A New Calcium Oral Controlled-Release System Based on Zeolite for Prevention of Osteoporosis
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of Calcium-Containing Zeolite Granules
2.2. Study of Calcium Release from Granules
2.3. Characterization of Granules by ATR-FTIR Spectroscopy
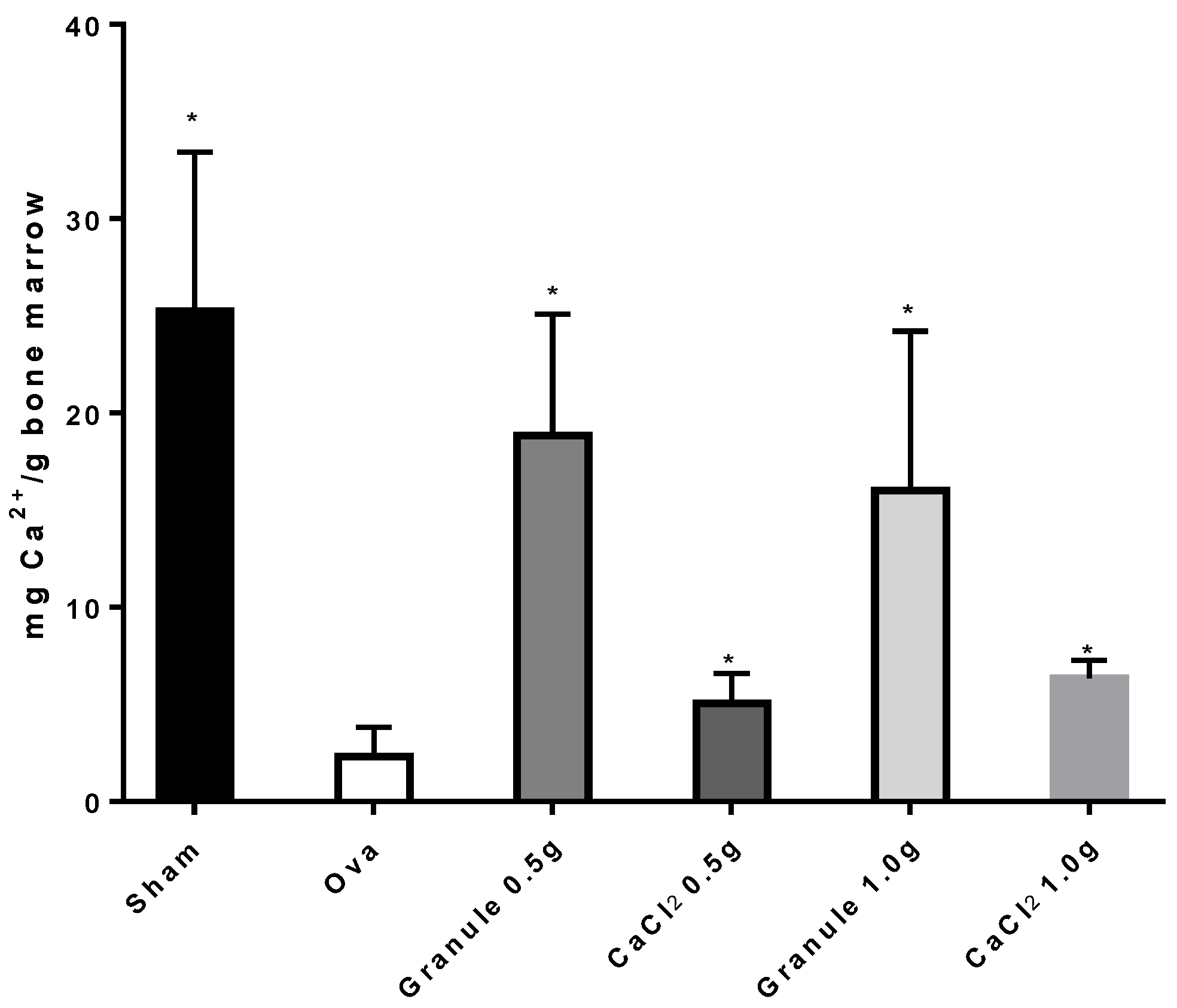
2.4. Experiments In Vivo
- Sham Group: nonovariectomized rats not treated with calcium formulations;
- Ova Group: ovariectomized rats not treated with calcium formulations;
- CaCl2 1.0 g Group: ovariectomized rats treated with 200 mg/Kg/die CaCl2, equivalent to a 1.0 g/die Ca2+ dose in humans;
- CaCl2 0.5 g Group: ovariectomized rats treated with 100 mg/Kg/die CaCl2, equivalent to a 0.5 g/die Ca2+ dose in humans;
- Granule 1.0 g Group: ovariectomized rats treated with 130 mg/Kg/die granules, equivalent to a 1.0 g/die Ca2+ dose in humans;
- Granule 0.5 g Group: ovariectomized rats treated with 65 mg/Kg/die granules, equivalent to a 0.5 g/die Ca2+ dose in humans.
