Impact of Two Forms of Daily Preventive Zinc or Therapeutic Zinc Supplementation for Diarrhea on Hair Cortisol Concentrations Among Rural Laotian Children: A Randomized Controlled Trial
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethical Approval
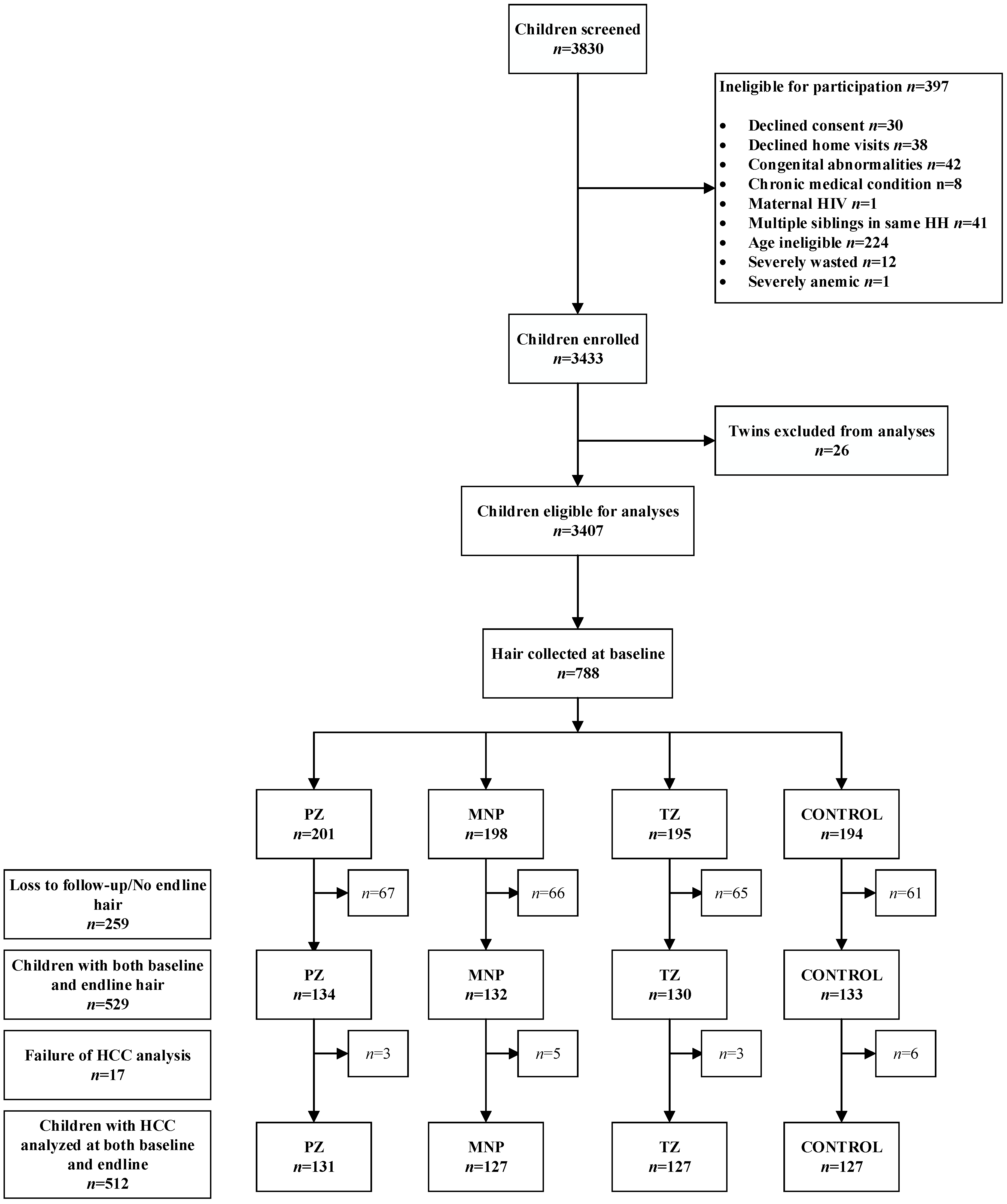
2.2. Study Design
2.3. Sample Size Estimation
2.4. Randomization and Intervention Products
2.5. Data Collection
2.6. Morbidity Surveillance and Supplement Administration
2.7. Hair Samples Collection and Hair Cortisol Analyses
2.8. Definitions
2.9. Statistical Analyses
3. Results
3.1. Study Population
3.2. Impact of the Study Interventions on HCC
3.3. Risk Factors Associated with HCC at Baseline
3.4. Association Between Endline HCC and Diarrhea and Fever in the Previous 3 Months
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Timmermans, M.; van Lier, P.A.; Koot, H.M. The role of stressful events in the development of behavioural and emotional problems from early childhood to late adolescence. Psychol. Med. 2010, 40, 1659–1668. [Google Scholar] [CrossRef] [PubMed]
- Shonkoff, J.P.; Boyce, W.T.; McEwen, B.S. Neuroscience, molecular biology, and the childhood roots of health disparities: Building a new framework for health promotion and disease prevention. JAMA 2009, 301, 2252–2259. [Google Scholar] [CrossRef] [PubMed]
- Danese, A.; McEwen, B.S. Adverse childhood experiences, allostasis, allostatic load, and age-related disease. Physiol. Behav. 2012, 106, 29–39. [Google Scholar] [CrossRef] [PubMed]
- Gunnar, M.; Quevedo, K. The neurobiology of stress and development. Annu. Rev. Psychol. 2007, 58, 145–173. [Google Scholar] [CrossRef] [PubMed]
- Doom, J.R.; Gunnar, M.R. Stress physiology and developmental psychopathology: Past, present, and future. Dev. Psychopathol. 2013, 25, 1359–1373. [Google Scholar] [CrossRef] [PubMed]
- Vanaelst, B.; De Vriendt, T.; Huybrechts, I.; Rinaldi, S.; De Henauw, S. Epidemiological approaches to measure childhood stress. Paediatr. Perinat. Epidemiol. 2012, 26, 280–297. [Google Scholar] [CrossRef] [PubMed]
- Ouellet-Morin, I.; Laurin, M.; Robitaille, M.P.; Brendgen, M.; Lupien, S.J.; Boivin, M.; Vitaro, F. Validation of an adapted procedure to collect hair for cortisol determination in adolescents. Psychoneuroendocrinology 2016, 70, 58–62. [Google Scholar] [CrossRef] [PubMed]
- Russell, E.; Koren, G.; Rieder, M.; Van Uum, S. Hair cortisol as a biological marker of chronic stress: Current status, future directions and unanswered questions. Psychoneuroendocrinology 2012, 37, 589–601. [Google Scholar] [CrossRef]
- Hanrahan, K.; McCarthy, A.M.; Kleiber, C.; Lutgendorf, S.; Tsalikian, E. Strategies for salivary cortisol collection and analysis in research with children. Appl. Nurs. Res. 2006, 19, 95–101. [Google Scholar] [CrossRef]
