Association between Vitamin D Deficiency and Single Nucleotide Polymorphisms in the Vitamin D Receptor and GC Genes and Analysis of Their Distribution in Mexican Postmenopausal Women
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants—Second Wave of the Health Workers Cohort Study
2.2. Data Collection and Blood Sample Collection
2.3. Anthropometric Measurements
2.4. Body Composition Measurements
2.5. Dietary Measurements
2.6. Biological Measurements
2.7. Measurement of Plasma 25(OH)D
2.8. Single Nucleotide Polymorphisms Selection and Genotyping
2.9. Construction of the Genetic Risk Score (GRS)
2.10. Statistical Analysis
3. Results
3.1. Participant Characteristics
3.2. Factors Asociated with Vitamin D Deficiency
3.3. Association between SNPs and Vitamin D Deficiency
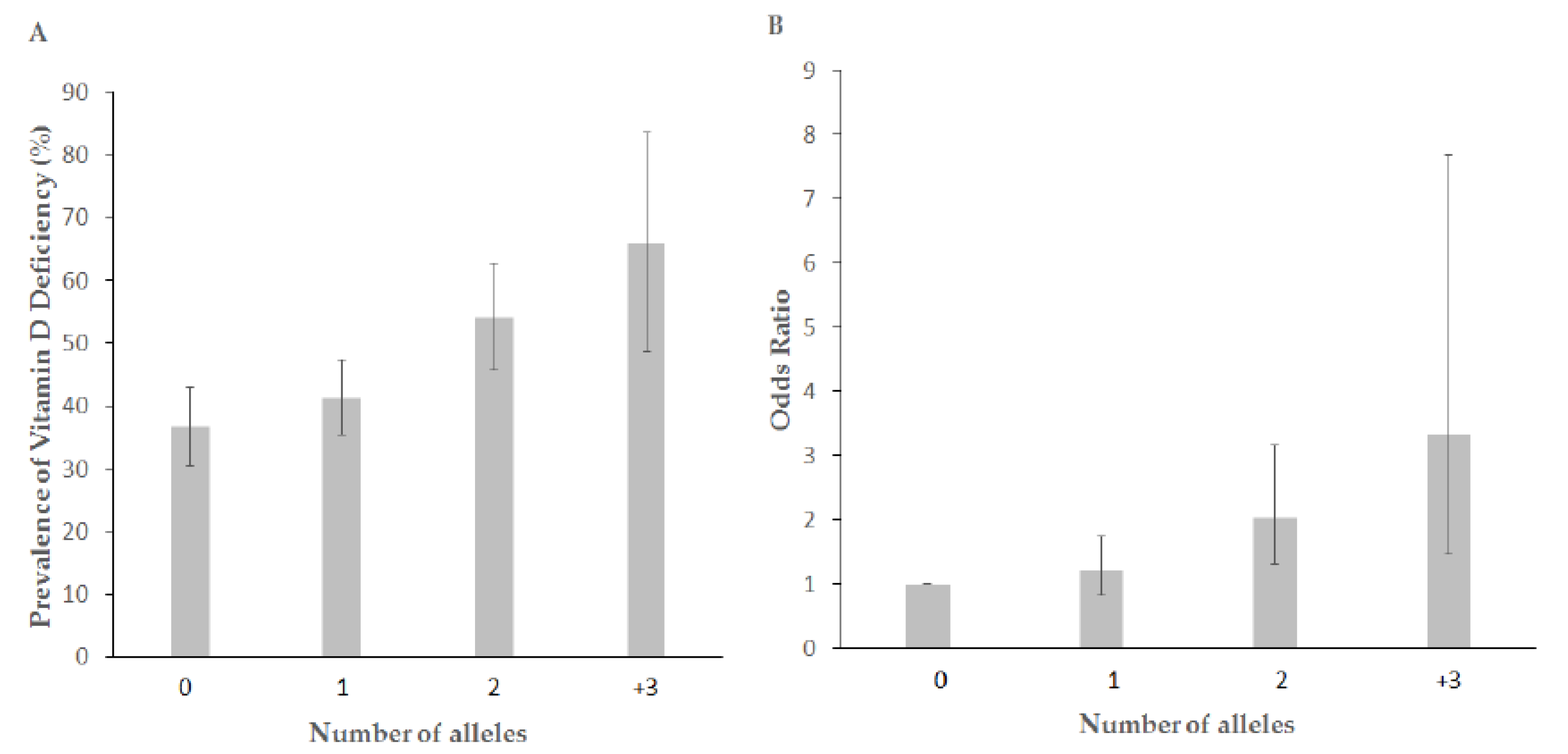
3.4. Association between GRS and Vitamin D Deficiency
