Tocotrienol-Rich Vitamin E from Palm Oil (Tocovid) and Its Effects in Diabetes and Diabetic Nephropathy: A Pilot Phase II Clinical Trial
Abstract
1. Introduction
2. Materials and Methods
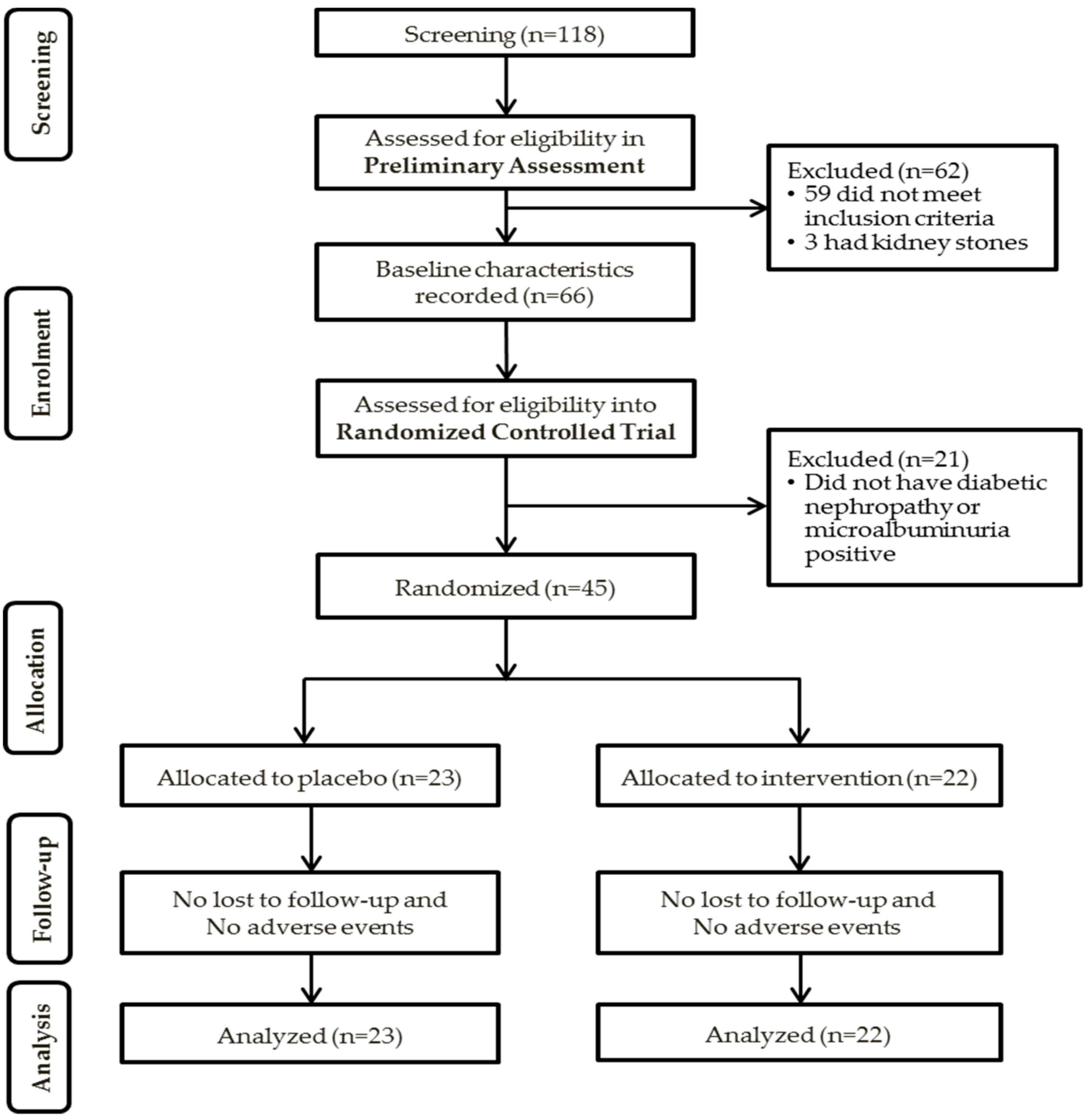
2.1. Participants and Study Enrolment
2.2. Preliminary Assessment
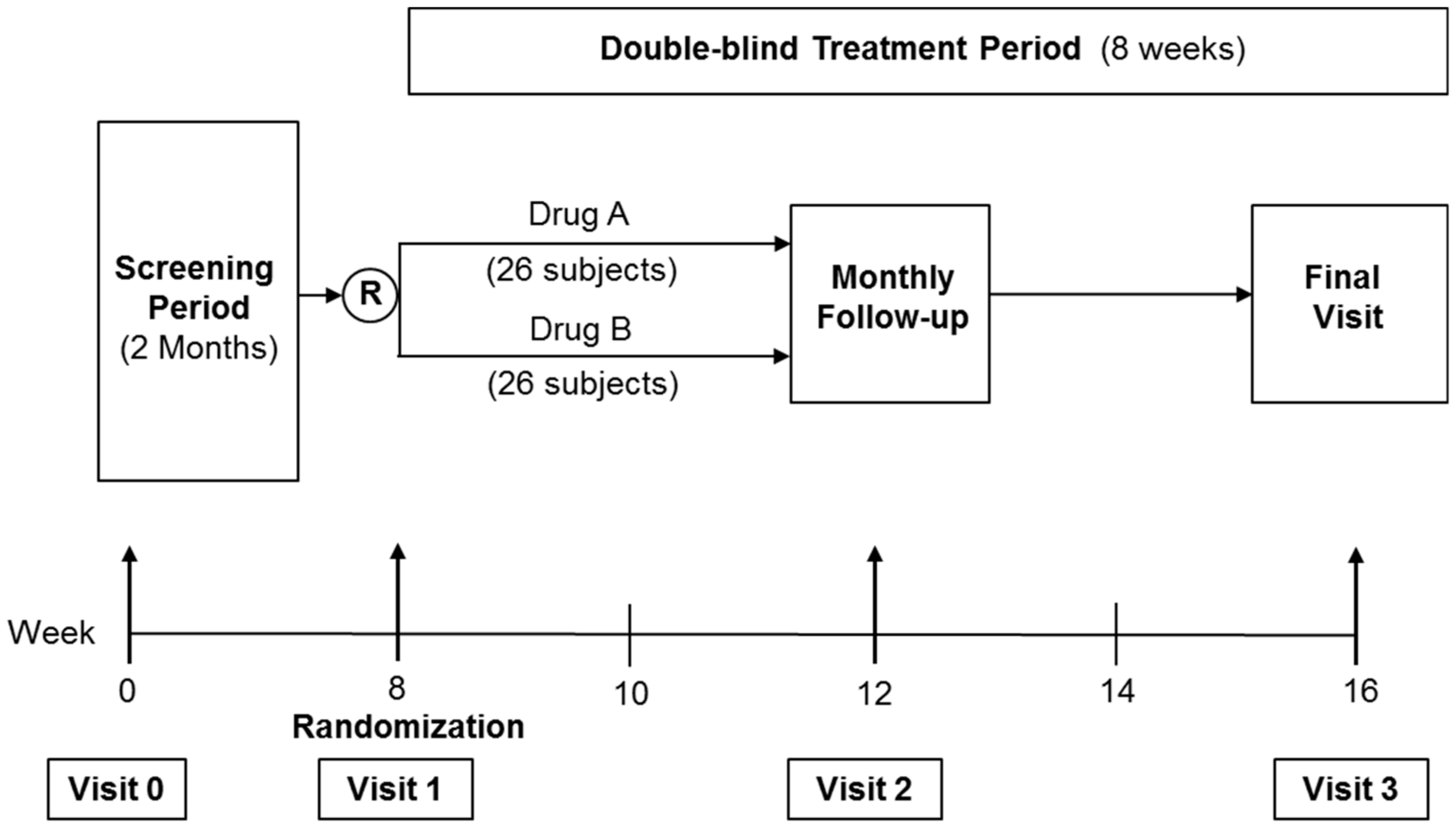
2.3. Randomized Controlled Trial (RCT)
2.4. Sample Size
2.5. Randomization
2.6. Follow-Up Visits
2.7. Assessment of Outcomes
2.8. HbA1c
2.9. BUSE Creatinine, Lipid Profile, and Liver Function Test
2.10. AGE, sRAGE, Nε-CML, and Cystatin C
2.11. UACR
2.12. Statistical Analysis
3. Results
3.1. Preliminary Assessment
3.1.1. Correlations with UACR Grades
3.1.2. Correlations with Serum Creatinine and eGFR
3.1.3. Correlations with Nε-CML
3.1.4. Univariate and Multivariate Analysis
3.2. Randomized Controlled Trial
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- World Health Organization (WHO). Global Report on Diabetes; World Health Organization: Geneva, Switzerland, 2016; ISBN 9789241565257. [Google Scholar]
- Bolignano, D.; Cernaro, V.; Gembillo, G.; Baggetta, R.; Buemi, M.; D’Arrigo, G. Antioxidant agents for delaying diabetic kidney disease progression: A systematic review and meta-analysis. PLoS ONE 2017, 12, e0178699. [Google Scholar] [CrossRef] [PubMed]
- Collins, A.J.; Foley, R.N.; Chavers, B.; Gilbertson, D.; Herzog, C.; Ishani, A.; Johansen, K.; Kasiske, B.L.; Kutner, N.; Liu, J.; et al. US Renal Data System 2013 Annual Data Report: Atlas of End-Stage Renal Disease in the United States. Am. J. Kidney Dis. 2014, 63, A4. [Google Scholar] [CrossRef] [PubMed]
- Weyer, C.; Bogardus, C.; Mott, D.M.; Pratley, R.E. The natural history of insulin secretory dysfunction and insulin resistance in the pathogenesis of type 2 diabetes mellitus. J. Clin. Investig. 1999, 104, 787–794. [Google Scholar] [CrossRef] [PubMed]
- Etgen, T.; Chonchol, M.; Forstl, H.; Sander, D. Chronic Kidney Disease and Cognitive Impairment: A Systematic Review and Meta-Analysis. Am. J. Nephrol. 2012, 35, 474–482. [Google Scholar] [CrossRef] [PubMed]
