Effects of β-Fructans Fiber on Bowel Function: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Formulation
2.2. Eligibility Criteria
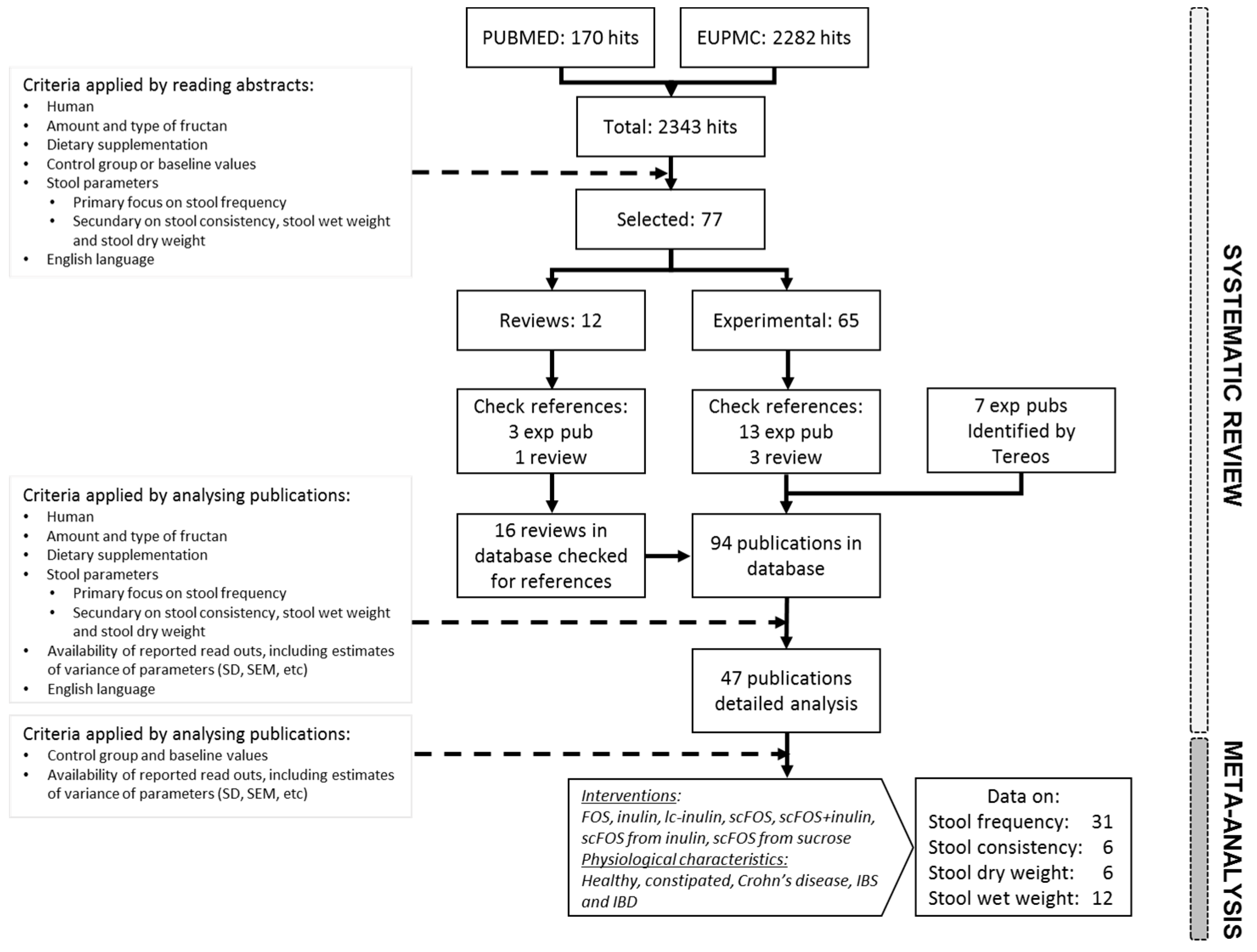
2.3. Literature Search and Study Selection
2.4. Data Extraction
2.5. Risk of Bias in Individual Studies and Across Studies
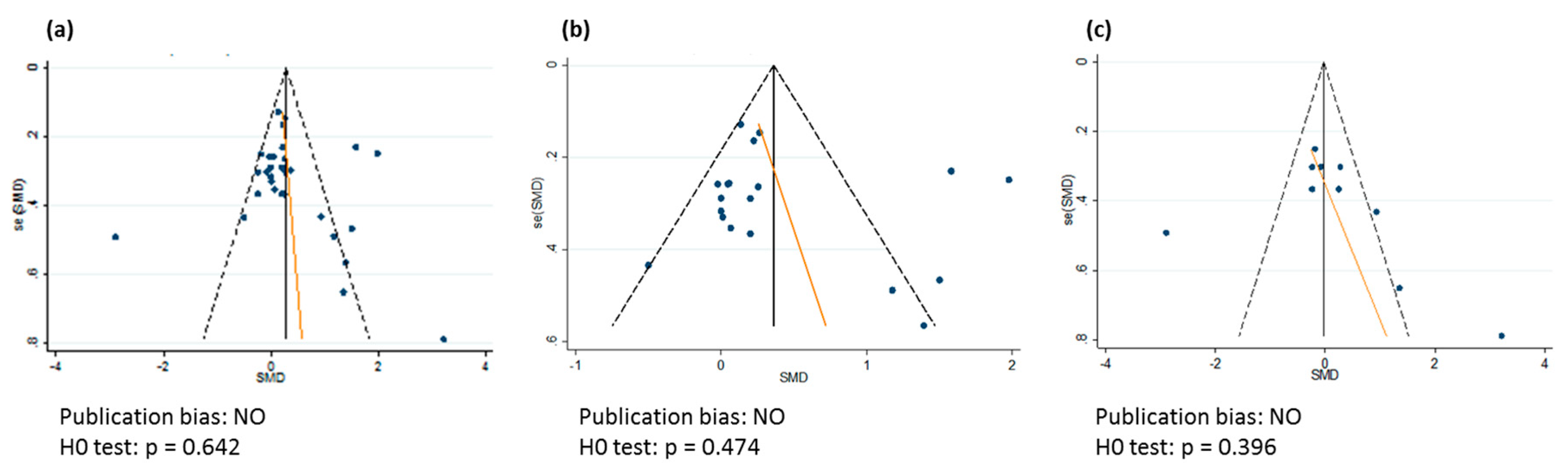
2.6. Statistical Analysis
3. Results
3.1. Study Selection
3.2. Study Characteristics
3.3. Meta-Analysis
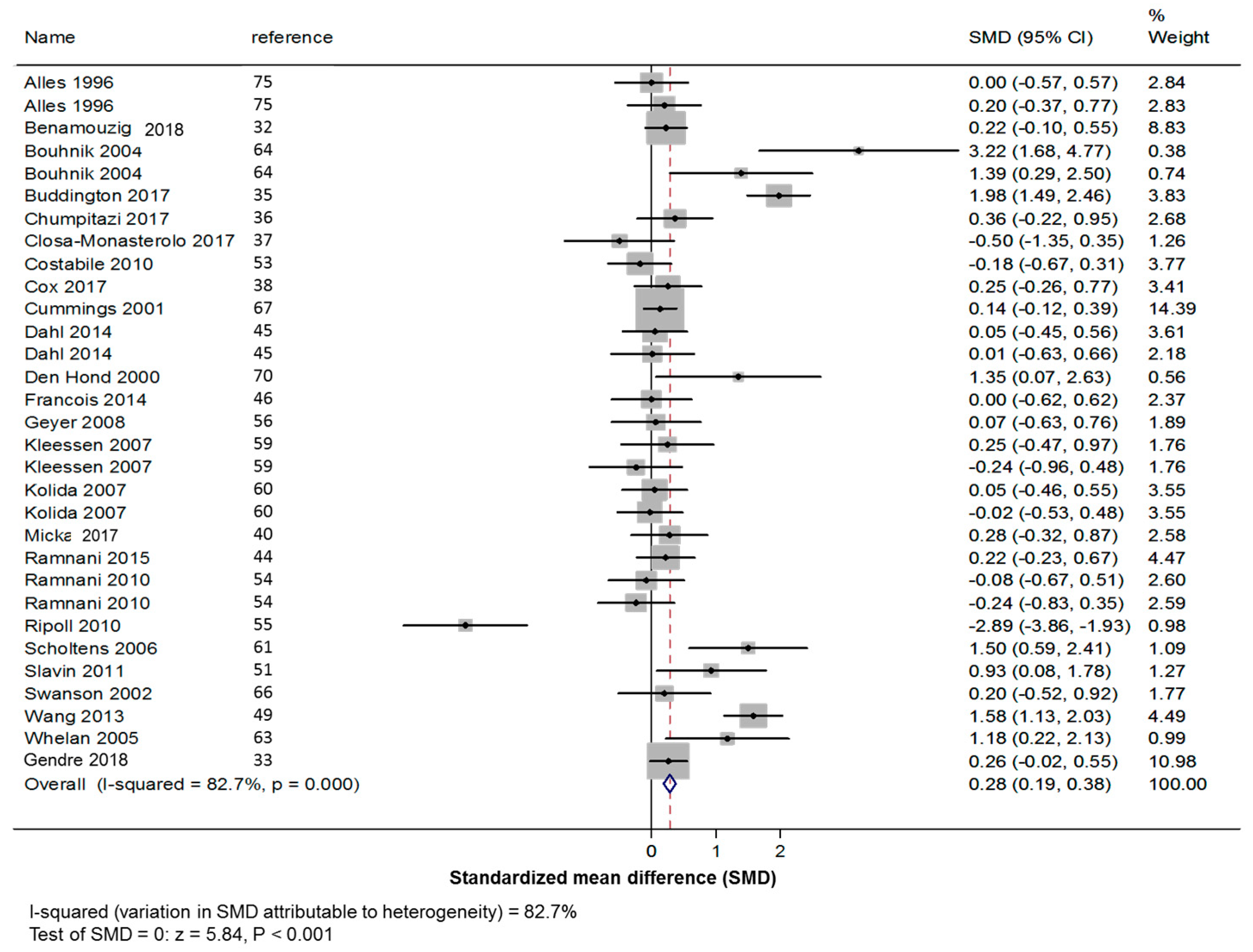
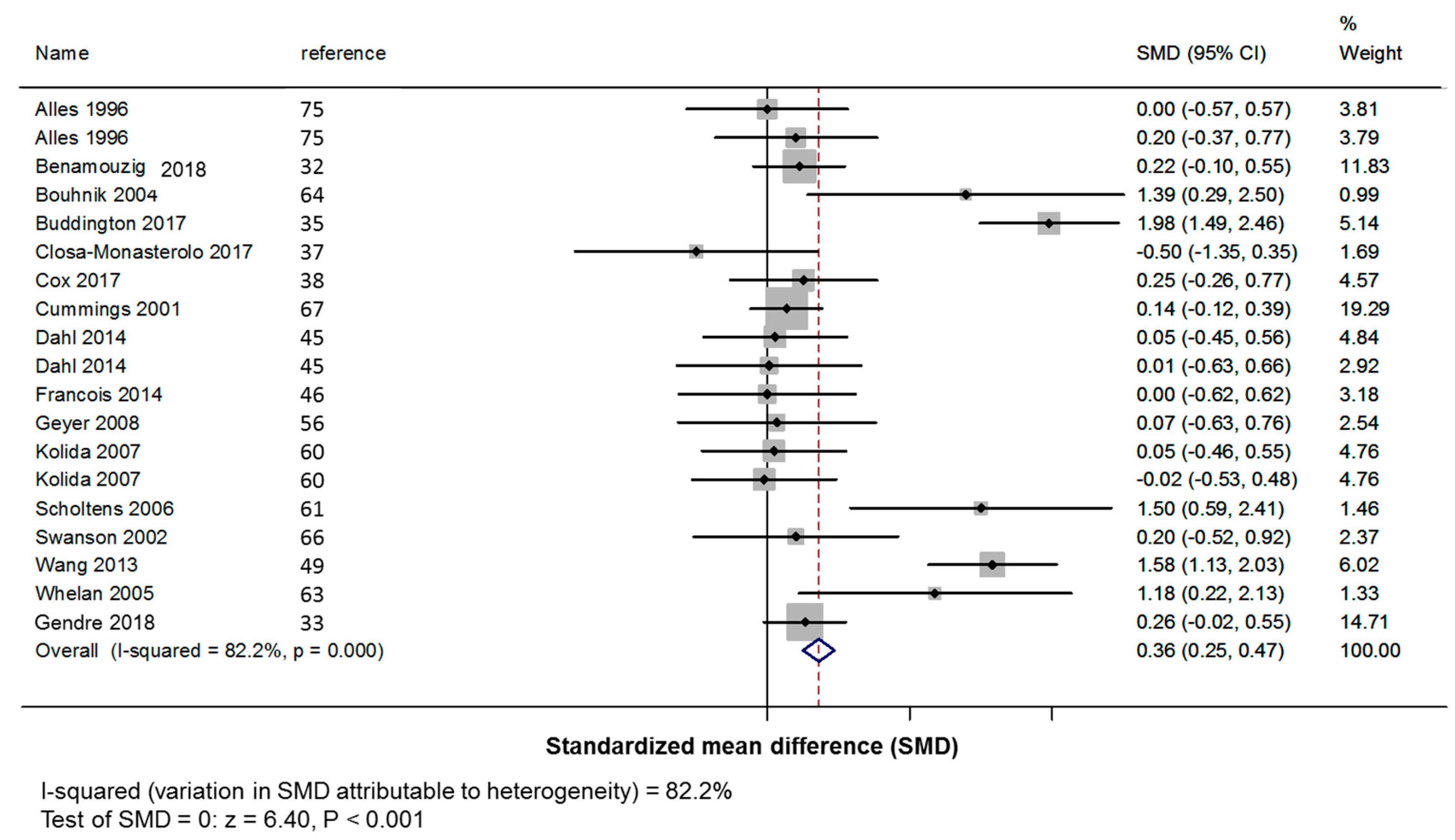
3.3.1. Primary Outcome: Stool Frequency
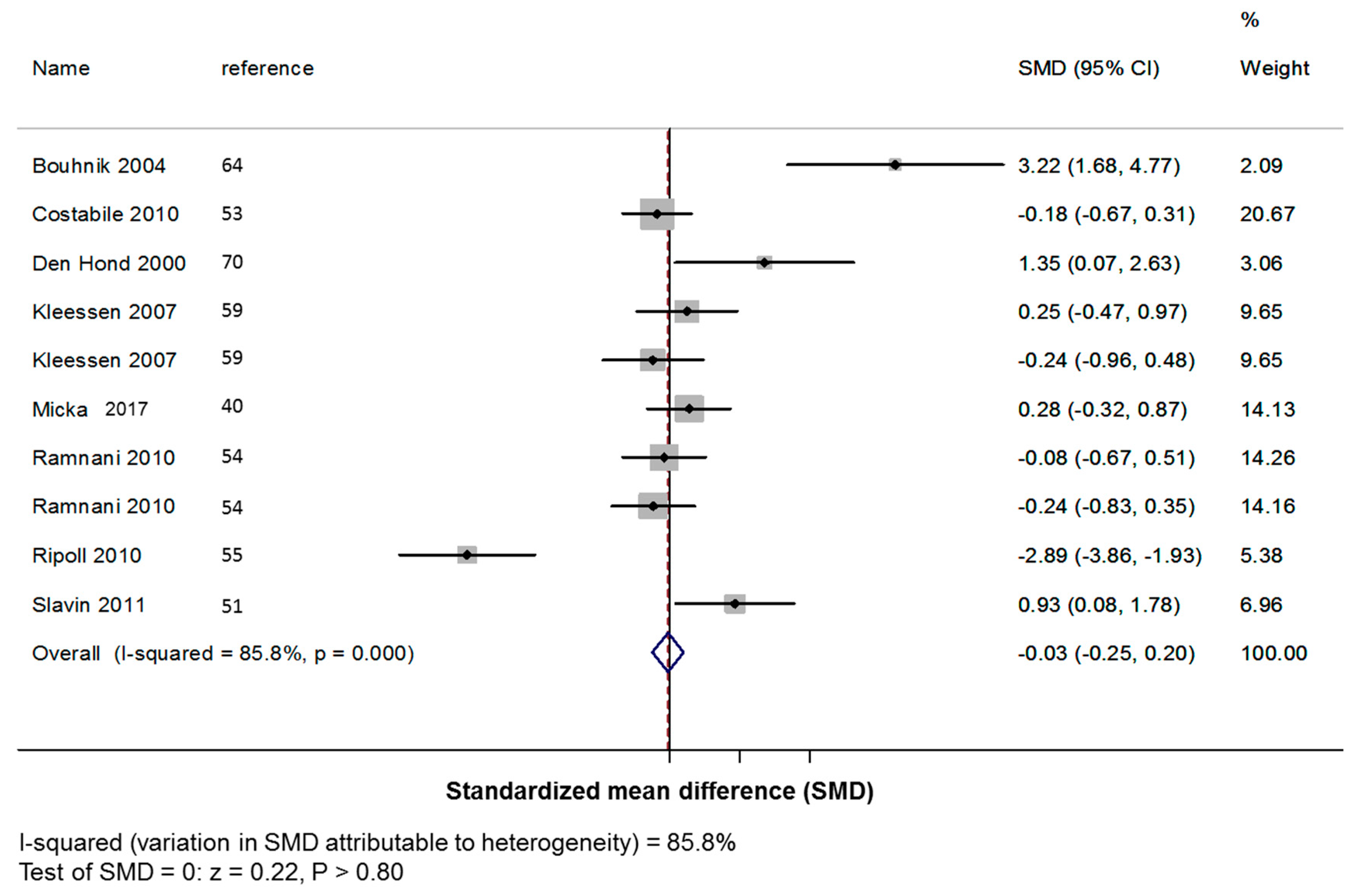
3.3.2. Secondary Outcomes: Stool Consistency, Fecal Dry and Wet Weights
3.4. Meta-Regression on Frequency of Bowel Movements
4. Discussion
4.1. Tolerance
4.2. Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Drossman, D.A. The functional gastrointestinal disorders and the Rome III process. Gastroenterology 2006, 130, 1377–1390. [Google Scholar] [CrossRef]
