Association Between Fatty Acids Profile and Cerebral Blood Flow: An Exploratory fNIRS Study on Children with and without ADHD
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.1.1. Children with ADHD
2.1.2. Typically Developing Children
2.2. Measures
2.2.1. Cognitive and Clinical-Behavioral Profile
2.2.2. Fatty Acids Profile
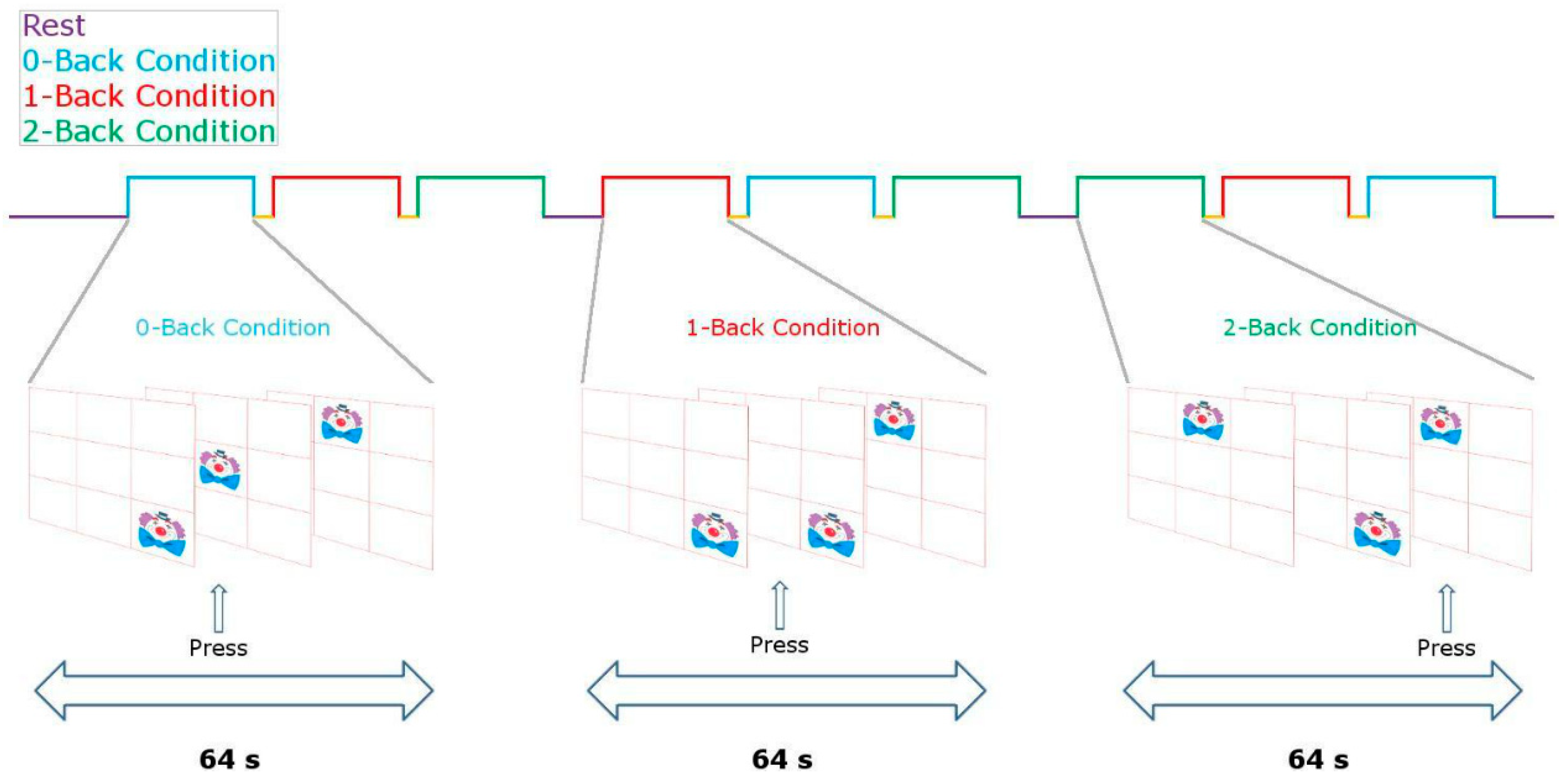
2.2.3. Stimulation Protocol
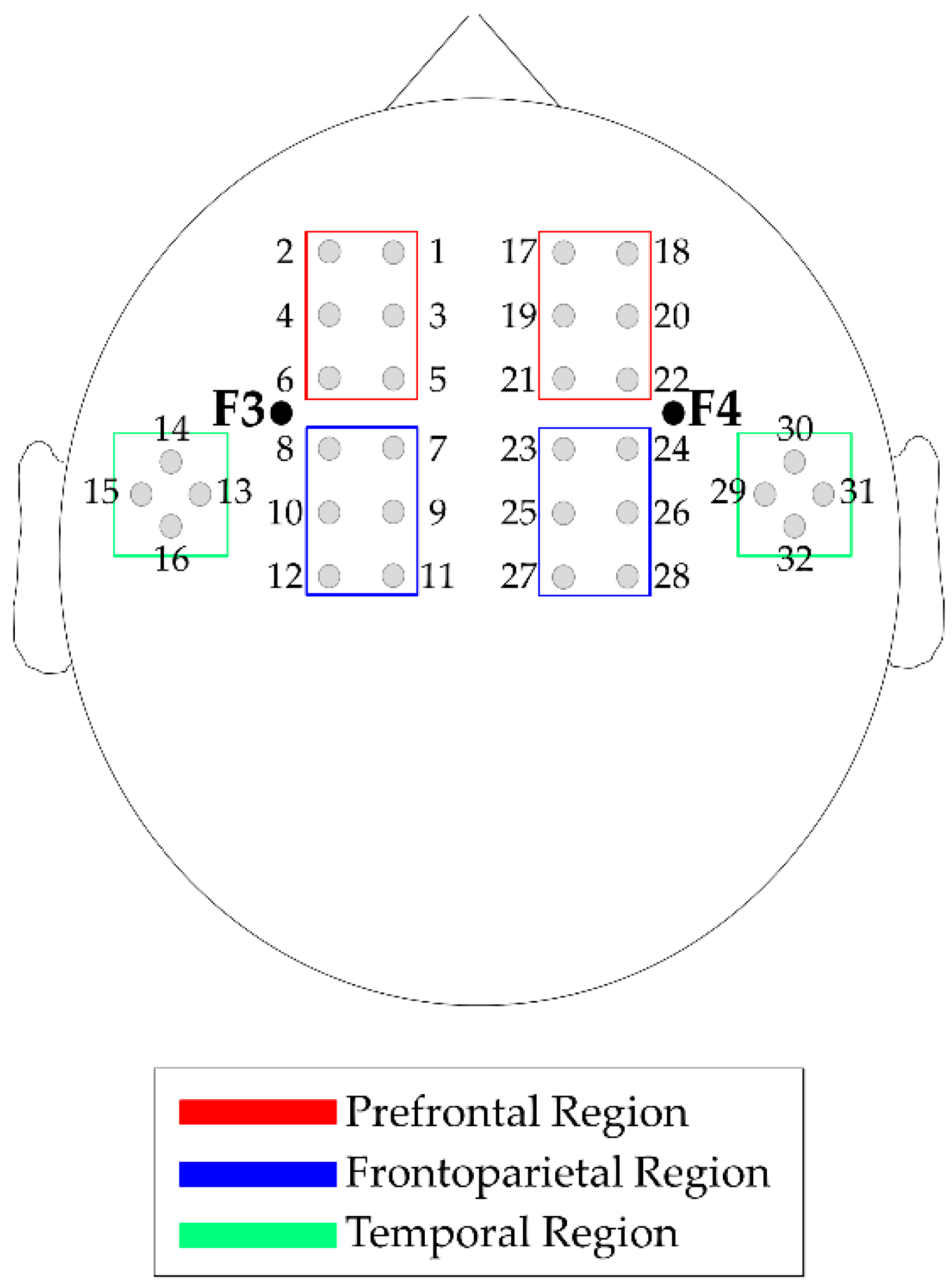
2.2.4. Neurophysiological Profile
2.2.5. Neurophysiological Profile
2.2.6. ADHD Severity-Neurophysiological Profile Association
2.2.7. Fatty Acids-Neurophysiological Profile Association
3. Results
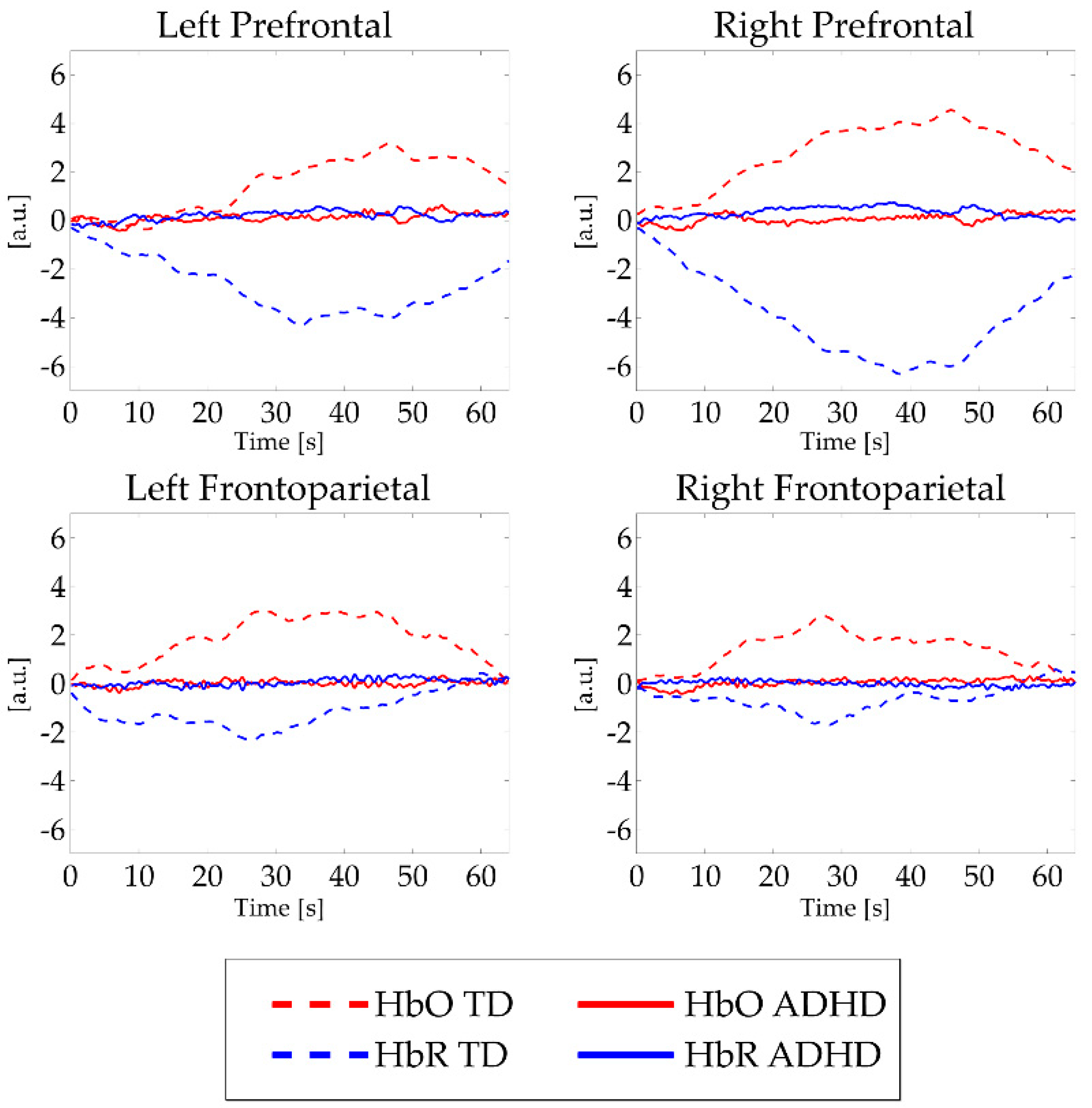
3.1. Neurophysiological Profile
3.2. ADHD Severity-Neurophysiological Profile Association.
3.3. Fatty Acids-Neurophysiological Profile Association
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Thomas, R.; Sanders, S.; Doust, J.; Beller, E.; Glasziou, P. Prevalence of Attention-Deficit/Hyperactivity Disorder: A Systematic Review and Meta-analysis. Pediatrics 2015, 135, 994–1001. [Google Scholar] [CrossRef] [PubMed]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders (DSM-5®); American Psychiatric Pub: Washington, DC, USA, 2013. [Google Scholar]
- Faraone, S.V.; Bonvicini, C.; Scassellati, C. Biomarkers in the Diagnosis of ADHD—Promising Directions. Curr. Psychiatry Rep. 2014, 16, 497. [Google Scholar] [CrossRef] [PubMed]
- Bloch, M.H.; Qawasmi, A. Omega-3 fatty acid supplementation for the treatment of children with attention-deficit/hyperactivity disorder symptomatology: Systematic review and meta-analysis. J. Am. Acad. Child Adolesc. Psychiatry 2011, 50, 991–1000. [Google Scholar] [CrossRef] [PubMed]
- Hawkey, E.; Nigg, J.T. Omega-3 fatty acid and ADHD: Blood level analysis and meta-analytic extension of supplementation trials. Clin. Psychol. Rev. 2014, 34, 496–505. [Google Scholar] [CrossRef] [PubMed]
- Sonuga-Barke, E.J.; Brandeis, D.; Cortese, S.; Daley, D.; Ferrin, M.; Holtmann, M.; Stevenson, J.; Danckaerts, M.; Van Der Oord, S.; Döpfner, M.; et al. Nonpharmacological Interventions for ADHD: Systematic Review and Meta-Analyses of Randomized Controlled Trials of Dietary and Psychological Treatments. Am. J. Psychiatry 2013, 170, 275–289. [Google Scholar] [CrossRef] [PubMed]
