The Role of Nutrients in Reducing the Risk for Noncommunicable Diseases during Aging
Abstract
1. Introduction
2. Musculoskeletal Health in the Older Adult
2.1. Vitamin D in Musculoskeletal Health
2.2. Vitamin K in Musculoskeletal Health
3. Cognitive Disorders
The Role of Nutrition in Dementia
4. Eye Disorders
5. Cardiovascular Disease
5.1. Cardiovascular Events
5.1.1. Diets
5.1.2. Vitamin D
5.1.3. B-Vitamins
5.1.4. Vitamin K
5.1.5. Omega-3 LCPUFA
5.1.6. Antioxidants
5.1.7. Vitamin E
5.1.8. Phenolics
5.2. Hypertension
5.2.1. Diets
5.2.2. Milk peptides
5.2.3. Omega-3 LCPUFAs
5.2.4. Vitamin C
5.2.5. Vitamin D
5.2.6. Flavonols
5.3. Diabetes
5.3.1. Vitamin D
5.3.2. Vitamin E
5.3.3. Omega-3 LCPUFA
5.3.4. Vitamin K
5.3.5. Chromium
6. Conclusions
Author Contributions
Funding
Acknowledgements
Conflicts of Interest
References
- World Health Organization (WHO). Global Health Observatory (GHO) Data, Life Expectancy. Available online: http://www.who.int/gho/mortality_burden_disease/life_tables/situation_trends/en/ (accessed on 20 September 2018).
- Figueira, I.; Fernandes, A.; Mladenovic Djordjevic, A.; Lopez-Contreras, A.; Henriques, C.M.; Selman, C.; Ferreiro, E.; Gonos, E.S.; Trejo, J.L.; Misra, J.; et al. Interventions for age-related diseases: Shifting the paradigm. Mech. Ageing Dev. 2016, 160, 69–92. [Google Scholar] [CrossRef]
- Troesch, B.; Biesalski, H.K.; Bos, R.; Buskens, E.; Calder, P.C.; Saris, W.H.; Spieldenner, J.; Verkade, H.J.; Weber, P.; Eggersdorfer, M. Increased Intake of Foods with High Nutrient Density Can Help to Break the Intergenerational Cycle of Malnutrition and Obesity. Nutrients 2015, 7, 6016–6037. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Global Action Plan for the Prevention and Control of NCDs 2013–2020. 2018. Available online: https://www.who.int/nmh/publications/ncd-action-plan/en/ (accessed on 3 January 2019).
- United Nations Economic and Social Council (ECOSOC). ECOSOC 2018 Task Force Resolution Urges Partners to Mobilize Resources for the Work of the Task Force. Available online: http://www.who.int/ncds/un-task-force/events/ecosoc-report-2018/en/ (accessed on 20 September 2018).
- World Health Organization (WHO). Health Statistics and Information Systems. Disease Burden and Mortality Estimates. Global Health Estimates 2016: DALYs by Age, Sex and Cause. Available online: http://www.who.int/healthinfo/global_burden_disease/estimates/en/index1.html (accessed on 20 September 2018).
- Eggersdorfer, M.; Walter, P. Emerging nutrition gaps in a world of affluence—Micronutrient intake and status globally. Int. J. Vitam. Nutr. Res. 2011, 81, 238–239. [Google Scholar] [CrossRef]
- Hoeft, B.; Weber, P.; Eggersdorfer, M. Micronutrients—A global perspective on intake, health benefits and economics. Int. J. Vitam. Nutr. Res. 2012, 82, 316–320. [Google Scholar] [CrossRef]
- Global Nutrition Report 2017, Nourishing the SDGs. Available online: http://165.227.233.32/wp-content/uploads/2017/11/Report_2017-2.pdf (accessed on 20 September 2018).
- Drewnowski, A. Nutrient density: Addressing the challenge of obesity. Br. J. Nutr. 2018, 120, S8–S14. [Google Scholar] [CrossRef]
- Mozaffarian, D.; Rosenberg, I.; Uauy, R. History of modern nutrition science-implications for current research, dietary guidelines, and food policy. BMJ 2018, 361, k2392. [Google Scholar] [CrossRef]
- Mertens, E.; Kuijsten, A.; Dofkova, M.; Mistura, L.; D’Addezio, L.; Turrini, A.; Dubuisson, C.; Favret, S.; Havard, S.; Trolle, E.; et al. Geographic and socioeconomic diversity of food and nutrient intakes: A comparison of four European countries. Eur. J. Nutr. 2018. [Google Scholar] [CrossRef]
- Candari, C.J.; Cylus, J.; Nolte, E. Assessing the Economic Costs of Unhealthy Diets and low Physical Activity: An Evidence Review and Proposed Framework. Available online: http://www.euro.who.int/en/publications/abstracts/assessing-the-economic-costs-of-unhealthy-diets-and-low-physical-activity-an-evidence-review-and-proposed-framework-2017 (accessed on 3 January 2019).
- Peter, S.; Saris, W.H.; Mathers, J.C.; Feskens, E.; Schols, A.; Navis, G.; Kuipers, F.; Weber, P.; Eggersdorfer, M. Nutrient Status Assessment in Individuals and Populations for Healthy Aging-Statement from an Expert Workshop. Nutrients 2015, 7, 10491–10500. [Google Scholar] [CrossRef]
- De Groot, L.C. Nutritional issues for older adults: Addressing degenerative ageing with long-term studies. Proc. Nutr. Soc. 2016, 75, 169–173. [Google Scholar] [CrossRef]
- Siddique, N.; O’Donoghue, M.; Casey, M.C.; Walsh, J.B. Malnutrition in the elderly and its effects on bone health—A review. Clin. Nutr. ESPEN 2017, 21, 31–39. [Google Scholar] [CrossRef]
- Leslie, W.; Hankey, C. Aging, Nutritional Status and Health. Healthcare (Basel) 2015, 3, 648–658. [Google Scholar] [CrossRef]
