Short Chain Fatty Acids and Fecal Microbiota Abundance in Humans with Obesity: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Literature Search
2.2. Inclusion and Exclusion Criteria
2.3. Data Extraction and Quality Assessment
2.4. Assessment of Quality of Evidence
2.5. Data Synthesis and Statistical Analyses
3. Results
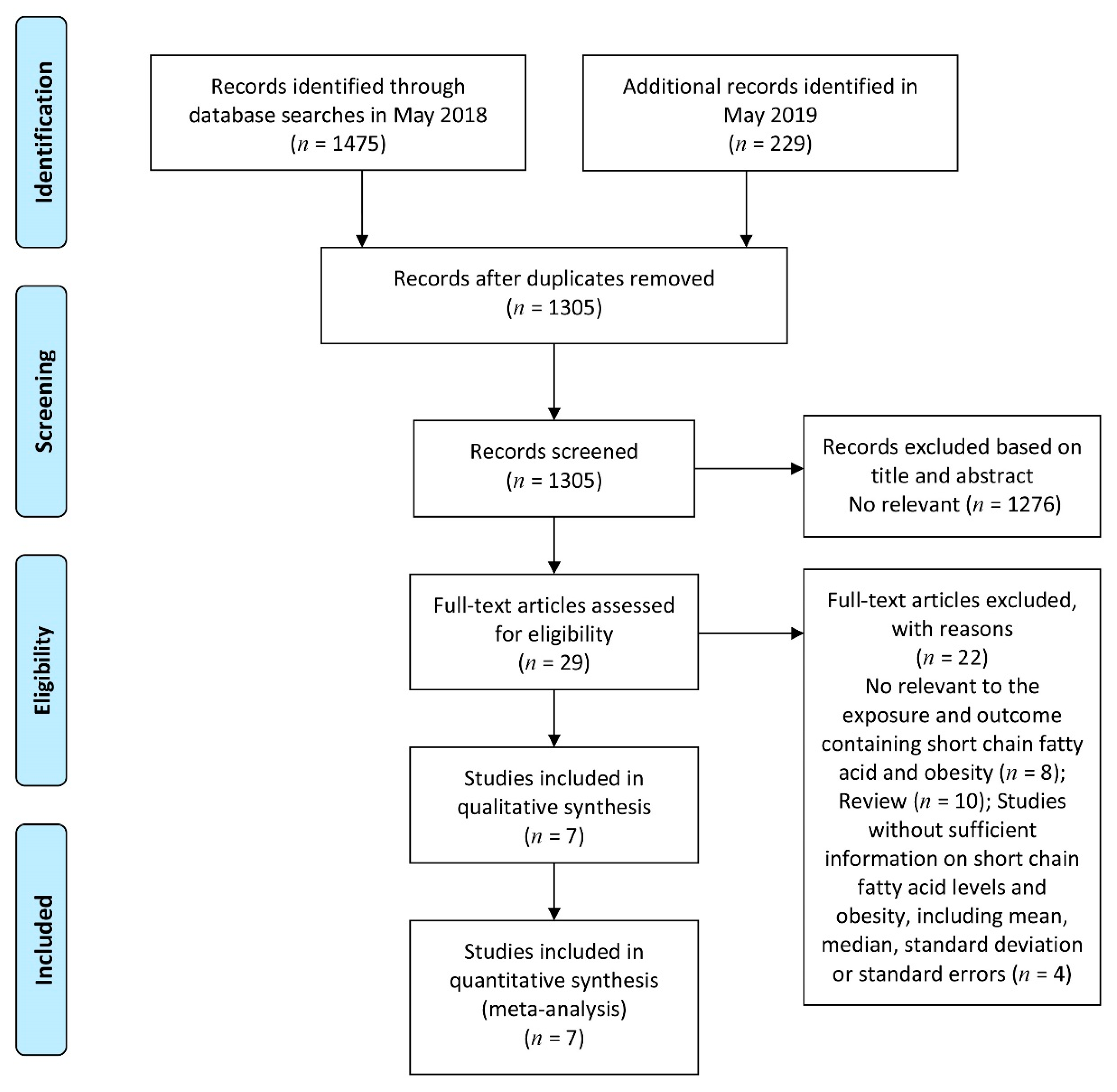
3.1. Study Search and Selection and Characteristics of Eligible Studies
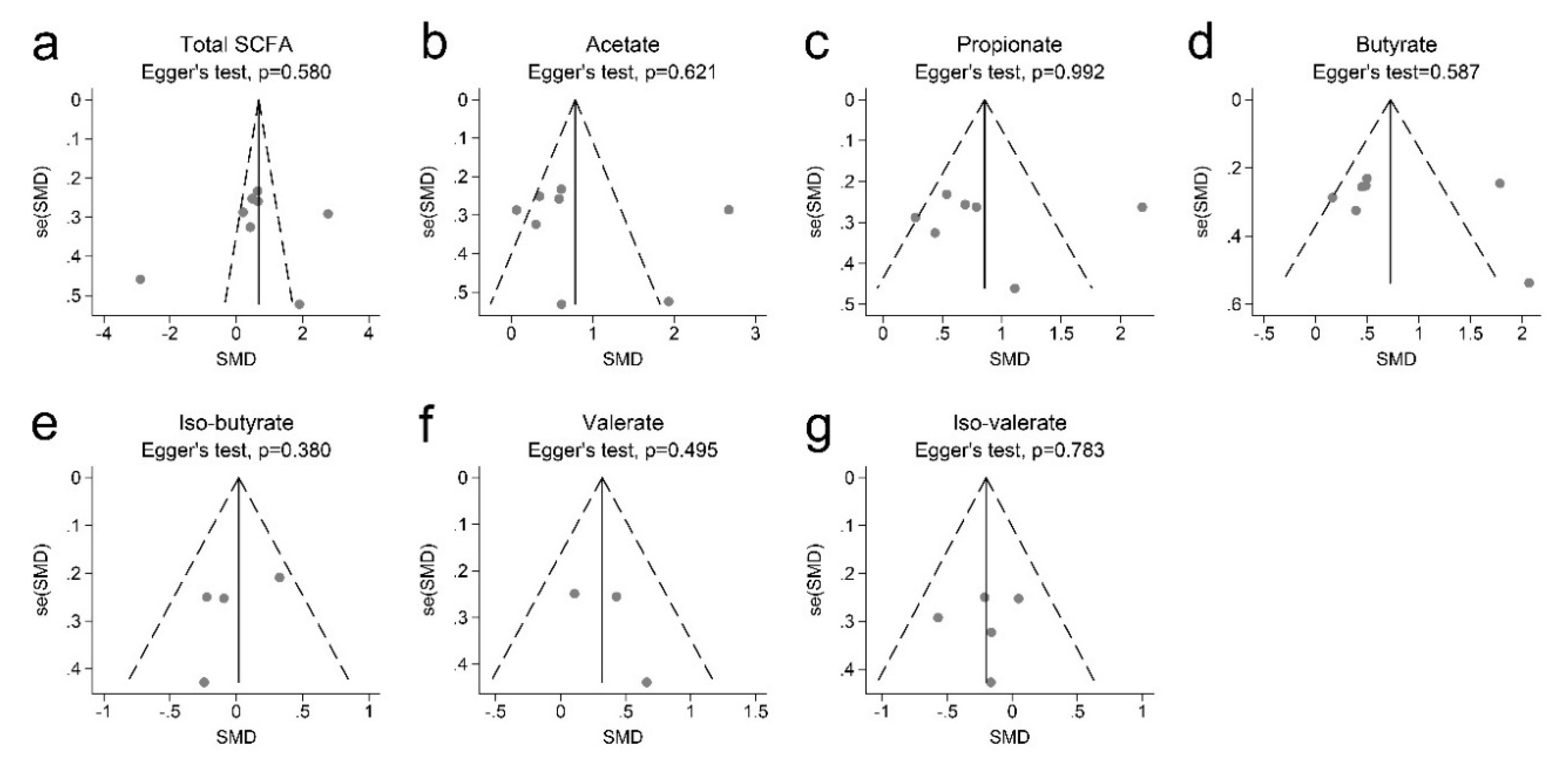
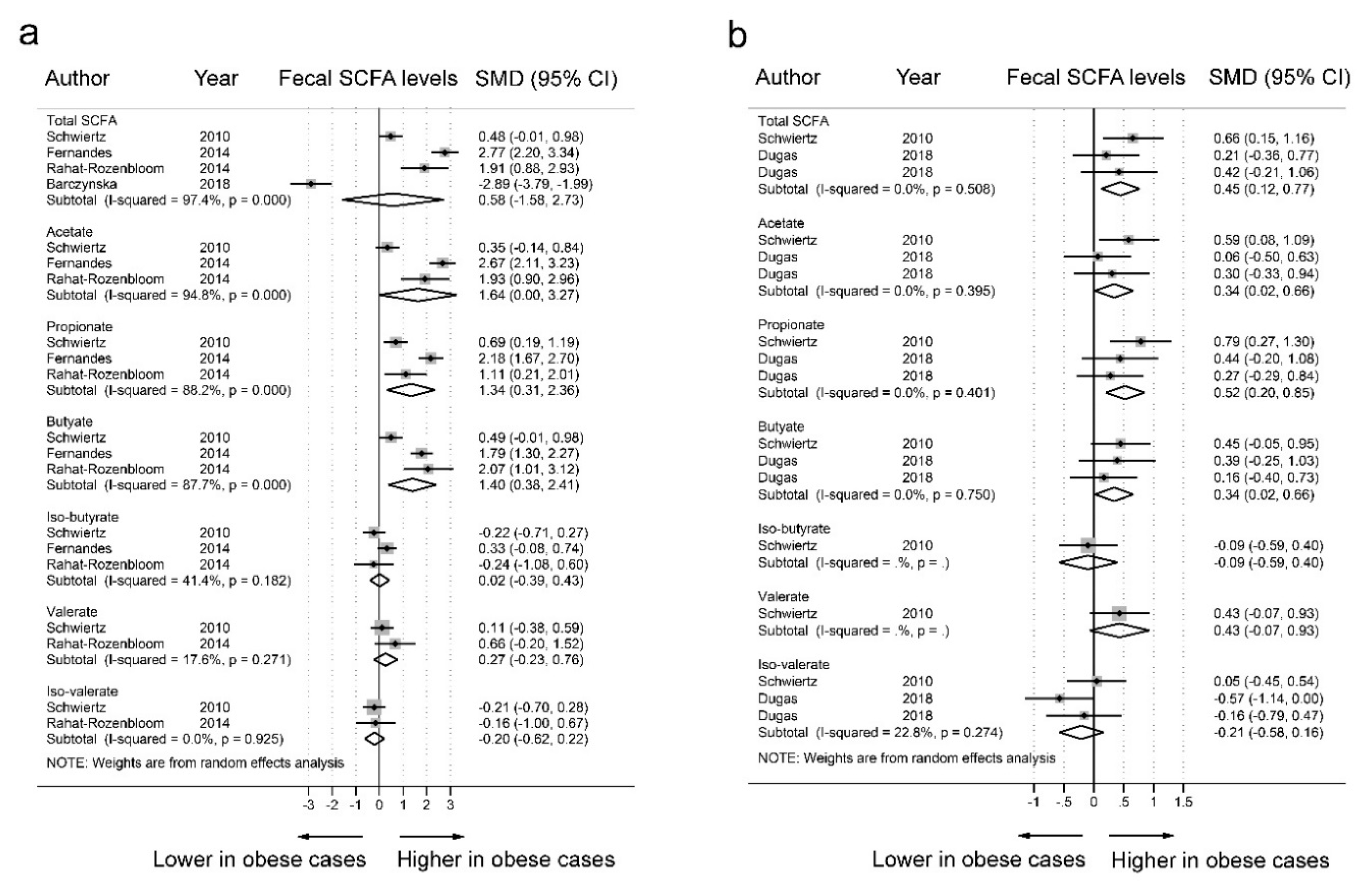
3.2. SCFAs and Obesity
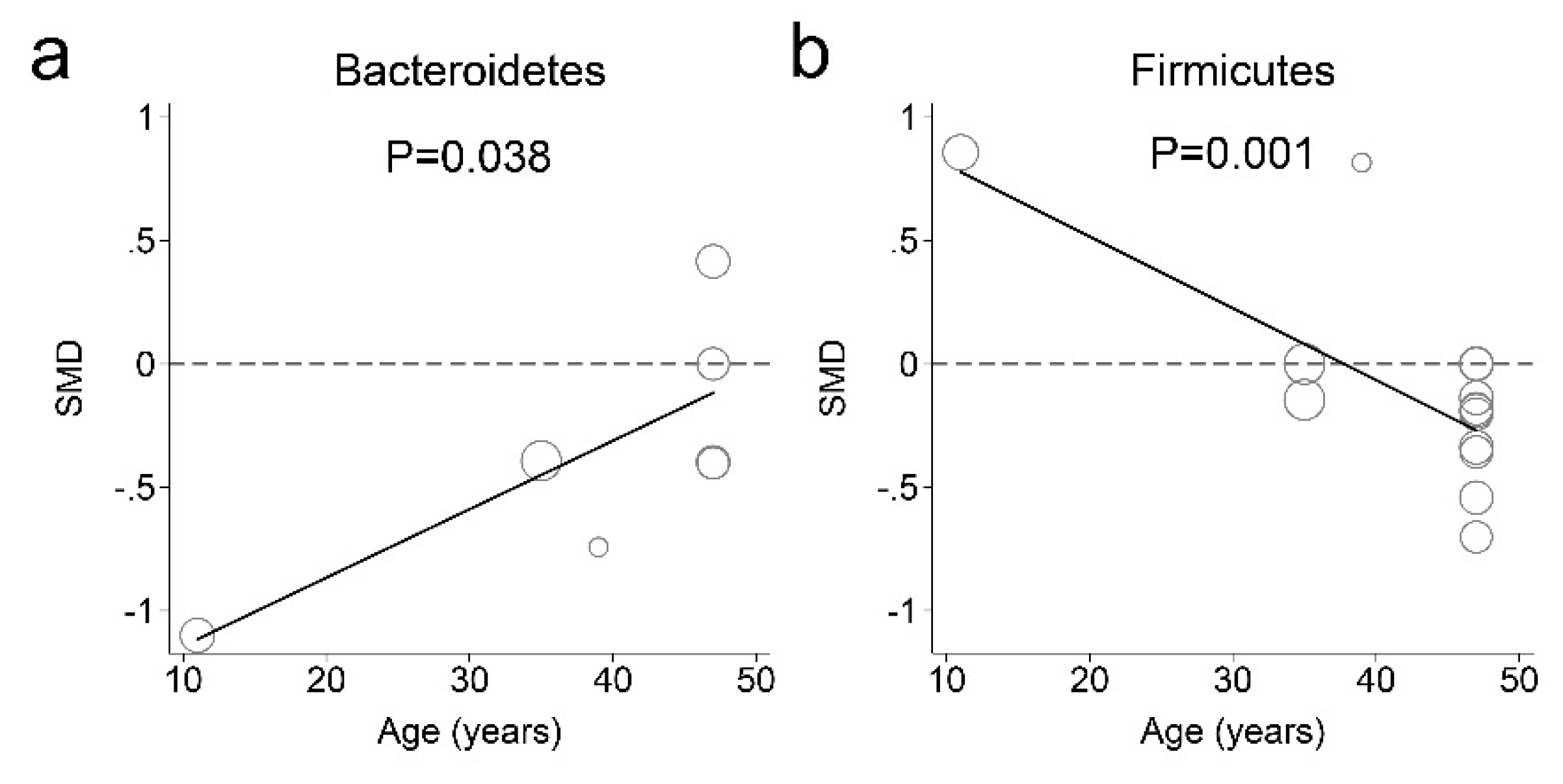
3.3. Fecal Microbiota and Obesity
3.4. Quality Assessment of Included Studies
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Upadhyay, J.; Farr, O.; Perakakis, N.; Ghaly, W.; Mantzoros, C. Obesity as a Disease. Med. Clin. N. Am. 2018, 102, 13–33. [Google Scholar] [CrossRef]
- Blaut, M. Gut microbiota and energy balance: Role in obesity. Proc. Nutr. Soc. 2015, 74, 227–234. [Google Scholar] [CrossRef] [PubMed]