- Kidd, S.; Midgley, P.; Lone, N.; Wallace, A.M.; Nicol, M.; Smith, J.; McIntosh, N. A re-investigation of saliva collection procedures that highlights the risk of potential positive interference in cortisol immunoassay. Steroids 2009, 74, 666–668. [Google Scholar] [CrossRef]
- Remer, T.; Maser-Gluth, C.; Wudy, S.A. Glucocorticoid measurements in health and disease--metabolic implications and the potential of 24-h urine analyses. Mini Rev. Med. Chem. 2008, 8, 153–170. [Google Scholar] [CrossRef] [PubMed]
- D’Anna-Hernandez, K.L.; Ross, R.G.; Natvig, C.L.; Laudenslager, M.L. Hair cortisol levels as a retrospective marker of hypothalamic-pituitary axis activity throughout pregnancy: Comparison to salivary cortisol. Physiol. Behav. 2011, 104, 348–353. [Google Scholar] [CrossRef] [PubMed]
- Kirschbaum, C.; Tietze, A.; Skoluda, N.; Dettenborn, L. Hair as a retrospective calendar of cortisol production-increased cortisol incorporation into hair in the third trimester of pregnancy. Psychoneuroendocrinology 2009, 34, 32–37. [Google Scholar] [CrossRef]
- Stalder, T.; Steudte, S.; Miller, R.; Skoluda, N.; Dettenborn, L.; Kirschbaum, C. Intraindividual stability of hair cortisol concentrations. Psychoneuroendocrinology 2012, 37, 602–610. [Google Scholar] [CrossRef]
- Short, S.J.; Stalder, T.; Marceau, K.; Entringer, S.; Moog, N.K.; Shirtcliff, E.A.; Wadhwa, P.D.; Buss, C. Correspondence between hair cortisol concentrations and 30-day integrated daily salivary and weekly urinary cortisol measures. Psychoneuroendocrinology 2016, 71, 12–18. [Google Scholar] [CrossRef]
- Grass, J.; Kirschbaum, C.; Miller, R.; Gao, W.; Steudte-Schmiedgen, S.; Stalder, T. Sweat-inducing physiological challenges do not result in acute changes in hair cortisol concentrations. Psychoneuroendocrinology 2015, 53, 108–116. [Google Scholar] [CrossRef]
- Sauve, B.; Koren, G.; Walsh, G.; Tokmakejian, S.; Van Uum, S.H. Measurement of cortisol in human hair as a biomarker of systemic exposure. Clin. Investig. Med. 2007, 30, E183–E191. [Google Scholar] [CrossRef]
- Stalder, T.; Kirschbaum, C. Analysis of cortisol in hair--state of the art and future directions. Brain Behav. Immun. 2012, 26, 1019–1029. [Google Scholar] [CrossRef]
- Vanaelst, B.; Huybrechts, I.; Bammann, K.; Michels, N.; de Vriendt, T.; Vyncke, K.; Sioen, I.; Iacoviello, L.; Gunther, K.; Molnar, D.; et al. Intercorrelations between serum, salivary, and hair cortisol and child-reported estimates of stress in elementary school girls. Psychophysiology 2012, 49, 1072–1081. [Google Scholar]
- Bates, R.; Salsberry, P.; Ford, J. Measuring stress in young children using hair cortisol: The state of the science. Biol. Res. Nurs. 2017, 19, 499–510. [Google Scholar] [CrossRef]
- King, J.C.R. Zinc. In Modern Nutrition in Health and Disease, 11th ed.; Shils, M.E., Shike, M., Ross, A.C., Caballero, B., Cousins, R.J., Eds.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2014; pp. 189–205. [Google Scholar]
