A Guide to Applying the Sex-Gender Perspective to Nutritional Genomics
Abstract
1. Importance of Sex/Gender Differences in Precision Medicine and Precision Nutrition
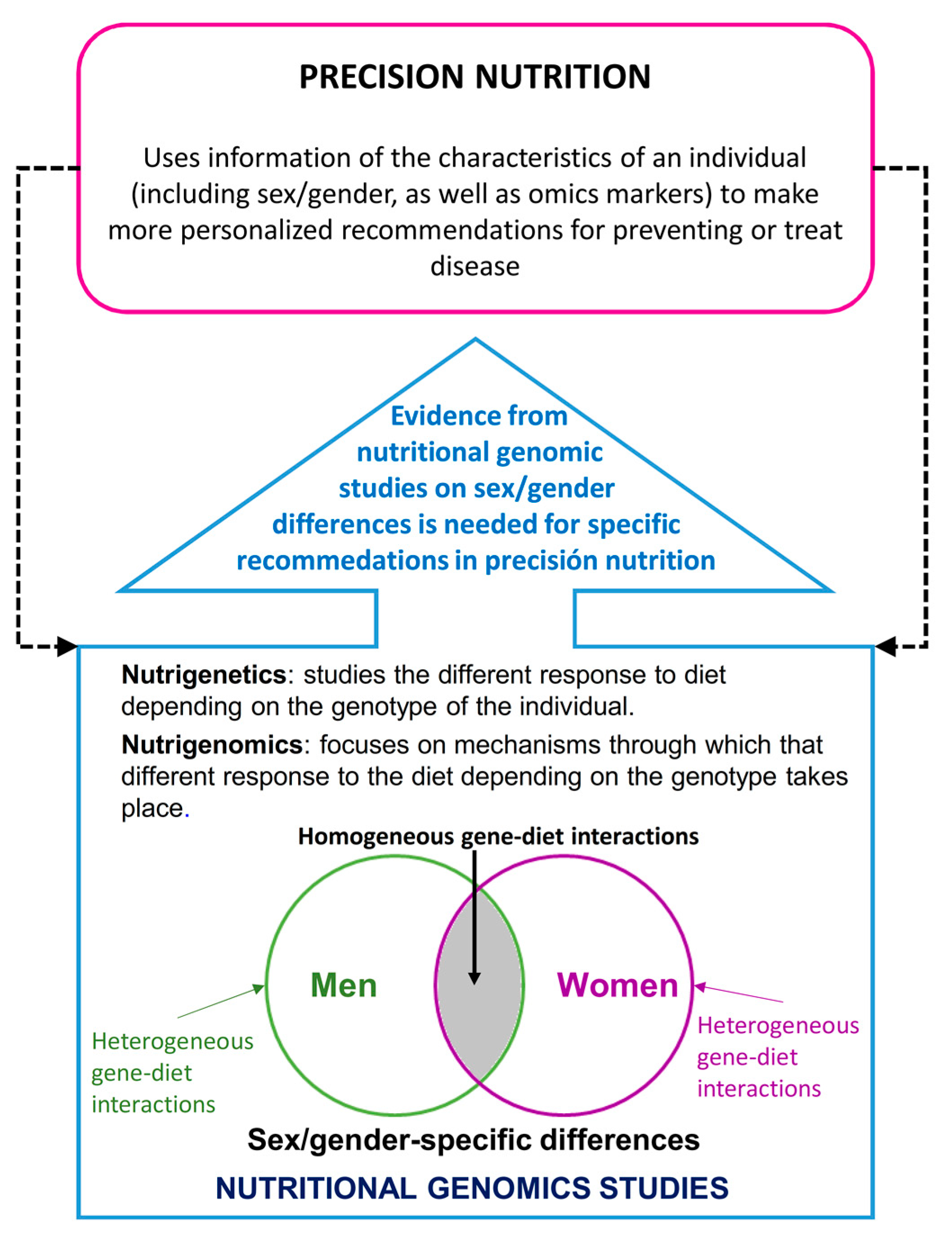
1.1. Precision Nutrition and Sex/Gender Differences
1.2. Nutritional Genomics and Sex/Gender Differences
2. General Context of Differences Per Sex and Gender Perspective in Biomedical Research and Nutritional Genomics
3. Difference Between Sex and Gender
3.1. Sex and Gender Identification in Studies on Nutritional Genomics
3.1.1. Measuring Sex in Nutritional Genomics Studies
3.1.2. Gender Measurement in Nutritional Genomics Studies
4. Measuring the Difference in Outcomes Depending on Sex/Gender
5. Gender Perspective in Genotype Analyses in Nutritional Genomics Studies
Sex-specific SNPs Associated with Disease, Current State of the Genome-Wide Association Studies
Methodology/Limitations
6. Specific Sex/Gender Gene-Diet Interactions
7. Improvement in Diet Measurement and Other Related Variables Using Gender Perspective in Nutritional Genomics
8. Recommendations on the Design, Analysis, and Presentation of Results in Nutritional Genomics Studies with a Gender Perspective
9. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Ramaswami, R.; Bayer, R.; Galea, S. Precision Medicine from a Public Health Perspective. Ann. Rev. Public Health 2018, 39, 153–168. [Google Scholar] [CrossRef] [PubMed]
- Collins, F.S.; Varmus, H. A New Initiative on Precision Medicine. New Engl. J. Med. 2015, 372, 793–795. [Google Scholar] [CrossRef] [PubMed]
- Ordovas, J.M.; Ferguson, L.R.; Tai, E.S.; Mathers, J.C. Personalised nutrition and health. BMJ 2018, 361, bmj.k2173. [Google Scholar] [CrossRef] [PubMed]
- De Toro-Martín, J.; Arsenault, B.; Després, J.-P.; Vohl, M.-C. Precision Nutrition: A Review of Personalized Nutritional Approaches for the Prevention and Management of Metabolic Syndrome. Nutrients 2017, 9, 913. [Google Scholar] [CrossRef]
- De Roos, B.; Brennan, L. Personalised Interventions-A Precision Approach for the Next Generation of Dietary Intervention Studies. Nutrients 2017, 9, 847. [Google Scholar] [CrossRef] [PubMed]
- Corella, D.; Coltell, O.; Macian, F.; Ordovas, J.M. Advances in Understanding the Molecular Basis of the Mediterranean Diet Effect. Annu. Rev. Food Sci. Technol. 2018, 9, 227–249. [Google Scholar] [CrossRef] [PubMed]
- Ordovas, J.M.; Corella, D. Nutritional genomics. Annu. Rev. Genomics Hum. Genet. 2004, 5, 71–118. [Google Scholar] [CrossRef]
- Whittle, K.L.; Inhorn, M.C. Rethinking Difference: A Feminist Reframing of Gender/Race/Class for the Improvement of Women’s Health Research. Inter. J. Health Serv. 2001, 31, 147–165. [Google Scholar] [CrossRef]
- Vidaver, R.M.; Lafleur, B.; Tong, C.; Bradshaw, R.; Marts, S.A. Women Subjects in NIH-Funded Clinical Research Literature: Lack of Progress in Both Representation and Analysis by Sex. J. Women’s Health Gender-Based Med. 2000, 9, 495–504. [Google Scholar] [CrossRef]
- Kim, M.-Y.; Kim, K.; Hong, C.H.; Lee, S.Y.; Jung, Y.-S. Sex Differences in Cardiovascular Risk Factors for Dementia. Biomol. Ther. 2018, 26, 521–532. [Google Scholar] [CrossRef]
- Adigun, R.O.; Boler, A.N.; Mankad, R. Disparities in Cardiac Care of Women: Current Data and Possible Solutions. Curr. Treat. Options in Cardiovasc. Med. 2018, 20. [Google Scholar] [CrossRef]
- Sobhani, K.; Nieves Castro, D.K.; Fu, Q.; Gottlieb, R.A.; Van Eyk, J.E.; Noel Bairey Merz, C. Sex differences in ischemic heart disease and heart failure biomarkers. Biol. Sex. Differ. 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- Daniels, L.B.; Maisel, A.S. Cardiovascular biomarkers and sex: The case for women. Nat. Rev. Cardiol. 2015, 12, 588–596. [Google Scholar] [CrossRef] [PubMed]
- Palmisano, B.T.; Zhu, L.; Eckel, R.H.; Stafford, J.M. Sex differences in lipid and lipoprotein metabolism. Mol. Metabol. 2018, 15, 45–55. [Google Scholar] [CrossRef] [PubMed]