- Perlman, R.; Finkelstein, F.; Liu, L.; Roys, E.; Kiser, M.; Eisele, G. Quality of life in chronic kidney disease (CKD): A cross-sectional analysis in the Renal Research Institute-CKD study. Am. J. Kidney Dis. 2005, 45, 658–666. [Google Scholar] [CrossRef] [PubMed]
- Chin, H.; Song, Y.; Lee, J.; Lee, S.; Kim, K.; Na, K. Moderately decreased renal function negatively affects the health-related quality of life among the elderly Korean population: A population-based study. Nephrol. Dial. Transplant. 2008, 23, 2810–2817. [Google Scholar] [CrossRef] [PubMed]
- Khatami, P.G.; Soleimani, A.; Sharifi, N.; Aghadavod, E.; Asemi, Z. The effects of high-dose vitamin E supplementation on biomarkers of kidney injury, inflammation, and oxidative stress in patients with diabetic nephropathy: A randomized, double-blind, placebo-controlled trial. J. Clin. Lipidol. 2016, 10, 922–929. [Google Scholar] [CrossRef] [PubMed]
- Peh, H.Y.; Tan, W.S.; Liao, W.; Wong, W.S. Vitamin E therapy beyond cancer: Tocopherol versus tocotrienol. Pharmacol. Ther. 2016, 162, 152–169. [Google Scholar] [CrossRef] [PubMed]
- Lonn, E.; Yusuf, S.; Hoogwerf, B.; Pogue, J.; Yi, Q.; Zinman, B.; Bosch, J.; Dagenais, G.; Mann, J.F.; Gerstein, H.C.; et al. Effects of vitamin E on cardiovascular and microvascular outcomes in high-risk patients with diabetes: Results of the HOPE study and MICRO-HOPE substudy. Diabetes Care 2002, 25, 1919–1927. [Google Scholar] [CrossRef] [PubMed]
- Siddiqui, S.; Ahsan, H.; Khan, M.R.; Siddiqui, W.A. Protective effects of tocotrienols against lipid-induced nephropathy in experimental type-2 diabetic rats by modulation in TGF-beta expression. Toxicol. Appl. Pharmacol. 2013, 273, 314–324. [Google Scholar] [CrossRef] [PubMed]
- Kuhad, A.; Chopra, K. Attenuation of diabetic nephropathy by tocotrienol: Involvement of NFkB signaling pathway. Life Sci. 2009, 84, 296–301. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.S.; Ton, S.H.; Tan, J.B.L.; Abdul Kadir, K. The Ameliorative Effects of a Tocotrienol-Rich Fraction on the AGE-RAGE Axis and Hypertension in High-Fat-Diet-Fed Rats with Metabolic Syndrome. Nutrients 2017, 9, 984. [Google Scholar] [CrossRef] [PubMed]