- Brusaferro, A.; Cozzali, R.; Orabona, C.; Biscarini, A.; Farinelli, E.; Cavalli, E.; Grohmann, U.; Principi, N.; Esposito, S. Is It Time to Use Probiotics to Prevent or Treat Obesity? Nutrients 2018, 10, 1613. [Google Scholar] [CrossRef] [PubMed]
- Morrison, D.J.; Preston, T. Formation of short chain fatty acids by the gut microbiota and their impact on human metabolism. Gut Microbes 2016, 7, 189–200. [Google Scholar] [CrossRef] [PubMed]
- den Besten, G.; Lange, K.; Havinga, R.; van Dijk, T.H.; Gerding, A.; van Eunen, K.; Muller, M.; Groen, A.K.; Hooiveld, G.J.; Bakker, B.M.; et al. Gut-derived short-chain fatty acids are vividly assimilated into host carbohydrates and lipids. Am. J. Physiol. 2013, 305, G900–G910. [Google Scholar] [CrossRef]
- Liou, A.P.; Paziuk, M.; Luevano, J.M., Jr.; Machineni, S.; Turnbaugh, P.J.; Kaplan, L.M. Conserved shifts in the gut microbiota due to gastric bypass reduce host weight and adiposity. Sci. Transl. Med. 2013, 5, 178. [Google Scholar] [CrossRef]
