Effects of a High-Protein/Moderate-Carbohydrate Diet on Appetite, Gut Peptides, and Endocannabinoids—A Preview Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Experimental Design
2.3. Anthropometric Measurements
2.4. Respiration Chamber
2.5. Diets and Energy Intake
2.6. Appetite Profile
2.7. Metabolic Parameters
2.7.1. Endocannabinoids and Endocannabinoid-Related Compounds
2.7.2. GLP-1 and PYY
2.7.3. Glucose and Insulin
2.7.4. β-Hydroxybutyrate
2.7.5. Triacylglycerol
2.8. Statistical Analysis
3. Results
3.1. Study Participants
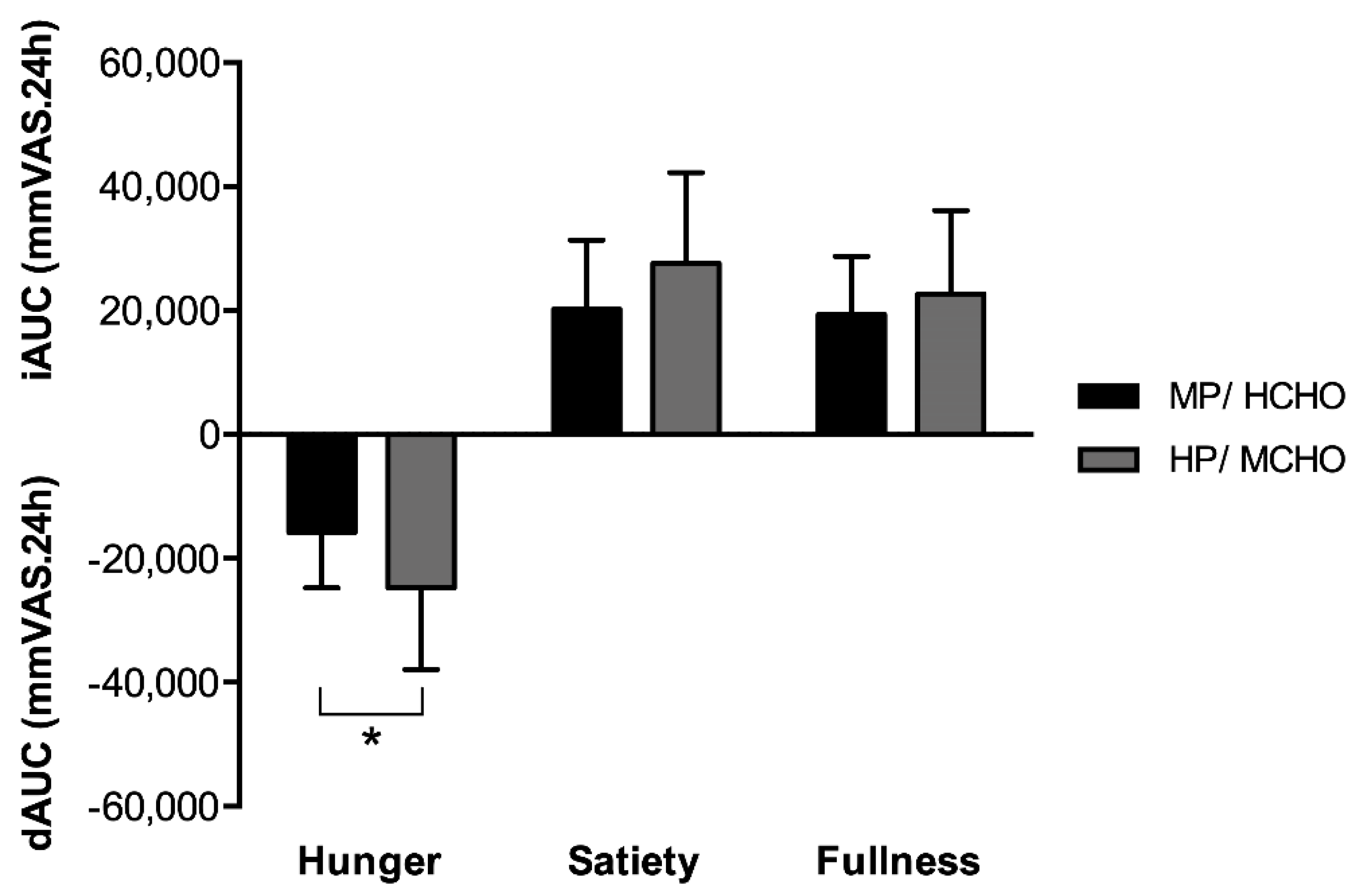
3.2. 24-Hour Hunger and Satiety Perception
3.3. Metabolic Parameters
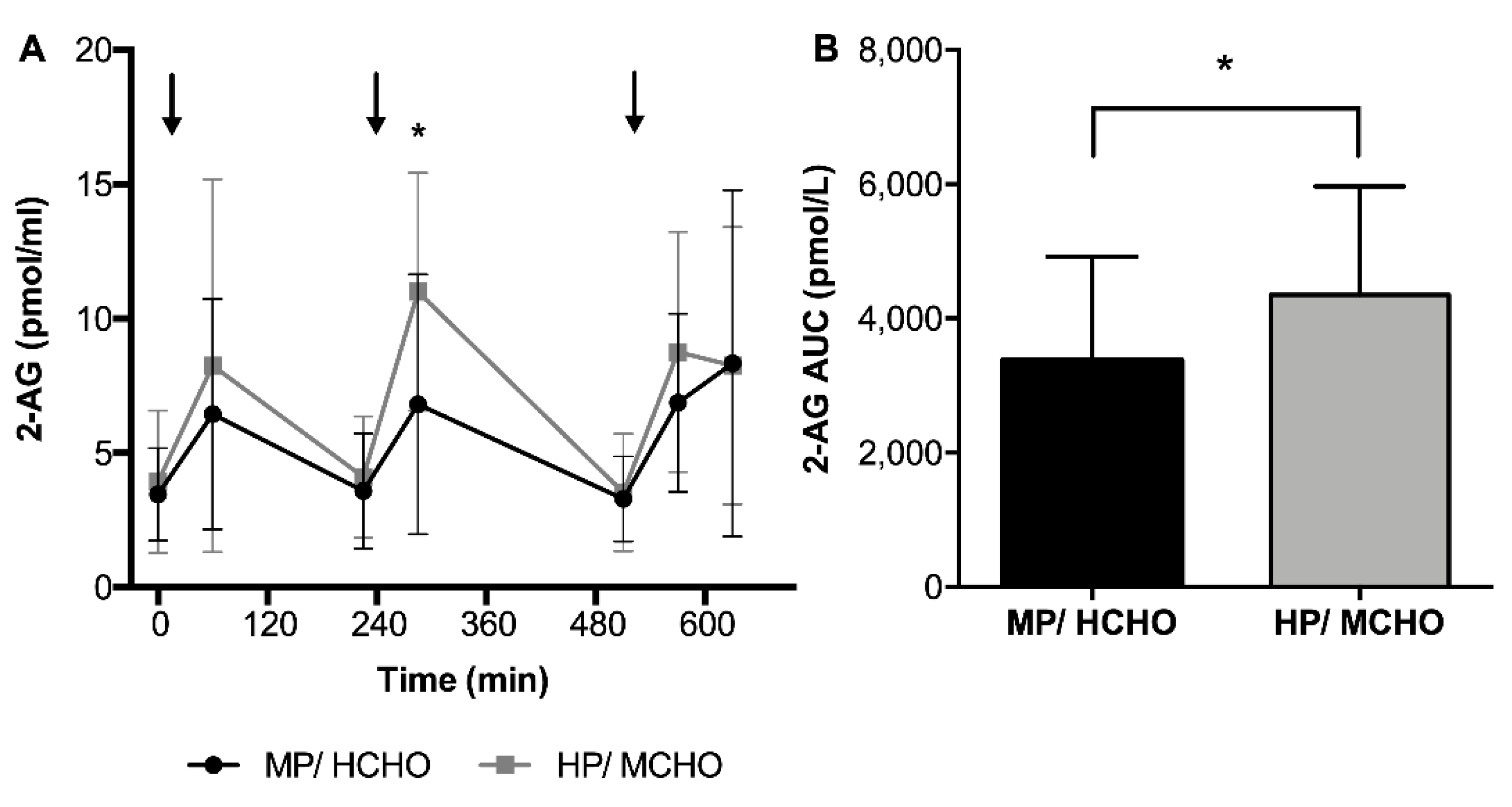
3.3.1. Endocannabinoids and Related Compounds Concentrations Throughout the Day
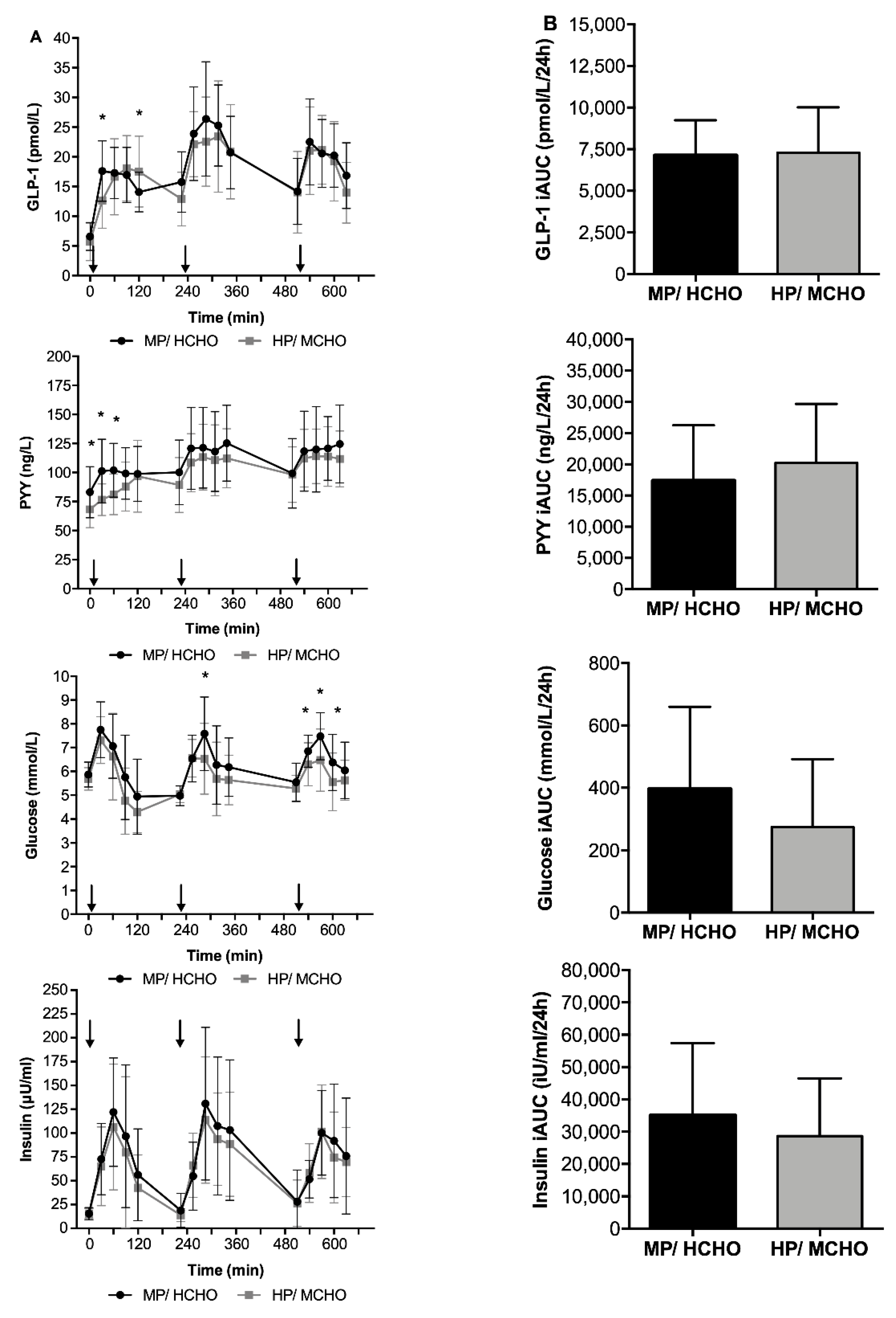