2.5. Determination of Calcium Concentration in Bone and Bone Marrow
2.6. Rat Femur Mechanical Characterization
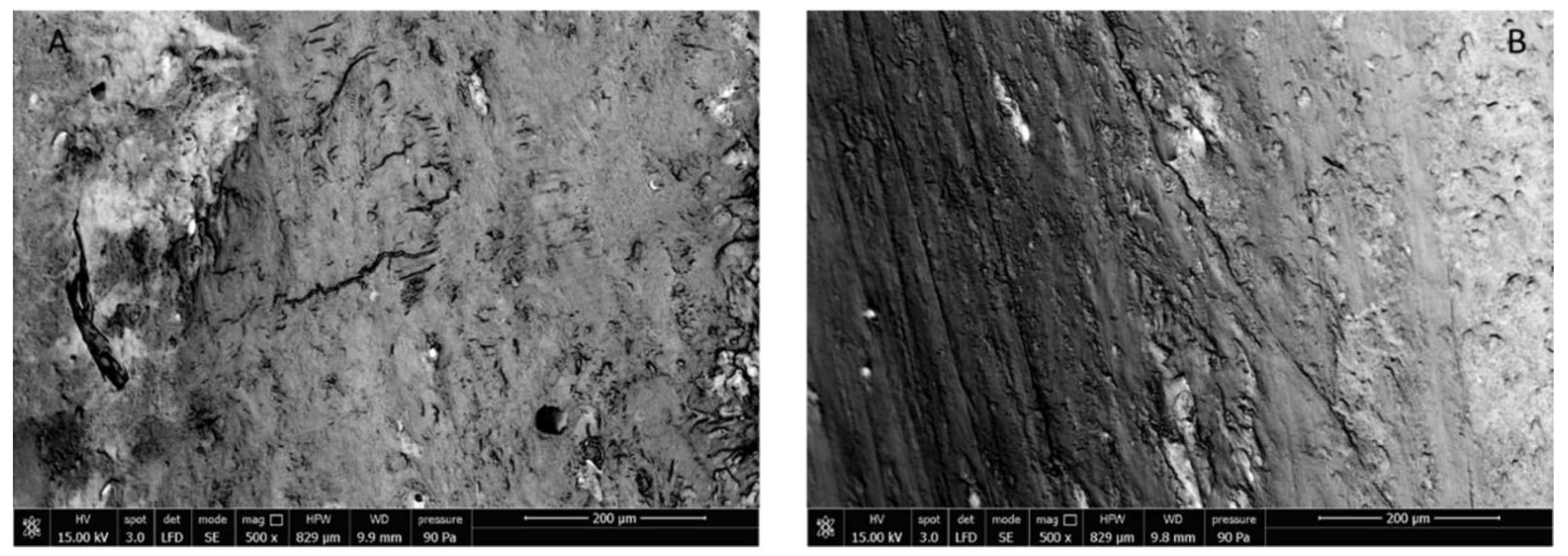
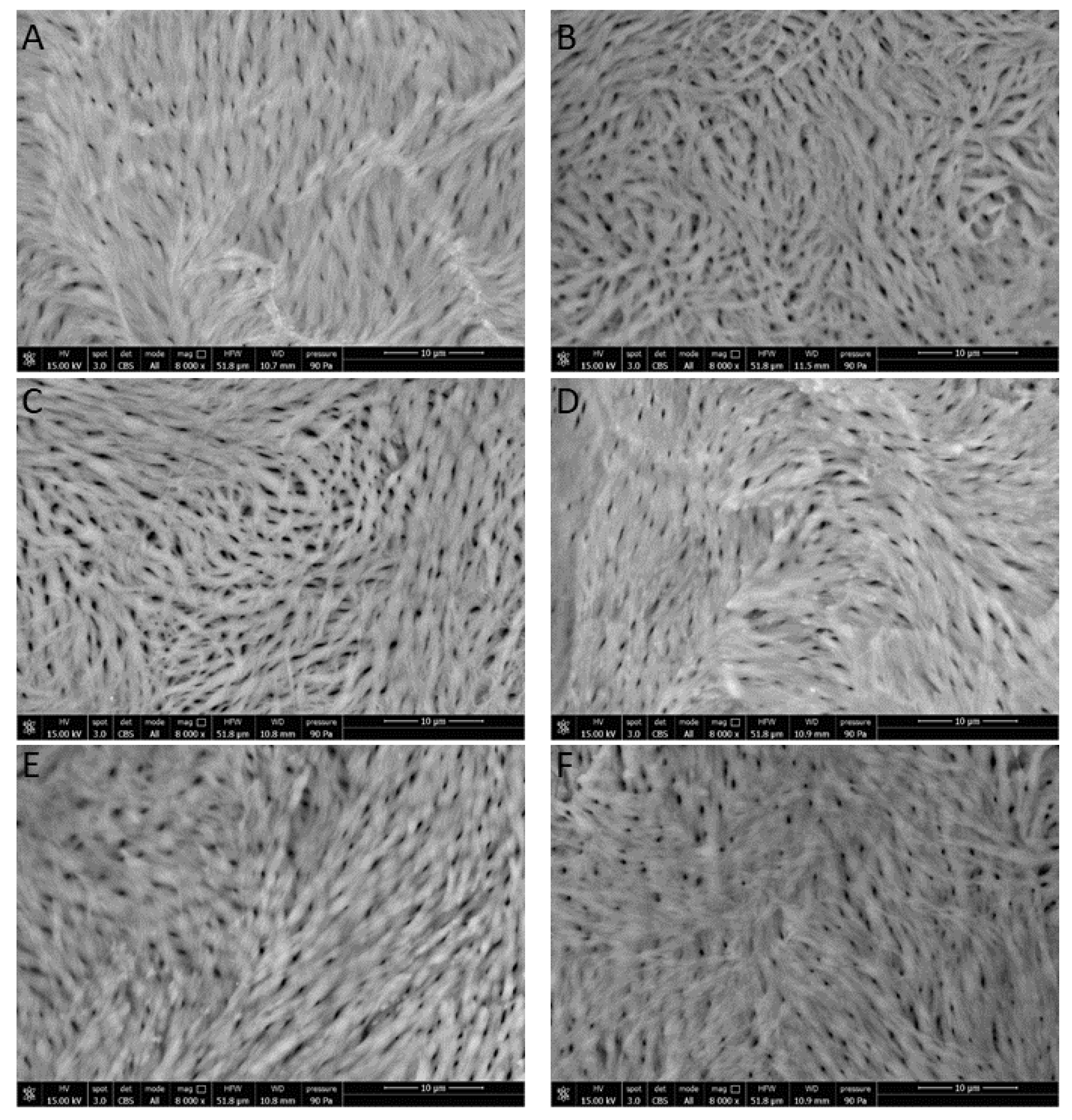
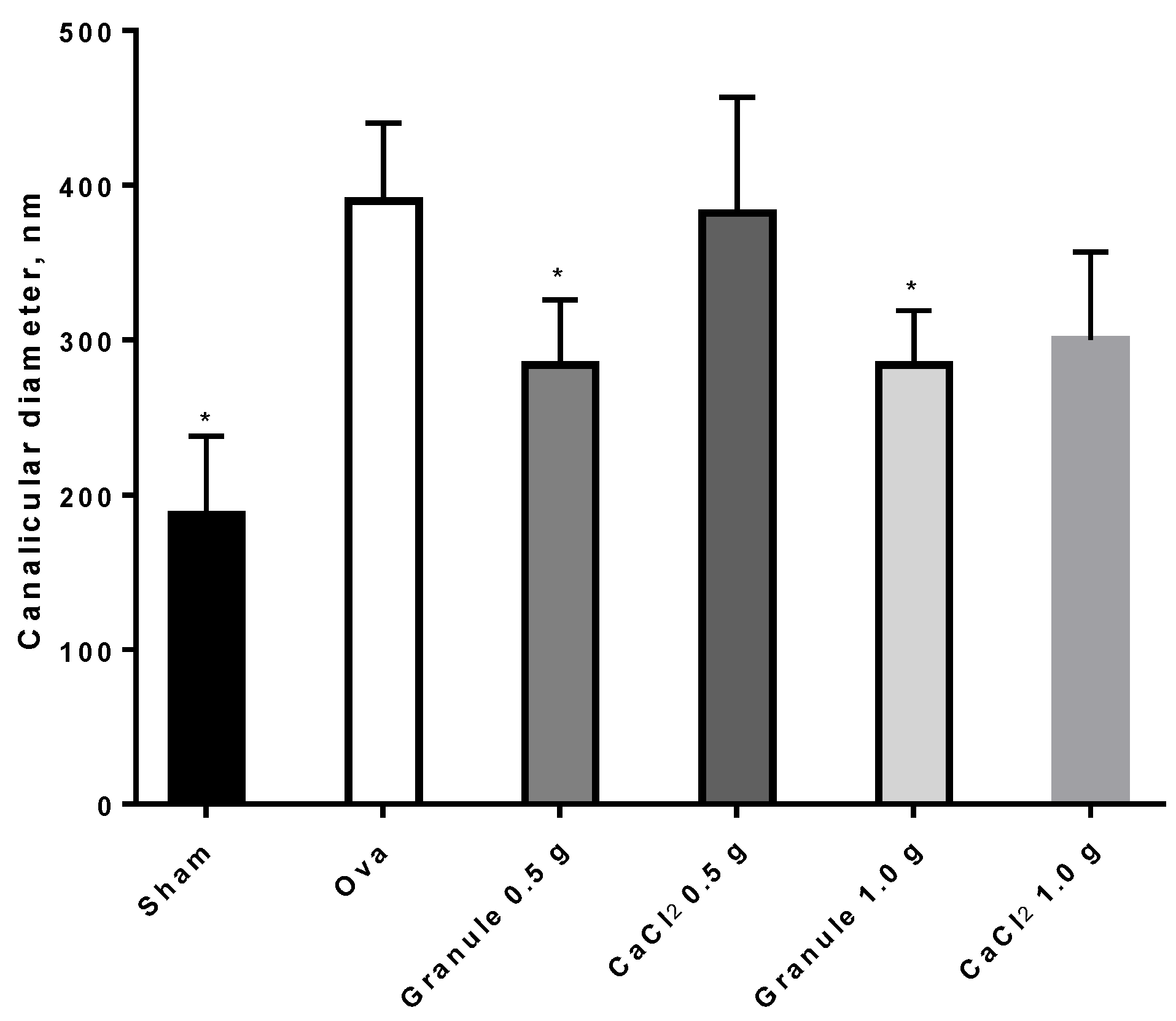
2.7. Rat Femur Morphological Characterization
2.8. Statistical Analysis
3. Results and Discussion
3.1. Calcium Release from Granules
3.2. Granule Characterization by ATR-FTIR Spectroscopy
3.3. Experiments In Vivo
3.3.1. Determination of Calcium Concentration in Rat Bone and Bone Marrow
3.3.2. Rat femur Mechanical Characterization
3.3.3. Rat Femur Morphological Characterization
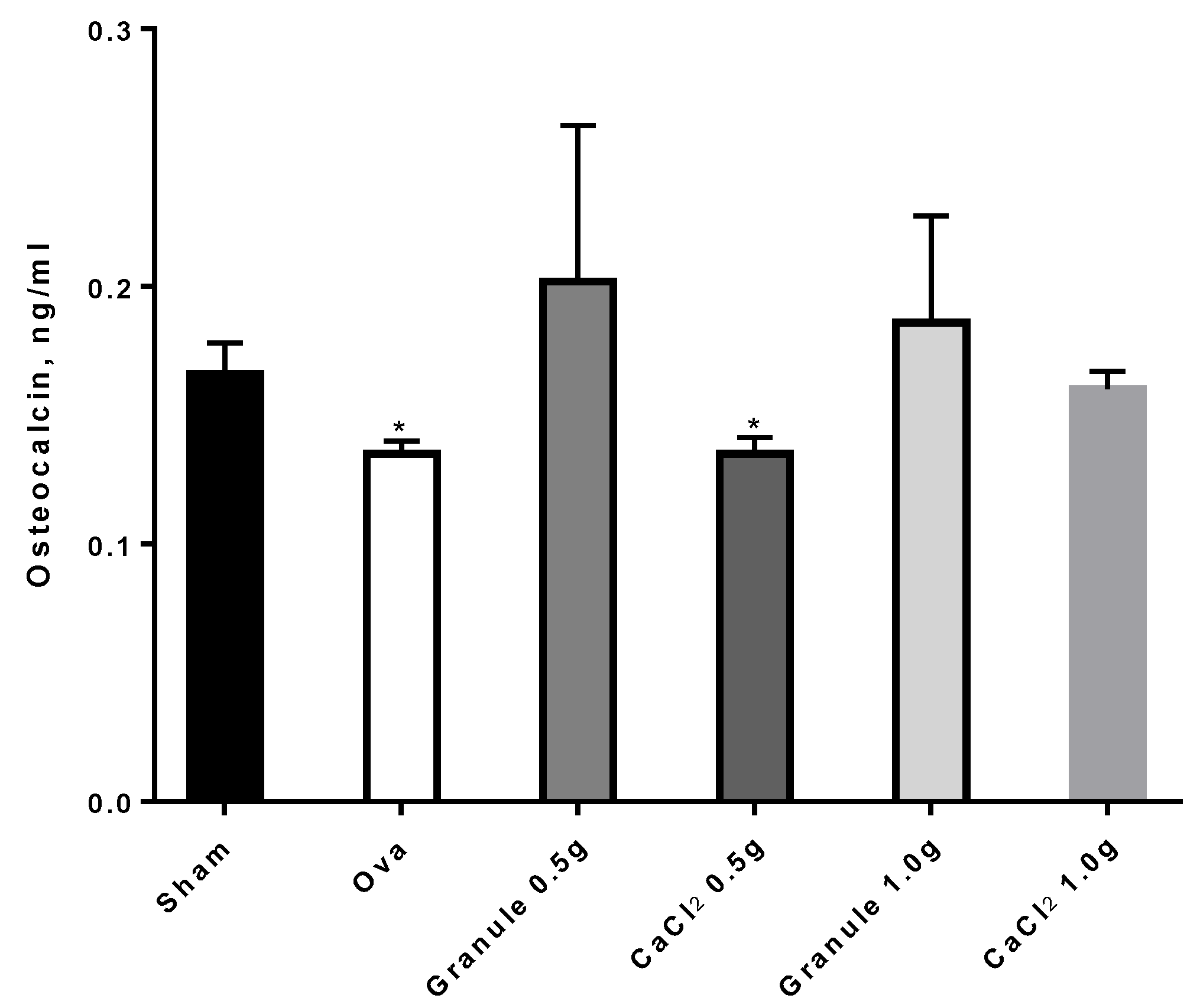
3.3.4. Plasmatic Marker of Osteopenia/Osteoporosis
4. Conclusions
5. Patents
Author Contributions
Funding
Conflicts of Interest
References
- Consensus Development Conference. Diagnosis, prophylaxis and treatment of osteoporosis. Am. J. Med. 1993, 94, 646–650. [Google Scholar] [CrossRef]
- Consensus Development Statement. Who are candidates for prevention and treatment for osteoporosis? Osteop. Int. 1997, 7, 1–6. [Google Scholar] [CrossRef]
- Osteoporosis Prevention, Diagnosis, and Therapy. Available online: https://jamanetwork.com/journals/jama/article-abstract/193534 (accessed on 12 October 2019).
- Black Sandler, R.; Slemenda, C.W.; LaPorte, R.E.; Cauley, J.A.; Schramm, M.M.; Barresi, M.L.; Kriska, A.M. Postemenopausal bone density and milk consumption in childhood and adolescence. Am. J. Clin. Nutr. 1985, 42, 270–274. [Google Scholar] [CrossRef] [PubMed]