- Perry, R.J.; Peng, L.; Barry, N.A.; Cline, G.W.; Zhang, D.; Cardone, R.L.; Petersen, K.F.; Kibbey, R.G.; Goodman, A.L.; Shulman, G.I. Acetate mediates a microbiome-brain-beta-cell axis to promote metabolic syndrome. Nature 2016, 534, 213–217. [Google Scholar] [CrossRef]
- Fernandes, J.; Su, W.; Rahat-Rozenbloom, S.; Wolever, T.M.; Comelli, E.M. Adiposity, gut microbiota and faecal short chain fatty acids are linked in adult humans. Nutr. Diabetes 2014, 4, e121. [Google Scholar] [CrossRef]
- Rahat-Rozenbloom, S.; Fernandes, J.; Gloor, G.B.; Wolever, T.M. Evidence for greater production of colonic short-chain fatty acids in overweight than lean humans. Int. J. Obes. 2014, 38, 1525–1531. [Google Scholar] [CrossRef]
- Riva, A.; Borgo, F.; Lassandro, C.; Verduci, E.; Morace, G.; Borghi, E.; Berry, D. Pediatric obesity is associated with an altered gut microbiota and discordant shifts in Firmicutes populations. Environ. Microbiol. 2017, 19, 95–105. [Google Scholar] [CrossRef]
