muscle atrophy by lowering oxidative stress in mice even in complete absence of CuZnSOD. Aging Cell 2012, 11, 770–782. [Google Scholar] [CrossRef] [Green Version]
- Wei, J.; Zhai, L.; Xu, J.; Wang, H. Role of Bmi1 in H2A ubiquitylation and Hox gene silencing. J. Biol. Chem. 2006, 281, 22537–22544. [Google Scholar] [CrossRef]
- Fulle, S.; Protasi, F.; Di Tano, G.; Pietrangelo, T.; Beltramin, A.; Boncompagni, S.; Vecchiet, L.; Fanò, G. The contribution of reactive oxygen species to sarcopenia and muscle ageing. Exp. Gerontol. 2004, 39, 17–24. [Google Scholar] [CrossRef]
- Ji, L.L. Antioxidant signaling in skeletal muscle: A brief review. Exp. Gerontol. 2007, 42, 582–593. [Google Scholar] [CrossRef]
- Pansarasa, O.; Bertorelli, L.; Vecchiet, J.; Felzani, G.; Marzatico, F. Age-dependent changes of antioxidant activities and markers of free radical damage in human skeletal muscle. Free Radic. Biol. Med. 1999, 27, 617–622. [Google Scholar] [CrossRef]
- Wei, Y.H.; Lee, H.C. Oxidative stress, mitochondrial DNA mutation, and impairment of antioxidant enzymes in aging. Exp. Biol. Med. 2002, 227, 671–682. [Google Scholar] [CrossRef]
- Sentman, M.L.; Granström, M.; Jakobson, H.; Reaume, A.; Basu, S.; Marklund, S.L. Phenotypes of mice lacking extracellular superoxide dismutase and copper- and zinc-containing superoxide dismutase. J. Biol. Chem. 2006, 281, 6904–6909. [Google Scholar] [CrossRef]
- Jang, Y.C.; Lustgarten, M.S.; Liu, Y.; Muller, F.L.; Bhattacharya, A.; Liang, H.; Salmon, A.B.; Brooks, S.V.; Larkin, L.; Hayworth, C.R.; et al. Increased superoxide in vivo accelerates age-associated muscle atrophy through mitochondrial dysfunction and neuromuscular junction degeneration. FASEB J. 2010, 24, 1376–1390. [Google Scholar] [CrossRef] [PubMed]
- Muller, F.L.; Song, W.; Liu, Y.; Chaudhuri, A.; Pieke-Dahl, S.; Strong, R.; Huang, T.T.; Epstein, C.J.; Roberts, L.J.; Csete, M.; et al. Absence of CuZn superoxide dismutase leads to elevated oxidative stress and acceleration of age-dependent skeletal muscle atrophy. Free Radic. Biol. Med. 2006, 40, 1993–2004. [Google Scholar] [CrossRef] [PubMed]
- Mecocci, P.; Fanó, G.; Fulle, S.; MacGarvey, U.; Shinobu, L.; Polidori, M.C.; Cherubini, A.; Vecchiet, J.; Senin, U.; Beal, M.F. Age-dependent increases in oxidative damage to DNA, lipids, and proteins in human skeletal muscle. Free Radic. Biol. Med. 1999, 26, 303–308. [Google Scholar] [CrossRef]
- Capel, F.; Buffière, C.; PatureauMirand, P.; Mosoni, L. Differential variation of mitochondrial H2O2 release during aging in oxidative and glycolytic muscles in rats. Mech. Ageing Dev. 2004, 125, 367–373. [Google Scholar] [CrossRef]
- Capel, F.; Rimbert, V.; Lioger, D.; Diot, A.; Rousset, P.; Mirand, P.P.; Boirie, Y.; Morio, B.; Mosoni, L. Due to reverse electron transfer, mitochondrial H2O2 release increases with age in human vastus lateralis muscle although oxidative capacity is preserved. Mech. Ageing Dev. 2005, 126, 505–511. [Google Scholar] [CrossRef]
- Demontis, F.; Piccirillo, R.; Goldberg, A.L.; Perrimon, N. Mechanisms of skeletal muscle aging: Insights from Drosophila and mammalian models. Dis. Model. Mech. 2013, 6, 1339–1352. [Google Scholar] [CrossRef]
