Sodium Intake from Foods Exceeds Recommended Limits in the Spanish Population: The ANIBES Study
Abstract
1. Introduction
2. Materials and Methods
3. Results
4. Discussion
4.1. Sodium Intakes by Gender and Age, Total and Per 1000 kcal
4.2. Sodium Intakes from Foods (Excluding Added Salt) Consumed at Different Eating Occasions by Age Groups
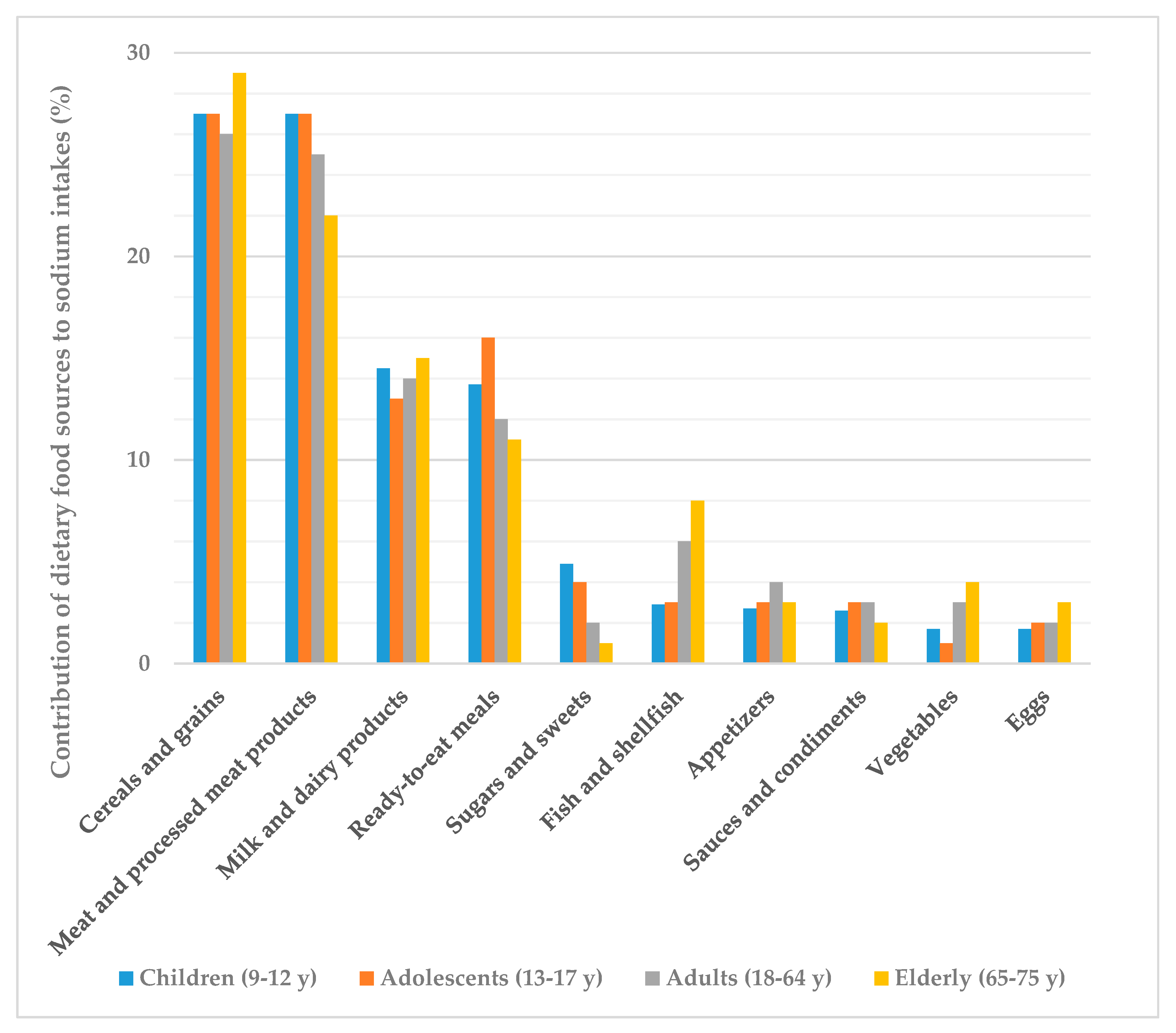
4.3. Contribution of Dietary Food Sources (Excluding Added Salt) to the ANIBES Population Daily Sodium Intakes: Groups and Subgroups by Age Group
4.4. Sodium Intakes According to Body Composition
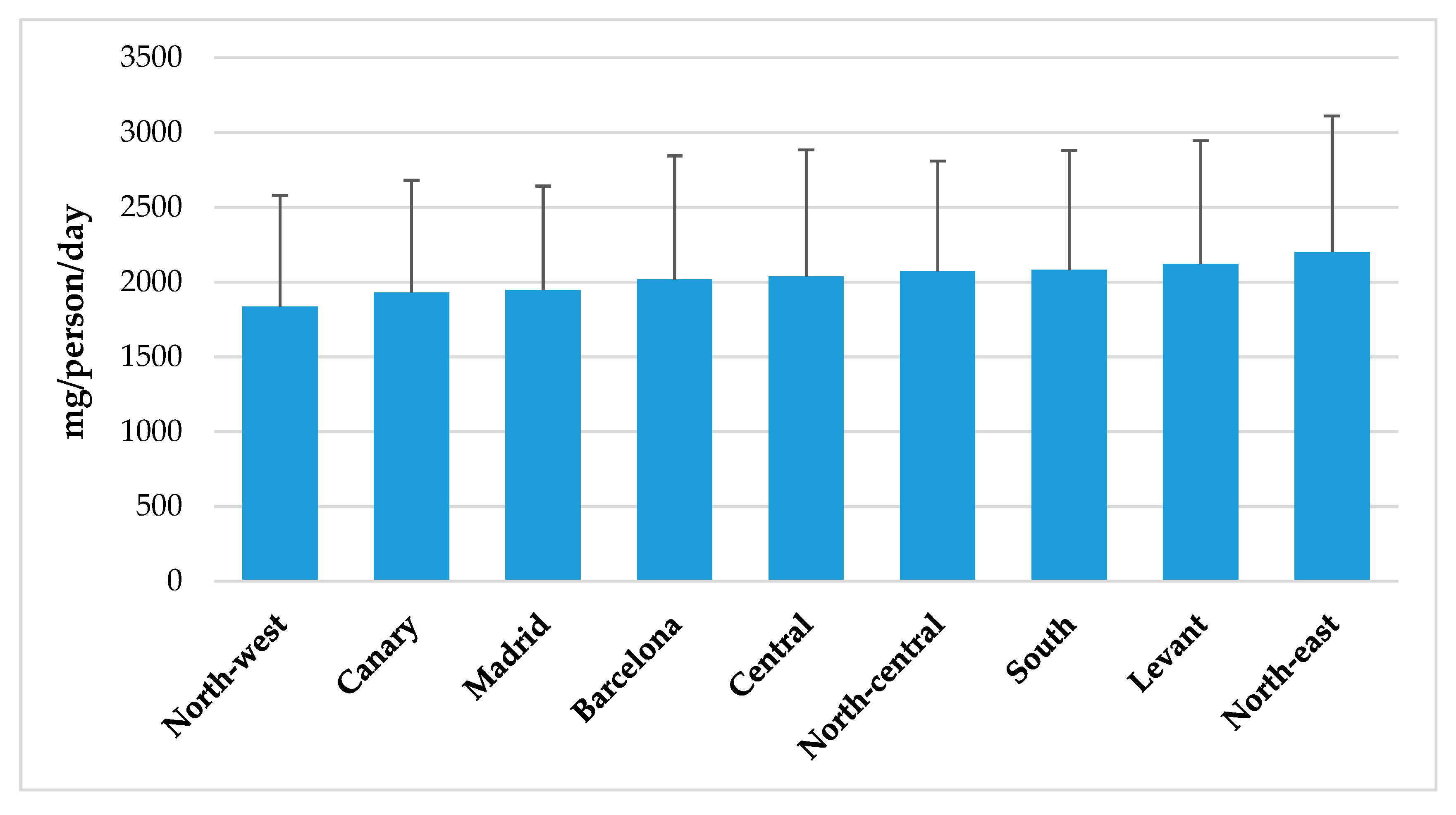
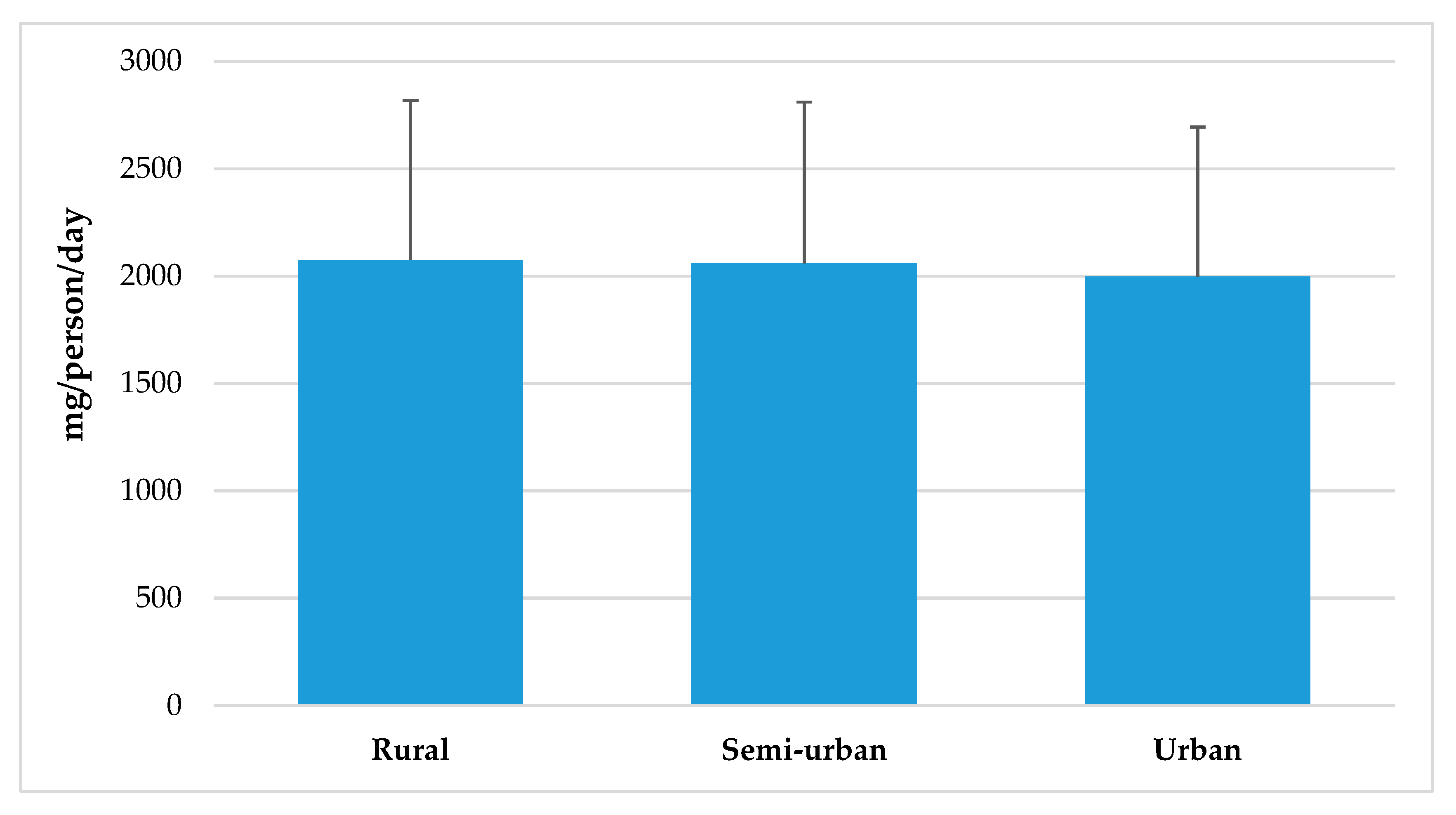
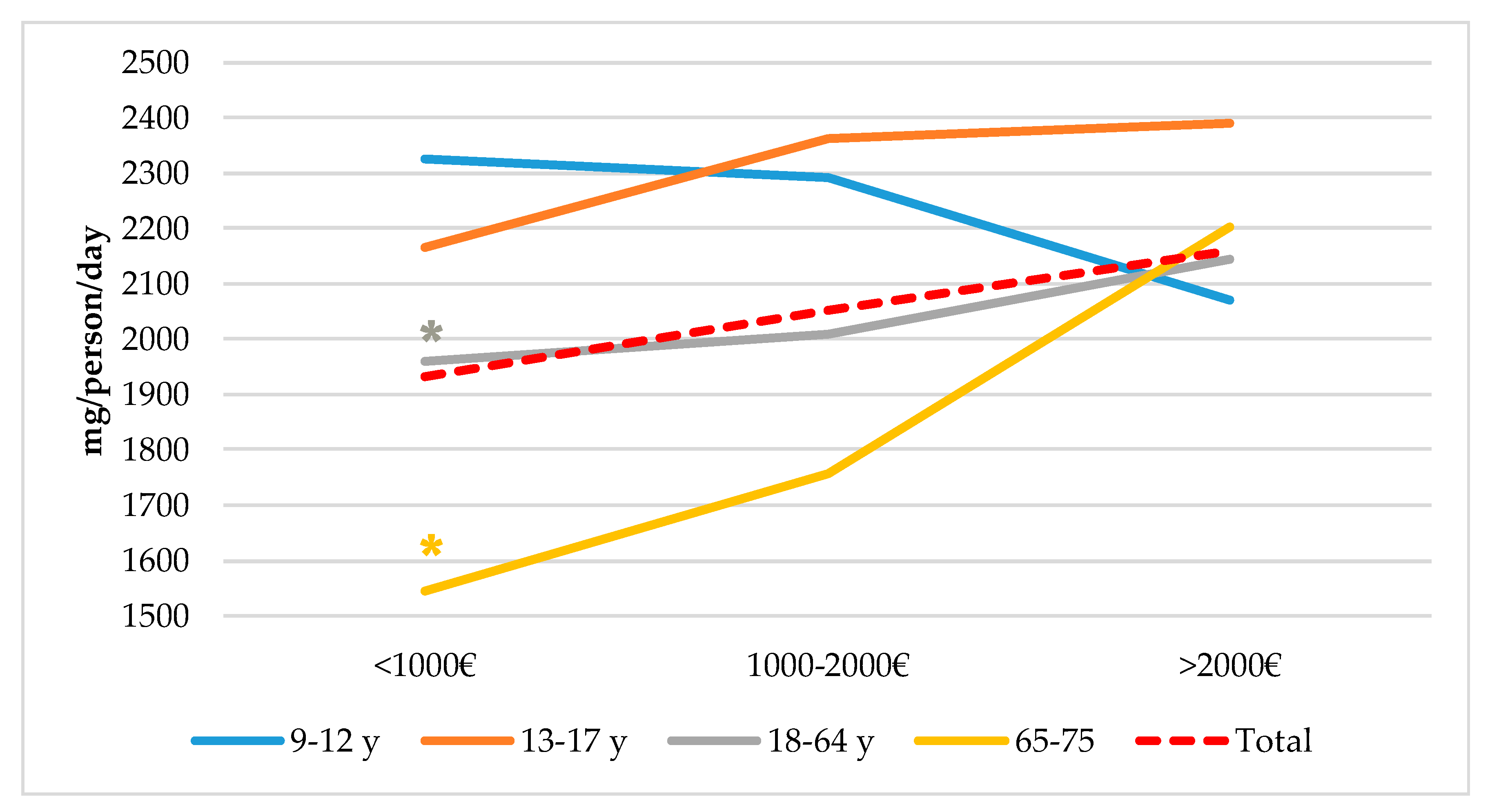
4.5. Sodium Intake from Total Population Segmented by Spanish Nielsen Areas, Habitat Size and by Income Level and Age Groups from the ANIBES Study Population
4.6. Strengths and Limitations
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lloyd-Jones, D.M.; Hong, Y.; Labarthe, D.; Mozaffarian, D.; Appel, L.J.; Horn, L.V.; Greenlund, K.; Daniels, S.; Nichol, G.; Tomaselli, G.F. Defining and Setting National Goals for Cardiovascular Health Promotion and Disease Reduction. Circulation 2010, 121, 586–613. [Google Scholar] [CrossRef] [PubMed]
- Benjamin, E.J.; Muntner, P.; Alonso, A.; Bittencourt, M.S.; Callaway, C.W.; Carson, A.P. Heart Disease and Stroke Statistics-2019 Update: A Report from the American Heart Association. Circulation 2019, 139, e56–e528. [Google Scholar] [CrossRef] [PubMed]