- Nowotny, K.; Jung, T.; Hohn, A.; Weber, D.; Grune, T. Advanced glycation end products and oxidative stress in type 2 diabetes mellitus. Biomolecules 2015, 5, 194–222. [Google Scholar] [CrossRef] [PubMed]
- Yamagishi, S.I.; Nakamura, N.; Matsui, T. Glycation and cardiovascular disease in diabetes: A perspective on the concept of metabolic memory. J. Diabetes 2017, 9, 141–148. [Google Scholar] [CrossRef] [PubMed]
- UK Prospective Diabetes Study (UKPDS). Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). Lancet 1998, 352, 837–853. [Google Scholar] [CrossRef]
- The Diabetes Control and Complications Trial Research Group (DCCT). The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. N. Engl. J. Med. 1993, 329, 977–986. [Google Scholar] [CrossRef] [PubMed]
- Nathan, D.M.; DCCT/EDIC Research Group. The Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications Study at 30 Years: Overview. Diabetes Care 2014, 37, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Holman, R.R.; Paul, S.K.; Bethel, M.A.; Matthews, D.R.; Neil, H.A. 10-Year Follow-up of Intensive Glucose Control in Type 2 Diabetes. N. Engl. J. Med. 2008, 359, 1577–1589. [Google Scholar] [CrossRef] [PubMed]
- Aso, Y.; Inukai, T.; Tayama, K.; Takemura, Y. Serum concentrations of advanced glycation endproducts are associated with the development of atherosclerosis as well as diabetic microangiopathy in patients with type 2 diabetes. Acta Diabetol. 2000, 37, 87–92. [Google Scholar] [CrossRef] [PubMed]
- Ono, Y.; Aoki, S.; Ohnishi, K.; Yasuda, T.; Kawano, K.; Tsukada, Y. Increased serum levels of advanced glycation end-products and diabetic complications. Diabetes Res. Clin. Pract. 1998, 41, 131–137. [Google Scholar] [CrossRef]
- Genuth, S.; Sun, W.; Cleary, P.; Sell, D.R.; Dahms, W.; Malone, J.; Sivitz, W.; Monnier, V.M.; DCCT Skin Collagen Ancillary Study Group. Glycation and carboxymethyllysine levels in skin collagen predict the risk of future 10-year progression of diabetic retinopathy and nephropathy in the diabetes control and complications trial and epidemiology of diabetes interventions and complications participants with type 1 diabetes. Diabetes 2005, 54, 3103–3111. [Google Scholar] [PubMed]