- McNamara, R.K.; Asch, R.H.; Lindquist, D.M.; Krikorian, R. Role of polyunsaturated fatty acids in human brain structure and function across the lifespan: An update on neuroimaging findings. Prostaglandins Leukot. Essent. Fat. Acids 2018, 136, 23–34. [Google Scholar] [CrossRef] [PubMed]
- Zárate, R.; El Jaber-Vazdekis, N.; Tejera, N.; Pérez, J.A.; Rodríguez, C. Significance of long chain polyunsaturated fatty acids in human health. Clin. Transl. Med. 2017, 6, 25. [Google Scholar] [CrossRef] [PubMed]
- Milte, C.M.; Sinn, N.; Howe, P.R.; Parletta, N. Polyunsaturated fatty acid status in attention deficit hyperactivity disorder, depression, and Alzheimer’s disease: Towards an omega-3 index for mental health? Nutr. Rev. 2009, 67, 573–590. [Google Scholar] [CrossRef]
- Agostoni, C.; Nobile, M.; Ciappolino, V.; DelVecchio, G.; Tesei, A.; Turolo, S.; Crippa, A.; Mazzocchi, A.; Altamura, C.A.; Brambilla, P. The Role of Omega-3 Fatty Acids in Developmental Psychopathology: A Systematic Review on Early Psychosis, Autism, and ADHD. Int. J. Mol. Sci. 2017, 18, 2608. [Google Scholar] [CrossRef]
- Sergeant, S.; Rahbar, E.; Chilton, F.H. Gamma-linolenic acid, Dihommo-gamma linolenic, Eicosanoids and Inflammatory Processes. Eur. J. Pharmacol. 2016, 785, 77–86. [Google Scholar] [CrossRef]
- Crippa, A.; Tesei, A.; Sangiorgio, F.; Salandi, A.; Trabattoni, S.; Grazioli, S.; Agostoni, C.; Molteni, M.; Nobile, M. Behavioral and cognitive effects of docosahexaenoic acid in drug-naïve children with attention-deficit/hyperactivity disorder: A randomized, placebo-controlled clinical trial. Eur. Child Adolesc. Psychiatry 2019, 28, 571–583. [Google Scholar] [CrossRef] [PubMed]
- Hadley, K.B.; Ryan, A.S.; Forsyth, S.; Gautier, S.; Salem, N. The Essentiality of Arachidonic Acid in Infant Development. Nutrients 2016, 8, 216. [Google Scholar] [CrossRef]
- Katsuki, H.; Okuda, S. Arachidonic acid as a neurotoxic and neurotrophic substance. Prog. Neurobiol. 1995, 46, 607–636. [Google Scholar] [CrossRef]