- Stanaway, J.D.; Afshin, A.; Gakidou, E.; Lim, S.S.; Abate, D.; Abate, K.H.; Abdela, J. Global, regional, and national comparative risk assessment of 84 behavioural, environmental and occupational, and metabolic risks or clusters of risks for 195 countries and territories, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet 2018, 392, 1923–1994. [Google Scholar] [CrossRef]
- Bifari, F.; Berton, V.; Pino, A.; Kusalo, M.; Malpeli, G.; Di Chio, M.; Bersan, E.; Amato, E.; Scarpa, A.; Krampera, M. Meninges harbor cells expressing neural precursor markers during development and adulthood. Front. Cell. Neurosci. 2015, 9, 383. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. European Food and Nutrition Action Plan 2015–2020; WHO Regional office for Europe: Copenhagen, Denmark, 2014. [Google Scholar]
- Afshin, A.; Sur, P.J.; Fay, K.A.; Cornaby, L.; Ferrara, G.; Salama, J.S.; Afarideh, M. Health effects of dietary risks in 195 countries, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet 2019, 393, 1958–1972. [Google Scholar] [CrossRef]
- Mozaffarian, D.; Fahimi, S.; Singh, G.M.; Micha, R.; Khatibzadeh, S.; Engell, R.E.; Lim, S.; Danae, G.; Ezzati, M.; Powles, J. Global sodium consumption and death from cardiovascular causes. N. Engl. J. Med. 2014, 371, 624–634. [Google Scholar] [CrossRef] [PubMed]
