Human Milk Oligosaccharide, Phospholipid, and Ganglioside Concentrations in Breast Milk from United Arab Emirates Mothers: Results from the MISC Cohort
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Setting and Population
2.2. Phospholipid, Ganglioside, and Oligosaccharide Analysis
2.3. Statistical Analysis
3. Results
3.1. HMO
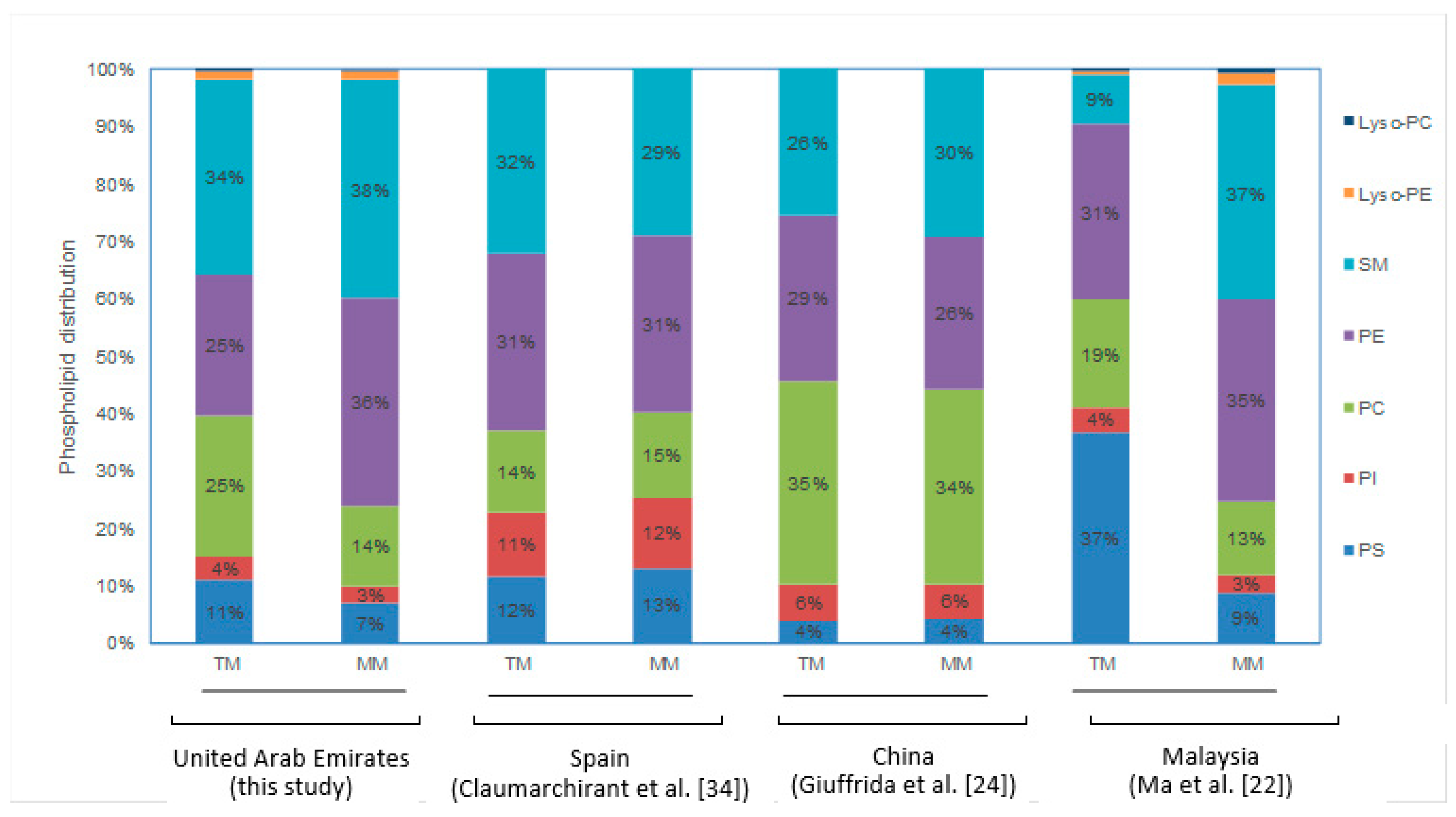
3.2. Phospholipids
3.3. Gangliosides
4. Discussion
4.1. Human Milk Oligosaccharides
4.2. Phospholipids
4.3. Gangliosides
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Michaelsen, K.F.; Skafte, L.; Badsberg, J.H.; Jorgensen, M. Variation in Macronutrients in Human bank milk-influencing factors and implications for human-milk banking. J. Pediatr. Gastroenterol. Nutr. 1990, 11, 229–239. [Google Scholar] [CrossRef] [PubMed]
- Kunz, C.; Rudloff, S.; Baier, W.; Klein, N.; Strobel, S. Oligosaccharides in human milk: Structural, functional, and metabolic aspects. Annu. Rev. Nutr. 2000, 20, 699–722. [Google Scholar] [CrossRef] [PubMed]
- Newburg, D.S.; Neubauer, S.H. Carbohydrates in milk: Analysis, quantities, and Significance. In Handbook of Milk Composition; Jensen, R.G., Ed.; Academic Press: London, UK, 1995; pp. 273–349. [Google Scholar]
- Gopal, P.K.; Gill, H.S. Oligosaccharides and glycoconjugates in bovine milk and colostrum. Br. J. Nutr. 2000, 84 (Suppl. S81), S69–S74. [Google Scholar] [CrossRef]
- Bode, L. Human milk oligosaccharides: Every baby needs a sugar mama. Glycobiology 2012, 22, 1147–1162. [Google Scholar] [CrossRef] [PubMed]
- Bode, L. The functional biology of human milk oligosaccharides. Early Hum. Dev. 2015, 91, 619–622. [Google Scholar] [CrossRef] [PubMed]
- Lin, A.E.; Autran, C.A.; Szyszka, A.; Escajadillo, T.; Huang, M.; Godula, K.; Prudden, A.R.; Boons, G.J.; Lewis, A.L.; Doran, K.S.; et al. Human milk oligosaccharides inhibit growth of group B Streptococcus. J. Biol. Chem. 2017, 292, 11243–11249. [Google Scholar] [CrossRef]
- Yu, H.; Lau, K.; Thon, V.; Autran, C.A.; Jantscher-Krenn, E.; Xue, M.; Li, Y.; Sugiarto, G.; Qu, J.; Mu, S.; et al. Synthetic disialyl hexasaccharides protect neonatal rats from necrotizing enterocolitis. Angew. Chem. Int. Ed. Engl. 2014, 53, 6687–6691. [Google Scholar] [CrossRef]
- Yu, H.; Yan, X.; Autran, C.A.; Li, Y.; Etzold, S.; Latasiewicz, J.; Robertson, B.M.; Li, J.; Bode, L.; Chen, X. Enzymatic and Chemoenzymatic Syntheses of Disialyl Glycans and Their Necrotizing Enterocolitis Preventing Effects. J. Org. Chem. 2017, 82, 13152–13160. [Google Scholar] [CrossRef]
- Ackerman, D.L.; Craft, K.M.; Doster, R.S.; Weitkamp, J.H.; Aronoff, D.M.; Gaddy, J.A.; Townsend, S.D. Antimicrobial and Antibiofilm Activity of Human Milk Oligosaccharides against Streptococcus agalactiae, Staphylococcus aureus, and Acinetobacter baumannii. ACS Infect. Dis. 2018, 4, 315–324. [Google Scholar] [CrossRef]