- Jackson, P.A.; Forster, J.S.; Bell, J.G.; Dick, J.R.; Younger, I.; Kennedy, D.O. DHA Supplementation Alone or in Combination with Other Nutrients Does not Modulate Cerebral Hemodynamics or Cognitive Function in Healthy Older Adults. Nutrients 2016, 8, 86. [Google Scholar] [CrossRef] [PubMed]
- Tesei, A.; Crippa, A.; Ceccarelli, S.B.; Mauri, M.; Molteni, M.; Agostoni, C.; Nobile, M. The potential relevance of docosahexaenoic acid and eicosapentaenoic acid to the etiopathogenesis of childhood neuropsychiatric disorders. Eur. Child Adolesc. Psychiatry 2017, 26, 1011–1030. [Google Scholar] [CrossRef]
- Faraone, S.V.; Asherson, P.; Banaschewski, T.; Biederman, J.; Buitelaar, J.K.; Ramos-Quiroga, J.A.; Franke, B. Attention-deficit/hyperactivity disorder. Nat. Rev. Dis. Primers 2015, 1, 15020. [Google Scholar] [CrossRef] [PubMed]
- Bos, D.J.; Van Montfort, S.J.; Oranje, B.; Durston, S.; Smeets, P.A.; Information, P.E.K.F.C. Effects of omega-3 polyunsaturated fatty acids on human brain morphology and function: What is the evidence? Eur. Neuropsychopharmacol. 2016, 26, 546–561. [Google Scholar] [CrossRef]
- Catalan, J.; Toru, M.; Slotnick, B.; Murthy, M.; Greiner, R.S.; Salem, N., Jr. Cognitive deficits in docosahexaenoic acid-deficient rats. Behav. Neurosci. 2002, 116, 1022. [Google Scholar] [CrossRef]
- Ferrari, M.; Quaresima, V. A brief review on the history of human functional near-infrared spectroscopy (fNIRS) development and fields of application. NeuroImage 2012, 63, 921–935. [Google Scholar] [CrossRef]
- Fallgatter, A.J.; Strik, W.K. Right frontal activation during the continuous performance test assessed with near-infrared spectroscopy in healthy subjects. Neurosci. Lett. 1997, 223, 89–92. [Google Scholar] [CrossRef]
- Fallgatter, A.J.; Strik, W.K. Frontal brain activation during the Wisconsin Card Sorting Test assessed with two-channel near-infrared spectroscopy. Eur. Arch. Psychiatry Clin. Neurosci. 1998, 248, 245–249. [Google Scholar] [CrossRef] [PubMed]
- Schecklmann, M.; Ehlis, A.C.; Plichta, M.M.; Fallgatter, A.J. Functional near-infrared spectroscopy: A long-term reliable tool for measuring brain activity during verbal fluency. NeuroImage 2008, 43, 147–155. [Google Scholar] [CrossRef] [PubMed]
