Lactoferrin in Human Milk of Prolonged Lactation
Abstract
1. Introduction
2. Material and Methods
2.1. Analysis of the Samples
2.2. Skim Milk Sample Preparation
2.3. Analysis of Lactoferrin in Skim Milk
2.4. Statistical Analysis
3. Results
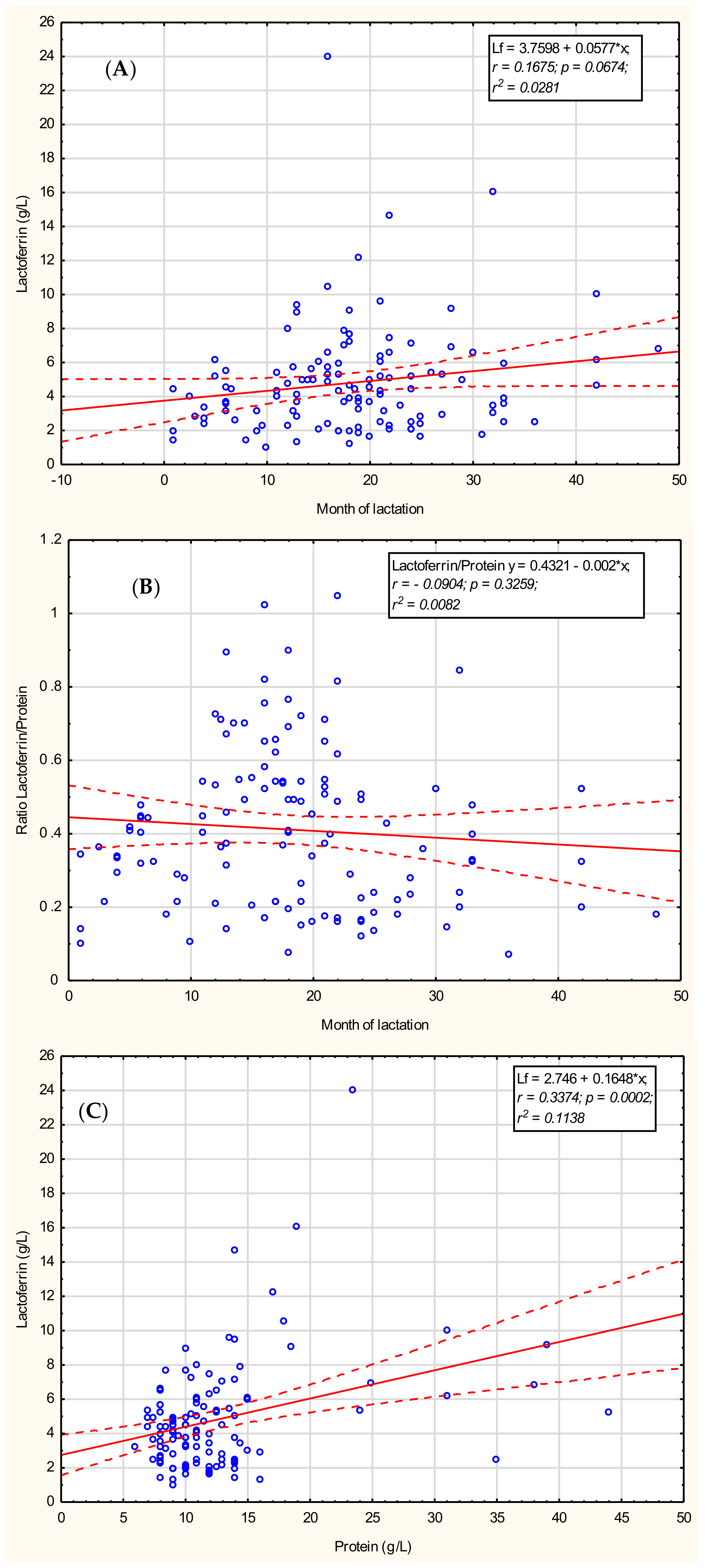
3.1. Lactoferrin
3.2. Total Protein
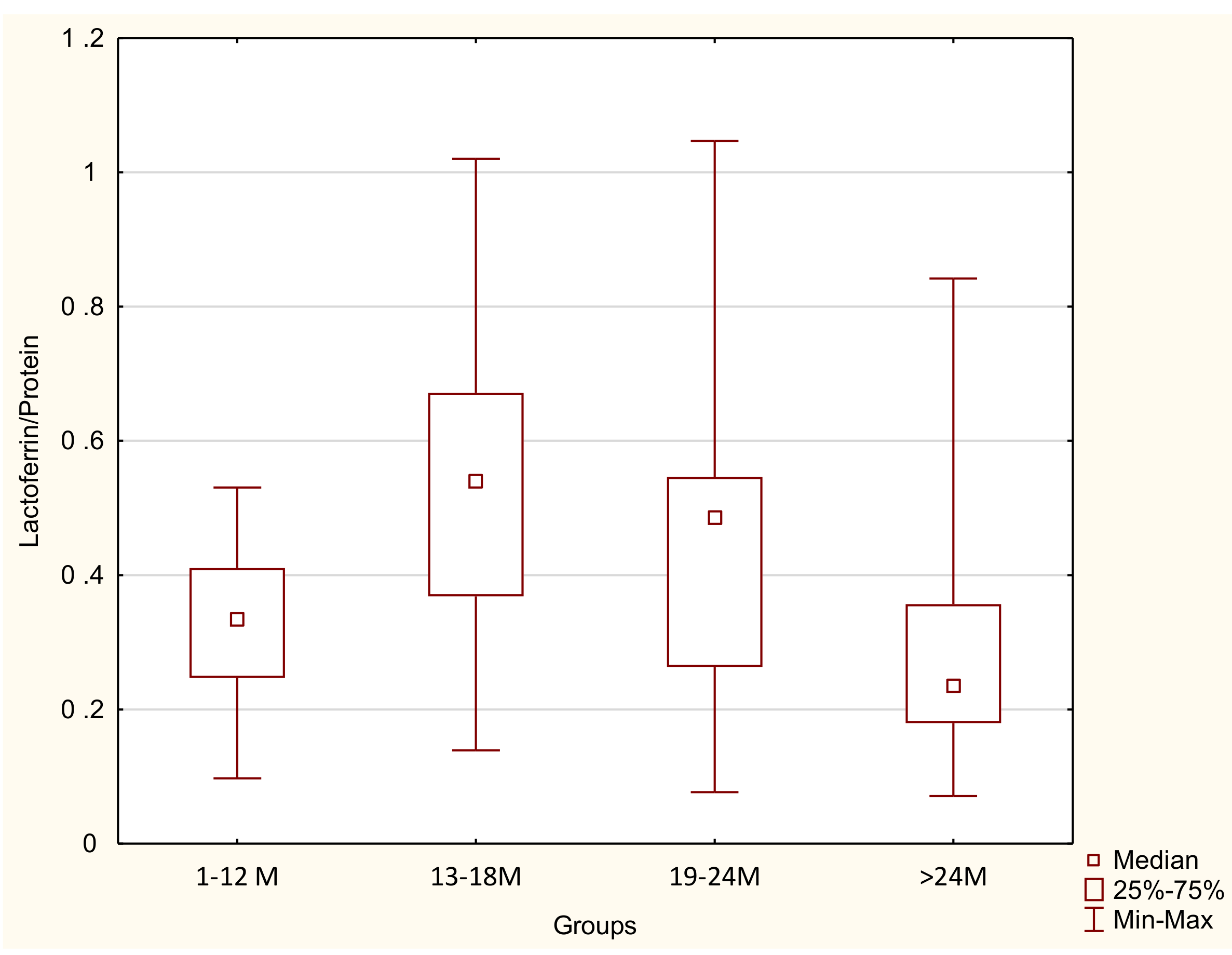
3.3. Lactoferrin/Protein Ratio
3.4. Lactoferrin Concentration in Relation to Macronutrients
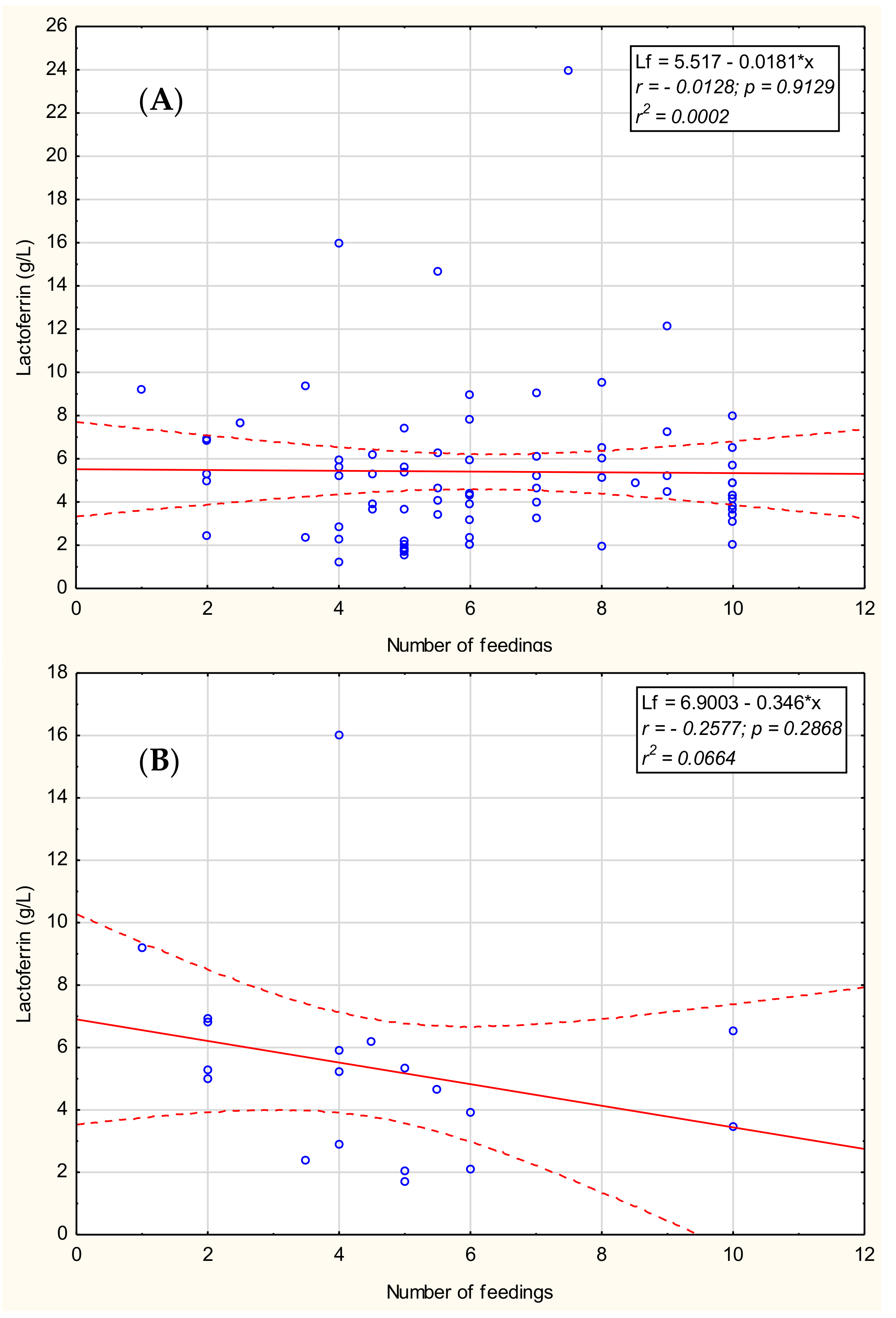
3.5. Lactoferrin Concentration in Breast Milk in Relation to the Number of Feedings
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ballard, O.; Morrow, A.L. Human milk composition: Nutrients and bioactive factors. Pediatr. Clin. N. Am. 2013, 60, 49–74. [Google Scholar] [CrossRef] [PubMed]
- Sorensen, M.; Sorensen, S.P.L. The proteins in whey. Compte Rendu Trav. Lab. Carlsberg 1939, 23, 55–99. [Google Scholar]
- Groves, M.L. The isolation of a red protein from milk. J. Am. Chem. Soc. 1960, 83, 3345–3350. [Google Scholar] [CrossRef]
- Johansson, B. Isolation of an iron-containing red protein from human milk. Acta Chem. Scand. 1960, 14, 510–512. [Google Scholar] [CrossRef]
- Montreuil, J.; Tonnelat, J.; Mullet, S. Preparation and properties of lactosiderophilin (lactotransferrin) of human milk. Biochim. Biophys. Acta 1960, 45, 413–421. [Google Scholar] [CrossRef]
- Rosa, L.; Cutone, A.; Lepanto, M.S.; Paesano, R.; Valenti, P. Lactoferrin: A Natural Glycoprotein Involved in Iron and Inflammatory Homeostasis. Int. J. Mol. Sci. 2017, 18, 1985. [Google Scholar] [CrossRef] [PubMed]
- Liao, Y.; Alvarado, R.; Phinney, B.; Lönnerdal, B. Proteomic characterization of human milk whey proteins during a twelve-month lactation period. J. Proteome Res. 2011, 10, 1746–1754. [Google Scholar] [CrossRef]
- Lönnerdal, B.; Iyer, S. Lactoferrin: Molecular structure and biological function. Annu. Rev. Nutr. 1995, 15, 93–110. [Google Scholar] [CrossRef]