- Arnim, C.A.F. Nutrition security in older adults: Status quo and future development. In Sustainable Nutrition in a Changing World; Biesalski, H.K., Drewnowski, A., Dwyer, J.T., Strain, J.J., Weber, P., Eggersdorfer, M., Eds.; Springer International Publishing: Basel, Switzerland, 2017; pp. 61–73. [Google Scholar]
- Kimokoti, R.W.; Millen, B.E. Nutrition for the Prevention of Chronic Diseases. Med. Clin. N. Am. 2016, 100, 1185–1198. [Google Scholar] [CrossRef]
- Bruins, M.J.; Bird, J.K.; Aebischer, C.P.; Eggersdorfer, M. Considerations for Secondary Prevention of Nutritional Deficiencies in High-Risk Groups in High-Income Countries. Nutrients 2018, 10, 47. [Google Scholar] [CrossRef]
- Wong, C.W. Vitamin B12 deficiency in the elderly: Is it worth screening? Hong Kong Med. J. 2015, 21, 155–164. [Google Scholar] [CrossRef]
- Bendik, I.; Friedel, A.; Roos, F.F.; Weber, P.; Eggersdorfer, M. Vitamin D: A critical and essential micronutrient for human health. Front. Physiol. 2014, 5, 248. [Google Scholar] [CrossRef]
- Stover, P.J. Influence of human genetic variation on nutritional requirements. Am. J. Clin. Nutr. 2006, 83, 436S–442S. [Google Scholar] [CrossRef]
- Prince, M.J.; Wu, F.; Guo, Y.; Gutierrez Robledo, L.M.; O’Donnell, M.; Sullivan, R.; Yusuf, S. The burden of disease in older people and implications for health policy and practice. Lancet 2015, 385, 549–562. [Google Scholar] [CrossRef]
- Avenell, A.; Mak, J.C.; O’Connell, D. Vitamin D and vitamin D analogues for preventing fractures in post-menopausal women and older men. Cochrane Database Syst. Rev. 2014, 14, CD000227. [Google Scholar] [CrossRef]
- Johnell, O.; Kanis, J.A. An estimate of the worldwide prevalence and disability associated with osteoporotic fractures. Osteoporos. Int. 2006, 17, 1726–1733. [Google Scholar] [CrossRef]
- World Health Organization (WHO). WHO Scientific Group on the Assessment of Osteoporosis at Primary Health Care Level; Summary Meeting Report, Brussels, Belgium, 5–7 May 2004; WHO: Geneva, Switzerland, 2004. [Google Scholar]
- Hernlund, E.; Svedbom, A.; Ivergard, M.; Compston, J.; Cooper, C.; Stenmark, J.; McCloskey, E.V.; Jonsson, B.; Kanis, J.A. Osteoporosis in the European Union: Medical management, epidemiology and economic burden. A report prepared in collaboration with the International Osteoporosis Foundation (IOF) and the European Federation of Pharmaceutical Industry Associations (EFPIA). Arch. Osteoporos. 2013, 8, 136. [Google Scholar] [CrossRef]
- Beaudart, C.; Buckinx, F.; Rabenda, V.; Gillain, S.; Cavalier, E.; Slomian, J.; Petermans, J.; Reginster, J.Y.; Bruyere, O. The effects of vitamin D on skeletal muscle strength, muscle mass, and muscle power: A systematic review and meta-analysis of randomized controlled trials. J. Clin. Endocrinol. Metab. 2014, 99, 4336–4345. [Google Scholar] [CrossRef]
- Thiering, E.; Bruske, I.; Kratzsch, J.; Hofbauer, L.C.; Berdel, D.; von Berg, A.; Lehmann, I.; Hoffmann, B.; Bauer, C.P.; Koletzko, S.; et al. Associations between serum 25-hydroxyvitamin D and bone turnover markers in a population based sample of German children. Sci. Rep. 2015, 5, 18138. [Google Scholar] [CrossRef]
- Quesada-Gomez, J.M.; Bouillon, R. Is calcifediol better than cholecalciferol for vitamin D supplementation? Osteoporos. Int. 2018, 29, 1697–1711. [Google Scholar] [CrossRef]
- Sai, A.J.; Walters, R.W.; Fang, X.; Gallagher, J.C. Relationship between vitamin D, parathyroid hormone, and bone health. J. Clin. Endocrinol. Metab. 2011, 96, E436–E446. [Google Scholar] [CrossRef]
- Heaney, R.P. Functional indices of vitamin D status and ramifications of vitamin D deficiency. Am. J. Clin. Nutr. 2004, 80, 1706S–1709S. [Google Scholar] [CrossRef]
- Cauley, J.A.; Parimi, N.; Ensrud, K.E.; Bauer, D.C.; Cawthon, P.M.; Cummings, S.R.; Hoffman, A.R.; Shikany, J.M.; Barrett-Connor, E.; Orwoll, E.; et al. Serum 25-hydroxyvitamin D and the risk of hip and nonspine fractures in older men. J. Bone Miner. Res. 2010, 25, 545–553. [Google Scholar] [CrossRef]
- Looker, A.C.; Mussolino, M.E. Serum 25-hydroxyvitamin D and hip fracture risk in older U.S. white adults. J. Bone Miner. Res. 2008, 23, 143–150. [Google Scholar] [CrossRef]
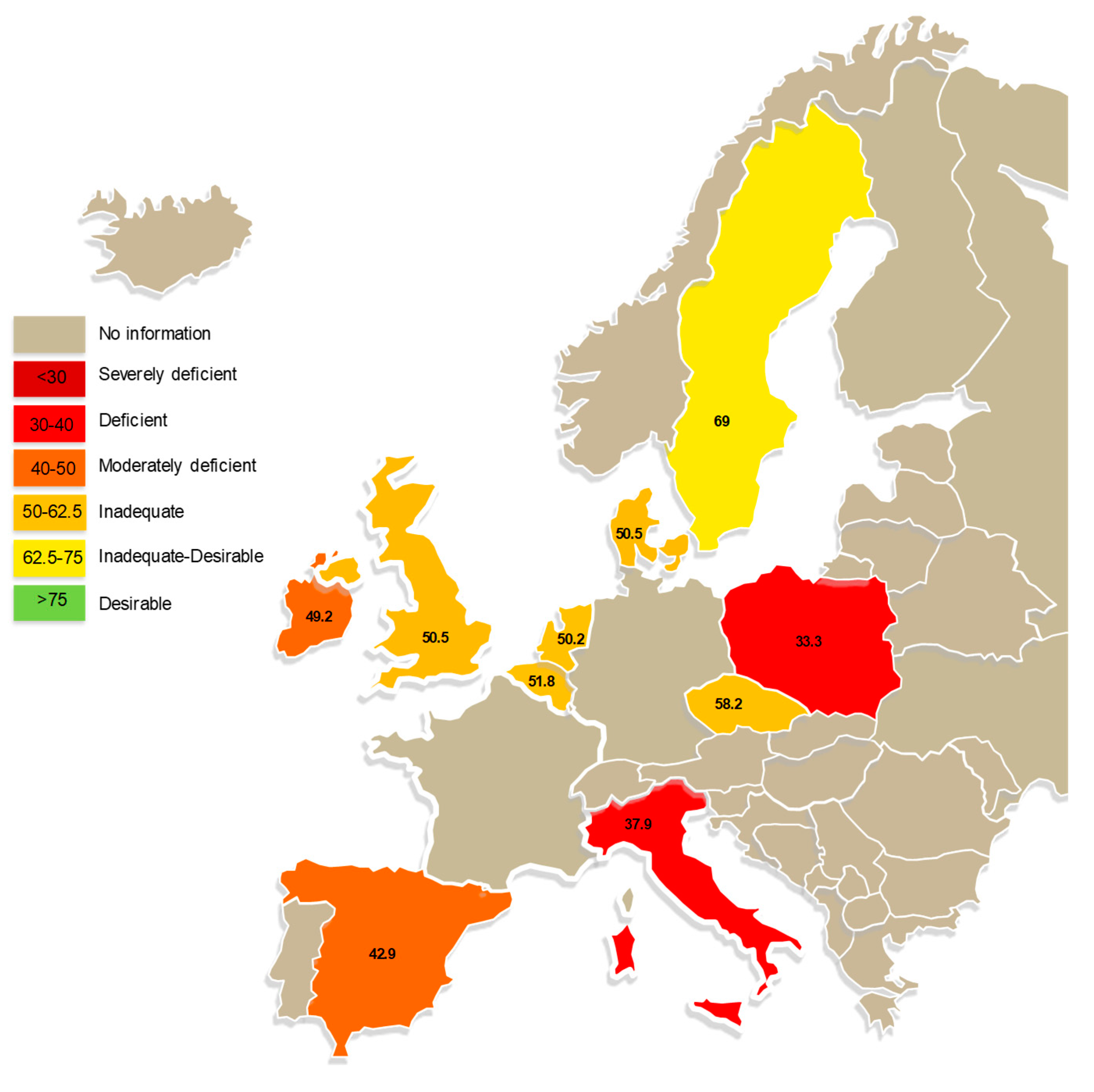
- Cashman, K.D.; Dowling, K.G.; Skrabakova, Z.; Gonzalez-Gross, M.; Valtuena, J.; De Henauw, S.; Moreno, L.; Damsgaard, C.T.; Michaelsen, K.F.; Molgaard, C.; et al. Vitamin D deficiency in Europe: Pandemic? Am. J. Clin. Nutr. 2016, 103, 1033–1044. [Google Scholar] [CrossRef]
- Hilger, J.; Friedel, A.; Herr, R.; Rausch, T.; Roos, F.; Wahl, D.A.; Pierroz, D.D.; Weber, P.; Hoffmann, K. A systematic review of vitamin D status in populations worldwide. Br. J. Nutr. 2014, 111, 23–45. [Google Scholar] [CrossRef]
- Wahl, D.A.; Cooper, C.; Ebeling, P.R.; Eggersdorfer, M.; Hilger, J.; Hoffmann, K.; Josse, R.; Kanis, J.A.; Mithal, A.; Pierroz, D.D.; et al. A global representation of vitamin D status in healthy populations: Reply to comment by Saadi. Arch. Osteoporos. 2013, 8, 122. [Google Scholar] [CrossRef]