- Barczynska, R.; Litwin, M.; Slizewska, K.; Szalecki, M.; Berdowska, A.; Bandurska, K.; Libudzisz, Z.; Kapusniak, J. Bacterial Microbiota and Fatty Acids in the Faeces of Overweight and Obese Children. Pol. J. Microbiol. 2018, 67, 339–345. [Google Scholar] [CrossRef] [PubMed]
- Furet, J.P.; Kong, L.C.; Tap, J.; Poitou, C.; Basdevant, A.; Bouillot, J.L.; Mariat, D.; Corthier, G.; Dore, J.; Henegar, C.; et al. Differential adaptation of human gut microbiota to bariatric surgery-induced weight loss: Links with metabolic and low-grade inflammation markers. Diabetes 2010, 59, 3049–3057. [Google Scholar] [CrossRef] [PubMed]
- Schwiertz, A.; Taras, D.; Schafer, K.; Beijer, S.; Bos, N.A.; Donus, C.; Hardt, P.D. Microbiota and SCFA in lean and overweight healthy subjects. Obesity 2010, 18, 190–195. [Google Scholar] [CrossRef] [PubMed]
- Duncan, S.H.; Lobley, G.E.; Holtrop, G.; Ince, J.; Johnstone, A.M.; Louis, P.; Flint, H.J. Human colonic microbiota associated with diet, obesity and weight loss. Int. J. Obes. 2008, 32, 1720–1724. [Google Scholar] [CrossRef]
