The Effect of Lutein on Eye and Extra-Eye Health
Abstract
1. Introduction
2. Characteristics of Lutein
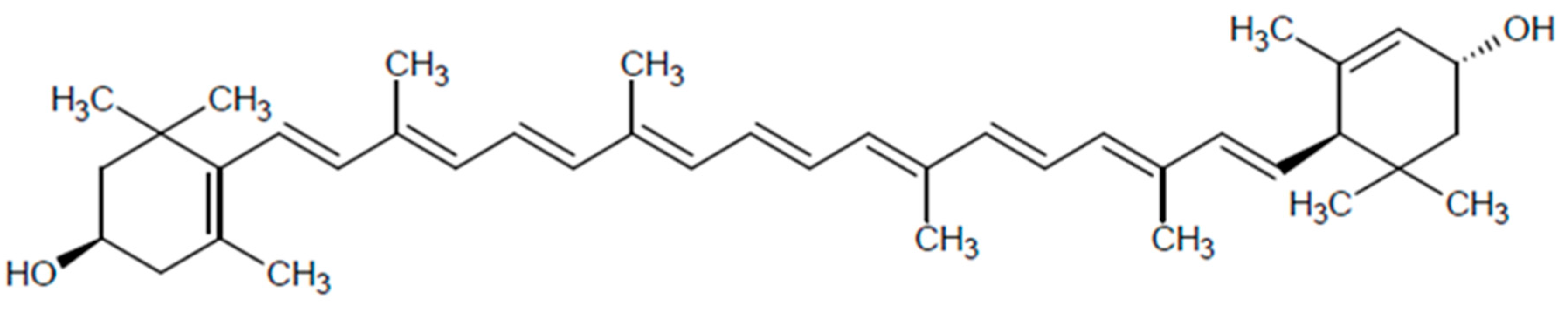
2.1. Structure and Distribution
2.2. Absorption and Metabolism
2.3. Mechanisms of Action
3. Dietary Lutein Intake
4. Lutein and Eye Disturbances
4.1. Age-Related Macular Degeneration
4.2. Cataract
4.3. Other Eye Diseases
5. Extra-Eye Actions
5.1. Cognitive Function
5.2. Cardiovascular Health
5.3. Cancer Risk
5.4. Other Systemic and Metabolic Effects
6. Lutein Safety and Toxicity
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Bjørklund, G.; Chirumbolo, S. Role of oxidative stress and antioxidants in daily nutrition and human health. Nutrition 2017, 33, 311–321. [Google Scholar] [CrossRef] [PubMed]
- Chrysohoou, C.; Panagiotakos, D.B.; Pitsavos, C.; Das, U.N.; Stefanadis, C. Adherence to the Mediterranean diet attenuates inflammation and coagulation process in healthy adults: The ATTICA Study. J. Am. Coll. Cardiol. 2004, 44, 152–158. [Google Scholar] [CrossRef] [PubMed]
- Di Daniele, N.; Noce, A.; Vidiri, M.F.; Moriconi, E.; Marrone, G.; Annicchiarico-Petruzzelli, M.; D’Urso, G.; Tesauro, M.; Rovella, V.; De Lorenzo, A. Impact of Mediterranean diet on metabolic syndrome, cancer and longevity. Oncotarget 2017, 8, 8947–8979. [Google Scholar] [CrossRef] [PubMed]
- Perry, A.; Rasmussen, H.; Johnson, E.J. Xanthophyll [lutein, zeaxanthin] content in fruits, vegetables and corn and egg products. J. Food Compos. Anal. 2009, 22, 9–15. [Google Scholar] [CrossRef]
- Fiedor, J.; Burda, K. Potential role of carotenoids as antioxidants in human health and disease. Nutrients 2014, 6, 466–488. [Google Scholar] [CrossRef] [PubMed]
- Young, A.J.; Lowe, G.M. Antioxidant and prooxidant properties of carotenoids. Arch. Biochem. Biophys. 2001, 385, 20–27. [Google Scholar] [CrossRef] [PubMed]
- Tapiero, H.; Townsend, D.M.; Tew, K.D. The role of carotenoids in the prevention of human pathologies. Biomed. Pharmacother. 2004, 58, 100–110. [Google Scholar] [CrossRef] [PubMed]
- Lima, V.C.; Rosen, R.B.; Farah, M. Macular pigment in retinal health and disease. Int. J. Retina Vitreous 2016, 2, 19. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.R.; Nakanishi, K.; Itagaki, Y.; Sparrow, J.R. Photooxidation of A2-PE, a photoreceptor outer segment fluorophore, and protection by lutein and zeaxanthin. Exp. Eye Res. 2006, 82, 828–839. [Google Scholar] [CrossRef] [PubMed]
- Seddon, J.M.; Ajani, U.A.; Sperduto, R.D.; Hiller, R.; Blair, N.; Burton, T.C.; Farber, M.D.; Gragoudas, E.S.; Haller, J.; Miller, D.T.; et al. Eye Disease Case-Control Study Group. Dietary carotenoids, vitamins A, C, and E, and advanced age-related macular degeneration. JAMA 1994, 272, 1413–1420. [Google Scholar] [CrossRef] [PubMed]
- Eroglu, A.; Harrison, E.H. Carotenoid metabolism in mammals, including man: Formation, occurrence, and function of apocarotenoids. J. Lipid Res. 2013, 54, 1719–1730. [Google Scholar] [CrossRef] [PubMed]
- Richer, S.; Stiles, W.; Statkute, L.; Pulido, J.; Frankowski, J.; Rudy, D.; Pei, K.; Tsipursky, M.; Nyland, J. Double-masked, placebo-controlled, randomized trial of lutein and antioxidant supplementation in the intervention of atrophic age-related macular degeneration: The Veterans LAST study (Lutein Antioxidant Supplementation Trial). Optometry 2004, 75, 216–230. [Google Scholar] [CrossRef]
- Parisi, V.; Tedeschi, M.; Gallinaro, G.; Varano, M.; Saviano, S.; Piermarocchi, S.; CARMIS Study Group. Carotenoids and antioxidants in age-related maculopathy Italian study: Multifocal electroretinogram modifications after 1 year. Ophthalmology 2008, 115, 324–333. [Google Scholar] [CrossRef] [PubMed]
- Weigert, G.; Kaya, S.; Pemp, B.; Sacu, S.; Lasta, M.; Werkmeister, R.M.; Dragostinoff, N.; Simader, C.; Garhöfer, G.; Schmidt-Erfurth, U.; Schmetterer, L. Effects of lutein supplementation on macular pigment optical density and visual acuity in patients with age-related macular degeneration. Investig. Ophthalmol. Vis. Sci. 2011, 52, 8174–8178. [Google Scholar] [CrossRef] [PubMed]
- Beatty, S.; Chakravarthy, U.; Nolan, J.M.; Muldrew, K.A.; Woodside, J.V.; Denny, F.; Stevenson, M.R. Secondary outcomes in a clinical trial of carotenoids with coantioxidants versus placebo in early age-related macular degeneration. Ophthalmology 2013, 120, 600–606. [Google Scholar] [CrossRef] [PubMed]
- Murray, I.J.; Makridaki, M.; van der Veen, R.L.; Carden, D.; Parry, N.R.; Berendschot, T.T. Lutein supplementation over a one-year period in early AMD might have a mild beneficial effect on visual acuity: The CLEAR study. Investig. Ophthalmol. Vis. Sci. 2013, 54, 1781–1788. [Google Scholar] [CrossRef] [PubMed]
- Christen, W.G.; Liu, S.; Glynn, R.J.; Gaziano, J.M.; Buring, J.E. Dietary carotenoids, vitamins C and E, and risk of cataract in women: A prospective study. Arch. Ophthalmol. 2008, 126, 102–109. [Google Scholar] [CrossRef] [PubMed]
- Vu, H.T.; Robman, L.; Hodge, A.; McCarty, C.A.; Taylor, H.R. Lutein and zeaxanthin and the risk of cataract: The Melbourne visual impairment project. Investig. Ophthalmol. Vis. Sci. 2006, 47, 3783–3786. [Google Scholar] [CrossRef] [PubMed]
- Moeller, S.M.; Voland, R.; Tinker, L.; Blodi, B.A.; Klein, M.L.; Gehrs, K.M.; Johnson, E.J.; Snodderly, D.M.; Wallace, R.B.; Chappell, R.J.; et al. CAREDS Study Group. Associations between age-related nuclear cataract and lutein and zeaxanthin in the diet and serum in the Carotenoids in the Age-Related Eye Disease Study, an Ancillary Study of the Women’s Health Initiative. Arch. Ophthalmol. 2008, 126, 354–364. [Google Scholar] [CrossRef] [PubMed]
- Brown, L.; Rimm, E.B.; Seddon, J.M.; Giovannucci, E.L.; Chasan-Taber, L.; Spiegelman, D.; Willett, W.C.; Hankinson, S.E. A prospective study of carotenoid intake and risk of cataract extraction in US men. Am. J. Clin. Nutr. 1999, 70, 517–524. [Google Scholar] [CrossRef] [PubMed]
- Chasan-Taber, L.; Willett, W.C.; Seddon, J.M.; Stampfer, M.J.; Rosner, B.; Colditz, G.A.; Speizer, F.E.; Hankinson, S.E. A prospective study of carotenoid and vitamin A intakes and risk of cataract extraction in US women. Am. J. Clin. Nutr. 1999, 70, 509–516. [Google Scholar] [CrossRef] [PubMed]
- Rosenthal, J.M.; Kim, J.; de Monasterio, F.; Thompson, D.J.; Bone, R.A.; Landrum, J.T.; de Moura, F.F.; Khachik, F.; Chen, H.; Schleicher, R.L.; Ferris, F.L., III; Chew, E.Y. Dose-ranging study of lutein supplementation in persons aged 60 years or older. Invest. Ophthalmol. Vis. Sci. 2006, 47, 5227–5233. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Lin, X.M.; Zou, Z.Y.; Xu, X.R.; Li, Y.; Xu, R. A 12-week lutein supplementation improves visual function in Chinese people with long-term computer display light exposure. Br. J. Nutr. 2009, 102, 186–190. [Google Scholar] [CrossRef] [PubMed]
- Sasamoto, Y.; Gomi, F.; Sawa, M.; Tsujikawa, M.; Nishida, K. Effect of 1-year lutein supplementation on macular pigment optical density and visual function. Graefes Arch. Clin. Exp. Ophthalmol. 2011, 249, 1847–1854. [Google Scholar] [CrossRef] [PubMed]
- Obana, A.; Tanito, M.; Gohto, Y.; Okazaki, S.; Gellermann, W.; Bernstein, P.S. Changes in macular pigment optical density and serum lutein concentration in Japanese subjects taking two different lutein supplements. PLoS ONE 2015, 10, e0139257. [Google Scholar] [CrossRef] [PubMed]
- Berrow, E.J.; Bartlett, H.E.; Eperjesi, F.; Gibson, J.M. The effects of a lutein-based supplement on objective and subjective measures of retinal and visual function in eyes with age-related maculopathy—A randomised controlled trial. Br. J. Nutr. 2013, 109, 2008–2014. [Google Scholar] [CrossRef] [PubMed]