- Sacks, F.M.; Svetkey, L.P.; Vollmer, W.M.; Appel, L.J.; Bray, G.A.; Harsha, D.; Obarzanek, E.; Conlin, P.R.; Miller, E.R.; Simons-Morton, D.G.; et al. Effects on blood pressure of reduced dietary sodium and the Dietary Approaches to Stop Hypertension (DASH) diet. N. Engl. J. Med. 2001, 344, 3–10. [Google Scholar] [CrossRef]
- Intersalt Cooperative Research Group. Intersalt: An international study of electrolyte excretion and blood pressure. Results for 24 h urinary sodium and potassium excretion. BMJ 1988, 297, 319–328. [Google Scholar] [CrossRef]
- Midgley, J.P.; Matthew, A.G.; Greenwood, C.M.T.; Logan, A.G. Effect of reduced dietary sodium on blood pressure: A meta-analysis of randomized controlled trials. Jama 1996, 275, 1590–1597. [Google Scholar] [CrossRef]
- Ndanuko, R.N.; Tapsell, L.C.; Charlton, K.E.; Neale, E.P.; Batterham, M.J. Dietary patterns and blood pressure in adults: A systematic review and meta-analysis of randomized controlled trials. Adv. Nutr. 2016, 7, 76–89. [Google Scholar] [CrossRef]
- Farquhar, W.B.; Edwards, D.G.; Jurkovitz, C.T.; Weintraub, W.S. Dietary sodium and health: More than just blood pressure. J. Am. Coll. Cardiol. 2015, 65, 1042–1050. [Google Scholar] [CrossRef] [PubMed]
- Kotchen, T.A.; Cowley, A.W.; Frohlich, E.D. Salt in Health and Disease—A Delicate Balance. N. Engl. J. Med. 2013, 368, 1229–1237. [Google Scholar] [CrossRef] [PubMed]