- Martin, C.; Dubouchaud, H.; Mosoni, L.; Chardigny, J.M.; Oudot, A.; Fontaine, E.; Vergely, C.; Keriel, C.; Rochette, L.; Leverve, X.; et al. Abnormalities of mitochondrial functioning can partly explain the metabolic disorders encountered in sarcopenic gastrocnemius. Aging Cell 2007, 6, 165–177. [Google Scholar] [CrossRef] [Green Version]
- Corcoran, M.P.; Lamon-Fava, S.; Fielding, R.A. Skeletal muscle lipid deposition and insulin resistance: Effect of dietary fatty acids and exercise. Am. J. Clin. Nutr. 2007, 85, 662–677. [Google Scholar]
- Pedersen, B.K. Muscle-to-fat interaction: A two-way street? J. Physiol. 2010, 588, 21. [Google Scholar] [CrossRef]
- Kien, C.L.; Everingham, K.I.; Stevens, R.D.; Fukagawa, N.K.; Muoio, D.M. Short-term effects of dietary fatty acids on muscle lipid composition and serum acylcarnitine profile in human subjects. Obesity 2011, 19, 305–311. [Google Scholar] [CrossRef]
- Lipina, C.; Hundal, H.S. Lipid modulation of skeletal muscle mass and function. J. Cachexia Sarcopenia Muscle 2017, 8, 190–201. [Google Scholar] [CrossRef] [PubMed]
- Baumgartner, R.; Koehler, K.; Gallagher, D.; Romero, L.; Heymsfield, S.B.; Ross, R.R.; Garry, P.J.; Lindeman, R.D. Epidemiology of sarcopenia among the elderly in New Mexico. Am. J. Epidemiol. 1998, 147, 755–763. [Google Scholar] [CrossRef] [PubMed]
- Folstein, M.F.; Folstein, S.E.; McHugh, P.R. “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J. Psychiatr. Res. 1975, 12, 189–198. [Google Scholar] [CrossRef]
- Rondanelli, M.; Klersy, C.; Terracol, G.; Talluri, J.; Maugeri, R.; Guido, D.; Faliva, M.A.; Solerte, B.S.; Fioravanti, M.; Lukaski, H.; et al. Whey protein, amino acids, and vitamin D supplementation with physical activity increases fat-free mass and strength, functionality, and quality of life and decreases inflammation in sarcopenic elderly. Am. J. Clin. Nutr. 2016, 103, 830–840. [Google Scholar] [CrossRef]
- Russell, R.M.; Rasmussen, H.; Lichtenstein, A.H. Modified food guide pyramid for people over seventy years age. J. Nutr. 1999, 129, 751–753. [Google Scholar] [CrossRef]
- Kim, H.K.; Suzuki, T.; Saito, K.; Yoshida, H.; Kobayashi, H.; Kato, H.; Katayama, M. Effects of Exercise and Amino Acid Supplementation on Body Composition and Physical Function in Community-Dwelling Elderly Japanese Sarcopenic Women: A Randomized Controlled Trial. J. Am. Geriatr. Soc. 2012, 60, 16–23. [Google Scholar] [CrossRef]
- American College of Sports Medicine. American College of Sports Medicine position stand. Progression models in resistance training for healthy adults. Med. Sci. Sports Exerc. 2009, 41, 687–708. [Google Scholar] [CrossRef]
- Borg, G.A. Psychophysical bases of perceived exertion. Med. Sci. Sports Exerc. 1982, 14, 377–381. [Google Scholar] [CrossRef]
- Folch, J.; Lees, M.; Sloane-Stanley, G.H. A simple method for the isolation and purification of total lipids from animal tissues. J. Biol. Chem. 1957, 226, 497–509. [Google Scholar]
- Rizzo, A.M.; Montorfano, G.; Negroni, M.; Adorni, L.; Berselli, P.; Corsetto, P.; Wahle, K.; Berra, B. A rapid method for determining arachidonic: Eicosapentaenoic acid ratios in whole blood lipids: Correlation with erythrocyte membrane ratios and validation in a large Italian population of various ages and pathologies. Lipids Health Dis. 2010, 9, 7. [Google Scholar] [CrossRef]
- Corsetto, P.A.; Cremona, A.; Montorfano, G.; Jovenitti, I.E.; Orsini, F.; Arosio, P.; Rizzo, A.M. Chemical-physical changes in cell membrane microdomains of breast cancer cells after omega-3 PUFA incorporation. Cell Biochem. 2012, 64, 45–59. [Google Scholar] [CrossRef] [PubMed]