3.5. Association between SNPs and Bone Mineral Density
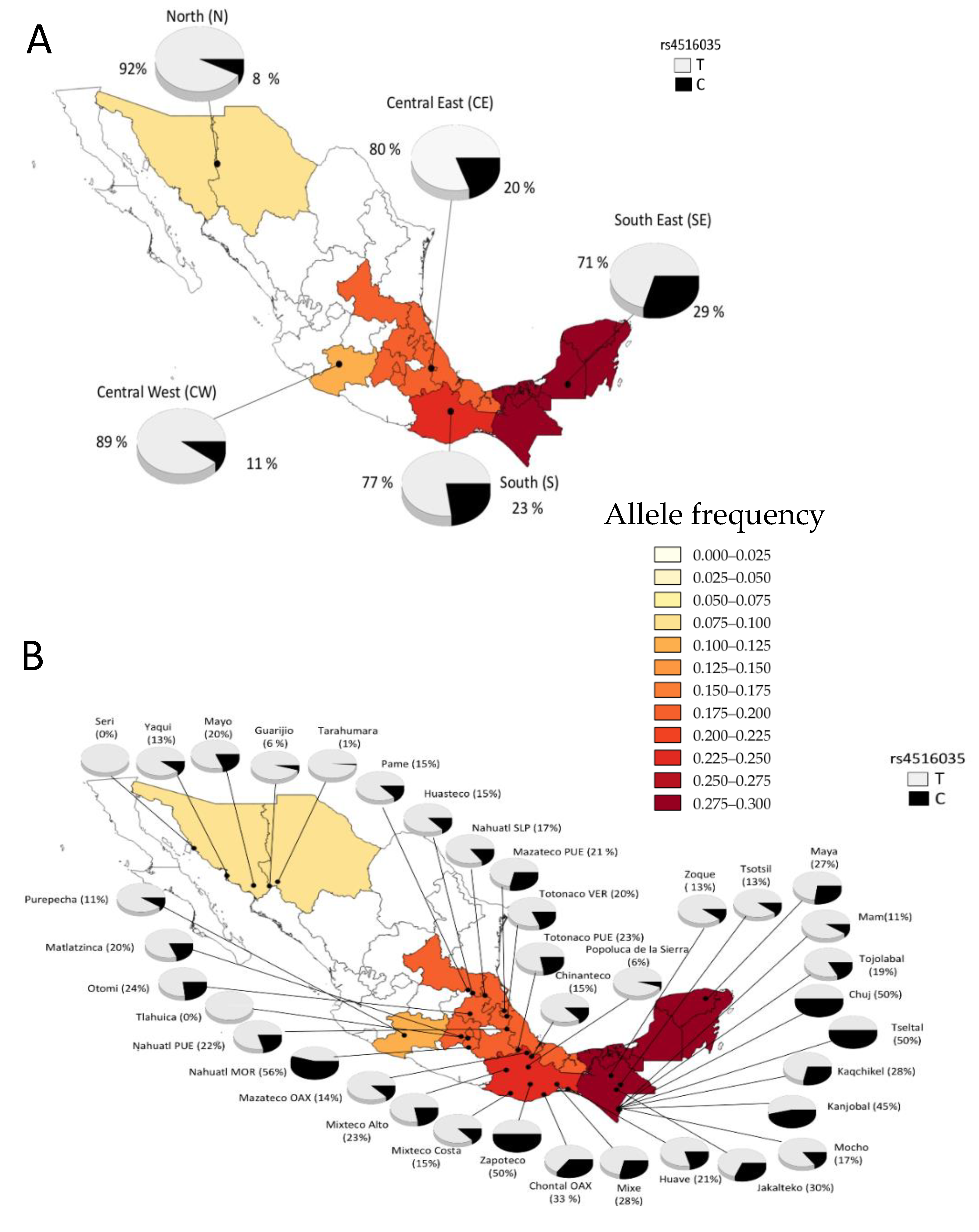
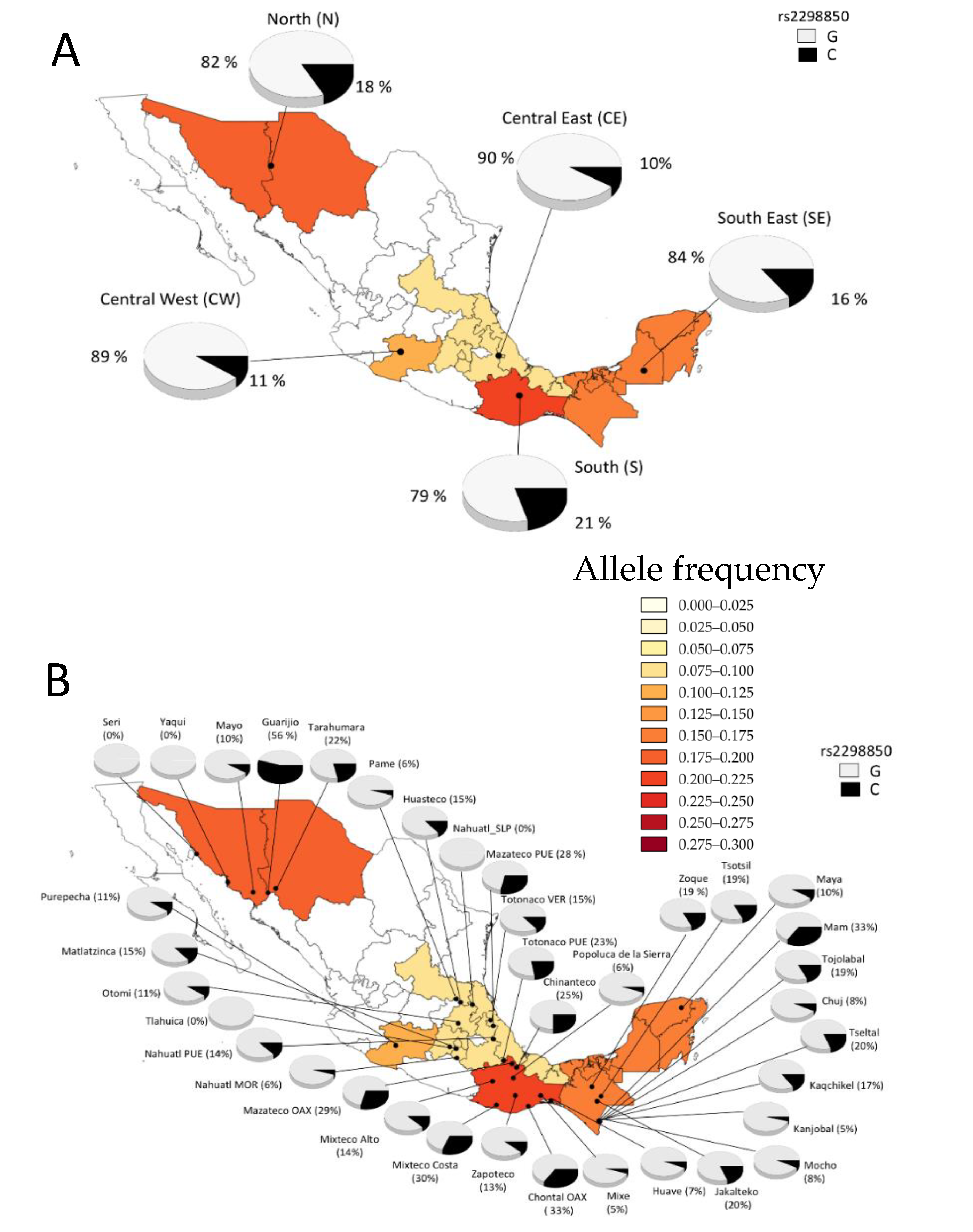
3.6. Minor Allele Frequency
3.7. Minor Allele Frequency from Different Ethnic Groups
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ojeda-Lavín, A. Consejo Nacional de Población (CONAPO). Available online: http://www.conapo.gob.mx/es/CONAPO/Proyecciones_Datos (accessed on 2 April 2018).