- Ackerman, D.L.; Doster, R.S.; Weitkamp, J.H.; Aronoff, D.M.; Gaddy, J.A.; Townsend, S.D. Human Milk Oligosaccharides Exhibit Antimicrobial and Antibiofilm Properties against Group B Streptococcus. ACS Infect. Dis. 2017, 3, 595–605. [Google Scholar] [CrossRef]
- Craft, K.M.; Thomas, H.C.; Townsend, S.D. Interrogation of Human Milk Oligosaccharide Fucosylation Patterns for Antimicrobial and Antibiofilm Trends in Group B Streptococcus. ACS Infect. Dis. 2018, 4, 1755–1765. [Google Scholar] [CrossRef] [PubMed]
- Craft, K.M.; Thomas, H.C.; Townsend, S.D. Sialylated variants of lacto-N-tetraose exhibit antimicrobial activity against Group B Streptococcus. Org. Biomol. Chem. 2019, 17, 1893–1900. [Google Scholar] [CrossRef] [PubMed]
- Kunz, C. Complex oligosaccharides in infant nutrition. Mon. Kinderheilkd. 1998, 146, S49–S56. [Google Scholar] [CrossRef]
- Kunz, C.; Rudloff, S. Biological Functions of Milk Oligosaccharides. Acta Paediatr. 1994, 83, 1042. [Google Scholar] [CrossRef]
- Craft, K.M.; Gaddy, J.A.; Townsend, S.D. Human Milk Oligosaccharides (HMOs) Sensitize Group B Streptococcus to Clindamycin, Erythromycin, Gentamicin, and Minocycline on a Strain Specific Basis. ACS Chem. Biol. 2018, 13, 2020–2026. [Google Scholar] [CrossRef] [PubMed]
- Urashima, T.; Kitaoka, M.; Terabayashi, T.; Fukuda, K.; Ohnishi, M.; Kobata, A. Milk Oligosaccharides. In Oligosaccharides: Sources, Properties and Application; Gordon, N.S., Ed.; Nova Science Publishers, Inc.: New York, NY, USA, 2011; pp. 1–58. [Google Scholar]
- Tonon, K.; de Morais, M.; Abrao, A.; Miranda, A.; Morais, T. Maternal and Infant Factors Associated with Human Milk Oligosaccharides Concentrations According to Secretor and Lewis Phenotypes. Nutrients 2019, 11, 1358. [Google Scholar] [CrossRef]
- Mozzi, R.; Buratta, S. Brain Phosphatidylserine: Metabolism and Functions. In Handbook of Neurochemistry and Molecular Neurobiology; Lajtha, A., Tettamanti, G., Goracci, G., Eds.; Springer: Boston, MA, USA, 2010; pp. 39–58. [Google Scholar] [CrossRef]
- Contarini, G.; Povolo, M. Phospholipids in Milk Fat: Composition, Biological and Technological Significance, and Analytical Strategies. Int. J. Mol. Sci. 2013, 14, 2808–2831. [Google Scholar] [CrossRef]
- Hirabayashi, Y.; Furuya, S. Roles of L-serine and sphingolipid synthesis in brain development and neuronal survival. Prog. Lipid Res. 2008, 47, 188–203. [Google Scholar] [CrossRef]
- Küllenberg, D.; Taylor, L.A.; Schneider, M.; Massing, U. Health effects of dietary phospholipids. Lipids Health Dis. 2012, 11. [Google Scholar] [CrossRef]