- Appel, L.J.; Frohlich, E.D.; Hall, J.E.; Pearson, T.A.; Sacco, R.L.; Seals, D.R.; Sacks, F.M.; Smith, S.C., Jr.; Vafiadis, D.K.; Van Horn, L.V. The Importance of Population-Wide Sodium Reduction as a Means to Prevent Cardiovascular Disease and Stroke. Circulation 2011, 123, 1138–1143. [Google Scholar] [CrossRef] [PubMed]
- He, F.J.; Campbell, N.R.; MacGregor, G.A. Reducing salt intake to prevent hypertension and cardiovascular disease. Rev. Panam. Salud Públ. 2012, 32, 293–300. [Google Scholar] [CrossRef] [PubMed]
- Widmaier, E.P.; Raff, H.; Strang, K.T. Vander’s Human Physiology: The Mechanisms of Body Function; McGraw-Hill Higher Education: Boston, MA, USA, 2008. [Google Scholar]
- Taormina, P.J. Implications of Salt and Sodium Reduction on Microbial Food Safety. Crit. Rev. Food Sci. Nutr. 2010, 50, 209–227. [Google Scholar] [CrossRef] [PubMed]
- Hasenegger, V.; Rust, P.; König, J.; Purtscher, A.E.; Erler, J.; Ekmekcioglu, C. Main Sources, Socio-Demographic and Anthropometric Correlates of Salt Intake in Austria. Nutrients 2018, 10, 311. [Google Scholar] [CrossRef]
- World Health Organization Regional Office for Europe. Mapping Salt Reduction Initiatives in the WHO European Region 2013. Available online: http://www.euro.who.int/__data/assets/pdf_file/0009/186462/Mapping-salt-reduction-initiatives-in-the-WHO-European-Region.pdf (accessed on 3 January 2018).