- Aoun, M.; Corsetto, P.A.; Nugue, G.; Montorfano, G.; Ciusani, E.; Crouzier, D.; Hogarth, P.; Gregory, A.; Hayflick, S.; Zorzi, G.; et al. Changes in Red Blood Cell membrane lipid composition: A new perspective into the pathogenesis of PKAN. Mol. Genet. Metab. 2017, 121, 180–189. [Google Scholar] [CrossRef] [PubMed]
- Rizzo, A.M.; Corsetto, P.A.; Montorfano, G.; Milani, S.; Zava, S.; Tavella, S.; Cancedda, R.; Berra, B. Effects of long-term space flight on erythrocytes and oxidative stress of rodents. PLoS ONE 2012, 7, e32361. [Google Scholar] [CrossRef] [PubMed]
- Beutler, E. Red Cell Metabolism. In A Manual of Biochemical Methods, 3rd ed.; Grune and Stratton: Orlando, FL, USA, 1984. [Google Scholar]
- Griffith, O.W. Glutathione and Glutathione Disulphide. In Methods of Enzymatic Analysis; Bergmeyer, H.U., Ed.; Academic Press: New York, NY, USA, 1985; Volume 8, pp. 521–529. [Google Scholar]
- Jackson, M.J. Reactive oxygen species in sarcopenia: Should we focus on excess oxidative damage or defective redox signalling? Mol. Asp. Med. 2016, 50, 33–40. [Google Scholar] [CrossRef]
- Bouzid, M.A.; Filaire, E.; McCall, A.; Fabre, C. Radical Oxygen Species, Exercise and Aging: An Update. Sports Med. 2015, 45, 1245–1261. [Google Scholar] [CrossRef]
- Vessby, B.; Lithell, H.; Gustafsson, I.B.; Boberg, J. Changes in the fatty acid composition of the plasma lipid esters during lipid-lowering treatment with diet, clofibrate and niceritrol. Reduction of the proportion of linoleate by clofibrate but not by niceritrol. Atherosclerosis 1980, 35, 51–65. [Google Scholar] [CrossRef]
- Zock, P.L.; Mensink, R.P.; Harryvan, J.; de Vries, J.H.; Katan, M.B. Fatty acids in serum cholesteryl esters as quantitative biomarkers of dietary intake in humans. Am. J. Epidemiol. 1997, 145, 1114–1122. [Google Scholar] [CrossRef]
- Glatz, J.F.; Soffers, A.E.; Katan, M.B. Fatty acid composition of serum cholesteryl esters and erythrocyte membranes as indicators of linoleic acid intake in man. Am. J. Clin. Nutr. 1989, 49, 269–276. [Google Scholar] [CrossRef] [Green Version]
- Fenton, J.I.; Gurzell, E.A.; Davidson, E.A.; Harris, W.S. Red blood cell PUFAs reflect the phospholipid PUFA composition of major organs. Prostaglandins Leukot. Essent. Fat. Acids 2016, 112, 12–23. [Google Scholar] [CrossRef]
- Pedersen, M.H.; Mølgaard, C.; Hellgren, L.I.; Lauritzen, L. Effects of fish oil supplementation on markers of the metabolic syndrome. J. Pediatr 2010, 157, 395–400. [Google Scholar] [CrossRef]
- López-Domínguez, J.A.; Khraiwesh, H.; González-Reyes, J.A.; López-Lluch, G.; Navas, P.; Ramsey, J.J.; De Cabo, R.; Burón, M.I.; Villalba, J.M. Dietary fat modifies mitochondrial and plasma membrane apoptotic signaling in skeletal muscle of calorie-restricted mice. Age 2013, 35, 2027–2044. [Google Scholar] [CrossRef] [PubMed]
- Ji, L.L.; Leeuwenburgh, C.; Leichtweis, S.; Gore, M.; Fiebig, R.; Hollander, J.; Bejma, J. Oxidative stress and aging. Role of exercise and its influences on antioxidant systems. Ann. N. Y. Acad. Sci. 1998, 854, 102–117. [Google Scholar] [CrossRef] [PubMed]
- Crown, S.B.; Marze, N.; Antoniewicz, M.R. Catabolism of Branched Chain Amino Acids Contributes Significantly to Synthesis of Odd-Chain and Even-Chain Fatty Acids in 3T3-L1 Adipocytes. PLoS ONE 2015, 10, e0145850. [Google Scholar] [CrossRef] [PubMed]