- Age-Related Eye Disease Study 2 Research Group. Lutein + zeaxanthin and omega-3 fatty acids for age-related macular degeneration: The Age-Related Eye Disease Study 2 [AREDS2] randomized clinical trial. JAMA 2013, 309, 2005–2015. [Google Scholar] [CrossRef] [PubMed]
- Korobelnik, J.F.; Rougier, M.B.; Delyfer, M.N.; Bron, A.; Merle, B.M.J.; Savel, H.; Chêne, G.; Delcourt, C.; Creuzot-Garcher, C. Effect of Dietary Supplementation with Lutein, Zeaxanthin, and ω-3 on Macular Pigment: A Randomized Clinical Trial. JAMA Ophthalmol. 2017, 135, 1259–1266. [Google Scholar] [CrossRef] [PubMed]
- Chong, E.W.; Wong, T.Y.; Kreis, A.J.; Simpson, J.A.; Guymer, R.H. Dietary antioxidants and primary prevention of age related macular degeneration: Systematic review and meta-analysis. BMJ 2007, 335, 755. [Google Scholar] [CrossRef] [PubMed]
- Cho, E.; Hankinson, S.E.; Rosner, B.; Willett, W.C.; Colditz, G.A. Prospective study of lutein/zeaxanthin intake and risk of age-related macular degeneration. Am. J. Clin. Nutr. 2008, 87, 1837–1843. [Google Scholar] [CrossRef] [PubMed]
- Trumbo, P.R.; Ellwood, K.C. Lutein and zeaxanthin intakes and risk of age-related macular degeneration and cataracts: An evaluation using the Food and Drug Administration’s evidence-based review system for health claims. Am. J. Clin. Nutr. 2006, 84, 971–974. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Jiang, C.; Zhang, Y.; Gong, Y.; Chen, X.; Zhang, M. Role of lutein supplementation in the management of age-related macular degeneration: Meta-analysis of randomized controlled trials. Ophthalmic Res. 2014, 52, 198–205. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Wang, T.; Zhang, B.; Qin, L.; Wu, C.; Li, Q.; Ma, L. Lutein and zeaxanthin supplementation and association with visual function in age-related macular degeneration. Investig. Ophthalmol. Vis. Sci. 2014, 56, 252–258. [Google Scholar] [CrossRef] [PubMed]
- Stringham, J.M.; Stringham, N.T. Serum and retinal responses to three different doses of macular carotenoids over 12 weeks of supplementation. Exp. Eye Res. 2016, 151, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Dawczynski, J.; Jentsch, S.; Schweitzer, D.; Hammer, M.; Lang, G.E.; Strobel, J. Long term effects of lutein, zeaxanthin and omega-3-LCPUFAs supplementation on optical density of macular pigment in AMD patients: The LUTEGA study. Graefes Arch. Clin. Exp. Ophthalmol. 2013, 251, 2711–2723. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.M.; Dou, H.L.; Huang, F.F.; Xu, X.R.; Zou, Z.Y.; Lin, X.M. Effect of supplemental lutein and zeaxanthin on serum, macular pigmentation, and visual performance in patients with early age-related macular degeneration. Biomed. Res. Int. 2015, 2015, 564738. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Yan, S.F.; Huang, Y.M.; Lu, X.R.; Qian, F.; Pang, H.L.; Xu, X.R.; Zou, Z.Y.; Dong, P.C.; Xiao, X.; et al. Effect of lutein and zeaxanthin on macular pigment and visual function in patients with early age-related macular degeneration. Ophthalmology 2012, 119, 2290–2297. [Google Scholar] [CrossRef] [PubMed]
- Bone, R.A.; Landrum, J.T. Dose-dependent response of serum lutein and macular pigment optical density to supplementation with lutein esters. Arch. Biochem. Biophys. 2010, 504, 50–55. [Google Scholar] [CrossRef] [PubMed]
- Chung, R.W.S.; Leanderson, P.; Lundberg, A.K.; Jonasson, L. Lutein exerts anti-inflammatory effects in patients with coronary artery disease. Atherosclerosis 2017, 262, 87–93. [Google Scholar] [CrossRef] [PubMed]
- Johnson, E.J.; Vishwanathan, R.; Johnson, M.A.; Hausman, D.B.; Davey, A.; Scott, T.M.; Green, R.C.; Miller, L.S.; Gearing, M.; Woodard, J.; et al. Relationship between serum and brain carotenoids, α-tocopherol, and retinol concentrations and cognitive performance in the oldest old from the Georgia Centenarian Study. J. Aging Res. 2013, 2013, 951786. [Google Scholar] [CrossRef] [PubMed]
- Slattery, M.L.; Benson, J.; Curtin, K.; Ma, K.N.; Schaeffer, D.; Potter, J.D. Carotenoids and colon cancer. Am. J. Clin. Nutr. 2000, 71, 575–582. [Google Scholar] [CrossRef] [PubMed]
- Regu, G.M.; Kim, H.; Kim, Y.J.; Paek, J.E.; Lee, G.; Chang, N.; Kwon, O. Association between dietary carotenoid intake and bone mineral density in Korean adults aged 30-75 years using data from the fourth and fifth Korean national health and nutrition examination surveys [2008–2011]. Nutrients 2017, 9, 1025. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.; Wang, C.; Liu, J.; Liu, Z.M.; Ling, W.H.; Chen, Y.M. Greater serum carotenoid levels associated with lower prevalence of nonalcoholic fatty liver disease in Chinese adults. Sci. Rep. 2015, 5, 12951. [Google Scholar] [CrossRef] [PubMed]
- Schünemann, H.J.; McCann, S.; Grant, B.J.; Trevisan, M.; Muti, P.; Freudenheim, J.L. Lung function in relation to intake of carotenoids and other antioxidant vitamins in a population-based study. Am. J. Epidemiol. 2002, 155, 463–471. [Google Scholar] [CrossRef] [PubMed]
- Bone, R.A.; Landrum, J.T.; Tarsis, S.L. Preliminary identification of the human macular pigment. Vis. Res. 1985, 25, 1531–1535. [Google Scholar] [CrossRef]
- Bone, R.A.; Landrum, J.T.; Hime, G.W.; Cains, A.; Zamor, J. Stereochemistry of the human macular carotenoids. Investig. Ophthalmol. Vis. Sci. 1993, 34, 2033–2040. [Google Scholar]
- Snodderly, D.M.; Brown, P.K.; Delori, F.C.; Auran, J.D. The macular pigment. I. Absorbance spectra, localization, and discrimination from other yellow pigments in primate retinas. Investig. Ophthalmol. Vis. Sci. 1984, 25, 660–673. [Google Scholar]
- Gass, J.D. Müller cell cone, an overlooked part of the anatomy of the fovea centralis: Hypotheses concerning its role in the pathogenesis of macular hole and foveomacular retinoschisis. Arch. Ophthalmol. 1999, 117, 821–823. [Google Scholar] [CrossRef] [PubMed]
- Bone, R.A.; Landrum, J.T.; Friedes, L.M.; Gomez, C.M.; Kilburn, M.D.; Menendez, E.; Vidal, I.; Wang, W. Distribution of lutein and zeaxanthin stereoisomers in the human retina. Exp. Eye Res. 1997, 64, 211–218. [Google Scholar] [CrossRef] [PubMed]
- Bone, R.A.; Landrum, J.T.; Fernandez, L.; Tarsis, S.L. Analysis of the macular pigment by HPLC: Retinal distribution and age study. Investig. Ophthalmol. Vis. Sci. 1988, 29, 843–849. [Google Scholar]
- Snodderly, D.M.; Auran, J.D.; Delori, F.C. The macular pigment. II. Spatial distribution in primate retinas. Investig. Ophthalmol. Vis. Sci. 1984, 25, 674–685. [Google Scholar]
- Bernstein, P.S.; Khachik, F.; Carvalho, L.S.; Muir, G.J.; Zhao, D.Y.; Katz, N.B. Identification and quantitation of carotenoids and their metabolites in the tissues of the human eye. Exp. Eye Res. 2001, 72, 215–223. [Google Scholar] [CrossRef] [PubMed]
- Sato, Y.; Kondo, Y.; Sumi, M.; Takekuma, Y.; Sugawara, M. Intracellular uptake mechanism of lutein in retinal pigment epithelial cells. J. Pharm. Pharm. Sci. 2013, 16, 494–501. [Google Scholar] [CrossRef] [PubMed]
- Vishwanathan, R.; Kuchan, M.J.; Sen, S.; Johnson, E.J. Lutein and preterm infants with decreased concentrations of brain carotenoids. J. Pediatr. Gastroenterol. Nutr. 2014, 59, 659–665. [Google Scholar] [CrossRef] [PubMed]
- Craft, N.E.; Haitema, T.B.; Garnett, K.M.; Fitch, K.A.; Dorey, C.K. Carotenoid, tocopherol, and retinol concentrations in elderly human brain. J. Nutr. Health Aging 2004, 8, 156–162. [Google Scholar] [PubMed]
- Parker, R.S. Absorption, metabolism, and transport of carotenoids. FASEB J. 1996, 10, 542–551. [Google Scholar] [CrossRef] [PubMed]
- Clevidence, B.A.; Bieri, J.G. Association of carotenoids with human plasma lipoproteins. Methods Enzymol. 1993, 214, 33–46. [Google Scholar] [CrossRef] [PubMed]