- Spiro, A.; Buttriss, J.L. Vitamin D: An overview of vitamin D status and intake in Europe. Nutr. Bull. 2014, 39, 322–350. [Google Scholar] [CrossRef]
- Van Ballegooijen, A.J.; Pilz, S.; Tomaschitz, A.; Grubler, M.R.; Verheyen, N. The Synergistic Interplay between Vitamins D and K for Bone and Cardiovascular Health: A Narrative Review. Int. J. Endocrinol. 2017, 2017, 7454376. [Google Scholar] [CrossRef]
- Cockayne, S.; Adamson, J.; Lanham-New, S.; Shearer, M.J.; Gilbody, S.; Torgerson, D.J. Vitamin K and the prevention of fractures: Systematic review and meta-analysis of randomized controlled trials. Arch. Intern. Med. 2006, 166, 1256–1261. [Google Scholar] [CrossRef]
- Bolton-Smith, C.; McMurdo, M.E.; Paterson, C.R.; Mole, P.A.; Harvey, J.M.; Fenton, S.T.; Prynne, C.J.; Mishra, G.D.; Shearer, M.J. Two-year randomized controlled trial of vitamin K1 (phylloquinone) and vitamin D3 plus calcium on the bone health of older women. J. Bone Miner. Res. 2007, 22, 509–519. [Google Scholar] [CrossRef]
- Je, S.H.; Joo, N.S.; Choi, B.H.; Kim, K.M.; Kim, B.T.; Park, S.B.; Cho, D.Y.; Kim, K.N.; Lee, D.J. Vitamin K supplement along with vitamin D and calcium reduced serum concentration of undercarboxylated osteocalcin while increasing bone mineral density in Korean postmenopausal women over sixty-years-old. J. Korean Med. Sci. 2011, 26, 1093–1098. [Google Scholar] [CrossRef]
- Moore, K.; Hughes, C.F.; Ward, M.; Hoey, L.; McNulty, H. Diet, nutrition and the ageing brain: Current evidence and new directions. Proc. Nutr. Soc. 2018, 77, 152–163. [Google Scholar] [CrossRef]
- Prince, M.; Bryce, R.; Albanese, E.; Wimo, A.; Ribeiro, W.; Ferri, C.P. The global prevalence of dementia: A systematic review and metaanalysis. Alzheimers Dement. 2013, 9, 63–75. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Fact Sheets: Dementia. Available online: http://www.who.int/news-room/fact-sheets/detail/dementia (accessed on 12 December 2017).
- Jiang, T.; Yu, J.T.; Tian, Y.; Tan, L. Epidemiology and etiology of Alzheimer’s disease: From genetic to non-genetic factors. Curr. Alzheimer Res. 2013, 10, 852–867. [Google Scholar] [CrossRef]
- Antal, M.; Péter, S.; Eggersdorfer, M. Alzheimer’s Disease: An epidemiologic disaster from nutritional perspective. Nutr. Health Food Sci. 2017, 5, 1–4. [Google Scholar]
- Reynolds, E. Vitamin B12, folic acid, and the nervous system. Lancet Neurol. 2006, 5, 949–960. [Google Scholar] [CrossRef]
- Reynolds, E.H. Folic acid, ageing, depression, and dementia. BMJ 2002, 324, 1512–1515. [Google Scholar] [CrossRef]
- McCleery, J.; Abraham, R.P.; Denton, D.A.; Rutjes, A.W.; Chong, L.Y.; Al-Assaf, A.S.; Griffith, D.J.; Rafeeq, S.; Yaman, H.; Malik, M.A.; et al. Vitamin and mineral supplementation for preventing dementia or delaying cognitive decline in people with mild cognitive impairment. Cochrane Database Syst. Rev. 2018, 11, CD011905. [Google Scholar] [CrossRef]
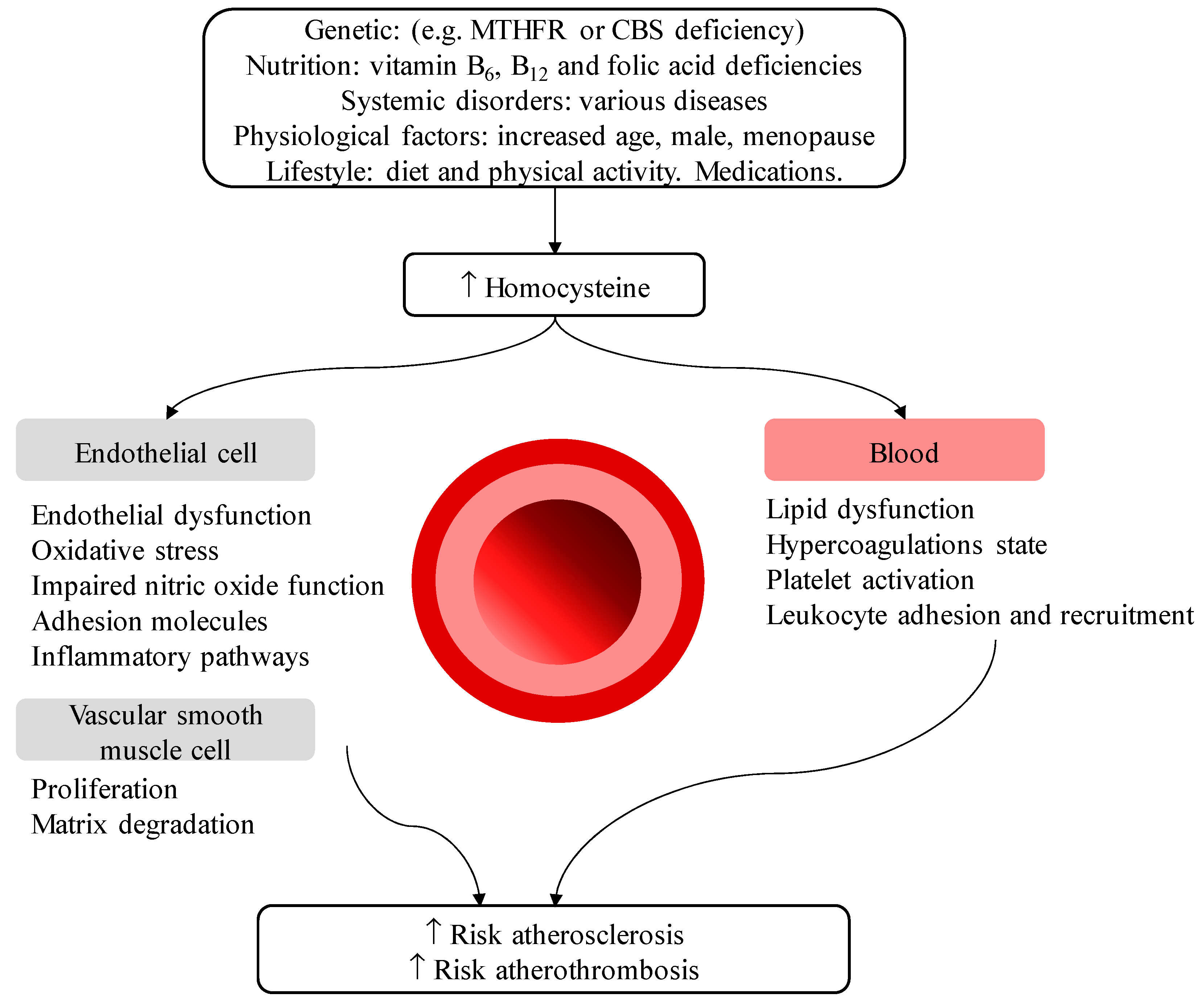
- Smith, A.D.; Refsum, H.; Bottiglieri, T.; Fenech, M.; Hooshmand, B.; McCaddon, A.; Miller, J.W.; Rosenberg, I.H.; Obeid, R. Homocysteine and Dementia: An International Consensus Statement. J. Alzheimers Dis. 2018, 62, 561–570. [Google Scholar] [CrossRef]
- Douaud, G.; Refsum, H.; de Jager, C.A.; Jacoby, R.; Nichols, T.E.; Smith, S.M.; Smith, A.D. Preventing Alzheimer’s disease-related gray matter atrophy by B-vitamin treatment. Proc. Natl. Acad. Sci. USA 2013, 110, 9523–9528. [Google Scholar] [CrossRef]
- Troesch, B.; Hoeft, B.; McBurney, M.; Eggersdorfer, M.; Weber, P. Dietary surveys indicate vitamin intakes below recommendations are common in representative Western countries. Br. J. Nutr. 2012, 108, 692–698. [Google Scholar] [CrossRef]
- Gugliandolo, A.; Bramanti, P.; Mazzon, E. Role of Vitamin E in the Treatment of Alzheimer’s Disease: Evidence from Animal Models. Int. J. Mol. Sci. 2017, 18, 2504. [Google Scholar] [CrossRef]
- Salem, N., Jr.; Vandal, M.; Calon, F. The benefit of docosahexaenoic acid for the adult brain in aging and dementia. Prostaglandins Leukot. Essent. Fatty Acids 2015, 92, 15–22. [Google Scholar] [CrossRef]
- Zhang, Y.J.; Gan, R.Y.; Li, S.; Zhou, Y.; Li, A.N.; Xu, D.P.; Li, H.B. Antioxidant Phytochemicals for the Prevention and Treatment of Chronic Diseases. Molecules 2015, 20, 21138–21156. [Google Scholar] [CrossRef]
- GBD 2015 DALYs and Hale Collaborators. Global, regional, and national disability-adjusted life-years (DALYs) for 315 diseases and injuries and healthy life expectancy (HALE), 1990–2015: A systematic analysis for the Global Burden of Disease Study 2015. Lancet 2016, 388, 1603–1658. [Google Scholar] [CrossRef]
- Quillen, D.A. Common causes of vision loss in elderly patients. Am. Fam. Physician 1999, 60, 99–108. [Google Scholar]
- Chen, Y.; Bedell, M.; Zhang, K. Age-related macular degeneration: Genetic and environmental factors of disease. Mol. Interv. 2010, 10, 271–281. [Google Scholar] [CrossRef]