- Johnston, C.C., Jr.; Miller, J.Z.; Slemenda, C.W.; Reister, T.K.; Hui, S.; Christian, J.C.; Peacock, M. Calcium supplementation and increases in bone mineral density in children. N. Engl. J. Med. 1992, 327, 82–85. [Google Scholar] [CrossRef] [PubMed]
- Weaver, C.M. Calcium requirements of physically active people. Am. J. Clin. Nutr. 2000, 72, 579S–584S. [Google Scholar] [CrossRef]
- Bronner, E.; Pansu, D.; Stein, W.D. An analysis of intestinal calcium transportacross the rat intestine. Am. J. Phys. 1986, 250, G561–G569. [Google Scholar] [CrossRef]
- Duflos, C.; Bellaton, C.; Pansu, D.; Bronner, F. Calcium solubility, intestinal sojourn time and paracellular permeability codetermine passive calcium absorption in rats. J. Nutr. 1995, 125, 2348–2355. [Google Scholar] [CrossRef]
- Hansen, C.; Werner, E.; Erbes, H.J.; Larrat, V.; Kaltwasser, J.P. Intestinal calcium absorption from different calcium preparations: Influence of anion and solubility. Osteoporos Int. 1996, 6, 386–393. [Google Scholar] [CrossRef]
- Heaney, R.P.; Recker, R.R.; Weaver, C.M. Absorbability of calcium sources – the limited role of solubility. Calcif Tissue Int. 1990, 46, 300–304. [Google Scholar] [CrossRef]
- Roth-Bassell, H.A.; Clydesdale, F.M. In vitro solubility characteristic of six calcium salts. J. Food Prot. 1992, 55, 1003–1005. [Google Scholar] [CrossRef]
