Favorable Effects of a Ketogenic Diet on Physical Function, Perceived Energy, and Food Cravings in Women with Ovarian or Endometrial Cancer: A Randomized, Controlled Trial
Abstract
1. Introduction
2. Methods
2.1. Participants
2.2. Protocol
2.3. Diet Interventions
2.4. Statistical Methods
3. Results
3.1. Patient Demographics
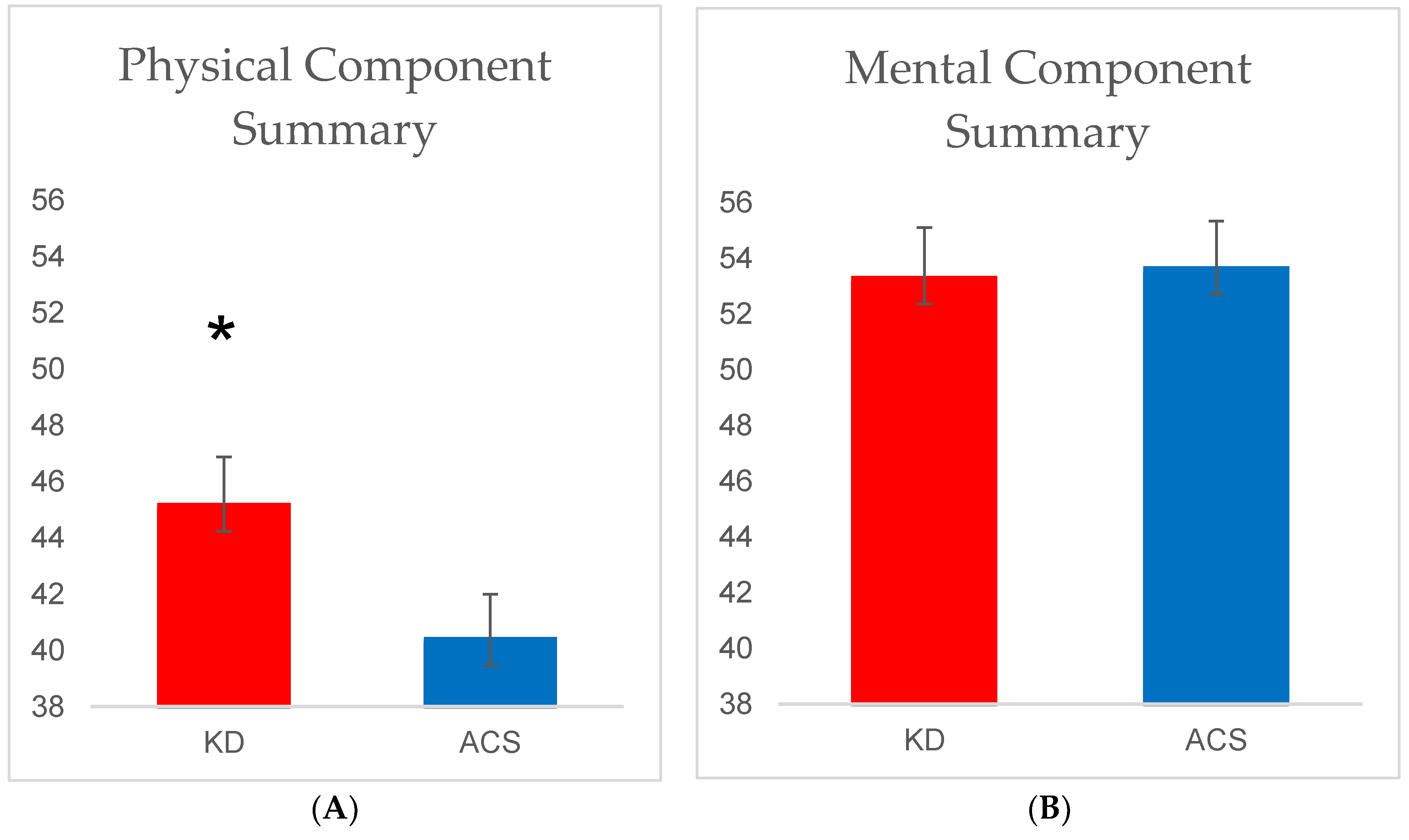
3.2. SF-12
3.3. VAS for Appetite
3.4. FCI
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| KD | ketogenic diet |
| ACS | American Cancer Society diet |
| CRF | cancer-related fatigue |
| SF-12 | Medical Outcomes Study Short Form-12 Health Survey |
| VAS | Visual Analog Scale for appetite |
| FCI | Food Craving Inventory |
| BHB | β-hydroxybutyrate |
References
- Zhou, W.; Mukherjee, P.; Kiebish, M.A.; Markis, W.T.; Mantis, J.G.; Seyfried, T.N. The calorically restricted ketogenic diet, an effective alternative therapy for malignant brain cancer. Nutr. Metab. 2007, 4, 5. [Google Scholar] [CrossRef] [PubMed]
- Zuccoli, G.; Marcello, N.; Pisanello, A.; Servadei, F.; Vaccaro, S.; Mukherjee, P.; Seyfried, T.N. Metabolic management of glioblastoma multiforme using standard therapy together with a restricted ketogenic diet: Case Report. Nutr. Metab. 2010, 7, 33. [Google Scholar] [CrossRef] [PubMed]
- Sremanakova, J.; Sowerbutts, A.M.; Burden, S. A systematic review of the use of ketogenic diets in adult patients with cancer. J. Hum. Nutr. Diet. 2018. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, D.P.; Kupelnick, B.; Miller, K.; Devine, D.; Lau, J. Evidence report on the occurrence, assessment, and treatment of fatigue in cancer patients. J. Natl. Cancer Inst. Monogr. 2004, 32, 40–50. [Google Scholar] [CrossRef] [PubMed]
- Servaes, P.; Verhagen, C.; Bleijenberg, G. Fatigue in cancer patients during and after treatment: Prevalence, correlates and interventions. Eur. J. Cancer 2002, 38, 27–43. [Google Scholar] [CrossRef]
- Cella, D.; Davis, K.; Breitbart, W.; Curt, G.; Fatigue, C. Cancer-related fatigue: Prevalence of proposed diagnostic criteria in a United States sample of cancer survivors. J. Clin. Oncol. 2001, 19, 3385–3391. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.S.; Zhao, F.; Fisch, M.J.; O’Mara, A.M.; Cella, D.; Mendoza, T.R.; Cleeland, C.S. Prevalence and characteristics of moderate to severe fatigue: A multicenter study in cancer patients and survivors. Cancer 2014, 120, 425–432. [Google Scholar] [CrossRef] [PubMed]
- Williams, L.A.; Agarwal, S.; Bodurka, D.C.; Saleeba, A.K.; Sun, C.C.; Cleeland, C.S. Capturing the patient’s experience: Using qualitative methods to develop a measure of patient-reported symptom burden: An example from ovarian cancer. J. Pain Symptom Manag. 2013, 46, 837–845. [Google Scholar] [CrossRef] [PubMed]