- Ohkuma, T.; Peters, S.A.E.; Woodward, M. Sex differences in the association between diabetes and cancer: A systematic review and meta-analysis of 121 cohorts including 20 million individuals and one million events. Diabetologia 2018, 61, 2140–2154. [Google Scholar] [CrossRef] [PubMed]
- Schiebinger, L.; Stefanick, M.L. Gender Matters in Biological Research and Medical Practice. J. Am. Coll. Cardiol. 2016, 67, 136–138. [Google Scholar] [CrossRef]
- Christianson, M.; Alex, L.; Wiklund, A.F.; Hammarström, A.; Lundman, B. Sex and Gender Traps and Springboards: A Focus Group Study Among Gender Researchers in Medicine and Health Sciences. Health Care for Women Inter. 2012, 33, 739–755. [Google Scholar] [CrossRef]
- Marino, M.; Masella, R.; Bulzomi, P.; Campesi, I.; Malorni, W.; Franconi, F. Nutrition and human health from a sex–gender perspective. Mol. Aspects of Med. 2011, 32, 1–70. [Google Scholar] [CrossRef]
- Doull, M.; Runnels, V.E.; Tudiver, S.; Boscoe, M. Appraising the Evidence: Applying Sex- and Gender-Based Analysis (SGBA) to Cochrane Systematic Reviews on Cardiovascular Diseases. J. Women’s Health 2010, 19, 997–1003. [Google Scholar] [CrossRef]
- Collins, F.S.; Tabak, L.A. Policy: NIH plans to enhance reproducibility. Nature 2014, 505, 612–613. [Google Scholar] [CrossRef]
- Consideration of Sex as a Biological Variable in NIH-funded Research. Available online: https://nccih.nih.gov/node/10486 (accessed on 7 November 2018).
- Tannenbaum, C.; Schwarz, J.M.; Clayton, J.A.; De Vries, G.J.; Sullivan, C. Evaluating sex as a biological variable in preclinical research: The devil in the details. Biol. Sex. Differ. 2016, 7. [Google Scholar] [CrossRef] [PubMed]
- Clayton, J.A. Studying both sexes: A guiding principle for biomedicine. FASEB J. 2016, 30, 519–524. [Google Scholar] [CrossRef] [PubMed]
- Mauvais-Jarvis, F.; Arnold, A.P.; Reue, K. A Guide for the Design of Pre-clinical Studies on Sex Differences in Metabolism. Cell. Metabol. 2017, 25, 1216–1230. [Google Scholar] [CrossRef] [PubMed]
- Klinge, I. Gender perspectives in European research. Pharmacol. Res. 2008, 58, 183–189. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, K.; Rieder, A. Methodologic and ethical ramifications of sex and gender differences in public health research. Gend. Med. 2007, 4, S96–S105. [Google Scholar] [CrossRef]
- Ordovas, J.M. Gender, a significant factor in the cross talk between genes, environment, and health. Gend. Med. 2007, 4, S111–S122. [Google Scholar] [CrossRef]
- Horizon 2020–European Commission. Available online: https://ec.europa.eu/programmes/horizon2020/en/official-documents (accessed on 7 November 2018).
- Gender, Sex and Health Research Guide: A Tool for CIHR Applicants|IGAR Tool. Available online: http://igar-tool.gender-net.eu/en/reference/gender-sex-and-health-research-guide-a-tool-for-cihr-applicants (accessed on 7 November 2018).
- WHO|WHO Gender Policy: Integrating Gender Perspectives in the Work of WHO. Available online: http://www.who.int/gender-equity-rights/knowledge/a78322/en/ (accessed on 7 November 2018).
- What is Gender Mainstreaming. Available online: https://eige.europa.eu/gender-mainstreaming/what-is-gender-mainstreaming (accessed on 7 November 2018).
- WHO|Gender. Available online: http://www.who.int/gender-equity-rights/understanding/gender-definition/en/ (accessed on 7 November 2018).
- Government of Canada, C.I. of H.R. What is Gender? What is Sex? CIHR. Available online: http://www.cihr-irsc.gc.ca/e/48642.html (accessed on 7 November 2018).
- Sex & Gender|Office of Research on Women’s Health. Available online: https://orwh.od.nih.gov/sex-gender (accessed on 7 November 2018).
- Concepts and Definitions. Available online: https://eige.europa.eu/gender-mainstreaming/concepts-and-definitions (accessed on 7 November 2018).
- Attorney General’s Department. Australian Government Guidelines on the Recognition of Sex and Gender. Available online: https://www.ag.gov.au/Publications/Pages/AustralianGovernmentGuidelinesontheRecognitionofSexandGender.aspx (accessed on 7 November 2018).