- Farrell, P.M. The development biochemistry of lung phospholipid metabolism. In Lung Development: Biological and Clinical Perspectives: Biochemistry and Physiology; Farrell, P.M., Ed.; Academic Press: Cambridge, MA, USA, 1982; Volume 1, pp. 223–235. [Google Scholar]
- McJarrow, P.; Schnell, N.; Jumpsen, J.; Clandinin, T. Influence of dietary gangliosides on neonatal brain development. Nutr. Rev. 2009, 67, 451–463. [Google Scholar] [CrossRef]
- Rueda, R. Gangliosides, immunity, infection and inflammation. In Diet, Immunity and Inflammation; Calder, P.C., Yaqoob, P., Eds.; Woodhead Publishing Ltd.: Cambridge, UK, 2013. [Google Scholar]
- Sonnino, S.; Mauri, L.; Ciampa, M.G.; Prinetti, A. Gangliosides as regulators of cell signaling: Ganglioside-protein interactions or ganglioside-driven membrane organization? J. Neurochem. 2013, 124, 432–435. [Google Scholar] [CrossRef] [PubMed]
- Thurl, S.; Munzert, M.; Henker, J.; Boehm, G.; Muller-Werner, B.; Jelinek, J.; Stahl, B. Variation of human milk oligosaccharides in relation to milk groups and lactational periods. Br. J. Nutr. 2010, 104, 1261–1271. [Google Scholar] [CrossRef] [PubMed]
- McGuire, M.K.; Meehan, C.L.; McGuire, M.A.; Williams, J.E.; Foster, J.; Sellen, D.W.; Kamau-Mbuthia, E.W.; Kamundia, E.W.; Mbugua, S.; Moore, S.E.; et al. What’s normal? Oligosaccharide concentrations and profiles in milk produced by healthy women vary geographically. Am. J. Clin. Nutr. 2017, 105, 1086–1100. [Google Scholar] [CrossRef] [PubMed]
- Austin, S.; De Castro, C.A.; Benet, T.; Hou, Y.F.; Sun, H.N.; Thakkar, S.K.; Vinyes-Pares, G.; Zhang, Y.M.; Wang, P.Y. Temporal Change of the Content of 10 Oligosaccharides in the Milk of Chinese Urban Mothers. Nutrients 2016, 8, 346. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; McJarrow, P.; Mohamed, H.; Liu, X.Y.; Welman, A.; Fong, B.Y. Lactational changes in the human milk oligosaccharide concentration in Chinese and Malaysian mothers’ milk. Int. Dairy J. 2018, 87, 1–10. [Google Scholar] [CrossRef]
- Ma, L.; MacGibbon, A.K.H.; Jan Mohamed, H.J.B.; Loy, S.; Rowan, A.; McJarrow, P.; Fong, B.Y. Determination of phospholipid concentrations in breast milk and serum using a high performance liquid chromatography–mass spectrometry–multiple reaction monitoring method. Int. Dairy J. 2017, 71, 50–59. [Google Scholar] [CrossRef]
- Thakkar, S.K.; Giuffrida, F.; Cristina, C.-H.; De Castro, C.A.; Mukherjee, R.; Tran, L.-A.; Steenhout, P.; Lee, L.Y.; Destaillats, F. Dynamics of human milk nutrient composition of women from Singapore with a special focus on lipids. Am. J. Hum. Biol. Off. J. Hum. Biol. Counc. 2013, 25, 770–779. [Google Scholar] [CrossRef]