- Ribaya-Mercado, J.D.; Ordovas, J.M.; Russell, R.M. Effect of beta-carotene supplementation on the concentrations and distribution of carotenoids, vitamin E, vitamin A, and cholesterol in plasma lipoprotein and non-lipoprotein fractions in healthy older women. J. Am. Coll. Nutr. 1995, 14, 614–620. [Google Scholar] [CrossRef] [PubMed]
- Bernstein, P.S.; Lia, B.; Vachalia, P.P.; Gorusupudia, A.; Shyama, R.; Henriksen, B.S.; Nolan, J.M. Lutein, Zeaxanthin, and meso-Zeaxanthin: The basic and clinical science underlying carotenoid-based nutritional interventions against ocular disease. Prog. Retin. Eye Res. 2016, 50, 34–66. [Google Scholar] [CrossRef] [PubMed]
- Castenmiller, J.J.; West, C.E.; Linssen, J.P.; van het Hof, K.H.; Voragen, A.G. The food matrix of spinach is a limiting factor in determining the bioavailability of beta-carotene and to a lesser extent of lutein in humans. J. Nutr. 1999, 129, 349–355. [Google Scholar] [CrossRef] [PubMed]
- Unlu, N.Z.; Bohn, T.; Clinton, S.K.; Schwartz, S.J. Carotenoid absorption from salad and salsa by humans is enhanced by the addition of avocado or avocado oil. J. Nutr. 2005, 135, 431–436. [Google Scholar] [CrossRef] [PubMed]
- Rock, C.L.; Swendseid, M.E. Plasma beta-carotene response in humans after meals supplemented with dietary pectin. Am. J. Clin. Nutr. 1992, 55, 96–99. [Google Scholar] [CrossRef] [PubMed]
- Riedl, J.; Linseisen, J.; Hoffmann, J.; Wolfram, G. Some dietary fibers reduce the absorption of carotenoids in women. J. Nutr. 1999, 129, 2170–2176. [Google Scholar] [CrossRef] [PubMed]
- Kostic, D.; White, W.S.; Olson, J.A. Intestinal absorption, serum clearance, and interactions between lutein and beta-carotene when administered to human adults in separate or combined oral doses. Am. J. Clin. Nutr. 1995, 62, 604–610. [Google Scholar] [CrossRef] [PubMed]
- Williams, A.W.; Boileau, T.W.; Erdman, J.W., Jr. Factors influencing the uptake and absorption of carotenoids. Proc. Soc. Exp. Biol. Med. 1998, 218, 106–108. [Google Scholar] [CrossRef] [PubMed]
- Marriage, B.J.; Williams, J.A.; Choe, Y.S.; Maki, K.C.; Vurma, M.; DeMichele, S.J. Mono- and diglycerides improve lutein absorption in healthy adults: A randomised, double-blind, cross-over, single-dose study. Br. J. Nutr. 2017, 118, 813–821. [Google Scholar] [CrossRef] [PubMed]
- Sundl, I.; Pail, E.; Mellitzer, K.; Toplak, H.; Winklhofer-Roob, B.M. Effects of orlistat therapy on plasma concentrations of oxygenated and hydrocarbon carotenoids. Lipids 2006, 41, 113–118. [Google Scholar] [CrossRef] [PubMed]
- Tang, G.; Serfaty-Lacrosniere, C.; Camilo, M.E.; Russell, R.M. Gastric acidity influences the blood response to a beta-carotene dose in humans. Am. J. Clin. Nutr. 1996, 64, 622–626. [Google Scholar] [CrossRef] [PubMed]
- Winklhofer-Roob, B.M.; Puhl, H.; Khoschsorur, G.; van’t Hof, M.A.; Esterbauer, H.; Shmerling, D.H. Enhanced resistance to oxidation of low density lipoproteins and decreased lipid peroxide formation during beta-carotene supplementation in cystic fibrosis. Free Radic. Biol. Med. 1995, 18, 849–859. [Google Scholar] [CrossRef]
- Alberg, A. The influence of cigarette smoking on circulating concentrations of antioxidant micronutrients. Toxicology 2002, 180, 121–137. [Google Scholar] [CrossRef]
- Albanes, D.; Virtamo, J.; Taylor, P.R.; Rautalahti, M.; Pietinen, P.; Heinonen, O.P. Effects of supplemental beta-carotene, cigarette smoking, and alcohol consumption on serum carotenoids in the Alpha-Tocopherol, Beta-Carotene Cancer Prevention Study. Am. J. Clin. Nutr. 1997, 66, 366–372. [Google Scholar] [CrossRef] [PubMed]
- Kijlstra, A.; Tian, Y.; Kelly, E.R.; Berendschot, T.T. Lutein: More than just a filter for blue light. Prog. Retin. Eye Res. 2012, 31, 303–315. [Google Scholar] [CrossRef] [PubMed]
- Junghans, A.; Sies, H.; Stahl, W. Macular pigments lutein and zeaxanthin as blue light filters studied in liposomes. Arch. Biochem. Biophys. 2001, 391, 160–164. [Google Scholar] [CrossRef] [PubMed]
- Subczynski, W.K.; Wisniewska, A.; Widomska, J. Location of macular xanthophylls in the most vulnerable regions of photoreceptor outer-segment membranes. Arch. Biochem. Biophys. 2010, 504, 61–66. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Liu, W.H.; Zhao, J.S.; Meng, F.Z.; Wang, H. Lutein protects against β-amyloid peptide-induced oxidative stress in cerebrovascular endothelial cells through modulation of Nrf-2 and NF-κb. Cell. Biol. Toxicol. 2017, 33, 57–67. [Google Scholar] [CrossRef] [PubMed]
- Chang, J.; Zhang, Y.; Li, Y.; Lu, K.; Shen, Y.; Guo, Y.; Qi, Q.; Wang, M.; Zhang, S. NrF2/ARE and NF-κB pathway regulation may be the mechanism for lutein inhibition of human breast cancer cell. Future Oncol. 2018, 14, 719–726. [Google Scholar] [CrossRef] [PubMed]
- Muriach, M.; Bosch-Morell, F.; Arnal, E.; Alexander, G.; Blomhoff, R.; Romero, F.J. Lutein prevents the effect of high glucose levels on immune system cells in vivo and in vitro. J. Physiol. Biochem. 2008, 64, 149–157. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Ding, Y.; Niu, Q.; Xu, S.; Pang, L.; Ma, R.; Jing, M.; Feng, G.; Tang, J.X.; Zhang, Q.; et al. Lutein has a protective effect on hepatotoxicity induced by arsenic via Nrf2 signaling. BioMed Res. Int. 2015, 2015, 315205. [Google Scholar] [CrossRef] [PubMed]
- Rafi, M.M.; Shafaie, Y. Dietary lutein modulates inducible nitric oxide synthase (iNOS) gene and protein expression in mouse macrophage cells (RAW 264.7). Mol. Nutr. Food Res. 2007, 51, 333–340. [Google Scholar] [CrossRef] [PubMed]
- Tian, Y.; Kijlstra, A.; van der Veen, R.L.; Makridaki, M.; Murray, I.J.; Berendschot, T.T. Lutein supplementation leads to decreased soluble complement membrane attack complex sC5b-9 plasma levels. Acta Ophthalmol. 2015, 93, 141–145. [Google Scholar] [CrossRef] [PubMed]
- Chung, H.Y.; Rasmussen, H.M.; Johnson, E.J. Lutein bioavailability is higher from lutein-enriched eggs than from supplements and spinach in men. J. Nutr. 2004, 134, 1887–1893. [Google Scholar] [CrossRef] [PubMed]
- Johnson, E.J.; Maras, J.E.; Rasmussen, H.M.; Tucker, K.L. Intake of lutein and zeaxanthin differ with age, sex, and ethnicity. J. Am. Diet. Assoc. 2010, 110, 1357–1362. [Google Scholar] [CrossRef] [PubMed]
- United States Department of Agriculture, Agricultural Research Service. What We Eat in America, NHANES 2011–2012; USDA ARS: Washington, DC, USA, 2014. Available online: http://www.ars.usda.gov/Services/docs.htm?docid=13793 (accessed on 1 July 2018).
- Olmedilla-Alonso, B.; Beltrán-de-Miguel, B.; Estévez-Santiago, R.; Cuadrado-Vives, C. Markers of lutein and zeaxanthin status in two age groups of men and women: Dietary intake, serum concentrations, lipid profile and macular pigment optical density. Nutr. J. 2014, 13, 52. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, M.E.; Carroll, Y.; Corridan, B.; Olmedilla, B.; Granado, F.; Blanco, I.; Van den Berg, H.; Hininger, I.; Rousell, A.M.; Chopra, M.; et al. A European carotenoid database to assess carotenoid intakes and its use in a five-country comparative study. Br. J. Nutr. 2001, 85, 499–507. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.S.; Cho, Y.H.; Park, J.; Shin, H.R.; Sung, M.K. Dietary intake of phytonutrients in relation to fruit and vegetable consumption in Korea. J. Acad. Nutr. Diet. 2013, 113, 1194–1199. [Google Scholar] [CrossRef] [PubMed]
- Le Marchand, L.; Hankin, J.H.; Bach, F.; Kolonel, L.N.; Wilkens, L.R.; Stacewicz-Sapuntzakis, M.; Bowen, P.E.; Beecher, G.R.; Laudon, F.; Baque, P.; et al. An ecological study of diet and lung cancer in the South Pacific. Int. J. Cancer 1995, 63, 18–23. [Google Scholar] [CrossRef] [PubMed]
- Available online: https://ndb.nal.usda.gov/ndb/nutrients/report/nutrientsfrm?max=25&offset=0&totCount=0&nutrient1=338&nutrient2=&nutrient3=&subset=0&sort=c&measureby=g (accessed on 1 July 2018).
