Wholegrain Intake and Risk of Type 2 Diabetes: Evidence from Epidemiological and Intervention Studies
Abstract
1. Introduction
2. Methods
3. Wholegrain Intake and T2DM Prevention
3.1. Epidemiological Studies
3.2. Intervention Trials
4. Wholegrain Intake and T2DM Risk Factors
4.1. Body Weight/Body Fat
4.2. Metabolic Syndrome/Insulin Resistance
- (1)
- The metabolic impact of wholegrain is mostly confined to the postprandial period; this has been clearly shown by an intervention trial from our group [61] that compared a diet based on wholegrain cereal foods with a refined cereal diet of identical nutrient composition. The wholegrain diet significantly reduced the postprandial insulin response by as much as 30%, in comparison with the control diet, despite similar postprandial glucose levels. In the same study, fasting insulin sensitivity was evaluated by both the HOMA index and the insulin sensitivity index measured during an Intravenous Glucose Tolerance Test was not at all influenced by the wholegrain intake [57]. The impact of wholegrain, specifically on postprandial insulin metabolism, has been recently confirmed in obese people at risk of T2DM [58]. Further support to the importance of the postprandial metabolism, in evaluating the impact of wholegrain on insulin sensitivity, comes from a study in which objective markers of wholegrain wheat or rye intake were employed and insulin sensitivity was evaluated after an oral glucose challenge. In this study, the alkylresorcinol C17:0/C21:0 ratio measured in non-fasting conditions was positively associated with insulin sensitivity indices measured after the oral glucose load—Matsuda ISI (p = 0.026) and disposition index (p = 0.022)—in a pooled analysis of the wholegrain and the control diet groups at the end of the intervention, and after adjustment for confounders [62].
- (2)
- Study duration has to be long enough to allow stable changes in the intestinal ecosystem that may eventually optimize the fermentation of fiber from wholegrain cereals.
- (3)
- The metabolic benefits may be more relevant for cereal types that provide larger amounts of indigestible carbohydrates, and particularly, types of dietary fiber that are more fermentable in the gut (i.e., oat, barley) [63].
4.3. Blood Glucose Regulation
5. Effects of Wholegrain Consumption in Patients with T2DM
5.1. Observational Studies
5.2. Intervention Trials
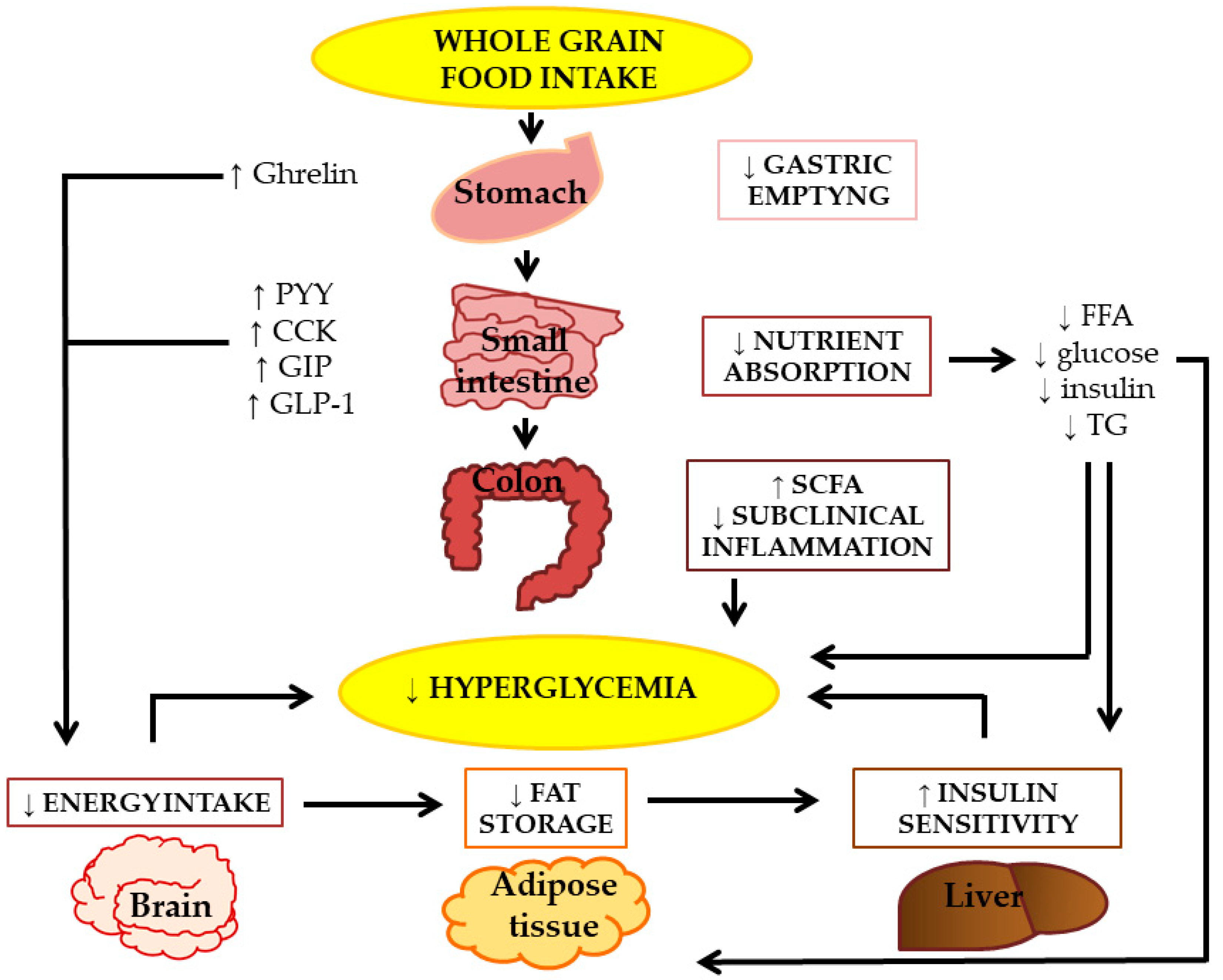
6. Plausible Mechanisms by Which Wholegrains Might Protect against T2DM
7. Conclusions
Author Contributions
Conflicts of Interest
Abbreviations
| T2DM | Type 2 Diabetes Mellitus |
| HbA1c | Glycated Hemoglobin A1c |
| BMI | Body Mass Index |
| LDL | Low Density Lipoprotein |
| HDL | High Density Lipoprotein |
| HOMA | Homeostatic Model Assessment |
References
- Ogurtsova, K.; da Rocha Fernandes, J.D.; Huang, Y.; Linnenkamp, U.; Guariguata, L.; Cho, N.H.; Cavan, D.; Shaw, J.E.; Makaroff, L.E. IDF Diabetes Atlas: Global estimates for the prevalence of diabetes for 2015 and 2040. Diabetes Res. Clin. Pract. 2017, 128, 40–50. [Google Scholar] [CrossRef] [PubMed]
- Bommer, C.; Heesemann, E.; Sagalova, V.; Manne-Goehler, J.; Atun, R.; Bärnighausen, T.; Vollmer, S. The global economic burden of diabetes in adults aged 20–79 years: A cost-of-illness study. Lancet Diabetes Endocrinol. 2017, 6, 423–430. [Google Scholar] [CrossRef]
- Skyler, J.S.; Bakris, G.L.; Bonifacio, E.; Darsow, T.; Eckel, R.H.; Groop, L.; Groop, P.H.; Handelsman, Y.; Insel, R.A.; Mathieu, C.; et al. Differentiation of diabetes by pathophysiology, natural history, and prognosis. Diabetes 2017, 66, 241–255. [Google Scholar] [CrossRef] [PubMed]
- Barroso, T.A.; Braga Marins, L.; Alves, R.; Souza Gonçalves, A.C.; Barroso, S.G.; de Souza Rocha, G. Association of central obesity with the incidence of cardiovascular diseases and risk factors. Int. J. Cardiovasc. Sci. 2017, 30, 416–424. [Google Scholar] [CrossRef]
- American Diabetes Association. Lifestyle Management: Standards of Medical Care in Diabetes. Diabetes Care 2018, 41, S51–S54. [Google Scholar] [CrossRef]
- Mozaffarrian, D. Dietary and policy priorities for cardiovascular disease, diabetes, and obesity—A comprehensive review. Circulation 2016, 2, 187–225. [Google Scholar] [CrossRef] [PubMed]
- Schwingshackl, L.; Hoffmann, G.; Lampousi, A.M.; Knüppel, S.; Iqbal, K.; Schwedhelm, C.; Bechthold, A.; Schlesinger, S.; Boeing, H. Food groups and risk of type 2 diabetes mellitus: A systematic review and meta-analysis of prospective studies. Eur. J. Epidemiol. 2017, 32, 363–375. [Google Scholar] [CrossRef] [PubMed]