- Tosini, G.; Ferguson, I.; Tsubota, K. Effects of blue light on the circadian system and eye physiology. Mol. Vis. 2016, 22, 61–72. [Google Scholar]
- Demmig-Adams, B.; Adams, R.B. Eye nutrition in context: Mechanisms, implementation, and future directions. Nutrients 2013, 5, 2483–2501. [Google Scholar] [CrossRef]
- Bernstein, P.S.; Li, B.; Vachali, P.P.; Gorusupudi, A.; Shyam, R.; Henriksen, B.S.; Nolan, J.M. Lutein, zeaxanthin, and meso-zeaxanthin: The basic and clinical science underlying carotenoid-based nutritional interventions against ocular disease. Prog. Retin. Eye Res. 2016, 50, 34–66. [Google Scholar] [CrossRef]
- Barker, F.M., II; Snodderly, D.M.; Johnson, E.J.; Schalch, W.; Koepcke, W.; Gerss, J.; Neuringer, M. Nutritional manipulation of primate retinas, V: Effects of lutein, zeaxanthin, and n-3 fatty acids on retinal sensitivity to blue-light-induced damage. Investig. Ophthalmol. Vis. Sci. 2011, 52, 3934–3942. [Google Scholar] [CrossRef]
- Hammond, B.R.; Fletcher, L.M.; Roos, F.; Wittwer, J.; Schalch, W. A double-blind, placebo-controlled study on the effects of lutein and zeaxanthin on photostress recovery, glare disability, and chromatic contrast. Investig. Ophthalmol. Vis. Sci. 2014, 55, 8583–8589. [Google Scholar] [CrossRef]
- Yao, Y.; Qiu, Q.H.; Wu, X.W.; Cai, Z.Y.; Xu, S.; Liang, X.Q. Lutein supplementation improves visual performance in Chinese drivers: 1-year randomized, double-blind, placebo-controlled study. Nutrition 2013, 29, 958–964. [Google Scholar] [CrossRef]
- Loughman, J.; Nolan, J.M.; Howard, A.N.; Connolly, E.; Meagher, K.; Beatty, S. The impact of macular pigment augmentation on visual performance using different carotenoid formulations. Investig. Ophthalmol. Vis. Sci. 2012, 53, 7871–7880. [Google Scholar] [CrossRef]
- Nolan, J.M.; Loughman, J.; Akkali, M.C.; Stack, J.; Scanlon, G.; Davison, P.; Beatty, S. The impact of macular pigment augmentation on visual performance in normal subjects: COMPASS. Vision Res. 2011, 51, 459–469. [Google Scholar] [CrossRef]
- Richer, S.P.; Stiles, W.; Graham-Hoffman, K.; Levin, M.; Ruskin, D.; Wrobel, J.; Park, D.W.; Thomas, C. Randomized, double-blind, placebo-controlled study of zeaxanthin and visual function in patients with atrophic age-related macular degeneration: The Zeaxanthin and Visual Function Study (ZVF) FDA IND #78, 973. Optometry 2011, 82, 667–680. [Google Scholar]
- Stringham, J.M.; Hammond, B.R. Macular pigment and visual performance under glare conditions. Optom. Vis. Sci. 2008, 85, 82–88. [Google Scholar] [CrossRef]
- Wenzel, A.J.; Fuld, K.; Stringham, J.M.; Curran-Celentano, J. Macular pigment optical density and photophobia light threshold. Vision Res. 2006, 46, 4615–4622. [Google Scholar] [CrossRef]
- Kvansakul, J.; Rodriguez-Carmona, M.; Edgar, D.F.; Barker, F.M.; Kopcke, W.; Schalch, W.; Barbur, J.L. Supplementation with the carotenoids lutein or zeaxanthin improves human visual performance. Ophthalmic. Physiol. Opt. 2006, 26, 362–371. [Google Scholar] [CrossRef]
- Feeney, J.; Finucane, C.; Savva, G.M.; Cronin, H.; Beatty, S.; Nolan, J.M.; Kenny, R.A. Low macular pigment optical density is associated with lower cognitive performance in a large, population-based sample of older adults. Neurobiol. Aging 2013, 34, 2449–2456. [Google Scholar] [CrossRef]
- Johnson, E.J.; Maras, J.E.; Rasmussen, H.M.; Tucker, K.L. Intake of lutein and zeaxanthin differ with age, sex, and ethnicity. J. Am. Diet. Assoc. 2010, 110, 1357–1362. [Google Scholar] [CrossRef]
- Nolan, J.M.; Power, R.; Stringham, J.; Dennison, J.; Stack, J.; Kelly, D.; Moran, R.; Akuffo, K.O.; Corcoran, L.; Beatty, S. Author Response: Comments on Enrichment of Macular Pigment Enhances Contrast Sensitivity in Subjects Free of Retinal Disease: CREST—Report 1. Investig. Ophthalmol. Vis. Sci. 2016, 57, 5416. [Google Scholar] [CrossRef]
- Olmedilla, B.; Granado, F.; Blanco, I.; Vaquero, M. Lutein, but not alpha-tocopherol, supplementation improves visual function in patients with age-related cataracts: A 2-y double-blind, placebo-controlled pilot study. Nutrition 2003, 19, 21–24. [Google Scholar] [CrossRef]
- Age-Related Eye Disease Study 2 Research Group. Lutein + zeaxanthin and omega-3 fatty acids for age-related macular degeneration: The Age-Related Eye Disease Study 2 (AREDS2) randomized clinical trial. JAMA 2013, 309, 2005–2015. [Google Scholar] [CrossRef]
- Age-Related Eye Disease Study 2 Research Group; Chew, E.Y.; Clemons, T.E.; Sangiovanni, J.P.; Danis, R.P.; Ferris, F.L., III; Elman, M.J.; Antoszyk, A.N.; Ruby, A.J.; Orth, D.; et al. Secondary analyses of the effects of lutein/zeaxanthin on age-related macular degeneration progression: AREDS2 report No. 3. JAMA Ophthalmol. 2014, 132, 142–149. [Google Scholar] [CrossRef]
- Liu, X.H.; Yu, R.B.; Liu, R.; Hao, Z.X.; Han, C.C.; Zhu, Z.H.; Ma, L. Association between lutein and zeaxanthin status and the risk of cataract: A meta-analysis. Nutrients 2014, 6, 452–465. [Google Scholar] [CrossRef]
