Exogenous Ketones Lower Blood Glucose Level in Rested and Exercised Rodent Models
Abstract
1. Introduction
2. Material and Methods
2.1. Animals
2.2. Diets and Ketogenic Compounds
2.3. Treatment Groups
2.4. Exercise with Accelerated Rotarod
2.5. Measurement of Blood R-βHB and Glucose
2.6. Statistics
3. Results
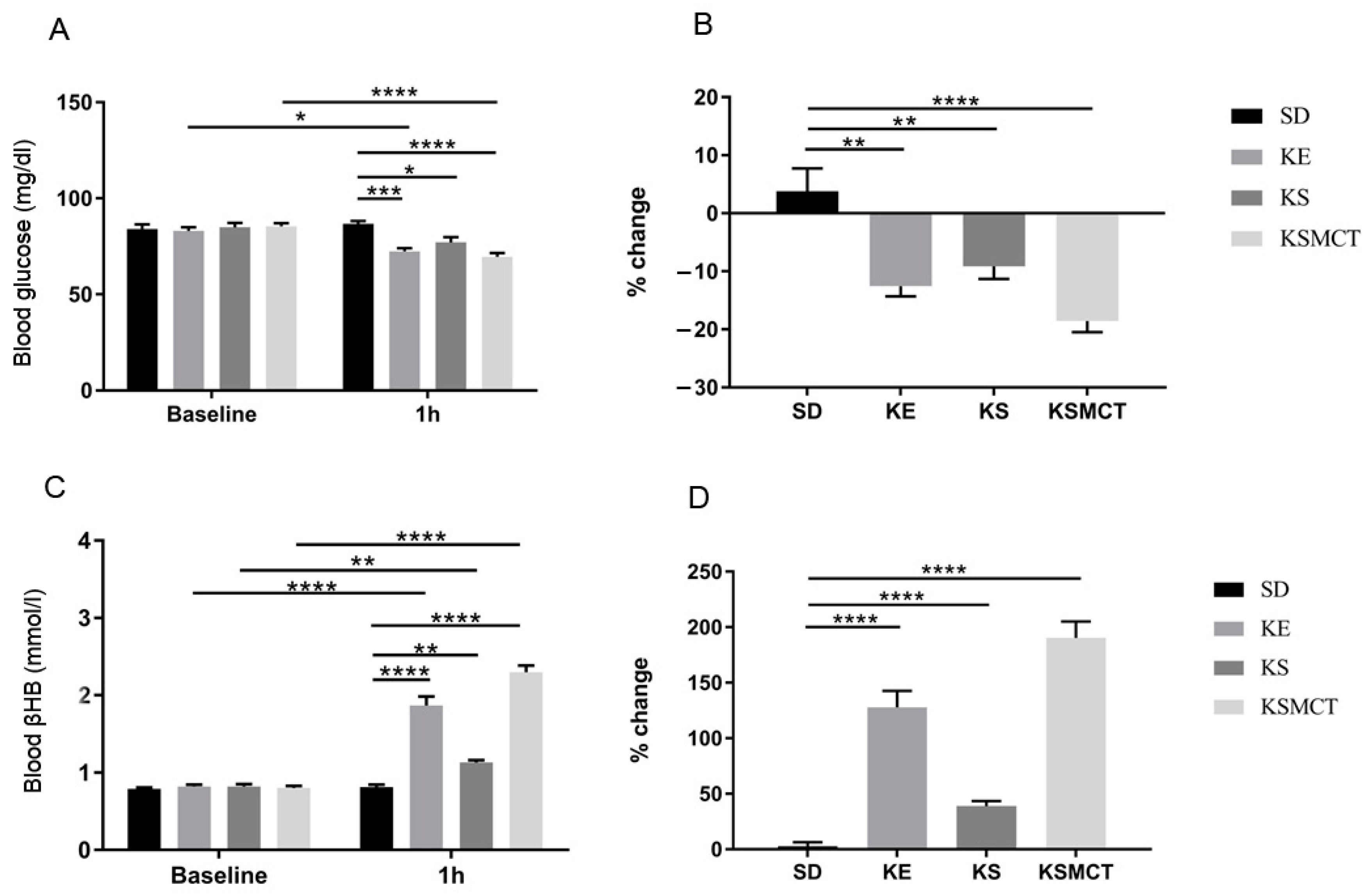
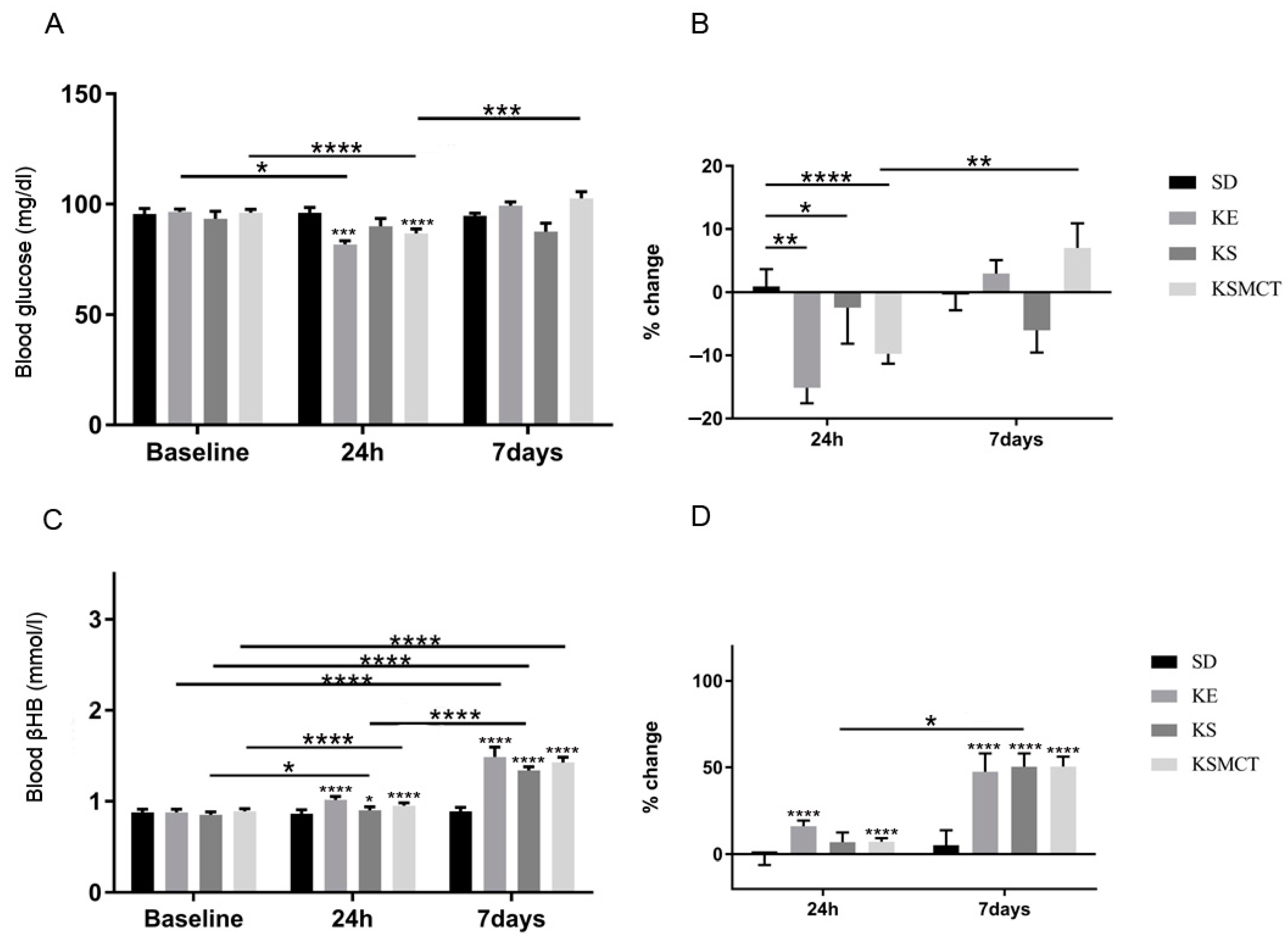
3.1. Acute Effect of Ketone Supplements on Blood Glucose and R-βHB Levels in Exercised Sprague-Dawley Rats
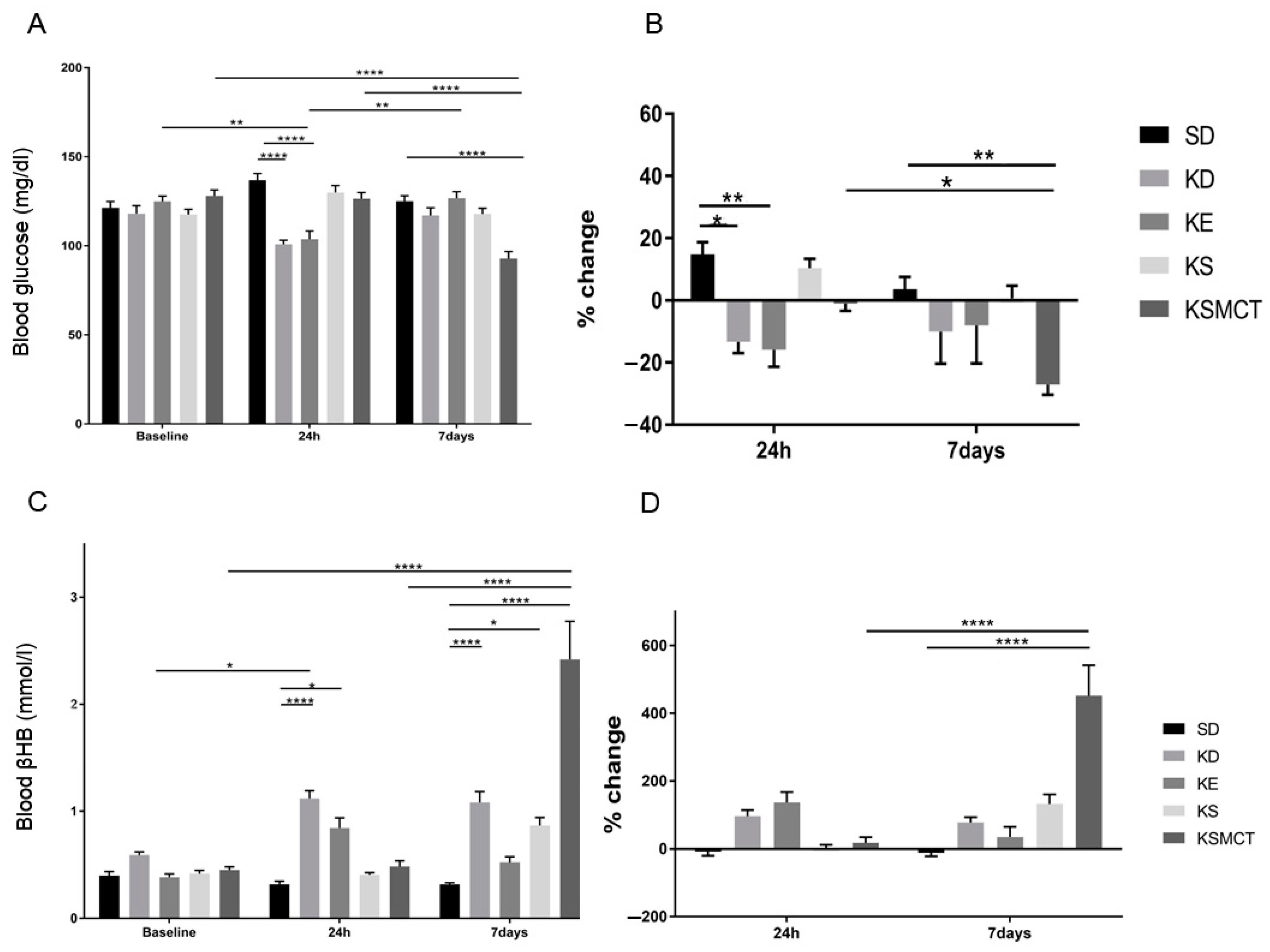
3.2. Sub-Chronic Effect of Ketone Supplements on Blood Glucose and R-βHB Levels in Exercised Sprague-Dawley Rats
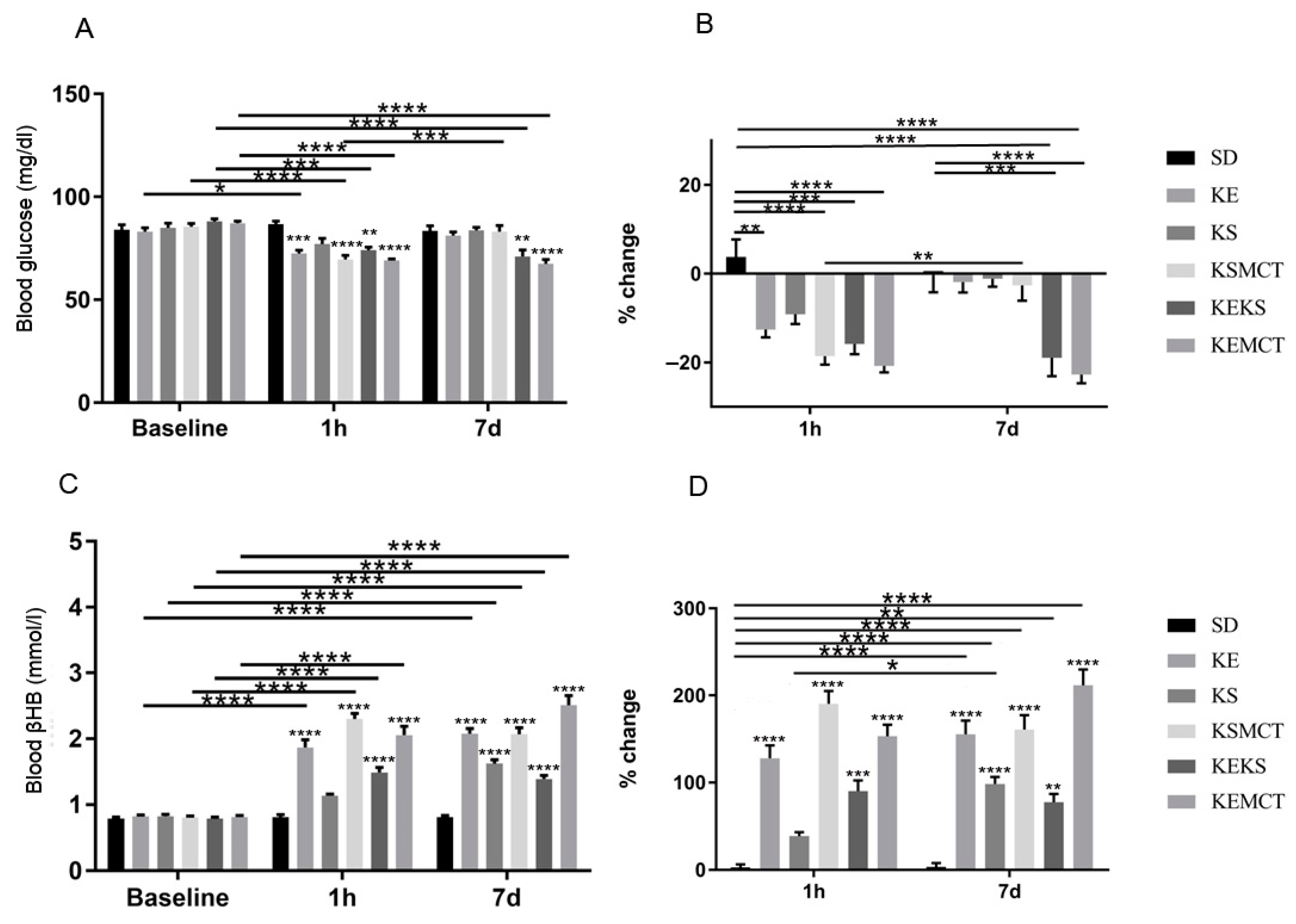
3.3. Acute Effect of Ketone Supplements on Blood Glucose and R-βHB Levels in Rested WAG/Rij Rats
3.4. Sub-Chronic Effects of Ketone Supplements on Blood Glucose and R-βHB Levels in Rested WAG/Rij Rats
3.5. Acute and Sub-Chronic Effects of Ketone Supplements on Blood Glucose and R-βHB Levels in Exercised WAG/Rij Rats
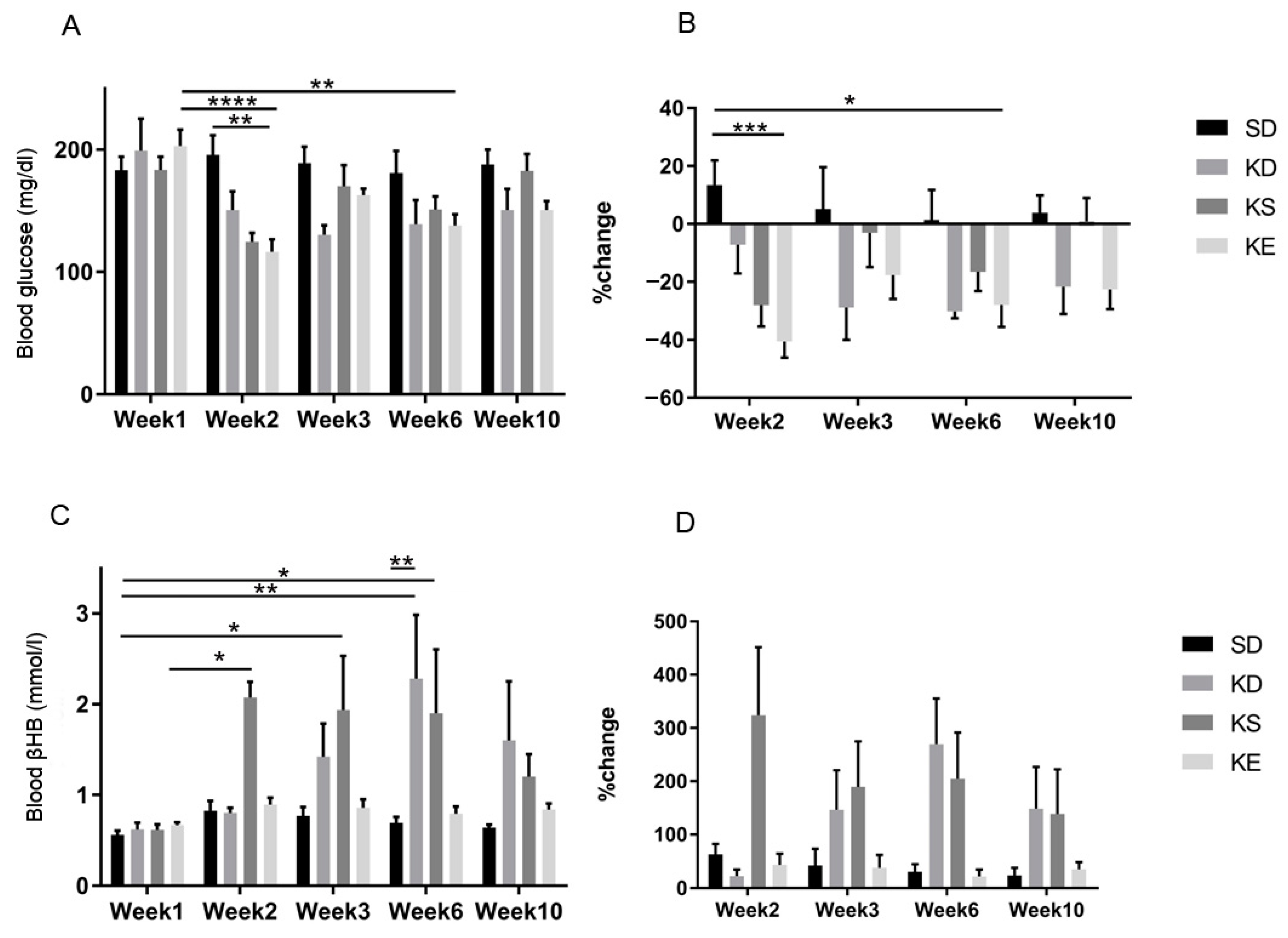
3.6. Chronic Effects of Ketone Supplements on Blood Glucose and R-βHB Levels in G1D Syndrome Mice
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| βHB | beta-hydroxybutyrate |
| GLUT1D | GLUT1 deficiency syndrome |
| KD | ketogenic diet |