- Lee, E.J.; Park, J.H. Receptor for Advanced Glycation Endproducts (RAGE), Its Ligands, and Soluble RAGE: Potential Biomarkers for Diagnosis and Therapeutic Targets for Human Renal Diseases. Genomics Inform. 2013, 11, 224–229. [Google Scholar] [CrossRef] [PubMed]
- Kajikawa, M.; Nakashima, A.; Fujimura, N.; Maruhashi, T.; Iwamoto, Y.; Iwamoto, A.; Matsumoto, T.; Oda, N.; Hidaka, T.; Kihara, Y.; et al. Ratio of serum levels of AGEs to soluble form of RAGE is a predictor of endothelial function. Diabetes Care 2015, 38, 119–125. [Google Scholar] [CrossRef] [PubMed]
- Campion, C.G.; Sanchez-Ferras, O.; Batchu, S.N. Potential Role of Serum and Urinary Biomarkers in Diagnosis and Prognosis of Diabetic Nephropathy. Can. J. Kidney Health Dis. 2017, 4, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Wan Nazaimoon, W.M.; Sakinah, O.; Gapor, A.; Khalid, B.A.K. Effects of palm olein tocopherol and tocotrienol on lipid peroxidation, lipid profiles and glycemic control in non-insulin diabetes mellitus patients. Nutr. Res. 1996, 16, 1901–1911. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Obesity: Preventing and Managing the Global Epidemic: Report of a WHO Consultation; WHO Technical Report Series 894; World Health Organ: Geneva, Switzerland, 2000; pp. 1–253. [Google Scholar]
- Wagner, Z.; Wittmann, I.; Mazak, I.; Schinzel, R.; Heidland, A. Nϵ-(carboxymethyl)lysine levels in patients with type 2 diabetes: Role of renal function. Am. J. Kidney Dis. 2001, 38, 785–791. [Google Scholar] [CrossRef] [PubMed]
- Hirata, K.; Kubo, K. Relationship between Blood levels of CML and pentosidine and the severity of Microangiopathy in Type 2 diabetes. Endocr. J. 2004, 51, 537–544. [Google Scholar] [CrossRef] [PubMed]
- Coughlan, M.T.; Patel, S.K.; Jerums, G.; Penfold, S.A.; Nguyen, T.V.; Sourris, K.C.; Panagiotopoulos, S.; Srivastava, P.M.; Cooper, M.E.; Burrell, L.M.; et al. Advanced glycation urinary protein-bound biomarkers and severity of diabetic nephropathy in man. Am. J. Nephrol. 2011, 34, 347–355. [Google Scholar] [CrossRef] [PubMed]