- Bower, J.E. Cancer-related fatigue—Mechanisms, risk factors, and treatments. Nat. Rev. Clin. Oncol. 2014, 11, 597–609. [Google Scholar] [CrossRef] [PubMed]
- Ebede, C.C.; Jang, Y.; Escalante, C.P. Cancer-related fatigue in cancer survivorship. Med. Clin. N. Am. 2017, 101, 1085–1097. [Google Scholar] [CrossRef] [PubMed]
- Tan-Shalaby, J.L.; Carrick, J.; Edinger, K.; Genovese, D.; Liman, A.D.; Passero, A.V.; Shah, R.B. Modified Atkins diet in advanced malignancies-final results of a safety and feasibility trial within the Veterans Affairs Pittsburgh Healthcare System. Nutr. Metab. 2016, 13, 52. [Google Scholar] [CrossRef] [PubMed]
- Toth, C.; Clemens, Z. Halted progression of soft palate cancer in a patient treated with the Paleolithic ketogenic diet alone: A 20-months follow-up. Am. J. Med. Case Rep. 2016, 4, 288–292. [Google Scholar]
- Klement, R.J. Beneficial effects of ketogenic diets for cancer patients: A realist review with focus on evidence and confirmation. Med. Oncol. 2017, 34, 132. [Google Scholar] [CrossRef] [PubMed]
- Breymeyer, K.L.; Lampe, J.W.; McGregor, B.A.; Neuhouser, M.L. Subjective mood and energy levels of healthy weight and overweight/obese healthy adults on high-and low-glycemic load experimental diets. Appetite 2016, 107, 253–259. [Google Scholar] [CrossRef] [PubMed]
- McClernon, F.J.; Yancy, Jr.W.S.; Eberstein, J.A.; Atkins, R.C.; Westman, E.C. The effects of a low-carbohydrate ketogenic diet and a low-fat diet on mood, hunger, and other self-reported symptoms. Obesity 2007, 15, 182–187. [Google Scholar] [CrossRef] [PubMed]
- Gibson, A.A.; Seimon, R.V.; Lee, C.M.; Ayre, J.; Franklin, J.; Markovic, T.P.; Caterson, I.D.; Sainsbury, A. Do ketogenic diets really suppress appetite? A systematic review and meta-analysis. Obes. Rev. 2015, 16, 64–76. [Google Scholar] [PubMed]
- Martin, C.K.; Rosenbaum, D.; Han, H.; Geiselman, P.J.; Wyatt, H.R.; Hill, J.O.; Brill, C.; Bailer, B.; Miller, B.V., III.; Stein, R.; et al. Change in food cravings, food preferences, and appetite during a low-carbohydrate and low-fat diet. Obesity 2011, 19, 1963–1970. [Google Scholar] [CrossRef] [PubMed]
- Cohen, C.W.; Fontaine, K.R.; Arend, R.C.; Alvarez, R.D.; Leath, C.A., III; Huh, W.K.; Bevis, K.S.; Kim, K.H.; Straughn, J.M., Jr.; Gower, B.A. A ketogenic diet reduces central obesity and serum insulin in women with ovarian or endometrial cancer. J. Nutr. 2018, 148, 1253–1260. [Google Scholar] [CrossRef] [PubMed]