- Vogel, H.J. Lactoferrin, a bird’s eye view. Biochem. Cell Biol. 2012, 90, 233–244. [Google Scholar] [CrossRef]
- Lönnerdal, B. Nutritional roles of lactoferrin. Curr. Opin. Clin. Nutr. Metab. Care 2009, 12, 293–297. [Google Scholar] [CrossRef]
- Zavaleta, N.; Figueroa, D.; Rivera, J.; Julia, S.; Segundo, A.; Bo, L. Efficacy of rice-based oral rehydration solution containing recombinant human lactoferrin and lysozyme in Peruvian children with acute diarrhea. J. Pediatr. Gastroenterol. Nutr. 2007, 44, 258–264. [Google Scholar] [CrossRef] [PubMed]
- Ochoa, T.J.; Chea-Woo, E.; Baiocchi, N.; Pecho, I.; Campos, M.; Prada, A.; Valdiviezo, G.; Lluque, A.; Lai, D.; Cleary, T.G. Randomized double-blind controlled trial of bovine lactoferrin for prevention of diarrhea in children. J. Pediatr. 2013, 162, 349–356. [Google Scholar] [CrossRef] [PubMed]
- Manzoni, P.; Rinaldi, M.; Cattani, S.; Pugni, L.; Romeo, M.G.; Messner, H.; Stolfi, I.; Decembrino, L.; Laforgia, N.; Vagnarelli, F.; et al. Bovine lactoferrin supplementation for prevention of late-onset sepsis in very low-birth-weight neonates: A randomized trial. JAMA 2009, 302, 1421–1428. [Google Scholar] [CrossRef] [PubMed]
- Manzoni, P.; Garcia Sanchez, R.; Meyer, M.; Stolfi, I.; Pugni, L.; Messner, H.; Cattani, S.; Betta, P.M.; Memo, L.; Decembrino, L. Exposure to gastric acid inhibitors increases the risk of infection in preterm very low birth weight infants but concomitant administration of lactoferrin counteracts this effect. J. Pediatr. 2018, 193, 62–67. [Google Scholar] [CrossRef] [PubMed]
- Pammi, M.; Suresh, G. Enteral lactoferrin supplementation for prevention of sepsis and necrotizing enterocolitis in preterm infants. Cochrane Database Syst. Rev. 2017, 6, CD007137. [Google Scholar] [CrossRef] [PubMed]