- Heidari, S.; Babor, T.F.; De Castro, P.; Tort, S.; Curno, M. Sex and Gender Equity in Research: Rationale for the SAGER guidelines and recommended use. Res. Integr. Peer Rev. 2016, 1. [Google Scholar] [CrossRef]
- Clayton, J.A.; Tannenbaum, C. Reporting Sex, Gender, or Both in Clinical Research? JAMA 2016, 316, 1863. [Google Scholar] [CrossRef]
- Francès, F.; Portolés, O.; González, J.I.; Coltell, O.; Verdú, F.; Castelló, A.; Corella, D. Amelogenin test: From forensics to quality control in clinical and biochemical genomics. Clin. Chim. Acta 2007, 386, 53–56. [Google Scholar] [CrossRef]
- Tobias, E.S.; McElreavey, K. Next Generation Sequencing for Disorders of Sex Development. In Endocrine Development; Hiort, O., Ahmed, S.F., Eds.; S. KARGER AG: Basel, Switzerland, 2014; Volume 27, pp. 53–62. ISBN 9783318025583. [Google Scholar]
- Hammarström, A.; Annandale, E. A Conceptual Muddle: An Empirical Analysis of the Use of ‘Sex’ and ‘Gender’ in ‘Gender-Specific Medicine’ Journals. PLoS ONE 2012, 7, e34193. [Google Scholar] [CrossRef]
- Alex, L.; Fjellman Wiklund, A.; Lundman, B.; Christianson, M.; Hammarström, A. Beyond a Dichotomous View of the Concepts of ‘Sex’ and ‘Gender’ Focus Group Discussions among Gender Researchers at a Medical Faculty. PLoS ONE 2012, 7, e50275. [Google Scholar] [CrossRef] [PubMed]
- On behalf of the COST Action BM1303 Working Group 1; Cools, M.; Nordenström, A.; Robeva, R.; Hall, J.; Westerveld, P.; Flück, C.; Köhler, B.; Berra, M.; Springer, A.; et al. Caring for individuals with a difference of sex development (DSD): A Consensus Statement. Nat. Rev. Endocrinol. 2018, 14, 415–429. [Google Scholar] [CrossRef] [PubMed]
- Day, S.; Mason, R.; Lagosky, S.; Rochon, P.A. Integrating and evaluating sex and gender in health research. Health Res. Policy Syst. 2016, 14. [Google Scholar] [CrossRef] [PubMed]
- Johnson, J.L.; Greaves, L.; Repta, R. Better science with sex and gender: Facilitating the use of a sex and gender-based analysis in health research. Inter. J. Equity in Health 2009, 8, 14. [Google Scholar] [CrossRef]
- Day, S.; Mason, R.; Tannenbaum, C.; Rochon, P.A. Essential metrics for assessing sex & gender integration in health research proposals involving human participants. PLoS ONE 2017, 12, e0182812. [Google Scholar] [CrossRef]
- Pelletier, R.; Khan, N.A.; Cox, J.; Daskalopoulou, S.S.; Eisenberg, M.J.; Bacon, S.L.; Lavoie, K.L.; Daskupta, K.; Rabi, D.; Humphries, K.H.; et al. Sex Versus Gender-Related Characteristics: Which Predicts Outcome After Acute Coronary Syndrome in the Young? J. Am. Coll. Cardiol. 2016, 67, 127–135. [Google Scholar] [CrossRef]
- Diemer, E.W.; Grant, J.D.; Munn-Chernoff, M.A.; Patterson, D.A.; Duncan, A.E. Gender Identity, Sexual Orientation, and Eating-Related Pathology in a National Sample of College Students. J. Adolesc. Health 2015, 57, 144–149. [Google Scholar] [CrossRef]
- Baggio, G.; Corsini, A.; Floreani, A.; Giannini, S.; Zagonel, V. Gender medicine: A task for the third millennium. Clin. Chem. Laborat. Med. 2013, 51. [Google Scholar] [CrossRef]
- Clayton, J.A.; Davis, A.F. Sex/Gender Disparities and Women’s Eye Health. Curr. Eye Res. 2015, 40, 102–109. [Google Scholar] [CrossRef]
- Doyal, L.; Das-Bhaumik, R.G. Sex, gender and blindness: A new framework for equity. BMJ Open Ophthalmol. 2018, 3, e000135. [Google Scholar] [CrossRef]
- Reusch, J.E.B.; Kumar, T.R.; Regensteiner, J.G.; Zeitler, P.S.; Conference Participants; Arany, Z.; Bairey Merz, C.N.; Barrett-Connor, E.; Boyle, K.; Brown, L.; et al. Identifying the Critical Gaps in Research on Sex Differences in Metabolism Across the Life Span. Endocrinology 2018, 159, 9–19. [Google Scholar] [CrossRef]
- Corella, D.; Coltell, O.; Mattingley, G.; Sorlí, J.V.; Ordovas, J.M. Utilizing nutritional genomics to tailor diets for the prevention of cardiovascular disease: A guide for upcoming studies and implementations. Exp. Rev. Mol. Diagn. 2017, 17, 495–513. [Google Scholar] [CrossRef]
- Corella, D.; Coltell, O.; Sorlí, J.; Estruch, R.; Quiles, L.; Martínez-González, M.; Salas-Salvadó, J.; Castañer, O.; Arós, F.; Ortega-Calvo, M.; et al. Polymorphism of the Transcription Factor 7-Like 2 Gene (TCF7L2) Interacts with Obesity on Type-2 Diabetes in the PREDIMED Study Emphasizing the Heterogeneity of Genetic Variants in Type-2 Diabetes Risk Prediction: Time for Obesity-Specific Genetic Risk Scores. Nutrients 2016, 8, 793. [Google Scholar] [CrossRef]
- Snell, D.M.; Turner, J.M.A. Sex Chromosome Effects on Male–Female Differences in Mammals. Curr. Biol. 2018, 28, R1313–R1324. [Google Scholar] [CrossRef] [PubMed]
- Shen, J.J.; Wang, T.-Y.; Yang, W. Regulatory and evolutionary signatures of sex-biased genes on both the X chromosome and the autosomes. Biol. Sex. Differ. 2017, 8. [Google Scholar] [CrossRef]
- BIOS Consortium; Luijk, R.; Wu, H.; Ward-Caviness, C.K.; Hannon, E.; Carnero-Montoro, E.; Min, J.L.; Mandaviya, P.; Müller-Nurasyid, M.; Mei, H.; et al. Autosomal genetic variation is associated with DNA methylation in regions variably escaping X-chromosome inactivation. Nat. Commun. 2018, 9. [Google Scholar] [CrossRef]
- Mauvais-Jarvis, F. Sex differences in metabolic homeostasis, diabetes, and obesity. Biol. Sex. Differ. 2015, 6. [Google Scholar] [CrossRef]
- Chen, X.; McClusky, R.; Chen, J.; Beaven, S.W.; Tontonoz, P.; Arnold, A.P.; Reue, K. The Number of X Chromosomes Causes Sex Differences in Adiposity in Mice. PLoS Genet. 2012, 8, e1002709. [Google Scholar] [CrossRef]
- Link, J.C.; Chen, X.; Arnold, A.P.; Reue, K. Metabolic impact of sex chromosomes. Adipocyte 2013, 2, 74–79. [Google Scholar] [CrossRef] [PubMed]
- Accounting for sex in the genome. Nat. Med. 2017, 23, 1243. [CrossRef] [PubMed]
- Wise, A.L.; Gyi, L.; Manolio, T.A. eXclusion: Toward Integrating the X Chromosome in Genome-wide Association Analyses. Am. J. Human Genet. 2013, 92, 643–647. [Google Scholar] [CrossRef] [PubMed]