- Wong, W.L.; Su, X.; Li, X.; Cheung, C.M.; Klein, R.; Cheng, C.Y.; Wong, T.Y. Global prevalence of age-related macular degeneration and disease burden projection for 2020 and 2040: A systematic review and meta-analysis. Lancet Glob. Health 2014, 2, e106–e116. [Google Scholar] [CrossRef]
- Lim, L.S.; Mitchell, P.; Seddon, J.M.; Holz, F.G.; Wong, T.Y. Age-related macular degeneration. Lancet 2012, 379, 1728–1738. [Google Scholar] [CrossRef]
- Abdel-Aal, E.-S.M.; Akhtar, H.; Zaheer, K.; Ali, R. Dietary sources of lutein and zeaxanthin carotenoids and their role in eye health. Nutrients 2013, 5, 1169–1185. [Google Scholar] [CrossRef] [PubMed]
- Akuffo, K.O.; Nolan, J.M.; Howard, A.N.; Moran, R.; Stack, J.; Klein, R.; Klein, B.E.; Meuer, S.M.; Sabour-Pickett, S.; Thurnham, D.I.; et al. Sustained supplementation and monitored response with differing carotenoid formulations in early age-related macular degeneration. Eye 2015, 29, 902–912. [Google Scholar] [CrossRef] [PubMed]
- Yao, Y.; Qiu, Q.H.; Wu, X.W.; Cai, Z.Y.; Xu, S.; Liang, X.Q. Lutein supplementation improves visual performance in Chinese drivers: 1-year randomized, double-blind, placebo-controlled study. Nutrition 2013, 29, 958–964. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.M.; Dou, H.L.; Huang, F.F.; Xu, X.R.; Zou, Z.Y.; Lu, X.R.; Lin, X.M. Changes following supplementation with lutein and zeaxanthin in retinal function in eyes with early age-related macular degeneration: A randomised, double-blind, placebo-controlled trial. Br. J. Ophthalmol. 2015, 99, 371–375. [Google Scholar] [CrossRef] [PubMed]
- Richer, S.P.; Stiles, W.; Graham-Hoffman, K.; Levin, M.; Ruskin, D.; Wrobel, J.; Park, D.W.; Thomas, C. Randomized, double-blind, placebo-controlled study of zeaxanthin and visual function in patients with atrophic age-related macular degeneration: The Zeaxanthin and Visual Function Study (ZVF) FDA IND #78, 973. Optometry 2011, 82, 667–680. [Google Scholar] [CrossRef] [PubMed]
- Fujimura, S.; Ueda, K.; Nomura, Y.; Yanagi, Y. Preliminary analysis of the relationship between serum lutein and zeaxanthin levels and macular pigment optical density. Clin. Ophthalmol. 2016, 10, 2149–2155. [Google Scholar] [CrossRef] [PubMed]
- Hammond, B.R.; Fletcher, L.M.; Roos, F.; Wittwer, J.; Schalch, W. A double-blind, placebo-controlled study on the effects of lutein and zeaxanthin on photostress recovery, glare disability, and chromatic contrast. Investig. Ophthalmol. Vis. Sci. 2014, 55, 8583–8589. [Google Scholar] [CrossRef] [PubMed]
- Nolan, J.M.; Loughman, J.; Akkali, M.C.; Stack, J.; Scanlon, G.; Davison, P.; Beatty, S. The impact of macular pigment augmentation on visual performance in normal subjects: COMPASS. Vis. Res. 2011, 51, 459–469. [Google Scholar] [CrossRef] [PubMed]
- Wolf-Schnurrbusch, U.E.; Zinkernagel, M.S.; Munk, M.R.; Ebneter, A.; Wolf, S. Oral lutein supplementation enhances macular pigment density and contrast sensitivity but not in combination with polyunsaturated fatty acids. Investig. Ophthalmol. Vis. Sci. 2015, 56, 8069–8074. [Google Scholar] [CrossRef] [PubMed]
- Sabour-Pickett, S.; Beatty, S.; Connolly, E.; Loughman, J.; Stack, J.; Howard, A.; Klein, R.; Klein, B.E.; Meuer, S.M.; Myers, C.E.; et al. Supplementation with three different macular carotenoid formulations in patients with early age-related macular degeneration. Retina 2014, 34, 1757–1766. [Google Scholar] [CrossRef] [PubMed]
- Cangemi, F.E. TOZAL Study: An open case control study of an oral antioxidant and omega-3 supplement for dry AMD. BMC Ophthalmol. 2007, 7, 3. [Google Scholar] [CrossRef] [PubMed]
- Piermarocchi, S.; Saviano, S.; Parisi, V.; Tedeschi, M.; Panozzo, G.; Scarpa, G.; Boschi, G.; Lo Giudice, G.; Carmis Study Group. Carotenoids in Age-related Maculopathy Italian Study [CARMIS]: Two-year results of a randomized study. Eur. J. Ophthalmol. 2012, 22, 216–225. [Google Scholar] [CrossRef] [PubMed]
- Eye Disease Case-Control Study Group. Antioxidant status and neovascular age-related macular degeneration. Arch. Ophthalmol. 1993, 111, 104–109. [Google Scholar] [CrossRef]
- Age-Related Eye Disease Study Research Group. A randomized, placebo-controlled, clinical trial of high dose supplementation with vitamins C and E, beta carotene, and zinc for age-related macular degeneration and vision loss: AREDS report no. 8. Arch. Ophthalmol. 2001, 119, 1417–1436. [Google Scholar] [CrossRef]
- Age-Related Eye Disease Study 2 (AREDS2) Research Group; Chew, E.Y.; Clemons, T.E.; Sangiovanni, J.P.; Danis, R.P.; Ferris, F.L., III; Elman, M.J.; Antoszyk, A.N.; Ruby, A.J.; Orth, D.; et al. Secondary analyses of the effects of lutein/zeaxanthin on age-related macular degeneration progression: AREDS2 report No. 3. JAMA Ophthalmol. 2014, 132, 142–149. [Google Scholar] [CrossRef] [PubMed]
- Albanes, D.; Heinonen, O.P.; Huttunen, J.K.; Taylor, P.R.; Virtamo, J.; Edwards, B.K.; Haapakoski, J.; Rautalahti, M.; Hartman, A.M.; Palmgren, J. Effects of alpha-tocopherol and beta-carotene supplements on cancer incidence in the Alpha-Tocopherol Beta-Carotene Cancer Prevention Study. Am. J. Clin. Nutr. 1995, 62, 1427S–1430S. [Google Scholar] [CrossRef] [PubMed]
- Omenn, G.S.; Goodman, G.E.; Thornquist, M.D.; Balmes, J.; Cullen, M.R.; Glass, A.; Keogh, J.P.; Meyskens, F.L., Jr.; Valanis, B.; Williams, J.H., Jr.; et al. Risk factors for lung cancer and for intervention effects in CARET, the Beta-Carotene and Retinol Efficacy Trial. J. Natl. Cancer Inst. 1996, 88, 1550–1559. [Google Scholar] [CrossRef] [PubMed]
- Ho, L.; van Leeuwen, R.; Witteman, J.C.; van Duijn, C.M.; Uitterlinden, A.G.; Hofman, A.; de Jong, P.T.; Vingerling, J.R.; Klaver, C.C. Reducing the genetic risk of age-related macular degeneration with dietary antioxidants, zinc, and ω-3 fatty acids: The Rotterdam study. Arch. Ophthalmol. 2011, 129, 758–766. [Google Scholar] [CrossRef] [PubMed]
- Chitchumroonchokchai, C.; Bomser, J.A.; Glamm, J.E.; Failla, M.L. Xanthophylls and alpha-tocopherol decrease UVB-induced lipid peroxidation and stress signaling in human lens epithelial cells. J. Nutr. 2004, 134, 3225–3232. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.C.; Wilkins, M.; Kim, T.; Malyugin, B.; Mehta, J.S. Cataracts. Lancet 2017, 390, 600–612. [Google Scholar] [CrossRef]
- Hu, Y.; Xu, Z.J. Effects of lutein on the growth and migration of bovine lens epithelial cells in vitro. Huazhong Univ. Sci. Technol. Med. Sci. 2008, 28, 360–363. [Google Scholar] [CrossRef] [PubMed]
- Jacques, P.F.; Taylor, A.; Moeller, S.; Hankinson, S.E.; Rogers, G.; Tung, W.; Ludovico, J.; Willett, W.C.; Chylack, L.T., Jr. Long-term nutrient intake and 5-year change in nuclear lens opacities. Arch. Ophthalmol. 2005, 123, 517–526. [Google Scholar] [CrossRef] [PubMed]
- Yeum, K.J.; Shang, F.M.; Schalch, W.M.; Russell, R.M.; Taylor, A. Fat-soluble nutrient concentrations in different layers of human cataractous lens. Curr. Eye Res. 1999, 19, 502–505. [Google Scholar] [CrossRef] [PubMed]
- Karppi, J.; Laukkanen, J.A.; Kurl, S. Plasma lutein and zeaxanthin and the risk of age-related nuclear cataract among the elderly Finnish population. Br. J. Nutr. 2012, 108, 148–154. [Google Scholar] [CrossRef] [PubMed]
- Gale, C.R.; Hall, N.F.; Phillips, D.I.; Martyn, C.N. Plasma antioxidant vitamins and carotenoids and age-related cataract. Ophthalmology 2001, 108, 1992–1998. [Google Scholar] [CrossRef]
- Olmedilla, B.; Granado, F.; Blanco, I.; Vaquero, M. Lutein, but not alpha-tocopherol, supplementation improves visual function in patients with age-related cataracts: A 2-y double-blind, placebo-controlled pilot study. Nutrition 2003, 19, 21–24. [Google Scholar] [CrossRef]
- Olmedilla, B.; Granado, F.; Blanco, I.; Vaquero, M.; Cajiga, L. Lutein in patients with cataracts and age-related macular degeneration: A long-term supplementation study. J. Sci. Food Agric. 2001, 81, 904–909. [Google Scholar] [CrossRef]
- Lyle, B.J.; Mares-Perlman, J.A.; Klein, B.E.; Klein, R.; Palta, M.; Bowen, P.E.; Greger, J.L. Serum carotenoids and tocopherols and incidence of age-related nuclear cataract. Am. J. Clin. Nutr. 1999, 69, 272–277. [Google Scholar] [CrossRef] [PubMed]
- Lyle, B.J.; Mares-Perlman, J.A.; Klein, B.E.; Klein, R.; Greger, J.L. Antioxidant intake and risk of incident age-related nuclear cataracts in the Beaver Dam Eye Study. Am. J. Epidemiol. 1999, 149, 801–809. [Google Scholar] [CrossRef] [PubMed]