- Mastromarino, P.; Capobianco, D.; Campagna, G.; Laforgia, N.; Drimaco, P.; Dileone, A.; Baldassarre, M.E. Correlation between lactoferrin and beneficial microbiota in breast milk and infant’s feces. Biometals 2014, 27, 1077–1086. [Google Scholar] [CrossRef] [PubMed]
- Broadhurst, M.; Beddis, K.; Black, J.; Henderson, H.; Nair, A.; Wheeler, T. Effect of gestation length on the levels of five innate defence proteins in human milk. Early Hum. Dev. 2015, 91, 7–11. [Google Scholar] [CrossRef]
- Villavicencio, A.; Rueda, M.S.; Turin, C.G.; Ochoa, T.J. Factors affecting lactoferrin concentration in human milk: How much do we know? Biochem. Cell Biol. 2016, 95, 12–21. [Google Scholar] [CrossRef]
- Ronayne de Ferrer, P.A.; Baroni, A.; Sambucetti, M.E.; López, N.E.; Cernadas, J.M.C. Lactoferrin levels in term and preterm milk. J. Am. Coll. Nutr. 2000, 19, 370–373. [Google Scholar] [CrossRef]
- Lönnerdal, B. Law infant nutrition: Bioactive proteins of human milk and implications for composition of infant formulas. Am. J. Clin. Nutr. 2014, 99, 712S–717S. [Google Scholar] [CrossRef]
- Law, B.A.; Reiter, B. The isolation and bacteriostatic properties of lactoferrin from bovine milk whey. J. Dairy Res. 1977, 44, 595–599. [Google Scholar] [CrossRef] [PubMed]
- Ostan, N.K.; Yu, R.H.; Ng, D.; Lai, C.C.-L.; Pogoutse, A.K.; Sarpe, V.; Hepburn, M.; Sheff, J.; Raval, S.; Schriemer, D.C.; et al. Lactoferrin binding protein B–a bi-functional bacterial receptor protein. PLoS Pathog. 2017, 13, e1006244. [Google Scholar] [CrossRef] [PubMed]
- Barbiroli, A.; Bonomi, F.; Capretti, G.I.; Iametti, S.; Manzoni, M.; Piergiovanni, L.; Rollini, M. Antimicrobial activity of lysozyme and lactoferrin incorporated in cellulose-based food packaging. Food Control 2012, 26, 387–392. [Google Scholar] [CrossRef]