- Giuffrida, F.; Cruz-Hernandez, C.; Bertschy, E.; Fontannaz, P.; Elmelegy, I.M.; Tavazzi, I.; Marmet, C.; Sanchez-Bridge, B.; Thakkar, S.K.; De Castro, C.A.; et al. Temporal changes of human breast milk lipids of Chinese mothers. Nutrients 2016, 8, 715. [Google Scholar] [CrossRef]
- Ma, L.; MacGibbon, A.K.H.; Jan Mohamed, H.J.B.; Loy, S.; Rowan, A.; McJarrow, P.; Fong, B.Y. Determination of ganglioside concentrations in breast milk and serum from Malaysian mothers using a high performance liquid chromatography-mass spectrometry-multiple reaction monitoring method. Int. Dairy J. 2015, 49, 62–71. [Google Scholar] [CrossRef]
- Ma, L.; Liu, X.; MacGibbon, A.K.H.; Rowan, A.; McJarrow, P.; Fong, B.Y. Lactational changes in concentration and distribution of ganglioside molecular species in human breast milk from Chinese mothers. Lipids 2015, 50, 1145–1154. [Google Scholar] [CrossRef]
- Radwan, H.; Hashim, M.; Obaid, R.S.; Hasan, H.; Naja, F.; Al Ghazal, H.; Mohamed, H.; Rizk, R.; Al Hilali, M.; Rayess, R.; et al. The Molther-Infant Study Cohort (MISC): Methodology, challenges, and baseline characteristics. PLoS ONE 2018, 13, e0198278. [Google Scholar] [CrossRef] [PubMed]
- Svennerholm, L.; Fredman, P. A procedure for the quantitative isolation of brain gangliosides. Biochim. Biophys. Acta 1980, 617, 97–109. [Google Scholar] [CrossRef]
- Fong, B.; Ma, L.; Norris, C. Analysis of phospholipids in infant formulas using high performance liquid chromatography-tandem mass spectrometry. J. Agric. Food Chem. 2013, 61, 858–865. [Google Scholar] [CrossRef] [PubMed]
- Azad, M.B.; Robertson, B.; Atakora, F.; Becker, A.B.; Subbarao, P.; Moraes, T.J.; Mandhane, P.J.; Turvey, S.E.; Lefebvre, D.L.; Sears, M.R.; et al. Human milk oligosaccharide concentrations are associated with multiple fixed and modifiable maternal characteristics, environmental factors, and feeding practices. J. Nutr. 2018, 148, 1733–1742. [Google Scholar] [CrossRef] [PubMed]
- Giuffrida, F.; Elmelegy, I.M.; Thakkar, S.K.; Marmet, C.; Destaillats, F. Longitudinal Evolution of the Concentration of Gangliosides GM3 and GD3 in Human Milk. Lipids 2014, 49, 997–1004. [Google Scholar] [CrossRef] [PubMed]
- Larsson, M.W.; Lind, M.V.; Laursen, R.P.; Yonemitsu, C.; Larnkjaer, A.; Molgaard, C.; Michaelsen, K.F.; Bode, L. Human Milk Oligosaccharide Composition Is Associated with Excessive Weight Gain During Exclusive Breastfeeding-An Explorative Study. Front. Pediatr. 2019, 7, 297. [Google Scholar] [CrossRef] [PubMed]
- Coppa, G.V.; Pierani, P.; Zampini, L.; Carloni, I.; Carlucci, A.; Gabrielli, O. Oligosaccharides in human milk during different phases of lactation. Acta Paediatr. Suppl. 1999, 88, 89–94. [Google Scholar] [CrossRef] [PubMed]