- Walters, W.A.; Xu, Z.; Knight, R. Meta-analyses of human gut microbes associated with obesity and IBD. FEBS Lett. 2014, 588, 4223–4233. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. Ann. Intern. Med. 2009, 151, 264–269. [Google Scholar] [CrossRef]
- Stroup, D.F.; Berlin, J.A.; Morton, S.C.; Olkin, I.; Williamson, G.D.; Rennie, D.; Moher, D.; Becker, B.J.; Sipe, T.A.; Thacker, S.B. Meta-analysis of observational studies in epidemiology: A proposal for reporting. Meta-analysis Of Observational Studies in Epidemiology (MOOSE) group. Jama 2000, 283, 2008–2012. [Google Scholar] [CrossRef]
- Guyatt, G.; Oxman, A.D.; Akl, E.A.; Kunz, R.; Vist, G.; Brozek, J.; Norris, S.; Falck-Ytter, Y.; Glasziou, P.; DeBeer, H.; et al. GRADE guidelines: 1. Introduction-GRADE evidence profiles and summary of findings tables. J. Clin. Epidemiol. 2011, 64, 383–394. [Google Scholar] [CrossRef]
- GRADEpro GDT: GRADEpro Guideline Development Tool [Software]. McMaster University, 2015 (Developed by Evidence Prime, Inc.). Available online: http://gradepro.org (accessed on 9 March 2019).