- Salas-Salvadó, J.; Bulló, M.; Estruch, R.; Ros, E.; Covas, M.I.; Ibarrola-Jurado, N.; Corella, D.; Arós, F.; Gómez-Gracia, E.; Ruiz-Gutiérrez, V.; et al. Prevention of diabetes with Mediterranean diets: A subgroup analysis of a randomized trial. Ann. Intern. Med. 2014, 160, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Seal, C.J.; Brownlee, I.A. Whole-grain foods and chronic disease: Evidence from epidemiological and intervention studies. Proc. Nutr. Soc. 2015, 74, 313–319. [Google Scholar] [CrossRef] [PubMed]
- American Association of Cereal Chemists International. Whole Grain Definition. Available online: http://www.aaccnet.org/initiatives/definitions/Pages/WholeGrain.aspx (accessed on 24 August 2018).
- Ross, A.B.; van der Kamp, J.W.; King, R.; Lê, K.A.; Mejborn, H.; Seal, K.J.; Thielecke, F. Perspective: A definition for whole-grain food products—Recommendations from the healthgrain forum. Adv. Nutr. 2017, 8, 525–531. [Google Scholar] [PubMed]
- van der Kamp, J.W.; Poutanen, K.; Seal, C.J.; Richardson, D.P. The HEALTHGRAIN definition of “whole grain”. Food Nutr. Res. 2014, 58, 22100. [Google Scholar] [CrossRef] [PubMed]
- Composition of Foods Raw, Processed, Prepared USDA National Nutrient Database for Standard Reference, Release 20. Available online: https://ndb.nal.usda.gov/ndb (accessed on 24 August 2018).
- Belobrajdic, D.P.; Bird, A.R. The potential role of phytochemicals in wholegrain cereals for the prevention of type-2 diabetes. Nutr. J. 2013, 12, 62. [Google Scholar] [CrossRef] [PubMed]
- Rouf Shah, T.; Prasad, K.; Kumar, P. Maize—A potential source of human nutrition and health: A review. Cogent Food Agric. 2016, 2, 1166995. [Google Scholar] [CrossRef]
- Mattila, P.; Pihlava, J.M.; Hellström, J. Contents of phenolic acids, alkyl- and alkenylresorcinols, and avenanthramides in commercial grain products. J. Agric. Food Chem. 2005, 53, 8290–8295. [Google Scholar] [CrossRef] [PubMed]
- Ferruzzi, M.G.; Jonnalagadda, S.S.; Liu, S.; Marquart, L.; McKeown, N.; Reicks, M.; Riccardi, G.; Seal, C.; Slavin, J.; Thielecke, F.; et al. Developing a standard definition of whole-grain foods for dietary recommendations: Summary report of a multidisciplinary expert roundtable discussion. Adv. Nutr. 2014, 5, 164–176. [Google Scholar] [CrossRef] [PubMed]
- Slavin, J. Why wholegrains are protective: Biological mechanisms. Proc. Nutr. Soc. 2003, 62, 129–134. [Google Scholar] [CrossRef] [PubMed]
- Develaraja, S.; Reddy, A.; Yadav, M.; Jain, S.; Yadav, H. Whole grains in amelioration of metabolic derangements. J. Nutr. Health Food Sci. 2016, 4, 1–11. [Google Scholar]
- Priebe, M.G.; van Binsbergen, J.J.; de Vos, R.; Vonk, R.J. Whole grain foods for the prevention of type 2 diabetes mellitus. Cochrane Database Syst. Rev. 2008, 23, CD006061. [Google Scholar]
- Ye, E.Q.; Chacko, S.A.; Chou, E.L.; Kugizaki, M.; Liu, S. Greater whole-grain intake is associated with lower risk of type 2 diabetes, cardiovascular disease, and weight gain. J. Nutr. 2012, 142, 1304–1313. [Google Scholar] [CrossRef] [PubMed]
- Aune, D.; Norat, T.; Romundstad, P.; Vatten, L.J. Whole grain and refined grain consumption and the risk of type 2 diabetes: A systematic review and dose-response meta-analysis of cohort studies. Eur. J. Epidemiol. 2013, 28, 845–858. [Google Scholar] [CrossRef] [PubMed]
- Sun, T.; Rong, Y.; Hu, X.; Zhu, Y.; Huang, H.; Chen, L.; Li, P.; Li, S.; Yang, W.; Cheng, J.; et al. Plasma alkylresorcinol metabolite, a biomarker of whole-grain wheat and rye intake, and risk of type 2 diabetes and impaired glucose regulation in a Chinese population. Diabetes Care 2018, 41, 440–445. [Google Scholar] [CrossRef] [PubMed]
- Savolainen, O.; Lind, M.V.; Bergström, G.; Fagerberg, B.; Sandberg, A.S.; Ross, A. Biomarkers of food intake and nutrient status are associated with glucose tolerance status and development of type 2 diabetes in older Swedish women. Am. J. Clin. Nutr. 2017, 106, 1302–1310. [Google Scholar] [CrossRef] [PubMed]
- Biskup, I.; Kyrø, C.; Marklund, M.; Olsen, A.; van Dam, R.M.; Tjønneland, A.; Overvad, K.; Lindahl, B.; Johansson, I.; Landberg, R. Plasma alkylresorcinols, biomarkers of whole-grain wheat and rye intake, and risk of type 2 diabetes in Scandinavian men and women. Am. J. Clin. Nutr. 2016, 104, 88–96. [Google Scholar] [CrossRef] [PubMed]
- Micha, R.; Khatibzadeh, S.; Shi, P.; Andrews, K.G.; Engell, R.E.; Mozaffarian, D.; Global Burden of Diseases Nutrition and Chronic Diseases Expert Group (NutriCoDE). Global, regional and national consumption of major food groups in 1990 and 2010: A systematic analysis including 266 country-specific nutrition surveys worldwide. BMJ Open 2015, 5, e008705. [Google Scholar] [CrossRef] [PubMed]