- Bao, Y.W.; Zhu, L.B.; Newburg, D.S. Simultaneous quantification of sialyloligosaccharides from human milk by capillary electrophoresis. Anal. Biochem. 2007, 370, 206–214. [Google Scholar] [CrossRef] [PubMed]
- Hong, Q.T.; Ruhaak, L.R.; Totten, S.M.; Smilowitz, J.T.; German, J.B.; Lebrilla, C.B. Label-Free Absolute Quantitation of Oligosaccharides Using Multiple Reaction Monitoring. Anal. Chem. 2014, 86, 2640–2647. [Google Scholar] [CrossRef] [PubMed]
- Garcia, C.; Millet, V.; Coste, T.C.; Mimoun, M.; Ridet, A.; Antona, C.; Simeoni, U.; Armand, M. French Mothers’ Milk Deficient in DHA Contains Phospholipid Species of Potential Interest for Infant Development. J. Pediatr. Gastroenterol. Nutr. 2011, 53, 206–212. [Google Scholar] [CrossRef] [PubMed]
- Claumarchirant, L.; Cilla, A.; Matencio, E.; Sanchez-Siles, L.M.; Castro-Gomez, P.; Fontecha, J.; Alegria, A.; Lagarda, M.J. Addition of milk fat globule membrane as an ingredient of infant formulas for resembling the polar lipids of human milk. Int. Dairy J. 2016, 61, 228–238. [Google Scholar] [CrossRef]
- Bitman, J.; Freed, L.M.; Neville, M.C.; Wood, D.L.; Hamosh, P.; Hamosh, M. Lipid-composition of prepartum human mammary secretion and postpartum milk. J. Pediatr. Gastroenterol. Nutr. 1986, 5, 608–615. [Google Scholar] [CrossRef] [PubMed]
- Sala-Vila, A.; Castellote, A.I.; Rodriguez-Palmero, M.; Campoy, C.; Lopez-Sabater, M.C. Lipid composition in human breast milk from Granada (Spain): Changes during lactation. Nutrition 2005, 21, 467–473. [Google Scholar] [CrossRef] [PubMed]
- Bitman, J.; Wood, L.; Hamosh, M.; Hamosh, P.; Mehta, N.R. Comparison of the lipid composition of breast milk from mothers of term and preterm infants. Am. J. Clin. Nutr. 1983, 38, 300–312. [Google Scholar] [CrossRef]
- Clark, R.M.; Ferris, A.M.; Fey, M.; Brown, P.B.; Hundrieser, K.E.; Jensen, R.G. Changes in the lipids of human milk from 2 to 16 weeks postpartum. J. Pediatr. Gastroenterol. Nutr. 1982, 1, 311–315. [Google Scholar] [CrossRef]
- Zou, X.Q.; Guo, Z.; Huang, J.H.; Jin, Q.Z.; Cheong, L.Z.; Wang, X.G.; Xu, X.B. Human Milk Fat Globules from Different Stages of Lactation: A Lipid Composition Analysis and Microstructure Characterization. J. Agric. Food Chem. 2012, 60, 7158–7167. [Google Scholar] [CrossRef]
- Harzer, G.; Haug, M.; Dieterich, I.; Gentner, P.R. Changing patterns of human milk lipids in the course of the lactation and during the day. Am. J. Clin. Nutr. 1983, 37, 612–621. [Google Scholar] [CrossRef]
- Jensen, R.G. F-Miscellaneous Factors Affecting Composition and Volume of Human and Bovine Milks. In Handbook of Milk Composition; Jensen, R.G., Ed.; Academic Press: San Diego, CA, USA, 1995; pp. 237–271. [Google Scholar] [CrossRef]