- Age-Related Eye Disease Study 2 Research Group; Chew, E.Y.; SanGiovanni, J.P.; Ferris, F.L.; Wong, W.T.; Agron, E.; Clemons, T.E.; Sperduto, R.; Danis, R.; Chandra, S.R.; et al. Lutein/zeaxanthin for the treatment of age-related cataract: AREDS2 randomized trial report no. 4. JAMA Ophthalmol. 2013, 131, 843–850. [Google Scholar] [CrossRef]
- Shanahan, C. The Economic Benefits of Using Lutein and Zeaxanthin Food Supplements in the European Union. Available online: https://ww2.frost.com/files/7015/0772/2735/HCCS_Lutein_AMD.2017.10.12.pdf (accessed on 3 January 2019).
- GBD Causes of Death Collaborators. Global, regional, and national age-sex specific mortality for 264 causes of death, 1980–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet 2017, 390, 1151–1210. [Google Scholar] [CrossRef]
- Mozaffarian, D. Dietary and Policy Priorities for Cardiovascular Disease, Diabetes, and Obesity: A Comprehensive Review. Circulation 2016, 133, 187–225. [Google Scholar] [CrossRef]
- Leening, M.J.; Ferket, B.S.; Steyerberg, E.W.; Kavousi, M.; Deckers, J.W.; Nieboer, D.; Heeringa, J.; Portegies, M.L.; Hofman, A.; Ikram, M.A.; et al. Sex differences in lifetime risk and first manifestation of cardiovascular disease: Prospective population based cohort study. BMJ 2014, 349, g5992. [Google Scholar] [CrossRef]
- The Institute for Health Metrics and Evaluation (IHME). Global Burden of Disease (GBD). GBD Data Visualizations. Available online: http://www.healthdata.org/gbd (accessed on 20 September 2018).
- Hankey, G.J. The Role of Nutrition in the Risk and Burden of Stroke: An Update of the Evidence. Stroke J. Cereb. Circ. 2017, 48, 3168–3174. [Google Scholar] [CrossRef]
- Van Staveren, W.A.; de Groot, L.C. Evidence-based dietary guidance and the role of dairy products for appropriate nutrition in the elderly. J. Am. Coll. Nutr. 2011, 30, 429S–437S. [Google Scholar] [CrossRef]
- Fleg, J.L.; Forman, D.E.; Berra, K.; Bittner, V.; Blumenthal, J.A.; Chen, M.A.; Cheng, S.; Kitzman, D.W.; Maurer, M.S.; Rich, M.W.; et al. Secondary prevention of atherosclerotic cardiovascular disease in older adults: A scientific statement from the American Heart Association. Circulation 2013, 128, 2422–2446. [Google Scholar] [CrossRef]
- Catapano, A.L.; Graham, I.; De Backer, G.; Wiklund, O.; Chapman, M.J.; Drexel, H.; Hoes, A.W.; Jennings, C.S.; Landmesser, U.; Pedersen, T.R.; et al. 2016 ESC/EAS Guidelines for the Management of Dyslipidaemias. Rev. Esp. Cardiol. (Engl. Ed.) 2017, 70, 115. [Google Scholar]
- Jellinger, P.S. American Association of Clinical Endocrinologists/American College of Endocrinology Management of Dyslipidemia and Prevention of Cardiovascular Disease Clinical Practice Guidelines. Diabetes Spectr. 2018, 31, 234–245. [Google Scholar] [CrossRef]
- Eckel, R.H.; Jakicic, J.M.; Ard, J.D.; de Jesus, J.M.; Houston Miller, N.; Hubbard, V.S.; Lee, I.M.; Lichtenstein, A.H.; Loria, C.M.; Millen, B.E.; et al. 2013 AHA/ACC guideline on lifestyle management to reduce cardiovascular risk: A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. Circulation 2014, 129, S76–S99. [Google Scholar] [CrossRef]
- Estruch, R.; Ros, E.; Salas-Salvado, J.; Covas, M.I.; Corella, D.; Aros, F.; Gomez-Gracia, E.; Ruiz-Gutierrez, V.; Fiol, M.; Lapetra, J.; et al. Primary Prevention of Cardiovascular Disease with a Mediterranean Diet Supplemented with Extra-Virgin Olive Oil or Nuts. N. Engl. J. Med. 2018, 378, e34. [Google Scholar] [CrossRef]
- De Lorgeril, M.; Salen, P.; Martin, J.L.; Monjaud, I.; Delaye, J.; Mamelle, N. Mediterranean diet, traditional risk factors, and the rate of cardiovascular complications after myocardial infarction: Final report of the Lyon Diet Heart Study. Circulation 1999, 99, 779–785. [Google Scholar] [CrossRef]
- Ornish, D.; Scherwitz, L.W.; Billings, J.H.; Brown, S.E.; Gould, K.L.; Merritt, T.A.; Sparler, S.; Armstrong, W.T.; Ports, T.A.; Kirkeeide, R.L.; et al. Intensive lifestyle changes for reversal of coronary heart disease. JAMA 1998, 280, 2001–2007. [Google Scholar] [CrossRef]
- Kheiri, B.; Abdalla, A.; Osman, M.; Ahmed, S.; Hassan, M.; Bachuwa, G. Vitamin D deficiency and risk of cardiovascular diseases: A narrative review. Clin. Hypertens. 2018, 24, 9. [Google Scholar] [CrossRef]
- Gepner, A.D.; Ramamurthy, R.; Krueger, D.C.; Korcarz, C.E.; Binkley, N.; Stein, J.H. A prospective randomized controlled trial of the effects of vitamin D supplementation on cardiovascular disease risk. PLoS ONE 2012, 7, e36617. [Google Scholar] [CrossRef]
- Ford, J.A.; MacLennan, G.S.; Avenell, A.; Bolland, M.; Grey, A.; Witham, M.; Group, R.T. Cardiovascular disease and vitamin D supplementation: Trial analysis, systematic review, and meta-analysis. Am. J. Clin. Nutr. 2014, 100, 746–755. [Google Scholar] [CrossRef]
- Vitamin D and Omega-3 Trial (VITAL). Available online: https://www.vitalstudy.org/findings.html (accessed on 20 December 2018).