3.3.2. Gut peptides, Glucose, and Insulin Concentrations Throughout the Day
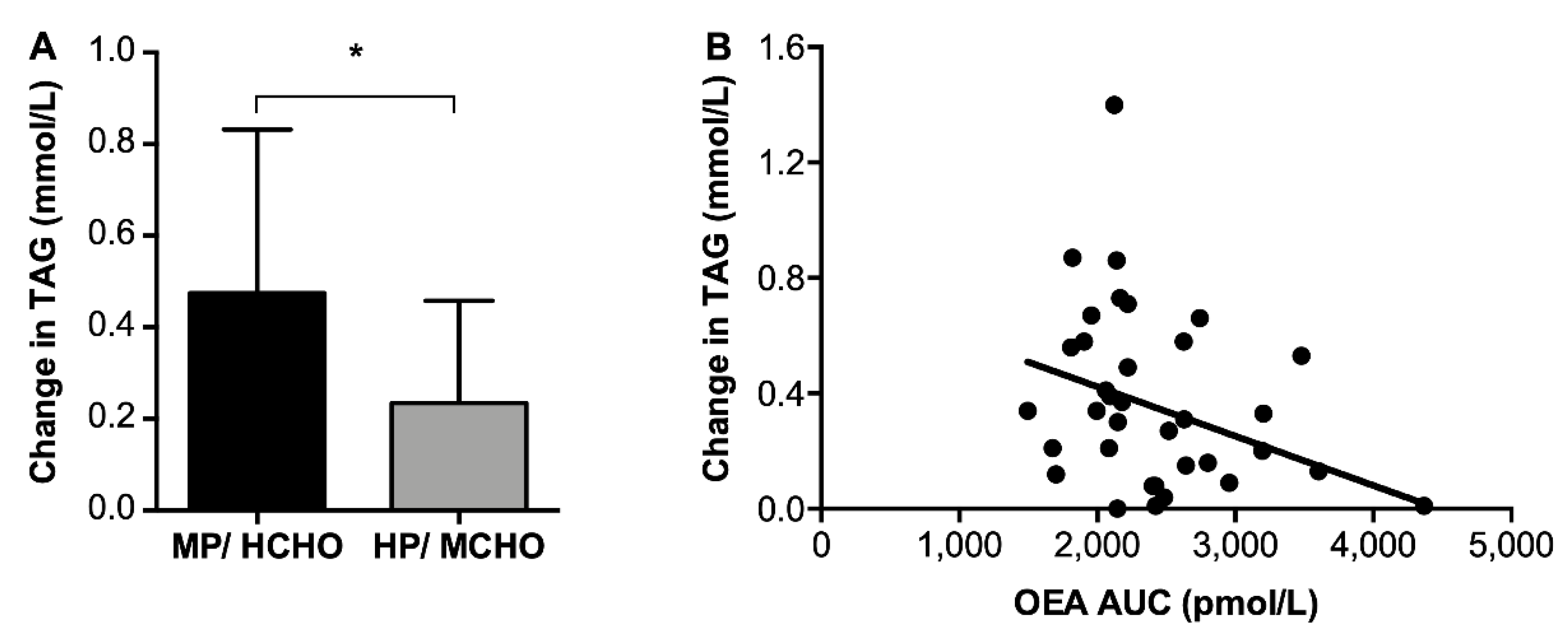
3.3.3. Fasted β-Hydroxybutyrate and Triacylglycerol Concentrations
3.4. Ad Libitum Energy Intake After Leaving the Chamber
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| 2-AG | 2-arachidonoylglycerol |
| AEA | anandamide |
| AUC | area under the curve |
| BHB | β-hydroxybutyrate |
| BMI | body mass index |
| BMR | basal metabolic rate |
| CB1 | cannabinoid receptor 1 |
| CB2 | cannabinoid receptor 2 |
| dAUC | decremental area under the curve |
| EDTA | ethylenediaminetetraacetic acid |
| EI | energy intake |
| FFM | fat-free mass |
| FM | fat mass |
| GC-MS | gas chromatography-mass spectrometry |
| GC-MS/MS | gas chromatography-tandem mass spectrometry |
| GI | glycemic index |
| GLP-1 | glucagon-like peptide-1 |
| HOMA-IR | homeostatic model assessment for insulin resistance |
| HP/MCHO | high protein/ moderate carbohydrate |
| iAUC | incremental area under the curve |
| MJ | megajoule |
| mmVAS | milimeter visiual analogue scale |
| MP/ HCHO | moderate-protein/ high-carbohydrate |
| OEA | oleoylethanolamide |
| OGTT | oral glucose tolerance test |
| PEA | palmitoylethanolamide |
| PMSF | phenylmethylsulfonyl fluoride |
| PREG | pregnenolone |
| PYY | polypeptide YY |
| TAG | triacylglycerol |
| T2D | type II diabetes |
| VAS | visual analogue scale |
References
- World Health Organization. Global Health Observatory (GHO) Data. Available online: https://www.who.int/gho/ncd/risk_factors/overweight/en/ (accessed on 5 August 2019).