- Boesten, D.M.P.H.J.; Elie, A.G.I.M.; Drittij-Reijnders, M.J.; den Hartog, G.J.M.; Bast, A. Effect of Nε-carboxymethyllysine on oxidative stress and the glutathione system in beta cells. Toxicol. Rep. 2014, 1, 973–980. [Google Scholar] [CrossRef] [PubMed]
- Fu, M.; Requena, J.; Jenkins, A.; Lyons, T.; Baynes, J.; Thorpe, S. The Advanced Glycation End Product, NGraphic-(Carboxymethyl)lysine, Is a Product of both Lipid Peroxidation and Glycoxidation Reactions. J. Biol. Chem. 1996, 271, 9982–9986. [Google Scholar] [CrossRef] [PubMed]
- Duckworth, W.; Abraira, C.; Moritz, T.; Reda, D.; Emanuele, N.; Reaven, P.D. Glucose Control and Vascular Complications in Veterans with Type 2 Diabetes. N. Engl. J. Med. 2009, 360, 129–139. [Google Scholar] [CrossRef] [PubMed]
- Margolis, K.L.; O’Connor, P.J.; Morgan, T.M.; Buse, J.B.; Cohen, R.M.; Cushman, W.C.; Cutler, J.A.; Evans, G.W.; Gerstein, H.C.; Grimm, R.H., Jr.; et al. Outcomes of combined cardiovascular risk factor management strategies in type 2 diabetes: The ACCORD randomized trial. Diabetes Care 2014, 37, 1721–1728. [Google Scholar] [CrossRef] [PubMed]
- ADVANCE Collaborative Group; Patel, A.; MacMahon, S.; Chalmers, J.; Neal, B.; Billot, L.; Woodward, M.; Marre, M.; Cooper, M.; Glasziou, P.; et al. Intensive blood glucose control and vascular outcomes in patients with type 2 diabetes. N. Engl. J. Med. 2008, 358, 2560–2572. [Google Scholar] [CrossRef] [PubMed]
- UK Prospective Diabetes Study (UKPDS). VIII. Study design, progress and performance. Diabetologia 1991, 34, 877–890. [Google Scholar]
| Baseline Characteristics | Participants (N = 66) |
|---|---|
| Gender, n (%) | |
| Male | 48 (72.7) |
| Female | 18 (27.3) |
| Race, n (%) | |
| Malay | 34 (51.5) |
| Chinese | 14 (21.2) |
| Indian | 18 (27.3) |
| Age (years) * | 61.6 ± 9.5 |
| Duration of DM (years) * | 18.5 ± 8.9 |
| HbA1c (%) * | 8.9 ± 1.7 |
| SBP (mmHg) * | 136.7 ± 15.3 |
| DBP (mmHg) * | 77.2 ± 9.1 |
| BMI (kg/m2) * | 29 ± 4.8 |