- Ware, J., Jr.; Kosinski, M.; Keller, S.D. A 12-Item Short-Form Health Survey: Construction of scales and preliminary tests of reliability and validity. Med. Care 1996, 34, 220–233. [Google Scholar] [CrossRef] [PubMed]
- Ware, J.E.; Kosinski, M.; Keller, S.D. How to Score the SF-12 Physical and Mental Health Survey Summary Scales, 2nd ed.; The Health Institute, New England Medical Center Press: Boston, MA, USA, 1995. [Google Scholar]
- Gandek, B.; Ware, J.E.; Aaronson, N.K.; Apolone, G.; Bjorner, J.B.; Brazier, J.E.; Bullinger, M.; Kaasa, S.; Leplege, A.; Prieto, L.; et al. Cross-validation of item selection and scoring for the SF-12 Health Survey in nine countries: Results from the IQOLA Project. J. Clin. Epidemiol. 1998, 51, 1171–1178. [Google Scholar] [CrossRef]
- Bhandari, N.R.; Kathe, N.; Hayes, C.; Payakachat, N. Reliability and validity of SF-12v2 among adults with self-reported cancer. Res. Soc. Adm. Pharm. 2018. [Google Scholar] [CrossRef] [PubMed]
- Flint, A.; Raben, A.; Blundell, J.E.; Astrup, A. Reproducibility, power and validity of visual analogue scales in assessment of appetite sensations in single test meal studies. Int. J. Obes. Relat. Metab. Disord. 2000, 24, 38–48. [Google Scholar] [CrossRef] [PubMed]
- Stock, S.; Leichner, P.; Wong, A.C.; Ghatei, M.A.; Kieffer, T.J.; Bloom, S.R.; Chanoine, J.P. Ghrelin, peptide YY, glucose-dependent insulinotropic polypeptide, and hunger responses to a mixed meal in anorexic, obese, and control female adolescents. J. Clin. Endocrinol. Metab. 2005, 90, 2161–2168. [Google Scholar] [CrossRef] [PubMed]
- White, M.A.; Whisenhunt, B.L.; Williamson, D.A.; Greenway, F.L.; Netemeyer, R.G. Development and validation of the food-craving inventory. Obes. Res. 2002, 10, 107–114. [Google Scholar] [CrossRef] [PubMed]
- Rock, C.L.; Doyle, C.; Demark-Wahnefried, W.; Meyerhardt, J.; Courneya, K.S.; Schwartz, A.L.; Bandera, E.V.; Hamilton, K.K.; Grant, B.; McCullough, M.; et al. Nutrition and physical activity guidelines for cancer survivors. Cancer J. Clin. 2012, 62, 275–276. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, K.K. Nutritional Needs of the Adult Oncology Patient, in Oncology Nutrition for Clinical Practice; Leser, M., Ledesma, N., Eds.; Academy of Nutrition and Dietetics: Chicago, IL, USA, 2013; pp. 33–39. [Google Scholar]
- Brown, J.C.; Harhay, M.O.; Harhay, M.N. Patient-reported versus objectively-measured physical function and mortality risk among cancer survivors. J. Geriatr. Oncol. 2016, 7, 108–115. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.C.; Harhay, M.O.; Harhay, M.N. Physical function as a prognostic biomarker among cancer survivors. Br. J. Cancer 2015, 112, 112–194. [Google Scholar] [CrossRef] [PubMed]