- Zarember, K.A.; Sugui, J.A.; Chang, Y.C.; Kwon-Chung, K.J.; Gallin, J.I. Human polymorphonuclear leukocytes inhibit Aspergillus fumigatus conidial growth by lactoferrin-mediated iron depletion. J. Immunol. 2007, 178, 6367–6373. [Google Scholar] [CrossRef]
- Legrand, D. Overview of Lactoferrin as a Natural Immune Modulator. J. Pediatr. 2016, 173, S10–S15. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Timilsena, Y.P.; Blanch, E.; Adhikari, B. Lactoferrin: Structure, Function, Denaturation and Digestion. Crit. Rev. Food Sci. Nutr. 2019, 59, 580–596. [Google Scholar] [CrossRef]
- Hassiotou, F.; Geddes, D.T. Immune Cell–Mediated Protection of the Mammary Gland and the Infant during Breastfeeding. Adv Nutr. 2015, 6, 267–275. [Google Scholar] [CrossRef] [PubMed]
- Vorbach, C.; Capecchi, M.R.; Penninger, J.M. Evolution of the mammary gland from the innate immune system? Bioessays 2006, 28, 606–616. [Google Scholar] [CrossRef]
- Czosnykowska-Łukacka, M.; Królak-Olejnik, B.; Orczyk-Pawiłowicz, M. Breast Milk Macronutrient Components in Prolonged Lactation. Nutrients 2018, 10, 1893. [Google Scholar] [CrossRef]
- Orczyk-Pawiłowicz, M.; Hirnle, L.; Berghausen-Mazur, M.; Kątnik-Prastowska, I.M. Lactation stage-related expression of sialylated and fucosylated glycotopes of human milk α-1-acid glycoprotein. Breastfeed Med. 2014, 9, 313–319. [Google Scholar] [CrossRef]
- Wesolowska, A.; Sinkiewicz-Darol, E.; Barbarska, O.; Strom, K.; Rutkowska, M.; Karzel, K.; Rosiak, E.; Oledzka, G.; Orczyk-Pawiłowicz, M.; Rzoska, S.; et al. New Achievements in High-Pressure Processing to Preserve Human Milk Bioactivity. Front. Pediatr. 2018, 16, 323. [Google Scholar] [CrossRef] [PubMed]
- Rai, D.; Adelman, A.S.; Zhuang, W.; Rai, G.P.; Boettcher, J.; Lönnerdal, B. Longitudinal changes in lactoferrin concentrations in human milk: A global systematic review. Crit. Rev. Food Sci. Nutr. 2014, 54, 1539–1547. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Jiang, R.; Chen, Q.; Wang, J.; Duan, Y.; Pang, X.; Jiang, S.; Bi, Y.; Zhang, H.; Lönnerdal, B.; et al. Concentration of Lactoferrin in Human Milk and Its Variation during Lactation in Different Chinese Populations. Nutrients 2018, 10, 1235. [Google Scholar] [CrossRef] [PubMed]