- Bharucha, A.E.; Pemberton, J.H.; Locke, G.R., 3rd. American Gastroenterological Association technical review on constipation. Gastroenterology 2013, 144, 218–238. [Google Scholar] [CrossRef] [PubMed]
- Peppas, G.; Alexiou, V.G.; Mourtzoukou, E.; Falagas, M. Epidemiology of constipation in Europe and Oceania: A systematic review. BMC Gastroenterol. 2008, 8, 5. [Google Scholar] [CrossRef] [PubMed]
- Higgins, P.D.; Johanson, J.F. Epidemiology of constipation in North America: A systematic review. Am. J. Gastroenterol. 2004, 99, 750–759. [Google Scholar] [CrossRef]
- Longstreth, G.F.; Thompson, W.G.; Chey, W.D.; Houghton, L.A.; Mearin, F.; Spiller, R.C. Functional bowel disorders. Gastroenterology 2006, 130, 1480–1491. [Google Scholar] [CrossRef] [PubMed]
- Thompson, W.G.; Longstreth, G.F.; Drossman, D.A.; Heaton, K.W.; Irvine, E.J.; Muller-Lissner, S.A. Functional bowel disorders and functional abdominal pain. Gut 1999, 45, 43–47. [Google Scholar] [CrossRef]
- Siproudhis, L.; Pigot, F.; Godeberge, P.; Damon, H.; Soudan, D.; Bigard, M.A. Defecation disorders: A French population survey. Dis. Colon Rectum 2006, 49, 219–227. [Google Scholar] [CrossRef]
- Tack, J.; Muller-Lissner, S.; Stanghellini, V.; Boeckxstaens, G.; Kamm, M.A.; Simren, M.; Galmiche, J.P.; Fried, M. Diagnosis and treatment of chronic constipation—A European perspective. Neurogastroenterol. Motil. 2011, 23, 697–710. [Google Scholar] [CrossRef]
- FAO/WHO. CODEX Alimentarius (CODEX) Guidelines on Nutrition Labeling CAC/GL 2–1985; FAO/WHO: Rome, Italy, 2009. [Google Scholar]
- WHO. Diet, Nutrition and the Prevention of Chronic Diseases; Technical Report 916; WHO: Geneva, Switzerland, 2003. [Google Scholar]
- EFSA Scientific Opinion on Dietary Reference Values for CHO and dietary fibres. EFSA J. 2010, 8, 1462.