- Vavrusova, M.; Skibsted, L. Calcium nutrition. Bioavaiability and fortification. LWT Food Sci. Technol. 2014, 59, 1198–1204. [Google Scholar] [CrossRef]
- Krajišnik, D.; Daković, A.; Janićijević, J.; Milić, J. Natural and Modified Silica-Based Materials as Carriers for NSAIDs. Microsized and Nanosized Carriers for Nonsteroidal Anti-Inflammatory Drugs: Formulation Challenges and Potential Benefits. Acad. Press 2017, 219–258. [Google Scholar] [CrossRef]
- Serri, C.; De Gennaro, B.; Quagliariello, V.; Iaffaioli, R.V.; De Rosa, G.; Catalanotti, L.; Biondi, M.; Mayol, L. Surface modified zeolite-based granulates for the sustained release of diclofenac sodium. Eur. J. Pharm. Sci. 2017, 99, 202–208. [Google Scholar] [CrossRef] [PubMed]
- Youssef, H.F.; Abdel-Aziz, M.S.; Fouda, F.K. Evaluation of antimicrobial activity of different silver-exchanged nano and micronized zeolites prepared by microwave technique. J. Porous Mater. 2017, 24, 947–957. [Google Scholar] [CrossRef]
- Prasai, T.P.; Walsh, K.B.; Bhattarai, S.P.; Midmore, D.J.; Van, T.T.H.; Moore, R.J.; Stanley, D. Zeolite food supplementation reduces abundance of enterobacteria. Microbiol Res. 2017, 195, 24–30. [Google Scholar] [CrossRef]
- Oral Controlled-Release Calcium Compound and Method for Preparing It. Available online: https://patents.google.com/patent/EP3482745A1/en (accessed on 12 October 2019).
