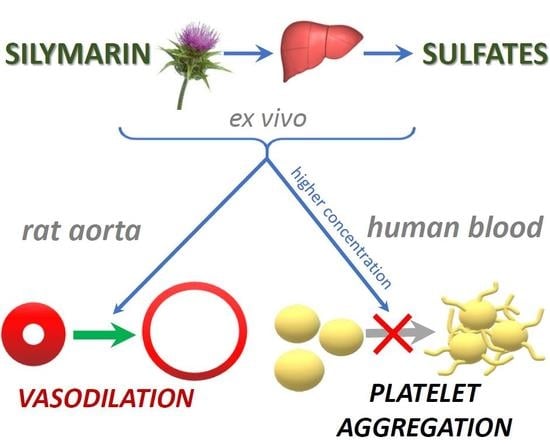
The Effect of Silymarin Flavonolignans and Their Sulfated Conjugates on Platelet Aggregation and Blood Vessels Ex Vivo
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
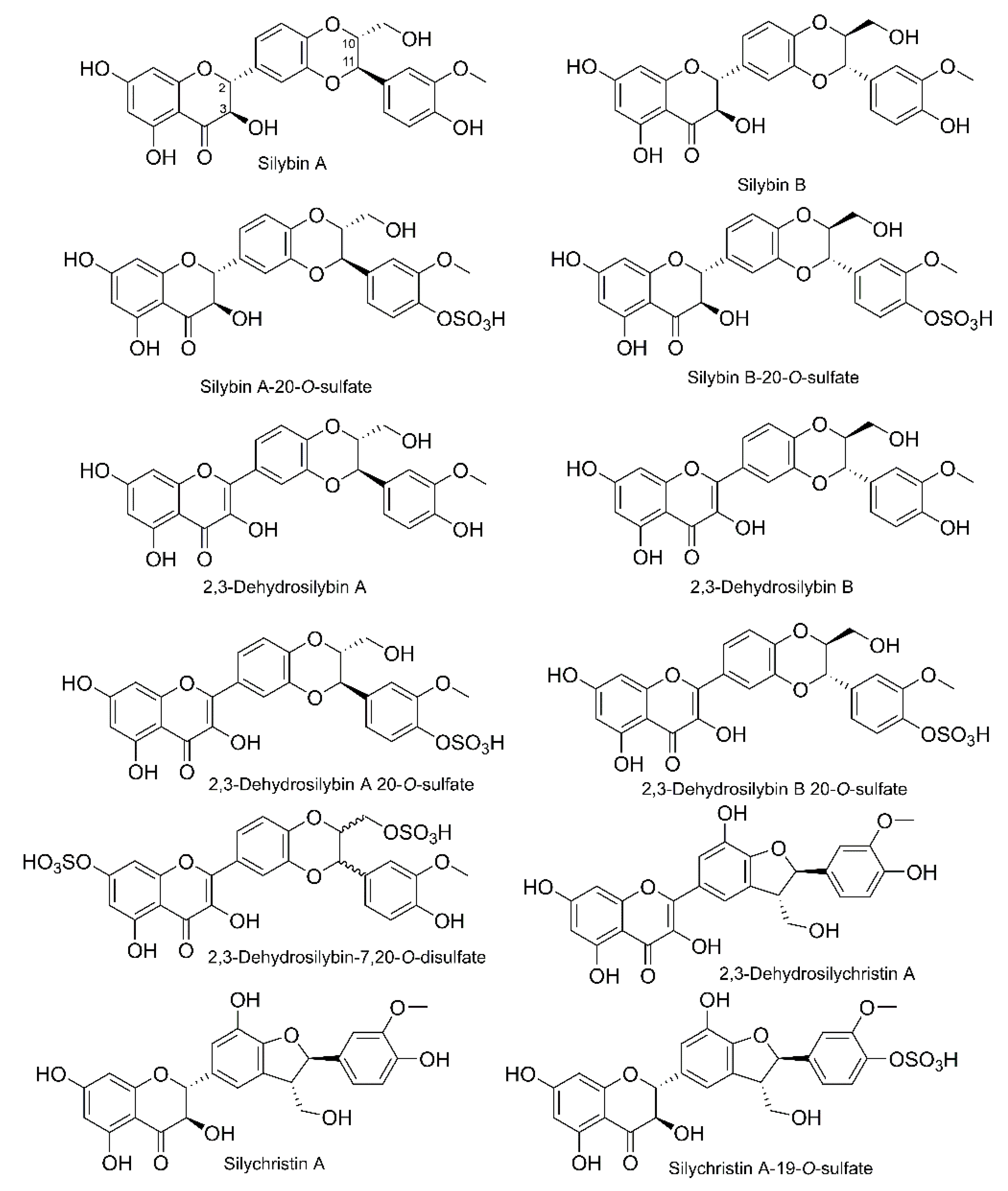
2.2. Silymarin Flavonolignans
2.3. Reagents
2.4. Ex Vivo Experiments on Isolated Rat Aortas
2.5. Platelet Aggregation Experiments
2.5.1. Blood Volunteers
2.5.2. Platelet Aggregation Induced by Collagen and Arachidonic Acid
2.5.3. Cyclooxygenase-1 (COX-1) Inhibition
2.5.4. Thromboxane A2 Synthase Inhibition
2.5.5. Antagonism at Thromboxane A2 Receptors
2.6. Statistical Analysis
3. Results
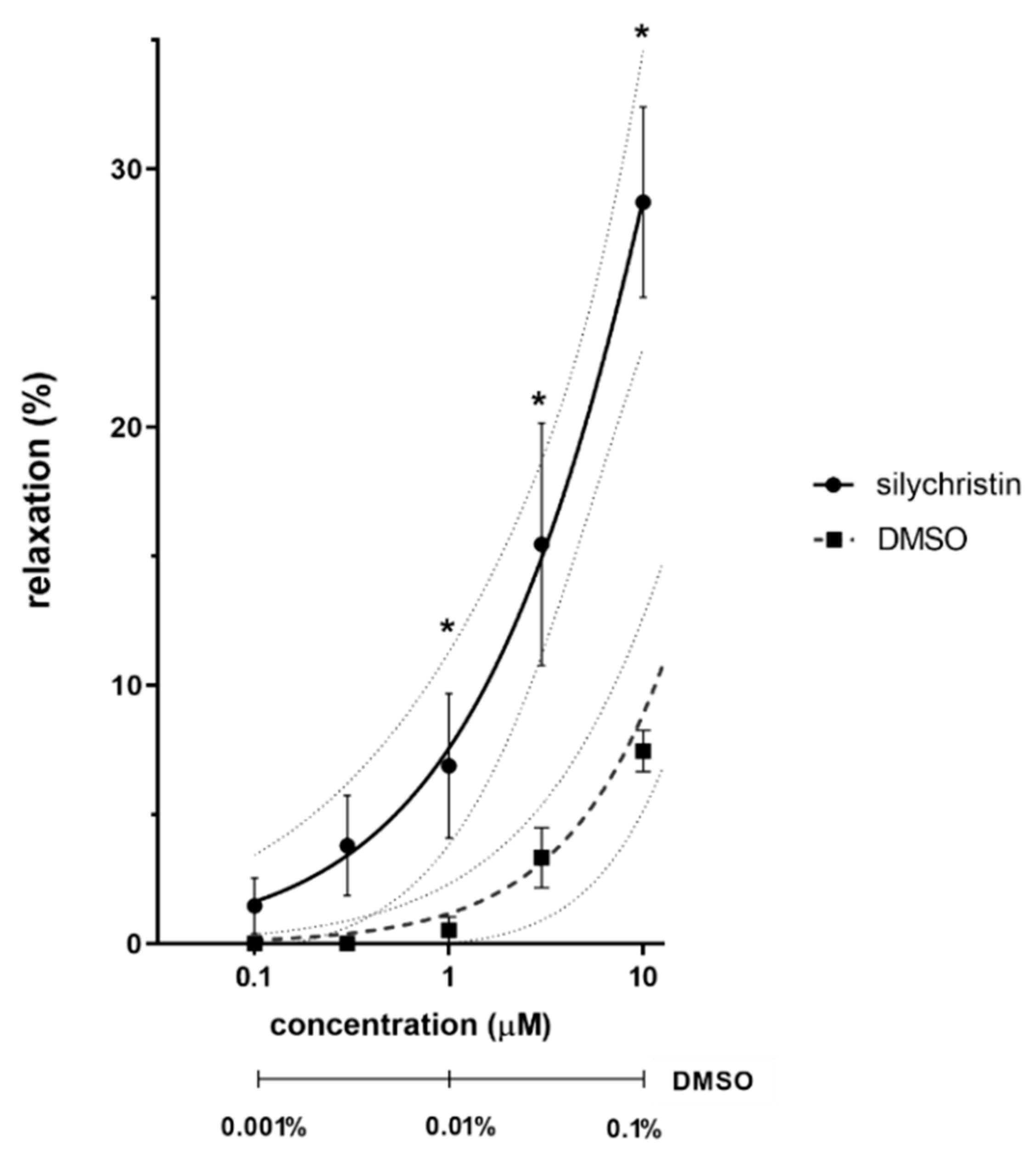
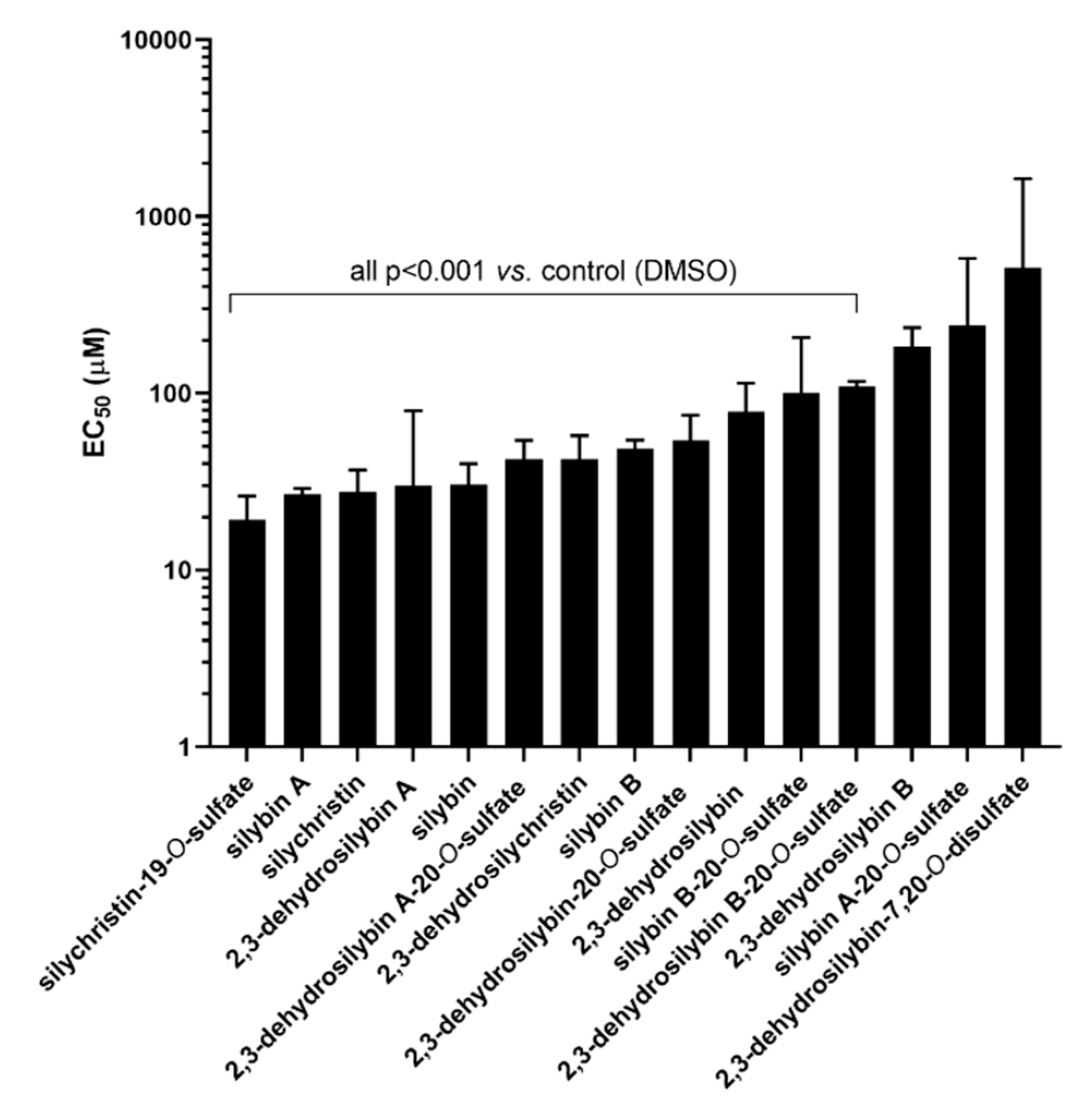
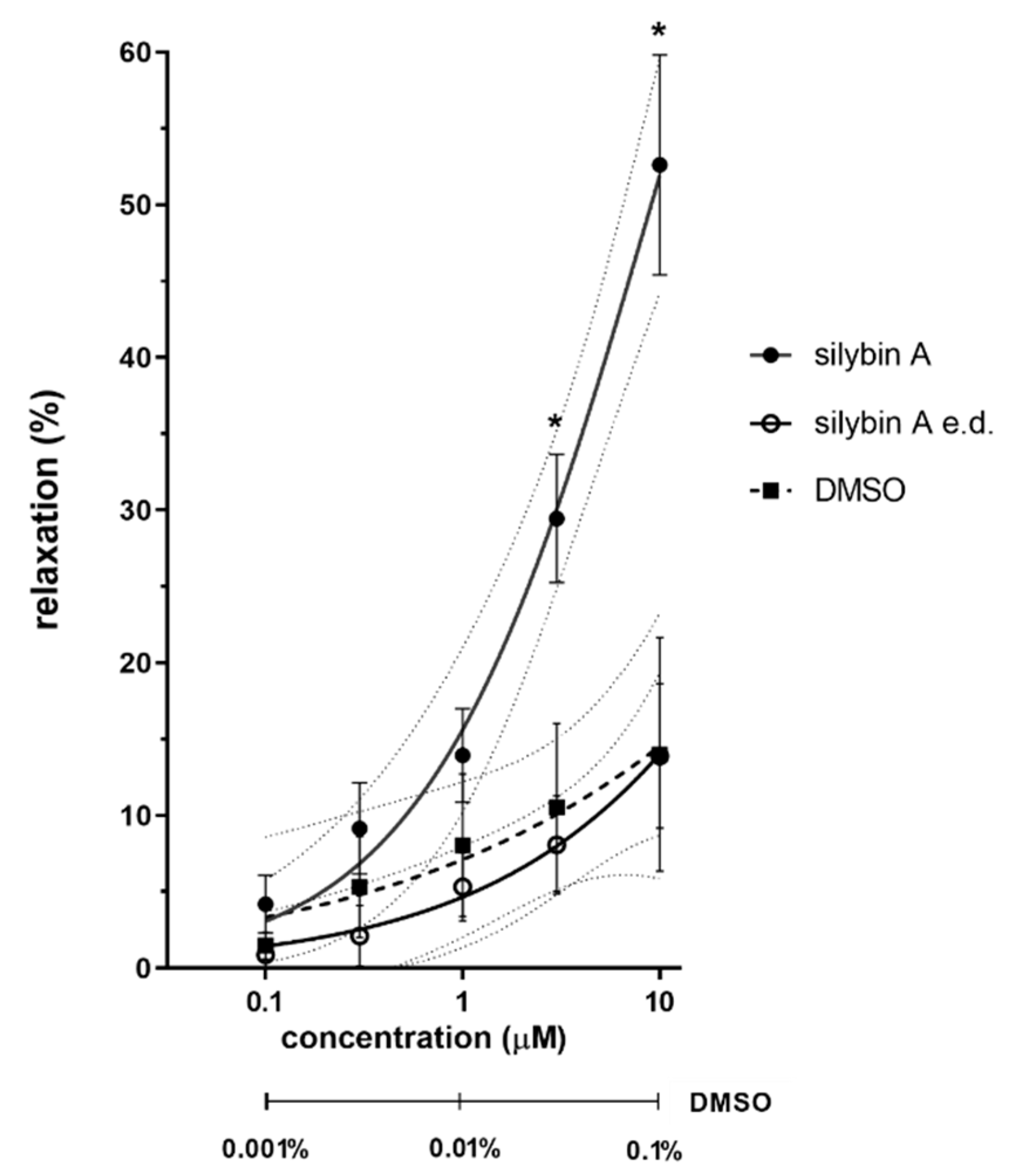
3.1. Ex Vivo Experiments on Isolated Rat Aorta
3.2. Platelet Aggregation Experiments
4. Discussion
4.1. Ex Vivo Experiments on Isolated Rat Aorta
4.2. Platelet Aggregation Experiments
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ASA | Acetylsalicylic acid |
| COX | Cyclooxygenase |
| DMSO | Dimethyl sulfoxide |
| DOCA | Deoxycorticosterone acetate |