- Haschke, F.; Haiden, N.; Thakkar, S.K. Nutritive and bioactive proteins in breastmilk. Ann. Nutr. Metab. 2016, 69, 16–26. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Rodenas, C.L.; De Castro, C.A.; Jenni, R.; Thakkar, S.K.; Beauport, L.; Tolsa, J.-F.; Fischer-Fumeaux, C.J.; Affolter, M. Temporal changes of major protein concentrations in preterm and term human milk. A prospective cohort study. Clin. Nutr. 2019, 38, 1844–1852. [Google Scholar] [CrossRef] [PubMed]
- Affolter, M.; Garcia-Rodenas, C.; Vinyes-Pares, G.; Jenni, R.; Roggero, I.; Avanti-Nigro, O.; de Castro, C.A.; Zhao, A.; Zhang, Y.; Wang, P.; et al. Temporal changes of protein composition in breast milk of Chinese Urban mothers and impact of caesarean section delivery. Nutrients 2016, 8, 504. [Google Scholar] [CrossRef] [PubMed]
- Trend, S.; Strunk, T.; Lloyd, M.; Kok, C.H.; Metcalfe, J.; Geddes, D.T.; Lai, C.T.; Richmond, P.; Doherty, D.A.; Simmer, K.; et al. Levels of innate immune factors in preterm and term mothers’ breast milk during the 1st month postpartum. Br. J. Nutr. 2016, 115, 1178–1193. [Google Scholar] [CrossRef]
- Woodman, T.; Strunk, T.; Patole, S.; Hartmann, B.; Simmer, K.; Currie, A. Effects of lactoferrin on neonatal pathogens and Bifidobacterium breve in human breast milk. PLoS ONE 2018, 13, e0201819. [Google Scholar] [CrossRef]
- Ochoa, T.J.; Pezo, A.; Cruz, K.; Chea-Woo, E.; Cleary, T.G. Clinical studies of lactoferrin in children. Biochem. Cell Biol. 2012, 90, 457–467. [Google Scholar] [CrossRef] [PubMed]