- Grazioli, S.; Mauri, M.; Crippa, A.; Maggioni, E.; Molteni, M.; Brambilla, P.; Nobile, M. Light up ADHD: II. Neuropharmacological effects measured by near infrared spectroscopy: Is there a biomarker? J. Affect. Disord. 2018, 244, 100–106. [Google Scholar] [CrossRef] [PubMed]
- Mauri, M.; Nobile, M.; Bellina, M.; Crippa, A.; Brambilla, P. Light up ADHD: I. Cortical hemodynamic responses measured by functional Near Infrared Spectroscopy (fNIRS). J. Affect. Disord. 2018, 234, 358–364. [Google Scholar] [CrossRef] [PubMed]
- Hamazaki-Fujita, N.; Hamazaki, K.; Tohno, H.; Itomura, M.; Terashima, Y.; Hamazaki, T.; Nakamura, N.; Yomoda, S. Polyunsaturated fatty acids and blood circulation in the forebrain during a mental arithmetic task. Brain Res. 2011, 1397, 38–45. [Google Scholar] [CrossRef] [PubMed]
- Pu, S.; Nakagome, K.; Yamada, T.; Matsumura, H.; Yokoyama, K.; Kaneko, K.; Kurosawa, Y. Association between Fish Consumption and Prefrontal Function during a Cognitive Task in Male Japanese Workers: A Multi-Channel Near-Infrared Spectroscopy Study. PLoS ONE 2015, 10, e0123972. [Google Scholar] [CrossRef] [PubMed]
- Jackson, P.A.; Reay, J.L.; Scholey, A.B.; Kennedy, D.O. DHA-rich oil modulates the cerebral haemodynamic response to cognitive tasks in healthy young adults: A near IR spectroscopy pilot study. Br. J. Nutr. 2012, 107, 1093–1098. [Google Scholar] [CrossRef] [PubMed]
- Jackson, P.A.; Reay, J.L.; Scholey, A.B.; Kennedy, D.O. Docosahexaenoic acid-rich fish oil modulates the cerebral hemodynamic response to cognitive tasks in healthy young adults. Boil. Psychol. 2012, 89, 183–190. [Google Scholar] [CrossRef]
- Bos, D.J.; Oranje, B.; Veerhoek, E.S.; Van Diepen, R.M.; Weusten, J.M.; Demmelmair, H.; Koletzko, B.; Velden, M.G.D.S.V.D.; Eilander, A.; Hoeksma, M.; et al. Reduced Symptoms of Inattention after Dietary Omega-3 Fatty Acid Supplementation in Boys with and without Attention Deficit/Hyperactivity Disorder. Neuropsychopharmacology 2015, 40, 2298–2306. [Google Scholar] [CrossRef]
- Cui, X.; Bray, S.; Bryant, D.M.; Glover, G.H.; Reiss, A.L. A quantitative comparison of NIRS and fMRI across multiple cognitive tasks. Neuroimage 2011, 54, 2808–2821. [Google Scholar] [CrossRef]