| EC50 | Half maximal effective concentrations |
| e.d. | Endothelium-denuded |
| IM CAS | Institute of Microbiology of the Czech Academy of Sciences |
| TXA2 | Thromboxane A2 |
| U-46619 | Synthetic analog of prostaglandin H2 |
References
- Gažák, R.; Walterová, D.; Křen, V. Silybin and silymarin--new and emerging applications in medicine. Curr. Med. Chem. 2007, 14, 315–338. [Google Scholar] [CrossRef] [PubMed]
- Tajmohammadi, A.; Razavi, B.M.; Hosseinzadeh, H. Silybum marianum (milk thistle) and its main constituent, silymarin, as a potential therapeutic plant in metabolic syndrome: A review. Phytother. Res. 2018, 32, 1933–1949. [Google Scholar] [CrossRef] [PubMed]
- Taleb, A.; Ahmad, K.A.; Ihsan, A.U.; Qu, J.; Lin, N.; Hezam, K.; Koju, N.; Hui, L.; Qilong, D. Antioxidant effects and mechanism of silymarin in oxidative stress induced cardiovascular diseases. Biomed. Pharmacother. 2018, 102, 689–698. [Google Scholar] [CrossRef] [PubMed]
- Vahabzadeh, M.; Amiri, N.; Karimi, G. Effects of silymarin on metabolic syndrome: A review. J. Sci. Food Agric. 2018, 98, 4816–4823. [Google Scholar] [CrossRef] [PubMed]