- Siqueiros-Cendón, T.; Arévalo-Gallegos, S.; Iglesias-Figueroa, B.F.; García-Montoya, I.A.; Salazar-Martínez, J.; Rascón-Cruz, Q. Immunomodulatory effects of lactoferrin. Acta Pharmacol. Sin. 2014, 35, 557–566. [Google Scholar] [CrossRef]
- Newburg, S.; Walker, W.A. Protection of the neonate by the innate immune system of developing gut and of human milk. Pediatr. Res. 2007, 61, 2. [Google Scholar] [CrossRef] [PubMed]
| Outcome and Exposure Variables Breastfeeding | Breastfeeding ≤12 Months n = 24% (n/N) | Breastfeeding >12 Months n = 96% (n/N) | χ2 | p-Value | |
|---|---|---|---|---|---|
| Maternal age % (n/N) | |||||
| 25–29 | 50 (12/24) | 33 (31/94) | 2.672 | 0.139 | NS |
| 30–34 | 41.7 (10/24) | 45 (41/94) | |||
| 35+ | 8.3 (2/24) | 22 (22/94) | |||
| Race/ethnicity | |||||
| Caucasian | 100 (24/24) | 100 (96/96) | - | - | - |
| Socioeconomic status and education | |||||
| Secondary education | 12.5 (3/24) | 20 (16/96) | 0.25 | 0.617 | NS |
| High education | 87.5 (21/24) | 83.3 (80/96) | |||
| Parity | |||||
| 1 | 79 (19/24) | 67.7 (65/96) | 1.628 | 0.22 | NS |
| 2 | 21 (5/24) | 33.3 (32/96) | |||
| 3 | 0 (0/24) | 2 (2/96) | |||
| Birth weight | |||||
| Appropriate for Gestational Age (AGA) | 100 (24/24) | 100 (96/96) | - | - | - |
| gestational age | |||||
| 34–37 | 4.2(1/24) | 1. (1/96) | 1.144 | 0.285 | NS |
| >37 | 95.8(23/24) | 99 (95/96) | |||
| Medicines during lactation | |||||
| No medications | 71(17/24) | 66 (63/96) | 0.884 | 0.643 | NS |
| Thyroxine | 21(5/24) | 29 (28/96) | |||
| Others | 8(2/24) | 5 (5/96) | |||
| Maternal diet during lactation | |||||
| Vegan/vegetarian | 0 (0/24) | 8 (8/96) | - | - | - |
| Dairy-free diet | 21 (5/24) | 7 (7/96) | |||
| Gluten-free diet | 0 (0/24) | 3 (3/96) | |||
| Complementary foods introduction | |||||
| above 6 months of life | NA | 94 (90/96) | - | - | - |
| Breast Milk. Lactoferrin/Protein Content | Lactation | |||
|---|---|---|---|---|
| 1–12 Months n = 24 | 13–18 Months n = 33 | 19–24 Months n = 37 | >24 Months n = 26 | |