- Stephen, A.M.; Champ, M.; Cloran, S.J.; Fleith, M.; van Lieshout, L.; Meiborn, H.; Burley, V.J. Dietary fibre in Europe: Current state of knowledge on definitions, sources, recommendations, intakes and relationships to health. Nutr. Res. Rev. 2017, 30, 149–190. [Google Scholar] [CrossRef]
- Tarrega, A.; Quiles, A.; Morell, P.; Fiszman, S.; Hernando, I. Importance of consumer perceptions in fiber-enriched food products. A case study with sponge cakes. Food Funct. 2017, 8, 574–583. [Google Scholar] [CrossRef] [PubMed]
- Ares, F.; Arrarte, E.; De Leon, T.; Ares, G.; Gambaro, A. Development of functional milk desserts enriched with resistant starch based on consumers’ perception. Food Sci. Technol. Int. 2012, 18, 465–475. [Google Scholar] [CrossRef] [PubMed]
- Wald, A.; Scarpignato, C.; Mueller-Lissner, S.; Kamm, M.A.; Hinkel, U.; Helfrich, I.; Schuijt, C.; Mandel, K.G. A multinational survey of prevalence and patterns of laxative use among adults with self-defined constipation. Aliment. Pharmacol. Ther 2008, 28, 917–930. [Google Scholar] [CrossRef] [PubMed]
- De Vries, J.; Miller, P.E.; Verbeke, K. Effects of cereal fiber on bowel function: A systematic review of intervention trials. World J. Gastroenterol. 2015, 21, 8952–8963. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Wang, H.-P.; Zhou, L.; Xu, C.-F. Effect of dietary fiber on constipation: A meta-analysis. World J. Gastroenterol. 2012, 18, 7378–7383. [Google Scholar] [CrossRef] [PubMed]
- FDA Review of the Scientific Evidence on the Physiological Effects of Certain Non-Digestible Carbohydrates. 2018. Available online: https://www.fda.gov/downloads/Food/LabelingNutrition/UCM610139.pdf (accessed on 14 June 2018).
- Roberfroid, M.B. Inulin-Type Fructans, Functional Food Ingredients. CRC Series in Modern Nutrition; Wolinsky, I., Hickson, J.F.J., Eds.; CRC Press: Boca Raton, FL, USA, 2005; p. 359. [Google Scholar]
- Scott, K.P.; Martin, J.C.; Duncan, S.H.; Flint, H.J. Prebiotic stimulation of human colonic butyrate-producing bacteria and bifidobacteria, in vitro. FEMS Microbiol. Ecol. 2014, 87, 30–40. [Google Scholar] [CrossRef] [PubMed]
- Valdes-Varela, L.; Ruas-Madieto, P.; Gueimonde, M. In vitro fermentation of different fructo-oligosaccharides by Bifidobacterium strains for the selection of synbiotic combinations. Int. J. Food Microbiol. 2017, 242, 19–23. [Google Scholar] [CrossRef]
- Roberfroid, M.B.; Gibson, G.R.; Hoyles, L.; McCartney, A.L.; Rastall, R.A.; Rowland, I.R.; Wolvers, D.; Watzl, B.; Szajewska, H.; Stahl, B.; et al. Prebiotic effects: Metabolic and health benefits. Br. J. Nutr. 2010, 104 (Suppl. 2), S1–S63. [Google Scholar] [CrossRef]
- EFSA Panel on Dietetic Products, Nutrition and Allergies (NDA). Scientific Opinion on the substantiation of a health claim related to “native chicory inulin” and maintenance of normal defecation by increasing stool frequency pursuant to Article 13(5) of Regulation (EC) No 1924/2006. EFSA J. 2015, 13, 3951. [Google Scholar] [CrossRef]
- Shimizu, T. Health claims on functional foods: The Japanese regulations and an international comparison. Nutr. Res. Rev. 2003, 16, 241–252. [Google Scholar] [CrossRef]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gotzsche, P.C.; Ioannidis, J.P.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: Explanation and elaboration. PLoS Med. 2009, 6, e1000100. [Google Scholar] [CrossRef] [PubMed]
- Lewis, S.; Heaton, K. Stool form scale as a useful guide to intestinal transit time. Scand. J. Gastroenterol. 1997, 32, 920–924. [Google Scholar] [CrossRef] [PubMed]
- Begg, C.B.; Mazumdar, M. Operating characteristics of a rank correlation test for publication bias. Biometrics 1994, 50, 1088–1101. [Google Scholar] [CrossRef] [PubMed]
- Egger, M.; Davey Smith, G.; Schneider, M.; Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997, 315, 629–634. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. BMJ 2003, 327, 557–560. [Google Scholar] [CrossRef] [PubMed]
- Thompson, S.G.; Higgins, J.P. How should meta-regression analyses be undertaken and interpreted? Stat. Med. 2002, 21, 1559–1573. [Google Scholar] [CrossRef]
- Higgins, J.P.; Thompson, S.G. Controlling the risk of spurious findings from meta-regression. Stat. Med. 2004, 23, 1663–1682. [Google Scholar] [CrossRef]
- Benamouzig, R.; Ait-Omar-Bourkeb, A.; Curis, E.; Nicolis, I.; Le Bourgot, C.; Glatt, D.; Louis, P.; Bornet, F.; Wagner, A.; Respondek, F. Dietary supplementation with scFOS increases frequency of bowel movements in subjects with functional constipation: A randomized, double-blind, placebo controlled study. Am. J. Clin. Nutr. 2018. Submitted. [Google Scholar]
- Gendre, D. Biofortis, Nantes, France. Unpublished work. 2018. [Google Scholar]
- Azpiroz, F.; Molne, L.; Mendez, S.; Nieto, A.; Manichanh, C.; Mego, M.; Accarino, A.; Santos, J.; Sailer, M.; Theis, S.; et al. Effect of Chicory-derived Inulin on Abdominal Sensations and Bowel Motor Function. J. Clin. Gastroenterol. 2017, 51, 619–625. [Google Scholar] [CrossRef]
- Buddington, R.K.; Kapadia, C.; Neumer, F.; Theis, S. Oligofructose Provides Laxation for Irregularity Associated with Low Fiber Intake. Nutrients 2017, 9, 1372. [Google Scholar] [CrossRef]