- Bahem, R.; Hoffmann, A.; Azonpi, A.; Caballero-George, C.; Vanderheyden, P. Modulation of calcium signaling of angiotensin AT1, endothelin ETA, and ETB receptors by silibinin, quercetin, crocin, diallyl sulfides, and ginsenoside Rb1. Planta Med. 2015, 81, 670–678. [Google Scholar] [CrossRef] [PubMed]
- Demirci, B.; Dost, T.; Gokalp, F.; Birincioglu, M. Silymarin improves vascular function of aged ovariectomized rats. Phytother. Res. 2014, 28, 868–872. [Google Scholar] [CrossRef] [PubMed]
- Li Volti, G.; Salomone, S.; Sorrenti, V.; Mangiameli, A.; Urso, V.; Siarkos, I.; Galvano, F.; Salamone, F. Effect of silibinin on endothelial dysfunction and ADMA levels in obese diabetic mice. Cardiovasc. Diabetol. 2011, 10, 62. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.S.; Park, S.K.; Yang, K.H.; Kim, H.M. Silymarin inhibits TNF-alpha-induced expression of adhesion molecules in human umbilical vein endothelial cells. FEBS Lett. 2003, 550, 89–93. [Google Scholar] [CrossRef]
- El-Shitany, N.A.; El-Haggar, S.; El-Desoky, K. Silymarin prevents adriamycin-induced cardiotoxicity and nephrotoxicity in rats. Food Chem. Toxicol. 2008, 46, 2422–2428. [Google Scholar] [CrossRef]