| KE | 1, 3 butanediol-acetoacetate diester |
| KS | ketone salt |
| SD | standard diet |
| SPD rats | Sprague-Dawley rats |
| WAG/Rij | Wistar Albino Glaxo/Rijswijk rats |
References
- Hawkins, R.A.; Biebuyck, J.F. Ketone bodies are selectively used by individual brain regions. Science 1979, 205, 325–327. [Google Scholar] [CrossRef] [PubMed]
- Veech, R.L. The therapeutic implications of ketone bodies: The effects of ketone bodies in pathological conditions: Ketosis, ketogenic diet, redox states, insulin resistance, and mitochondrial metabolism. Prostaglandins Leukot. Essent. Fat. Acids 2004, 70, 309–319. [Google Scholar] [CrossRef] [PubMed]
- Yudkoff, M.; Daikhin, Y.; Melø, T.M.; Nissim, I.; Sonnewald, U.; Nissim, I. The Ketogenic Diet and Brain Metabolism of Amino Acids: Relationship to the Anticonvulsant Effect. Annu. Rev. Nutr. 2007, 27, 415–430. [Google Scholar] [CrossRef] [PubMed]
- Achanta, L.B.; Rae, C.D. β-Hydroxybutyrate in the Brain: One Molecule, Multiple Mechanisms. Neurochem. Res. 2017, 42, 35–49. [Google Scholar] [CrossRef]
- Halestrap, A.P.; Price, N.T. The proton-linked monocarboxylate transporter (MCT) family: Structure, function and regulation. Biochem. J. 1999, 343, 281–299. [Google Scholar] [CrossRef]