- Crippa, A.; Salvatore, C.; Molteni, E.; Mauri, M.; Salandi, A.; Trabattoni, S.; Agostoni, C.; Molteni, M.; Nobile, M.; Castiglioni, I. The Utility of a Computerized Algorithm Based on a Multi-Domain Profile of Measures for the Diagnosis of Attention Deficit/Hyperactivity Disorder. Front. Psychol. 2017, 8, 189. [Google Scholar] [CrossRef] [PubMed]
- Goodman, R.; Ford, T.; Richards, H.; Gatward, R.; Meltzer, H. The Development and Well-Being Assessment: Description and Initial Validation of an Integrated Assessment of Child and Adolescent Psychopathology. J. Child Psychol. Psychiatry 2000, 41, 645–655. [Google Scholar] [CrossRef] [PubMed]
- Wechsler, D. Wechsler Intelligence Scale for Children–III (WISC-III) Italian Edition; Organizzazioni Speciali: Florence, Italy, 2006. [Google Scholar]
- Groth-Marnat, G. Handbook of Psychological Assessment, 3rd ed.; John Wiley and Sons: New York, NY, USA, 1997. [Google Scholar]
- Hollingshead, A.B. Four Factor Index of Social Status; Yale University: New Haven, CT, USA, 1975; Unpublished work. [Google Scholar]
- De Sonneville, L.M.J. ANT 2.1—Amsterdam Neuropsychological Tasks; Sonar: Amstelveen, The Netherlands, 2000. [Google Scholar]
- Conners, C.K. Conners’ Rating Scales—Revised: User’s Manual; Multi-Health Systems, Incorporated: North Tonawanda, NY, USA, 1997. [Google Scholar]
- DuPaul, G.J.; Power, T.J.; Anastopoulos, A.D.; Reid, R. ADHD Rating Scale—IV: Checklists, Norms, and Clinical Interpretation; Guilford Press: New York, NY, USA, 1998. [Google Scholar]
- Crippa, A.; Agostoni, C.; Mauri, M.; Molteni, M.; Nobile, M. Polyunsaturated fatty acids are associated with behavior but not with cognition in children with and without ADHD: An Italian study. J. Atten. Disord. 2018, 22, 971–983. [Google Scholar] [CrossRef] [PubMed]
- Agostoni, C.; Galli, C.; Riva, E.; Risé, P.; Colombo, C.; Giovannini, M.; Marangoni, F. Whole blood fatty acid composition at birth: From the maternal compartment to the infant. Clin. Nutr. 2011, 30, 503–505. [Google Scholar] [CrossRef] [PubMed]
- Marangoni, F.; Colombo, C.; Galli, C. A method for the direct evaluation of the fatty acid status in a drop of blood from a fingertip in humans: Applicability to nutritional and epidemiological studies. Anal. Biochem. 2004, 326, 267–272. [Google Scholar] [CrossRef] [PubMed]