- Cheung, A.S.; Rooy, C.D.; Hoermann, R.; Joon, D.L.; Zajac, J.D.; Grossmann, M. Quality of life decrements in men with prostate cancer undergoing androgen deprivation therapy. Clin. Endocrinol. 2017, 86, 388–394. [Google Scholar] [CrossRef] [PubMed]
- Patel, A.R.; Lester, R.T.; Marra, C.A.; van der Kop, M.L.; Ritvo, P.; Engel, L.; Karanja, S.; Lynd, L.D. The validity of the SF-12 and SF-6D instruments in people living with HIV/AIDS in Kenya. Health Qual. Life Outcomes 2017, 15, 143. [Google Scholar] [CrossRef] [PubMed]
- Bennett, S.J.; Oldridge, N.B.; Eckert, G.J.; Embree, J.L.; Browning, S.; Hou, N.; Chui, M.; Deer, M. Murray MD Comparison of quality of life measures in heart failure. Nurs. Res. 2003, 52, 207–216. [Google Scholar] [CrossRef] [PubMed]
- Kunitake, H.; Russell, M.M.; Zheng, P.; Yothers, G.; Land, S.R.; Petersen, L.; Fehrenbacher, L.; Giguere, J.K.; Wickerham, D.L.; Ko, C.Y.; et al. Quality of life and symptoms in long-term survivors of colorectal cancer: Results from NSABP protocol LTS-01. J. Cancer Surv. 2017, 11, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Schulman-Green, D.; Ercolano, E.; Dowd, M.; Schwartz, P.; McCorkle, R. Quality of life among women after surgery for ovarian cancer. Palliat. Support Care 2008, 6, 239–247. [Google Scholar] [CrossRef] [PubMed]
- Harvey, C.; Schofield, G.; Williden, M.; The lived experience of healthy adults following a ketogenic diet: A qualitative study. J. Holist. Perform. 2018. Available online: http://www.holisticperformance.org/article/3638-the-lived-experience-of-healthy-adults-following-a-ketogenic-diet-a-qualitative-study (accessed on 10 May 2018).
- Sarafidis, P.A.; Bakris, G.L. The antinatriuretic effect of insulin: An unappreciated mechanism for hypertension associated with insulin resistance? Am. J. Nephrol. 2007, 27, 44–54. [Google Scholar] [CrossRef] [PubMed]
- Rabast, U.; Vornberger, K.H.; Ehl, M. Loss of weight, sodium and water in obese persons consuming a high- or low-carbohydrate diet. Ann. Nutr. Metab. 1981, 25, 341–349. [Google Scholar] [CrossRef] [PubMed]
- Kolanowski, J. On the mechanisms of fasting natriuresis and of carbohydrate-induced sodium retention. Diabete Metab. 1977, 3, 131–143. [Google Scholar] [PubMed]
| KD | ACS |
|---|---|
|
|
| ACS (n = 20) | KD (n = 23 a) | ||||||
|---|---|---|---|---|---|---|---|
| WEEK 0 | WEEK 12 | Δ | WEEK 0 | WEEK 12 | Δ | p | |
| Hunger (mm) | 41.6 ± 26.9 | 46.1 ± 24.5 | 4.5 ± 30.8 | 37.0 ± 25.2 | 39.6 ± 31.8 | 2.6 ± 28.6 | 0.58 |
| Satisfaction (mm) | 23.4 ± 17.9 | 26.1 ± 23.4 | 2.8 ± 22.0 | 30.5 ± 27.7 | 35.0 ± 31.4 | 4.5 ± 26.8 | 0.61 |
| Fullness (mm) | 16.1 ± 22.0 | 21.8 ± 22.1 | 5.6 ± 22.1 | 26.8 ± 27.7 | 28.3 ± 32.3 | 1.4 ± 36.1 | 0.79 |