- Tukiainen, T.; Pirinen, M.; Sarin, A.-P.; Ladenvall, C.; Kettunen, J.; Lehtimäki, T.; Lokki, M.-L.; Perola, M.; Sinisalo, J.; Vlachopoulou, E.; et al. Chromosome X-Wide Association Study Identifies Loci for Fasting Insulin and Height and Evidence for Incomplete Dosage Compensation. PLoS Genet. 2014, 10, e1004127. [Google Scholar] [CrossRef] [PubMed]
- Russo, P.; Siani, A.; Miller, M.A.; Karanam, S.; Esposito, T.; Gianfrancesco, F.; Barba, G.; Lauria, F.; Strazzullo, P.; Cappuccio, F.P. Genetic Variants of Y Chromosome Are Associated With a Protective Lipid Profile in Black Men. Arterioscler. Thromb. Vasc. Biol. 2008, 28, 1569–1574. [Google Scholar] [CrossRef] [PubMed]
- Estrada, K.; Styrkarsdottir, U.; Evangelou, E.; Hsu, Y.-H.; Duncan, E.L.; Ntzani, E.E.; Oei, L.; Albagha, O.M.E.; Amin, N.; Kemp, J.P.; et al. Genome-wide meta-analysis identifies 56 bone mineral density loci and reveals 14 loci associated with risk of fracture. Nat. Genet. 2012, 44, 491–501. [Google Scholar] [CrossRef] [PubMed]
- Ohlsson, C.; Wallaschofski, H.; Lunetta, K.L.; Stolk, L.; Perry, J.R.B.; Koster, A.; Petersen, A.-K.; Eriksson, J.; Lehtimäki, T.; Huhtaniemi, I.T.; et al. Genetic Determinants of Serum Testosterone Concentrations in Men. PLoS Genet. 2011, 7, e1002313. [Google Scholar] [CrossRef] [PubMed]
- Winkler, T.W.; Justice, A.E.; Graff, M.; Barata, L.; Feitosa, M.F.; Chu, S.; Czajkowski, J.; Esko, T.; Fall, T.; Kilpeläinen, T.O.; et al. The Influence of Age and Sex on Genetic Associations with Adult Body Size and Shape: A Large-Scale Genome-Wide Interaction Study. PLoS Genet. 2015, 11, e1005378. [Google Scholar] [CrossRef]
- Randall, J.C.; Winkler, T.W.; Kutalik, Z.; Berndt, S.I.; Jackson, A.U.; Monda, K.L.; Kilpeläinen, T.O.; Esko, T.; Mägi, R.; Li, S.; et al. Sex-stratified Genome-wide Association Studies Including 270,000 Individuals Show Sexual Dimorphism in Genetic Loci for Anthropometric Traits. PLoS Genet. 2013, 9, e1003500. [Google Scholar] [CrossRef]
- Heid, I.M.; Jackson, A.U.; Randall, J.C.; Winkler, T.W.; Qi, L.; Steinthorsdottir, V.; Thorleifsson, G.; Zillikens, M.C.; Speliotes, E.K.; Mägi, R.; et al. Meta-analysis identifies 13 new loci associated with waist-hip ratio and reveals sexual dimorphism in the genetic basis of fat distribution. Nat. Genet. 2010, 42, 949–960. [Google Scholar] [CrossRef]
- Porcu, E.; Medici, M.; Pistis, G.; Volpato, C.B.; Wilson, S.G.; Cappola, A.R.; Bos, S.D.; Deelen, J.; Den Heijer, M.; Freathy, R.M.; et al. A Meta-Analysis of Thyroid-Related Traits Reveals Novel Loci and Gender-Specific Differences in the Regulation of Thyroid Function. PLoS Genet. 2013, 9, e1003266. [Google Scholar] [CrossRef]
- Kitamoto, A.; Kitamoto, T.; So, R.; Matsuo, T.; Nakata, Y.; Hyogo, H.; Ochi, H.; Nakamura, T.; Kamohara, S.; Miyatake, N.; et al. ADIPOQ polymorphisms are associated with insulin resistance in Japanese women. Endocr. J. 2015, 62, 513–521. [Google Scholar] [CrossRef]
- Mittelstrass, K.; Ried, J.S.; Yu, Z.; Krumsiek, J.; Gieger, C.; Prehn, C.; Roemisch-Margl, W.; Polonikov, A.; Peters, A.; Theis, F.J.; et al. Discovery of Sexual Dimorphisms in Metabolic and Genetic Biomarkers. PLoS Genet. 2011, 7, e1002215. [Google Scholar] [CrossRef] [PubMed]
- Teslovich, T.M.; Musunuru, K.; Smith, A.V.; Edmondson, A.C.; Stylianou, I.M.; Koseki, M.; Pirruccello, J.P.; Ripatti, S.; Chasman, D.I.; Willer, C.J.; et al. Biological, clinical and population relevance of 95 loci for blood lipids. Nature 2010, 466, 707–713. [Google Scholar] [CrossRef] [PubMed]
- Aung, L.H.H.; Yin, R.-X.; Wu, D.-F.; Wang, W.; Wu, J.-Z.; Liu, C.-W. Sex-specific Association of the Zinc Finger Protein 259 rs2075290 Polymorphism and Serum Lipid Levels. Inter. J. Med. Sci. 2014, 11, 471–478. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.; Kwon, D.Y.; Park, J. The impacts of the interaction of genetic variation, CYP11β2 and NEDD4L, with sodium intake on pediatric obesity with gender difference: A 3-year panel study. Int. J. Obes. (Lond) 2017, 41, 542–550. [Google Scholar] [CrossRef]
- Nilsson, L.; Olsson, A.H.; Isomaa, B.; Groop, L.; Billig, H.; Ling, C. A common variant near the PRL gene is associated with increased adiposity in males. Mol. Genet. Metab. 2011, 102, 78–81. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Jiang, F.; Zhang, R.; Tang, S.; Chen, M.; Peng, D.; Yan, J.; Wang, T.; Wang, S.; Bao, Y.; et al. Serum uric acid levels are associated with polymorphisms in the SLC2A9, SF1, and GCKR genes in a Chinese population. Acta Pharmacol. Sin. 2014, 35, 1421–1427. [Google Scholar] [CrossRef] [PubMed]
- Sung, Y.J.; Pérusse, L.; Sarzynski, M.A.; Fornage, M.; Sidney, S.; Sternfeld, B.; Rice, T.; Terry, J.G.; Jacobs, D.R.; Katzmarzyk, P.; et al. Genome-wide association studies suggest sex-specific loci associated with abdominal and visceral fat. Int. J. Obes. (Lond) 2016, 40, 662–674. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Yang, J.; Li, Y.; Ma, X.; Li, R. Sex chromosome abnormalities and psychiatric diseases. Oncotarget 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Ng, H.K.T.; Li, J.; Liu, Q.; Huang, H. Detecting associated single-nucleotide polymorphisms on the X chromosome in case control genome-wide association studies. Stat. Methods in Med. Res. 2017, 26, 567–582. [Google Scholar] [CrossRef]
- Clayton, D. Testing for association on the X. chromosome. Biostatistics 2008, 9, 593–600. [Google Scholar] [CrossRef]
- Thornton, T.; Zhang, Q.; Cai, X.; Ober, C.; McPeek, M.S. XM: Association Testing on the X-Chromosome in Case-Control Samples With Related Individuals: X-Chromosome Association in Pedigrees. Genet. Epidemiol. 2012, 36, 438–450. [Google Scholar] [CrossRef] [PubMed]
- Purcell, S.; Neale, B.; Todd-Brown, K.; Thomas, L.; Ferreira, M.A.R.; Bender, D.; Maller, J.; Sklar, P.; De Bakker, P.I.W.; Daly, M.J.; et al. PLINK: A Tool Set for Whole-Genome Association and Population-Based Linkage Analyses. Am. J. Human Genet. 2007, 81, 559–575. [Google Scholar] [CrossRef] [PubMed]
- O’Neil, A.; Scovelle, A.J.; Milner, A.J.; Kavanagh, A. Gender/Sex as a Social Determinant of Cardiovascular Risk. Circulation 2018, 137, 854–864. [Google Scholar] [CrossRef] [PubMed]