- Taghiabadi, E.; Imenshahidi, M.; Abnous, K.; Mosafa, F.; Sankian, M.; Memar, B.; Karimi, G. Protective effect of silymarin against acrolein-induced cardiotoxicity in mice. Evid. Based Complement. Alternat. Med. 2012, 2012, 352091. [Google Scholar] [CrossRef]
- Metwally, M.A.A.; El-Gellal, A.M.; El-Sawaisi, S.M. Effects of silymarin on lipid metabolism in rats. World Appl. Sci. J. 2009, 6, 1634–1637. [Google Scholar]
- Heidarian, E.; Rafieian-Kopaei, M. Effect of silymarin on liver phoshpatidate phosphohydrolase in hyperlipidemic rats. Biosci. Res. 2012, 9, 59–67. [Google Scholar]
- Krečman, V.; Škottová, N.; Walterová, D.; Ulrichová, J.; Šimánek, V. Silymarin inhibits the development of diet-induced hypercholesterolemia in rats. Planta Med. 1998, 64, 138–142. [Google Scholar] [CrossRef]
- Škottová, N.; Krečman, V. Dietary silymarin improves removal of low density lipoproteins by the perfused rat liver. Acta Univ. Palacki. Olomouc. Fac. Med. 1998, 141, 39–40. [Google Scholar]
- Bijak, M.; Saluk-Bijak, J. Flavonolignans inhibit the arachidonic acid pathway in blood platelets. BMC Complement. Altern. Med. 2017, 17, 396–403. [Google Scholar] [CrossRef]