- Chumpitazi, B.P.; McMeans, A.R.; Vaughan, A.; Ali, A.; Orlando, S.; Elsaadi, A.; Shulman, R.J. Fructans Exacerbate Symptoms in a Subset of Children with Irritable Bowel Syndrome. Clin. Gastroenterol. Hepatol. 2018, 16, 219–225. [Google Scholar] [CrossRef] [PubMed]
- Closa-Monasterolo, R.; Ferre, N.; Castillejo-DeVillasante, G.; Luque, V.; Gispert-Llaurado, M.; Zaragoza-Jordana, M.; Theis, S.; Escribano, J. The use of inulin-type fructans improves stool consistency in constipated children. A randomised clinical trial: Pilot study. Int. J. Food Sci. Nutr. 2017, 68, 587–594. [Google Scholar] [CrossRef] [PubMed]
- Cox, S.R.; Prince, A.C.; Myers, C.E.; Irving, P.M.; Lindsay, J.O.; Lomer, M.C.; Whelan, K. Fermentable Carbohydrates [FODMAPs] Exacerbate Functional Gastrointestinal Symptoms in Patients with Inflammatory Bowel Disease: A Randomised, Double-blind, Placebo-controlled, Cross-over, Re-challenge Trial. J. Crohns Colitis 2017, 11, 1420–1429. [Google Scholar] [CrossRef]
- Jinno, S.; Toshimitsu, T.; Nakamura, Y.; Kubota, T.; Igoshi, Y.; Ozawa, N.; Suzuki, S.; Nakano, T.; Morita, Y.; Arima, T.; et al. Maternal Prebiotic Ingestion Increased the Number of Fecal Bifidobacteria in Pregnant Women but Not in Their Neonates Aged One Month. Nutrients 2017, 9, 196. [Google Scholar] [CrossRef] [PubMed]
- Micka, A.; Siepelmeyer, A.; Holz, A.; Theis, S.; Schon, C. Effect of consumption of chicory inulin on bowel function in healthy subjects with constipation: A randomized, double-blind, placebo-controlled trial. Int. J. Food Sci. Nutr. 2017, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Clarke, S.T.; Green-Johnson, J.M.; Brooks, S.P.; Ramdath, D.D.; Bercik, P.; Avila, C.; Inglis, G.D.; Green, J.; Yanke, L.J.; Selinger, L.B.; et al. beta2-1 Fructan supplementation alters host immune responses in a manner consistent with increased exposure to microbial components: Results from a double-blinded, randomised, cross-over study in healthy adults. Br. J. Nutr. 2016, 115, 1748–1759. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Peris, P.; Velasco, C.; Hernandez, M.; Lozano, M.A.; Paron, L.; de la Cuerda, C.; Breton, I.; Camblor, M.; Guarner, F. Effect of inulin and fructo-oligosaccharide on the prevention of acute radiation enteritis in patients with gynecological cancer and impact on quality-of-life: A randomized, double-blind, placebo-controlled trial. Eur. J. Clin. Nutr. 2016, 70, 170–174. [Google Scholar] [CrossRef]
- Meksawan, K.; Chaotrakul, C.; Leeaphorn, N.; Gonlchanvit, S.; Eiam-Ong, S.; Kanjanabuch, T. Effects of fructo-oligosaccharide supplementation on constipation in elderly continuous ambulatory peritoneal dialysis patients. Perit. Dial. Int. 2016, 36, 60–66. [Google Scholar] [CrossRef]
- Ramnani, P.; Costabile, A.; Bustillo, A.G.; Gibson, G.R. A randomised, double- blind, cross-over study investigating the prebiotic effect of agave fructans in healthy human subjects. J. Nutr. Sci. 2015, 4, e10. [Google Scholar] [CrossRef]
- Dahl, W.J.; Wright, A.R.; Specht, G.J.; Christman, M.; Mathews, A.; Meyer, D.; Boileau, T.; Willis, H.J.; Langkamp-Henken, B. Consuming foods with added oligofructose improves stool frequency: A randomised trial in healthy young adults. J. Nutr. Sci. 2014, 3, e7. [Google Scholar] [CrossRef]
- Francois, I.E.; Lescroart, O.; Veraverbeke, W.S.; Windey, K.; Verbeke, K.; Broekaert, W.F. Tolerance and the effect of high doses of wheat bran extract, containing arabinoxylan-oligosaccharides, and oligofructose on faecal output: A double-blind, randomised, placebo-controlled, cross-over trial. J. Nutr. Sci. 2014, 3, e49. [Google Scholar] [CrossRef]
- Majid, H.A.; Cole, J.; Emery, P.W.; Whelan, K. Additional oligofructose/inulin does not increase faecal bifidobacteria in critically ill patients receiving enteral nutrition: A randomised controlled trial. Clin. Nutr. 2014, 33, 966–972. [Google Scholar] [CrossRef] [PubMed]
- Respondek, F.; Hilpipre, C.; Chauveau, P.; Cazaubiel, M.; Gendre, D.; Maudet, C.; Wagner, A. Digestive tolerance and postprandial glycaemic and insulinaemic responses after consumption of dairy desserts containing maltitol and fructo-oligosaccharides in adults. Eur. J. Clin. Nutr. 2014, 68, 575–580. [Google Scholar] [CrossRef]
- Wang, Y.; Zeng, T.; Wang, S.; Li, F.; Guo, X.-Y.; Jin, J.; Yu, H.-X. Laxative effect of fructooligosaccharides in mice and humans. Curr. Top. Nutr. Res. 2013, 11, 1–9. [Google Scholar]
- Benjamin, J.L.; Hedin, C.R.; Koutsoumpas, A.; Ng, S.C.; McCarthy, N.E.; Hart, A.L.; Kamm, M.A.; Sanderson, J.D.; Knight, S.C.; Forbes, A.; et al. Randomised, double-blind, placebo-controlled trial of fructo-oligosaccharides in active Crohn’s disease. Gut 2011, 60, 923–929. [Google Scholar] [CrossRef] [PubMed]
- Slavin, J.; Feirtag, J. Chicory inulin does not increase stool weight or speed up intestinal transit time in healthy male subjects. Food Funct. 2011, 2, 72–77. [Google Scholar] [CrossRef] [PubMed]
- Yen, C.H.; Kuo, Y.W.; Tseng, Y.H.; Lee, M.C.; Chen, H.L. Beneficial effects of fructo-oligosaccharides supplementation on faecal bidifidobacteria and index of peroxidation status in constipated nursing-home residents—A placebo-controlled, diet-controlled trial. Nutrition 2011, 27, 323–328. [Google Scholar] [CrossRef] [PubMed]