- Jasper, H.H. The ten-twenty electrode system of the International Federation. Electroencephalogr. Clin. Neurophysiol. 1958, 10, 370–375. [Google Scholar]
- Scholkmann, F.; Kleiser, S.; Metz, A.J.; Zimmermann, R.; Pavia, J.M.; Wolf, U.; Wolf, M. A review on continuous wave functional near-infrared spectroscopy and imaging instrumentation and methodology. NeuroImage 2014, 85, 6–27. [Google Scholar] [CrossRef] [PubMed]
- Huppert, T.J.; Diamond, S.G.; Franceschini, M.A.; Boas, D.A. HomER: A review of time-series analysis methods for near-infrared spectroscopy of the brain. Appl. Opt. 2009, 48, D280–D298. [Google Scholar] [CrossRef]
- Piazza, C.; Bacchetta, A.; Crippa, A.; Mauri, M.; Grazioli, S.; Reni, G.; Nobile, M.; Bianchi, A.M. Preprocessing Pipeline for fNIRS Data in Children. In Proceedings of the XV Mediterranean Conference on Medical and Biological Engineering and Computing – MEDICON 2019, Coimbra, Portugal, 26–28 September 2019; Henriques, J., Neves, N., de Carvalho, P., Eds.; Springer: Cham, Germany, 2019; Volume 76. [Google Scholar]
- Ichikawa, H.; Nakato, E.; Kanazawa, S.; Shimamura, K.; Sakuta, Y.; Sakuta, R.; Yamaguchi, M.K.; Kakigi, R. Hemodynamic response of children with attention-deficit and hyperactive disorder (ADHD) to emotional facial expressions. Neuropsychologia 2014, 63, 51–58. [Google Scholar] [CrossRef]
- Field, A. Discovering Statistics Using IBM SPSS Statistics; Sage: Newcastle, UK, 2013. [Google Scholar]
- Barbaranelli, C. Analisi Dei Dati: Tecniche Multivariate Per la Ricerca Psicologica e Sociale; Edizioni Universitarie di Lettere Economia Diritto: Milano, Italy, 2007. [Google Scholar]
- Morse, N. A meta-analysis of blood fatty acids in people with learning disorders with particular interest in arachidonic acid. ProstaglandinS Leukot. Essent. Fat. Acids 2009, 81, 373–389. [Google Scholar] [CrossRef]
- Parletta, N.; Niyonsenga, T.; Duff, J. Omega-3 and Omega-6 Polyunsaturated Fatty Acid Levels and Correlations with Symptoms in Children with Attention Deficit Hyperactivity Disorder, Autistic Spectrum Disorder and Typically Developing Controls. PLoS ONE 2016, 11, e0156432. [Google Scholar] [CrossRef] [PubMed]