- Ordovas, J.M.; Corella, D.; Cupples, L.A.; Demissie, S.; Kelleher, A.; Coltell, O.; Wilson, P.W.; Schaefer, E.J.; Tucker, K. Polyunsaturated fatty acids modulate the effects of the APOA1 G-A polymorphism on HDL-cholesterol concentrations in a sex-specific manner: The Framingham Study. Am. J. Clin. Nutr. 2002, 75, 38–46. [Google Scholar] [CrossRef] [PubMed]
- Ribalta, J. Additive Effects of the PPAR, APOE, and FABP-2 Genes in Increasing Daylong Triglycerides of Normolipidemic Women to Concentrations Comparable to Those in Men. Clin. Chem. 2005, 51, 864–871. [Google Scholar] [CrossRef]
- Méplan, C.; Crosley, L.K.; Nicol, F.; Beckett, G.J.; Howie, A.F.; Hill, K.E.; Horgan, G.; Mathers, J.C.; Arthur, J.R.; Hesketh, J.E. Genetic polymorphisms in the human selenoprotein P gene determine the response of selenoprotein markers to selenium supplementation in a gender-specific manner (the SELGEN study). FASEB J. 2007, 21, 3063–3074. [Google Scholar] [CrossRef] [PubMed]
- Hu, M.; Li, Z.; Fang, D.Z. A high carbohydrate diet induces the beneficial effect of the CC genotype of hepatic lipase C-514T polymorphism on the apoB100/apoAI ratio only in young Chinese males. Scand. J. Clin. Laborat. Investig. 2012, 72, 563–569. [Google Scholar] [CrossRef]
- Shatwan, I.; Minihane, A.-M.; Williams, C.; Lovegrove, J.; Jackson, K.; Vimaleswaran, K. Impact of Lipoprotein Lipase Gene Polymorphism, S447X, on Postprandial Triacylglycerol and Glucose Response to Sequential Meal Ingestion. Inter. J. Mol. Sci. 2016, 17, 397. [Google Scholar] [CrossRef]
- Jacobo-Albavera, L.; Posadas-Romero, C.; Vargas-Alarcón, G.; Romero-Hidalgo, S.; Posadas-Sánchez, R.; González-Salazar, M.D.C.; Carnevale, A.; Canizales-Quinteros, S.; Medina-Urrutia, A.; Antúnez-Argüelles, E.; et al. Dietary fat and carbohydrate modulate the effect of the ATP-binding cassette A1 (ABCA1) R230C variant on metabolic risk parameters in premenopausal women from the Genetics of Atherosclerotic Disease (GEA) Study. Nutr. Metabol. 2015, 12. [Google Scholar] [CrossRef]
- Zhang, Z.; Gong, R.R.; Du, J.; Xiao, L.Y.; Duan, W.; Zhou, X.D.; Fang, D.Z. Associations of the SREBP-1c gene polymorphism with gender-specific changes in serum lipids induced by a high-carbohydrate diet in healthy Chinese youth. Appl. Physiol. Nutr. Metabol. 2011, 36, 226–232. [Google Scholar] [CrossRef]
- Barragán, R.; Coltell, O.; Portolés, O.; Asensio, E.; Sorlí, J.; Ortega-Azorín, C.; González, J.; Sáiz, C.; Fernández-Carrión, R.; Ordovas, J.; et al. Bitter, Sweet, Salty, Sour and Umami Taste Perception Decreases with Age: Sex-Specific Analysis, Modulation by Genetic Variants and Taste-Preference Associations in 18 to 80 Year-Old Subjects. Nutrients 2018, 10, 1539. [Google Scholar] [CrossRef] [PubMed]
- Lauritzen, L.; Sørensen, L.B.; Harsløf, L.B.; Ritz, C.; Stark, K.D.; Astrup, A.; Dyssegaard, C.B.; Egelund, N.; Michaelsen, K.F.; Damsgaard, C.T. Mendelian randomization shows sex-specific associations between long-chain PUFA–related genotypes and cognitive performance in Danish schoolchildren. Am. J. Clin. Nutr. 2017, 106, 88–95. [Google Scholar] [CrossRef] [PubMed]
- Obregón, A.M.; Valladares, M.; Goldfield, G. Association of the dopamine D2 receptor rs1800497 polymorphism and eating behavior in Chilean children. Nutrition 2017, 35, 139–145. [Google Scholar] [CrossRef] [PubMed]
- Roumans, N.J.T.; Vink, R.G.; Gielen, M.; Zeegers, M.P.; Holst, C.; Wang, P.; Astrup, A.; Saris, W.H.; Valsesia, A.; Hager, J.; et al. Variation in extracellular matrix genes is associated with weight regain after weight loss in a sex-specific manner. Genes Nutr. 2015, 10. [Google Scholar] [CrossRef] [PubMed]
- Ericson, U.; Rukh, G.; Stojkovic, I.; Sonestedt, E.; Gullberg, B.; Wirfält, E.; Wallström, P.; Orho-Melander, M. Sex-specific interactions between the IRS1 polymorphism and intakes of carbohydrates and fat on incident type 2 diabetes. Am. J. Clin. Nutr. 2013, 97, 208–216. [Google Scholar] [CrossRef]
- Dedoussis, G.V.; Manios, Y.; Kourlaba, G.; Kanoni, S.; Lagou, V.; Butler, J.; Papoutsakis, C.; Scott, R.A.; Yannakoulia, M.; Pitsiladis, Y.P.; et al. An age-dependent diet-modified effect of the PPARγ Pro12Ala polymorphism in children. Metabolism 2011, 60, 467–473. [Google Scholar] [CrossRef]
- Nettleton, J.A.; Volcik, K.A.; Hoogeveen, R.C.; Boerwinkle, E. Carbohydrate intake modifies associations between ANGPTL4[E40K] genotype and HDL-cholesterol concentrations in White men from the Atherosclerosis Risk in Communities (ARIC) study. Atherosclerosis 2009, 203, 214–220. [Google Scholar] [CrossRef]
- Gastaldi, M.; Dizière, S.; Defoort, C.; Portugal, H.; Lairon, D.; Darmon, M.; Planells, R. Sex-specific association of fatty acid binding protein 2 and microsomal triacylglycerol transfer protein variants with response to dietary lipid changes in the 3-mo Medi-RIVAGE primary intervention study. Am. J. Clin. Nutr. 2007, 86, 1633–1641. [Google Scholar] [CrossRef]
- Alkhalaf, A.; Landman, G.W.D.; Van Hateren, K.J.J.; Groenier, K.H.; Mooyaart, A.L.; De Heer, E.; Gans, R.O.B.; Navis, G.J.; Bakker, S.J.L.; Kleefstra, N.; et al. Sex specific association between carnosinase gene CNDP1 and cardiovascular mortality in patients with type 2 diabetes (ZODIAC-22). J. Nephrol. 2015, 28, 201–207. [Google Scholar] [CrossRef]
- Van Dijk, S.J.; Zhou, J.; Peters, T.J.; Buckley, M.; Sutcliffe, B.; Oytam, Y.; Gibson, R.A.; McPhee, A.; Yelland, L.N.; Makrides, M.; et al. Effect of prenatal DHA supplementation on the infant epigenome: Results from a randomized controlled trial. Clin. Epigenet. 2016, 8. [Google Scholar] [CrossRef]
- Gonzalez-Nahm, S.; Mendez, M.; Robinson, W.; Murphy, S.K.; Hoyo, C.; Hogan, V.; Rowley, D. Low maternal adherence to a Mediterranean diet is associated with increase in methylation at the MEG3-IG differentially methylated region in female infants. Environ. Epigenet. 2017, 3. [Google Scholar] [CrossRef] [PubMed]