- Hawke, R.L.; Schrieber, S.J.; Soule, T.A.; Wen, Z.; Smith, P.C.; Reddy, K.R.; Wahed, A.S.; Belle, S.H.; Afdhal, N.H.; Navarro, V.J.; et al. Silymarin ascending multiple oral dosing phase I study in noncirrhotic patients with chronic hepatitis C. J. Clin. Pharmacol. 2010, 50, 434–449. [Google Scholar] [CrossRef]
- Chambers, C.S.; Holečková, V.; Petrásková, L.; Biedermann, D.; Valentová, K.; Buchta, M.; Křen, V. The silymarin composition... and why does it matter??? Food Res. Int. 2017, 100, 339–353. [Google Scholar] [CrossRef]
- Biedermann, D.; Vavříková, E.; Cvak, L.; Křen, V. Chemistry of silybin. Nat. Prod. Rep. 2014, 31, 1138–1157. [Google Scholar] [CrossRef]
- Gažák, R.; Marhol, P.; Purchartová, K.; Monti, D.; Biedermann, D.; Riva, S.; Cvak, L.; Křen, V. Large-scale separation of silybin diastereoisomers using lipases. Process Biochem. 2010, 45, 1657–1663. [Google Scholar] [CrossRef]
- Křenek, K.; Marhol, P.; Peikerová, Ž.; Křen, V.; Biedermann, D. Preparatory separation of the silymarin flavonolignans by Sephadex LH-20 gel. Food Res. Int. 2014, 65, 115–120. [Google Scholar] [CrossRef]