| UACR Grade (mg/mmol) | N (%) | AGE (µg/mL) | sRAGE (pg/mL) | Nε-CML *,† (ng/mL) | Cystatin C (ng/mL) |
|---|---|---|---|---|---|
| 0–29 | 29 (43.9) | 61.4 ± 95.8 | 956.5 ± 467.8 | 1.46 ± 0.36 | 2235.1 ± 956.7 |
| 30–149 | 29 (43.9) | 159.3 ± 191.6 | 1131.9 ± 395.4 | 2.85 ± 0.36 | 1933.2 ± 906.8 |
| 150–299 | 5 (7.6) | 90.9 ± 65.3 | 1301.9 ± 643.8 | 1.92 ± 0.90 | 1767.7 ± 1114.9 |
| ≥300 | 3 (4.5) | 12.7 ± 9.9 | 1020.6 ± 443.4 | 3.41 ± 1.10 | 2043.8 ± 928.2 |
| UACR Grade (mg/mmol) | N (%) | HbA1c (%) | SBP (mmHg) | DBP (mmHg) | Age (years) | Duration of Diabetes (years) |
|---|---|---|---|---|---|---|
| 0–29 | 29 (43.9) | 8.7 ± 1.6 | 136 ± 11 | 76 ± 8 | 62.5 ± 7.8 | 17.8 ± 9.4 |
| 30–149 | 29 (43.9) | 8.9 ± 1.9 | 136 ± 18 | 79 ± 10 | 61.7 ± 10.4 | 18.8 ± 8.6 |
| 150–299 | 5 (7.6) | 10.2 ± 2.2 | 140 ± 23 | 75 ± 8 | 54.6 ± 14.8 | 20 ± 11.2 |
| ≥300 | 3 (4.5) | 8.9 ± 0.4 | 143 ± 8 | 81 ± 10 | 62.3 ± 2.1 | 20.3 ± 5.7 |
| Baseline Parameters | Serum Creatinine (mmHg) | eGFR (mL/min/1.73 m2) | ||
|---|---|---|---|---|
| Correlation, r | p-Value | Correlation, r | p-Value | |
| AGE (µg/mL) | 0.140 | 0.287 | −0.145 | 0.269 |
| sRAGE (pg/mL) | −0.199 | 0.121 | 0.185 | 0.151 |
| Nε-CML (ng/mL) | 0.31 | 0.015 * | −0.30 | 0.032 * |
| Cystatin C (ng/mL) | −0.238 | 0.061 | 0.164 | 0.199 |
| HbA1c (%) † | 0.116 | 0.369 | −0.049 | 0.704 |
| SBP (mmHg) ‡ | 0.182 | 0.153 | −0.126 | 0.325 |
| DBP (mmHg) ‡ | 0.086 | 0.504 | −0.017 | 0.893 |
| Baseline Parameters | Nε-CML (ng/mL) | |
|---|---|---|
| Correlation, r | p-Value | |
| HbA1c (%) | −0.08 | 0.522 |
| Age (years) | −0.20 | 0.107 |
| Duration of diabetes (years) | 0.06 | 0.625 |
| AGE (µg/mL) ‡ | 0.500 | 0.000 * |
| sRAGE (pg/mL) ‡ | −0.094 | 0.476 |
| Cystatin C (ng/mL) ‡ | −0.180 | 0.169 |
| Baseline Parameters | Simple Logistic Regression | Multiple Logistic Regression a | ||||
|---|---|---|---|---|---|---|
| B | Crude OR (95% CI) | p-Value | B | Adjusted OR ‡ (95% CI) | p-Value | |
| HbA1c (%) | 0.132 | 1.141 (0.852, 1.527) | 0.377 | |||
| SBP (mmHg) | 0.006 | 0.730 (0.974, 1.039) | 0.730 | |||
| DBP (mmHg) | 0.033 | 1.033 (0.978, 1.092) | 0.246 * | |||