- Borgo, F.; Garbossa, S.; Riva, A.; Severgnini, M.; Luigiano, C.; Benetti, A.; Pontiroli, A.E.; Morace, G.; Borghi, E. Body Mass Index and Sex Affect Diverse Microbial Niches within the Gut. Front. Microbiol. 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- Bolnick, D.I.; Snowberg, L.K.; Hirsch, P.E.; Lauber, C.L.; Org, E.; Parks, B.; Lusis, A.J.; Knight, R.; Caporaso, J.G.; Svanbäck, R. Individual diet has sex-dependent effects on vertebrate gut microbiota. Nat. Commun. 2014, 5. [Google Scholar] [CrossRef] [PubMed]
- Naska, A.; Lagiou, A.; Lagiou, P. Dietary assessment methods in epidemiological research: Current state of the art and future prospects. F1000Research 2017, 6, 926. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Kang, M.; Song, W.O.; Shim, J.E.; Paik, H.Y. Gender analysis in the development and validation of FFQ: A systematic review. Br. J. Nutr. 2016, 115, 666–671. [Google Scholar] [CrossRef]
- Zenun Franco, R.; Fallaize, R.; Lovegrove, J.A.; Hwang, F. Online dietary intake assessment using a graphical food frequency app (eNutri): Usability metrics from the EatWellUK study. PLoS ONE 2018, 13, e0202006. [Google Scholar] [CrossRef] [PubMed]
- Blasco-Blasco, M.; Ruiz-Cantero, M.T.; Juárez-Herrera y Cairo, L.A.; Jovaní, V.; Pascual, E. Sex and Gender Interactions in the Lives of Patients with Spondyloarthritis in Spain: A Quantitative-qualitative Study. J. Rheumatol. 2017, 44, 1429–1435. [Google Scholar] [CrossRef] [PubMed]
- Maliqueo, M.; Cruz, G.; Espina, C.; Contreras, I.; García, M.; Echiburú, B.; Crisosto, N. Obesity during pregnancy affects sex steroid concentrations depending on fetal gender. Inter. J. Obes. 2017, 41, 1636–1645. [Google Scholar] [CrossRef] [PubMed]
- Mauvais-Jarvis, F. Menopause, Estrogens, and Glucose Homeostasis in Women. In Sex and Gender Factors Affecting Metabolic Homeostasis, Diabetes and Obesity; Mauvais-Jarvis, F., Ed.; Springer International Publishing: Cham, Switzerland, 2017; Volume 1043, pp. 217–225. ISBN 9783319701776. [Google Scholar]
- Ellingjord-Dale, M.; Vos, L.; Tretli, S.; Hofvind, S.; dos-Santos-Silva, I.; Ursin, G. Parity, hormones and breast cancer subtypes–results from a large nested case-control study in a national screening program. Breast Cancer Res. 2017, 19. [Google Scholar] [CrossRef]
| Sex Definition | Gender Definition | Reference |
|---|---|---|
| The different biological and physiological characteristics of males and females, such as reproductive organs, chromosomes, hormones, etc. | Refers to the socially constructed characteristics of women and men–such as norms, roles and relationships of and between groups of women and men. It varies from society to society and can be changed—including how they should interact with others of the same or opposite sex within households, communities and work places. | WHO [32] |
| Refers to a set of biological attributes in humans and animals. It is primarily associated with physical and physiological features including chromosomes, gene expression, hormone levels and function, and reproductive/sexual anatomy. Sex is usually categorized as female or male but there is variation in the biological attributes that comprise sex and how those attributes are expressed. | Refers to the socially constructed roles, behaviours, expressions and identities of girls, women, boys, men, and gender diverse people. It influences how people perceive themselves and each other, how they act and interact, and the distribution of power and resources in society. Gender is usually conceptualized as a binary (girl/woman and boy/man) yet there is considerable diversity in how individuals and groups understand, experience, and express it. | CIHR [33] |
| Refers to biological differences between females and males, including chromosomes, sex organs, and endogenous hormonal profiles. | Refers to socially constructed and enacted roles and behaviors which occur in a historical and cultural context and vary across societies and over time. All individuals act in many ways that fulfill the gender expectations of their society. With continuous interaction between sex and gender, health is determined by both biology and the expression of gender. | NIH [34] |
| Biological and physiological characteristics that define humans as female or male. | Social attributes and opportunities associated with being female and male and to the relationships between women and men and girls and boys, as well as to the relations between women and those between men. | EIGE [35] |
| Refers to the chromosomal, gonadal and anatomical characteristics associated with biological sex. | It is a part of a person’s personal and social identity. It refers to the way a person feels, presents and is recognized within the community. A person’s gender may be reflected in outward social markers, including their name, outward appearance, mannerisms and dress. | Australian Government [36] |
| Author | SNP/Loci/Gene | Trait | Sex-Specific | Chromosome |
|---|---|---|---|---|
| Tukiainen et al., 2014 [63] | ITM2A, FGF16 ATRX/MAGT1 (Xq23) (no associated gene) | Height Fasting insulin levels | Female (height) Male (fasting insulin) | X |
| Russo Paola et al., 2008 [64] | TBL1Y, USP9Y | Lower triglycerides (TG) and higher HDL | Male | Y |
| Estrada et al., 2012 [65] | Rs5934507 (Xp22.31, closest gene FAM9B/KAL1 | Bone metabolism | Male | X |
| Ohlsson et al., 2011 [66] | rs5934505 in the X Chromosome near FAM9B | Lower testosterone levels | Male | X |
| Winkler et al., 2015 [67] | 44 loci | Changes in body mass index (BMI), measures of body size, or waist-to-hip ratio | Female (28) and male (5) and 11 opposite effects | Autosome chromosomes |
| Randall et al., 2013 [68] | GRB14/COBLL1, LYPLAL1/SLC30A10, VEGFA, ADAMTS9, MAP3K1, HSD17B4, PPARG | Anthropometrics (specifically waist phenotypes) | Female | 2,1,6,3,5,5,3 |
| Heid et al., 2010 [69] | RSPO3, VEGFA, GRB14, LYPLA1, ITPR2-SSPN, ADAMTS9 | Waist-to-hip ratio | Female | 6,6,2,1,12,3 |