- Costabile, A.; Kolida, S.; Klinder, A.; Gietl, E.; Bauerlein, M.; Frohberg, C.; Landschutze, V.; Gibson, G.R. A double-blind, placebo-controlled, cross-over study to establish the bifidogenic effect of a very-long-chain inulin extracted from globe artichoke (Cynara scolymus) in healthy human subjects. Br. J. Nutr. 2010, 104, 1007–1017. [Google Scholar] [CrossRef]
- Ramnani, P.; Gaudier, E.; Bingham, M.; van Bruggen, P.; Tuohy, K.M.; Gibson, G.R. Prebiotic effect of fruit and vegetable shots containing Jerusalem artichoke inulin: A human intervention study. Br. J. Nutr. 2010, 104, 233–240. [Google Scholar] [CrossRef]
- Ripoll, C.; Flourie, B.; Megnien, S.; Hermand, O.; Janssens, M. Gastrointestinal tolerance to an inulin-rich soluble roasted chicory extract after consumption in healthy subjects. Nutrition 2010, 26, 799–803. [Google Scholar] [CrossRef]
- Geyer, M.; Manrique, I.; Degen, L.; Beglinger, C. Effects of yacon (smallanthus sonchifolius) on colonic transit time in healthy vonlunterres. Digestion 2008, 78, 30–33. [Google Scholar] [CrossRef] [PubMed]
- Bouhnik, Y.; Achour, L.; Paineau, D.; Riottot, M.; Attar, A.; Bornet, F. Four-week short chain fructo-oligosaccharides ingestion leads to an increase in faecal bifidobacteria and cholesterol excretion elderly volunteers. Nutr. J. 2007, 6, 42. [Google Scholar] [CrossRef] [PubMed]
- De Preter, V.; Vanhoutte, T.; Huys, G.; Swings, J.; De Vuyst, L.; Rutgeerts, P.; Verbeke, K. Effects of Lactobacillus casei Shirota, Bifidobacterium breve, and oligofructose-enriched inulin on colonic nitrogen-protein metabolism in healthy humans. Am. J. Physiol. Gastrointest. Liver Physiol. 2007, 292, G358–G368. [Google Scholar] [CrossRef]
- Kleessen, B.; Schwarz, S.; Boehm, A.; Fuhrmann, H.; Richter, A.; Henle, T.; Krueger, M. Jerusalem artichokes and chicory inulin in bakery products affect faecal microbiota of healthy volunteers. Br. J. Nutr. 2007, 98, 540–549. [Google Scholar] [CrossRef] [PubMed]
- Kolida, S.; Meyer, D.; Gibson, G.R. A double-blind placebo-controlled study to establish the bifidogenic dose of inulin in healthy humans. Eur. J. Clin. Nutr. 2007, 61, 1189–1195. [Google Scholar] [CrossRef] [PubMed]
- Scholtens, P.; Alles, M.; Willemsen, L.; van den Braak, C.; Bindels, J.; Boehm, G.; Govers, M. Dietary fructo-oligosaccharides in healthy adults do not negatively affect faecal cytotoxicity: A randomised, double-blind, placebo-controlled crossover trial. Br. J. Nutr. 2006, 95, 1143–1149. [Google Scholar] [CrossRef] [PubMed]
- Dahl, W.J.; Whiting, S.J.; Isaac, T.M.; Weeks, S.J.; Arnold, C.J. Effects of thickened beverages fortified with inulin on beverage acceptance, gastrointestinal function, and bone resorption in institutionalized adults. Nutrition 2005, 21, 308–311. [Google Scholar] [CrossRef] [PubMed]
- Whelan, K.; Judd, P.A.; Preedy, V.R.; Simmering, R.; Jann, A.; Taylor, M. Fructooligosaccharides and fiber partially prevent the alterations in fecal microbiota and short-chain fatty acid concentrations caused by standard enteral formula in healthy humans. J. Nutr. 2005, 135, 1896–1902. [Google Scholar] [CrossRef]
- Bouhnik, Y.; Raskine, L.; Simoneau, G.; Vicaut, E.; Neut, C.; Flourie, B.; Brouns, F.; Bornet, F. The capacity of nondigestible carbohydrates to stimulate fecal bifidobacteria in healthy humans: A double-blind, randomized, placebo-controlled, parallel-group, dose-response relation study. Am. J. Clin. Nutr. 2004, 80, 1658–1664. [Google Scholar] [CrossRef]
- Grasten, S.; Liukkonen, K.H.; Chrevatidis, A.; El-Nezami, H.; Poutanen, K.; Mykkanen, H. Effects of wheat pentosan and inulin on the metabolic activity of fecal microbiota and on bowel function in healthy humans. Nut. Res. 2003, 23, 1503–1514. [Google Scholar] [CrossRef]
- Swanson, K.S.; Grieshop, C.M.; Flickinger, E.A.; Bauer, L.L.; Wolf, B.W.; Chow, J.; Garleb, K.A.; Williams, J.A.; Fahey, G.C. Fructooligosaccharides and Lactobacillus acidophilus Modify Bowel Function and Protein Catabolites Excreted by Healthy Humans. J. Nutr. 2002, 132, 3042–3050. [Google Scholar] [CrossRef] [PubMed]
- Cummings, J.; Christie, S.; Cole, T. A study of fructo oligosaccharides in the prevention of travellers’ diarrhoea. Aliment. Pharmacol. Ther. 2001, 15, 1139–1145. [Google Scholar] [CrossRef]
- Tahiri, M.; Tressol, J.C.; Arnaud, J.; Bornet, F.; Bouteloup-Demange, C.; Feillet-Coudray, C.; Ducros, V.; Pepin, D.; Brouns, F.; Rayssiguier, A.M.; et al. Five-week intake of short-chain fructo-oligosaccharides increases intestinal absorption and status of magnesium in postmenopausal women. J. Bone Min. Res. 2001, 11, 2152–2160. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Lu, Y.; Lin, J.; Ko, L. Effects of fructooligosaccharide on bowel function and indicators of nutritional status in constipated elderly men. Nutr. Res. 2000, 20, 1725–1733. [Google Scholar] [CrossRef]
- Den Hond, E.; Geypens, B.; Ghoos, Y. Effect of high performance chicory inulin on constipation. Nutr. Res. 2000, 20, 731–736. [Google Scholar] [CrossRef]
- Brighenti, F.; Casiraghi, M.; Canzi, E.; Ferrari, A. Effect of consumption of a ready-to-eat breakfast cereal containing inulin on the intestinal milieu and blood lipids in healthy male volunteers. Eur. J. Clin. Nutr. 1999, 53, 726–733. [Google Scholar] [CrossRef]