- Clark, P.; Tamayo, J.A.; Cisneros, F.; Rivera, F.C.; Valdés, M. Epidemiology of osteoporosis in Mexico. Present and future directions. Rev. Investig. Clin. 2013, 65, 183–191. [Google Scholar]
- Clark, P.; Lavielle, P.; Franco-Marina, F.; Ramírez, E.; Salmerón, J.; Kanis, J.A.; Cummings, S.R. Incidence rates and life-time risk of hip fractures in Mexicans over 50 years of age: A population-based study. Osteoporos. Int. 2005, 16, 2025–2030. [Google Scholar] [CrossRef] [PubMed]
- Kuchuck, N.O.; Pluijm, S.M.; Van Schoor, N.M.; Smit, J.H.; Lips, P. Relationships of serum 25-hydroxyvitamin D to bone mineral density and serum parathyroid hormone and markers of bone turnover in older persons. J. Clin. Endocrinol. Metab. 2009, 94, 1244–1250. [Google Scholar] [CrossRef] [PubMed]
- Hwang, S.; Choi, H.S.; Kim, K.M.; Rhee, Y.; Lim, S.K. Association between serum 25-hydroxyvitramin D and bone mineral density and proximal geometry in Korean: The Korean National Health and Nutrition Examinations Surveys (KNHANES) 2008–2009. Osteoporos. Int. 2014, 26, 163–171. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F.; Chen, T.C. Vitamin D deficiency: A worldwide problem with health consequences. Am. J. Clin. Nutr. 2008, 87, S1080–S1086. [Google Scholar] [CrossRef] [PubMed]
- Elizondo-Montemayor, E.; Ugalde-Asas, P.; Serrano-González, M.; Cuello-García, C.; Borbolla-Escoboza, J. Serum 25-hyroxyvitamin D concentration, life factors and obesity in Mexican Children. Obesity 2010, 18, 1805–1811. [Google Scholar] [CrossRef] [PubMed]
- Flores, M.; Barquera, S.; Sánchez, L.; Macías, N.; Lozada, A.; Díaz, E.; Barquera, S. Concentraciones de Vitaminas D en niños, Adolescentes y Adultos Mexicanos Resultados de la ENSANUT-2006. Available online: http://www.cdi.salud.gob.mx:8080/BasesCDI/Archivos/EstudioseInvestigaciones/ReporteVitaminaD.pdf (accessed on 10 January 2018).
- Carrillo-Vega, M.F.; García-Peña, C.; Gutiérrez-Robledo, L.M.; Pérez-Zepeda, M.U. Vitamin D deficiency in older adults and its associated factors: A cross-sectional analysis of the Mexican Health and Aging Study. Arch. Osteoporos. 2017, 12, 8. [Google Scholar] [CrossRef] [PubMed]
- McMillan, A.; Hicks, J.; Isabella, C.; Higa, G.M. A critical analysis of the (near) legendary status of vitamin D. Expert Rev. Endocrinol. 2012, 7, 103–119. [Google Scholar] [CrossRef]
- Nair, R.; Maseeh, A. Vitamin D: The “sunshine” vitamin. J. Pharmacol. Pharmacother. 2012, 3, 118–126. [Google Scholar] [CrossRef] [PubMed]
- Hunter, D.; de Lange, M.; Snieder, H.; MacGregor, A.J.; Swaminathan, R.; Thakker, R.V.; Spector, T.D. Genetic contribution to bone metabolism, calcium excretion, and vitamin D and parathyroid hormone regulation. J. Bone Miner. Res. 2001, 16, 371–378. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.J.; Zhang, F.; Richards, J.B.; Kestenbaum, B.; Meurs, J.B.; Berry, D.; Kiel, D.; Streeten, E.A.; Ohlsson, C.; Koller, D.L.; et al. Common genetic determinant of vitamin d insufficiency: A genome-wide association study. Lancet 2010, 376, 180–188. [Google Scholar] [CrossRef]
- Wang, W.; Ingles, S.A.; Torres-Mejía, G.; Stern, M.C.; Stanczyc, F.; Schwartz, G.G.; Nelson, D.O.; Fejerman, L.; Wolff, R.K.; Slattery, M.L.; et al. Genetic variants and non-genetic factors predict circulating vitamin D levels in Hispanic and non-Hispanic white women: The breast cancer disparities study. In. J. Mol. Epidemiol. Genet. 2014, 5, 31–46. [Google Scholar]
- Denova-Gutiérrez, E.; Flores, Y.N.; Gallegos-Carrillo, K.; Ramírez-Palacios, P.; Rivera-Paredez, B.; Muñoz-Aguirre, P.; Velázquez-Cruz, R.; Torres-Ibarra, L.; Meneses-León, J.; Méndez-Hernández, P.; et al. Health workers cohort study: Methods and study design. Salud Publica Mex. 2016, 58, 708–716. [Google Scholar] [CrossRef] [PubMed]