- Age-Related Eye Disease Study 2 (AREDS2) Research Group; Chew, E.Y.; San Giovanni, J.P.; Ferris, F.L.; Wong, W.T.; Agron, E.; Clemons, T.E.; Sperduto, R.; Danis, R.; Chandra, S.R.; et al. Lutein/zeaxanthin for the treatment of age-related cataract: AREDS2 randomized trial report no. 4. JAMA Ophthalmol. 2013, 131, 843–850. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.H.; Yu, R.B.; Liu, R.; Hao, Z.X.; Han, C.C.; Zhu, Z.H.; Ma, L. Association between lutein and zeaxanthin status and the risk of cataract: A meta-analysis. Nutrients 2014, 6, 452–465. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Hao, Z.X.; Liu, R.R.; Yu, R.B.; Shi, Q.; Pan, J.P. A dose-response meta-analysis of dietary lutein and zeaxanthin intake in relation to risk of age-related cataract. Graefes Arch. Clin. Exp. Ophthalmol. 2014, 252, 63–70. [Google Scholar] [CrossRef] [PubMed]
- Mares-Perlman, J.A.; Millen, A.E.; Ficek, T.L.; Hankinson, S.E. The body of evidence to support a protective role for lutein and zeaxanthin in delaying chronic disease. Overview. J. Nutr. 2002, 132, 518S–524S. [Google Scholar] [CrossRef] [PubMed]
- Weikel, K.A.; Garber, C.; Baburins, A.; Taylor, A. Nutritional modulation of cataract. Nutr. Rev. 2014, 72, 30–47. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.C.; Wu, C.R.; Wang, Z.L.; Wang, L.Y.; Han, Y.; Sun, S.L.; Li, Q.S.; Ma, L. Effect of lutein supplementation on visual function in nonproliferative diabetic retinopathy. Asia Pac. J. Clin. Nutr. 2017, 26, 406–411. [Google Scholar] [CrossRef] [PubMed]
- Sahli, M.W.; Mares, J.A.; Meyers, K.J.; Klein, R.; Brady, W.E.; Klein, B.E.; Ochs-Balcom, H.M.; Donahue, R.P.; Millen, A.E. Dietary intake of lutein and diabetic retinopathy in the Atherosclerosis Risk in Communities Study (ARIC). Ophthalmic Epidemiol. 2016, 23, 99–108. [Google Scholar] [CrossRef] [PubMed]
- Bahrami, H.; Melia, M.; Dagnelie, G. Lutein supplementation in retinitis pigmentosa: PC-based vision assessment in a randomized double-masked placebo-controlled clinical trial [NCT00029289]. BMC Ophthalmol. 2006, 6, 23. [Google Scholar] [CrossRef] [PubMed]
- Aleman, T.S.; Duncan, J.L.; Bieber, M.L.; de Castro, E.; Marks, D.A.; Gardner, L.M.; Steinberg, J.D.; Cideciyan, A.V.; Maguire, M.G.; Jacobson, S.G. Macular pigment and lutein supplementation in retinitis pigmentosa and Usher syndrome. Investig. Ophthalmol. Vis. Sci. 2001, 42, 1873–1881. [Google Scholar]
- Adackapara, C.A.; Sunness, J.S.; Dibernardo, C.W.; Melia, B.M.; Dagnelie, G. Prevalence of cystoid macular edema and stability in oct retinal thickness in eyes with retinitis pigmentosa during a 48-week lutein trial. Retina 2008, 28, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Smith, L.E. Retinopathy of prematurity. Angiogenesis 2007, 10, 133–140. [Google Scholar] [CrossRef] [PubMed]
- Fu, Z.; Meng, S.S.; Burnim, S.B.; Smith, L.E.; Lo, A.C. Lutein facilitates physiological revascularization in a mouse model of retinopathy of prematurity. Clin. Exp. Ophthalmol. 2017, 45, 529–538. [Google Scholar] [CrossRef] [PubMed]
- Dani, C.; Lori, I.; Favelli, F.; Frosini, S.; Messner, H.; Wanker, P.; De Marini, S.; Oretti, C.; Boldrini, A.; Ciantelli, M.; et al. Lutein and zeaxanthin supplementation in preterm infants to prevent retinopathy of prematurity: A randomized controlled study. J. Maternal Fetal Neonatal Med. 2012, 25, 523–527. [Google Scholar] [CrossRef] [PubMed]
- Feeney, J.; O’Leary, N.; Moran, R.; O’Halloran, A.M.; Nolan, J.M.; Beatty, S.; Young, I.S.; Kenny, R.A. Plasma lutein and zeaxanthin are associated with better cognitive function across multiple domains in a large population-based sample of older adults: Findings from the Irish Longitudinal Study on Aging. J. Gerontol. A Biol. Sci. Med. Sci. 2017, 72, 1431–1436. [Google Scholar] [CrossRef] [PubMed]
- Zamroziewicz, M.K.; Paul, E.J.; Zwilling, C.E.; Johnson, E.J.; Kuchan, M.J.; Cohen, N.J.; Barbey, A.K. Parahippocampal cortex mediates the relationship between lutein and crystallized intelligence in healthy, older adults. Front. Aging Neurosci. 2016, 8, 297. [Google Scholar] [CrossRef] [PubMed]
- Picone, S.; Ritieni, A.; Fabiano, A.; Graziani, G.; Paolillo, P.; Livolti, G.; Galvano, F.; Gazzolo, D. Lutein levels in arterial cord blood correlate with neuroprotein activin A in healthy preterm and term newborns: A trophic role for lutein? Clin. Biochem. 2018, 52, 80–84. [Google Scholar] [CrossRef] [PubMed]
- Lai, M.; Sirimanne, E.; Williams, C.E.; Gluckman, P.D. Sequential patterns of inhibin subunit gene expression following hypoxic-ischemic injury in the rat brain. Neuroscience 1996, 70, 1013–1024. [Google Scholar] [CrossRef]
- Lai, M.; Gluckman, P.; Dragunow, M.; Hughes, P.E. Focal brain injury increases activin betaA mRNA expression in hippocampal neurons. Neuroreport 1997, 8, 2691–2694. [Google Scholar] [CrossRef] [PubMed]
- Johnson, E.J.; McDonald, K.; Caldarella, S.M.; Chung, H.Y.; Troen, A.M.; Snodderly, D.M. Cognitive findings of an exploratory trial of docosahexaenoic acid and lutein supplementation in older women. Nutr. Neurosci. 2008, 11, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Power, R.; Coen, R.F.; Beatty, S.; Mulcahy, R.; Moran, R.; Stack, J.; Howard, A.N.; Nolan, J.M. Supplemental retinal carotenoids enhance memory in healthy individuals with low levels of macular pigment in a randomized, double-blind, placebo-controlled clinical trial. J. Alzheimer’s Dis 2018, 61, 947–961. [Google Scholar] [CrossRef] [PubMed]
- Renzi-Hammond, L.M.; Bovier, E.R.; Fletcher, L.M.; Miller, L.S.; Mewborn, C.M.; Lindbergh, C.A.; Baxter, J.H.; Hammond, B.R. Effects of a lutein and zeaxanthin intervention on cognitive function: A randomized, double-masked, placebo-controlled trial of younger healthy adults. Nutrients 2017, 9, 1246. [Google Scholar] [CrossRef] [PubMed]
- Lindbergh, C.A.; Renzi-Hammond, L.M.; Hammond, B.R.; Terry, D.P.; Mewborn, C.M.; Puente, A.N.; Miller, L.S. Lutein and zeaxanthin influence brain function in older adults: A randomized controlled trial. J. Int. Neuropsychol. Soc. 2018, 24, 77–90. [Google Scholar] [CrossRef] [PubMed]
- Lindbergh, C.A.; Mewborn, C.M.; Hammond, B.R.; Renzi-Hammond, L.M.; Curran-Celentano, J.M.; Miller, L.S. Relationship of lutein and zeaxanthin levels to neurocognitive functioning: An fMRI study of older adults. J. Int. Neuropsychol. Soc. 2017, 23, 11–22. [Google Scholar] [CrossRef] [PubMed]
- Feeney, J.; Finucane, C.; Savva, G.M.; Cronin, H.; Beatty, S.; Nolan, J.M.; Kenny, R.A. Low macular pigment optical density is associated with lower cognitive performance in a large, population-based sample of older adults. Neurobiol. Aging 2013, 34, 2449–2456. [Google Scholar] [CrossRef] [PubMed]
- Vishwanathan, R.; Iannaccone, A.; Scott, T.M.; Kritchevsky, S.B.; Jennings, B.J.; Carboni, G.; Forma, G.; Satterfield, S.; Harris, T.; Johnson, K.C.; et al. Macular pigment optical density is related to cognitive function in older people. Age Ageing 2014, 43, 271–275. [Google Scholar] [CrossRef] [PubMed]
- Kuchan, M.; Wang, F.; Geng, Y.; Feng, B.; Lai, C. Lutein stimulates the differentiation of human stem cells to neural progenitor cells in vitro. In Advances and Controversies in Clinical Nutrition; Abstract No. 23; ILSI North America: Washington, DC, USA, 2013. [Google Scholar]
- Nataraj, J.; Manivasagam, T.; Thenmozhi, A.J.; Essa, M.M. Lutein protects dopaminergic neurons against MPTP-induced apoptotic death and motor dysfunction by ameliorating mitochondrial disruption and oxidative stress. Nutr. Neurosci. 2016, 19, 237–246. [Google Scholar] [CrossRef] [PubMed]
- Morris, M.C.; Wang, Y.; Barnes, L.L.; Bennett, D.A.; Dawson-Hughes, B.; Booth, S.L. Nutrients and bioactives in green leafy vegetables and cognitive decline: Prospective study. Neurology 2018, 90, e214–e222. [Google Scholar] [CrossRef] [PubMed]
- Chew, E.Y.; Clemons, T.E.; Agrón, E.; Launer, L.J.; Grodstein, F.; Bernstein, P.S.; Age-Related Eye Disease Study 2 (AREDS2) Research Group. Effect of omega-3 fatty acids, lutein/zeaxanthin, or other nutrient supplementation on cognitive function: The AREDS2 randomized clinical trial. JAMA 2015, 314, 791–801. [Google Scholar] [CrossRef] [PubMed]
- Libby, P.; Tabas, I.; Fredman, G.; Fisher, E.A. Inflammation and its resolution as determinants of acute coronary syndromes. Circ. Res. 2014, 114, 1867–1879. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.E.; Leite, J.O.; DeOgburn, R.; Smyth, J.A.; Clark, R.M.; Fernandez, M.L. A lutein-enriched diet prevents cholesterol accumulation and decreases oxidized LDL and inflammatory cytokines in the aorta of guinea pigs. J. Nutr. 2011, 141, 1458–1463. [Google Scholar] [CrossRef] [PubMed]