- World Health Organization. Global Atlas on Cardiovascular Disease Prevention and Control; Shanti Mendis, P.P., Bo, N., Eds.; World Health Organization: Geneva, Switzerland, 2011; pp. 3–18. [Google Scholar]
- Anderson, G.H.; Moore, S.E. Dietary proteins in the regulation of food intake and body weight in humans. J. Nutr. 2004, 134, 974S–979S. [Google Scholar] [CrossRef] [PubMed]
- Astrup, A. The satiating power of protein—A key to obesity prevention? Am. J. Clin. Nutr 2005, 82, 1–2. [Google Scholar] [CrossRef] [PubMed]
- Halton, T.L.; Hu, F.B. The effects of high protein diets on thermogenesis, satiety and weight loss: A critical review. J. Am. Coll. Nutr. 2004, 23, 373–385. [Google Scholar] [CrossRef] [PubMed]
- Paddon-Jones, D.; Westman, E.; Mattes, R.D.; Wolfe, R.R.; Astrup, A.; Westerterp-Plantenga, M. Protein, weight management, and satiety. Am. J. Clin. Nutr. 2008, 87, 1558s–1561s. [Google Scholar] [CrossRef]
- Reid, M.; Hetherington, M. Relative effects of carbohydrates and protein on satiety—A review of methodology. Neurosci. Biobehav. Rev. 1997, 21, 295–308. [Google Scholar] [CrossRef]
- Veldhorst, M.A.; Nieuwenhuizen, A.G.; Hochstenbach-Waelen, A.; Westerterp, K.R.; Engelen, M.P.; Brummer, R.J.; Deutz, N.E.; Westerterp-Plantenga, M.S. A breakfast with α-lactalbumin, gelatin, or gelatin + TRP lowers energy intake at lunch compared with a breakfast with casein, soy, whey, or whey-GMP. Clin. Nutr. 2009, 28, 147–155. [Google Scholar] [CrossRef] [PubMed]
- Martens, E.A.; Gonnissen, H.K.; Gatta-Cherifi, B.; Janssens, P.L.; Westerterp-Plantenga, M.S. Maintenance of energy expenditure on high-protein vs. high-carbohydrate diets at a constant body weight may prevent a positive energy balance. Clin. Nutr. 2015, 34, 968–975. [Google Scholar] [CrossRef]
- Veldhorst, M.A.; Westerterp, K.R.; Westerterp-Plantenga, M.S. Gluconeogenesis and protein-induced satiety. Br. J. Nutr. 2012, 107, 595–600. [Google Scholar] [CrossRef]
- Soenen, S.; Martens, E.A.; Hochstenbach-Waelen, A.; Lemmens, S.G.; Westerterp-Plantenga, M.S. Normal protein intake is required for body weight loss and weight maintenance, and elevated protein intake for additional preservation of resting energy expenditure and fat free mass. J. Nutr. 2013, 143, 591–596. [Google Scholar] [CrossRef]
- Westerterp-Plantenga, M.S.; Lejeune, M.P.; Nijs, I.; van Ooijen, M.; Kovacs, E.M. High protein intake sustains weight maintenance after body weight loss in humans. Int. J. Obes. Relat. Metab. Disord. 2004, 28, 57–64. [Google Scholar] [CrossRef]
- Lejeune, M.P.; Westerterp, K.R.; Adam, T.C.; Luscombe-Marsh, N.D.; Westerterp-Plantenga, M.S. Ghrelin and glucagon-like peptide 1 concentrations, 24-h satiety, and energy and substrate metabolism during a high-protein diet and measured in a respiration chamber. Am. J. Clin. Nutr. 2006, 83, 89–94. [Google Scholar] [CrossRef]
- Westerterp-Plantenga, M.S.; Rolland, V.; Wilson, S.A.; Westerterp, K.R. Satiety related to 24 h diet-induced thermogenesis during high protein/carbohydrate vs high fat diets measured in a respiration chamber. Eur. J. Clin. Nutr. 1999, 53, 495–502. [Google Scholar] [CrossRef] [PubMed]
- Mazier, W.; Saucisse, N.; Gatta-Cherifi, B.; Cota, D. The Endocannabinoid System: Pivotal Orchestrator of Obesity and Metabolic Disease. Trends Endocrinol. Metab. 2015, 26, 524–537. [Google Scholar] [CrossRef]
- Silvestri, C.; Di Marzo, V. The endocannabinoid system in energy homeostasis and the etiopathology of metabolic disorders. Cell Metab. 2013, 17, 475–490. [Google Scholar] [CrossRef]
- Simon, V.; Cota, D. Mechanisms in Endocrinology: Endocannabinoids and metabolism: Past, present and future. Eur. J. Endocrinol. 2017, 176, R309–R324. [Google Scholar] [CrossRef]
- Bluher, M.; Engeli, S.; Kloting, N.; Berndt, J.; Fasshauer, M.; Batkai, S.; Pacher, P.; Schon, M.R.; Jordan, J.; Stumvoll, M. Dysregulation of the peripheral and adipose tissue endocannabinoid system in human abdominal obesity. Diabetes 2006, 55, 3053–3060. [Google Scholar] [CrossRef]