- Johnsen, N.F.; Frederiksen, K.; Christensen, J.; Skeie, G.; Lund, E.; Landberg, R.; Johansson, I.; Nilsson, L.M.; Halkjær, J.; Olsen, A.; et al. Whole-grain products and whole-grain types are associated with lower all-cause and cause-specific mortality in the Scandinavian HELGA cohort. Br. J. Nutr. 2015, 114, 608–623. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, D.R., Jr.; Meyer, K.A.; Kushi, L.H.; Folsom, A.R. Is whole grain intake associated with reduced total and cause-specific death rates in older women? The Iowa Women’s Health Study. Am. J. Public Health 1999, 89, 322–329. [Google Scholar] [CrossRef] [PubMed]
- Muhihi, A.; Gimbi, D.; Njelekela, M.; Shemaghembe, E.; Mwambene, K.; Chiwanga, F.; Malik, V.S.; Wedick, N.M.; Spiegelman, D.; Hu, F.B.; et al. Consumption and acceptability of whole grain staples for lowering markers of diabetes risk among overweight and obese Tanzanian adults. Glob. Health 2013, 9, 26. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.; Pan, A.; Zong, G.; Yu, Z.; Wu, H.; Chen, X.; Tang, L.; Feng, Y.; Zhou, H.; Chen, X.; et al. Substituting white rice with brown rice for 16 weeks does not substantially affect metabolic risk factors in middle-aged Chinese men and women with diabetes or a high risk for diabetes. J. Nutr. 2011, 141, 1685–1690. [Google Scholar] [CrossRef] [PubMed]
- Meyer, K.A.; Kushi, L.H.; Jacobs, D.R., Jr.; Slavin, J.; Sellers, T.A.; Folsom, A.R. Carbohydrates, dietary fiber, and incident type 2 diabetes in older women. Am. J. Clin. Nutr. 2000, 71, 921–930. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Manson, J.E.; Stampfer, M.J.; Hu, F.B.; Giovannucci, E.; Colditz, G.A.; Hennekens, C.H.; Willett, W.C. A prospective study of whole-grain intake and risk of type 2 diabetes mellitus in US women. Am. J. Public Health 2000, 90, 1409–1415. [Google Scholar] [PubMed]
- Fung, T.T.; Hu, F.B.; Pereira, M.A.; Liu, S.; Stampfer, M.J.; Colditz, G.A.; Willett, W.C. Whole-grain intake and the risk of type 2 diabetes: A prospective study in men. Am. J. Clin. Nutr. 2002, 76, 535–540. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Willett, W.C.; Manson, J.E.; Hu, F.B.; Rosner, B.; Colditz, G. Relation between changes in intakes of dietary fiber and grain products and changes in weight and development of obesity among middle-aged women. Am. J. Clin. Nutr. 2003, 78, 920–927. [Google Scholar] [CrossRef] [PubMed]
- McKeown, N.M.; Yoshida, M.; Shea, M.K.; Jacques, P.F.; Lichtenstein, A.H.; Rogers, G.; Booth, S.L.; Saltzman, E. Whole-grain intake and cereal fiber are associated with lower abdominal adiposity in older adults. J. Nutr. 2009, 139, 1950–1955. [Google Scholar] [CrossRef] [PubMed]
- Koh-Banerjee, P.; Franz, M.; Sampson, L.; Liu, S.; Jacobs, D.R., Jr.; Spiegelman, D.; Willett, W.; Rimm, E. Changes in whole-grain, bran, and cereal fiber consumption in relation to 8-year weight gain among men. Am. J. Clin. Nutr. 2004, 80, 1237–1245. [Google Scholar] [CrossRef] [PubMed]
- Giacco, R.; Della Pepa, G.; Luongo, D.; Riccardi, G. Whole grain intake in relation to body weight: From epidemiological evidence to clinical trials. Nutr. Metab. Cardiovasc. Dis. 2011, 21, 901–908. [Google Scholar] [CrossRef] [PubMed]
- Pol, K.; Christensen, R.; Bartels, E.M.; Raben, A.; Tetens, I.; Kristensen, M. Whole grain and body weight changes in apparently healthy adults: A systematic review and meta-analysis of randomized controlled studies. Am. J. Clin. Nutr. 2013, 98, 872–884. [Google Scholar] [CrossRef] [PubMed]
- Kirwan, J.P.; Malin, S.K.; Scelsi, A.R.; Kullman, E.L.; Navaneethan, S.D.; Pagadala, M.R.; Haus, J.M.; Filion, J.; Godin, J.P.; Kochhar, S.; et al. A whole-grain diet reduces cardiovascular risk factors in overweight and obese adults: A randomized controlled trial. J. Nutr. 2016, 146, 2244–2251. [Google Scholar] [CrossRef] [PubMed]
- Suhr, J.; Vuholm, S.; Iversen, K.N.; Landberg, R.; Kristensen, M. Wholegrain rye, but not wholegrain wheat, lowers body weight and fat mass compared with refined wheat: A 6-week randomized study. Eur. J. Clin. Nutr. 2017, 71, 959–967. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Cai, X.; Ma, X.; Jing, L.; Gu, J.; Bao, L.; Jun, Li.; Xu, M.; Zhang, Z.; Li, Y. Short- and Long-Term Effects of wholegrain oat intake on weight management and glucolipid metabolism in overweight type-2 diabetics: A randomized control trial. Nutrients 2016, 8, 549. [Google Scholar] [CrossRef] [PubMed]
- Karl, J.P.; Meydani, M.; Barnett, J.B.; Vanegas, S.M.; Goldin, B.; Kane, A.; Rasmussen, H.; Saltzman, E.; Vangay, P.; Knights, D.; et al. Substituting whole grains for refined grains in a 6-week randomized trial favorably affects energy-balance metrics in healthy men and postmenopausal women. Am. J. Clin. Nutr. 2017, 105, 589–599. [Google Scholar] [CrossRef] [PubMed]
- Grundy, S.M.; Becker, D.; Clark, L.T.; Cooper, R.S.; Denke, M.A.; Howard, J.; Hunninghake, D.B.; Illingworth, D.R.; Luepker, R.V.; McBride, P.; et al. Detection, evaluation, and treatment of high blood cholesterol in adults (Adult Treatment Panel III) final report. Circulation 2002, 106, 3143–3421. [Google Scholar]