- Džubák, P.; Hajdúch, M.; Gažák, R.; Svobodová, A.; Psotová, J.; Walterová, D.; Sedmera, P.; Křen, V. New derivatives of silybin and 2,3-dehydrosilybin and their cytotoxic and P-glycoprotein modulatory activity. Bioorg. Med. Chem. 2006, 14, 3793–3810. [Google Scholar] [CrossRef]
- Valentová, K.; Purchartová, K.; Rydlová, L.; Roubalová, L.; Biedermann, D.; Petrásková, L.; Křenková, A.; Pelantová, H.; Holečková-Moravcová, V.; Tesařová, E.; et al. Sulfated metabolites of flavonolignans and 2,3-dehydroflavonolignans: Preparation and properties. Int. J. Mol. Sci. 2018, 19, 2349. [Google Scholar] [CrossRef]
- Company, C.C. COX (Ovine/Human) Inhibitor Screening Assay Kit. Available online: https://www.caymanchem.com/product/560131 (accessed on 15 July 2019).
- Chang, T.S.; Kim, H.M.; Lee, K.S.; Khil, L.Y.; Mar, W.C.; Ryu, C.K.; Moon, C.K. Thromboxane A2 synthase inhibition and thromboxane A2 receptor blockade by 2-[(4-cyanophenyl)amino]-3-chloro-1,4-naphthalenedione (NQ-Y15) in rat platelets. Biochem. Pharmacol. 1997, 54, 259–268. [Google Scholar] [CrossRef]
- Company, C.C. Thromboxane B2 ELISA Kit. Available online: https://www.caymanchem.com/product/501020 (accessed on 15 July 2019).