- Di Colo, G.; Baggiani, A.; Zambito, Y.; Mollica, G.; Geppi, M.; Serafini, M.F. A new hydrogel for the extended and complete prednisolone release in the GI tract. Int. J. Pharm. 2006, 310, 154–161. [Google Scholar] [CrossRef]
- Grynkiewicz, G.; Poenie, M.; Tsien, R.Y. A new generation of Ca2+ indicators with greatly improved fluorescence properties. J. Biol. Chem. 1985, 260, 3440–3450. [Google Scholar]
- McGrath, J.C.; Lilley, E. Implementing guidelines on reporting research using animals (ARRIVE etc.): New requirements for publication in BJP. Br. J. Pharmacol. 2015, 172, 3189–3193. [Google Scholar] [CrossRef]
- Erfanian, A.; Mirhosseini, H.; Rasti, B.; Hair-Bejo, M.; Bin Mustafa, S.; Abd Manap, M.Y. Absorption and Bioavailability of Nano-Size Reduced Calcium Citrate Fortified Milk Powder in Ovariectomized and Ovariectomized-Osteoporosis Rats. J. Agric. Food Chem. 2015, 63, 5795–5804. [Google Scholar] [CrossRef]
- Zafar, T.A.; Weaver, C.M.; Zhao, Y.; Martin, B.R.; Wastney, M.E. Nondigestible oligosaccharides increase calcium absorption and suppress bone resorption in ovariectomized rats. J. Nutr. 2004, 134, 399–402. [Google Scholar] [CrossRef]
- Tozaki, H.; Odoriba, T.; Okada, N.; Fujita, T.; Terabe, A.; Suzuki, T.; Okabe, S.; Muranishi, S.; Yamamoto, A. Chitosan capsules for colon-specific drug delivery: Enhanced localization of 5-aminosalicylic acid in the large intestine accelerates healing of TNBS-induced colitis in rats. J. Control. Release 2002, 82, 51–61. [Google Scholar] [CrossRef]