| Porcu et al., 2013 [70] | PDE8B, PDE10A, MAF/LOC440389, LPCT2/CAPNS2, NETO1/FBXO15 | Affect thyroid stimulating hormone (TSH) and Free thyroxine (FT4) levels | Male (first 4) and Female (last 1) | 5,6,16,16,18 |
| Kitamoto et al., 2015 [71] | ADIPOQ (2 SNPs out of 7) | Decrease serum adiponectin levels which are correlated with decreased homeostatic model assessment-insulin resistance (HOMA-IR) | Female | 3 |
| Mittelstrass et al., 2011 [72] | CSP1 | Sex-specific association with glycine | Female and male | 2 |
| Teslovich et al., 2010 [73] | KLF14, ABCA8 | Sex-specific association with TG, LDL-C | Female | 7,17 |
| Aung et al., 2014 [74] | ZNF259 | Many lipid associations | Male and Female | 11 |
| Lee, Kwon, & Park, 2017 [75] | CYP11β2 | Salt sensitive gene leads to obesity from salt-intake? | Female | 8 |
| Nilsson et al., 2011 [76] | PRL | Sex-specific obesity | Male | 6 |
| Sun et al., 2014 [77] | GCKR, SLC2A9, SF1 | Associated with serum uric acid concentrations | Male | 2,4,9 |
| Sung et al., 2016 [78] | BBS9, ADCY8, KCNK9 MLLT10/DNAJC1/EBLN1 | Visceral fat Subcutaneous fat | Women | 10,8 |
| Author | Study Characteristics and Aims | Findings |
|---|---|---|
| Ordovas JM et al., 2002 [85] | Examined whether dietary fat modulates the association between the APOA1 G-A polymorphism and HDL-in men and women from the Framingham Study. | We found a significant gene-diet interaction associated with the APOA1 G-A polymorphism. In women carriers of the A allele, higher polyunsaturated fatty acid (PUFA) intakes were associated with higher HDL-cholesterol concentrations, whereas the opposite effect was observed in G/G women. |
| Ribalta J et al., 2005 [86] | Identification of gender-specific genetic influences on fasting and postprandial TG concentrations under typical living conditions in healthy, lean, normolipidemics. | An adverse combination of common alleles of the FABP-2, APOE, and PPARgamma genes in women increases their TGs to values comparable to those seen in men. Although this influence is not appreciable when studying fasting plasma TGs, it becomes apparent with use of a more sensitive index. |
| Méplan C et al., 2007 [87] | Analysis of polymorphisms in the selenoprotein P gene determine the response of selenoprotein markers to selenium (Se) supplementation in a gender-specific manner (the SELGEN study). | Two common functional SNPs within the human SePP gene that may predict behavior of biomarkers of Se status and response to supplementation and thus susceptibility to disease. Both SNPs and gender were associated with differences in scavenger glutathione peroxidase 3 (GPx3) activity and other markers. |
| Hu M et al., 2012 [88] | Intervention with a high-carbohydrate (high-CHO) diet for a short-term and investigation of the interactions with the hepatic lipase G-250A promoter polymorphism to affect the ratios of plasma lipids and apolipoproteins. | The high-CHO diet induced the positive effects on the lipid ratios in general, only except the TG/HDL-C ratio in females. Noticeably, the decreased apoB100/apoAI ratio was associated with the A allele of hepatic lipase G-250A polymorphism only in males. |
| Shatwan IM et al., 2016 [89] | Analysis of the influence of two commonly studied LPL polymorphisms (rs320, HindIII; rs328, S447X) on postprandial lipaemia, in 261 participants using a standard sequential meal challenge. | Novel finding on the effect of the LPL S447X polymorphism on the postprandial glucose and gender-specific impact of the polymorphism on fasting and postprandial TAG concentrations in response to sequential meal challenge in healthy participants. The sex-specific results were only detected in men. |
| Jacobo-Albavera L et al., 2015 [90] | Analysis of whether gender, menopausal status and macronutrient proportions of diet modulate the effect of the (ABCA1) R230C variant on various metabolic parameters. | First study reporting a gender-specific interaction between ABCA1/R230C variant and dietary carbohydrate and fat percentages affecting Visceral adipose tissue (VAT) / subcutaneous adipose tissue (SAT) ratio, gamma-glutamyl transpeptidase (GGT), alkaline phosphatase (ALP), adiponectin levels and HOMA index. This study confirmed a previously reported gender-specific ABCA1-diet interaction affecting HDL-C levels. |
| Zhang Z et al., 2011 [91] | Investigation of the association between the sterol regulatory element-binding protein-1c gene (SREBP-1c) rs2297508 and the changes in lipid profiles in a high-carbohydrate and low-fat diet in a Chinese population. | The C allele of the rs2297508 polymorphism was associated with a retardation of the increases in serum triacylglycerol, serum insulin, and HOMA-IR in females and with the elevated serum HDL-C in males after the high-carbohydrate/low fat (high-CHO/LF) diet. |
| Barragán R et al., 2018 [92] | Analyzed the age influence on the intensity rating of the five basic tastes: sweet, salty, bitter, sour and umami (separately and jointly in a "total taste score") and their modulation by sex and genetics in a relatively healthy population. | Women perceived taste significantly more intense than men (p = 1.4 × 10−8 for total taste score). Significant associations were, found between a higher perception of sour taste and a higher preference for it in women. In contrast, the higher perception of sweet was significantly associated with a higher preference for bitter in both, men and women. The TAS2R38-rs713598 SNP had a significant interaction with sex. |
| Lauritzen L et al., 2017 [93] | Mendelian randomization study to explore whether SNPs in fatty acid desaturase (FADS) and elongase (ELOVL) genes were associated with school performance in a sex-specific manner. | Associations between rs1535 minor allele homozygosity and rs174448 major allele carriage and improved performance in boys but not in girls was found, thereby counteracting existing sex differences. |