- Frazier, T.H.; DiBaise, J.K.; McClain, C.J. Gut microbiota, intestinal permeability, obesity-induced inflammation, and liver injury. JPEN J. Parenter. Enteral. Nutr. 2011, 35, 14S–20S. [Google Scholar] [CrossRef]
- Aeberli, I.; Gerber, P.A.; Hochuli, M.; Kohler, S.; Haile, S.R.; Gouni-Berthold, I.; Berthold, H.K.; Spinas, G.A.; Berneis, K. Low to moderate sugar-sweetened beverage consumption impairs glucose and lipid metabolism and promotes inflammation in healthy young men: A randomized controlled trial. Am. J. Clin. Nutr. 2011, 94, 479–485. [Google Scholar] [CrossRef]
- Jameel, F.; Phang, M.; Wood, L.G.; Garg, M.L. Acute effects of feeding fructose, glucose and sucrose on blood lipid levels and systemic inflammation. Lipids Health Dis. 2014, 13, 195. [Google Scholar] [CrossRef]
- Buyken, A.E.; Flood, V.; Empson, M.; Rochtchina, E.; Barclay, A.W.; Brand-Miller, J.; Mitchell, P. Carbohydrate nutrition and inflammatory disease mortality in older adults. Am. J. Clin. Nutr. 2010, 92, 634–643. [Google Scholar] [CrossRef]
- Kleen, J.K.; Holmes, G.L. Brain inflammation initiates seizures. Nat. Med. 2008, 14, 1309–1310. [Google Scholar] [CrossRef]
- Shimada, T.; Takemiya, T.; Sugiura, H.; Yamagata, K. Role of Inflammatory Mediators in the Pathogenesis of Epilepsy. Available online: https://www.hindawi.com/journals/mi/2014/901902/ (accessed on 27 August 2019).