- Brown, K.H.; Peerson, J.M.; Baker, S.K.; Hess, S.Y. Preventive zinc supplementation among infants, preschoolers, and older prepubertal children. Food Nutr. Bull. 2009, 30, S12–S40. [Google Scholar] [CrossRef] [PubMed]
- Mayo-Wilson, E.; Junior, J.A.; Imdad, A.; Dean, S.; Chan, X.H.; Chan, E.S.; Jaswal, A.; Bhutta, Z.A. Zinc supplementation for preventing mortality, morbidity, and growth failure in children aged 6 months to 12 years of age. Cochrane Database Syst. Rev. 2014, CD009384. [Google Scholar] [CrossRef]
- Aggarwal, R.; Sentz, J.; Miller, M.A. Role of zinc administration in prevention of childhood diarrhea and respiratory illnesses: A meta-analysis. Pediatrics 2007, 119, 1120–1130. [Google Scholar] [CrossRef]
- Yakoob, M.Y.; Theodoratou, E.; Jabeen, A.; Imdad, A.; Eisele, T.P.; Ferguson, J.; Jhass, A.; Rudan, I.; Campbell, H.; Black, R.E.; et al. Preventive zinc supplementation in developing countries: Impact on mortality and morbidity due to diarrhea, pneumonia and malaria. BMC Public Health 2011, 11 (Suppl. 3), S23. [Google Scholar] [CrossRef] [PubMed]
- De-Regil, L.M.; Suchdev, P.S.; Vist, G.E.; Walleser, S.; Pena-Rosas, J.P. Home fortification of foods with multiple micronutrient powders for health and nutrition in children under two years of age. Cochrane Database Syst. Rev. 2011, CD008959. [Google Scholar] [CrossRef] [PubMed]
- Salam, R.A.; MacPhail, C.; Das, J.K.; Bhutta, Z.A. Effectiveness of micronutrient powders (mnp) in women and children. BMC Public Health 2013, 13 (Suppl. 3), S22. [Google Scholar]
- World Health Organization. Clinical Management of Acute Diarrhoea. Who/Unicef Joint Statement. Report No.: Who/fch/cah/04.7; WHO: Geneva, Switzerland, 2004. [Google Scholar]
- Lazzerini, M.; Ronfani, L. Oral zinc for treating diarrhoea in children. Cochrane Database Syst. Rev. 2013, CD005436. [Google Scholar] [CrossRef]
- Vaghri, Z.; Guhn, M.; Weinberg, J.; Grunau, R.E.; Yu, W.; Hertzman, C. Hair cortisol reflects socio-economic factors and hair zinc in preschoolers. Psychoneuroendocrinology 2013, 38, 331–340. [Google Scholar] [CrossRef]
- Wessells, K.R.; Brown, K.H.; Kounnavong, S.; Barffour, M.A.; Hinnouho, G.; Sayasone, S.; Stephensen, C.B.; Ratsavong, R.; Larson, C.P.; Arnold, C.D.; et al. Comparison of two forms of daily preventive zinc supplementation versus therapeutic zinc supplementation for diarrhea on young children’s physical growth and risk of infection: Study design and rationale for a randomized controlled trial. BMC Nutr. 2018, 4, 39. [Google Scholar] [CrossRef]
- WHO Multicentre Growth Reference Study Group. Who Child Growth Standards: Length/height-For-Age, Weight-For-Age, Weight-For-Length, Weight-For-Height and Body Mass Index-For-Age: Methods and Development; World Health Organization: Geneva, Switzerland, 2006. [Google Scholar]
- Adu-Afarwuah, S.; Lartey, A.; Brown, K.H.; Zlotkin, S.; Briend, A.; Dewey, K.G. Home fortification of complementary foods with micronutrient supplements is well accepted and has positive effects on infant iron status in ghana. Am. J. Clin. Nutr. 2008, 87, 929–938. [Google Scholar] [CrossRef]