- Huang, T.; Chen, Y.; Yang, B.; Yang, J.; Wahlqvist, M.L.; Li, D. Meta-analysis of B vitamin supplementation on plasma homocysteine, cardiovascular and all-cause mortality. Clin. Nutr. 2012, 31, 448–454. [Google Scholar] [CrossRef]
- Yang, H.T.; Lee, M.; Hong, K.S.; Ovbiagele, B.; Saver, J.L. Efficacy of folic acid supplementation in cardiovascular disease prevention: An updated meta-analysis of randomized controlled trials. Eur. J. Intern. Med. 2012, 23, 745–754. [Google Scholar] [CrossRef]
- Wald, D.S.; Law, M.; Morris, J.K. Homocysteine and cardiovascular disease: Evidence on causality from a meta-analysis. BMJ 2002, 325, 1202. [Google Scholar] [CrossRef]
- Li, Y.; Huang, T.; Zheng, Y.; Muka, T.; Troup, J.; Hu, F.B. Folic Acid Supplementation and the Risk of Cardiovascular Diseases: A Meta-Analysis of Randomized Controlled Trials. J. Am. Heart Assoc. 2016, 5, e003768. [Google Scholar] [CrossRef]
- Fezeu, L.K.; Ducros, V.; Gueant, J.L.; Guilland, J.C.; Andreeva, V.A.; Hercberg, S.; Galan, P. MTHFR 677C→T genotype modulates the effect of a 5-year supplementation with B-vitamins on homocysteine concentration: The SU.FOL.OM3 randomized controlled trial. PLoS ONE 2018, 13, e0193352. [Google Scholar] [CrossRef]
- DiNicolantonio, J.J.; Bhutani, J.; O’Keefe, J.H. The health benefits of vitamin K. Open Heart 2015, 2, e000300. [Google Scholar] [CrossRef]
- Innes, J.K.; Calder, P.C. The Differential Effects of Eicosapentaenoic Acid and Docosahexaenoic Acid on Cardiometabolic Risk Factors: A Systematic Review. Int. J. Mol. Sci. 2018, 19, 532. [Google Scholar] [CrossRef]
- Thota, R.N.; Ferguson, J.J.A.; Abbott, K.A.; Dias, C.B.; Garg, M.L. Science behind the cardio-metabolic benefits of omega-3 polyunsaturated fatty acids: Biochemical effects vs. clinical outcomes. Food Funct. 2018, 9, 3576–3596. [Google Scholar] [CrossRef]
- Karalis, D.G. A Review of Clinical Practice Guidelines for the Management of Hypertriglyceridemia: A Focus on High Dose Omega-3 Fatty Acids. Adv. Ther. 2017, 34, 300–323. [Google Scholar] [CrossRef]
- Alexander, D.D.; Miller, P.E.; Van Elswyk, M.E.; Kuratko, C.N.; Bylsma, L.C. A Meta-Analysis of Randomized Controlled Trials and Prospective Cohort Studies of Eicosapentaenoic and Docosahexaenoic Long-Chain Omega-3 Fatty Acids and Coronary Heart Disease Risk. Mayo Clin. Proc. 2017, 92, 15–29. [Google Scholar] [CrossRef]
- Abdelhamid, A.S.; Brown, T.J.; Brainard, J.S.; Biswas, P.; Thorpe, G.C.; Moore, H.J.; Deane, K.H.; AlAbdulghafoor, F.K.; Summerbell, C.D.; Worthington, H.V.; et al. Omega-3 fatty acids for the primary and secondary prevention of cardiovascular disease. Cochrane Database Syst. Rev. 2018, 7, CD003177. [Google Scholar]
- Meyer, B.J.; Groot, R.H.M. Effects of Omega-3 Long Chain Polyunsaturated Fatty Acid Supplementation on Cardiovascular Mortality: The Importance of the Dose of DHA. Nutrients 2017, 9, 1305. [Google Scholar] [CrossRef]
- Kromhout, D.; Giltay, E.J.; Geleijnse, J.M.; Alpha Omega Trial, G. n-3 fatty acids and cardiovascular events after myocardial infarction. N. Engl. J. Med. 2010, 363, 2015–2026. [Google Scholar] [CrossRef]
- Writing Group for the AREDS2 Research Group; Bonds, D.E.; Harrington, M.; Worrall, B.B.; Bertoni, A.G.; Eaton, C.B.; Hsia, J.; Robinson, J.; Clemons, T.E.; Fine, L.J.; et al. Effect of long-chain omega-3 fatty acids and lutein + zeaxanthin supplements on cardiovascular outcomes: Results of the Age-Related Eye Disease Study 2 (AREDS2) randomized clinical trial. JAMA Intern. Med. 2014, 174, 763–771. [Google Scholar]
- Hidayat, K.; Yang, J.; Zhang, Z.; Chen, G.C.; Qin, L.Q.; Eggersdorfer, M.; Zhang, W. Effect of omega-3 long-chain polyunsaturated fatty acid supplementation on heart rate: A meta-analysis of randomized controlled trials. Eur. J. Clin. Nutr. 2018, 72, 805–817. [Google Scholar] [CrossRef]
- Zhang, G.Q.; Zhang, W. Heart rate, lifespan, and mortality risk. Ageing Res. Rev. 2009, 8, 52–60. [Google Scholar] [CrossRef]
- Chen, Y.; Michalak, M.; Agellon, L.B. Importance of Nutrients and Nutrient Metabolism on Human Health. Yale J. Biol. Med. 2018, 91, 95–103. [Google Scholar]
- Rangel-Huerta, O.D.; Pastor-Villaescusa, B.; Aguilera, C.M.; Gil, A. A Systematic Review of the Efficacy of Bioactive Compounds in Cardiovascular Disease: Phenolic Compounds. Nutrients 2015, 7, 5177–5216. [Google Scholar] [CrossRef]
- Ye, Y.; Li, J.; Yuan, Z. Effect of antioxidant vitamin supplementation on cardiovascular outcomes: A meta-analysis of randomized controlled trials. PLoS ONE 2013, 8, e56803. [Google Scholar] [CrossRef]