- Cote, M.; Matias, I.; Lemieux, I.; Petrosino, S.; Almeras, N.; Despres, J.P.; Di Marzo, V. Circulating endocannabinoid levels, abdominal adiposity and related cardiometabolic risk factors in obese men. Int. J. Obes. 2007, 31, 692–699. [Google Scholar] [CrossRef]
- Engeli, S.; Lehmann, A.C.; Kaminski, J.; Haas, V.; Janke, J.; Zoerner, A.A.; Luft, F.C.; Tsikas, D.; Jordan, J. Influence of dietary fat intake on the endocannabinoid system in lean and obese subjects. Obesity 2014, 22, E70–E76. [Google Scholar] [CrossRef]
- Piomelli, D. A fatty gut feeling. Trends Endocrinol. Metab. 2013, 24, 332–341. [Google Scholar] [CrossRef]
- Vallee, M.; Vitiello, S.; Bellocchio, L.; Hebert-Chatelain, E.; Monlezun, S.; Martin-Garcia, E.; Kasanetz, F.; Baillie, G.L.; Panin, F.; Cathala, A.; et al. Pregnenolone can protect the brain from cannabis intoxication. Science 2014, 343, 94–98. [Google Scholar] [CrossRef]
- Pagotto, U.; Marsicano, G.; Cota, D.; Lutz, B.; Pasquali, R. The emerging role of the endocannabinoid system in endocrine regulation and energy balance. Endocr. Rev. 2006, 27, 73–100. [Google Scholar] [CrossRef]
- Batterham, R.L.; Heffron, H.; Kapoor, S.; Chivers, J.E.; Chandarana, K.; Herzog, H.; Le Roux, C.W.; Thomas, E.L.; Bell, J.D.; Withers, D.J. Critical role for peptide YY in protein-mediated satiation and body-weight regulation. Cell Metab. 2006, 4, 223–233. [Google Scholar] [CrossRef]
- Westerterp-Plantenga, M.S.; Lejeune, M.P.; Smeets, A.J.; Luscombe-Marsh, N.D. Sex differences in energy homeostatis following a diet relatively high in protein exchanged with carbohydrate, assessed in a respiration chamber in humans. Physiol. Behav. 2009, 97, 414–419. [Google Scholar] [CrossRef]
- De Graaf, C.; Blom, W.A.; Smeets, P.A.; Stafleu, A.; Hendriks, H.F. Biomarkers of satiation and satiety. Am. J. Clin. Nutr. 2004, 79, 946–961. [Google Scholar] [CrossRef]
- Leidy, H.J.; Armstrong, C.L.; Tang, M.; Mattes, R.D.; Campbell, W.W. The influence of higher protein intake and greater eating frequency on appetite control in overweight and obese men. Obesity 2010, 18, 1725–1732. [Google Scholar] [CrossRef]
- Lemmens, S.G.; Martens, E.A.; Kester, A.D.; Westerterp-Plantenga, M.S. Changes in gut hormone and glucose concentrations in relation to hunger and fullness. Am. J. Clin. Nutr. 2011, 94, 717–725. [Google Scholar] [CrossRef]
- Fogelholm, M.; Larsen, T.M.; Westerterp-Plantenga, M.; Macdonald, I.; Martinez, J.A.; Boyadjieva, N.; Poppitt, S.; Schlicht, W.; Stratton, G.; Sundvall, J.; et al. Preview: Prevention of Diabetes through Lifestyle Intervention and Population Studies in Europe and around the World. Design, Methods, and Baseline Participant Description of an Adult Cohort Enrolled into a Three-Year Randomised Clinical Trial. Nutrients 2017, 9. [Google Scholar] [CrossRef]
- Drummen, M.; Tischmann, L.; Gatta-Cherifi, B.; Fogelholm, M.; Raben, A.; Adam, T.C.; Westerterp-Plantenga, M. High versus moderate protein intake reduces adaptive thermogenesis and induces a negative energy balance during long-term weight loss maintenance—A preview study. Under Review. 2019. [Google Scholar]
- Schoffelen, P.F.; Westerterp, K.R.; Saris, W.H.; Ten Hoor, F. A dual-respiration chamber system with automated calibration. J. Appl. Physiol. 1997, 83, 2064–2072. [Google Scholar] [CrossRef]
- Weir, J.B. New methods for calculating metabolic rate with special reference to protein metabolism. J. Physiol. 1949, 109, 1–9. [Google Scholar] [CrossRef]
- Hochstenbach-Waelen, A.; Westerterp-Plantenga, M.S.; Veldhorst, M.A.; Westerterp, K.R. Single-protein casein and gelatin diets affect energy expenditure similarly but substrate balance and appetite differently in adults. J. Nutr. 2009, 139, 2285–2292. [Google Scholar] [CrossRef]
- Flint, A.; Raben, A.; Blundell, J.E.; Astrup, A. Reproducibility, power and validity of visual analogue scales in assessment of appetite sensations in single test meal studies. Int. J. Obes. Relat. Metab. Disord. 2000, 24, 38–48. [Google Scholar] [CrossRef]