- Monteiro, C.A.; Cannon, G.; Moubarac, J.C.; Levy, R.B.; Louzada, M.L.C.; Jaime, P.C. The UN Decade of Nutrition, the NOVA food classification and the trouble with ultra-processing. Public Health Nutr. 2017, 21, 5–17. [Google Scholar] [CrossRef]
- James, W.P.; Ralph, A.; Sanchez-Castillo, C. The dominance of salt in manufactured food in the sodium intake of affluent societies. Lancet 1987, 329, 426–429. [Google Scholar] [CrossRef]
- Brown, I.J.; Tzoulaki, I.; Candeias, V.; Elliott, P. Salt intakes around the world: Implications for public health. Int. J. Epidemiol. 2009, 38, 791–813. [Google Scholar] [CrossRef]
- Necchi, D.; Scherini, E. The malformation of the cerebellar fissura prima: A tool for studying histogenetic processes. Cerebellum 2002, 1, 137–142. [Google Scholar] [CrossRef]
- McLean, R. Measuring population sodium intake: A review of methods. Nutrients 2014, 6, 4651–4662. [Google Scholar] [CrossRef] [PubMed]
- Powles, J.; Fahimi, S.; Micha, R.; Khatibzadeh, S.; Shi, P.; Ezzati, M.; Engell, R.E.; Lim, S.S.; Danaei, G.; Mozaffarian, D. Global, regional and national sodium intakes in 1990 and 2010: A systematic analysis of 24 h urinary sodium excretion and dietary surveys worldwide. BMJ Open 2013, 3, e003733. [Google Scholar] [CrossRef] [PubMed]
- Ortega, R.M.; López-Sobaler, A.M.; Ballesteros, J.M.; Pérez-Farinós, N.; Rodríguez-Rodríguez, E.; Aparicio, A.; Perea, J.M.; Andrés, P. Estimation of salt intake by 24 h urinary sodium excretion in a representative sample of Spanish adults. Br. J. Nutr. 2011, 105, 787–794. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. WHO Guideline: Sodium Intake for Adults and Children; Report, i-46; WHO Press: Geneva, Switzerland, 2012. [Google Scholar]
- World Health Organization. A Comprehensive Global Monitoring Framework including Indicators and a Set of Voluntary Global Targets for the Prevention and Control of Noncommunicable Diseases; World Health Organization: Geneva, Switzerland, 2012. [Google Scholar]
- Commission, E.E. National Salt Initiatives; Implementing the EU Framework for Salt Reduction Initiatives; European Commission Brussels: Brussels, Belgium, 2009. [Google Scholar]
- Stoughton, R.L.; del Cerro, M.; Walker, J.R.; Swarz, J.R. Presence of displaced neural elements within rat cerebellar fissures. Brain Res. 1978, 148, 15–29. [Google Scholar] [CrossRef]
- Yeung, C.H.C.; Gohil, P.; Rangan, A.M.; Flood, V.M.; Arcot, J.; Gill, T.P.; Louie, J.C.Y. Modelling of the impact of universal added sugar reduction through food reformulation. Sci. Rep. 2017, 7, 17392. [Google Scholar] [CrossRef]
- Ministry of Health, Social Services and Equality. Spanish Agency for Food Safety and Nutrition. Collaboration Plan for the Improvement of the Composition of Food and Beverages and Other Measures 2020. Available online: http://www.aecosan.msssi.gob.es/AECOSAN/docs/documentos/nutricion/Plan_Colaboracion_INGLES.pdf (accessed on 10 January 2018).
- Ruiz, E.; Ávila, J.; Castillo, A.; Valero, T.; del Pozo, S.; Rodriguez, P.; Bartrina, J.A.; Gil, A.; González-Gross, M.; Ortega, R.M.; et al. The ANIBES Study on Energy Balance in Spain: Design, Protocol and Methodology. Nutrients 2015, 7, 970–998. [Google Scholar] [CrossRef]
- Ruiz, E.; Ávila, J.M.; Valero, T.; Del Pozo, S.; Rodriguez, P.; Aranceta-Bartrina, J.; Gil, A.; González-Gross, M.; Ortega, R.M.; Serra-Majem, L.; et al. Energy intake, profile, and dietary sources in the Spanish population: Findings of the ANIBES study. Nutrients 2015, 7, 4739–4762. [Google Scholar] [CrossRef]
- Moreiras, O.; Carbajal, Á.; Cabrera, L.; Cuadrado, C. Tablas de Composición de Alimentos 16. a Ed; Pirámide: Madrid, Spain, 2013. [Google Scholar]
- Fernández, C.; Lorenzo, H.; Vrotsou, K.; Aresti, U.; Rica, I.; Sánchez, E. Estudio de Crecimiento de Bilbao: Curvas y Tablas de Crecimiento (Estudio Transversal); Fundación Faustino Orbegozo Eizaguirre: Bilbao, Spain, 2011. [Google Scholar]
- World Health Organization. Obesity: Preventing and Managing the Global Epidemic; World Health Organization: Geneva, Switzerland, 2000. [Google Scholar]
- Browning, L.M.; Hsieh, S.D.; Ashwell, M. A systematic review of waist-to-height ratio as a screening tool for the prediction of cardiovascular disease and diabetes: 0·5 could be a suitable global boundary value. Nutr. Res. Rev. 2010, 23, 247–269. [Google Scholar] [CrossRef]
- World Health Organization. Strategies to Monitor and Evaluate Population Sodium Consumption and Sources of Sodium in the Diet; Report of a Joint Technical Meeting Convened by WHO and the Government of Canada; World Health Organization: Geneva, Switzerland, 2011. [Google Scholar]
- European Commission; Directorate-General Health and Consumers. Survey on Members States’ Implementation of the EU Salt Reduction Framework. 2014. Available online: http://ec.europa.eu/health/nutrition_physical_activity/docs/salt_report1_en.pdf (accessed on 21 February 2019).