- Frayn, K.N.; Hodson, L.; Karpe, F. Dietary fat and insulin sensitivity. Diabetologia 2010, 53, 799–801. [Google Scholar] [CrossRef]
- Frayn, K.N. Fat as a fuel: Emerging understanding of the adipose tissue-skeletal muscle axis. Acta Physiol. 2010, 199, 509–518. [Google Scholar] [CrossRef]
- Sitnick, M.; Bodine, S.C.; Rutledge, J.C. Chronic high fat feeding attenuates load-induced hypertrophy in mice. J. Physiol. 2009, 587, 5753–5765. [Google Scholar] [CrossRef]
- Welch, A.A.; MacGregor, A.J.; Minihane, A.M.; Skinner, J.; Valdes, A.A.; Spector, T.D.; Cassidy, A. Dietary fat and fatty acid profile are associated with indices of skeletal muscle mass in women aged 18–79 years. J. Nutr. 2014, 144, 327–334. [Google Scholar] [CrossRef]
- Naples, S.P.; Borengasser, S.J.; Rector, R.S.; Uptergrove, G.M.; Morris, E.M.; Mikus, C.R.; Koch, L.G.; Britton, S.L.; Ibdah, J.A.; Thyfault, J.P. Skeletal muscle mitochondrial and metabolic responses to a high-fat diet in female rats bred for high and low aerobic capacity. Appl. Physiol. Nutr. Metab. 2010, 35, 151–162. [Google Scholar] [CrossRef]
- Andersson, A.; Sjödin, A.; Olsson, R.; Vessby, B. Effects of physical exercise on phospholipid fatty acid composition in skeletal muscle. Am. J. Physiol. Endocrinol. Metab. 1998, 274, E432–E438. [Google Scholar] [CrossRef]
- Laurentius, T.; Kob, R.; Fellner, C.; Nourbakhsh, M.; Bertsch, T.; Sieber, C.C.; Bollheimer, L.C. Long-Chain Fatty Acids and Inflammatory Markers Coaccumulate in the Skeletal Muscle of Sarcopenic Old Rats. Dis. Markers 2019, 2019, 9140789. [Google Scholar] [CrossRef]
- Abreu, P.; Pinheiro, C.H.; Vitzel, K.F.; Vasconcelos, D.A.; Torres, R.P.; Fortes, M.S.; Marzuca-Nassr, G.N.; Mancini-Filho, J.; Hirabara, S.M.; Curi, R. Contractile function recovery in severely injured gastrocnemius muscle of rats treated with either oleic or linoleic acid. Exp. Physiol. 2016, 101, 1392–1405. [Google Scholar] [CrossRef] [PubMed]
- Hlais, S.; El-Bistami, D.; El Rahi, B.; Mattar, M.A.; Obeid, O.A. Combined fish oil and high oleic sunflower oil supplements neutralize their individual effects on the lipid profile of healthy men. Lipids 2013, 48, 853–861. [Google Scholar] [CrossRef] [PubMed]
- Perez-Martinez, P.; Garcia-Rios, A.; Delgado-Lista, J.; Perez-Jimenez, F.; Lopez-Miranda, J. Mediterranean diet rich in olive oil and obesity, metabolic syndrome and diabetes mellitus. Curr. Pharm. Des. 2011, 17, 769–777. [Google Scholar] [CrossRef] [PubMed]
- Kwon, B.; Lee, H.K.; Querfurth, H.W. Oleate prevents palmitate-induced mitochondrial dysfunction, insulin resistance and inflammatory signaling in neuronal cells. Biochim. Biophys. Acta 2014, 1843, 1402–1413. [Google Scholar] [CrossRef] [Green Version]
- Salvadó, L.; Coll, T.; Gomez-Foix, A.M.; Salmeron, E.; Barroso, E.; Palomer, X.; Vazquez-Carrera, M. Oleate prevents saturated-fatty-acid-induced ER stress, inflammation and insulin resistance in skeletal muscle cells through an AMPK-dependent mechanism. Diabetologia 2013, 56, 1372–1382. [Google Scholar] [CrossRef]
- Sales-Campos, H.; Souza, P.R.; Peghini, B.C.; da Silva, J.S.; Cardoso, C.R. An overview of the modulatory effects of oleic acid in health and disease. Mini Rev. Med. Chem. 2013, 13, 201–210. [Google Scholar]