- Han, H.; Cui, W.; Wang, L.; Xiong, Y.; Liu, L.; Sun, X.; Hao, L. Lutein prevents high fat diet-induced atherosclerosis in ApoE-deficient mice by inhibiting NADPH oxidase and increasing PPAR expression. Lipids 2015, 50, 261–273. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Wang, M.; Zhang, S.; Zhao, L. Oxidative stress in rats with hyperhomo-cysteinemia and intervention effect of lutein. Eur. Rev. Med. Pharmacol. Sci. 2014, 18, 359–364. [Google Scholar] [PubMed]
- Boushey, C.J.; Beresford, S.A.; Omenn, G.S.; Motulsky, A.G. A quantitative assessment of plasma homocysteine as a risk factor for vascular disease. Probable benefits of increasing folic acid intakes. JAMA 1995, 274, 1049–1057. [Google Scholar] [CrossRef] [PubMed]
- Homocysteine Studies Collaboration. Homocysteine and risk of ischemic heart disease and stroke: A meta-analysis. JAMA 2002, 288, 2015–2022. [Google Scholar] [CrossRef]
- Sharavana, G.; Joseph, G.S.; Baskaran, V. Lutein attenuates oxidative stress markers and ameliorates glucose homeostasis through polyol pathway in heart and kidney of STZ-induced hyperglycemic rat model. Eur. J. Nutr. 2017, 56, 2475–2485. [Google Scholar] [CrossRef] [PubMed]
- Lind, M.; Hayes, A.; Caprnda, M.; Petrovic, D.; Rodrigo, L.; Kruzliak, P.; Zulli, A. Inducible nitric oxide synthase: Good or bad? Biomed. Pharmacother. 2017, 93, 370–375. [Google Scholar] [CrossRef] [PubMed]
- Howard, A.N.; Thurnham, D.I. Lutein and atherosclerosis: Belfast versus Toulouse revisited. Med. Hypotheses 2017, 98, 63–68. [Google Scholar] [CrossRef] [PubMed]
- Tian, Y.; Kijlstra, A.; van der Veen, R.L.; Makridaki, M.; Murray, I.J.; Berendschot, T.T. The effect of lutein supplementation on blood plasma levels of complement factor D, C5a and C3d. PLoS ONE 2013, 8, e73387. [Google Scholar] [CrossRef] [PubMed]
- Vlaicu, S.I.; Tatomir, A.; Rus, V.; Mekala, A.P.; Mircea, P.A.; Niculescu, F.; Rus, H. The role of complement activation in atherogenesis: The first 40 years. Immunol. Res. 2016, 64, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.X.; Jiao, J.H.; Li, Z.Y.; Liu, R.R.; Shi, Q.; Ma, L. Lutein supplementation reduces plasma lipid peroxidation and C-reactive protein in healthy nonsmokers. Atherosclerosis 2013, 227, 380–385. [Google Scholar] [CrossRef] [PubMed]
- Karppi, J.; Nurmi, T.; Kurl, S.; Rissanen, T.H.; Nyyssönen, K. Lycopene, lutein and beta-carotene as determinants of LDL conjugated dienes in serum. Atherosclerosis 2010, 209, 565–572. [Google Scholar] [CrossRef] [PubMed]
- Karppi, J.; Kurl, S.; Mäkikallio, T.H.; Ronkainen, K.; Laukkanen, J.A. Low levels of plasma carotenoids are associated with an increased risk of atrial fibrillation. Eur. J. Epidemiol. 2013, 28, 45–53. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.R.; Zou, Z.Y.; Xiao, X.; Huang, Y.M.; Wang, X.; Lin, X.M. Effects of lutein supplement on serum inflammatory cytokines, ApoE and lipid profiles in early atherosclerosis population. J. Atheroscler. Thromb. 2013, 20, 170–177. [Google Scholar] [CrossRef] [PubMed]
- Zou, Z.Y.; Xu, X.R.; Lin, X.M.; Zhang, H.B.; Xiao, X.; Ouyang, L.; Huang, Y.M.; Wang, X.; Liu, Y.Q. Effects of lutein and lycopene on carotid intima-media thickness in Chinese subjects with subclinical atherosclerosis: A randomised, double-blind, placebo-controlled trial. Br. J. Nutr. 2014, 111, 474–480. [Google Scholar] [CrossRef] [PubMed]
- Polak, J.F.; Pencina, M.J.; Pencina, K.M.; O’Donnell, C.J.; Wolf, P.A.; D’Agostino Sr, R.B. Carotid-wall intima-media thickness and cardiovascular events. N. Engl. J. Med. 2011, 365, 213–221. [Google Scholar] [CrossRef] [PubMed]
- Sen, A.; Marsche, G.; Freudenberger, P.; Schallert, M.; Toeglhofer, A.M.; Nagl, C.; Schmidt, R.; Launer, L.J.; Schmidt, H. Association between higher plasma lutein, zeaxanthin, and vitamin C concentrations and longer telomere length: Results of the Austrian Stroke Prevention Study. J. Am. Geriatr. Soc. 2014, 62, 222–229. [Google Scholar] [CrossRef] [PubMed]
- Brouilette, S.; Singh, R.K.; Thompson, J.R.; Goodall, A.H.; Samani, N.J. White cell telomere length and risk of premature myocardial infarction. Arterioscler. Thromb. Vasc. Biol. 2003, 23, 842–846. [Google Scholar] [CrossRef] [PubMed]
- Bonds, D.E.; Harrington, M.; Worrall, B.B.; Bertoni, A.G.; Eaton, C.B.; Hsia, J.; Robinson, J.; Clemons, T.E.; Fine, L.J.; Chew, E.Y. Effect of long-chain ω-3 fatty acids and lutein + zeaxanthin supplements on cardiovascular outcomes: Results of the Age-Related Eye Disease Study 2 (AREDS2) randomized clinical trial. JAMA Intern. Med. 2014, 174, 763–771. [Google Scholar] [CrossRef] [PubMed]
- Leermakers, E.T.; Kiefte-de Jong, J.C.; Hofman, A.; Jaddoe, V.W.; Franco, O.H. Lutein intake at the age of 1 year and cardiometabolic health at the age of 6 years: The Generation R Study. Br. J. Nutr. 2015, 114, 970–978. [Google Scholar] [CrossRef] [PubMed]
- Leermakers, E.T.; Darweesh, S.K.; Baena, C.P.; Moreira, E.M.; Melo van Lent, D.; Tielemans, M.J.; Muka, T.; Vitezova, A.; Chowdhury, R.; Bramer, W.M.; et al. The effects of lutein on cardiometabolic health across the life course: A systematic review and meta-analysis. Am. J. Clin. Nutr. 2016, 103, 481–494. [Google Scholar] [CrossRef] [PubMed]
- Teymouri, M.; Pirro, M.; Fallarino, F.; Gargaro, M.; Sahebkar, A. IL-35, a hallmark of immune-regulation in cancer progression, chronic infections and inflammatory diseases. Int. J. Cancer 2018. [Google Scholar] [CrossRef] [PubMed]
- Mantovani, A.; Barajon, I.; Garlanda, C. IL-1 and IL-1 regulatory pathways in cancer progression and therapy. Immunol. Rev. 2018, 281, 57–61. [Google Scholar] [CrossRef] [PubMed]
- Jansen, R.J.; Robinson, D.P.; Stolzenberg-Solomon, R.Z.; Bamlet, W.R.; de Andrade, M.; Oberg, A.L.; Rabe, K.G.; Anderson, K.E.; Olson, J.E.; Sinha, R.; et al. Nutrients from fruit and vegetable consumption reduce the risk of pancreatic cancer. J. Gastrointest. Cancer 2013, 44, 152–161. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Li, B.; Pan, M.X.; Mo, X.F.; Chen, Y.M.; Zhang, C.X. Specific carotenoid intake is inversely associated with the risk of breast cancer among Chinese women. Br. J. Nutr. 2014, 111, 1686–1695. [Google Scholar] [CrossRef] [PubMed]
- Bae, J.M. Reinterpretation of the results of a pooled analysis of dietary carotenoid intake and breast cancer risk by using the interval collapsing method. Epidemiol. Health 2016, 38, e2016024. [Google Scholar] [CrossRef] [PubMed]
- Bakker, M.F.; Peeters, P.H.; Klaasen, V.M.; Bueno-de-Mesquita, H.B.; Jansen, E.H.; Ros, M.M.; Travier, N.; Olsen, A.; Tjønneland, A.; Overvad, K.; et al. Plasma carotenoids, vitamin C, tocopherols, and retinol and the risk of breast cancer in the European Prospective Investigation into Cancer and Nutrition cohort. Am. J. Clin. Nutr. 2016, 103, 454–464. [Google Scholar] [CrossRef] [PubMed]
- Leoncini, E.; Edefonti, V.; Hashibe, M.; Parpinel, M.; Cadoni, G.; Ferraroni, M.; Serraino, D.; Matsuo, K.; Olshan, A.F.; Zevallos, J.P.; et al. Carotenoid intake and head and neck cancer: A pooled analysis in the International Head and Neck Cancer Epidemiology Consortium. Eur. J. Epidemiol. 2016, 31, 369–383. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Hu, J.; Liu, P.; Li, J.; Wei, Z.; Liu, P. Carotenoid intake and risk of non-Hodgkin lymphoma: A systematic review and dose-response meta-analysis of observational studies. Ann. Hematol. 2017, 96, 957–965. [Google Scholar] [CrossRef] [PubMed]
- Ge, X.X.; Xing, M.Y.; Yu, L.F.; Shen, P. Carotenoid intake and esophageal cancer risk: A meta-analysis. Asian Pac. J. Cancer Prev. 2013, 14, 1911–1918. [Google Scholar] [CrossRef] [PubMed]
- Grudzinski, W.; Piet, M.; Luchowski, R.; Reszczynska, E.; Welc, R.; Paduch, R.; Gruszecki, W.I. Different molecular organization of two carotenoids, lutein and zeaxanthin, in human colon epithelial cells and colon adenocarcinoma cells. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2018, 188, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Jiang, W.; Shao, L.; Zhong, D.; Wu, Y.; Cai, J. Association between intake of antioxidants and pancreatic cancer risk: A meta-analysis. Int. J. Food Sci. Nutr. 2016, 67, 744–753. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Wang, T.; Meng, Q.; Zhai, S. Association of carotenoids with risk of gastric cancer: A meta-analysis. Clin. Nutr. 2016, 35, 109–116. [Google Scholar] [CrossRef] [PubMed]
- Ho, W.J.; Simon, M.S.; Yildiz, V.O.; Shikany, J.M.; Kato, I.; Beebe-Dimmer, J.L.; Cetnar, J.P.; Bock, C.H. Antioxidant micronutrients and the risk of renal cell carcinoma in the Women’s Health Initiative cohort. Cancer 2015, 121, 580–588. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Lu, M.S.; Fang, Y.J.; Xu, M.; Huang, W.Q.; Pan, Z.Z.; Chen, Y.M.; Zhang, C.X. Serum carotenoids and colorectal cancer risk: A case-control study in Guangdong, China. Mol. Nutr. Food Res. 2017, 61. [Google Scholar] [CrossRef] [PubMed]