- Matthews, J.N.; Altman, D.G.; Campbell, M.J.; Royston, P. Analysis of serial measurements in medical research. BMJ 1990, 300, 230–235. [Google Scholar] [CrossRef]
- Gatta-Cherifi, B.; Matias, I.; Vallee, M.; Tabarin, A.; Marsicano, G.; Piazza, P.V.; Cota, D. Simultaneous postprandial deregulation of the orexigenic endocannabinoid anandamide and the anorexigenic peptide YY in obesity. Int. J. Obes. 2012, 36, 880–885. [Google Scholar] [CrossRef]
- Kingsley, P.J.; Marnett, L.J. LC-MS-MS analysis of neutral eicosanoids. Methods Enzymol. 2007, 433, 91–112. [Google Scholar] [CrossRef]
- Orskov, C.; Rabenhoj, L.; Wettergren, A.; Kofod, H.; Holst, J.J. Tissue and plasma concentrations of amidated and glycine-extended glucagon-like peptide I in humans. Diabetes 1994, 43, 535–539. [Google Scholar] [CrossRef]
- Levy, J.C.; Matthews, D.R.; Hermans, M.P. Correct homeostasis model assessment (HOMA) evaluation uses the computer program. Diabetes Care 1998, 21, 2191–2192. [Google Scholar] [CrossRef]
- Matias, I.; Gonthier, M.P.; Orlando, P.; Martiadis, V.; De Petrocellis, L.; Cervino, C.; Petrosino, S.; Hoareau, L.; Festy, F.; Pasquali, R.; et al. Regulation, function, and dysregulation of endocannabinoids in models of adipose and β-pancreatic cells and in obesity and hyperglycemia. J. Clin. Endocrinol. Metab. 2006, 91, 3171–3180. [Google Scholar] [CrossRef]
- Monteleone, P.; Piscitelli, F.; Scognamiglio, P.; Monteleone, A.M.; Canestrelli, B.; Di Marzo, V.; Maj, M. Hedonic eating is associated with increased peripheral levels of ghrelin and the endocannabinoid 2-arachidonoyl-glycerol in healthy humans: A pilot study. J. Clin. Endocrinol. Metab 2012, 97, E917–E924. [Google Scholar] [CrossRef]
- Monteleone, A.M.; Di Marzo, V.; Monteleone, P.; Dalle Grave, R.; Aveta, T.; Ghoch, M.E.; Piscitelli, F.; Volpe, U.; Calugi, S.; Maj, M. Responses of peripheral endocannabinoids and endocannabinoid-related compounds to hedonic eating in obesity. Eur. J. Nutr. 2016, 55, 1799–1805. [Google Scholar] [CrossRef]
- Rigamonti, A.E.; Piscitelli, F.; Aveta, T.; Agosti, F.; De Col, A.; Bini, S.; Cella, S.G.; Di Marzo, V.; Sartorio, A. Anticipatory and consummatory effects of (hedonic) chocolate intake are associated with increased circulating levels of the orexigenic peptide ghrelin and endocannabinoids in obese adults. Food Nutr. Res. 2015, 59, 29678. [Google Scholar] [CrossRef]
- DiPatrizio, N.V.; Astarita, G.; Schwartz, G.; Li, X.; Piomelli, D. Endocannabinoid signal in the gut controls dietary fat intake. Proc. Natl. Acad. Sci. USA 2011, 108, 12904–12908. [Google Scholar] [CrossRef]
- Pintus, S.; Murru, E.; Carta, G.; Cordeddu, L.; Batetta, B.; Accossu, S.; Pistis, D.; Uda, S.; Elena Ghiani, M.; Mele, M.; et al. Sheep cheese naturally enriched in α-linolenic, conjugated linoleic and vaccenic acids improves the lipid profile and reduces anandamide in the plasma of hypercholesterolaemic subjects. Br. J. Nutr. 2013, 109, 1453–1462. [Google Scholar] [CrossRef]
- Hillard, C.J. Circulating Endocannabinoids: From Whence Do They Come and Where are They Going? Neuropsychopharmacology 2018, 43, 155–172. [Google Scholar] [CrossRef]
- Rodriguez de Fonseca, F.; Navarro, M.; Gomez, R.; Escuredo, L.; Nava, F.; Fu, J.; Murillo-Rodriguez, E.; Giuffrida, A.; LoVerme, J.; Gaetani, S.; et al. An anorexic lipid mediator regulated by feeding. Nature 2001, 414, 209–212. [Google Scholar] [CrossRef]
- Racz, B.; Duskova, M.; Vondra, K.; Sramkova, M.; Hill, M.; Starka, L. Daily profiles of steroid hormones and their metabolites related to food intake. Physiol. Res. 2015, 64, S219–S225. [Google Scholar]
- Fu, J.; Gaetani, S.; Oveisi, F.; Lo Verme, J.; Serrano, A.; Rodriguez De Fonseca, F.; Rosengarth, A.; Luecke, H.; Di Giacomo, B.; Tarzia, G.; et al. Oleylethanolamide regulates feeding and body weight through activation of the nuclear receptor PPAR-α. Nature 2003, 425, 90–93. [Google Scholar] [CrossRef]