| Lactoferrin (g/L) | 3.39 ± 1.43 3.24 2.30–4.47 | 5.55 ± 4.00 * 4.92 3.69–6.08 p < 0.006 | 5.02 ± 2.97 4.42 3.16–6.53 | 4.90 ± 3.18 4.29 2.49–6.18 |
| Protein (g/L) | 10.5 ± 2.3 10.0 8.5–12.8 | 10.4 ± 3.4 9.0 8.0–11.0 | 11.2 ± 2.7 ** 11.0 9.5–12.0 p < 0.05 | 19.1 ± 10.7 *** 14.3 12.0–25.0 p < 0.0001 |
| Lactoferrin/Protein ratio | 0.32 ± 0.12 0.33 0.25–0.41 | 0.49 ± 0.21 * 0.54 0.36–0.65 p < 0.002 | 0.43 ± 0.21 0.45 0.22–0.54 | 0.29 ± 0.17 *** 0.24 0.18–0.36 p < 0.009 |
| Correlation Coefficient (r Value *) | |||||
|---|---|---|---|---|---|
| Lactoferrin | |||||
| Duration of lactation | 1–48 months | 1–12 months | 13–18 months | 19–24 months | Over 24 months |
| NS | NS | NS | NS | 0.39 | |
| Carbohydrate | –0.32 | NS | NS | NS | –0.50 |
| Fat | 0.19 | NS | NS | NS | 0.58 |
| Protein | 0.25 | NS | NS | NS | 0.56 |
| True protein | 0.24 | NS | NS | NS | 0.58 |
| Dry mass | 0.18 | NS | NS | NS | 0.65 |
| Energy | 0.19 | NS | NS | NS | 0.65 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Czosnykowska-Łukacka, M.; Orczyk-Pawiłowicz, M.; Broers, B.; Królak-Olejnik, B. Lactoferrin in Human Milk of Prolonged Lactation. Nutrients 2019, 11, 2350. https://doi.org/10.3390/nu11102350
Czosnykowska-Łukacka M, Orczyk-Pawiłowicz M, Broers B, Królak-Olejnik B. Lactoferrin in Human Milk of Prolonged Lactation. Nutrients. 2019; 11(10):2350. https://doi.org/10.3390/nu11102350
Chicago/Turabian StyleCzosnykowska-Łukacka, Matylda, Magdalena Orczyk-Pawiłowicz, Barbara Broers, and Barbara Królak-Olejnik. 2019. "Lactoferrin in Human Milk of Prolonged Lactation" Nutrients 11, no. 10: 2350. https://doi.org/10.3390/nu11102350
APA StyleCzosnykowska-Łukacka, M., Orczyk-Pawiłowicz, M., Broers, B., & Królak-Olejnik, B. (2019). Lactoferrin in Human Milk of Prolonged Lactation. Nutrients, 11(10), 2350. https://doi.org/10.3390/nu11102350