- Candow, D.G.; Forbes, S.C.; Little, J.P.; Cornish, S.M.; Pinkoski, C.; Chilibeck, P.D. Effect of nutritional interventions and resistance exercise on aging muscle mass and strength. Biogerontology 2012, 13, 345–358. [Google Scholar] [CrossRef]
- Mu, L.; Mukamal, K.J.; Naqvi, A.Z. Erythrocyte saturated fatty acids and systemic inflammation in adults. Nutrition 2014, 30, 1404–1408. [Google Scholar] [CrossRef] [Green Version]
- Sergeant, S.; Rahbar, E.; Chilton, F.H. Gamma-linolenic acid, Dihommo-gamma linolenic, Eicosanoids and Inflammatory Processes. Eur. J. Pharm. 2016, 785, 77–86. [Google Scholar] [CrossRef] [Green Version]
- Cruzat, V.F.; Krause, M.; Newsholme, P. Amino acid supplementation and impact on immune function in the context of exercise. J. Int. Soc. Sports Nutr. 2014, 11, 61. [Google Scholar] [CrossRef]
- Derbré, F.; Gratas-Delamarche, A.; Gómez-Cabrera, M.C.; Viña, J. Inactivity-induced oxidative stress: A central role in age-related sarcopenia? Eur. J. Sport Sci. 2014, 14, S98–S108. [Google Scholar] [CrossRef] [PubMed]
- Thompson, L.V. Effects of age and training on skeletal-muscle physiology and performance. Phys. Ther. 1994, 74, 71–81. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, I.V.T.H.; Krajewski, K.M.; Larkin, L.M.; Reid, P.; Halter, J.B.; Supiano, M.A.; Dengel, D.R. Effect of age on skeletal muscle proteolysis in extensor digitorum longus muscles of B6C3F1 mice. J. Gerontol. A Biol. Sci. Med. Sci. 2002, 57, B198–B201. [Google Scholar] [CrossRef] [PubMed]
- Yarasheski, K.E. Exercise, aging, and muscle protein metabolism. J. Gerontol. Biol. Sci. Med. Sci. 2003, 58, M918–M922. [Google Scholar] [CrossRef] [PubMed]
- Zinna, E.M.; Yarasheski, K.E. Exercise treatment to counteract protein wasting of chronic diseases. Curr. Opin. Clin. Nutr. Metab. Care 2003, 6, 87–93. [Google Scholar] [CrossRef] [PubMed]
- Ji, L.L. Exercise-induced modulation of antioxidant defense. Ann. N. Y. Acad. Sci. 2002, 959, 82–92. [Google Scholar] [CrossRef]
- Pollack, M.; Leeuwenburgh, C. Apoptosis and aging: Role of the mitochondria. J. Gerontol. A Biol. Sci. Med. Sci. 2001, 56, B475–B482. [Google Scholar] [CrossRef]
- Pollack, M.; Phaneuf, S.; Dirks, A.; Leeuwenburgh, C. The role of apoptosis in the normal aging brain, skeletal muscle, and heart. Ann. N. Y. Acad. Sci. 2002, 959, 93–107. [Google Scholar] [CrossRef]
- Jain, S.K.; Micinski, D. Vitamin D upregulates glutamate cysteine ligase and glutathione reductase, and GSH formation, and decreases ROS and MCP-1 and IL-8 secretion in high-glucose exposed U937 monocytes. Biochem. Res. Commun 2013, 437, 7–11. [Google Scholar] [CrossRef] [Green Version]
- Ruggiero, C.; Cherubini, A.; Ble, A.; Bos, A.J.; Maggio, M.; Dixit, V.D.; Lauretani, F.; Bandinelli, S.; Senin, U.; Ferrucci, L. Uric acid and inflammatory markers. Eur. Heart J. 2006, 27, 1174–1181. [Google Scholar] [CrossRef] [Green Version]
- Zoccali, C.; Maio, R.; Mallamaci, F.; Sesti, G.; Perticone, F. Uric acid and endothelial dysfunction in essential hypertension. J. Am. Soc. Nephrol. 2006, 17, 1466–1471. [Google Scholar] [CrossRef] [PubMed]
- Sundström, J.; Sullivan, L.; D’Agostino, R.B.; Levy, D.; Kannel, W.B.; Vasan, R.S. Relations of serum uric acid to longitudinal blood pressure tracking and hypertension incidence. Hypertension 2005, 45, 28–33. [Google Scholar] [CrossRef] [PubMed]