- Leyvraz, M.; Chatelan, A.; da Costa, B.R.; Taffé, P.; Paradis, G.; Bovet, P.; Bochud, M.; Chiolero, A. Sodium intake and blood pressure in children and adolescents: A systematic review and meta-analysis of experimental and observational studies. Int. J. Epidemiol. 2018, 47, 1796–1810. [Google Scholar] [CrossRef]
- Aparicio, A.; Rodríguez-Rodríguez, E.; Cuadrado-Soto, E.; Navia, B.; López-Sobaler, A.; Ortega, R. Estimation of salt intake assessed by urinary excretion of sodium over 24 h in Spanish subjects aged 7–11 years. Eur. J. Nutr. 2017, 56, 171–178. [Google Scholar] [CrossRef]
- Quader, Z.S.; Gillespie, C.; Sliwa, S.A.; Ahuja, J.K.C.; Burdg, J.P.; Moshfegh, A.; Pehrsson, P.R.; Gunn, J.P.; Mugavero, K.; Cogswell, M.E. Sodium Intake among US School-Aged Children: National Health and Nutrition Examination Survey, 2011–2012. J. Acad. Nutr. Diet. 2017, 117, 39–47. [Google Scholar] [CrossRef] [PubMed]
- Dickinson, K.M.; Chan, L.; Moores, C.J.; Miller, J.; Thomas, J.; Yaxley, A.; Jackson, K.; Mehta, K.; Matwiejczyk, L.; Wray, A.; et al. Eating occasions and the contribution of foods to sodium and potassium intakes in adults. Public Health Nutr. 2018, 21, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Cuadrado-Soto, E.; Peral-Suarez, Á.; Aparicio, A.; Perea, J.; Ortega, R.; López-Sobaler, A. Sources of Dietary Sodium in Food and Beverages Consumed by Spanish Schoolchildren between 7 and 11 Years Old by the Degree of Processing and the Nutritional Profile. Nutrients 2018, 10, 1880. [Google Scholar] [CrossRef] [PubMed]
- Neal, B.C.; Capelin, C.; Dunford, E.K.; Webster, J.L.; Jebb, S.A.; Ni Mhurchu, C. Sodium content of processed foods in the United Kingdom: Analysis of 44,000 foods purchased by 21,000 households1–3. Am. J. Clin. Nutr. 2011, 93, 594–600. [Google Scholar] [CrossRef]
- Quilez, J.; Salas-Salvado, J. Salt in bread in Europe: Potential benefits of reduction. Nutr. Rev. 2012, 70, 666–678. [Google Scholar] [CrossRef]
- Quílez, J.; Salas-Salvadó, J. La ingesta de sal y el consumo de pan. Una visión amplia de la situación en España. Rev. Esp. Nutr. Hum. Diet. 2013, 17, 61–72. [Google Scholar] [CrossRef]
- Pérez Farinós, N.; Santos Sanz, S.; Dal Re, M.Á.; Yusta Boyo, J.; Robledo, T.; Castrodeza, J.J.; Campos Amado, J.; Villar, C. Salt content in bread in Spain, 2014. Nutr. Hosp. 2018, 35, 650–654. [Google Scholar] [CrossRef]
- Pérez-Farinós, N.; Robledo de Dios, T.; Re-Saavedra, D.; Ángeles, M.; Villar Villalba, C.; Labrado Mendo, E. Exceso de sal y salud. Consumo y fuentes alimentarias de sal en España. Nutr. Hosp. 2014, 30, 36–38. [Google Scholar]
- Pascual, V.C.; Sánchez, A.M.; Toledano, F.L.; del Moral, A.M.; de Victoria Muñoz, E.M.; Martínez, G.P.; Se-gura, C.P.; Solé, R.M.P.; Martínez, A.P.; Cañavate, J.L.R.; et al. Informe del Comité Científico de la Agencia Española de Consumo, Seguridad Alimentaria y Nutrición (AECOSAN) sobre objetivos y re-comendaciones nutricionales y de actividad física frente a la obesidad en el marco de la Estrategia NAOS. Revista del Comité Científico de la AESAN 2014, 19, 95–209. [Google Scholar]
- European Food Safety Authority. Opinion of the scientific panel on dietetic products, nutrition and allergies on a request from the commission related to the tolerable upper intake level of sodium. EFSA J. 2005, 209, 1–26. Available online: http://www.efsa.europa.eu/sites/default/files/scientific_output/files/main_documents/209.pdf (accessed on 22 February 2019).