- Fabiano, A.; Brilli, E.; Mattii, L.; Testai, L.; Moscato, S.; Citi, V.; Tarantino, G.; Zambito, Y. Ex vivo and in vivo study of sucrosomial® iron intestinal absorption and bioavailability. Int. J. Mol. Sci. 2018, 19. [Google Scholar] [CrossRef] [PubMed]
- Erfanian, A.; Rasti, B.; Manap, Y. Comparing the calcium bioavailability from two types of nano-sized enriched milk using in-vivo assay. Food Chem. 2017, 214, 606–613. [Google Scholar] [CrossRef] [PubMed]
- Okano, T.; Kimura, T.; Tsugawa, N.; Fujiwara, M.; Yamamoto, M.; Kobayashi, T. Bioavailability of calcium from bovine-bone-marrow calcium (BBMCa) and calcium carbonate in vitamin D-deficient rats. Food Chem. 1994, 51, 61–67. [Google Scholar] [CrossRef]
- Bottini, M.; Mebarek, S.; Anderson, K.L.; Strzelecka-Kiliszek, A.; Bozycki, L.; Simão, A.M.S.; Bolean, M.; Ciancaglini, P.; Pikula, J.B.; Pikula, S.; et al. Matrix vesicles from chondrocytes and osteoblasts: Their biogenesis, properties, functions and biomimetic models. Biochim. Biophys Acta Gen. Subj. 2018, 1862, 532–546. [Google Scholar] [CrossRef]
- Cui, L.; Houston, D.A.; Farquharson, C.; MacRae, V.E. Characterisation of matrix vesicles in skeletal and soft tissue mineralisation. Bone 2016, 87, 147–158. [Google Scholar] [CrossRef]
- Hu, F.; Pan, L.; Zhang, K.; Xing, F.; Wang, X.; Lee, I.; Zhang, X.; Xu, J. Elevation of extracellular Ca2+ induces store-operated calcium entry via calcium-sensing receptors: A pathway contributes to the proliferation of osteoblasts. PLoS ONE 2014, 9, e107217. [Google Scholar] [CrossRef]
- Nollet, E.; Van Craenenbroeck, E.M.; Martinet, W.; Rodrigus, I.; De Bock, D.; Berneman, Z.; Pintelon, I.; Ysebaert, D.; Vrints, C.J.; Conraads, V.M.; et al. Bone matrix vesicle-bound alkaline phosphatase for the assessment of peripheral blood admixture to human bone marrow aspirates. Clin. Chim. Acta 2015, 446, 253–260. [Google Scholar] [CrossRef]