- Tominaga, S.; Hirayama, M.; Adachi, T.; Tokunaga, T.; Iino, H. Effects of ingested fructooligosaccharides on stool frequency in healthy female volunteers: A placebo-controlled study. Biosci. Microflora 1999, 18, 49–53. [Google Scholar] [CrossRef]
- Van Dokkum, W.; Wezendonk, B.; Srikumar, T.; Van den Heuvel, E. Effect of nondigestible oligosaccharides on large-bowel functions, blood lipid concentrations and glucose absorption in young healthy male subjects. Eur. J. Clin. Nutr. 1999, 53, 1–7. [Google Scholar] [CrossRef]
- Sobotka, L.; Bratova, M.; Slemrova, M.; Manak, J.; Vizd’a, J.; Zadak, Z. Inulin as the soluble fiber in liquid enteral nutrition. Nutrition 1997, 13, 21–25. [Google Scholar] [CrossRef]
- Alles, M.S.; Hautvast, J.G.; Nagengast, F.M.; Hartemink, R.; van Laere, K.M.; Jansen, J.B. Fate of fructo-oligosaccharides in the Human intestine. Br. J. Nutr. 1996, 76, 211–221. [Google Scholar] [CrossRef]
- Bouhnik, Y.; Flourie, B.; Riottot, M.; Bisetti, N.; Gailing, M.F.; Guibert, A.; Bornet, F.; Rambaud, J.C. Effects of fructo-oligosaccharides ingestion on fecal bifidobacteria and selected metabolic indexes of colon carcinogenesis in healthy humans. Nutr. Cancer 1996, 26, 21–29. [Google Scholar] [CrossRef] [PubMed]
- Molis, C.; Flourie, B.; Ouarne, F.; Gailing, M.F.; Lartigue, S.; Guibert, A.; Bornet, F.; Galmiche, J.P. Digestion, excretion, and energy value of fructooligosaccharides in healthy humans. Am. J. Clin. Nutr. 1996, 64, 324–328. [Google Scholar] [CrossRef] [PubMed]
- Gibson, G.; Beatty, E.; Xin Wang, J.; Cummings, J. Selective stimulation of bifidobacteria in the human colon by oligofructose and inulin. Gastroenterology 1995, 108, 975–982. [Google Scholar] [CrossRef]
- Cummings, J.H.; Macfarlane, G.T.; Englyst, H.N. Prebiotic digestion and fermentation. Am. J. Clin. Nutr. 2001, 73, 415S–420S. [Google Scholar] [CrossRef] [PubMed]
- Briet, F.; Achour, L.; Flourie, B.; Beaugerie, L.; Pellier, P.; Franchisseur, C.; Bornet, F.; Rambaud, J.C. Symptomatic response to varying levels of fructo-oligosaccharides consumed occasionally or regularly. Eur. J. Clin. Nutr. 1995, 49, 501–507. [Google Scholar]
- Carabin, I.G.; Flamm, W.G. Evaluation of safety of inulin and oligofructose as dietary fiber. Regul. Toxicol. Pharmacol. RTP 1999, 30, 268–282. [Google Scholar] [CrossRef] [PubMed]
- Bonnema, A.L.; Kolberg, L.W.; Thomas, W.; Slavin, J.L. Gastrointestinal tolerance of chicory inulin products. J. Am. Diet. Assoc. 2010, 110, 865–868. [Google Scholar] [CrossRef]
- Azpiroz, F.; Dubray, C.; Bernalier-Donadille, A.; Cardot, J.-M.; Accarino, A.; Serra, J.; Wagner, A.; Respondek, F.; Dapoigny, M. Effects of scFOS on the composition of fecal microbiota and anxiety in patients with irritable bowel syndrome: A randomized, double blind, placebo controlled study. Neurogastroenterol. Motil. 2017, 29, e12911. [Google Scholar] [CrossRef]
- Paineau, D.; Payen, F.; Panserieu, S.; Coulombier, G.; Sobaszek, A.; Lartigau, I.; Brabet, M.; Galmiche, J.P.; Tripodi, D.; Sacher-Huvelin, S.; et al. The effects of regular consumption of short-chain fructo-oligosaccharides on digestive comfort of subjects with minor functional bowel disorders. Br. J. Nutr. 2008, 99, 311–318. [Google Scholar] [CrossRef]

| References | Experimental Design 1 | N | Participants | Intervention 2 | Comparator | Dose (g/Day) | Duration (Day) | Stool Frequency | Stool Consistency | Fecal Dry Weight | Fecal Wet Weight | Included in Meta-Analysis |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Benamouzig, 2018 [32] | Parallel | 150 | Constipated | DP < 10 scFOS | Maltodextrin | 5 | 42 | Yes | Yes | No | No | Yes |
| Gendre, 2018 [33] | Parallel | 187 | Constipated | DP < 10 scFOS | Maltodextrin | 5 | 42 | Yes | Yes | Yes | Yes | Yes |
| Azpiroz, 2017 [34] | Parallel | 36 | IBS a | DP ≥ 10 | Maltodextrin | 8 | 28 | No | No | No | No | No |
| Buddington, 2017 [35] | Parallel | 97 | Constipated | DP < 10 OF | Maltodextrin | 15 | 84 | Yes | No | No | No | Yes |
| Chumpitazi, 2018 [36] | Cross-over | 23 | IBS a | DP ≥ 10 | Maltodextrin | 6.2 | 3 | Yes | Yes | No | No | Yes |
| Closa-Monasterolo, 2017 [37] | Parallel | 22 | Constipated | DP < 10 OF | Maltodextrin | 4 | 42 | Yes | Yes | No | No | Yes |
| Cox, 2017 [38] | Cross-over | 32 | IBD b | DP < 10 OF | Glucose | 12 | 3 | Yes | Yes | No | No | Yes |
| Jinno, 2017 [39] | Parallel | 64 | Pregnant | DP < 10 scFOS | Sucrose | 8 | 105 | Yes | Yes | No | No | No |
| Micka, 2017 [40] | Cross-over | 44 | Constipated | DP ≥ 10 | Maltodextrin | 12 | 28 | Yes ** | Yes | No | No | Yes |
| Clarke, 2016 [41] | Cross-over | 30 | Healthy | Mixture | Maltodextrin | 15 | 28 | No | No | No | No | No d |
| Garcia-Perris, 2016 [42] | Parallel | 38 | Gynecological cancer | Mixture | Maltodextrin | 12 | 28 | Yes | Yes | No | No | No |
| Meksawan, 2016 [43] | Cross-over | 9 | Peritoneal dialysis | DP < 10 OF | Placebo | 20 | 30 | Yes | Yes | No | No | No |
| Ramnani, 2015 [44] | Cross-over | 38 | Healthy | No detail | Maltodextrin | 4.7 | 21 | Yes | Yes* | No | No | Yes |
| Dahl, 2014 [45] | Parallel | 98 | Healthy | DP < 10 OF | Placebo | 15.6 & 15.3 | 56 | Yes | No | No | No | Yes |
| François, 2014 [46] | Cross-over NB | 20 | Healthy | DP < 10 OF | Placebo | 30 | 14 | Yes | Yes | No | Yes | Yes |
| Majid, 2014 [47] | Parallel | 22 | Healthy with enteral nutrition | DP ≥ 10 | Maltodextrin | 7 | 7 | Yes | Yes | No | No | No |