- Cogill, B. Anthropometric Indicators Measurement Guide; Food and Nutrition Technical Assistance: Washington, DC, USA, 2003. [Google Scholar]
- Erhardt, J.G.; Estes, J.E.; Pfeiffer, C.M.; Biesalski, H.K.; Craft, N.E. Combined measurement of ferritin, soluble transferrin receptor, retinol binding protein, and c-reactive protein by an inexpensive, sensitive, and simple sandwich enzyme-linked immunosorbent assay technique. J. Nutr. 2004, 134, 3127–3132. [Google Scholar] [CrossRef] [PubMed]
- Hess, S.; Barffour, M.; Hinnouho, G. Lao Zinc Study. Open Science Framework. Available online: https://osf.io/5bq9c (accessed on 6 April 2018).
- Vyas, S.; Kumaranayake, L. Constructing socio-economic status indices: How to use principal components analysis. Health Policy Plan. 2006, 21, 459–468. [Google Scholar] [CrossRef] [PubMed]
- Coates, J. Anne Swindale and Paula Bilinsky. Household Food Insecurity Access Scale (Hfias) for Measurement of Household food Access: Indicator Guide (V. 3); FHI 360/FANTA: Washington, DC, USA, 2007. [Google Scholar]
- World Health Organization. Indicators for Assessing Infant and Young Child Feeding Practices. Part. I: Definition; World Health Organization: Geneva, Switzerland, 2008. [Google Scholar]
- World Health Organization. Indicators for Assessing Infant and Young Child Feeding Practices. Part. II: Measurement; World Health Organization: Geneva, Switzerland, 2010. [Google Scholar]
- Barffour, M.A.; Hinnouho, G.M.; Kounnavong, S.; Wessells, K.R.; Ratsavong, K.; Bounheuang, B.; Chanhthavong, B.; Sitthideth, D.; Sengnam, K.; Arnold, C.D.; et al. Effects of daily preventive zinc supplementation, daily supplementation with a high-zinc, low-iron containing multiple micronutrient powder or therapeutic zinc supplementation for diarrhea, on physical growth, anemia and zinc and iron status in rural Laotian children: A randomized controlled trial. J. Pediatr. 2018. [Google Scholar] [CrossRef]
- Barffour, M.A.; Hinnouho, G.M.; Kounnavong, S.; Wessells, K.R.; Ratsavong, K.; Bounheuang, B.; Chanhthavong, B.; Sitthideth, D.; Khanpaseuth, S.; Arnold, C.D.; et al. Effects of two forms of daily preventive zinc and therapeutic zinc supplementation for diarrhea on diarrhea and acute respiratory tract infections in laotian children. Curr. Dev. Nutr. 2018, in press. [Google Scholar]
- Hess, S.Y.; Wessells, K.R.; Hinnouho, G.M.; Barffour, M.A.; Sanchaisuriya, K.; Arnold, C.D.; Ratsavong, K.; Brown, K.H.; Larson, C.P.; Fucharoen, S.; et al. Associations of genetic hemoglobin disorders and iron status with linear growth and morbidity among young lao children receiving micronutrient powders. Curr. Dev. Nutr. 2018, in press. [Google Scholar]