| AGE (µg/mL) | 0.004 | 1.004 (1.000, 1.009) | 0.049 * | |||
| sRAGE (pg/mL) | 0.001 | 1.001 (1.000, 1.002) | 0.099 * | |||
| Nε-CML (ng/mL) | 0.357 | 1.429 (1.106, 1.845) | 0.006 * | 0.389 | 1.476 (1.112, 1.996) | 0.008 ** |
| Cystatin C (ng/mL) | 0.000 | 1.000 (0.999, 1.000) | 0.143 * | |||
| General Characteristics | Placebo Group (N = 23) | Tocovid Group (N = 22) | p-Value |
|---|---|---|---|
| Gender | 0.586 | ||
| Male (%) | 15 (65.2) | 16 (72.7) | |
| Female (%) | 8 (34.8) | 6 (27.30 | |
| Race | 0.895 | ||
| Malay (%) | 14 (60.9) | 12 (54.5) | |
| Chinese (%) | 5 (21.7) | 6 (27.3) | |
| Indian (%) | 4 (17.4) | 4 (18.2) | |
| Age (years) | 63.3 ± 10.42 | 59.9 ± 10.24 | 0.283 |
| Duration of DM (years) | 17.9 ± 7.65 | 18.2 ± 10 | 0.893 |
| HbA1c (%) | 8.7 ± 1.5 | 9.0 ± 2 | 0.611 |
| SBP (mmHg) | 138.8 ± 15 | 136.2 ± 18.4 | 0.601 |
| DBP (mmHg) | 78.5 ± 9.4 | 77.0 ± 10.2 | 0.617 |
| Weight (kg) | 78.3 ± 12.8 | 78.2 ± 16.5 | 0.983 |
| BMI (kg/m2) | 29.3 ± 4.7 | 29.4 ± 5.4 | 0.978 |
| Renal Parameters: | |||
| UACR (mg/mmol) | 128.7 ± 164.7 | 66.4 ± 61.8 | 0.101 |
| Serum Creatinine (umol/L) | 125.5 ± 56.6 | 120.2 ± 57.9 | 0.761 |
| eGFR (mL/min/1.73 m²) | 57.5 ± 25.1 | 63.1 ± 24.1 | 0.445 |
| Biomarkers: | |||
| AGE (µg/mL) | 112.2 ± 149.5 | 136.1 ± 188.4 | 0.646 |
| sRAGE (pg/mL) | 1060.9 ± 438.8 | 1099.9 ± 408 | 0.759 |
| Nε-CML (ng/mL) | 2.4 ± 2.2 | 2.7 ± 2.5 | 0.695 |
| Cystatin C (ng/mL) | 1947.5 ± 1078 | 1941.3 ± 837 | 0.983 |
| Safety Tests: | |||
| Urea (mmol/L) | 8.4 ± 4.7 | 6.3 ± 3.5 | 0.095 |
| Total chol (mmol/L) | 4.2 ± 0.9 | 4.6 ± 0.9 | 0.192 |
| HDL (mmol/L) | 1.1 ± 0.3 | 1.2 ± 0.2 | 0.728 |
| AST (UI/L) | 19.2 ± 6.9 | 25.4 ± 9.1 | 0.013 * |
| ALT (UI/L) | 19.3 ± 11 | 29 ± 15.7 | 0.020 * |
| Analytes | Placebo Group | Tocovid Group | Mean Difference | p-Value † |
|---|---|---|---|---|
| HbA1c (%) | 8.61 ± 0.17 | 8.45 ± 0.17 | −0.16 ± 0.24 | 0.518 |
| SBP (mmHg) | 137.09 ± 2.81 | 130.48 ± 2.88 | −6.62 ± 4.03 | 0.108 |
| DBP (mmHg) | 77.61 ± 1.78 | 77.91 ± 1.82 | 0.297 ± 2.57 | 0.909 |
| Weight (kg) | 78.22 ± 0.84 | 79.47 ± 0.86 | 1.24 ± 1.20 | 0.308 |