- Sharma, D.; Ciani, C.; Ramirez Marin, P.A.; Levy, J.D.; Doty, S.B.; Fritton, S.P. Alterations in the Osteocyte Lacunar-Canalicular Microenvironment due to Estrogen Deficiency. Bone 2012, 51, 488–497. [Google Scholar] [CrossRef]
- Bailey, S.; Karsenty, G.; Gundberg, C.; Vashishth, D. Osteocalcin and osteopontin influence bone morphology and mechanical properties. Ann. N. Y. Acad. Sci. 2017, 1409, 79–84. [Google Scholar] [CrossRef]
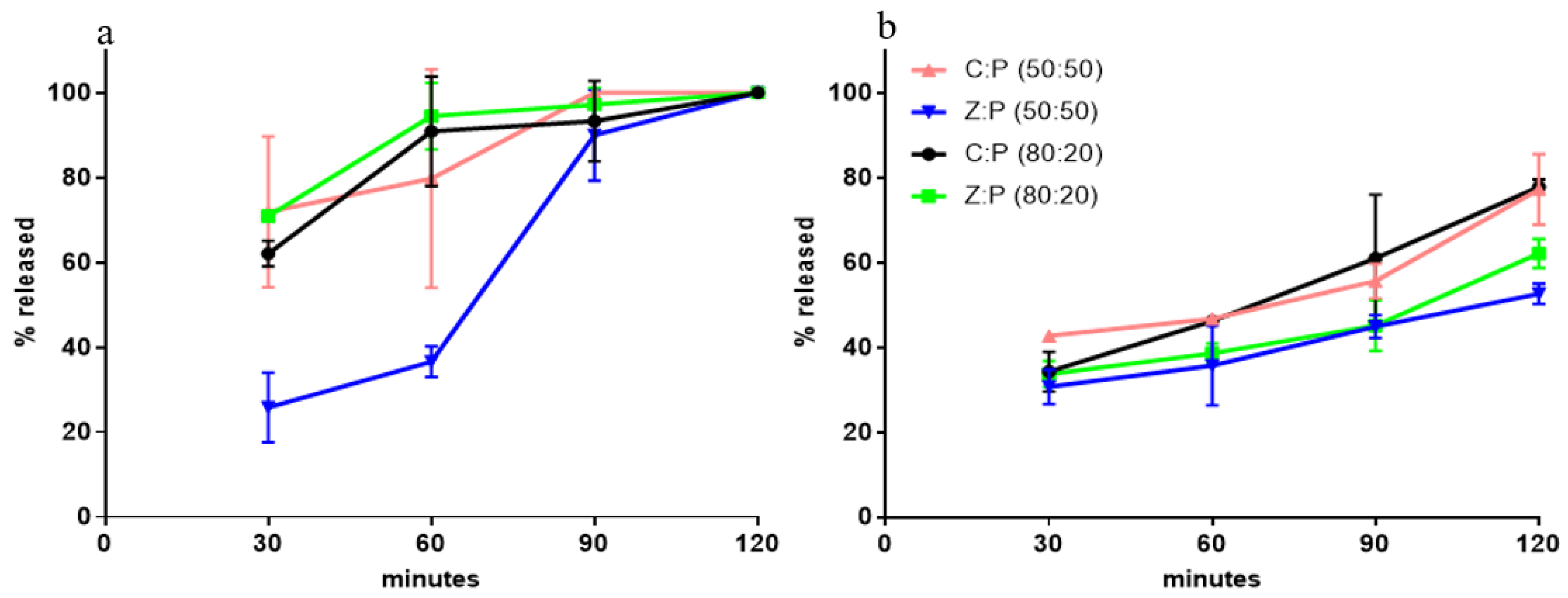
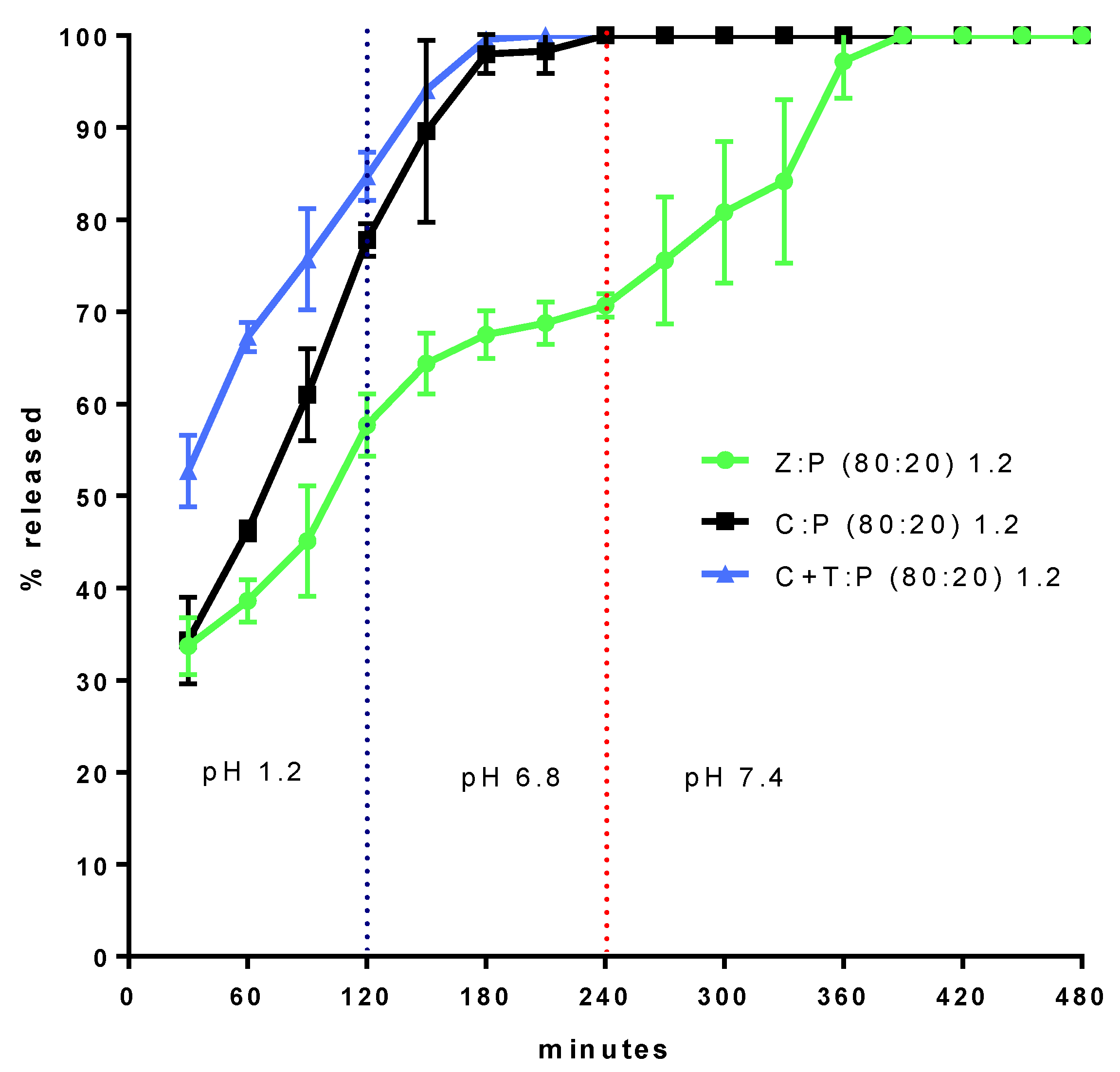
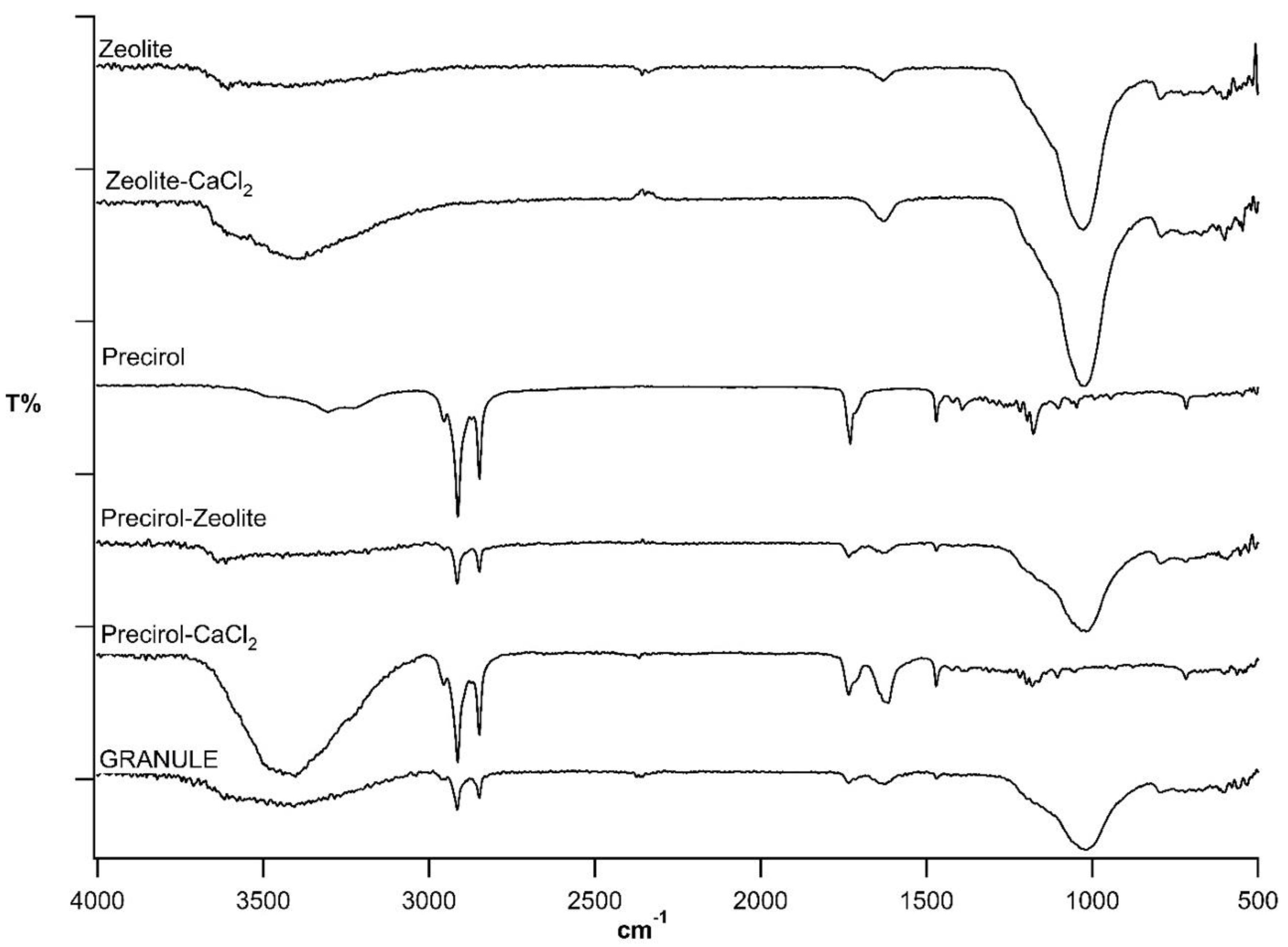
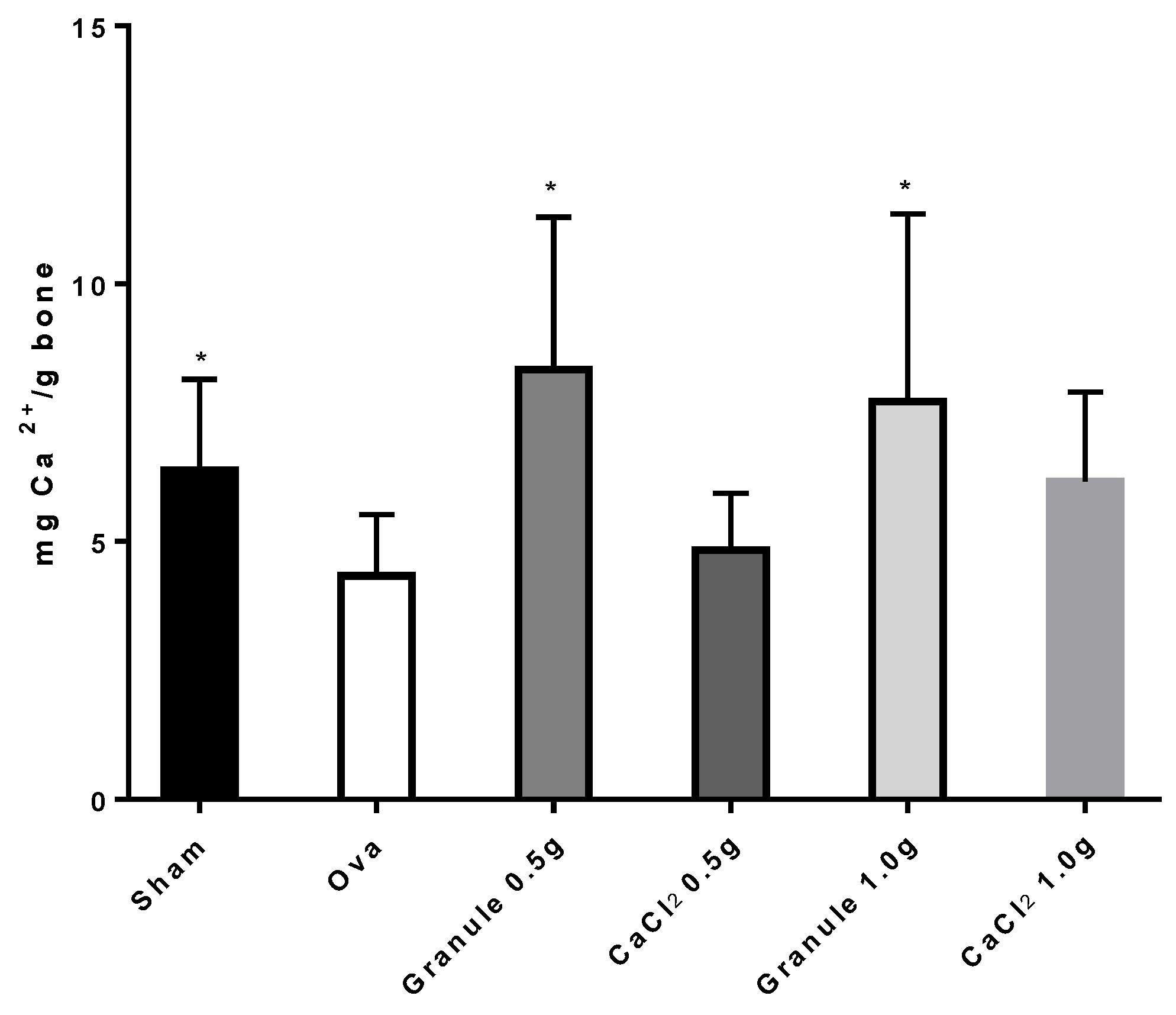
| Code | Precirol % | Ca2+ % | Zeolite % | Talc % | Granule Size (mm) |
|---|---|---|---|---|---|
| Z:P (80:20) 0.9 | 20 | 61 | 19 | 0.9 | |
| Z:P (50:50) 0.9 | 50 | 37.8 | 12.2 | 0.9 | |
| C:P (80:20) 0.9 | 20 | 80 | 0.9 | ||
| C:P (50:50) 0.9 | 50 | 50 | 0.9 | ||
| Z:P (80:20) 1.2 a | 20 | 61 | 19 | 1.2 | |
| Z:P (50:50) 1.2 | 50 | 37.8 | 12.2 | 1.2 | |
| C:P (80:20) 1.2 | 20 | 80 | 1.2 | ||
| C:P (50:50) 1.2 | 50 | 50 | 1.2 | ||
| (C+T):P (80:20) 1.2 | 20 | 61 | 19 | 1.2 |
| Components | Hydration H2O | νas C–H, νs C–H | νs C=O | Hydration H2O | δas C–H | δs C–H | Silicate νas e νs Si–O | |||
|---|---|---|---|---|---|---|---|---|---|---|
| Zeolite | 3600 | 1632 | 1027 | 795 | ||||||
| Zeolite CaCl2 | 3400 | 1631 | 1027 | 795 | ||||||
| Precirol® | 3304 | 2955 | 2913 | 2848 | 1730 | 1471 | 1392 | |||
| Precirol® Zeolite | 3600 | 2955 | 2915 | 2849 | 1734 | 1634 | 1471 | 1392 | 1027 | 795 |