- Council of the European Union. Council conclusions on action to reduce population salt intake for better health. In Proceedings of the 3019th Employment, Social Policy Health and Consumer Affairs Council Meeting, Luxembourg, 8 June 2010; Available online: http://www.consilium.europa.eu/uedocs/cms_Data/docs/pressdata/en/lsa/114998.pdf (accessed on 21 February 2019).
- Araújo, J.R.; Martel, F.; Borges, N.; Araújo, J.M.; Keating, E. Folates and aging: Role in mild cognitive impairment, dementia and depression. Ageing Res. Rev. 2015, 22, 9–19. [Google Scholar] [CrossRef] [PubMed]
- Navia, B.; Aparicio, A.; Perea, J.M.; Pérez-Farinós, N.; Villar-Villalba, C.; Labrado, E.; Ortega, R.M. Sodium intake may promote weight gain; results of the FANPE study in a representative sample of the adult Spanish population. Nutr. Hosp. 2014, 29, 1283–1289. [Google Scholar] [PubMed]
- Cocores, J.A.; Gold, M.S. The Salted Food Addiction Hypothesis may explain overeating and the obesity epidemic. Med. Hypotheses 2009, 73, 892–899. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; He, F.J.; MacGregor, G.A. High salt intake: Independent risk factor for obesity? Hypertension 2015, 66, 843–849. [Google Scholar] [CrossRef]
- Cappuccio, F.P.; Ji, C.; Donfrancesco, C.; Palmieri, L.; Ippolito, R.; Vanuzzo, D.; Giampaoli, S.; Strazzullo, P. Geographic and socioeconomic variation of sodium and potassium intake in Italy: Results from the MINISAL-GIRCSI programme. BMJ Open 2015, 5, e007467. [Google Scholar] [CrossRef]
| N | Total Sodium Intake (mg/day) | |||
|---|---|---|---|---|
| Total Population (9–75 y) | Total | 2009 | 2025 ± 805 | |
| Men | 1013 | 2218 ± 868 * | ||
| Women | 996 | 1828 ± 682 | ||
| Age | 9–12 y | Total | 213 | 2247 ± 735 a |
| Men | 126 | 2350 ± 798 * | ||
| Women | 87 | 2098 ± 608 | ||
| 13–17 y | Total | 211 | 2351 ± 842 a | |
| Men | 137 | 2517 ± 868 * | ||
| Women | 74 | 2043 ± 698 | ||
| 18–64 y | Total | 1655 | 2026 ± 805 b | |
| Men | 798 | 2219 ± 876 * | ||
| Women | 857 | 1846 ± 686 | ||
| 65–75 y | Total | 206 | 1693 ± 640 c | |
| Men | 99 | 1841 ± 668 * | ||
| Women | 107 | 1556 ± 583 | ||
| N | Sodium (mg/day) Per 1000 kcal | |||
|---|---|---|---|---|
| Total Population (9–75 y) | Total | 2009 | 1118 ± 315 | |
| Men | 1013 | 1130 ± 307 | ||
| Women | 996 | 1106 ± 322 | ||
| Age | 9–12 y | Total | 213 | 1146 ± 265 a |
| Men | 126 | 1169 ± 266 | ||
| Women | 87 | 1113 ± 263 | ||
| 13–17 y | Total | 211 | 1166 ± 303 a | |
| Men | 137 | 1190 ± 304 | ||
| Women | 74 | 1122 ± 299 | ||
| 18–64 y | Total | 1655 | 1116 ± 317 a,b | |
| Men | 798 | 1125 ± 306 | ||
| Women | 857 | 1109 ± 327 | ||
| 65–75 y | Total | 206 | 1052 ± 299 b | |
| Men | 99 | 1048 ± 307 | ||
| Women | 107 | 1056 ± 294 | ||
| Sodium Intakes by Eating Occasion/Period (mg/day). | Children | Adolescents | Adults | Elderly |
|---|---|---|---|---|
| 9–12 Years Old | 13–17 Years Old | 18–64 Years Old | 65–75 Years Old | |
| n | 213 | 211 | 1655 | 206 |
| Breakfast | 376.4 ± 188.6 | 363.9 ± 229.9 | 309.6 ± 246.0 | 305.9 ± 203.6 |
| Mid-morning | 246.8 ± 266.6 | 278.6 ± 335.6 | 111.6 ± 225.8 | 61.8 ± 156.6 |
| Lunch | 569.7 ± 278.7 | 623.1 ± 382.2 | 692.6 ± 406.3 | 635.2 ± 341.5 |
| Afternoon | 309.6 ± 260.1 | 202.3 ± 244.8 | 101.1 ± 186.3 | 75.9 ± 142.2 |
| Dinner | 695.6 ± 373.7 | 824.9 ± 487.9 | 756.6 ± 453.1 | 599 ± 360.3 |
| Other occasions | 48.9 ± 112.2 | 58 ± 184.8 | 54.1 ± 141.7 | 15.1 ± 48.1 |
| Working days | 2348.8 ± 883.7 | 2382.5 ± 998.8 | 2014.5 ± 907.3 | 1712.9 ± 729.3 |
| Non-working days | 2043.4 ± 1021.1 | 2287.1 ± 1092.2 | 2047.6 ± 1129.6 | 1653 ± 880.7 |