- Steffen, L.M.; Jacobs, D.R., Jr.; Murtaugh, M.A.; Moran, A.; Steinberger, J.; Hong, C.P.; Sinaiko, A.R. Whole grain intake is associated with lower body mass and greater insulin sensitivity among adolescents. Am. J. Epidemiol. 2003, 158, 243–250. [Google Scholar] [CrossRef] [PubMed]
- McKeown, N.M.; Meigs, J.B.; Liu, S.; Wilson, P.W.; Jacques, P.F. Whole-grain intake is favorably associated with metabolic risk factors for type 2 diabetes and cardiovascular disease in the Framingham offspring study. Am. J. Clin. Nutr. 2002, 76, 390–398. [Google Scholar] [CrossRef] [PubMed]
- Esmaillzadeh, A.; Mirmiran, P.; Azizi, F. Whole-grain consumption and the metabolic syndrome: A favorable association in Tehranian adults. Eur. J. Clin. Nutr. 2005, 59, 353–362. [Google Scholar] [CrossRef] [PubMed]
- Sahyoun, N.R.; Jacques, P.F.; Zhang, X.L.; Juan, W.; McKeown, N.M. Whole grain intake is inversely associated with the metabolic syndrome and mortality in older adults. Am. J. Clin. Nutr. 2006, 83, 124–131. [Google Scholar] [CrossRef] [PubMed]
- Liese, A.D.; Roach, A.K.; Sparks, K.C.; Marquart, L.; D’Agostino, R.B., Jr.; Mayer-Davis, E.J. Whole-grain intake and insulin sensitivity: The insulin resistance atherosclerosis study. Am. J. Clin. Nutr. 2003, 78, 965–971. [Google Scholar] [CrossRef] [PubMed]
- Damsgaard, C.T.; Biltoft-Jensen, A.; Tetens, I.; Michaelsen, K.F.; Lind, M.V.; Astrup, A.; Landberg, R. Whole-grain intake, reflected by dietary records and biomarkers, is inversely associated with circulating insulin and other cardiometabolic markers in 8- to 11-year-old children. J. Nutr. 2017, 147, 816–824. [Google Scholar] [CrossRef] [PubMed]
- Pereira, M.A.; Jacobs, D.R., Jr.; Pins, J.J.; Raatz, S.K.; Gross, M.D.; Slavin, J.L.; Seaquist, E.R. Effect of whole grains on insulin sensitivity in overweight hyperinsulinemic adults. Am. J. Clin. Nutr. 2002, 75, 848–855. [Google Scholar] [CrossRef] [PubMed]
- Juntunen, K.S.; Laaksonen, D.E.; Poutanen, K.S.; Niskanen, L.K.; Mykkänen, H.M. High fiber rye bread and insulin secretion and sensitivity in healthy postmenopausal women. Am. J. Clin. Nutr. 2003, 77, 385–391. [Google Scholar] [CrossRef] [PubMed]
- McIntosh, G.H.; Noakes, M.; Royle, P.J.; Foster, P.R. Whole grain rye and wheat foods and markers of bowel health in overweight middle aged men. Am. J. Clin. Nutr. 2003, 77, 967–974. [Google Scholar] [CrossRef] [PubMed]
- Andersson, A.; Tengblad, S.; Karlström, B.; Kamal Eldin, A.; Landberg, R.; Basu, S.; Aman, P.; Vessby, B. Whole grain foods do not affect insulin sensitivity or markers of lipid peroxidation and inflammation in healthy, moderately overweight subjects. J. Nutr. 2007, 137, 1401–1407. [Google Scholar] [CrossRef] [PubMed]
- Katcher, H.I.; Legro, R.S.; Kunselman, A.R.; Gillies, P.J.; Demers, L.M.; Bagshaw, D.M.; Kris-Etherton, P.M. The effects of a whole grain enriched hypocaloric diet on cardiovascular disease risk factors in men and women with metabolic syndrome. Am. J. Clin. Nutr. 2008, 87, 79–90. [Google Scholar] [CrossRef] [PubMed]
- Giacco, R.; Clemente, G.; Cipriano, D.; Luongo, D.; Viscovo, D.; Patti, L.; Di Marino, L.; Giacco, A.; Naviglio, D.; Bianchi, M.A.; et al. Effects of the regular consumption of wholemeal wheat foods on cardiovascular risk factors in healthy people. Nutr. Metab. Cardiovasc. Dis. 2010, 20, 186–194. [Google Scholar] [CrossRef] [PubMed]
- Brownlee, I.A.; Moore, C.; Chatfield, M.; Richardson, D.P.; Ashby, P.; Kuznesof, S.A.; Jebb, S.A.; Seal, C.J. Markers of cardiovascular risk are not changed by increased whole grain intake: The WHOLEheart study, a randomised, controlled dietary intervention. Br. J. Nutr. 2010, 104, 125–134. [Google Scholar] [CrossRef] [PubMed]
- Giacco, R.; Lappi, J.; Costabile, G.; Kolehmainen, M.; Schwab, U.; Landberg, R.; Uusitupa, M.; Poutanen, K.; Pacini, G.; Rivellese, A.A.; et al. Effects of rye and whole wheat versus refined cereal foods on metabolic risk factors: A randomised controlled two-centre intervention study. Clin. Nutr. 2013, 32, 941–949. [Google Scholar] [CrossRef] [PubMed]
- Malin, S.K.; Kullman, E.L.; Scelsi, A.R.; Haus, J.M.; Filion, J.; Pagadala, M.R.; Godin, J.P.; Kochhar, S.; Ross, A.B.; Kirwan, J.P. A whole-grain diet reduces peripheral insulin resistance and improves glucose kinetics in obese adults: A randomized-controlled trial. Metabolism 2018, 82, 111–117. [Google Scholar] [CrossRef] [PubMed]
- He, L.X.; Zhao, J.; Huang, Y.S.; Li, Y. The difference between oats and beta-glucan extract intake in the management of HbA1c, fasting glucose and insulin sensitivity: A meta-analysis of randomized controlled trials. Food Funct. 2016, 7, 1413–1428. [Google Scholar] [CrossRef] [PubMed]