- Kobel, M.; Bechtel, N.; Weber, P.; Specht, K.; Klarhöfer, M.; Scheffler, K.; Opwis, K.; Penner, I.K. Effects of methylphenidate on working memory functioning in children with attention deficit/hyperactivity disorder. Eur. J. Paediatr. Neurol. 2009, 13, 516–523. [Google Scholar] [CrossRef] [PubMed]
- McNamara, R.K.; Able, J.; Jandacek, R.; Rider, T.; Tso, P.; Eliassen, J.C.; Alfieri, D.; Weber, W.; Jarvis, K.; DelBello, M.P.; et al. Docosahexaenoic acid supplementation increases prefrontal cortex activation during sustained attention in healthy boys: A placebo-controlled, dose-ranging, functional magnetic resonance imaging study. Am. J. Clin. Nutr. 2010, 91, 1060–1067. [Google Scholar] [CrossRef] [PubMed]
- Ehlis, A.C.; Bähne, C.G.; Jacob, C.P.; Herrmann, M.J.; Fallgatter, A.J. Reduced lateral prefrontal activation in adult patients with attention-deficit/hyperactivity disorder (ADHD) during a working memory task: A functional near-infrared spectroscopy (fNIRS) study. J. Psychiatr. Res. 2008, 42, 1060–1067. [Google Scholar] [CrossRef] [PubMed]
- Sublette, M.E.; Milak, M.S.; Hibbeln, J.R.; Freed, P.J.; Oquendo, M.A.; Malone, K.M.; Parsey, R.V.; Mann, J.J. Plasma polyunsaturated fatty acids and regional cerebral glucose metabolism in major depression. Prostaglandins Leukot. Essent. Fat. Acids 2009, 80, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Almeida, D.M.; Jandacek, R.J.; Weber, W.A.; McNamara, R.K. Docosahexaenoic acid biostatus is associated with event-related functional connectivity in cortical attention networks of typically developing children. Nutr. Neurosci. 2017, 20, 246–254. [Google Scholar] [CrossRef] [PubMed]
- Lauritzen, L.; Brambilla, P.; Mazzocchi, A.; Harsløf, L.B.S.; Ciappolino, V.; Agostoni, C. DHA Effects in Brain Development and Function. Nutrients 2016, 8, 6. [Google Scholar] [CrossRef] [PubMed]
| ADHD | TD | Value | p | |
|---|---|---|---|---|
| N | 24 | 21 | - | - |
| F:M | 0:24 | 1:20 | 1.169 a | 0.467 |
| Age | 11.5 ± 1.5 | 11.3 ± 1.8 | −0.485 b | 0.630 |
| IQ | 101.8 ± 11.1 | 110 ± 20 | 1.683 b | 0.103 |