- Stafstrom, C.E. Hyperglycemia Lowers Seizure Threshold. Epilepsy Curr. 2003, 3, 148–149. [Google Scholar] [CrossRef] [PubMed]
- Moien-Afshari, F.; Téllez-Zenteno, J.F. Occipital seizures induced by hyperglycemia: A case report and review of literature. Seizure 2009, 18, 382–385. [Google Scholar] [CrossRef] [PubMed]
- Sabitha, K.M.; Girija, A.S.; Vargese, K.S. Seizures in hyperglycemic patients. J. Assoc. Physicians India 2001, 49, 723–726. [Google Scholar]
- Chiang, J.L.; Kirkman, M.S.; Laffel, L.M.B.; Peters, A.L. Type 1 Diabetes Through the Life Span: A Position Statement of the American Diabetes Association. Diabetes Care 2014, 37, 2034–2054. [Google Scholar] [CrossRef] [PubMed]
- Forbes, J.M.; Cooper, M.E. Mechanisms of diabetic complications. Physiol. Rev. 2013, 93, 137–188. [Google Scholar] [CrossRef] [PubMed]
- Prato, S.D. Role of glucotoxicity and lipotoxicity in the pathophysiology of Type 2 diabetes mellitus and emerging treatment strategies. Diabet. Med. 2009, 26, 1185–1192. [Google Scholar] [CrossRef] [PubMed]
- Poitout, V.; Robertson, R.P. Glucolipotoxicity: Fuel Excess and β-Cell Dysfunction. Endocr. Rev. 2008, 29, 351–366. [Google Scholar] [CrossRef]
- Cernea, S.; Dobreanu, M. Diabetes and beta cell function: From mechanisms to evaluation and clinical implications. Biochem. Med. Biochem. Med. 2013, 23, 266–280. [Google Scholar] [CrossRef]
- Roseman, H.M. Progression from obesity to type 2 diabetes: Lipotoxicity, glucotoxicity, and implications for management. J. Manag. Care Pharm. 2005, 11, S3–S11. [Google Scholar]
- Johnson, E.L. Glycemic Variability in Type 2 Diabetes Mellitus. In Diabetes: An Old Disease, a New Insight; Ahmad, S.I., Ed.; Advances in Experimental Medicine and Biology; Springer: New York, NY, USA, 2013; pp. 139–154. ISBN 978-1-4614-5441-0. [Google Scholar]
- Yan, L.J. Pathogenesis of Chronic Hyperglycemia: From Reductive Stress to Oxidative Stress. Available online: https://www.hindawi.com/journals/jdr/2014/137919/ (accessed on 27 August 2019).
- Giacco, F.; Brownlee, M. Oxidative stress and diabetic complications. Circ. Res. 2010, 107, 1058–1070. [Google Scholar] [CrossRef]
- Bocci, V.; Zanardi, I.; Huijberts, M.S.P.; Travagli, V. An integrated medical treatment for type-2 diabetes. Diabetes Metab. Syndr. Clin. Res. Rev. 2014, 8, 57–61. [Google Scholar] [CrossRef] [PubMed]
- Ye, J. Mechanisms of insulin resistance in obesity. Front. Med. 2013, 7, 14–24. [Google Scholar] [CrossRef] [PubMed]
- Butkowski, E.G.; Jelinek, H.F. Hyperglycaemia, oxidative stress and inflammatory markers. Redox Rep. 2017, 22, 257–264. [Google Scholar] [CrossRef] [PubMed]
- Capes, S.E.; Hunt, D.; Malmberg, K.; Gerstein, H.C. Stress hyperglycaemia and increased risk of death after myocardial infarction in patients with and without diabetes: A systematic overview. Lancet 2000, 355, 773–778. [Google Scholar] [CrossRef]
- Shimoyama, T.; Kimura, K.; Uemura, J.; Saji, N.; Shibazaki, K. Elevated glucose level adversely affects infarct volume growth and neurological deterioration in non-diabetic stroke patients, but not diabetic stroke patients. Eur. J. Neurol. 2014, 21, 402–410. [Google Scholar] [CrossRef] [PubMed]
- Richards, J.E.; Kauffmann, R.M.; Obremskey, W.T.; May, A.K. Stress-Induced Hyperglycemia as a Risk Factor for Surgical-Site Infection in Non-diabetic Orthopaedic Trauma Patients Admitted to the Intensive Care Unit. J. Orthop. Trauma 2013, 27, 16–21. [Google Scholar] [CrossRef] [PubMed]
- Salim, A.; Hadjizacharia, P.; Dubose, J.; Brown, C.; Inaba, K.; Chan, L.S.; Margulies, D. Persistent Hyperglycemia in Severe Traumatic Brain Injury: An Independent Predictor of Outcome. Am. Surg. 2009, 75, 25–29. [Google Scholar]
- Mitchell, T.H.; Abraham, G.; Schiffrin, A.; Leiter, L.A.; Marliss, E.B. Hyperglycemia After Intense Exercise in IDDM Subjects During Continuous Subcutaneous Insulin Infusion. Diabetes Care 1988, 11, 311–317. [Google Scholar] [CrossRef]
- LaMonte, M.J.; Blair, S.N.; Church, T.S. Physical activity and diabetes prevention. J. Appl. Physiol. 2005, 99, 1205–1213. [Google Scholar] [CrossRef]
- Hirsch, I.B.; Marker, J.C.; Smith, L.J.; Spina, R.J.; Parvin, C.A.; Holloszy, J.O.; Cryer, P.E. Insulin and glucagon in prevention of hypoglycemia during exercise in humans. Am. J. Physiol. Endocrinol. Metab. 1991, 260, E695–E704. [Google Scholar] [CrossRef]
- Wasserman, D.H.; Spalding, J.A.; Lacy, D.B.; Colburn, C.A.; Goldstein, R.E.; Cherrington, A.D. Glucagon is a primary controller of hepatic glycogenolysis and gluconeogenesis during muscular work. Am. J. Physiol. Endocrinol. Metab. 1989, 257, E108–E117. [Google Scholar] [CrossRef] [PubMed]
- Marliss, E.B.; Vranic, M. Intense exercise has unique effects on both insulin release and its roles in glucoregulation: Implications for diabetes. Diabetes 2002, 51, S271–S283. [Google Scholar] [CrossRef] [PubMed]