- Higgins, J.P.T.; Green, S. Cochrane Handbook for Systematic Reviews of Interventions; John Wiley & Sons: Hoboken, NJ, USA, 2008; Volume 5. [Google Scholar]
- DerSimonian, R.; Laird, N. Meta-analysis in clinical trials. Controll. Clin. Trials 1986, 7, 177–188. [Google Scholar] [CrossRef]
- Higgins, J.P.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. BMJ 2003, 327, 557–560. [Google Scholar] [CrossRef]
- Egger, M.; Davey Smith, G.; Schneider, M.; Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997, 315, 629–634. [Google Scholar] [CrossRef] [PubMed]
- Dugas, L.R.; Bernabe, B.P.; Priyadarshini, M.; Fei, N.; Park, S.J.; Brown, L.; Plange-Rhule, J.; Nelson, D.; Toh, E.C.; Gao, X.; et al. Decreased microbial co-occurrence network stability and SCFA receptor level correlates with obesity in African-origin women. Sci. Rep. 2018, 8, 17135. [Google Scholar] [CrossRef] [PubMed]
- Todesco, T.; Zamboni, M.; Armellini, F.; Bissoli, L.; Turcato, E.; Piemonte, G.; Rao, A.V.; Jenkins, D.J.; Bosello, O. Plasma acetate levels in a group of obese diabetic, obese normoglycemic, and control subjects and their relationships with other blood parameters. Am. J. Gastroenterol. 1993, 88, 751–755. [Google Scholar] [PubMed]
- Flint, H.J.; Bayer, E.A.; Rincon, M.T.; Lamed, R.; White, B.A. Polysaccharide utilization by gut bacteria: Potential for new insights from genomic analysis. Nat. Rev. Microbiol. 2008, 6, 121–131. [Google Scholar] [CrossRef]
- Riley, L.W.; Raphael, E.; Faerstein, E. Obesity in the United States - dysbiosis from exposure to low-dose antibiotics? Front. Public Health 2013, 1, 69. [Google Scholar] [CrossRef]
- Diamant, M.; Blaak, E.E.; de Vos, W.M. Do nutrient-gut-microbiota interactions play a role in human obesity, insulin resistance and type 2 diabetes? Obes. Rev. 2011, 12, 272–281. [Google Scholar] [CrossRef]
- De la Cuesta-Zuluaga, J.; Mueller, N.T.; Alvarez-Quintero, R.; Velasquez-Mejia, E.P.; Sierra, J.A.; Corrales-Agudelo, V.; Carmona, J.A.; Abad, J.M.; Escobar, J.S. Higher Fecal Short-Chain Fatty Acid Levels Are Associated with Gut Microbiome Dysbiosis, Obesity, Hypertension and Cardiometabolic Disease Risk Factors. Nutrients 2018, 11, 51. [Google Scholar] [CrossRef]
- Andersen-Nissen, E.; Smith, K.D.; Strobe, K.L.; Barrett, S.L.; Cookson, B.T.; Logan, S.M.; Aderem, A. Evasion of Toll-like receptor 5 by flagellated bacteria. Proc. Natl. Acad. Sci. USA 2005, 102, 9247–9252. [Google Scholar] [CrossRef]
- Forstneric, V.; Ivicak-Kocjan, K.; Plaper, T.; Jerala, R.; Bencina, M. The role of the C-terminal D0 domain of flagellin in activation of Toll like receptor 5. PLoS Pathog. 2017, 13, e1006574. [Google Scholar] [CrossRef]
- Vijay-Kumar, M.; Aitken, J.D.; Carvalho, F.A.; Cullender, T.C.; Mwangi, S.; Srinivasan, S.; Sitaraman, S.V.; Knight, R.; Ley, R.E.; Gewirtz, A.T. Metabolic syndrome and altered gut microbiota in mice lacking Toll-like receptor 5. Science 2010, 328, 228–231. [Google Scholar] [CrossRef]