- Theodosiou, E.; Purchartová, K.; Stamatis, H.; Kolisis, F.; Křen, V. Bioavailability of silymarin flavonolignans: Drug formulations and biotransformation. Phytochem. Rev. 2014, 13, 1–18. [Google Scholar] [CrossRef]
- Vrba, J.; Papoušková, B.; Roubalová, L.; Zatloukalová, M.; Biedermann, D.; Křen, V.; Valentová, K.; Ulrichová, J.; Vacek, J. Metabolism of flavonolignans in human hepatocytes. J. Pharm. Biomed. Anal. 2018, 152, 94–101. [Google Scholar] [CrossRef]
- Marhol, P.; Bednář, P.; Kolářová, P.; Večeřa, R.; Ulrichova, J.; Tesařová, E.; Vavříková, E.; Kuzma, M.; Křen, V. Pharmacokinetics of pure silybin diastereoisomers and identification of their metabolites in rat plasma. J. Funct. Foods 2015, 14, 570–580. [Google Scholar] [CrossRef]
- Han, Y.H.; Lou, H.X.; Ren, D.M.; Sun, L.R.; Ma, B.; Ji, M. Stereoselective metabolism of silybin diastereoisomers in the glucuronidation process. J. Pharm. Biomed. Anal. 2004, 34, 1071–1078. [Google Scholar] [CrossRef]
- Jančová, P.; Šiller, M.; Anzenbacherová, E.; Křen, V.; Anzenbacher, P.; Šimánek, V. Evidence for differences in regioselective and stereoselective glucuronidation of silybin diastereomers from milk thistle (Silybum marianum) by human UDP-glucuronosyltransferases. Xenobiotica 2011, 41, 743–751. [Google Scholar] [CrossRef]
- Křen, V.; Ulrichová, J.; Kosina, P.; Stevenson, D.; Sedmera, P.; Přikrylová, V.; Halada, P.; Šimánek, V. Chemoenzymatic preparation of silybin β-glucuronides and their biological evaluation. Drug Metab. Dispos. 2000, 28, 1513–1517. [Google Scholar]
- Najmanová, I.; Pourová, J.; Vopršalová, M.; Pilarová, V.; Semecký, V.; Novaková, L.; Mladěnka, P. Flavonoid metabolite 3-(3-hydroxyphenyl)propionic acid formed by human microflora decreases arterial blood pressure in rats. Mol. Nutr. Food Res. 2016, 60, 981–991. [Google Scholar] [CrossRef]
- Pourová, J.; Najmanová, I.; Vopršalová, M.; Migkos, T.; Pilarová, V.; Applová, L.; Novaková, L.; Mladěnka, P. Two flavonoid metabolites, 3,4-dihydroxyphenylacetic acid and 4-methylcatechol, relax arteries ex vivo and decrease blood pressure in vivo. Vascul. Pharmacol. 2018, 111, 36–43. [Google Scholar] [CrossRef]
- Ribaudo, G.; Pagano, M.A.; Pavan, V.; Redaelli, M.; Zorzan, M.; Pezzani, R.; Mucignat-Caretta, C.; Vendrame, T.; Bova, S.; Zagotto, G. Semi-synthetic derivatives of natural isoflavones from Maclura pomifera as a novel class of PDE-5A inhibitors. Fitoterapia 2015, 105, 132–138. [Google Scholar] [CrossRef]
- Fermini, B.; Ramirez, D.S.; Sun, S.; Bassyouni, A.; Hemkens, M.; Wisialowski, T.; Jenkinson, S. L-type calcium channel antagonism - Translation from in vitro to in vivo. J. Pharmacol. Toxicol. Methods 2017, 84, 86–92. [Google Scholar] [CrossRef]
- Egea, J.; Fabregat, I.; Frapart, Y.M.; Ghezzi, P.; Görlach, A.; Kietzmann, T.; Kubaichuk, K.; Knaus, U.G.; Lopez, M.G.; Olaso-Gonzalez, G.; et al. European contribution to the study of ROS: A summary of the findings and prospects for the future from the COST action BM1203 (EU-ROS). Redox Biol. 2017, 13, 94–162. [Google Scholar] [CrossRef]
- Parveen, R.; Baboota, S.; Ali, J.; Ahuja, A.; Vasudev, S.S.; Ahmad, S. Oil based nanocarrier for improved oral delivery of silymarin: In vitro and in vivo studies. Int. J. Pharm. 2011, 413, 245–253. [Google Scholar] [CrossRef]
- Jadhav, G.B.; Upasani, C.D. Antihypertensive effect of Silymarin on DOCA salt induced hypertension in unilateral nephrectomized rats. Orient. Pharm. Exp. Med. 2011, 11, 101–106. [Google Scholar] [CrossRef]