- Marventano, S.; Vetrani, C.; Vitale, M.; Godos, J.; Riccardi, G.; Grosso, G. Whole grain intake and glycaemic control in healthy subjects: A systematic review and meta-analysis of randomized controlled trials. Nutrients 2017, 19, 769. [Google Scholar] [CrossRef] [PubMed]
- Giacco, R.; Costabile, G.; Della Pepa, G.; Anniballi, G.; Griffo, E.; Mangione, A.; Cipriano, P.; Viscovo, D.; Clemente, G.; Landberg, R.; et al. A whole-grain cereal-based diet lowers postprandial plasma insulin and triglyceride levels in individuals with metabolic syndrome. Nutr. Metab. Cardiovasc. Dis. 2014, 24, 837–844. [Google Scholar] [CrossRef] [PubMed]
- Magnusdottir, O.K.; Landberg, R.; Gunnarsdottir, I.; Cloetens, L.; Akesson, B.; Landin-Olsson, M.; Rosqvist, F.; Iggman, D.; Schwab, U.; Herzig, K.H.; et al. Plasma alkylresorcinols C17:0/C21:0 ratio, a biomarker of relative whole-grain rye intake, is associated to insulin sensitivity: A randomized study. Eur. J. Clin. Nutr. 2014, 68, 453–458. [Google Scholar] [CrossRef] [PubMed]
- Bao, L.; Cai, X.; Xu, M.; Li, Y. Effect of oat intake on glycaemic control and insulin sensitivity: A meta-analysis of randomised controlled trials. Br. J. Nutr. 2014, 112, 457–466. [Google Scholar] [CrossRef] [PubMed]
- Wirström, T.; Hilding, A.; Gu, H.F.; Östenson, C.G.; Björklund, A. Consumption of whole grain reduces risk of deteriorating glucose tolerance, including progression to prediabetes. Am. J. Clin. Nutr. 2013, 97, 179–187. [Google Scholar] [CrossRef] [PubMed]
- Lappi, J.; Salojärvi, J.; Kolehmainen, M.; Mykkänen, H.; Poutanen, K.; de Vos, W.M.; Salonen, A. Intake of whole-grain and fiber-rich rye bread versus refined wheat bread does not differentiate intestinal microbiota composition. J. Nutr. 2013, 143, 648–655. [Google Scholar] [CrossRef] [PubMed]
- European Food Safety Authority. Scientific opinion on the substantiation of health claims related to beta glucans and maintenance or achievement of normal blood glucose concentrations (ID 756, 802, 2935) pursuant to Article 13(1) of Regulation (EC) No 1924/2006. EFSA J. 2010, 8, 1482. [Google Scholar]
- Kdnudsen, K.E. Fiber and nonstarch polysaccharide content and variation in common crops used in broiler diets. Poult. Sci. 2014, 9, 2380–2393. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, D.; Kendall, C.W.; Augustin, L.S.; Martini, M.C.; Axelsen, M.; Faulkner, D.; Vidgen, E.; Parker, T.; Lau, H.; Connelly, P.W.; et al. Effect of wheat bran on glycemic control and risk factors for cardiovascular disease in type 2 diabetes. Diabetes Care 2002, 25, 1522–1528. [Google Scholar] [CrossRef] [PubMed]
- Silva, F.M.; Kramer, C.K.; de Almeida, J.C.; Steemburgo, T.; Gross, J.L.; Azevedo, M.J. Fiber intake and glycemic control in patients with type 2 diabetes mellitus: A systematic review with meta-analysis of randomized controlled trials. Nutr. Rev. 2013, 71, 790–801. [Google Scholar] [CrossRef] [PubMed]
- Hou, Q.; Li, Y.; Li, L.; Cheng, G.; Sun, X.; Li, S.; Tian, H. The metabolic effects of oats intake in patients with type 2 diabetes: A systematic review and meta-analysis. Nutrients 2015, 7, 10369–10387. [Google Scholar] [CrossRef] [PubMed]
- Shen, X.L.; Zhao, T.; Zhou, Y.; Shi, X.; Zou, Y.; Zhao, G. Effect of oat β-glucan intake on glycaemic control and insulin sensitivity of diabetic patients: A meta-analysis of randomized controlled trials. Nutrients 2016, 8, 39. [Google Scholar] [CrossRef] [PubMed]
- Fardet, A. New hypotheses for the health-protective mechanisms of whole-grain cereals: What is beyond fibre? Nutr. Res. Rev. 2010, 23, 65–134. [Google Scholar] [CrossRef] [PubMed]
- Karimi, G.; Azadbakht, L.; Haghighatdoost, F.; Esmaillzadeh, A. Low energy density diet, weight loss maintenance, and risk of cardiovascular disease following a recent weight reduction program: A randomized control trial. J. Res. Med. Sci. 2016, 21, 32. [Google Scholar] [PubMed]
- Wanders, A.J.; Van de Borne, J.J.; de Graaf, C.; Hulshof, T.; Jonathan, M.C.; Kristensen, M.; Mars, M.; Schols, H.A.; Feskens, E.J. Effects of dietary fibre on subjective appetite, energy intake and body weight: A systematic review of randomized controlled trials. Obes. Rev. 2011, 12, 724–739. [Google Scholar] [CrossRef] [PubMed]
- Westerterp-Plantenga, M.S. Effects of energy density of daily food intake on long-term energy intake. Physiol. Behav. 2004, 81, 765–771. [Google Scholar] [CrossRef] [PubMed]
- McRorie, J.W. Evidence-based approach to fiber supplements and clinically meaningful health benefits, What to look for and how to recommend an effective fiber therapy. Nutr. Today 2015, 50, 82–89. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, D.; Miguel, M.; Aleixandre, A. Dietary fiber, gut peptides, and adipocytokines. J. Med. Food 2012, 15, 223–230. [Google Scholar] [CrossRef] [PubMed]