- Kawamoto, R.; Tabara, Y.; Kohara, K.; Kusunoki, T.; Abe, M.; Miki, T. Serum uric acid is more strongly associated with impaired fasting glucose in women than in men from a community-dwelling population. PLoS ONE 2013, 8, e65886. [Google Scholar] [CrossRef] [PubMed]
- Meisinger, C.; Koenig, W.; Baumert, J.; Döring, A. Uric acid levels are associated with all-cause and cardiovascular disease mortality independent of systemic inflammation in men from the general population: The MONICA/KORA cohort study. Arterioscler. Thromb. Vasc. Biol. 2008, 28, 1186–1192. [Google Scholar] [CrossRef] [PubMed]
- Ames, B.N.; Cathcart, R.; Schwiers, E.; Hochstein, P. Uric acid provides an antioxidant defense in humans against oxidant- and radical-caused aging and cancer: A hypothesis. Proc. Natl. Acad. Sci. USA 1981, 78, 6858–6862. [Google Scholar] [CrossRef] [PubMed]
- Glantzounis, G.K.; Tsimoyiannis, E.C.; Kappas, A.M.; Galaris, D.A. Uric acid and oxidative stress. Curr. Pharm. Des. 2005, 11, 4145–4151. [Google Scholar] [CrossRef]
- Macchi, C.; Molino-Lova, R.; Polcaro, P.; Guarducci, L.; Lauretani, F.; Cecchi, F.; Bandinelli, S.; Guralnik, J.M.; Ferrucci, L. Higher circulating levels of uric acid are prospectively associated with better muscle function in older persons. Mech. Ageing 2008, 129, 522–527. [Google Scholar] [CrossRef]
- Wu, Y.; Zhang, D.; Pang, Z.; Jiang, W.; Wang, S.; Tan, Q. Association of serum uric acid level with muscle strength and cognitive function among Chinese aged 50–74 years. Geriatrgerontolol. Int. 2013, 13, 672–677. [Google Scholar] [CrossRef]
- Kawamoto, R.; Ninomiya, D.; Kasai, Y.; Kusunoki, T.; Ohtsuka, N.; Kumagi, T.; Abe, M. Serum uric acid is positively associated with handgrip strength among Japanese community-dwelling elderly women. PLoS ONE 2016, 11, e0151044. [Google Scholar] [CrossRef]


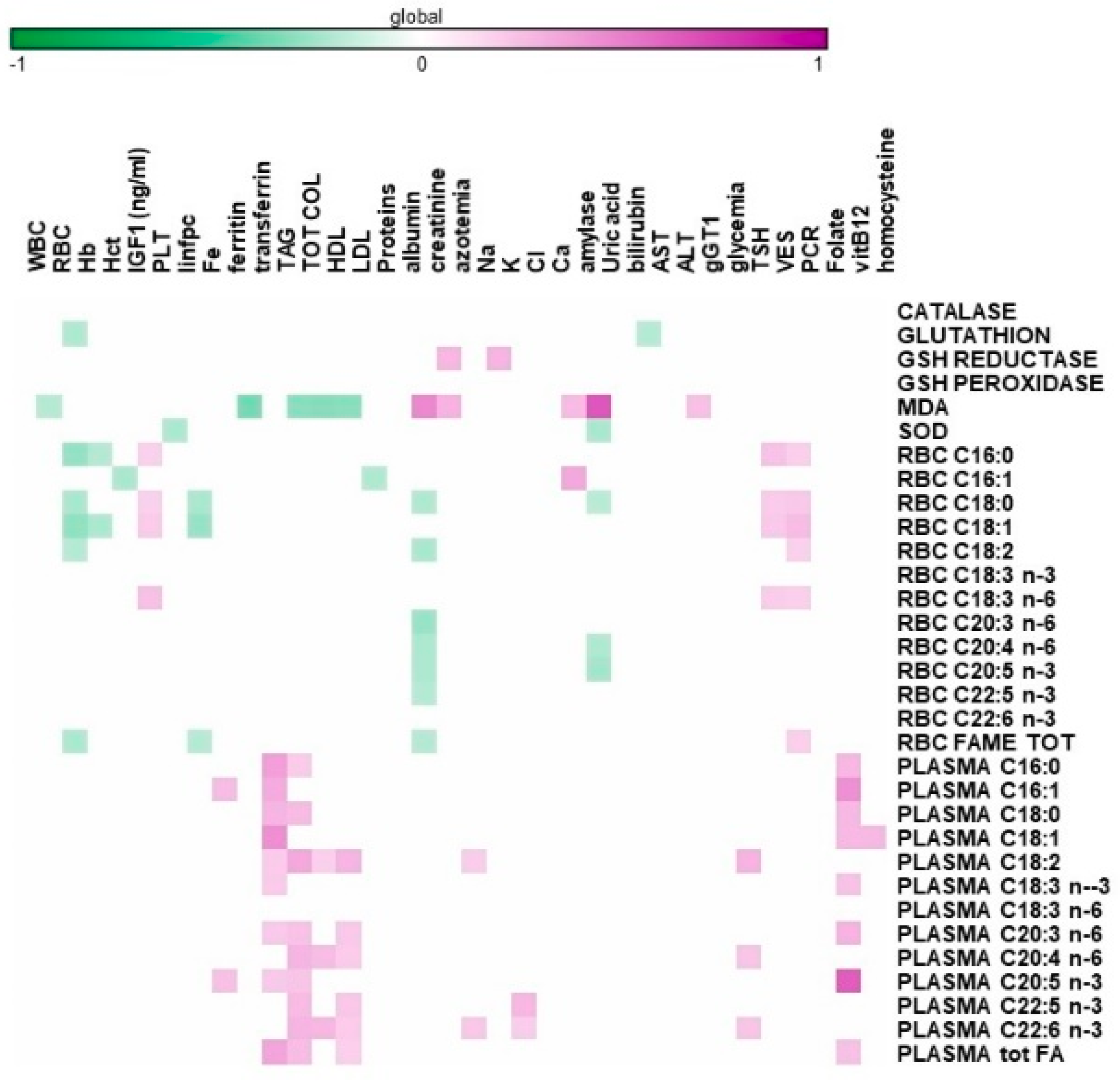

| Supplement Group (69 Subjects) | Placebo Group (61 Subjects) | |
|---|---|---|
| Characteristics | ||
| Age (y) | 80.77 ± 6.29 | 80.21 ± 8.54 |
| Male N (%) | 29 (42%) | 24 (39%) |
| Smokers N (%) | 3 (4%) | 5 (8%) |
| Level of schooling (years) | 7 (3–11) | 5 (2–9) |
| Body composition | ||
| Fat free mass (kg) | 39.90 ± 8.13 | 38.71 ± 8.37 |
| Fat mass (kg) | 17.81 ± 6.78 | 19.21 ± 9.18 |
| Gynoid (%) | 35.79 ± 9.67 | 37.67 ± 10.60 |
| Android (%) | 34.21 ± 10.76 | 34.26 ± 12.85 |
| RSMM (kg/m2) | 6.60 ± 1.19 | 6.36 ± 1.32 |
| MNA (score) | 17.84 ± 3.07 | 17.84 ± 3.57 |
| Weight (kg) | 59.47 ± 11.16 | 59.39 ± 13.51 |
| BMI (kg/m2) | 23.85 ± 3.63 | 23.93 ± 4.60 |
| Wrist circumference (cm) | 16.29 ± 1.75 | 16.08 ± 1.42 |
| Arm circumference (cm) | 25.22 ± 3.36 | 25.02 ± 3.80 |
| Calf circumference (cm) | 30.43 ± 3.13 | 29.95 ± 4.55 |
| Waist circumference (cm) | 88.95 ± 9.74 | 89.01 ± 10.15 |
| Handgrip (kg) | 16.63 ± 4.99 | 19.62 ± 6.01 |
| Psychological scores | ||
| MMSE (score) | 21.78 ± 3.70 | 20.5 ± 4.93 |
| ADL (score) | 3.97 ± 1.19 | 4.03 ± 1.08 |
| SF-36 MCS (score) | 46.65 ± 10.7 | 44.0 ± 9.7 |
| SF-36 PCS (score) | 34.1 ± 10.2 | 37.1 ± 11.0 |
| Hematology | Supplemented Group | Placebo Group |
|---|---|---|
| Proteins (g/dL) | 6.67 ± 0.55 | 6.56 ± 0.61 |
| Albumin (g/dL) | 3.76 ± 0.54 | 3.6 ± 0.55 |
| Albumin (%) | 55.92 ± 5.82 | 54.83 ± 6.00 |
| Creatinine (mg/dL) | 0.95 ± 0.7 | 0.91 ± 0.38 |
| Azotaemia (mg/dL) | 45.00 ± 35.41 | 46.92 ± 22.42 |