| Precirol® CaCl2 | 3403 | 2956 | 2914 | 2849 | 1734 | 1623 | 1471 | 1392 | ||
| GRANULE | 3400 | 2960 | 2916 | 2848 | 1735 | 1626 | 1470 | 1396 | 1020 | 798 |
| Rat Groups | Stiffness (N/mm) | Load Max (N) |
|---|---|---|
| Sham | 159 ± 17 | 107 ± 6 |
| Ova | 113 ± 24 | 93 ± 18 |
| Granule 0.5 g | 136 ± 38 | 106 ± 9 |
| CaCl2 0.5 g | 118 ± 51 | 101 ± 13 |
| Granule 1.0 g | 136 ± 19 | 101 ± 7 |
| CaCl2 1.0 g | 128 ± 11 | 93 ± 11 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fabiano, A.; Piras, A.M.; Calderone, V.; Testai, L.; Flori, L.; Puppi, D.; Chiellini, F.; Zambito, Y. A New Calcium Oral Controlled-Release System Based on Zeolite for Prevention of Osteoporosis. Nutrients 2019, 11, 2467. https://doi.org/10.3390/nu11102467
Fabiano A, Piras AM, Calderone V, Testai L, Flori L, Puppi D, Chiellini F, Zambito Y. A New Calcium Oral Controlled-Release System Based on Zeolite for Prevention of Osteoporosis. Nutrients. 2019; 11(10):2467. https://doi.org/10.3390/nu11102467
Chicago/Turabian StyleFabiano, Angela, Anna Maria Piras, Vincenzo Calderone, Lara Testai, Lorenzo Flori, Dario Puppi, Federica Chiellini, and Ylenia Zambito. 2019. "A New Calcium Oral Controlled-Release System Based on Zeolite for Prevention of Osteoporosis" Nutrients 11, no. 10: 2467. https://doi.org/10.3390/nu11102467
APA StyleFabiano, A., Piras, A. M., Calderone, V., Testai, L., Flori, L., Puppi, D., Chiellini, F., & Zambito, Y. (2019). A New Calcium Oral Controlled-Release System Based on Zeolite for Prevention of Osteoporosis. Nutrients, 11(10), 2467. https://doi.org/10.3390/nu11102467