- Chen, H.; Chen, S.C.; Zhang, T.H.; Tian, H.C.; Guan, Y.; Su, D.F. Protective effects of silybin and tetrandrine on the outcome of spontaneously hypertensive rats subjected to acute coronary artery occlusion. Int. J. Cardiol. 1993, 41, 103–108. [Google Scholar] [CrossRef]
- Mosua, A.M.; Numan, A.T.; Saeed, B.N. Adjuvant use of silymarin in patients with hypertension and microalbuminuria. Int. Res. J. Pharm. 2012, 3, 95–96. [Google Scholar]
- Calani, L.; Brighenti, F.; Bruni, R.; Del Rio, D. Absorption and metabolism of milk thistle flavanolignans in humans. Phytomedicine 2012, 20, 40–46. [Google Scholar] [CrossRef]
- Jackson, S.P. The growing complexity of platelet aggregation. Blood 2007, 109, 5087–5095. [Google Scholar] [CrossRef]
- Bijak, M.; Dziedzic, A.; Saluk-Bijak, J. Flavonolignans reduce the response of blood platelet to collagen. Int. J. Biol. Macromol. 2018, 106, 878–884. [Google Scholar] [CrossRef]
- Bijak, M.; Szelenberger, R.; Saluk, J.; Nowak, P. Flavonolignans inhibit ADP induced blood platelets activation and aggregation in whole blood. Int. J. Biol. Macromol. 2017, 95, 682–688. [Google Scholar] [CrossRef]
- Dehmlow, C.; Murawski, N.; de Groot, H. Scavenging of reactive oxygen species and inhibition of arachidonic acid metabolism by silibinin in human cells. Life Sci. 1996, 58, 1591–1600. [Google Scholar] [CrossRef]
- Rui, Y.C. Advances in pharmacological studies of silymarin. Mem. Inst. Oswaldo Cruz 1991, 86 (Suppl. 2), 79–85. [Google Scholar] [CrossRef]
- Litvinov, R.I.; Weisel, J.W. Role of red blood cells in haemostasis and thrombosis. ISBT Sci. Ser. 2017, 12, 176–183. [Google Scholar] [CrossRef]
- Karlíčková, J.; Říha, M.; Filipský, T.; Macaková, K.; Hrdina, R.; Mladěnka, P. Antiplatelet effects of flavonoids mediated by inhibition of arachidonic acid based pathway. Planta Med. 2016, 82, 76–83. [Google Scholar] [CrossRef]
- Applová, L.; Karlíčková, J.; Říha, M.; Filipský, T.; Macáková, K.; Spilková, J.; Mladěnka, P. The isoflavonoid tectorigenin has better antiplatelet potential than acetylsalicylic acid. Phytomedicine 2017, 35, 11–17. [Google Scholar] [CrossRef]



© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pourová, J.; Applová, L.; Macáková, K.; Vopršalová, M.; Migkos, T.; Bentanachs, R.; Biedermann, D.; Petrásková, L.; Tvrdý, V.; Hrubša, M.; et al. The Effect of Silymarin Flavonolignans and Their Sulfated Conjugates on Platelet Aggregation and Blood Vessels Ex Vivo. Nutrients 2019, 11, 2286. https://doi.org/10.3390/nu11102286
Pourová J, Applová L, Macáková K, Vopršalová M, Migkos T, Bentanachs R, Biedermann D, Petrásková L, Tvrdý V, Hrubša M, et al. The Effect of Silymarin Flavonolignans and Their Sulfated Conjugates on Platelet Aggregation and Blood Vessels Ex Vivo. Nutrients. 2019; 11(10):2286. https://doi.org/10.3390/nu11102286
Chicago/Turabian StylePourová, Jana, Lenka Applová, Kateřina Macáková, Marie Vopršalová, Thomas Migkos, Roger Bentanachs, David Biedermann, Lucie Petrásková, Václav Tvrdý, Marcel Hrubša, and et al. 2019. "The Effect of Silymarin Flavonolignans and Their Sulfated Conjugates on Platelet Aggregation and Blood Vessels Ex Vivo" Nutrients 11, no. 10: 2286. https://doi.org/10.3390/nu11102286
APA StylePourová, J., Applová, L., Macáková, K., Vopršalová, M., Migkos, T., Bentanachs, R., Biedermann, D., Petrásková, L., Tvrdý, V., Hrubša, M., Karlíčková, J., Křen, V., Valentová, K., & Mladěnka, P. (2019). The Effect of Silymarin Flavonolignans and Their Sulfated Conjugates on Platelet Aggregation and Blood Vessels Ex Vivo. Nutrients, 11(10), 2286. https://doi.org/10.3390/nu11102286