| Food Groups | Food Subgroups | Sodium Contribution (mg/day) | Sodium Contribution (%) |
|---|---|---|---|
| Meat and meat products | 530.3 ± 410.0 | 27 | |
| Sausages and other processed meats products | 403.8 ± 371.9 | ||
| Fresh meat | 125.0 ± 157.5 | ||
| Viscera and offal | 1.5 ± 9.2 | ||
| Cereals and Grains | 522.0 ± 271.1 | 26 | |
| Bread | 400.4 ± 244.4 | ||
| Bakery and pastry | 84.3 ± 108.2 | ||
| Breakfast cereals and cereal bars | 24.7 ± 71.2 | ||
| Grains and flours | 4.7 ± 13.7 | ||
| Tubers | 4.4 ± 10.4 | ||
| Pasta | 3.6 ± 24.0 | ||
| Milk and dairy products | 280.5 ± 232.4 | 14 | |
| Cheese | 150.0 ± 209.2 | ||
| Milk | 88.9 ± 70.2 | ||
| Yogurt and fermented milk | 29.3 ± 40.2 | ||
| Other dairy products | 12.3 ± 29.4 | ||
| Ready-to-eat meals | 261.9 ± 330.2 | 13 | |
| Fish and shellfish | 120.1 ± 198.4 | 6 | |
| Canned fish and shellfish | 74.1 ± 182.3 | ||
| Shellfish | 23.4 ± 56.3 | ||
| Fish | 23.4 ± 33.9 | ||
| Appetisers | 77.1 ± 168.0 | 4 | |
| Sauces and condiments | 59.5 ± 125.1 | 3 | |
| Vegetables | 49.1 ± 49.7 | 2 | |
| Sugars and sweets | 37.9 ± 70.9 | 2 | |
| Chocolates | 37.3 ± 70.9 | ||
| Jams and similar | 0.4 ± 1.3 | ||
| Sugars | 0.2 ± 0.6 | ||
| Eggs | 36.4 ± 39.1 | 2 |
| Sodium Intakes (mg/d) | ||||||
|---|---|---|---|---|---|---|
| Total Population | Children 9–12 y | Adolescents 13–17 y | Adults 18–64 y | Elderly 65–75 y | ||
| BMI | Underweight | 2245.7± 771.5 | 2252.3 ± 681.2 | 2327.1 ± 793.8 | 2151.1 ± 983.8 | - |
| n = 143 | n = 85 | n = 28 | n = 30 | |||
| Normal | 2129.3 ± 840.5 | 2205.1 ± 779.0 | 2392.9 ± 905.9 | 2084.4 ± 827.2 | 1725.6 ±733.1 | |
| n = 1000 | n = 112 | n = 146 | n = 704 | n = 38 | ||
| Overweight | 1953.8 ± 745.9 | 2543.0 ± 694.4 * | 2177.4 ± 561.7 | 1970.2 ± 761.4 | 1694.9 ± 619.5 | |
| n = 740 | n = 15 | n = 33 | n = 592 | n = 100 | ||
| Obesity | 1938.6 ± 788.7 | - | 2404.0 ± 688.6 | 1987.8 ± 810.6 | 1671.7 ± 622.7 | |
| n = 402 | n = 4 | n = 329 | n = 68 | |||
| Waist/Height | No risk | 2174.8 ± 845.2 | 2247.9 ± 761 | 2387.1 ± 876.2 | 2107.1 ± 846.2 | 1842.5 ± 831.7 |
| n = 1043 | n = 162 | n = 181 | n = 689 | n = 11 | ||
| Risk | 1938.3 ± 750.1 | 2243.9 ± 654.3 | 2131.3 ± 554.9 | 1967.4 ± 769.9 | 1684.5 ± 629.0 | |
| n = 1242 | n = 51 | n = 30 | n = 966 | n = 195 | ||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Partearroyo, T.; Samaniego-Vaesken, M.d.L.; Ruiz, E.; Aranceta-Bartrina, J.; Gil, Á.; González-Gross, M.; Ortega, R.M.; Serra-Majem, L.; Varela-Moreiras, G. Sodium Intake from Foods Exceeds Recommended Limits in the Spanish Population: The ANIBES Study. Nutrients 2019, 11, 2451. https://doi.org/10.3390/nu11102451
Partearroyo T, Samaniego-Vaesken MdL, Ruiz E, Aranceta-Bartrina J, Gil Á, González-Gross M, Ortega RM, Serra-Majem L, Varela-Moreiras G. Sodium Intake from Foods Exceeds Recommended Limits in the Spanish Population: The ANIBES Study. Nutrients. 2019; 11(10):2451. https://doi.org/10.3390/nu11102451
Chicago/Turabian StylePartearroyo, Teresa, Mª de Lourdes Samaniego-Vaesken, Emma Ruiz, Javier Aranceta-Bartrina, Ángel Gil, Marcela González-Gross, Rosa M. Ortega, Lluis Serra-Majem, and Gregorio Varela-Moreiras. 2019. "Sodium Intake from Foods Exceeds Recommended Limits in the Spanish Population: The ANIBES Study" Nutrients 11, no. 10: 2451. https://doi.org/10.3390/nu11102451
APA StylePartearroyo, T., Samaniego-Vaesken, M. d. L., Ruiz, E., Aranceta-Bartrina, J., Gil, Á., González-Gross, M., Ortega, R. M., Serra-Majem, L., & Varela-Moreiras, G. (2019). Sodium Intake from Foods Exceeds Recommended Limits in the Spanish Population: The ANIBES Study. Nutrients, 11(10), 2451. https://doi.org/10.3390/nu11102451