| Uric Acid (mg/dL) | 5.00 ± 2.20 | 5.15 ± 1.89 |
| CRP (mg/L) | 0.30 (0.14–1.23) | 0.33 (0.16–1.03) |
| IGF-I (ng/mL) | 80.6 ± 33.8 | 82.7 ± 38.8 |
| White Blood Cell (109/L) | 6.96 ± 2.06 | 7.39 ± 3.08 |
| Red Blood Cell (1012/L) | 4.27 ± 0.48 | 4.24 ± 0.84 |
| Hemoglobin (g/dL) | 12.60 ± 1.47 | 12.19 ± 1.77 |
| Hematocrit % | 38.20 ± 4.25 | 36.91 ± 6.31 |
| Platelet count (109/L) | 245.94 ± 85.38 | 240.14 ± 147.75 |
| Lymphocyte count (109/L) | 1.80 ± 0.59 | 2.38 ± 3.88 |
| Lymphocytes % | 27.18 ± 8.51 | 25.92 ± 10.88 |
| Iron (µg/dL) | 72.46 ± 30.65 | 65.60 ± 30.79 |
| Transferrin (g/L) | 2.19 ± 0.50 | 2.15 ± 0.76 |
| Glycaemia mmol/L | 5.96 ± 2.12 | 5.51 ± 1.78 |
| Triglyceride mmol/L | 3.01 ± 1.09 | 3.32 ± 1.92 |
| Total Cholesterol mmol/L | 5.05 ± 1.21 | 4.9 ± 1.42 |
| High-Density Lipoprotein mmol/L | 1.35 ± 0.41 | 1.17 ± 0.38 |
| Low-Density Lipoprotein mmol/L | 3.14 ± 1.02 | 2.71 ± 1.48 |
| Sodium (mEq/L) | 138.88 ± 2.74 | 139.43 ± 2.92 |
| Potassium (mEq/L) | 4.06 ± 0.42 | 4.25 ± 0.61 |
| Chlorine (mEq/L) | 103.71 ± 3.86 | 104.83 ± 4.24 |
| Calcium (mg/dL) | 9.15 ± 0.63 | 9.02 ± 0.54 |
| Amylase (U/L) | 32.53 ± 24.31 | 28.60 ± 12.40 |
| Total Bilirubin (mg/dL) | 0.74 ± 0.47 | 0.71 ± 0.35 |
| AST (U/L) | 19.01 ± 10.54 | 21.52 ± 19.19 |
| ALT (U/L) | 16.35 ± 11.94 | 20.08 ± 18.22 |
| gGT (U/L) | 32.31 ± 38.43 | 36.70 ± 42.17 |
| Fatty Acid | Placebo Group | Supplemented Group | ||||
|---|---|---|---|---|---|---|
| PRE | POST | p Value | PRE | POST | p Value | |
| Palmitic acid (C16:0) | 30.1 ± 3.52 | 28.1 ± 3.70 | 0.007 | 29.2 ± 3.01 | 29.3 ± 2.95 | 0.843 |
| Palmitoleic acid (C16:1) | 3.63 ± 1.14 | 3.83 ± 1.52 | 0.209 | 3.63 ± 1.25 | 3.55 ± 0.99 | 0.528 |
| Stearic acid (C18:0) | 5.75 ± 0.95 | 5.76 ± 1.02 | 0.952 | 5.61 ± 0.69 | 6.08 ± 0.85 | 0.001 |
| Oleic acid (C18:1) | 24.2 ± 6.44 | 25.4 ± 3.73 | 0.265 | 24.1 ± 3.21 | 25.0 ± 2.79 | 0.046 |
| Linoleic acid (C18:2) | 22.9 ± 4.19 | 22.1 ± 3.80 | 0.190 | 23.2 ± 3.23 | 21.6 ± 3.81 | 0.002 |
| α-linolenic acid (C18:3 n-3) | 0.44 ± 0.17 | 0.48 ± 0.18 | 0.206 | 0.53 ± 0.29 | 0.60 ± 0.52 | 0.347 |
| γ-linolenic acid (C18:3 n-6) | 0.23 ± 0.18 | 0.30 ± 0.29 | 0.191 | 0.25 ± 0.30 | 0.28 ± 0.25 | 0.504 |
| Dihomo-γ-linolenic (C20:3 n-6) | 1.30 ± 0.34 | 1.44 ± 0.39 | 0.014 | 1.36 ± 0.91 | 1.44 ± 0.32 | 0.496 |
| Arachidonic acid (C20:4 n-6) | 9.24 ± 2.48 | 10.31 ± 7.27 | 0.325 | 9.64 ± 2.73 | 9.81 ± 2.67 | 0.655 |
| Eicosapentaenoic acid (C20:5 n-3) | 0.34 ± 0.19 | 0.43 ± 0.17 | 0.076 | 0.42 ± 0.15 | 0.45 ± 0.18 | 0.199 |
| Docosapentaenoic acid (C22:5 n-3) | 0.42 ± 0.18 | 0.41 ± 0.10 | 0.593 | 0.42 ± 0.12 | 0.43 ± 0.10 | 0.694 |
| Docosahexaenoic acid (C22:6 n-3) | 1.50 ± 0.42 | 1.49 ± 0.41 | 0.927 | 1.57 ± 0.58 | 1.51 ± 0.48 | 0.424 |
| AA/EPA | 29.3 ± 12.3 | 26.3 ± 16.6 | 0.299 | 24.9 ± 8.56 | 24.5 ± 9.77 | 0.809 |
| Saturated fatty acids | 35.8 ± 3.75 | 33.9 ± 3.94 | 0.012 | 34.8 ± 2.99 | 35.4 ± 3.21 | 0.229 |
| Monounsaturated fatty acids | 27.8 ± 6.47 | 29.2 ± 4.21 | 0.168 | 27.8 ± 3.65 | 28.5 ± 3.02 | 0.112 |
| Polyunsaturated fatty acids | 36.4 ± 5.4 | 36.9 ± 6.21 | 0.604 | 37.6 ± 4.56 | 36.1 ± 4.27 | 0.024 |
| Omega-6 | 33.6 ± 5.23 | 34.1 ± 6.21 | 0.654 | 34.5 ± 4.35 | 33.1 ± 4.13 | 0.028 |
| Omega-3 | 2.73 ± 0.57 | 2.81 ± 0.54 | 0.381 | 2.93 ± 0.80 | 3.00 ± 0.83 | 0.618 |
| Omega-6/Omega-3 ratio | 12.9 ± 3.97 | 12.7 ± 4.32 | 0.809 | 12.4 ± 2.84 | 11.8 ± 3.23 | 0.155 |