- Da Cunha, J.; da Costa, T.H.M.; Ito, M.K. Influences of maternal dietary intake and suckling on breast milk lipid and fatty acid composition in low-income women from Brasilia, Brazil. Early Hum. Dev. 2005, 81, 303–311. [Google Scholar] [CrossRef]
- Jensen, R.G. Lipids in human milk. Lipids 1999, 34, 1243–1271. [Google Scholar] [CrossRef]
- Jensen, R.G. Handbook of Milk Composition; Academic Press: London, UK, 1995. [Google Scholar]
- Michalski, M.C.; Briard, V.; Michel, F.; Tasson, F.; Poulain, P. Size distribution of fat globules in human colostrum, breast milk, and infant formula. J. Dairy Sci. 2005, 88, 1927–1940. [Google Scholar] [CrossRef]
- Cohen, B.C.; Shamay, A.; Argov-Argaman, N. Regulation of Lipid Droplet Size in Mammary Epithelial Cells by Remodeling of Membrane Lipid Composition-A Potential Mechanism. PLoS ONE 2015, 10. [Google Scholar] [CrossRef] [PubMed]
- Takamizawa, K.; Iwamori, M.; Mutai, M.; Nagai, Y. Gangliosides of Bovine Buttermilk-Isolation and Characterization of a Novel Monosialoganglioside with a New Branching Structure. J. Biol. Chem. 1986, 261, 5625–5630. [Google Scholar]
- Nakano, T.; Sugawara, M.; Kawakami, H. Sialic acid in human milk: Composition and functions. Acta Paediatr. Taiwanica 2001, 42, 11–17. [Google Scholar]
- Rueda, R.; Puente, R.; Hueso, P.; Maldonado, J.; Gil, A. New data on content and distribution of gangliosides in human milk. Biol. Chem. Hoppe-Seyler 1995, 376, 723–727. [Google Scholar] [CrossRef] [PubMed]
- Pan, X.L.; Izumi, T. Chronological changes in the ganglioside composition of human milk during lactation. Early Hum. Dev. 1999, 55, 1–8. [Google Scholar] [CrossRef]
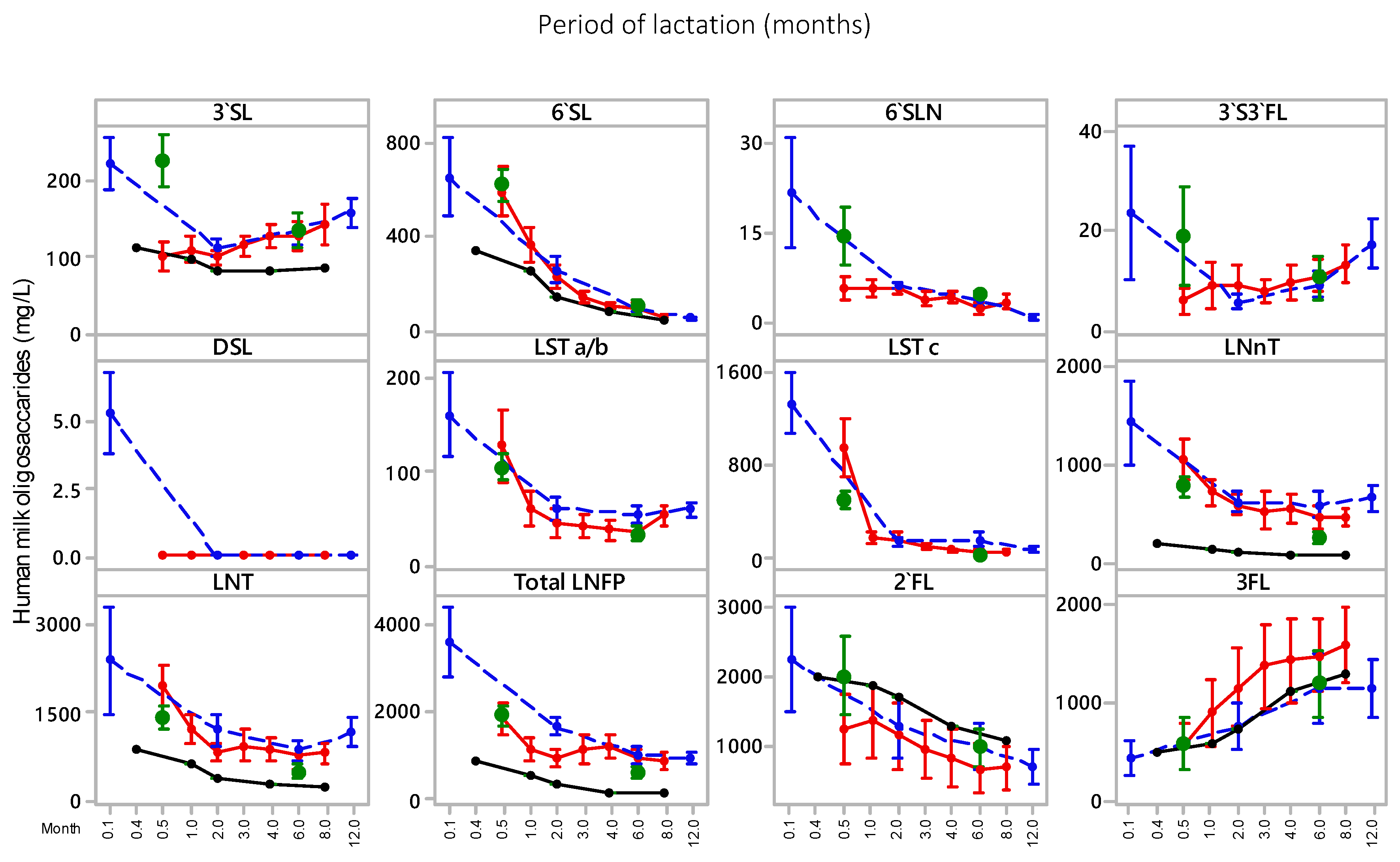
| Sample | 3’SL | 6’SL | 6’SLN | DSL | 3’S3FL | LSTa/b | LSTc | 2’FL | 3FL | LNnT | LNT | LNFP | Total HMO |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Transitional (all) (n = 41) | 226 ± 107 a | 621 ± 212 a | 15 ± 15 a | 2.2 ± 2.3 a | 19 ± 21 a | 104 ± 46 a | 488 ± 224 a | 2021 ± 1776 a | 581 ± 868 a | 765 ± 350 a | 1429 ± 693 a | 1932 ± 762 a | 8204 ± 2388 a |
| Non-secretor | 256 ± 144 | 562 ± 232 | 20 ± 23 | 1.8 ± 1.9 | 48 ± 49 | 124 ± 52 | 456 ± 243 | 4.3 ± 8.6 | 1599 ± 119 | 990 ± 524 | 1917 ± 973 | 1490 ± 528 | 7466 ± 1812 |
| Secretor | 216 ± 91 | 643 ± 204 | 13 ± 11 | 2.4 ± 2.4 | 8.1 ± 7.0 | 98 ± 42 | 500 ± 220 | 2761 ± 1497 # | 208 ± 112 # | 682 ± 220 | 1250 ± 460 | 2094 ± 777 # | 8475 ± 2541 |
| Mature (all) (n = 40) | 134 ± 69 b | 91 ± 108 b | 5 ± 1 b | 0.2 ± 0.4 b | 10 ± 14 a | 31 ± 25 b | 11 ± 8 b | 997 ± 885 a | 1194 ± 106 b | 250 ± 188 b | 504 ± 337 b | 650 ± 416 b | 3876 ± 1403 b |
| Non-secretor | 181 ± 98 | 81 ± 52 | 4.9 ± 2.2 | 0.1 ± 0.2 | 25 ± 20 | 28 ± 18 | 6.7 ± 5.3 | 4.0 ± 2.8 | 2526 ± 113 | 187 ± 113 | 420 ± 276 | 548 ± 300 | 4009 ± 1104 |