- Ramakrishnan, U.; Nguyen, P.; Martorell, R. Effects of micronutrients on growth of children under 5 y of age: Meta-analyses of single and multiple nutrient interventions. Am. J. Clin. Nutr. 2009, 89, 191–203. [Google Scholar] [CrossRef] [PubMed]
- Brown, K.H.; Peerson, J.M.; Rivera, J.; Allen, L.H. Effect of supplemental zinc on the growth and serum zinc concentrations of prepubertal children: A meta-analysis of randomized controlled trials. Am. J. Clin. Nutr. 2002, 75, 1062–1071. [Google Scholar] [CrossRef] [PubMed]
- Flom, M.; St John, A.M.; Meyer, J.S.; Tarullo, A.R. Infant hair cortisol: Associations with salivary cortisol and environmental context. Dev. Psychobiol. 2017, 59, 26–38. [Google Scholar] [CrossRef]
- Noppe, G.; Van Rossum, E.F.; Koper, J.W.; Manenschijn, L.; Bruining, G.J.; de Rijke, Y.B.; van den Akker, E.L. Validation and reference ranges of hair cortisol measurement in healthy children. Horm. Res. Paediatr. 2014, 82, 97–102. [Google Scholar] [CrossRef]
- Dettenborn, L.; Tietze, A.; Kirschbaum, C.; Stalder, T. The assessment of cortisol in human hair: Associations with sociodemographic variables and potential confounders. Stress 2012, 15, 578–588. [Google Scholar] [CrossRef]
- Helfrecht, C.; Hagen, E.H.; DeAvila, D.; Bernstein, R.M.; Dira, S.J.; Meehan, C.L. Dheas patterning across childhood in three sub-saharan populations: Associations with age, sex, ethnicity, and cortisol. Am. J. Hum. Biol. 2018, 30, e23090. [Google Scholar] [CrossRef] [PubMed]
- Gerber, M.; Brand, S.; Lindwall, M.; Elliot, C.; Kalak, N.; Herrmann, C.; Puhse, U.; Jonsdottir, I.H. Concerns regarding hair cortisol as a biomarker of chronic stress in exercise and sport science. J. Sports Sci. Med. 2012, 11, 571–581. [Google Scholar] [PubMed]
- Dowlati, Y.; Herrmann, N.; Swardfager, W.; Thomson, S.; Oh, P.I.; Van Uum, S.; Koren, G.; Lanctot, K.L. Relationship between hair cortisol concentrations and depressive symptoms in patients with coronary artery disease. Neuropsychiatr. Dis. Treat. 2010, 6, 393–400. [Google Scholar] [PubMed]
- Skoluda, N.; Dettenborn, L.; Stalder, T.; Kirschbaum, C. Elevated hair cortisol concentrations in endurance athletes. Psychoneuroendocrinology 2012, 37, 611–617. [Google Scholar] [CrossRef] [PubMed]
- Vliegenthart, J.; Noppe, G.; van Rossum, E.F.; Koper, J.W.; Raat, H.; van den Akker, E.L. Socioeconomic status in children is associated with hair cortisol levels as a biological measure of chronic stress. Psychoneuroendocrinology 2016, 65, 9–14. [Google Scholar] [CrossRef] [PubMed]
- Rippe, R.C.; Noppe, G.; Windhorst, D.A.; Tiemeier, H.; van Rossum, E.F.; Jaddoe, V.W.; Verhulst, F.C.; Bakermans-Kranenburg, M.J.; van, I.M.H.; van den Akker, E.L. Splitting hair for cortisol? Associations of socio-economic status, ethnicity, hair color, gender and other child characteristics with hair cortisol and cortisone. Psychoneuroendocrinology 2016, 66, 56–64. [Google Scholar] [CrossRef]