- Marliss, E.B.; Simantirakis, E.; Miles, P.D.; Hunt, R.; Gougeon, R.; Purdon, C.; Halter, J.B.; Vranic, M. Glucose turnover and its regulation during intense exercise and recovery in normal male subjects. Clin. Investig. Med. 1992, 15, 406–419. [Google Scholar]
- Marliss, E.B.; Simantirakis, E.; Miles, P.D.; Purdon, C.; Gougeon, R.; Field, C.J.; Halter, J.B.; Vranic, M. Glucoregulatory and hormonal responses to repeated bouts of intense exercise in normal male subjects. J. Appl. Physiol. 1991, 71, 924–933. [Google Scholar] [CrossRef] [PubMed]
- Sigal, R.J.; Purdon, C.; Fisher, S.J.; Halter, J.B.; Vranic, M.; Marliss, E.B. Hyperinsulinemia prevents prolonged hyperglycemia after intense exercise in insulin-dependent diabetic subjects. J. Clin. Endocrinol. Metab. 1994, 79, 1049–1057. [Google Scholar] [PubMed]
- Bracken, R.M.; Edavalath, M.; Morton, R.; West, D.; Fielding, A.; Luzio, S.; Underwood, P.; Stephens, J.W. Exercise-induced hyperglycaemia in the absence of diabetes. Diabet. Med. 2010, 27, 723–724. [Google Scholar] [CrossRef]
- Krinsley, J.S.; Preiser, J.C. Time in blood glucose range 70 to 140 mg/dl > 80% is strongly associated with increased survival in non-diabetic critically ill adults. Crit. Care 2015, 19, 179. [Google Scholar] [CrossRef]
- Singh, M.; Kumar, A. Risks Associated with SGLT2 Inhibitors: An Overview. Curr. Drug Saf. 2018, 13, 84–91. [Google Scholar] [CrossRef]
- Danne, T.; Garg, S.; Peters, A.L.; Buse, J.B.; Mathieu, C.; Pettus, J.H.; Alexander, C.M.; Battelino, T.; Ampudia-Blasco, F.J.; Bode, B.W.; et al. International Consensus on Risk Management of Diabetic Ketoacidosis in Patients with Type 1 Diabetes Treated with Sodium–Glucose Cotransporter (SGLT) Inhibitors. Diabetes Care 2019, 42, 1147–1154. [Google Scholar] [CrossRef]
- Paoli, A.; Rubini, A.; Volek, J.S.; Grimaldi, K.A. Beyond weight loss: A review of the therapeutic uses of very-low-carbohydrate (ketogenic) diets. Eur. J. Clin. Nutr. 2013, 67, 789–796. [Google Scholar] [CrossRef]
- Allen, F.M.; Stillman, E.; Fitz, R. Total Dietary Regulation in the Treatment of Diabetes; Rockefeller Institute for Medical Research: New York, NY, USA, 1919. [Google Scholar]
- Wilder, R.M.; Winter, M.D. The Threshold of Ketogenesis; The Mayo Clinic: Rotchester, NY, USA, 1922. [Google Scholar]
- Wheless, J.W. History of the ketogenic diet. Epilepsia 2008, 49, 3–5. [Google Scholar] [CrossRef] [PubMed]
- Neal, E.G.; Chaffe, H.; Schwartz, R.H.; Lawson, M.S.; Edwards, N.; Fitzsimmons, G.; Whitney, A.; Cross, J.H. The ketogenic diet for the treatment of childhood epilepsy: A randomised controlled trial. Lancet Neurol. 2008, 7, 500–506. [Google Scholar] [CrossRef]
- Freeman, J.M.; Kossoff, E.H. Ketosis and the Ketogenic Diet, 2010: Advances in Treating Epilepsy and Other Disorders. Adv. Pediatr. 2010, 57, 315–329. [Google Scholar] [CrossRef] [PubMed]
- Westman, E.C.; Yancy, W.S.; Mavropoulos, J.C.; Marquart, M.; McDuffie, J.R. The effect of a low-carbohydrate, ketogenic diet versus a low-glycemic index diet on glycemic control in type 2 diabetes mellitus. Nutr. Metab. 2008, 5, 36. [Google Scholar] [CrossRef]
- Seyfried, T.N.; Mukherjee, P. Targeting energy metabolism in brain cancer: Review and hypothesis. Nutr. Metab. 2005, 2, 30. [Google Scholar] [CrossRef]
- Poff, A.; Ari, C.; Arnold, P.; Seyfried, T.; D’Agostino, D. Ketone supplementation decreases tumor cell viability and prolongs survival of mice with metastatic cancer. Int. J. Cancer 2014, 135, 1711–1720. [Google Scholar] [CrossRef]
- Zhao, Z.; Lange, D.J.; Voustianiouk, A.; MacGrogan, D.; Ho, L.; Suh, J.; Humala, N.; Thiyagarajan, M.; Wang, J.; Pasinetti, G.M. A ketogenic diet as a potential novel therapeutic intervention in amyotrophic lateral sclerosis. BMC Neurosci. 2006, 7, 29. [Google Scholar]
- Ari, C.; Poff, A.M.; Held, H.E.; Landon, C.S.; Goldhagen, C.R.; Mavromates, N.; D’Agostino, D.P. Metabolic Therapy with Deanna Protocol Supplementation Delays Disease Progression and Extends Survival in Amyotrophic Lateral Sclerosis (ALS) Mouse Model. PLoS ONE 2014, 9, e103526. [Google Scholar] [CrossRef]
- Prins, M.L. Cerebral Metabolic Adaptation and Ketone Metabolism after Brain Injury. J. Cereb. Blood Flow Metab. 2008, 28, 1–16. [Google Scholar] [CrossRef]
- Henderson, S.T.; Vogel, J.L.; Barr, L.J.; Garvin, F.; Jones, J.J.; Costantini, L.C. Study of the ketogenic agent AC-1202 in mild to moderate Alzheimer’s disease: A randomized, double-blind, placebo-controlled, multicenter trial. Nutr. Metab. 2009, 6, 31. [Google Scholar] [CrossRef]
- Lennerz, B.S.; Barton, A.; Bernstein, R.K.; Dikeman, R.D.; Diulus, C.; Hallberg, S.; Rhodes, E.T.; Ebbeling, C.B.; Westman, E.C.; Yancy, W.S.; et al. Management of Type 1 Diabetes with a Very Low-Carbohydrate Diet. Pediatrics 2018, 141, e20173349. [Google Scholar] [CrossRef] [PubMed]