- Bodnaruc, A.M.; Prud’homme, D.; Blanchet, R.; Giroux, I. Nutritional modulation of endogenous glucagon-like peptide-1 secretion: A review. Nutr. Metab. 2016, 13, 92. [Google Scholar] [CrossRef] [PubMed]
- Costabile, G.; Griffo, E.; Cipriano, P.; Vetrani, C.; Vitale, M.; Mamone, G.; Rivellese, A.A.; Riccardi, G.; Giacco, R. Subjective satiety and plasma PYY concentration after wholemeal pasta. Appetite 2018, 125, 172–181. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, A.C.; Johansson-Boll, E.V.; Björck, I.M. Increased gut hormones and insulin sensitivity index following a 3-d intervention with a barley kernel-based product: A randomised cross-over study in healthy middle-aged subjects. Br. J. Nutr. 2015, 114, 899–907. [Google Scholar] [CrossRef] [PubMed]
- Bozzetto, L.; Costabile, G.; Della Pepa, G.; Ciciola, P.; Vetrani, C.; Vitale, M.; Rivellese, A.A.; Annuzzi, G. Dietary fibre as a unifying remedy for the whole spectrum of obesity-associated cardiovascular risk. Nutrients 2018, 10, 943. [Google Scholar] [CrossRef] [PubMed]
- Hartvigsen, M.L.; Gregersen, S.; Lærke, H.N.; Holst, J.J.; Bach Knudsen, K.E.; Hermansen, K. Effects of concentrated arabinoxylan and β-glucan compared with refined wheat and whole grain rye on glucose and appetite in subjects with the metabolic syndrome: A randomized study. Eur. J. Clin. Nutr. 2014, 1, 84–90. [Google Scholar] [CrossRef] [PubMed]
- Hartvigsen, M.L.; Lærke, H.N.; Overgaard, A.; Holst, J.J.; Bach Knudsen, K.E.; Hermansen, K. Postprandial effects of test meals including concentrated arabinoxylan and whole grain rye in subjects with the metabolic syndrome: A randomised study. Eur. J. Clin. Nutr. 2014, 5, 567–574. [Google Scholar] [CrossRef] [PubMed]
- Canfora, E.E.; Jocken, J.W.; Blaak, E.E. Short-chain fatty acids in control of body weight and insulin sensitivity. Nat. Rev. Endocrinol. 2015, 11, 577–591. [Google Scholar] [CrossRef] [PubMed]
- Vetrani, C.; Costabile, G.; Luongo, D.; Naviglio, D.; Rivellese, A.A.; Riccardi, G.; Giacco, R. Effects of whole-grain cereal foods on plasma short chain fatty acid concentrations in individuals with the metabolic syndrome. Nutrition 2016, 32, 217–221. [Google Scholar] [CrossRef] [PubMed]
- Vitaglione, P.; Mennella, I.; Ferracane, R.; Rivellese, A.A.; Giacco, R.; Ercolini, D.; Gibbons, S.M.; La Storia, A.; Gilbert, J.A.; Jonnalagadda, S.; et al. Whole-grain wheat consumption reduces inflammation in a randomized controlled trial on overweight and obese subjects with unhealthy dietary and lifestyle behaviors: Role of polyphenols bound to cereal dietary fiber. Am. J. Clin. Nutr. 2015, 101, 251–261. [Google Scholar] [CrossRef] [PubMed]
- De Angelis, M.; Montemurno, E.; Vannini, L.; Cosola, C.; Cavallo, N.; Gozzi, G.; Maranzano, V.; Di Cagno, R.; Gobbetti, M.; Gesualdo, L. Effect of whole-grain barley on the human fecal microbiota and metabolome. Appl. Environ. Microbiol. 2015, 81, 7945–7956. [Google Scholar] [CrossRef] [PubMed]
- Adom, K.K.; Sorrells, M.E.; Liu, R.H. Phytochemicals and antioxidant activity of milled fractions of different wheat varieties. J. Agric. Food Chem. 2005, 53, 2297–2306. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Sharma, S. Bioactive components and functional properties of biologically activated cereal grains: A bibliographic review. Crit. Rev. Food Sci. Nutr. 2017, 14, 3051–3071. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Zhao, X.; Ran, L.; Wan, J.; Wang, X.; Qin, J.; Shu, F.; Gao, Y.; Yuan, L.; Zhang, Q.; et al. Resveratrol improves insulin resistance, glucose and lipid metabolism in patients with non-alcoholic fatty liver disease: A randomized controlled trial. Dig. Liver Dis. 2015, 47, 226–232. [Google Scholar] [CrossRef] [PubMed]
- Bozzetto, L.; Annuzzi, G.; Pacini, G.; Costabile, G.; Vetrani, V.; Vitale, M.; Griffo, G.; Giacco, A.; De Natale, C.; Cocozza, S.; et al. Polyphenol-rich diets improve glucose metabolism in people at high cardiometabolic risk: A controlled randomized intervention trial. Diabetologia 2015, 58, 1551–1560. [Google Scholar] [CrossRef] [PubMed]
- Via, M. The malnutrition of obesity: Micronutrient deficiencies that promote diabetes. ISRN Endocrinol. 2012, 2012, 103472. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Bailo, B.; El-Sohemy, A.; Haddad, P.S.; Arora, P.; BenZaied, F.; Karmali, M.; Badawi, A. Vitamins D, C, and E in the prevention of type 2 diabetes mellitus: Modulation of inflammation and oxidative stress. Biologics 2011, 5, 7–19. [Google Scholar] [PubMed]
- Costabile, G.; Della Pepa, G.; Bozzetto, L.; Annuzzi, G.; Vetrani, C.; Giacco, R.; Della Corte, G.; Conte, F.S.; Di Marino, L.; Rivellese, A.A. Urine 8-isoprostane in relation to adiposity and insulin resistance in individuals at high cardiometabolic risk. Metab. Syndr. Relat. Disord. 2015, 13, 187–191. [Google Scholar] [CrossRef] [PubMed]