- Raederstorff, D.; Wyss, A.; Calder, P.C.; Weber, P.; Eggersdorfer, M. Vitamin E function and requirements in relation to PUFA. Br. J. Nutr. 2015, 114, 1113–1122. [Google Scholar] [CrossRef]
- Lee, I.M.; Cook, N.R.; Gaziano, J.M.; Gordon, D.; Ridker, P.M.; Manson, J.E.; Hennekens, C.H.; Buring, J.E. Vitamin E in the primary prevention of cardiovascular disease and cancer: The Women’s Health Study: A randomized controlled trial. JAMA 2005, 294, 56–65. [Google Scholar] [CrossRef]
- Glynn, R.J.; Ridker, P.M.; Goldhaber, S.Z.; Zee, R.Y.; Buring, J.E. Effects of random allocation to vitamin E supplementation on the occurrence of venous thromboembolism: Report from the Women’s Health Study. Circulation 2007, 116, 1497–1503. [Google Scholar] [CrossRef]
- Farbstein, D.; Levy, A.P. The genetics of vascular complications in diabetes mellitus. Cardiol. Clin. 2010, 28, 477–496. [Google Scholar] [CrossRef]
- He, H.Y.; Liu, M.Z.; Zhang, Y.L.; Zhang, W. Vitamin Pharmacogenomics: New Insight into Individual Differences in Diseases and Drug Responses. Genom. Proteom. Bioinform. 2017, 15, 94–100. [Google Scholar] [CrossRef]
- Huxley, R.R.; Neil, H.A. The relation between dietary flavonol intake and coronary heart disease mortality: A meta-analysis of prospective cohort studies. Eur. J. Clin. Nutr. 2003, 57, 904–908. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.M.; Nie, Z.L.; Zhou, B.; Lian, X.Q.; Zhao, H.; Gao, W.; Wang, Y.S.; Jia, E.Z.; Wang, L.S.; Yang, Z.J. Flavonols intake and the risk of coronary heart disease: A meta-analysis of cohort studies. Atherosclerosis 2012, 222, 270–273. [Google Scholar] [CrossRef] [PubMed]
- Castro, I.; Waclawovsky, G.; Marcadenti, A. Nutrition and physical activity on hypertension: Implication of current evidence and guidelines. Curr. Hypertens. Rev. 2015, 11, 91–99. [Google Scholar] [CrossRef] [PubMed]
- Miller, A.P.; Navar, A.M.; Roubin, G.S.; Oparil, S. Cardiovascular care for older adults: Hypertension and stroke in the older adult. J. Geriatr. Cardiol. 2016, 13, 373–379. [Google Scholar] [PubMed]
- James, P.A.; Oparil, S.; Carter, B.L.; Cushman, W.C.; Dennison-Himmelfarb, C.; Handler, J.; Lackland, D.T.; LeFevre, M.L.; MacKenzie, T.D.; Ogedegbe, O.; et al. 2014 evidence-based guideline for the management of high blood pressure in adults: Report from the panel members appointed to the Eighth Joint National Committee (JNC 8). JAMA 2014, 311, 507–520. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, H.; Odelola, O.A.; Rangaswami, J.; Amanullah, A. A review of nutritional factors in hypertension management. Int. J. Hypertens. 2013, 2013, 698940. [Google Scholar] [CrossRef] [PubMed]
- Perez, V.; Chang, E.T. Sodium-to-potassium ratio and blood pressure, hypertension, and related factors. Adv. Nutr. 2014, 5, 712–741. [Google Scholar] [CrossRef]
- Cicero, A.F.; Colletti, A.; Rosticci, M.; Cagnati, M.; Urso, R.; Giovannini, M.; Borghi, C.; D’Addato, S. Effect of Lactotripeptides (Isoleucine-Proline-Proline/Valine-Proline-Proline) on Blood Pressure and Arterial Stiffness Changes in Subjects with Suboptimal Blood Pressure Control and Metabolic Syndrome: A Double-Blind, Randomized, Crossover Clinical Trial. Metab. Syndr. Relat. Disord. 2016, 14, 161–166. [Google Scholar] [CrossRef]
- Miller, P.E.; Van Elswyk, M.; Alexander, D.D. Long-chain omega-3 fatty acids eicosapentaenoic acid and docosahexaenoic acid and blood pressure: A meta-analysis of randomized controlled trials. Am. J. Hypertens. 2014, 27, 885–896. [Google Scholar] [CrossRef]
- Geleijnse, J.M.; Giltay, E.J.; Grobbee, D.E.; Donders, A.R.; Kok, F.J. Blood pressure response to fish oil supplementation: Metaregression analysis of randomized trials. J. Hypertens. 2002, 20, 1493–1499. [Google Scholar] [CrossRef]
- Juraschek, S.P.; Guallar, E.; Appel, L.J.; Miller, E.R., III. Effects of vitamin C supplementation on blood pressure: A meta-analysis of randomized controlled trials. Am. J. Clin. Nutr. 2012, 95, 1079–1088. [Google Scholar] [CrossRef] [PubMed]
- Forman, J.P.; Scott, J.B.; Ng, K.; Drake, B.F.; Suarez, E.G.; Hayden, D.L.; Bennett, G.G.; Chandler, P.D.; Hollis, B.W.; Emmons, K.M.; et al. Effect of vitamin D supplementation on blood pressure in blacks. Hypertension 2013, 61, 779–785. [Google Scholar] [CrossRef] [PubMed]
- International Diabetes Federation (IDF). IDF Diabetes Atlas 8th Edition. Available online: http://www.diabetesatlas.org/ (accessed on 20 September 2018).