- Hemingway, C.; Freeman, J.M.; Pillas, D.J.; Pyzik, P.L. The Ketogenic Diet: A 3- to 6-Year Follow-Up of 150 Children Enrolled Prospectively. Pediatrics 2001, 108, 898–905. [Google Scholar] [CrossRef] [PubMed]
- Coppola, G.; Veggiotti, P.; Cusmai, R.; Bertoli, S.; Cardinali, S.; Dionisi-Vici, C.; Elia, M.; Lispi, M.L.; Sarnelli, C.; Tagliabue, A.; et al. The ketogenic diet in children, adolescents and young adults with refractory epilepsy: An Italian multicentric experience. Epilepsy Res. 2002, 48, 221–227. [Google Scholar] [CrossRef]
- Dansinger, M.L.; Gleason, J.A.; Griffith, J.L.; Selker, H.P.; Schaefer, E.J. Comparison of the Atkins, Ornish, Weight Watchers, and Zone diets for weight loss and heart disease risk reduction: A randomized trial. JAMA 2005, 293, 43–53. [Google Scholar] [CrossRef]
- Gardner, C.D.; Trepanowski, J.F.; Del Gobbo, L.C.; Hauser, M.E.; Rigdon, J.; Ioannidis, J.P.A.; Desai, M.; King, A.C. Effect of Low-Fat vs Low-Carbohydrate Diet on 12-Month Weight Loss in Overweight Adults and the Association with Genotype Pattern or Insulin Secretion: The DIETFITS Randomized Clinical Trial. JAMA 2018, 319, 667–679. [Google Scholar] [CrossRef]
- Pfeifer, H.H.; Thiele, E.A. Low-glycemic-index treatment: A liberalized ketogenic diet for treatment of intractable epilepsy. Neurology 2005, 65, 1810–1812. [Google Scholar] [CrossRef]
- Yancy, W.S.; Olsen, M.K.; Guyton, J.R.; Bakst, R.P.; Westman, E.C. A Low-Carbohydrate, Ketogenic Diet versus a Low-Fat Diet to Treat Obesity and Hyperlipidemia: A Randomized, Controlled Trial. Ann. Intern. Med. 2004, 140, 769. [Google Scholar] [CrossRef]
- Kesl, S.L.; Poff, A.M.; Ward, N.P.; Fiorelli, T.N.; Ari, C.; Van Putten, A.J.; Sherwood, J.W.; Arnold, P.; D’Agostino, D.P. Effects of exogenous ketone supplementation on blood ketone, glucose, triglyceride, and lipoprotein levels in Sprague–Dawley rats. Nutr. Metab. 2016, 13, 9. [Google Scholar] [CrossRef]
- Ari, C.; Kovács, Z.; Murdun, C.; Koutnik, A.P.; Goldhagen, C.R.; Rogers, C.; Diamond, D.; D’Agostino, D.P. Nutritional ketosis delays the onset of isoflurane induced anesthesia. BMC Anesthesiol. 2018, 18, 85. [Google Scholar] [CrossRef]
- Ari, C.; Koutnik, A.P.; DeBlasi, J.; Landon, C.; Rogers, C.Q.; Vallas, J.; Bharwani, S.; Puchowicz, M.; Bederman, I.; Diamond, D.M.; et al. Delaying latency to hyperbaric oxygen-induced CNS oxygen toxicity seizures by combinations of exogenous ketone supplements. Physiol. Rep. 2019, 7, e13961. [Google Scholar] [CrossRef]
- Cox, P.J.; Kirk, T.; Ashmore, T.; Willerton, K.; Evans, R.; Smith, A.; Murray, A.J.; Stubbs, B.; West, J.; McLure, S.W.; et al. Nutritional Ketosis Alters Fuel Preference and Thereby Endurance Performance in Athletes. Cell Metab. 2016, 24, 256–268. [Google Scholar] [CrossRef] [PubMed]
- O’Malley, T.; Myette-Cote, E.; Durrer, C.; Little, J.P. Nutritional ketone salts increase fat oxidation but impair high-intensity exercise performance in healthy adult males. Appl. Physiol. Nutr. Metab. 2017, 42, 1031–1035. [Google Scholar] [CrossRef] [PubMed]
- Rodger, S.; Plews, D.; Laursen, P.; Driller, M.W. Oral β-hydroxybutyrate salt fails to improve 4-min cycling performance following submaximal exercise. Res. Commons. 2017, 6, 26–31. [Google Scholar]
- Evans, M.; Patchett, E.; Nally, R.; Kearns, R.; Larney, M.; Egan, B. Effect of acute ingestion of β-hydroxybutyrate salts on the response to graded exercise in trained cyclists. Eur. J. Sport Sci. 2018, 18, 376–386. [Google Scholar] [CrossRef]
- Waldman, H.S.; Basham, S.A.; Price, F.G.; Smith, J.W.; Chander, H.; Knight, A.C.; Krings, B.M.; McAllister, M.J. Exogenous ketone salts do not improve cognitive responses after a high-intensity exercise protocol in healthy college-aged males. Appl. Physiol. Nutr. Metab. 2018, 43, 711–717. [Google Scholar] [CrossRef]
- Evans, M.; Egan, B. Intermittent Running and Cognitive Performance after Ketone Ester Ingestion. Med. Sci. Sports Exerc. 2018, 50, 2330–2338. [Google Scholar] [CrossRef]
- Leckey, J.J.; Ross, M.L.; Quod, M.; Hawley, J.A.; Burke, L.M. Ketone Diester Ingestion Impairs Time-Trial Performance in Professional Cyclists. Front. Physiol. 2017, 8, 806. [Google Scholar] [CrossRef]
- Evans, M.; McSwiney, F.; Brady, A.; Egan, B. No Benefit of Ingestion of a Ketone Monoester Supplement on 10-km Running Performance. Med. Sci. Sports Exerc. 2019, 1. [Google Scholar] [CrossRef]
- Thomas, D.; Burke, L.; Erdman, K. Nutrition and Athletic Performance. Med. Sci. 2016, 48, 543–568. [Google Scholar]
- Shaw, D.M.; Merien, F.; Braakhuis, A.; Plews, D.; Laursen, P.; Dulson, D.K. The Effect of 1,3-Butanediol on Cycling Time-Trial Performance. Int. J. Sport Nutr. Exerc. Metab. 2019, 29, 466–473. [Google Scholar] [CrossRef]