| Respondek, 2014 [48] | Cross-over | 36 | Healthy | DP < 10 scFOS | Dextrose | 11 | 1 | Yes | Yes | No | No | No |
| Wang, 2013 [49] | Parallel | 100 | Constipated | DP < 10 OF | Placebo | 1.26 | 10 | Yes | No | No | No | Yes |
| Benjamin, 2011 [50] | Parallel | 103 | Crohn’s disease c | DP < 10 OF | Maltodextrin | 15 | 28 | No | No | No | No | No e |
| Slavin, 2011 [51] | Cross-over | 12 | Healthy | DP ≥ 10 | Placebo | 20 | 21 | Yes | Yes | No | Yes | Yes |
| Yen, 2010 [52] | Parallel | 10 | Constipated | DP < 10 scFOS | Placebo | 10 | 28 | Yes | No | Yes | No | Yes |
| Costabile, 2010 [53] | Cross-over | 32 | Healthy | DP ≥ 10 | Maltodextrin | 10 | 21 | Yes | Yes | No | No | Yes |
| Ramnani, 2010 [54] | Parallel | 66 | Healthy | DP ≥ 10 | Placebo | 5 | 21 | Yes | Yes | No | No | Yes |
| Ripoll, 2010 [55] | Parallel | 35 | Healthy | DP ≥ 10 | Sucrose | 5 | 28 | Yes | Yes | No | No | Yes |
| Geyer, 2008 [56] | Cross-over | 16 | Healthy | DP < 10 OF | Placebo | 6.4 | 14 | Yes | Yes * | No | No | Yes |
| Bouhnik, 2007 [57] | Single arm | 12 | Healthy | DP < 10 scFOS | None | 8 | 28 | No | No | Yes | Yes | No |
| De Preter, 2007 [58] | Cross-over | 20 | Healthy | DP ≥ 10 | Maltodextrin | 20 | 28 | No | No | Yes * | Yes * | Yes |
| Kleessen, 2007 [59] | Parallel | 45 | Healthy | DP ≥ 10 | Placebo | 15 | 21 | Yes ** | Yes ** | No | No | Yes |
| Kolida, 2007 [60] | Sequential | 90 | Healthy | DP < 10 OF | Maltodextrin | 5 & 8 | 14 | Yes | No | No | No | No |
| Scholtens, 2006 [61] | Cross-over | 12 | Healthy | DP < 10 OF | Maltodextrin | 30 | 14 | Yes | Yes | Yes | Yes | Yes |
| Dahl, 2005 [62] | Cross-over | 15 | Dysphagia | DP ≥ 10 | Placebo | 12.6 | 21 | Yes | No | No | No | No |
| Whelan, 2005 [63] | Cross-over | 10 | Healthy | DP < 10 OF | Placebo | 9.5 | 14 | Yes | No | No | Yes | Yes |
| Bouhnik, 2004 [64] | Parallel | 24 | Healthy | DP < 10 scFOS DP ≥ 10 | 50:50 Malto/sucrose | 10 | 7 | Yes | Yes | No | No | Yes |
| Grasten, 2003 [65] | Parallel | 14 | Healthy | DP ≥ 10 | Arabinoxylan-OS | 13.3 | 21 | Yes * | Yes * | No | No | No |
| Swanson, 2002 [66] | Parallel | 68 | Healthy | DP < 10 scFOS | Sucrose | 3 | 28 | Yes | Yes * | Yes * | No | Yes |
| Cummings, 2001 [67] | Parallel | 244 | Healthy | DP < 10 OF | Maltodextrin | 10 | 28 | Yes | Yes | No | No | Yes |
| Tahiri, 2001 [68] | Cross-over | 11 | Healthy | DP < 10 OF | Sucrose | 10 | 35 | No | No | Yes | Yes | Yes |
| Chen, 2000 [69] | Sequential | 5 | Constipated | DP < 10 scFOS | None | 10 | 30 | Yes | No | Yes | Yes | No |
| Den Hond, 2000 [70] | Cross-over | 6 | Constipated | DP ≥ 10 | Sucrose | 15 | 14 | Yes | Yes * | Yes | Yes | Yes |
| Brighenti, 1999 [71] | Sequential | 12 | Healthy | DP < 10 OF | None | 9 | 28 | Yes | No | Yes | Yes | No |
| Tominaga, 1999 [72] | Parallel NB | 34 | Healthy | DP < 10 scFOS | Placebo | 3 | 14 | Yes | Yes | No | No | No f |
| Van Dokkum, 1999 [73] | Cross-over | 12 | Healthy | DP < 10 OF & DP ≥ 10 | Placebo | 15 | 21 | No | No | Yes | Yes | Yes |
| Sobotka, 1997 [74] | Sequential | 9 | Other | DP ≥ 10 | None | 30 | 7 | Yes | Yes | No | No | No |
| Alles, 1996 [75] | Cross-over NB | 24 | Healthy | DP < 10 OF | Glucose | 5 & 15 | 7 | Yes | Yes | Yes * | Yes | Yes |
| Bouhnik, 1996 [76] | Parallel | 20 | Healthy | DP < 10 scFOS | Saccharose | 12.5 | 12 | No | No | No | Yes | Yes |
| Molis, 1996 [77] | Cross-over NB | 6 | Healthy | DP < 10 scFOS | 30:70 Malto/sucrose | 20.1 | 11 | No | No | No | Yes | No |
| Gibson, 1995 [78] | Sequential | 12 | Healthy | DP < 10 scFOS & DP < 10 OF | None | 15 | 15 | Yes | No | Yes | Yes | No |
| All β-Fructans | Long-Chain β-Fructans (DP ≥ 10) | Short-Chain β-Fructans (DP < 10) | |
|---|---|---|---|
| Stool consistency (Bristol stool scale score) | |||
| N observations | 6 | 1 | 5 |
| Overall standardized mean difference (95% IC) | 0.23 (0.05, 0.40) | NA | 0.26 (0.08, 0.45) |
| p-value | 0.01 | NA | 0.006 |
| Stool dry weight (g/day) | |||
| N observations | 6 | 2 | 4 |
| Overall standardized mean difference (95% IC) | 0.18 (−0.05, 0.42) | 0.31 (−0.35, 0.98) | 0.17 (−0.08, 0.41) |
| p-value | 0.12 | 0.35 | 0.19 |
| Stool wet weight (g/day) | |||
| N observations | 12 | 3 | 9 |
| Overall standardized mean difference (95% IC) | 0.24 (0.06, 0.43) | 0.29 (−0.22, 0.80) | 0.24 (0.04, 0.43) |
| p-value | 0.008 | 0.262 | 0.016 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
De Vries, J.; Le Bourgot, C.; Calame, W.; Respondek, F. Effects of β-Fructans Fiber on Bowel Function: A Systematic Review and Meta-Analysis. Nutrients 2019, 11, 91. https://doi.org/10.3390/nu11010091
De Vries J, Le Bourgot C, Calame W, Respondek F. Effects of β-Fructans Fiber on Bowel Function: A Systematic Review and Meta-Analysis. Nutrients. 2019; 11(1):91. https://doi.org/10.3390/nu11010091
Chicago/Turabian StyleDe Vries, Jan, Cindy Le Bourgot, Wim Calame, and Frédérique Respondek. 2019. "Effects of β-Fructans Fiber on Bowel Function: A Systematic Review and Meta-Analysis" Nutrients 11, no. 1: 91. https://doi.org/10.3390/nu11010091
APA StyleDe Vries, J., Le Bourgot, C., Calame, W., & Respondek, F. (2019). Effects of β-Fructans Fiber on Bowel Function: A Systematic Review and Meta-Analysis. Nutrients, 11(1), 91. https://doi.org/10.3390/nu11010091