| SES | 54 ± 20.2 | 56 ± 18.7 | 0.344 b | 0.734 |
| ADHD | TD | Value a | p | |
|---|---|---|---|---|
| LA | 22.80 ± 2.34 | 22.63 ± 2.47 | −0.242 | 0.810 |
| AA | 9.34 ± 1.19 | 10.07 ± 0.94 | 2.264 | 0.029 |
| EPA | 0.98 ± 0.56 | 1.13 ± 0.46 | 0.963 | 0.341 |
| DHA | 1.75 ± 0.49 | 1.92 ± 0.54 | 1.107 | 0.274 |
| AA and EPA → rFP HbO, 0B | Regression Coefficients | ||
|---|---|---|---|
| β [Bootstrap c.i.] | t | p | |
| Age | −0.323 [−0.936; 0.051] | −1.976 | 0.055 |
| IQ | 0.042 [−0.050; 0.057] | 0.259 | 0.810 |
| SES | 0.216 [−0.006; 0.061] | 1.351 | 0.094 |
| AA | −0.030 [−0.860; 0.562] | −0.208 | 0.840 |
| EPA | −0.494 [−4.678; −0.813] | −3.280 | 0.015 |
| Model summary | |||
| Adjusted R2 | F | p | |
| Age, IQ, SES | 0.070 | 1.955 | 0.139 |
| AA and EPA | 0.268 | 3.786 | 0.008 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Grazioli, S.; Crippa, A.; Mauri, M.; Piazza, C.; Bacchetta, A.; Salandi, A.; Trabattoni, S.; Agostoni, C.; Molteni, M.; Nobile, M. Association Between Fatty Acids Profile and Cerebral Blood Flow: An Exploratory fNIRS Study on Children with and without ADHD. Nutrients 2019, 11, 2414. https://doi.org/10.3390/nu11102414
Grazioli S, Crippa A, Mauri M, Piazza C, Bacchetta A, Salandi A, Trabattoni S, Agostoni C, Molteni M, Nobile M. Association Between Fatty Acids Profile and Cerebral Blood Flow: An Exploratory fNIRS Study on Children with and without ADHD. Nutrients. 2019; 11(10):2414. https://doi.org/10.3390/nu11102414
Chicago/Turabian StyleGrazioli, Silvia, Alessandro Crippa, Maddalena Mauri, Caterina Piazza, Andrea Bacchetta, Antonio Salandi, Sara Trabattoni, Carlo Agostoni, Massimo Molteni, and Maria Nobile. 2019. "Association Between Fatty Acids Profile and Cerebral Blood Flow: An Exploratory fNIRS Study on Children with and without ADHD" Nutrients 11, no. 10: 2414. https://doi.org/10.3390/nu11102414
APA StyleGrazioli, S., Crippa, A., Mauri, M., Piazza, C., Bacchetta, A., Salandi, A., Trabattoni, S., Agostoni, C., Molteni, M., & Nobile, M. (2019). Association Between Fatty Acids Profile and Cerebral Blood Flow: An Exploratory fNIRS Study on Children with and without ADHD. Nutrients, 11(10), 2414. https://doi.org/10.3390/nu11102414