- Bozzetto, L.; Annuzzi, G.; Ragucci, M.; Di Donato, O.; Della Pepa, G.; Della Corte, G.; Griffo, E.; Anniballi, G.; Giacco, A.; Mancini, M.; et al. Insulin resistance, postprandial GLP-1 and adaptive immunity are the main predictors of NAFLD in a homogeneous population at high cardiovascular risk. Nutr. Metab. Cardiovasc. Dis. 2016, 26, 623–629. [Google Scholar] [CrossRef] [PubMed]
- Barbagallo, M.; Dominguez, L.J. Magnesium and type 2 diabetes. World J. Diabetes 2015, 6, 1152–1157. [Google Scholar] [CrossRef] [PubMed]
- Heer, M.; Egert, S. Nutrients other than carbohydrates: Their effects on glucose homeostasis in humans. Diabetes Metab. Res. Rev. 2015, 31, 14–35. [Google Scholar] [CrossRef] [PubMed]
- O’Neil, C.E.; Nicklas, T.A.; Zanovec, M.; Cho, S. Whole grain consumption is associated with diet quality and nutrient intake in adults: The national health and nutrition examination survey, 1999–2004. J. Am. Diet. Assoc. 2010, 110, 1461–1468. [Google Scholar] [CrossRef] [PubMed]
- Authors/Task Force Members; Rydén, L.; Grant, P.J.; Anker, S.D.; Berne, C.; Cosentino, F.; Danchin, N.; Deaton, C.; Escaned, J.; Hammes, H.P.; et al. ESC guidelines on diabetes, pre-diabetes, and cardiovascular diseases developed in collaboration with the EASD: The task force on diabetes, pre-diabetes, and cardiovascular diseases of the European Society of Cardiology (ESC) and developed in collaboration with the European Association for the Study of Diabetes (EASD). Eur. Heart. J. 2013, 39, 3035–3087. [Google Scholar]
| Nutrient | Reference | Wheat | Brown Rice | Maize | Oat | Rye | Barley |
|---|---|---|---|---|---|---|---|
| Macronutrient (g/100 g) | [13] | ||||||
| Carbohydrate | 71.2 | 76.2 | 74.3 | 66.3 | 75.9 | 73.4 | |
| Lipid | 1.5 | 3.2 | 4.7 | 6.9 | 1.6 | 2.3 | |
| Protein | 12.6 | 7.5 | 9.4 | 16.9 | 10.3 | 12.5 | |
| Fiber | 12.2 | 3.6 | 7.3 | 10.6 | 15.1 | 17.3 | |
| Minerals (mg/100 g) | [13] | ||||||
| Calcium | 29 | 33 | 7 | 54 | 24 | 33 | |
| Iron | 3.19 | 1.80 | 2.71 | 4.72 | 2.63 | 3.60 | |
| Magnesium | 126 | 143 | 127 | 177 | 110 | 133 | |
| Phosphorus | 288 | 264 | 210 | 523 | 332 | 264 | |
| Potassium | 363 | 268 | 287 | 429 | 510 | 452 | |
| Sodium | 2 | 4 | 35 | 2 | 2 | 12 | |
| Zinc | 2.65 | 2.02 | 2.21 | 3.97 | 2.65 | 2.77 | |
| Vitamins (mg/100 g) | [13] | ||||||
| Thiamin | 0.383 | 0.413 | 0.385 | 0.763 | 0.316 | 0.646 | |
| Riboflavin | 0.115 | 0.043 | 0.201 | 0.139 | 0.251 | 0.285 | |
| Niacin | 5.464 | 4.308 | 3.627 | 0.961 | 4.270 | 4.604 | |
| Vitamin B6 | 0.300 | 0.509 | 0.622 | 0.119 | 0.294 | 0.318 | |
| Folate | 0.038 | 0.020 | 0.019 | 0.056 | 0.038 | 0.019 | |
| Vitamin E | 1.010 | n.a. | 0.490 | n.a. | 0.850 | 0.570 | |
| Vitamin K | 0.019 | n.a. | 0.003 | n.a. | 0.060 | 0.022 | |
| Phytochemicals § (mg/100 g) | [14,15,16] | ||||||
| Phenolic acids | 1.342 | 0.286 | 0.601 | 0.472 | 1.364 | 0.898 | |
| Ferulic acid | 114 | 30 | 174 | 2 | 4 | 115 | |
| Flavonoids | 36 | n.a. | n.a. | 7 | 7 | 15 | |
| Betaine | 156 | 0.5 | n.a. | 55 | n.a. | 58 | |
| Carotenoids | 0.33 | 0.04 | 1.45 | 0.03 | n.a. | 0.06 | |
| Alkylresorcinol | 0.47 | n.a. | n.a. | n.a. | 1.89 | 0.75 | |
| Phytosterols | 77.5 | n.a. | 14.8 | n.a. | n.a. | 102 |
| Author (Reference) | Study Design | Study Population Participants Age BMI Health Status | Intervention and Doses | Duration Weeks | Observed Effects in Wholegrain Group |
|---|---|---|---|---|---|
| Pol et al., 2013 [38] | Meta-analyses | 2060 M/F 18–70 years 18–35 kg/m2 - | wholegrain (mostly oat, wheat, barley, rye, rice: 18–136 g/day) vs. refined grain | 2–16 | = BW = WC ↓ Body fat |
| Kirwan et al., 2016 [39] | RCT, crossover | 40 M/F 40 years 33 kg/m2 Healthy | wholegrain (wheat, rice, oat: 93 g/day) vs. refined grain | 6 | = BW =WC |
| Suhr et al., 2017 [40] | RCT, parallel | 75 M/F 30–65 years 28 kg/m2 Healthy | ad libitum wholegrain rye-based foods (124 ± 12 g/day) vs. ad libitum wholegrain wheat-based foods (145 ± 12 g/day) vs. ad libitum refined wheat-based foods | 6 | Whole rye: ↓ BW = WC Whole wheat: = BW = WC |
| Li et al., 2016 [41] | RCT, parallel | 287 M/F 59 years 27 kg/m2 T2DM | wholegrain oat-based foods (100 g/day) vs. wholegrain oat-based foods (50 g/day) vs. usual dietary habits | 48 | ↓ BW |
| Author (Reference) | Study Design | Study Population Participants Age BMI Health Status | Intervention and Doses | Duration Weeks | Observed Effects in Wholegrain Group |
|---|---|---|---|---|---|
| Pereira et al., 2002 [50] | RCT cross-over | 11 M/F 41.6 years 30.2 kg/m2 Hyperinsulinemic | wholegrain foods (mostly wheat, rice, rye, corn, oat, burley: 386 g/day) vs. refined grain | 6 | ↑ IS (euglycemic hyperinsulinemic clamp tests) |
| Juntunen et al., 2003 [51] | RCT cross-over | 20 F 59 years 28 kg/m2 Healthy | high fiber rye bread (208 g/day) vs. white wheat bread | 8 | = IS (FSIGT) |