| Prospective Food Consumption (mm) | 64.1 ± 23.5 | 54.5 ± 19.0 | −9.6 ± 24.7 | 54.2 ± 27.0 | 44.8 ± 23.8 | −9.5 ± 31.4 | 0.27 |
| Desire for something sweet b (mm) | 60.3 ± 34.1 | 66.3 ± 28.9 | 6.0 ± 32.9 | 65.8 ± 31.2 | 71.6 ± 27.7 | 5.9 ± 40.0 | 0.64 |
| Desire for something salty b (mm) | 61.6 ± 26.6 | 65.5 ± 23.3 | 3.9 ± 28.1 | 60.5 ± 23.4 | 49.8 ± 23.7 | −10.8 ± 24.6 * | 0.03 |
| Desire for something savory b (mm) | 40.3 ± 28.7 | 42.0 ± 22.2 | 1.8 ± 33.5 | 49.1 ± 26.9 | 42.3 ± 23.1 | −6.8 ± 25.4 | 0.73 |
| Desire for something fatty b (mm) | 69.4 ± 29.5 | 68.1 ± 30.2 | −1.3 ± 30.2 | 61.3 ± 27.7 | 58.9 ± 20.0 | −2.4 ± 30.2 | 0.39 |
| ACS (n = 20) | KD (n = 25) | ||||||
|---|---|---|---|---|---|---|---|
| WEEK 0 | WEEK 12 | Δ | WEEK 0 | WEEK 12 | Δ | p | |
| Starches a | 1.7 ± 0.7 | 1.7 ± 0.6 | 0.0 ± 0.8 | 1.8 ± 0.6 | 1.3 ± 0.6 | −0.4 ± 0.6 * | 0.03 |
| High−fat foods a | 2.2 ± 0.5 | 2.2 ± 0.5 | 0.0 ± 0.4 | 2.2 ± 0.5 | 2.0 ± 0.5 | −0.1 ± 0.4 | 0.33 |
| Sweets | 2.2 ± 0.6 | 1.8 ± 1.0 | −0.4 ± 1.2 | 2.2 ± 0.7 | 1.9 ± 0.8 | −0.3 ± 0.7 * | 0.84 |
| Fast food fats | 2.1 ± 0.7 | 2.1 ± 0.6 | 0.0 ± 0.7 | 2.1 ± 0.4 | 1.7 ± 0.7 | −0.4 ± 0.6 * | 0.04 |
| Overall cravings a | 2.0 ± 0.4 | 1.9 ± 0.5 | −0.1 ± 0.6 | 2.0 ± 0.4 | 1.7 ± 0.5 | −0.3 ± 0.4 * | 0.11 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cohen, C.W.; Fontaine, K.R.; Arend, R.C.; Soleymani, T.; Gower, B.A. Favorable Effects of a Ketogenic Diet on Physical Function, Perceived Energy, and Food Cravings in Women with Ovarian or Endometrial Cancer: A Randomized, Controlled Trial. Nutrients 2018, 10, 1187. https://doi.org/10.3390/nu10091187
Cohen CW, Fontaine KR, Arend RC, Soleymani T, Gower BA. Favorable Effects of a Ketogenic Diet on Physical Function, Perceived Energy, and Food Cravings in Women with Ovarian or Endometrial Cancer: A Randomized, Controlled Trial. Nutrients. 2018; 10(9):1187. https://doi.org/10.3390/nu10091187
Chicago/Turabian StyleCohen, Caroline W., Kevin R. Fontaine, Rebecca C. Arend, Taraneh Soleymani, and Barbara A. Gower. 2018. "Favorable Effects of a Ketogenic Diet on Physical Function, Perceived Energy, and Food Cravings in Women with Ovarian or Endometrial Cancer: A Randomized, Controlled Trial" Nutrients 10, no. 9: 1187. https://doi.org/10.3390/nu10091187
APA StyleCohen, C. W., Fontaine, K. R., Arend, R. C., Soleymani, T., & Gower, B. A. (2018). Favorable Effects of a Ketogenic Diet on Physical Function, Perceived Energy, and Food Cravings in Women with Ovarian or Endometrial Cancer: A Randomized, Controlled Trial. Nutrients, 10(9), 1187. https://doi.org/10.3390/nu10091187