| Renal parameters: | ||||
| UACR (mg/mmol) | 66.93 ± 8.93 | 85.43 ± 9.14 | 18.45 ± 12.97 | 0.161 |
| Sr Creatinine (μmol/L) | 131.04 ± 2.92 | 119.76 ± 2.92 | −11.28 ± 4.31 | * 0.014 ‡ |
| eGFR (mL/min/1.73 m²) | 74.89 ± 11.51 | 63.57 ± 11.77 | −11.31 ± 16.51 | 0.497 |
| Serum biomarkers: | ||||
| AGE (µg/mL) | 83.66 ± 27.20 | 89.82 ± 27.20 | 6.16 ± 38.50 | 0.874 |
| sRAGE (pg/mL) | 1088.32 ± 111.65 | 1246.36 ± 114.16 | 158.05 ± 159.77 | 0.328 |
| Nε-CML (ng/mL) | 2.56 ± 0.48 | 2.59 ± 0.49 | 0.28 ± 0.69 | 0.967 |
| Cystatin C (ng/mL) | 2172.45 ± 181.56 | 2031.99 ± 185.64 | −140.46 ± 259.66 | 0.591 |
| Safety tests: | ||||
| Urea (mmol/L) | 7.33 ± 0.37 | 7.26 ± 0.38 | −0.07 ± 0.54 | 0.896 |
| Total chol (mmol/L) | 4.52 ± 0.12 | 4.67 ± 0.12 | 0.15 ± 0.17 | 0.384 |
| HDL (mmol/L) | 1.17 ± 0.03 | 1.18 ± 0.03 | 0.01 ± 0.04 | 0.772 |
| AST (IU/L) | 21.21 ± 0.91 | 19.41 ± 0.93 | −1.80 ± 1.35 | 0.190 |
| ALT (IU/L) | 26.45 ± 1.67 | 22.17 ± 1.71 | −4.28 ± 2.46 | 0.089 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tan, S.M.Q.; Chiew, Y.; Ahmad, B.; Kadir, K.A. Tocotrienol-Rich Vitamin E from Palm Oil (Tocovid) and Its Effects in Diabetes and Diabetic Nephropathy: A Pilot Phase II Clinical Trial. Nutrients 2018, 10, 1315. https://doi.org/10.3390/nu10091315
Tan SMQ, Chiew Y, Ahmad B, Kadir KA. Tocotrienol-Rich Vitamin E from Palm Oil (Tocovid) and Its Effects in Diabetes and Diabetic Nephropathy: A Pilot Phase II Clinical Trial. Nutrients. 2018; 10(9):1315. https://doi.org/10.3390/nu10091315
Chicago/Turabian StyleTan, Suzanne May Quinn, Yilynn Chiew, Badariah Ahmad, and Khalid Abdul Kadir. 2018. "Tocotrienol-Rich Vitamin E from Palm Oil (Tocovid) and Its Effects in Diabetes and Diabetic Nephropathy: A Pilot Phase II Clinical Trial" Nutrients 10, no. 9: 1315. https://doi.org/10.3390/nu10091315
APA StyleTan, S. M. Q., Chiew, Y., Ahmad, B., & Kadir, K. A. (2018). Tocotrienol-Rich Vitamin E from Palm Oil (Tocovid) and Its Effects in Diabetes and Diabetic Nephropathy: A Pilot Phase II Clinical Trial. Nutrients, 10(9), 1315. https://doi.org/10.3390/nu10091315