| Secretor | 116 ± 44 | 95 ± 124 | 4.4 ± 0.6 | 0.3 ± 0.5 | 4.6 ± 2.4 # | 32 ± 28 | 13 ± 8 # | 1374 ± 746 # | 688 ± 398 # | 273 ± 207 | 536 ± 357 | 689 ± 451 | 3826 ± 1515 |
| Time Point | Phospholipids | Gangliosides | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| PI | PE | PC | SM | PS | L-PE | L-PC | Total PL | GM3 | GD3 | Total GA | |
| Transitional (n = 41) | 11.2 ± 5.5 a (4%) | 66.3 ± 27.16 a (25%) | 66.4 ± 32.87 a (25%) | 91.2 ± 26.38 a (34%) | 28.5 ± 13.29 a (7%) | 3.7 ± 2.37 a (1.4%) | 1.7 ± 0.98 a (0.6%) | 269.0 ± 89.2 a | 9.47 ± 8.37 a (45%) | 11.71 ± 9.46 a (55%) | 21.18 ± 11.46 a |
| Mature (n = 40) | 6.5 ± 3.61 b (3%) | 80.0 ± 35.35 a (36%) # | 30.2 ± 22.07 b (14%) # | 82.9 ± 29.21 a (38%) | 16.1 ± 6.99 b (7%) | 3.1 ± 1.99 a (1.4%) | 0.9 ± 0.63 b (0.4%) | 219.6 ± 85.0 b | 18.62 ± 9.69 b (92%) # | 1.57 ± 2.24 b (8%) # | 20.18 ± 9.75 a |
| Reference | Milk (Post Partum) | n | 3’SL | 6’SL | 6’SLN | DSL | 3’S3FL | LSTa/b | LSTc | 2’FL | 3FL | LNnT | LNT | Total LNFP |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Larsson et al. [41] a,b Denmark | Mature (5 month) | 15 | 492 | 156 | 149 | 33 | 2989 | 229 | 578 | 703 | 2692 | |||
| Coppa et al. [42] Italy | Transition (4 days) | 18 | 3930 | 340 | 2040 | 840 | 1650 | |||||||
| Bao et al. [43] USA | Transition (9–21 days) | 76 | 396 | 74 c | 148 | |||||||||
| Austin et al. [20] (Table 4) China | Transition (5–11 days) | 90 | 110 | 340 | 2600 | 510 | 180 | 880 | 1157 | |||||
| Mature (4–8 months) | 90 | 83 | 45 | 1300 | 1300 | 59 | 250 | 199 | ||||||
| Ma et al. [21] China | Transition (14 days) | 20 | 100 | 593 | 6 | 3 | 6 | 127 | 941 | 1281 | 543 | 1033 | 1979 | 1870 |
| Mature (6 months) | 20 | 127 | 83 | 2 | 0 | 11 | 33 | 47 | 704 | 1476 | 446 | 785 | 945 | |
| Ma et al. [21] Malaysia | Mature (6 months) | 21 | 135 | 84 | 4 | 0 | 9 | 84 | 145 | 1003 | 1146 | 571 | 867 | 1036 |
| This study UAE | Transition (6–14 days) | 41 | 226 | 621 | 15 | 2 | 19 | 104 | 488 | 2021 | 581 | 765 | 1429 | 1932 |
| This study UAE | Mature (6 months) | 40 | 134 | 91 | 5 | 0 | 10 | 31 | 11 | 997 | 1194 | 250 | 504 | 650 |
| Total PL Ranges (mg/L) | Country | Reference | |
|---|---|---|---|
| Transitional Milk | Mature Milk | ||
| 390 ± 50‒440 ± 73 (8‒15, 20) | 370 ± 106‒405 ± 80 (22–36, 40) | Germany | Harzer et al. [52] |
| 310 ± 30 (11, 5) | 270 ± 30 (23, 5 pooled) | USA | Bitman et al. [47] |
| 158 (11, 17) | 114 (23, 19) | Spain | Sala-Vila et al. [48] |
| 148 (7, 6) | 133–227 (20–84, 6) | USA | Bitman et al. [49] |
| 973 (14, 10) | 1023‒1298 (42‒112, 10) | USA | Clark et al. [50] |
| 185 (6‒15, 45) | 182 (>16, 45) | Denmark | Zou et al. [51] |