- Palmer, F.B.; Anand, K.J.; Graff, J.C.; Murphy, L.E.; Qu, Y.; Volgyi, E.; Rovnaghi, C.R.; Moore, A.; Tran, Q.T.; Tylavsky, F.A. Early adversity, socioemotional development, and stress in urban 1-year-old children. J. Pediatr. 2013, 163, 1733–1739. [Google Scholar] [CrossRef] [PubMed]
- Karlen, J.; Frostell, A.; Theodorsson, E.; Faresjo, T.; Ludvigsson, J. Maternal influence on child hpa axis: A prospective study of cortisol levels in hair. Pediatrics 2013, 132, e1333–e1340. [Google Scholar] [CrossRef]
- Karlen, J.; Ludvigsson, J.; Hedmark, M.; Faresjo, A.; Theodorsson, E.; Faresjo, T. Early psychosocial exposures, hair cortisol levels, and disease risk. Pediatrics 2015, 135, e1450–e1457. [Google Scholar] [CrossRef]
- Larsen, S.C.; Fahrenkrug, J.; Olsen, N.J.; Heitmann, B.L. Association between hair cortisol concentration and adiposity measures among children and parents from the “healthy start” study. PLoS ONE 2016, 11, e0163639. [Google Scholar] [CrossRef]
- Wester, V.L.; van Rossum, E.F. Clinical applications of cortisol measurements in hair. Eur. J. Endocrinol. 2015, 173, M1–M10. [Google Scholar] [CrossRef] [PubMed]
- Cooper, G.A.; Kronstrand, R.; Kintz, P. Society of hair testing guidelines for drug testing in hair. Forensic Sci. Int. 2012, 218, 20–24. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Xie, Q.; Gao, W.; Xu, Y.; Wang, S.; Deng, H.; Lu, Z. Time course of cortisol loss in hair segments under immersion in hot water. Clin. Chim. Acta 2012, 413, 434–440. [Google Scholar] [CrossRef] [PubMed]
- Gow, R.; Thomson, S.; Rieder, M.; Van Uum, S.; Koren, G. An assessment of cortisol analysis in hair and its clinical applications. Forensic Sci. Int. 2010, 196, 32–37. [Google Scholar] [CrossRef] [PubMed]
- Manenschijn, L.; Koper, J.W.; Lamberts, S.W.; van Rossum, E.F. Evaluation of a method to measure long term cortisol levels. Steroids 2011, 76, 1032–1036. [Google Scholar] [CrossRef] [PubMed]
- Abbeddou, S.; Hess, S.Y.; Yakes Jimenez, E.; Some, J.W.; Vosti, S.A.; Guissou, R.M.; Ouedraogo, J.B.; Brown, K.H. Comparison of methods to assess adherence to small-quantity lipid-based nutrient supplements (sq-lns) and dispersible tablets among young burkinabe children participating in a community-based intervention trial. Matern. Child. Nutr 2015, 11 (Suppl. 4), 90–104. [Google Scholar] [CrossRef]
| Characteristics | All (n = 512) | PZ (n = 131) | MNP (n = 127) | TZ (n = 127) | CONTROL (n = 127) |
|---|---|---|---|---|---|
| Age, mo | 15.6 ± 5.0 | 15.3 ± 5.2 | 15.9 ± 5.0 | 15.8 ± 4.8 | 15.4 ± 5.0 |
| Gender, female | 283 (55.3) | 79 (60.3) | 64 (50.4) | 73 (57.5) | 67 (52.8) |
| Breastfeeding | 298 (64.4) | 72 (63.7) | 70 (62.5) | 78 (65.6) | 78 (65.6) |
| 2 Adequate dietary diversity | 167 (35.9) | 39 (34.5) | 39 (34.5) | 38 (31.7) | 51 (42.9) |
| 3 Minimum meal frequency | 258 (55.7) | 63 (55.8) | 60 (53.6) | 65 (54.6) | 70 (58.8) |
| Consumption of iron rich foods | 371 (79.8) | 83 (73.5) | 93 (82.3) | 101 (84.2) | 94 (79.0) |