| Obregón AM et al., 2017 [94] | Analysis of the association between the DRD2 rs1800497 polymorphism and eating behavior in Chilean children. | In the sex-specific analysis, the TaqI A1 allele was associated with higher scores on Satiety Responsiveness and Emotional Undereating subscales in obese girls, and higher scores of Enjoyment of Food subscale in boys. |
| Roumans NJ et al., 2015 [95] | Investigation of whether genetic variation in extracellular matrix (ECM) -related genes is associated with weight regain among participants of the European DiOGenes study. | Variants of ECM genes were associated with weight regain after weight loss in a sex-specific manner. |
| Ericson U et al., 2013 [96] | Interaction analysis between IRS1 rs2943641 and macronutrient intakes on incident T2D and percentage body fat in the Malmö Diet and Cancer cohort. | The IRS1 rs2943641 interacted with carbohydrate and fat intakes on incident T2D in a sex-specific manner. A protective association between the rs2943641 T allele and T2D was restricted to women with low carbohydrate intake and to men with low fat intake. |
| Dedoussis GV et al., 2011 [97] | Investigation of the age-related association between the Pro12Ala variant (rs1801282) and diet in obesity-related traits in children. | Adiposity in children was influenced by the Pro12Ala polymorphism in a sex-specific and age-dependent manner. |
| Nettleton JA et al., 2009 [98] | Analysis of whether dietary macronutrient intake modified associations between ANGPTL4[E40K] variation and TG and HDL-C in White men and women from the Atherosclerosis Risk in Communities study. | In men, but not women, the inverse association between carbohydrate and HDL-C was stronger in A allele carriers (beta+/-S.E. -1.80+/-0.54) than non-carriers (beta+/-S.E. -0.54+/-0.11, p(interaction) = 0.04 in men and 0.69 in women; p 3-way interaction = 0.14). |
| Gastaldi M et al., 2007 [99] | Analysis of the effect of fatty acid binding protein 2 (FABP2) Ala54Thr and microsomal triacylglycerol transfer protein (MTTP) -493G/T variations on plasma lipid markers, at baseline and on the response to the 3-mo Medi-RIVAGE study. | These 2 polymorphic loci are thus differently associated with the baseline lipid markers as well as with the response to nutritional recommendations, but both presented a marked sex-specific profile, with the response to diet being particularly efficient in men homozygous for the MTTP -493T allele. |
| Alkhalaf A et al., 2015 [100] | This study investigated whether 5L-5L in the CNDP1 gene was associated with mortality and progression of renal function loss and to what extent this effect was modified by sex. | The association between CNDP1 and cardiovascular mortality was sex-specific, with a higher risk in women with 5L-5L genotype. CNDP1 was not associated with all-cause mortality or change in epidermal growth factor receptor (EGFR). |
| van Dijk SJ et al., 2016 [101] | A double-blind randomized placebo-controlled trial in pregnant women to test whether a defined nutritional exposure in utero, docosahexaenoic acid (DHA), could alter the infant epigenome. | Maternal DHA supplementation during the second half of pregnancy had small effects on DNA methylation of infants. However, the number of differential methylated loci (DMRs) at birth was greater in males (127 DMRs) than in females (72 DMRs) separately, indicating a gender-specific effect. |
| Gonzalez-Nahm S et al., 2017 [102] | Association between maternal adherence to a Mediterranean diet pattern during pregnancy and infant DNA methylation at birth. | There was an association between overall diet pattern and methylation at the 9 DMRs analyzed and suggests that maternal diet can have a sex-specific impact on infant DNA methylation at specific imprinted DMRs. |
| Borgo F et al., 2018 [103] | Comparison of the gut microbiota is in at least two separate microbial populations, the lumen-associated (LAM) and the mucosa-associated microbiota (MAM). Next generation sequencing was used). | LAM and MAM communities seemed to be influenced by different host factors, such as diet and sex. Female MAM was enriched in Actinobacteria (with an increased trend of the genus Bifidobacterium), and a significant depletion in Veillonellaceae. |
| Bolnick DI et al., 2014 [104] | Analysis of the factors related to the composition of gut microbiota, mainly the host genotyping. | The results indicated that microbiota composition depends on interactions between host diet and sex within populations of wild and laboratory fish, laboratory mice and humans. The diet-microbiota associations were sex dependent. Further experimental work confirmed that microbiota was different males versus females and suggested that therapies to treat dysbiosis might have sex-specific effects |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Corella, D.; Coltell, O.; Portolés, O.; Sotos-Prieto, M.; Fernández-Carrión, R.; Ramirez-Sabio, J.B.; Zanón-Moreno, V.; Mattei, J.; Sorlí, J.V.; Ordovas, J.M. A Guide to Applying the Sex-Gender Perspective to Nutritional Genomics. Nutrients 2019, 11, 4. https://doi.org/10.3390/nu11010004
Corella D, Coltell O, Portolés O, Sotos-Prieto M, Fernández-Carrión R, Ramirez-Sabio JB, Zanón-Moreno V, Mattei J, Sorlí JV, Ordovas JM. A Guide to Applying the Sex-Gender Perspective to Nutritional Genomics. Nutrients. 2019; 11(1):4. https://doi.org/10.3390/nu11010004
Chicago/Turabian StyleCorella, Dolores, Oscar Coltell, Olga Portolés, Mercedes Sotos-Prieto, Rebeca Fernández-Carrión, Judith B. Ramirez-Sabio, Vicente Zanón-Moreno, Josiemer Mattei, José V. Sorlí, and Jose M. Ordovas. 2019. "A Guide to Applying the Sex-Gender Perspective to Nutritional Genomics" Nutrients 11, no. 1: 4. https://doi.org/10.3390/nu11010004
APA StyleCorella, D., Coltell, O., Portolés, O., Sotos-Prieto, M., Fernández-Carrión, R., Ramirez-Sabio, J. B., Zanón-Moreno, V., Mattei, J., Sorlí, J. V., & Ordovas, J. M. (2019). A Guide to Applying the Sex-Gender Perspective to Nutritional Genomics. Nutrients, 11(1), 4. https://doi.org/10.3390/nu11010004