- Qiu, X.; Gao, D.H.; Xiang, X.; Xiong, Y.F.; Zhu, T.S.; Liu, L.G.; Sun, X.F.; Hao, L.P. Ameliorative effects of lutein on non-alcoholic fatty liver disease in rats. World J. Gastroenterol. 2015, 21, 8061–8072. [Google Scholar] [CrossRef] [PubMed]
- Melo van Lent, D.; Leermakers, E.T.M.; Darweesh, S.K.L.; Moreira, E.M.; Tielemans, M.J.; Muka, T.; Vitezova, A.; Chowdhury, R.; Bramer, W.M.; Brusselle, G.G.; et al. The effects of lutein on respiratory health across the life course: A systematic review. Clin. Nutr. ESPEN 2016, 13, e1–e7. [Google Scholar] [CrossRef] [PubMed]
- Bliuc, D.; Nguyen, N.D.; Alarkawi, D.; Nguyen, T.V.; Eisman, J.A.; Center, J.R. Accelerated bone loss and increased post-fracture mortality in elderly women and men. Osteoporos. Int. 2015, 26, 1331–1339. [Google Scholar] [CrossRef] [PubMed]
- Takeda, H.; Tominari, T.; Hirata, M.; Watanabe, K.; Matsumoto, C.; Grundler, F.M.W.; Inada, M.; Miyaura, C. Lutein Enhances Bone Mass by Stimulating Bone Formation and Suppressing Bone Resorption in Growing Mice. Biol. Pharm. Bull. 2017, 40, 716–721. [Google Scholar] [CrossRef] [PubMed]
- Tominari, T.; Matsumoto, C.; Watanabe, K.; Hirata, M.; Grundler, F.M.; Inada, M.; Miyaura, C. Lutein, a carotenoid, suppresses osteoclastic bone resorption and stimulates bone formation in cultures. Biosci. Biotechnol. Biochem. 2017, 81, 302–306. [Google Scholar] [CrossRef] [PubMed]
- Hayhoe, R.P.G.; Lentjes, M.A.H.; Mulligan, A.A.; Luben, R.N.; Khaw, K.T.; Welch, A.A. Carotenoid dietary intakes and plasma concentrations are associated with heel bone ultrasound attenuation and osteoporotic fracture risk in the European Prospective Investigation into Cancer and Nutrition (EPIC)-Norfolk cohort. Br. J. Nutr. 2017, 117, 1439–1453. [Google Scholar] [CrossRef] [PubMed]
- Grether-Beck, S.; Marini, A.; Jaenicke, T.; Stahl, W.; Krutmann, J. Molecular evidence that oral supplementation with lycopene or lutein protects human skin against ultraviolet radiation: Results from a double-blinded, placebo-controlled, crossover study. Br. J. Dermatol. 2017, 176, 1231–1240. [Google Scholar] [CrossRef] [PubMed]
- Palombo, P.; Fabrizi, G.; Ruocco, V.; Ruocco, E.; Fluhr, J.; Roberts, R.; Morganti, P. Beneficial long-term effects of combined oral/topical antioxidant treatment with the carotenoids lutein and zeaxanthin on human skin: A double-blind, placebo-controlled study. Skin Pharmacol. Physiol. 2007, 20, 199–210. [Google Scholar] [CrossRef] [PubMed]
- Morganti, P.; Bruno, C.; Guarneri, F.; Cardillo, A.; Del Ciotto, P.; Valenzano, F. Role of topical and nutritional supplement to modify the oxidative stress. Int. J. Cosmet. Sci. 2002, 24, 331–339. [Google Scholar] [CrossRef] [PubMed]
- Lorenzoni, F.; Giampietri, M.; Ferri, G.; Lunardi, S.; Madrigali, V.; Battini, L.; Boldrini, A.; Ghirri, P. Lutein administration to pregnant women with gestational diabetes mellitus is associated to a decrease of oxidative stress in newborns. Gynecol. Endocrinol. 2013, 29, 901–903. [Google Scholar] [CrossRef] [PubMed]
- Cohen, J.M.; Kramer, M.S.; Platt, R.W.; Basso, O.; Evans, R.W.; Kahn, S.R. The association between maternal antioxidant levels in midpregnancy and preeclampsia. Am. J. Obstet. Gynecol. 2015, 213, 695.e1-13. [Google Scholar] [CrossRef] [PubMed]
- Zielińska, M.A.; Wesołowska, A.; Pawlus, B.; Hamułka, J. Health effects of carotenoids during pregnancy and lactation. Nutrients 2017, 9, 838. [Google Scholar] [CrossRef] [PubMed]
- European Food Safety Authority. Scientific opinion on the re-evaluation of lutein [e 161b] as a food additive. EFSA J. 2010, 8, 1678. [Google Scholar] [CrossRef]
- Harikumar, K.B.; Nimita, C.V.; Preethi, K.C.; Kuttan, R.; Deshpande, J. Toxicity profile of lutein and lutein ester isolated from marigold flowers (Tagetes erecta). Int. J. Toxicol. 2008, 27, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Ravikrishnan, R.; Rusia, S.; Ilamurugan, G.; Salunkhe, U.; Deshpande, J.; Shankaranarayanan, J.; Shankaranarayana, M.L.; Soni, M.G. Safety assessment of lutein and zeaxanthin (Lutemax 2020): Subchronic toxicity and mutagenicity studies. Food Chem. Toxicol. 2011, 49, 2841–2848. [Google Scholar] [CrossRef] [PubMed]
- Amengual, J.; Lobo, G.P.; Golczak, M.; Li, H.N.; Klimova, T.; Hoppel, C.L.; Wyss, A.; Palczewski, K.; von Lintig, J. A mitochondrial enzyme degrades carotenoids and protects against oxidative stress. FASEB J. 2011, 25, 948–959. [Google Scholar] [CrossRef] [PubMed]
- Institute of Medicine (US) Panel on Dietary Antioxidants and Related Compounds. Dietary Reference Intakes for Vitamin C, Vitamin E, Selenium, and Carotenoids; National Academies Press (US): Washington, DC, USA, 2000. [Google Scholar]
- Joint, F.A.O. Evaluation of Certain Food Additives: Sixty-Third Report of the Joint FAO/WHO Expert Committee on Food Additives; World Health Organization: Geneva, Switzerland, 2005; pp. 23–26. [Google Scholar]
- Landrum, J.T.; Bone, R.A.; Joa, H.; Kilburn, M.D.; Moore, L.L.; Sprague, K.E. A one year study of the macular pigment: The effect of 140 days of a lutein supplement. Exp. Eye Res. 1997, 65, 57–62. [Google Scholar] [CrossRef] [PubMed]
- Dagnelie, G.; Zorge, I.S.; McDonald, T.M. Lutein improves visual function in some patients with retinal degeneration: A pilot study via the Internet. Optometry 2000, 71, 147–164. [Google Scholar] [PubMed]
- European Food Safety Authority (EFSA). Safety, bioavailability and suitability of lutein for the particular nutritional use by infants and young children—Scientific Opinion of the Panel on Dietetic Products, Nutrition and Allergies. EFSA J. 2008, 823, 1–24. [Google Scholar]
- Zheng, Y.F.; Bae, S.H.; Kwon, M.J.; Park, J.B.; Choi, H.D.; Shin, W.G.; Bae, S.K. Inhibitory effects of astaxanthin, b-cryptoxanthin, canthaxanthin, lutein, and zeaxanthin on cytochrome P450 enzyme activities. Food Chem. Toxicol. 2013, 59, 78–85. [Google Scholar] [CrossRef] [PubMed]
- Olmedilla, B.; Granado, F.; Southon, S.; Wright, A.J.; Blanco, I.; Gil-Martinez, E.; van den Berg, H.; Thurnham, D.; Corridan, B.; Chopra, M.; et al. A European multicentre, placebo-controlled supplementation study with alpha-tocopherol, carotene-rich palm oil, lutein or lycopene: Analysis of serum responses. Clin. Sci. 2002, 102, 447–456. [Google Scholar] [CrossRef] [PubMed]
- Satia, J.A.; Littman, A.; Slatore, C.G.; Galanko, J.A.; White, E. Long-term use of beta-carotene, retinol, lycopene, and lutein supplements and lung cancer risk: Results from the Vitamins and Lifestyle (VITAL) study. Am. J. Epidemiol. 2009, 169, 815–828, Erratum in Am. J. Epidemiol. 2009, 169, 1409. [Google Scholar] [CrossRef] [PubMed]
- Choi, R.Y.; Chortkoff, S.C.; Gorusupudi, A.; Bernstein, P.S. Crystalline maculopathy associated with high-dose lutein supplementation. JAMA Ophthalmol. 2016, 134, 1445–1448. [Google Scholar] [CrossRef] [PubMed]
| Food | L + Z Content (mg/100 g of Food) | L + Z Content (mg/Household) | ||
|---|---|---|---|---|
| Raw | Cooked | Raw | Cooked | |
| Paprika | 18.94 | 0.43 t | ||
| Sweet potato leaves | 14.72 | 11.45 | 5.15 c | 7.32 c |
| Dandelion | 13.61 | 9.16 | 7.48 c | 9.61 c |
| Pepper | 13.16 | 0.23 t | ||
| Turnip greens | 12.83 | 8.44 | 7.05 c | 12.15 c |
| Cress | 12.50 | 8.40 | 6.25 c | 11.34 c |
| Spinach | 12.20 | 11.31 | 3.66 c | 20.35 c |
| Chard | 11.00 | 11.02 | 3.96 c | 19.28 c |
| Chicory | 10.30 | 2.99 c | ||
| Radicchio | 8.83 | 3.53 c | ||
| Kale | 6.26 | 4.98 | 1.32 c | 5.88 c |
| Basil | 5.65 | 0.14 l | ||
| Parsley | 5.56 | 3.34 c | ||
| Collards | 4.32 | 6.2 | 1.56 c | 11.77 c |
| Mustard greens | 3.73 | 10.4 | 2.09 c | 14.56 c |
| Arugula | 3.56 | |||
| Peas | 2.48 | 2.59 | 3.59 c | 4.15 c |
| Lettuce | 2.31 | 1.09 c | ||
| Squash | 2.13 | 2.25 | 2.40 c | 4.05 c |
| Egg yolk | 1.09 | 0.19 e | ||
| Study (year) | Design (Number of Participants) | Intervention and Lutein Supplementation | Effects |
|---|---|---|---|
| AREDS2 (2013) [27] | RCT, participants with bilateral drusen or AMD in 1 eye (4176); 4 groups: G1 (1007); G2 (1038); G3 (1062); G4 (1069) | G1: AREDS formulation; G2: AREDS + L 10 mg + Z 2 mg; G3: AREDS + DHA 350 mg + EPA 650 mg G4: AREDS + L 10 mg + Z 2 mg + DHA 350 mg + EPA 650 mg | No effect in reducing progression to advanced AMD. No effect in improving VA. In the lowest quintile of L dietary intake, L + Z had significant effect vs. no L + Z in reducing progression to advanced AMD. |
| AREDS2 (2014) [105] | RCT, participants with bilateral drusen or AMD in 1 eye (3335 eyes analyzed); 3 groups: G1 (1114 eyes); G2 (1104 eyes); G3 (1117 eyes) | G1: AREDS + L 10 mg + Z 2 mg without beta-carotene; G2: AREDS + L 10 mg + Z 2 mg; G3: AREDS formulation | G1 (compared to G3) significantly reduced progression to advanced AMD and neovascular AMD, no effect for CGA. No difference between G2 vs. G3. |