| Child anthropometric measures | |||||
| Length, cm | 73.8 ± 5.4 | 73.3 ± 5.6 | 74.0 ± 5.2 | 73.6 ± 5.2 | 74.5 ± 5.6 |
| Weight, kg | 8.5 ± 1.3 | 8.3 ± 1.3 | 8.5 ± 1.3 | 8.5 ± 1.3 | 8.8 ± 1.3 |
| MUAC, cm | 13.9 ± 1.0 | 13.8 ± 1.0 | 13.9 ± 1.0 | 14.0 ± 1.0 | 14.1 ± 1.0 |
| LAZ | −0.71 ± 1.08 | −1.79 ± 1.10 | −1.79 ± 1.10 | −1.86 ± 1.07 | −1.41 ± 1.07 |
| WAZ | −1.42 ± 0.99 | −1.52 ± 1.00 | −1.52 ± 1.04 | −1.51 ± 0.96 | −1.14 ± 0.93 |
| WLZ | −0.74 ± 0.86 | −0.81 ± 0.89 | −0.82 ± 0.86 | −0.76 ± 0.87 | −0.57 ± 0.82 |
| Stunting | 195 (38.2) | 52 (40.0) | 52 (40.9) | 54 (42.5) | 37 (29.1) |
| Wasting | 32 (6.3) | 11 (8.5) | 12 (9.5) | 7 (5.5) | 2 (1.6) |
| Underweight | 139 (27.2) | 40 (30.8) | 41 (32.3) | 38 (29.9) | 20 (15.8) |
| Elevated CRP (>5 mg/L) | 55 (11.3) | 16 (12.6) | 9 (7.44) | 14 (11.5) | 16 (13.9) |
| Elevated AGP (>1 g/L) | 97 (20.0) | 27 (21.3) | 24 (19.8) | 22 (18.0) | 24 (20.9) |
| Hair cortisol concentrations, pg/mg | 21.7 (13.8–35.1) | 27.0 (14.7–38.3) | 19.4 (12.5–29.7) | 21.0 (13.4–34.4) | 21.5 (14.0–34.0) |
| Maternal education, primary or lower | 267 (53.3) | 72 (56.7) | 67 (52.8) | 64 (51.6) | 64 (52.0) |
| Maternal BMI, kg/m2 | 22.0 ± 3.3 | 21.8 ± 3.1 | 22.2 ± 3.2 | 21.9 ± 3.4 | 22.0 ± 3.5 |
| HFIAS | |||||
| Food secure | 131 (25.9) | 35 (27.3) | 35 (28.2) | 25 (19.7) | 36 (28.6) |
| Mildly food insecure | 148 (29.3) | 35 (27.3) | 37 (29.8) | 39 (30.7) | 37 (29.4) |
| Moderately food insecure | 141 (27.9) | 26 (20.3) | 36 (29.0) | 42 (33.1) | 37 (29.4) |
| Severely food insecure | 85 (16.8) | 32 (25.0) | 16 (12.9) | 21 (16.5) | 16 (12.7) |
| n | PZ | MNP | TZ | CONTROL | P | |
|---|---|---|---|---|---|---|
| 2 HCC at endline, pg/mg | ||||||
| Minimally adjusted model | 512 | 14.3 (12.5; 16.4) | 13.3 (11.6; 15.3) | 16.1 (14.0; 18.4) | 13.6 (11.8; 15.6) | 0.231 |
| Fully adjusted model | 499 | 13.6 (11.8; 15.6) | 13.1 (11.4; 15.1) | 15.9 (13.8; 18.3) | 13.7 (11.9; 15.9) | 0.260 |
| 3 Change in HCC, pg/mg | ||||||
| Minimally adjusted model | 512 | −8.9 (−11.2; −6.6) | −7.8 (−10.2; −5.4) | −5.0 (−7.4; −2.6) | −7.8 (−10.2; −5.4) | 0.121 |
| Fully adjusted model | 499 | −9.4 (−11.7; −7.0) | −7.7 (−10.1; −5.4) | −4.6 (−7.0; −2.3) | −7.2 (−9.7; −4.8) | 0.078 |
| Variables | Percent Change 2 (95% CI) | p |
|---|---|---|
| Child | ||
| Weight, kg | −6.7 (−13.9; 1.3) | 0.094 |
| Length, cm | −2.4 (−5.0; 0.3) | 0.083 |
| MUAC, cm | −6.5 (−13.8; 1.4) | 0.106 |
| LAZ | −5.6 (−12.4; 1.8) | 0.132 |
| WAZ | −6.0 (−13.2; 1.9) | 0.130 |
| WLZ | −3.0 (−11.6; 6.3) | 0.512 |
| Stunting | −6.7 (−20.7; 9.9) | 0.408 |
| Wasting | 7.0 (−22.8; 48.3) | 0.683 |
| Underweight | 3.3 (−13.5; 23.5) | 0.717 |
| Low MUAC (MUAC ≤ 12.5 cm) | 32.4 (−4.8; 84.1) | 0.095 |
| Breastfeeding | 19.0 (−1.5; 43.8) | 0.071 |
| Iron rich foods | −23.9 (−38.8; −5.4) | 0.014 |
| Adequate dietary diversity | −11.4 (−25.9; 6.0) | 0.186 |
| Minimum meal frequency | −10.1 (−24.5; 7.0) | 0.231 |