- Poffé, C.; Ramaekers, M.; Thienen, R.V.; Hespel, P. Ketone ester supplementation blunts overreaching symptoms during endurance training overload. J. Physiol. 2019, 597, 3009–3027. [Google Scholar] [CrossRef] [PubMed]
- Breckenridge, J.; Seibert, J.; Jackson, D.; Prins, P.; Buxton, J.; Ault, D. Effects of an Exogenous Ketone Supplement on Five-Kilometer Running Performance. Int. J. Exerc. Sci. Conf. Proc. 2019, 9, 11. [Google Scholar]
- Kovács, Z.; Kékesi, K.A.; Szilágyi, N.; Abrahám, I.; Székács, D.; Király, N.; Papp, E.; Császár, I.; Szego, E.; Barabás, K.; et al. Facilitation of spike-wave discharge activity by lipopolysaccharides in Wistar Albino Glaxo/Rijswijk rats. Neuroscience 2006, 140, 731–742. [Google Scholar] [CrossRef] [PubMed]
- Kovács, Z.; Czurkó, A.; Kékesi, K.A.; Juhász, G. Intracerebroventricularly administered lipopolysaccharide enhances spike-wave discharges in freely moving WAG/Rij rats. Brain Res. Bull. 2011, 85, 410–416. [Google Scholar] [CrossRef]
- Kovács, Z.; D’Agostino, D.P.; Dobolyi, A.; Ari, C. Adenosine A1 Receptor Antagonism Abolished the Anti-seizure Effects of Exogenous Ketone Supplementation in Wistar Albino Glaxo Rijswijk Rats. Front. Mol. Neurosci. 2017, 10, 235. [Google Scholar] [CrossRef]
- Kovács, Z.; Dobolyi, A.; Juhász, G.; Kékesi, K.A. Lipopolysaccharide induced increase in seizure activity in two animal models of absence epilepsy WAG/Rij and GAERS rats and Long Evans rats. Brain Res. Bull. 2014, 104, 7–18. [Google Scholar] [CrossRef][Green Version]
- Russo, E.; Andreozzi, F.; Iuliano, R.; Dattilo, V.; Procopio, T.; Fiume, G.; Mimmi, S.; Perrotti, N.; Citraro, R.; Sesti, G.; et al. Early molecular and behavioral response to lipopolysaccharide in the WAG/Rij rat model of absence epilepsy and depressive-like behavior, involves interplay between AMPK, AKT/mTOR pathways and neuroinflammatory cytokine release. Brain Behav. Immun. 2014, 42, 157–168. [Google Scholar] [CrossRef]
- Tang, M.; Gao, G.; Rueda, C.B.; Yu, H.; Thibodeaux, D.N.; Awano, T.; Engelstad, K.M.; Sanchez-Quintero, M.J.; Yang, H.; Li, F.; et al. Brain microvasculature defects and Glut1 deficiency syndrome averted by early repletion of the glucose transporter-1 protein. Nat. Commun. 2017, 8, 14152. [Google Scholar] [CrossRef]
- Veggiotti, P.; De Giorgis, V. Dietary Treatments and New Therapeutic Perspective in GLUT1 Deficiency Syndrome. Curr. Treat. Opt. Neurol. 2014, 16, 291. [Google Scholar] [CrossRef]
- Wang, D.; Pascual, J.M.; Yang, H.; Engelstad, K.; Jhung, S.; Sun, R.P.; De Vivo, D.C. Glut-1 deficiency syndrome: Clinical, genetic, and therapeutic aspects. Ann. Neurol. 2005, 57, 111–118. [Google Scholar] [CrossRef]
- Ari, C.; Kovács, Z.; Juhasz, G.; Murdun, C.; Goldhagen, C.R.; Koutnik, A.P.; Poff, A.M.; Kesl, S.L.; D’Agostino, D.P. Exogenous Ketone Supplements Reduce Anxiety-Related Behavior in Sprague-Dawley and Wistar Albino Glaxo/Rijswijk Rats. Front. Mol. Neurosci. 2017, 9, 137. [Google Scholar] [CrossRef] [PubMed]
- Coulston, A.M.; Liu, G.C.; Reaven, G.M. Plasma glucose, insulin and lipid responses to high-carbohydrate low-fat diets in normal humans. Metab. Clin. Exp. 1983, 32, 52–56. [Google Scholar] [CrossRef]
- Gougeon, R.; Carrington, M.; Field, C.J. The Impact of Low-carbohydrate Diets on Glycemic Control and Weight Management in Patients with Type 2 Diabetes. Can. J. Diabetes 2006, 30, 269–277. [Google Scholar] [CrossRef]
- Laaksonen, D.E.; Toppinen, L.K.; Juntunen, K.S.; Autio, K.; Liukkonen, K.H.; Poutanen, K.S.; Niskanen, L.; Mykkänen, H.M. Dietary carbohydrate modification enhances insulin secretion in persons with the metabolic syndrome. Am. J. Clin. Nutr. 2005, 82, 1218–1227. [Google Scholar] [CrossRef] [PubMed]
- Shin, Y.; Park, S.; Choue, R. Comparison of time course changes in blood glucose, insulin and lipids between high carbohydrate and high fat meals in healthy young women. Nutr. Res. Pract. 2009, 3, 128–133. [Google Scholar] [CrossRef] [PubMed]
- Kalant, N.; Leibovici, D.; Leibovici, T.; Fukushima, N. Effect of Age on Glucose Utilization and Responsiveness to Insulin in Forearm Muscle. J. Am. Geriatr. Soc. 1980, 28, 304–307. [Google Scholar] [CrossRef] [PubMed]
- Elahi, D.; Muller, D.C.; McAloon-Dyke, M.; Tobin, J.D.; Andres, R. The effect of age on insulin response and glucose utilization during four hyperglycemic plateaus. Exp. Gerontol. 1993, 28, 393–409. [Google Scholar] [CrossRef]
- Brewer, R.A.; Gibbs, V.K.; Smith, D.L. Targeting glucose metabolism for healthy aging. Nutr. Healthy Aging 2016, 4, 31–46. [Google Scholar] [CrossRef]
- D’Agostino, D.P.; Pilla, R.; Held, H.E.; Landon, C.S.; Puchowicz, M.; Brunengraber, H.; Ari, C.; Arnold, P.; Dean, J.B. Therapeutic ketosis with ketone ester delays central nervous system oxygen toxicity seizures in rats. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2013, 304, R829–R836. [Google Scholar] [CrossRef]