| Akuffo et al. (2015) [92] | Intervention trial, participants with AMD (67); 3 groups with different dosages | G1: L 20 mg + Z 2 mg; G2: L 10 mg + Z 2 mg + MZ 10 mg; G3: L 3 mg + Z 2 mg + MZ 17 mg | After 3 years, all the groups showed a significant increase in MPOD but no effects in reducing progression to advanced AMD or improving VA. CS significantly increased, mainly in G3. |
| Beatty et al. (2013) [15] | RCT, participants with at least bilateral early AMD (433); intervention group (216) vs. placebo (217) | Intervention group: formulation containing L 12 mg | No significant improvement in CS. Significant VA enhancement not before 24 months. |
| Berrow et al. (2013) [26] | RCT, participants with AMD (14); treatment group (8) vs. no treatment (6) | Treatment group: L 12 mg | After 40 weeks, no clinical effects; only minimal improvement in mfEGR. |
| Bone (2010) [38] | Intervention trial, healthy participants (87); 4 groups: G1 (10); G2 (17); G3 (22); G4 (38) | G1: placebo; G2: L 5 mg;G3: L 10 mg; G4: 20 mg | MPOD increased in a linear, dose-dependent manner. L did not increase MPOD in all the participants. |
| Cangemi (2007) [101] | Intervention trial, participants with at least 1 eye with dry AMD (37) | Formulation containing L 8 mg | Significant improvement in VA after 6 months. |
| Dawczynski et al. (2013) [35] | RCT, participants with non-exudative AMD (145); 3 groups: G1 (50); G2 (55); G3 (40) | G1: L 10 mg + Z 1 mg + DHA 100 mg + EPA 30 mg; G2: L 20 mg + Z 2 mg + DHA 200 mg + EPA 60 mg; G3: placebo | Significant increase in MPOD and improvement in VA both in G1 and G2; MPOD decreased in G3. No significant differences in MPOD accumulation between G1 and G2. |
| Fujimura et al. (2016) [96] | Intervention trial, participants with at least 1 eye with wet AMD or early AMD (20) | Formulation containing L 20 mg + Z 1 mg + DHA 200 mg | After 6 months, significant increase in MPOD and CS. Linear correlation between MPOD and serum concentrations of L. |
| Hammond et al. (2014) [97] | RCT, healthy participants (115); intervention group (58) vs. placebo (57) | Intervention group: formulation containing L 10 mg + Z 2 mg | After 1 year, significant increase in MPOD, recovery from photostress and chromatic contrast. |
| Huang et al. (2015) [94] | RCT, participants with early AMD (108); 4 groups: G1 (28); G2 (26); G3 (27); G4 (27) | G1: placebo; G2: L 10 mg; G3: L 20 mg; G4: L 10 mg + Z 10 mg | After 2 years, significant increase in MPOD and mean retinal sensitivity. |
| Huang et al. (2015) [36] | RCT, participants with early AMD (108); 4 groups: G1 (28)]; G2 (26); G3 (27); G4 (27) | G1: placebo; G2: L 10 mg; G3: L 20 mg; G4: L 10 mg + Z 10 mg | After 2 years, significant increase in MPOD and CS, no effect in VA and flash recovery time. Same efficacy in all treatment groups. |
| Ma et al. (2009) [23] | Intervention trial, healthy participants (37); 3 groups: G1 (12); G2 (12); G3 (13) | G1: placebo; G2: L 6 mg; G3: L 12 mg | After 12 weeks, no effect in improving VA and glare sensitivity. CS significantly increased in both G2 and G3, but much more in G3. |
| Ma et al. (2012) [37] | RCT, participants with early AMD (108); 4 groups: G1 (27); G2 (27); G3 (27); G4 (27); group of healthy controls (36) | G1: placebo; G2: L 10 mg; G3: L 20 mg; G4: L 10 mg + Z 10 mg | After 48 weeks, both G3 and G4 effectively increased MPOD; CS only improved on G3. Positive correlation between MPOD increase, VA and CS. Significant dose-response effect following L supplementation. |
| Murray et al. (2013) [16] | RCT, participants with early AMD (72); intervention group (36) vs. placebo (36) | Intervention group: formulation containing L 10 mg | Significant effect on MPOD. No improvement in VA, but VA decreased on placebo. Changes in VA were significant between L and placebo. |
| Nolan et al. (2011) [98] | RCT, healthy participants (121); intervention group (61) vs. placebo (60) | Intervention group: formulation containing L 12 mg + Z 1 mg | After 1 year, significant effect on MPOD but no improvement in VA, CS, glare disability, recovery from photostress. |
| Obana et al. (2015) [25] | RCT, healthy participants (36) | L 10 mg + Z 1 mg | After 6 months, no effect on MPOD. Only a subgroup of 13 participants had an effective increase both in serum levels of L and MPOD. |
| Parisi et al. (2008) [13] | RCT, participants with non-advanced AMD (27); treatment group (15) vs. no treatment [12] | Treatment group: formulation containing L 10 mg + Z 1 mg | After 1 year, significant improvement in central retina dysfunction but no effect in peripheral retina. |
| Piermarocchi et al. (2012) [102] | Intervention trial, participants with dry AMD [109]; treatment group [84] vs. no treatment [26] | Treatment group: formulation containing L 10 mg + Z 1 mg | Significant improvement in VA and CS after 2 years. |
| Richer et al. (2004) [12] | RCT, participants with atrophic AMD (90); 3 groups: G1 (29); G2 (30); G3 (31) | G1: L 10 mg; G2: formulation containing L 10 mg; G3: placebo | After 1 year, both G1 and G2 showed significantly increased MPOD, VA and CS. |
| Richer et al. (2011) [95] | RCT, participants with non-advanced AMD (60); 3 groups: G1 (10); G2 (25); G3 (25) | G1: L 9 mg; G2: Z 8 mg; G3: L 9 mg + Z 8 mg | After 1 year, both G1 and G2 showed effectively increased MPOD and CS; no improvement in G3. |
| Rosenthal et al. (2006) [22] | Intervention trial, participants with AMD (45); 3 groups: G1 (15); G2 (15)]; G3 (15) | G1: L 2.5 mg; G2: L 5 mg; G3: L 10 mg | After 6 months, no effect in VA. 10 mg were safely administered without toxicity or adverse events. |
| Sabour-Pickett et al. (2014) [100] | Intervention trial, participants with AMD (52); 3 groups: G1 (17); G2 (21); G3 (14) | G1: L 20 mg + Z 2 mg; G2: L 10 mg + Z 2 mg + MZ 10 mg; G3: L 3 mg + Z 2 mg + MZ 17 mg | After 1 year, MPOD increased in all groups; the significant improvement in CS was much more effective in G3. |
| Sasamoto et al. (2011) [24] | Intervention trial, healthy controls (5), participants with AMD (33) and participants with central serous chorioretinopathy (5) | Formulation containing L 6 mg | After 1 year, no effect in MPOD. Significant improvement in CS and retinal sensitivity. |
| Stringham et al. (2016) [34] | RCT, healthy participants (28); 4 groups: G1 (5); G2 (7); G3 (8); G4 (8) | G1: placebo; G2: L 6 mg + Z 0,7 mg + MZ 0,5 mg; G3: L 10.9 mg+Z 1.3 mg + MZ 0.9 mg; G4: L 22 mg + Z 2.7 mg + MZ 2 mg | All the intervention groups showed a significant effect in MPOD at 12 weeks, G3 was much more effective. |
| Weigert et al. (2011) [14] | RCT, participants with AMD (126); 2 groups: G1 (84); G2 (42) | G1: L 20 mg for the first 3 months, L 10 mg for the last 3 ones; G2: placebo | After 6 months, MPOD increased by nearly 28% vs. placebo in G1. No improvement in VA and macular function. The lower MPOD at baseline, the greater the improvement. Linear correlation between changes in MPOD, VA and macular function. |
| Wolf-Schnurrbusch et al. (2015) [99] | Intervention trial, participants with AMD (79); 2 groups: G1 (40); G2 (39) | G1: formulation containing L 10 mg + Z 1 mg; G2: formulation containing L 10 mg + Z 1 mg + DHA and EPA 130 mg | After 6 months and 1 year, MPOD and CS [not VA] significantly increased G1. No significant effect on G2. |
| Yao et al. (2013) [93] | RCT, healthy participants (120); treatment group (60) vs. placebo (60) | Treatment group: L 20 mg | After 1 year, significant improvement in MPOD, CS and glare sensitivity vs. placebo. No effect in VA. |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Buscemi, S.; Corleo, D.; Di Pace, F.; Petroni, M.L.; Satriano, A.; Marchesini, G. The Effect of Lutein on Eye and Extra-Eye Health. Nutrients 2018, 10, 1321. https://doi.org/10.3390/nu10091321
Buscemi S, Corleo D, Di Pace F, Petroni ML, Satriano A, Marchesini G. The Effect of Lutein on Eye and Extra-Eye Health. Nutrients. 2018; 10(9):1321. https://doi.org/10.3390/nu10091321
Chicago/Turabian StyleBuscemi, Silvio, Davide Corleo, Francesco Di Pace, Maria Letizia Petroni, Angela Satriano, and Giulio Marchesini. 2018. "The Effect of Lutein on Eye and Extra-Eye Health" Nutrients 10, no. 9: 1321. https://doi.org/10.3390/nu10091321
APA StyleBuscemi, S., Corleo, D., Di Pace, F., Petroni, M. L., Satriano, A., & Marchesini, G. (2018). The Effect of Lutein on Eye and Extra-Eye Health. Nutrients, 10(9), 1321. https://doi.org/10.3390/nu10091321