| Plasma Malondialdehyde (µM) | 3.78 ± 1.91 | 4.20 ± 1.98 | 0.084 | 3.42 ± 1.68 | 3.51 ± 1.75 | 0.663 |
| Superoxide dismutase (U/g Hb) | 4724 ± 2499 | 4800 ± 2868 | 0.891 | 4413 ± 2520 | 4445 ± 2269 | 0.937 |
| Total GSH (nmol/mg Hb) | 4.08 ± 1.45 | 4.33 ± 1.93 | 0.408 | 4.35 ± 1.23 | 4.34 ± 1.61 | 0.981 |
| Glutathione Reductase (U/g Hb) | 3.06 ± 1.16 | 3.23 ± 1.13 | 0.352 | 2.59 ± 0.66 | 2.85 ± 0.88 | 0.031 |
| Glutathione Peroxidase (U/g Hb) | 28.6 ± 16.2 | 30.2 ± 16.0 | 0.553 | 36.3 ± 13.6 | 31.2 ± 18.1 | 0.084 |
| Variables | Δ (95%) Between Groups | p Value Treatment Effect |
|---|---|---|
| Palmitic acid | 1.73 (0.13 to 3.33) | 0.034 |
| Palmitoleic acid | −0.31 (−0.71 to 0.10) | 0.137 |
| Stearic acid | 0.58 (0.14 to 1.02) | 0.011 |
| Oleic acid | −0.69 (−2.80 to 1.42) | 0.520 |
| Linoleic acid | −0.25 (−1.88 to 1.39) | 0.765 |
| α-linolenic acid | 0.02 (−0.17 to 0.22) | 0.799 |
| γ-linolenic acid | 0.03 (−0.10 to 0.16) | 0.656 |
| Di-homo-γ-linolenic | −0.08 (−0.39 to 0.23) | 0.622 |
| Arachidonic acid | −0.96 (−3.05 to 1.14) | 0.366 |
| Eicosapentaenoic acid | −0.02 (−0.09 to 0.05) | 0.622 |
| Docosapentaenoic acid | −0.00 (−0.05 to 0.05) | 0.912 |
| Docosahexaenoic acid | -0.09 (-0.28 to 0.10) | 0.349 |
| AA/EPA | 1.19 (−4.89 to 7.27) | 0.699 |
| Saturated fatty acids | 2.31 (0.61 to 4.01) | 0.008 |
| Monounsaturated fatty acids | −0.99 (−3.11 to 1.12) | 0.354 |
| Polyunsaturated fatty acids | −1.47 (−3.87 to 0.93) | 0.226 |
| Omega-6 | −1.25 (−3.66 to 1.15) | 0.303 |
| Omega-3 | −0.07 (−0.40 to 0.26) | 0.673 |
| Omega-6/Omega-3 | 0.27 (−1.48 to 2.01) | 0.763 |
| Malondialdehyde | −0.36 (−0.98 to 0.25) | 0.243 |
| Superoxide dismutase | 702 (−682 to 2086) | 0.316 |
| Glutathione | 0.02 (−0.54 to 0.58) | 0.951 |
| Glutathione reductase | −0.002 (−0.41 to 0.41) | 0.993 |
| Glutathione peroxidase | −7.71 (−15.92 to 0.49) | 0.065 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Corsetto, P.A.; Montorfano, G.; Klersy, C.; Massimino, L.; Infantino, V.; Iannello, G.; Anna Faliva, M.; Lukaski, H.; Perna, S.; Alalwan, T.A.; et al. Fatty Acid Profile and Antioxidant Status Fingerprint in Sarcopenic Elderly Patients: Role of Diet and Exercise. Nutrients 2019, 11, 2569. https://doi.org/10.3390/nu11112569
Corsetto PA, Montorfano G, Klersy C, Massimino L, Infantino V, Iannello G, Anna Faliva M, Lukaski H, Perna S, Alalwan TA, et al. Fatty Acid Profile and Antioxidant Status Fingerprint in Sarcopenic Elderly Patients: Role of Diet and Exercise. Nutrients. 2019; 11(11):2569. https://doi.org/10.3390/nu11112569
Chicago/Turabian StyleCorsetto, Paola Antonia, Gigliola Montorfano, Catherine Klersy, Luca Massimino, Vittoria Infantino, Giancarlo Iannello, Milena Anna Faliva, Henry Lukaski, Simone Perna, Tariq A. Alalwan, and et al. 2019. "Fatty Acid Profile and Antioxidant Status Fingerprint in Sarcopenic Elderly Patients: Role of Diet and Exercise" Nutrients 11, no. 11: 2569. https://doi.org/10.3390/nu11112569
APA StyleCorsetto, P. A., Montorfano, G., Klersy, C., Massimino, L., Infantino, V., Iannello, G., Anna Faliva, M., Lukaski, H., Perna, S., Alalwan, T. A., Rizzo, A. M., & Rondanelli, M. (2019). Fatty Acid Profile and Antioxidant Status Fingerprint in Sarcopenic Elderly Patients: Role of Diet and Exercise. Nutrients, 11(11), 2569. https://doi.org/10.3390/nu11112569