- Witkamp, R.F. The role of fatty acids and their endocannabinoid-like derivatives in the molecular regulation of appetite. Mol. Aspects Med. 2018, 64, 45–67. [Google Scholar] [CrossRef]
- Guzman, M.; Lo Verme, J.; Fu, J.; Oveisi, F.; Blazquez, C.; Piomelli, D. Oleoylethanolamide stimulates lipolysis by activating the nuclear receptor peroxisome proliferator-activated receptor α (PPAR-α). J. Biol. Chem. 2004, 279, 27849–27854. [Google Scholar] [CrossRef]
- Gatta-Cherifi, B.; Cota, D. Endocannabinoids and Metabolic Disorders. Handb. Exp. Pharmacol. 2015, 231, 367–391. [Google Scholar] [CrossRef]
- El Khoury, D.; El-Rassi, R.; Azar, S.; Hwalla, N. Postprandial ghrelin and PYY responses of male subjects on low carbohydrate meals to varied balancing proportions of proteins and fats. Eur. J. Nutr. 2010, 49, 493–500. [Google Scholar] [CrossRef]
- Paddon-Jones, D.; Leidy, H. Dietary protein and muscle in older persons. Curr. Opin. Clin. Nutr. Metab. Care 2014, 17, 5–11. [Google Scholar] [CrossRef]
- Smeets, A.J.; Soenen, S.; Luscombe-Marsh, N.D.; Ueland, O.; Westerterp-Plantenga, M.S. Energy expenditure, satiety, and plasma ghrelin, glucagon-like peptide 1, and peptide tyrosine-tyrosine concentrations following a single high-protein lunch. J. Nutr. 2008, 138, 698–702. [Google Scholar] [CrossRef]
- Raben, A.; Agerholm-Larsen, L.; Flint, A.; Holst, J.J.; Astrup, A. Meals with similar energy densities but rich in protein, fat, carbohydrate, or alcohol have different effects on energy expenditure and substrate metabolism but not on appetite and energy intake. Am. J. Clin. Nutr. 2003, 77, 91–100. [Google Scholar] [CrossRef]
- Van der Klaauw, A.A.; Keogh, J.M.; Henning, E.; Trowse, V.M.; Dhillo, W.S.; Ghatei, M.A.; Farooqi, I.S. High protein intake stimulates postprandial GLP1 and PYY release. Obesity 2013, 21, 1602–1607. [Google Scholar] [CrossRef]
- Gennuso, K.P.; Gangnon, R.E.; Thraen-Borowski, K.M.; Colbert, L.H. Dose-response relationships between sedentary behaviour and the metabolic syndrome and its components. Diabetologia 2015, 58, 485–492. [Google Scholar] [CrossRef]
- Henson, J.; Yates, T.; Biddle, S.J.; Edwardson, C.L.; Khunti, K.; Wilmot, E.G.; Gray, L.J.; Gorely, T.; Nimmo, M.A.; Davies, M.J. Associations of objectively measured sedentary behaviour and physical activity with markers of cardiometabolic health. Diabetologia 2013, 56, 1012–1020. [Google Scholar] [CrossRef]
- Uebanso, T.; Taketani, Y.; Fukaya, M.; Sato, K.; Takei, Y.; Sato, T.; Sawada, N.; Amo, K.; Harada, N.; Arai, H.; et al. Hypocaloric high-protein diet improves fatty liver and hypertriglyceridemia in sucrose-fed obese rats via two pathways. Am. J. Physiol. Endocrinol. Metab. 2009, 297, E76–E84. [Google Scholar] [CrossRef]
- Margolis, L.M.; Rivas, D.A.; Ezzyat, Y.; Gaffney-Stomberg, E.; Young, A.J.; McClung, J.P.; Fielding, R.A.; Pasiakos, S.M. Calorie Restricted High Protein Diets Downregulate Lipogenesis and Lower Intrahepatic Triglyceride Concentrations in Male Rats. Nutrients 2016, 8, 571. [Google Scholar] [CrossRef]
 ) and HP/MCHO (
) and HP/MCHO ( ) group. Regression lines are shown in black (MP/HCHO) and grey (HP/MCHO, r = 0.710, p = 0.001). MP/HCHO = moderate protein/high carbohydrate; HP/MCHO = high protein/moderate carbohydrate; iAUC = incremental area under the curve; dAUC = decremental area under the curve; PYY = peptide YY.
) group. Regression lines are shown in black (MP/HCHO) and grey (HP/MCHO, r = 0.710, p = 0.001). MP/HCHO = moderate protein/high carbohydrate; HP/MCHO = high protein/moderate carbohydrate; iAUC = incremental area under the curve; dAUC = decremental area under the curve; PYY = peptide YY.
 ) and HP/MCHO (
) and HP/MCHO ( ) group. Regression lines are shown in black (MP/HCHO) and grey (HP/MCHO, r = 0.710, p = 0.001). MP/HCHO = moderate protein/high carbohydrate; HP/MCHO = high protein/moderate carbohydrate; iAUC = incremental area under the curve; dAUC = decremental area under the curve; PYY = peptide YY.