- Priyadarshini, M.; Kotlo, K.U.; Dudeja, P.K.; Layden, B.T. Role of Short Chain Fatty Acid Receptors in Intestinal Physiology and Pathophysiology. Compr. Physiol. 2018, 8, 1091–1115. [Google Scholar] [CrossRef] [PubMed]
- Arora, T.; Sharma, R.; Frost, G. Propionate. Anti-obesity and satiety enhancing factor? Appetite 2011, 56, 511–515. [Google Scholar] [CrossRef] [PubMed]
- Diraison, F.; Dusserre, E.; Vidal, H.; Sothier, M.; Beylot, M. Increased hepatic lipogenesis but decreased expression of lipogenic gene in adipose tissue in human obesity. Am. J. Physiol. Endocrinol. Metab. 2002, 282, E46–E51. [Google Scholar] [CrossRef] [PubMed]
- Kasubuchi, M.; Hasegawa, S.; Hiramatsu, T.; Ichimura, A.; Kimura, I. Dietary gut microbial metabolites, short-chain fatty acids, and host metabolic regulation. Nutrients 2015, 7, 2839–2849. [Google Scholar] [CrossRef]
- Cani, P.D.; Bibiloni, R.; Knauf, C.; Waget, A.; Neyrinck, A.M.; Delzenne, N.M.; Burcelin, R. Changes in gut microbiota control metabolic endotoxemia-induced inflammation in high-fat diet-induced obesity and diabetes in mice. Diabetes 2008, 57, 1470–1481. [Google Scholar] [CrossRef]
- Chambers, E.S.; Viardot, A.; Psichas, A.; Morrison, D.J.; Murphy, K.G.; Zac-Varghese, S.E.; MacDougall, K.; Preston, T.; Tedford, C.; Finlayson, G.S.; et al. Effects of targeted delivery of propionate to the human colon on appetite regulation, body weight maintenance and adiposity in overweight adults. Gut 2015, 64, 1744–1754. [Google Scholar] [CrossRef]
- Le Chatelier, E.; Nielsen, T.; Qin, J.; Prifti, E.; Hildebrand, F.; Falony, G.; Almeida, M.; Arumugam, M.; Batto, J.M.; Kennedy, S.; et al. Richness of human gut microbiome correlates with metabolic markers. Nature 2013, 500, 541–546. [Google Scholar] [CrossRef]


| BMI Category, | Sex, Age | SCFA | Specimen | Obese Individuals | Nonobese Individuals | Measure | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| Study | (mean, y) | unit | n | Mean | SD | n | Mean | SD | SCFA | |
| BMI-Z (mean) | 2.14–5 (3) | −2.12–1.56 (0.3) | ||||||||
| Riva, | M and F | Total SCFA | Feces | 42 | 65.3 | 32.4 | 36 | 47.5 | 20.4 | CE |
| 2017, Italy | 9–16 (11) | Acetate | µmol/g | 42 | 40.4 | 18.9 | 36 | 30.3 | 13 | |
| Propionate | 42 | 12.5 | 7.7 | 36 | 8.8 | 5.8 | ||||
| Butyrate | 42 | 12.4 | 9.8 | 36 | 8.4 | 5.3 | ||||
| BMI (kg/m2) | >25 | <25 | ||||||||
| Barczyńska, | M and F | Total SCFA | Feces | 20 | 3.59 | 0.49 | 20 | 5.44 | 0.76 | HPLC |
| 2018, Poland | 6–15 (10) | mg/g | ||||||||
| BMI (kg/m2) | >25 | <25 | ||||||||
| Fernandes, | M and F | Total SCFA | Feces | 42 | 89.7 | 4.2 | 52 | 77.6 | 4.5 | GC |
| 2014, Canada | 18–67 (35) | Acetate | mmol/kg | 42 | 48 | 2.3 | 52 | 41.4 | 2.6 | |
| Propionate | 42 | 17.6 | 1.2 | 52 | 15.1 | 1.1 | ||||
| Butyrate | 42 | 16.1 | 1 | 52 | 14 | 1.3 | ||||
| Iso-butyrate | 42 | 3 | 0.4 | 52 | 2.9 | 0.2 | ||||
| BMI (kg/m2) | >25 | <25 | ||||||||
| Rahat-Rozenbloom, | M and F | Total SCFA | Feces | 11 | 81.3 | 7.4 | 11 | 64.1 | 10.4 | GC |
| 2014, Canada | 17 < (39) | Acetate | mmol/kg | 11 | 45.3 | 4.3 | 11 | 35.1 | 6.1 | |