- Wang, D.; Pascual, J.M.; Yang, H.; Engelstad, K.; Mao, X.; Cheng, J.; Yoo, J.; Noebels, J.L.; De Vivo, D.C. A mouse model for Glut-1 haploinsufficiency. Hum. Mol. Genet. 2006, 15, 1169–1179. [Google Scholar] [CrossRef]
- Gilbert, D.L.; Pyzik, P.L.; Freeman, J.M. The Ketogenic Diet: Seizure Control Correlates Better With Serum β-Hydroxybutyrate Than With Urine Ketones. J. Child Neurol. 2000, 15, 787–790. [Google Scholar] [CrossRef] [PubMed]
- Clarke, K.; Tchabanenko, K.; Pawlosky, R.; Carter, E.; Todd King, M.; Musa-Veloso, K.; Ho, M.; Roberts, A.; Robertson, J.; VanItallie, T.B.; et al. Kinetics, safety and tolerability of (R)-3-hydroxybutyl (R)-3-hydroxybutyrate in healthy adult subjects. Regul. Toxicol. Pharmacol. 2012, 63, 401–408. [Google Scholar] [CrossRef] [PubMed]
- Clarke, K.; Tchabanenko, K.; Pawlosky, R.; Carter, E.; Knight, N.S.; Murray, A.J.; Cochlin, L.E.; King, M.T.; Wong, A.W.; Roberts, A.; et al. Oral 28-day and developmental toxicity studies of (R)-3-hydroxybutyl (R)-3-hydroxybutyrate. Regul. Toxicol. Pharmacol. 2012, 63, 196–208. [Google Scholar] [CrossRef] [PubMed]
- Moss, S.E.; Klein, R.; Klein, B.E.K.; Meuer, S.M. The Association of Glycemia and Cause-Specific Mortality in a Diabetic Population. Arch. Intern. Med. 1994, 154, 2473–2479. [Google Scholar] [CrossRef]
- Hallberg, S.J.; McKenzie, A.L.; Williams, P.T.; Bhanpuri, N.H.; Peters, A.L.; Campbell, W.W.; Hazbun, T.L.; Volk, B.M.; McCarter, J.P.; Phinney, S.D.; et al. Effectiveness and Safety of a Novel Care Model for the Management of Type 2 Diabetes at 1 Year: An Open-Label, Non-Randomized, Controlled Study. Diabetes Ther. 2018, 9, 583–612. [Google Scholar] [CrossRef]
- McKenzie, A.L.; Hallberg, S.J.; Creighton, B.C.; Volk, B.M.; Link, T.M.; Abner, M.K.; Glon, R.M.; McCarter, J.P.; Volek, J.S.; Phinney, S.D. A Novel Intervention Including Individualized Nutritional Recommendations Reduces Hemoglobin A1c Level, Medication Use, and Weight in Type 2 Diabetes. JMIR Diabetes 2017, 2, e5. [Google Scholar] [CrossRef]
- Kawahito, S.; Kitahata, H.; Oshita, S. Problems associated with glucose toxicity: Role of hyperglycemia-induced oxidative stress. World J. Gastroenterol. 2009, 15, 4137–4142. [Google Scholar] [CrossRef]
- Stubbs, B.J.; Cox, P.J.; Evans, R.D.; Santer, P.; Miller, J.J.; Faull, O.K.; Magor-Elliott, S.; Hiyama, S.; Stirling, M.; Clarke, K. On the Metabolism of Exogenous Ketones in Humans. Front. Physiol. 2017, 8, 848. [Google Scholar] [CrossRef]
- Srivastava, S.; Kashiwaya, Y.; King, M.T.; Baxa, U.; Tam, J.; Niu, G.; Chen, X.; Clarke, K.; Veech, R.L. Mitochondrial biogenesis and increased uncoupling protein 1 in brown adipose tissue of mice fed a ketone ester diet. FASEB J. 2012, 26, 2351–2362. [Google Scholar] [CrossRef]
- Binkiewicz, A.; Sadeghi-Najad, A.; Hochman, H.; Loridan, L.; Senior, B. An effect of ketones on the concentrations of glucose and of free fatty acids in man independent of the release of insulin. J. Pediatr. 1974, 84, 226–231. [Google Scholar] [CrossRef]
- Klepper, J. Glucose transporter deficiency syndrome (GLUT1DS) and the ketogenic diet. Epilepsia 2008, 49, 46–49. [Google Scholar] [CrossRef] [PubMed]
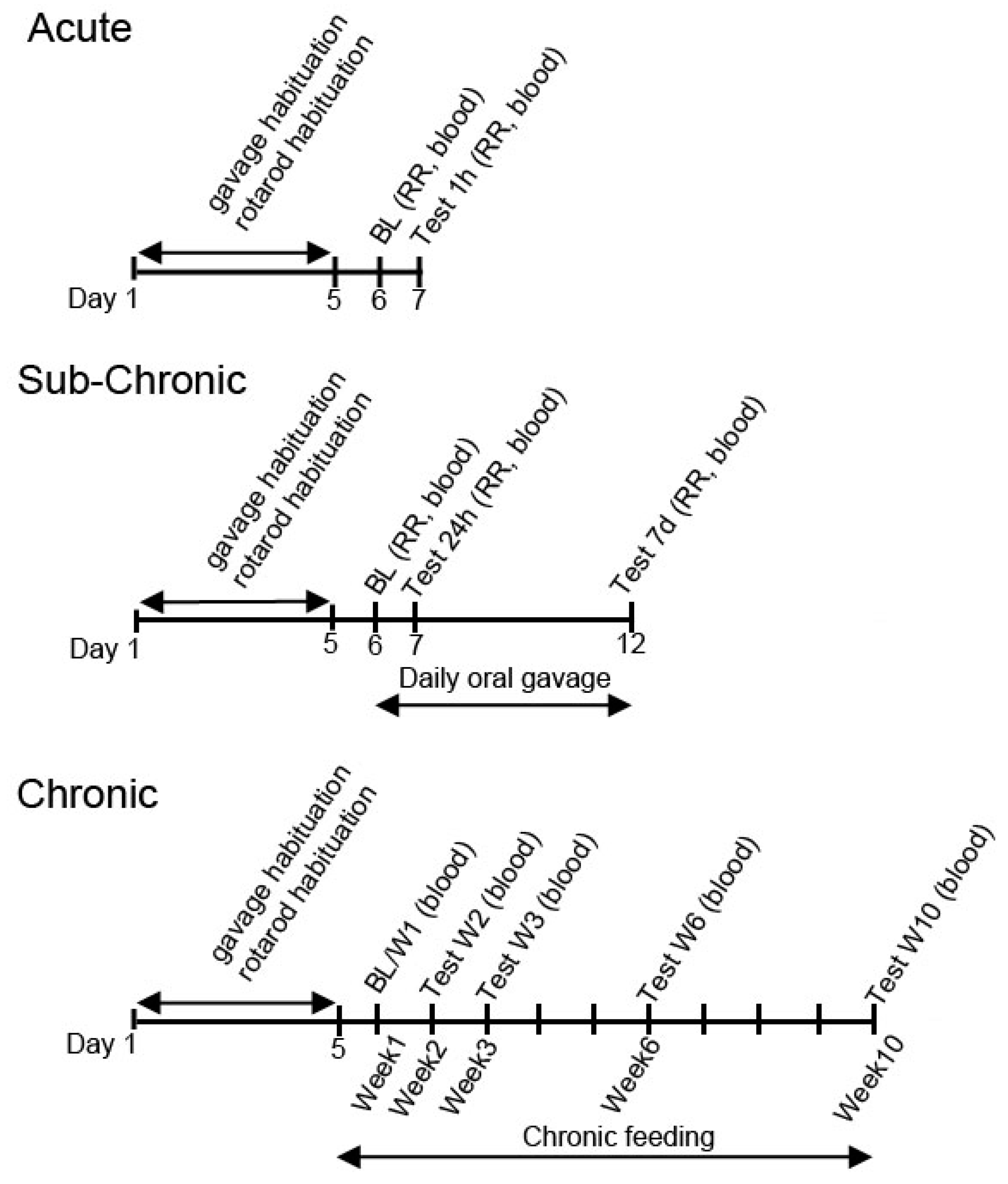

| Acute | Sub-Chronic | Chronic | |
|---|---|---|---|
| SPD | (R)Ex | (R)Ex | (R) |
| WR | R, Ex | R, Ex | |
| GLUT1D | R |
| Macronutrient Information | Standard Diet (SD) | Ketogenic Diet (KD) |
|---|---|---|
| % Cal from Fat | 18.0 | 77.1 |
| % Cal from Protein | 24.0 | 22.4 |
| % Cal from Carbohydrate | 58.0 | 0.5 |
| Caloric Density (Kcal/g) | 3.1 | 4.7 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ari, C.; Murdun, C.; Koutnik, A.P.; Goldhagen, C.R.; Rogers, C.; Park, C.; Bharwani, S.; Diamond, D.M.; Kindy, M.S.; D’Agostino, D.P.; et al. Exogenous Ketones Lower Blood Glucose Level in Rested and Exercised Rodent Models. Nutrients 2019, 11, 2330. https://doi.org/10.3390/nu11102330
Ari C, Murdun C, Koutnik AP, Goldhagen CR, Rogers C, Park C, Bharwani S, Diamond DM, Kindy MS, D’Agostino DP, et al. Exogenous Ketones Lower Blood Glucose Level in Rested and Exercised Rodent Models. Nutrients. 2019; 11(10):2330. https://doi.org/10.3390/nu11102330
Chicago/Turabian StyleAri, Csilla, Cem Murdun, Andrew P. Koutnik, Craig R. Goldhagen, Christopher Rogers, Collin Park, Sahil Bharwani, David M. Diamond, Mark S. Kindy, Dominic P. D’Agostino, and et al. 2019. "Exogenous Ketones Lower Blood Glucose Level in Rested and Exercised Rodent Models" Nutrients 11, no. 10: 2330. https://doi.org/10.3390/nu11102330
APA StyleAri, C., Murdun, C., Koutnik, A. P., Goldhagen, C. R., Rogers, C., Park, C., Bharwani, S., Diamond, D. M., Kindy, M. S., D’Agostino, D. P., & Kovács, Z. (2019). Exogenous Ketones Lower Blood Glucose Level in Rested and Exercised Rodent Models. Nutrients, 11(10), 2330. https://doi.org/10.3390/nu11102330