- Samocha-Bonet, D.; Debs, S.; Greenfield, J.R. Prevention and Treatment of Type 2 Diabetes: A Pathophysiological-Based Approach. Trends Endocrinol. Metab. 2018, 29, 370–379. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Liu, Y.; Zheng, Y.; Wang, P.; Zhang, Y. The Effect of Vitamin D Supplementation on Glycemic Control in Type 2 Diabetes Patients: A Systematic Review and Meta-Analysis. Nutrients 2018, 10, 375. [Google Scholar] [PubMed]
- Upreti, V.; Maitri, V.; Dhull, P.; Handa, A.; Prakash, M.S.; Behl, A. Effect of oral vitamin D supplementation on glycemic control in patients with type 2 diabetes mellitus with coexisting hypovitaminosis D: A parellel group placebo controlled randomized controlled pilot study. Diabetes Metab. Syndr. 2018, 12, 509–512. [Google Scholar] [CrossRef] [PubMed]
- Milman, U.; Blum, S.; Shapira, C.; Aronson, D.; Miller-Lotan, R.; Anbinder, Y.; Alshiek, J.; Bennett, L.; Kostenko, M.; Landau, M.; et al. Vitamin E supplementation reduces cardiovascular events in a subgroup of middle-aged individuals with both type 2 diabetes mellitus and the haptoglobin 2-2 genotype: A prospective double-blinded clinical trial. Arterioscler. Thromb. Vasc. Biol. 2008, 28, 341–347. [Google Scholar] [CrossRef] [PubMed]
- Asleh, R.; Briasoulis, A.; Berinstein, E.M.; Wiener, J.B.; Palla, M.; Kushwaha, S.S.; Levy, A.P. Meta-analysis of the association of the haptoglobin genotype with cardiovascular outcomes and the pharmacogenomic interactions with vitamin E supplementation. Pharmacogenom. Pers. Med. 2018, 11, 71–82. [Google Scholar] [CrossRef]
- Chen, C.; Yu, X.; Shao, S. Effects of Omega-3 Fatty Acid Supplementation on Glucose Control and Lipid Levels in Type 2 Diabetes: A Meta-Analysis. PLoS ONE 2015, 10, e0139565. [Google Scholar] [CrossRef]
- Beulens, J.W.; van der, A.D.; Grobbee, D.E.; Sluijs, I.; Spijkerman, A.M.; van der Schouw, Y.T. Dietary phylloquinone and menaquinones intakes and risk of type 2 diabetes. Diabetes Care 2010, 33, 1699–1705. [Google Scholar] [CrossRef]
- Yoshida, M.; Jacques, P.F.; Meigs, J.B.; Saltzman, E.; Shea, M.K.; Gundberg, C.; Dawson-Hughes, B.; Dallal, G.; Booth, S.L. Effect of vitamin K supplementation on insulin resistance in older men and women. Diabetes Care 2008, 31, 2092–2096. [Google Scholar] [CrossRef]
- Suksomboon, N.; Poolsup, N.; Yuwanakorn, A. Systematic review and meta-analysis of the efficacy and safety of chromium supplementation in diabetes. J. Clin. Pharm. Ther. 2014, 39, 292–306. [Google Scholar] [CrossRef] [PubMed]
- Blum, S.; Vardi, M.; Brown, J.B.; Russell, A.; Milman, U.; Shapira, C.; Levy, N.S.; Miller-Lotan, R.; Asleh, R.; Levy, A.P. Vitamin E reduces cardiovascular disease in individuals with diabetes mellitus and the haptoglobin 2-2 genotype. Pharmacogenomics 2010, 11, 675–684. [Google Scholar] [CrossRef] [PubMed]
| % Meeting EAR or AI | EAR or AI | Denmark n = 2025 people | Czech Republic n = 1869 people | Italy n = 2831 people | France n = 2624 people |
|---|---|---|---|---|---|
| Protein, g/d | 0.66 g/kg BW |  84% 84% |  88% 88% |  99% 99% |  98% 98% |
| MUFA, E% | 10–20 E% |  69% 69% |  92% 92% |  75% 75% |  77% 77% |
| Dietary fiber, g/d | 25 |  19% 19% |  4% 4% |  12% 12% |  9% 9% |
| Calcium, mg/d | 750 |  70% 70% |  31% 31% |  43% 43% |  62% 62% |
| Iron, mg/d | M: 6; F: 7 |  92% 92% |  96% 96% |  98% 98% |  98% 98% |
| Potassium, mg/d | 3500 |  31% 31% |  4% 4% |  19% 19% |  18% 18% |
| Magnesium, mg/d | M: 350; F: 300 |  46% 46% |  25% 25% |  20% 20% |  23% 23% |
| Zinc, mg/d | M: 7.5; F: 6.2 |  90% 90% |  48% 48% |  97% 97% |  91% 91% |
| Vitamin A, µg RE/d | M: 570; F490 |  77% 77% |  38% 38% |  66% 66% |  77% 77% |
| Vitamin C, mg/d | M: 90; F: 80 |  50% 50% |  35% 35% |  62% 62% |  44% 44% |
| Vitamin E, mg/d | M: 13; F: 11 |  5% 5% |  44% 44% |  47% 47% |  34% 34% |
| Vitamin D, µg/d | 15 |  3% 3% |  1% 1% |  1% 1% |  1% 1% |
| Vitamin B1, mg/d | 0.6 |  97% 97% |  98% 98% |  47% 47% |  100% 100% |
| Vitamin B2, mg/d | M: 1.1; F: 0.9 |  80% 80% |  35% 35% |  84% 84% |  92% 92% |
| Vitamin B12, µg/d | 4 |  55% 55% |  36% 36% |  52% 52% |  50% 50% |
| Folate, µg DFE/d | 250 |  59% 59% |  24% 24% |  77% 77% |  51% 51% |
| % exceeding MRV | MRV | ||||
| SFA, E% | <10 E% |  86% 86% |  80% 80% |  62% 62% |  91% 91% |
| Added sugar, E% | <10 E% |  32% 32% |  21% 21% |  24% 24% | |
| Sodium, mg/d | <2400 mg/d |  80% 80% |  98% 98% |  13% 13% |  85% 85% |
| Micronutrient | Challenges, Clinical Signs, and Symptoms in Older Adults |
|---|---|
| Vitamin B12 (cobalamin) | Deficiencies common in older adults, often underdiagnosed. Role in reducing elevated homocysteine, a cardiovascular risk factor. Absorption decreases mainly due to high prevalence of age-related atrophic gastritis. Among the common causes of anaemia in older adults, leading to weakness and fatigue. Low status increases the risk for cardiovascular disease and cognitive impairment. |
| Folate | Deficiencies common in older adults. Role in reducing elevated homocysteine, a cardiovascular risk factor. Closely related to vitamin B12 and B6. Among the common causes of anaemia in older adults, leading to weakness and fatigue. Deficiencies linked to depression and dementia. |
| Vitamin B6 | Deficiencies common in older adults. Role in reducing elevated homocysteine, a cardiovascular risk factor. Closely related to vitamin B12 and folate. |
| Thiamine (vitamin B1) | Deficiencies common in older adults, often underdiagnosed. Risk factor for heart failure, peripheral neuropathy, and encephalopathy. |
| Calcium | Deficiencies common in senior women. Mean intake decreases with age, probably related to general change in diet. Associated with low bone mass, rapid bone loss, and high fracture rates. |
| Vitamin D | Older adults are less exposed to sun and have diminished ability of the skin to synthesize vitamin and the liver and kidney to hydrolyze vitamin D with age. Deficiency is a risk factor low bone mass, rapid bone loss, high fracture rates, and muscle weakness. |
| Vitamin C | Prevalence of inadequate intake is very high among adults. May help elderly maintain immune cells and function. Smoking increases need. |
| Iron | Women’s iron requirements decrease after the menopause. Deficiencies are mainly seen among hospitalized, institutionalized, or chronically ill older adults. Among the common causes of anaemia in older adults, leading to weakness and fatigue. |
| Zinc | Deficiency is common in the elderly. Risk factor for immune deficiency and susceptibility to infection in the elderly. |
| Selenium | Deficiency deficiency may increase risk of diseases of aging such as cardiovascular disease, reduced immune response, and cognitive decline. |
| Magnesium | Often deficient in older adults. Maintains muscle integrity and function. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bruins, M.J.; Van Dael, P.; Eggersdorfer, M. The Role of Nutrients in Reducing the Risk for Noncommunicable Diseases during Aging. Nutrients 2019, 11, 85. https://doi.org/10.3390/nu11010085
Bruins MJ, Van Dael P, Eggersdorfer M. The Role of Nutrients in Reducing the Risk for Noncommunicable Diseases during Aging. Nutrients. 2019; 11(1):85. https://doi.org/10.3390/nu11010085
Chicago/Turabian StyleBruins, Maaike J., Peter Van Dael, and Manfred Eggersdorfer. 2019. "The Role of Nutrients in Reducing the Risk for Noncommunicable Diseases during Aging" Nutrients 11, no. 1: 85. https://doi.org/10.3390/nu11010085
APA StyleBruins, M. J., Van Dael, P., & Eggersdorfer, M. (2019). The Role of Nutrients in Reducing the Risk for Noncommunicable Diseases during Aging. Nutrients, 11(1), 85. https://doi.org/10.3390/nu11010085