| McIntosh et al., 2003 [52] | RCT cross-over | 28 M 40–65 years 30 kg/m2 Healthy | wholegrain rye-based foods (230 g/day) vs. wholegrain wheat-based foods (230 g/day) vs. low fiber diet | 4 | = IS (HOMA) |
| Andersson et al., 2007 [53] | RCT cross-over | 30 M/F 59 years 28.3 kg/m2 One criteria of MS | wholegrain foods (mostly wheat, oat, rye, rice: 112 g/day) vs. refined grain | 6 | = IS (euglycemic hyperinsulinemic clamp tests) |
| Katcher et al., 2008 [54] | RCT parallel | 47 M/F 46 years 36 kg/m2 MS | wholegrain foods (mostly wheat, oat, rye, rice: 218 g/day) vs. refined grain | 12 | = IS (ISI during OGTT) |
| Giacco et al., 2010 [55] | RCT crossover | 15 M/F 55 years 27 kg/m2 Healthy | wholegrain wheat-based foods (283 g/day) vs. refined grain | 3 | = IS (HOMA) |
| Brownlee et al., 2010 [56] | RCT parallel | 216 M/F 46 years 30 kg/m2 Healthy | Wholegrain foods (wheat, oat, rice: 120 g/day) vs. wholegrain foods (wheat, oat, rice: 60 g/day) vs. refined grain | 16 | = IS (QUICKI) |
| Giacco et al., 2013 [57] | RCT parallel | 133 M/F 40–65 years 31.4 kg/m2 MS | wholegrain foods (rye, wheat: 232 g/day) vs. refined grain | 12 | = IS (FSIGT) |
| Malin et al., 2018 [58] | RCT crossover | 14 M/F 38 years 34 kg/m2 Healthy | wholegrain foods (wheat, oat, rice: 90 g/day) vs. refined grain | 8 | ↑ IS (OGTT with isotopic tracer) |
| He et al., 2016 [59] | Meta-analyses | 298 M/F 53 years 26 kg/m2 Overweight/T2DM | wholegrain oat-based foods (20–136 g/day) vs. refined grain foods | 8 | ↑ IS (HOMA) |
| Author (Reference) | Study Design | Study Population Participants Age BMI Health Status | Intervention and Doses | Duration Weeks | Observed Effects in Wholegrain Group |
|---|---|---|---|---|---|
| Fasting condition | |||||
| Marventano et al., 2017 [60] | Meta-analyses | 377 M/F 50 years 28 kg/m2 Healthy | Wholegrain foods (mostly wheat, rye, rice, barley, maize and oat) vs. refined grain | 2–16 | = glucose |
| Postprandial condition | |||||
| Marventano et al., 2017 [60] | Meta-analyses | 377 M/F 50 years 28 kg/m2 Healthy | wholegrain foods (mostly rye, oat and barley) vs. refined grain foods | Acute meal studies | ↓ glucose AUC |
| Lappi et al., 2013 [65] | RCT, Crossover | 21 M/F 38–65 years 19–30 kg/m2 Healthy | wholegrain rye bread (180–300 g/day) vs. refined wheat bread | 4 | = glucose AUC |
| Giacco et al., 2014 [61] | RCT, Parallel | 54 M/F 56 years 31.7 kg/m2 MS | wholegrain foods (wheat, oat, rye, barley: 268 g/day) vs. refined grain | 12 | = glucose AUC |
| Author (Reference) | Study Design | Study Population Participants Age BMI Health Status | Intervention and Doses | Duration Weeks | Observed Effects in Wholegrain Group |
|---|---|---|---|---|---|
| Fasting condition | |||||
| Hou et al., 2015 [70] | Meta-analyses | 306 M/F 60 years - T2DM | wholegrain oat-based foods (50–100 g/day) vs. refined grain foods | 1–4 | ↓ glucose = insulin ↓ HbA1c = Insulin resistance (HOMA) |
| Shen et al., 2016 [71] | Meta-analyses | 350 M/F 61 years 28 kg/m2 T2DM | wholegrain oat-based foods (2.5–5 g/day) vs. refined grain foods | 3–8 | ↓ glucose = insulin ↓ HbA1c |
| Li et al., 2016 [41] | RCT, parallel | 287 M/F 59 years 27 kg/m2 T2DM | wholegrain oat-based foods (100 g/day) vs. wholegrain oat-based foods (50 g/day) vs. usual dietary habits | 48 | ↓ glucose ↓ insulin ↓ HbA1c |
| Postprandial condition | |||||
| Hou et al., 2015 [70] | Meta-analyses | 306 M/F 60 years - T2DM | wholegrain oat-based foods (50–100 g/day) vs. refined grain foods | 1–4 | ↓ AUC glucose |
| Li et al., 2016 [41] | RCT, parallel | 287 M/F 59 years 27 kg/m2 T2DM | wholegrain oat-based foods (100 g/day) vs. wholegrain oat-based foods (50 g/day) vs. usual dietary habits | 48 | ↓ glucose AUC |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Della Pepa, G.; Vetrani, C.; Vitale, M.; Riccardi, G. Wholegrain Intake and Risk of Type 2 Diabetes: Evidence from Epidemiological and Intervention Studies. Nutrients 2018, 10, 1288. https://doi.org/10.3390/nu10091288
Della Pepa G, Vetrani C, Vitale M, Riccardi G. Wholegrain Intake and Risk of Type 2 Diabetes: Evidence from Epidemiological and Intervention Studies. Nutrients. 2018; 10(9):1288. https://doi.org/10.3390/nu10091288
Chicago/Turabian StyleDella Pepa, Giuseppe, Claudia Vetrani, Marilena Vitale, and Gabriele Riccardi. 2018. "Wholegrain Intake and Risk of Type 2 Diabetes: Evidence from Epidemiological and Intervention Studies" Nutrients 10, no. 9: 1288. https://doi.org/10.3390/nu10091288
APA StyleDella Pepa, G., Vetrani, C., Vitale, M., & Riccardi, G. (2018). Wholegrain Intake and Risk of Type 2 Diabetes: Evidence from Epidemiological and Intervention Studies. Nutrients, 10(9), 1288. https://doi.org/10.3390/nu10091288