| 550 ± 260 (6–10, 7) | 450 ± 260 (30, 16) | France | Gracia et al. [45] |
| 230 ± 49–242 ± 82 (30–120, 50) | Singapore | Thakkar et al. [32] | |
| 437 ± 23–535 ± 26 (6–15, 44) | 260 ± 3–422 ± 13 (16–360, 44) | Spain | Claumarchirant et al. [46] |
| 266 ± 57 (6‒14, 12) | 170 ± 80‒219 ± 92 (60–365, 132) | Malaysia | Ma et al. [31] |
| 285 ± 144 (6–15, 81) | 242 ± 114 (16–240, 345) | China | Guiffrida et al. [33] |
| 269 ± 89 (6–14, 41) | 220 ± 85 (180, 40) | UAE | This study |
| Reference | Milk | n | GM3 | GD3 | Total GA |
|---|---|---|---|---|---|
| Giuffrida et al. [40] China | Colostrum/Transition (0–11 days) | 450 | 3.8 ± 0.4 (47) | 4.3 ± 0.9 (53) | 8.1 |
| Ma et al. [34] Malaysia | Transition | 12 | 8.3 ± 4.8 (44) | 10.6 ± 4.3 (56) | 18.9 ± 6.6 |
| This Study UAE | Transition (5–15 days) | 41 | 9.5 ± 8.4 (45) | 11.7 ± 9.5 (55) | 21.2 ± 11.5 |
| Ma et al. [34] Malaysia | Mature (6 months) | 42 | 21.4 ± 13 (85) | 4.3 ± 5.5 (15) | 25.3 ± 15.7 |
| Ma et al. [35] China | Mature (6 months) | 20 | 21.4 ± 9.5 (93) | 1.5 ± 1.4 (7) | 22.9 ± 9.9 |
| This Study UAE | Mature (6 Months) | 40 | 18.6 ± 9.7 (92) | 1.6 ± 2.2 (8) | 20.2 ± 9.8 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
McJarrow, P.; Radwan, H.; Ma, L.; MacGibbon, A.K.H.; Hashim, M.; Hasan, H.; Obaid, R.S.; Naja, F.; Mohamed, H.J.J.; Al Ghazal, H.; et al. Human Milk Oligosaccharide, Phospholipid, and Ganglioside Concentrations in Breast Milk from United Arab Emirates Mothers: Results from the MISC Cohort. Nutrients 2019, 11, 2400. https://doi.org/10.3390/nu11102400
McJarrow P, Radwan H, Ma L, MacGibbon AKH, Hashim M, Hasan H, Obaid RS, Naja F, Mohamed HJJ, Al Ghazal H, et al. Human Milk Oligosaccharide, Phospholipid, and Ganglioside Concentrations in Breast Milk from United Arab Emirates Mothers: Results from the MISC Cohort. Nutrients. 2019; 11(10):2400. https://doi.org/10.3390/nu11102400
Chicago/Turabian StyleMcJarrow, Paul, Hadia Radwan, Lin Ma, Alastair K.H. MacGibbon, Mona Hashim, Hayder Hasan, Reyad Shaker Obaid, Farah Naja, Hamid Jan Jan Mohamed, Hessa Al Ghazal, and et al. 2019. "Human Milk Oligosaccharide, Phospholipid, and Ganglioside Concentrations in Breast Milk from United Arab Emirates Mothers: Results from the MISC Cohort" Nutrients 11, no. 10: 2400. https://doi.org/10.3390/nu11102400
APA StyleMcJarrow, P., Radwan, H., Ma, L., MacGibbon, A. K. H., Hashim, M., Hasan, H., Obaid, R. S., Naja, F., Mohamed, H. J. J., Al Ghazal, H., & Fong, B. Y. (2019). Human Milk Oligosaccharide, Phospholipid, and Ganglioside Concentrations in Breast Milk from United Arab Emirates Mothers: Results from the MISC Cohort. Nutrients, 11(10), 2400. https://doi.org/10.3390/nu11102400