| CRP, mg/L | 5.4 (−0.2; 11.4) | 0.058 |
| Elevated CRP (CRP > 5 mg/L) | 19.6 (−7.5; 54.7) | 0.172 |
| AGP, g/L | 30.3 (12.4; 51.1) | <0.001 |
| Elevated AGP (AGP > 1 g/L) | 39.2 (13.2; 70.6) | 0.002 |
| RBP, mg/dL | −14.3 (−36.5; 15.8) | 0.316 |
| Ferritin, µg/L | 6.9 (−4.0; 19.1) | 0.221 |
| Low ferritin (pF < 12 µg/L) | −4.7 (−24.5; 20.3) | 0.687 |
| sTfR, mg/L | 6.9 (−11.7; 29.4) | 0.495 |
| High sTfR (sTfR > 8.3 mg/L) | 23.5 (4.1; 46.7) | 0.016 |
| Maternal | ||
| Age | −1.0 (−2.3; 0.3) | 0.135 |
| Education | −4.3 (−18.5; 12.3) | 0.589 |
| Marital status (couple) | 25.0 (−23.0; 103.0) | 0.367 |
| BMI | 1.8 (−0.8; 4.4) | 0.172 |
| Household | ||
| SES index | −2.9 (−6.9; 1.3) | 0.175 |
| HFIAS | ||
| Food secure | Ref | |
| Mildly food insecure | −5.6 (−24.0; 17.3) | 0.604 |
| Moderately food insecure | 0.9 (−19.4; 26.3) | 0.939 |
| Severely food insecure | −7.0 (−28.1; 20.2) | 0.578 |
| Variables | n (%) | Percent Change 2 (95% CI) | p |
|---|---|---|---|
| Reported at least 1 episode of diarrhea | 238 (46.5) | 3.1 (−10.4; 18.7) | 0.666 |
| Reported at least 1 episode of fever | 382 (74.6) | 5.4 (−10.3; 23.9) | 0.520 |
| Measured at least 1 episode of fever | 60 (11.7) | 6.7 (−14.2; 32.6) | 0.560 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hinnouho, G.-M.; Bernstein, R.M.; Barffour, M.A.; Arnold, C.D.; Wessells, K.R.; Ratsavong, K.; Bounheuang, B.; Kounnavong, S.; Hess, S.Y. Impact of Two Forms of Daily Preventive Zinc or Therapeutic Zinc Supplementation for Diarrhea on Hair Cortisol Concentrations Among Rural Laotian Children: A Randomized Controlled Trial. Nutrients 2019, 11, 47. https://doi.org/10.3390/nu11010047
Hinnouho G-M, Bernstein RM, Barffour MA, Arnold CD, Wessells KR, Ratsavong K, Bounheuang B, Kounnavong S, Hess SY. Impact of Two Forms of Daily Preventive Zinc or Therapeutic Zinc Supplementation for Diarrhea on Hair Cortisol Concentrations Among Rural Laotian Children: A Randomized Controlled Trial. Nutrients. 2019; 11(1):47. https://doi.org/10.3390/nu11010047
Chicago/Turabian StyleHinnouho, Guy-Marino, Robin M. Bernstein, Maxwell A. Barffour, Charles D. Arnold, K. Ryan Wessells, Kethmany Ratsavong, Bangone Bounheuang, Sengchanh Kounnavong, and Sonja Y. Hess. 2019. "Impact of Two Forms of Daily Preventive Zinc or Therapeutic Zinc Supplementation for Diarrhea on Hair Cortisol Concentrations Among Rural Laotian Children: A Randomized Controlled Trial" Nutrients 11, no. 1: 47. https://doi.org/10.3390/nu11010047
APA StyleHinnouho, G.-M., Bernstein, R. M., Barffour, M. A., Arnold, C. D., Wessells, K. R., Ratsavong, K., Bounheuang, B., Kounnavong, S., & Hess, S. Y. (2019). Impact of Two Forms of Daily Preventive Zinc or Therapeutic Zinc Supplementation for Diarrhea on Hair Cortisol Concentrations Among Rural Laotian Children: A Randomized Controlled Trial. Nutrients, 11(1), 47. https://doi.org/10.3390/nu11010047