| Propionate | 11 | 15.4 | 2 | 11 | 12.7 | 2.8 | ||||
| Butyrate | 11 | 15.4 | 1.7 | 11 | 11.1 | 2.4 | ||||
| Iso-butyrate | 11 | 1.4 | 0.3 | 11 | 1.5 | 0.5 | ||||
| Valerate | 11 | 1.9 | 0.4 | 11 | 1.6 | 0.5 | ||||
| Iso-valerate | 11 | 2 | 0.5 | 11 | 2.1 | 0.7 | ||||
| BMI (kg/m2) | >25 | <25 | ||||||||
| Schwiertz, | M and F | Total SCFA | Feces | 35 | 98.7 | 33.9 | 30 | 84.6 | 22.9 | GC |
| 2010, Germany | 14–74 (47) | Acetate | mmol/L | 35 | 56 | 18.2 | 30 | 50.5 | 12.6 | |
| Propionate | 35 | 18.3 | 7.9 | 30 | 13.6 | 5.2 | ||||
| Butyrate | 35 | 18.5 | 10.1 | 30 | 14.1 | 7.6 | ||||
| Iso-butyrate | 35 | 1.6 | 0.9 | 30 | 1.8 | 0.9 | ||||
| Valerate | 35 | 2 | 1.1 | 30 | 1.9 | 0.7 | ||||
| Iso-valerate | 35 | 2.3 | 1.7 | 30 | 2.7 | 2.1 | ||||
| BMI (kg/m2) | >30 | <25 | ||||||||
| Total SCFA | 33 | 103.9 | 34.3 | 30 | 84.6 | 22.9 | ||||
| Acetate | 33 | 59.8 | 18.3 | 30 | 50.5 | 12.6 | ||||
| Propionate | 33 | 19.3 | 8.7 | 30 | 13.6 | 5.2 | ||||
| Butyrate | 33 | 18.1 | 10 | 30 | 14.1 | 7.6 | ||||
| Iso-butyrate | 33 | 1.7 | 1.2 | 30 | 1.8 | 0.9 | ||||
| Valerate | 33 | 2.3 | 1.1 | 30 | 1.9 | 0.7 | ||||
| Iso-valerate | 33 | 2.8 | 2 | 30 | 2.7 | 2.1 | ||||
| BMI (kg/m2) | >30 | <25 | ||||||||
| Dugas, | F | Total SCFA | Feces | 21 | 5.48 | 1.35 | 29 | 5.09 | 2.19 | GC/MC |
| 2018, Ghana | 25–45 | Acetate | µg/mL | 21 | 2.12 | 0.44 | 29 | 2.08 | 0.73 | |
| Propionate | 21 | 1.28 | 0.49 | 29 | 1.11 | 0.7 | ||||
| Butyrate | 21 | 1.79 | 0.8 | 29 | 1.65 | 0.89 | ||||
| Iso-butyrate | 21 | 0.09 | 0.04 | 29 | 0.12 | 0.06 | ||||
| BMI (kg/m2) | >30 | <25 | ||||||||
| Dugas, | Total SCFA | 37 | 3.76 | 1.94 | 13 | 3.01 | 1.13 | |||
| 2018, USA | Acetate | 37 | 1.6 | 0.74 | 13 | 1.39 | 0.53 | |||
| Propionate | 37 | 0.71 | 0.43 | 13 | 0.54 | 0.22 | ||||
| Butyrate | 37 | 1.18 | 0.85 | 13 | 0.85 | 0.57 | ||||
| Iso-butyrate | 37 | 0.1 | 0.07 | 13 | 0.11 | 0.03 | ||||
| BMI (mean, kg/m2) | 36.6 | 22.6 | ||||||||
| Todescol, | M and F | Acetate | Blood | 8 | 3.6 | 1.4 | 7 | 2.9 | 0.7 | GC |
| 1993, Canada | O (33.4), | µmol/dL | ||||||||
| NO (29.0) | ||||||||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, K.N.; Yao, Y.; Ju, S.Y. Short Chain Fatty Acids and Fecal Microbiota Abundance in Humans with Obesity: A Systematic Review and Meta-Analysis. Nutrients 2019, 11, 2512. https://doi.org/10.3390/nu11102512
Kim KN, Yao Y, Ju SY. Short Chain Fatty Acids and Fecal Microbiota Abundance in Humans with Obesity: A Systematic Review and Meta-Analysis. Nutrients. 2019; 11(10):2512. https://doi.org/10.3390/nu11102512
Chicago/Turabian StyleKim, Kyu Nam, Yao Yao, and Sang Yhun Ju. 2019. "Short Chain Fatty Acids and Fecal Microbiota Abundance in Humans with Obesity: A Systematic Review and Meta-Analysis" Nutrients 11, no. 10: 2512. https://doi.org/10.3390/nu11102512
APA StyleKim, K. N., Yao, Y., & Ju, S. Y. (2019). Short Chain Fatty Acids and Fecal Microbiota Abundance in Humans with Obesity: A Systematic Review and Meta-Analysis. Nutrients, 11(10), 2512. https://doi.org/10.3390/nu11102512