) group. Regression lines are shown in black (MP/HCHO) and grey (HP/MCHO, r = 0.710, p = 0.001). MP/HCHO = moderate protein/high carbohydrate; HP/MCHO = high protein/moderate carbohydrate; iAUC = incremental area under the curve; dAUC = decremental area under the curve; PYY = peptide YY.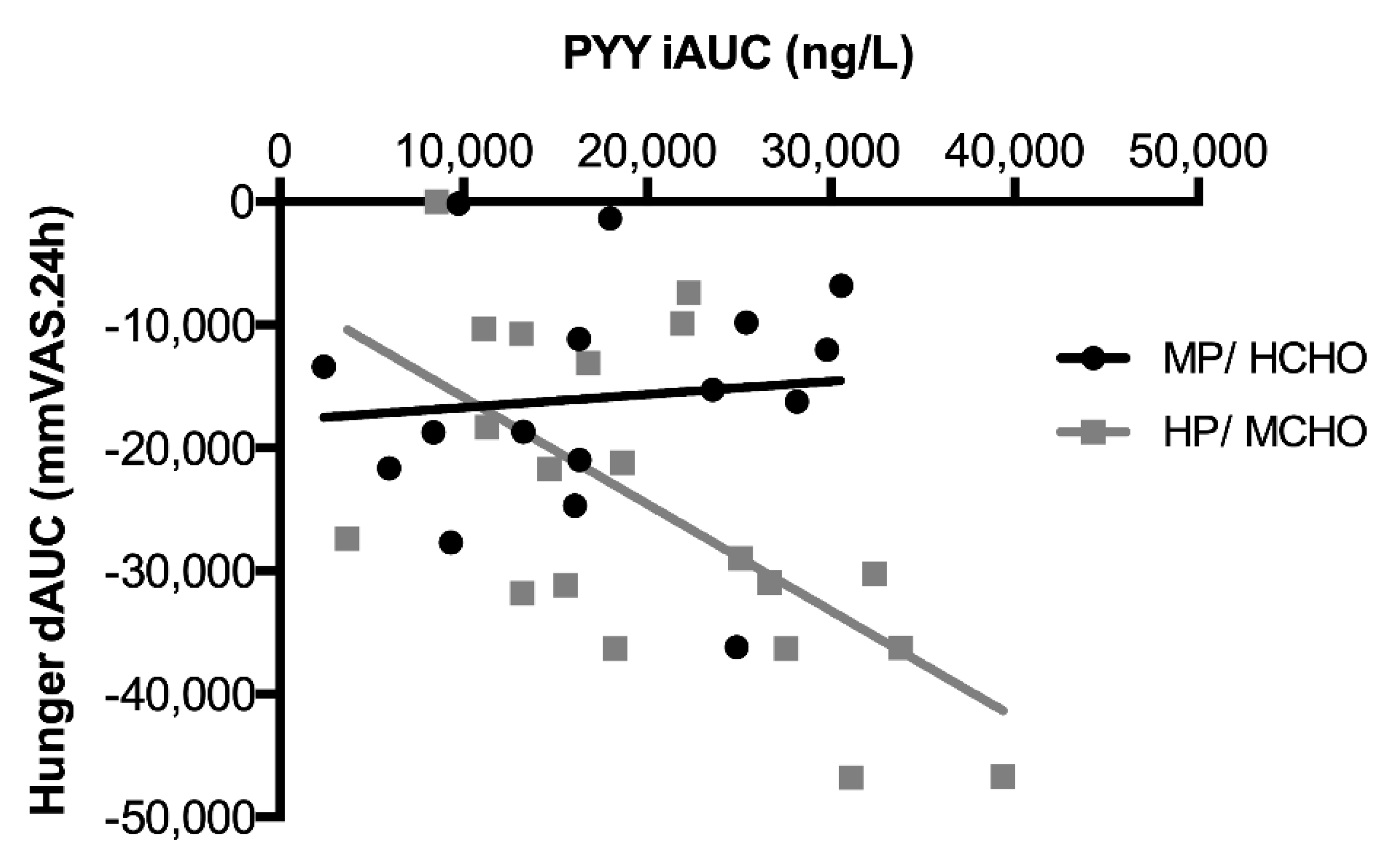
| MP/ HCHO (n = 18) | HP/MCHO (n = 20) | |
|---|---|---|
| N (f/m) | 18 (9/9) | 20 (13/7) |
| Age (year) | 65.1 ± 5.8 | 64.0 ± 6.2 |
| BMI (kg/m2) | 29.0 ± 3.8 | 28.9 ± 4.0 |
| Body-fat (%) | 39.5 ± 8.1 | 40.7 ± 7.7 |
| Fat mass (kg) | 33.9 ± 7.1 | 34.8 ± 8.8 |
| Fat-free mass (kg) | 52.5 ± 10.9 | 50.8 ± 11.3 |
| Glucose (mmol/L) | 5.8 ± 0.4 | 5.7 ± 0.5 |
| Insulin (μU/mL) | 14.8 ± 7.6 | 14.4 ± 4.9 |
| HOMA-IR | 3.8 ± 1.8 | 3.6 ± 1.3 |
| TAG (mmol/L) | 1.5 ± 0.7 | 1.2 ± 0.6 * |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tischmann, L.; Drummen, M.; Gatta-Cherifi, B.; Raben, A.; Fogelholm, M.; Hartmann, B.; Holst, J.J.; Matias, I.; Cota, D.; Mensink, R.P.; et al. Effects of a High-Protein/Moderate-Carbohydrate Diet on Appetite, Gut Peptides, and Endocannabinoids—A Preview Study. Nutrients 2019, 11, 2269. https://doi.org/10.3390/nu11102269
Tischmann L, Drummen M, Gatta-Cherifi B, Raben A, Fogelholm M, Hartmann B, Holst JJ, Matias I, Cota D, Mensink RP, et al. Effects of a High-Protein/Moderate-Carbohydrate Diet on Appetite, Gut Peptides, and Endocannabinoids—A Preview Study. Nutrients. 2019; 11(10):2269. https://doi.org/10.3390/nu11102269
Chicago/Turabian StyleTischmann, Lea, Mathijs Drummen, Blandine Gatta-Cherifi, Anne Raben, Mikael Fogelholm, Bolette Hartmann, Jens J. Holst, Isabelle Matias, Daniela Cota, Ronald P. Mensink, and et al. 2019. "Effects of a High-Protein/Moderate-Carbohydrate Diet on Appetite, Gut Peptides, and Endocannabinoids—A Preview Study" Nutrients 11, no. 10: 2269. https://doi.org/10.3390/nu11102269
APA StyleTischmann, L., Drummen, M., Gatta-Cherifi, B., Raben, A., Fogelholm, M., Hartmann, B., Holst, J. J., Matias, I., Cota, D., Mensink, R. P., Joris, P. J., Westerterp-Plantenga, M. S., & Adam, T. C. (2019). Effects of a High-Protein/Moderate-Carbohydrate Diet on Appetite, Gut Peptides, and Endocannabinoids—A Preview Study. Nutrients, 11(10), 2269. https://doi.org/10.3390/nu11102269