- Contreras-Cubas, C.; Sánchez-Hernández, B.E.; García-Ortiz, H.; Martínez-Hernández, A.; Barajas-Olmos, F.; Cid, M.; Mendoza-Caamal, E.C.; Centeno-Cruz, F.; Ortiz-Cruz, G.; Jiménez-López, J.C. Heterogenous distribution of mthfr gene variants among mestizos and diverse amerindian groups from mexico. PLoS ONE 2016, 11, e0163248. [Google Scholar] [CrossRef] [PubMed]
- Ojeda-Granados, C.; Panduro, A.; Gonzalez-Aldaco, K.; Sepulveda-Villegas, M.; Rivera-Iñiguez, I.; Roman, S. Tailoring nutritional advice for mexicans based on prevalence profiles of diet-related adaptive gene polymorphisms. J. Pers. Med. 2017, 7, 16. [Google Scholar] [CrossRef] [PubMed]
- Rubi-Castellanos, R.; Martinez-Cortes, G.; Munoz-Valle, J.F.; Gonzalez-Martin, A.; Cerda-Flores, R.M.; Anaya-Palafox, M.; Rangel-Villalobos, H. Pre-Hispanic Mesoamerican demography approximates the present-day ancestry of Mestizos throughout the territory of Mexico. Am. J. Phys. Anthropol. 2009, 139, 284–294. [Google Scholar] [CrossRef] [PubMed]
- Wolf, A.M.; Hunter, D.J.; Colditz, G.A.; Manson, J.E.; Stampfer, M.J.; Corsano, K.A.; Rosner, B.; Kriska, A.; Willett, W.C. Reproducibility and validity of a self-administered physical activity questionnaire. Int. J. Epidemiol. 1994, 23, 991–999. [Google Scholar] [CrossRef] [PubMed]
- Martínez, G.M.A.; López, F.C.; Varo, J.J.; Sánchez, V.A.; Martinez, J.A. Validation of the Spanish version of the physical activity questionnaire used in the Nurses “Health Study and the Health Professionals” Follow-up Study. Public Health Nutr. 2005, 8, 920–927. [Google Scholar] [CrossRef]
- Lohman, T.; Roche, A.; Martorell, R. Anthropometric Standardization Reference Manual; Human Kinetics: Champaign, IL, USA, 1998. [Google Scholar]
- National Center for Health Statistics. National Health and Nutrition Examination Survey. Available online: https://www.cdc.gov/nchs/data/nhanes/nhanes_07_08/manual_dexa.pdf consulted (accessed on 15 January 2018).
- WHO Scientific Group on the Assessment of Osteoporosis at Primary Health Care Level. Available online: http://www.who.int/chp/topics/Osteoporosis.pdf (accessed on 15 February 2018).
- Hernández-Avila, M.; Romieu, I.; Parra, S.; Hernández-Avila, J.; Madrigal, H.; Willett, W. Validity and reproducibility of a food frequency questionnaire to assess dietary intake of women living in Mexico City. Salud Publica Mex. 1998, 40, 133–140. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Avila, M.; Resoles, M.; Parra, S.; Romieu, I. Software: Sistema de Evaluación de Hábitos Nutricionales y Consumo de Nutrimentos. Available online: http://www.insp.mx/snut2003/ (accessed on 18 August 2017).
- Tate, J.R.; Berg, K.; Couderc, R.; Dati, F.; Kostner, G.M.; Marcovina, S.M.; Rifai, N.; Sakurabayashi, I.; Steinmetz, A. International Federation of Clinical Chemistry and Laboratory Medicine (IFCC) Standardization Project for the Measurement of Lipoprotein(a). Phase 2: Selection and properties of a proposed secondary reference material for lipoprotein(a). Clin. Chem. Lab. Med. 1999, 37, 949–958. [Google Scholar] [CrossRef] [PubMed]
- Freeman, J.; Wilson, K.; Spears, R.; Shalhoub, V.; Sibley, P. Performance evaluation of four 25-hydroxyvitamin D assays to measure 25-hydroxyvitamin D2. Clin. Biochem. 2015, 48, 1097–1104. [Google Scholar] [CrossRef] [PubMed]
- Flores, A.; Flores, M.; Macias, N.; Hernández-Barrera, L.; Rivera, M.; Contreras, A.; Villalpando, S. Vitamin D deficiency is common and is associated with overweight in Mexican children aged 1–11 years. Public Health Nutr. 2017, 20, 1807–1815. [Google Scholar] [CrossRef] [PubMed]
- Ahn, J.; Yu, K.; Stolzenberg-Solomon, R.; Simon, K.C.; McCullough, M.L.; Gallicchio, L.; Jacobs, E.J.; Ascherio, A.; Helzlsouer, K.; Jacobs, K.B.; et al. Genome-wide association study of circulating vitamin D levels. Hum. Mol. Genet. 2010, 19, 2739–2745. [Google Scholar] [CrossRef] [PubMed]
- Engelman, C.D.; Fingerlin, T.E.; Langefeld, C.D.; Hicks, P.J.; Rich, S.S.; Wagenknecht, L.E.; Bowden, D.W.; Norris, J.M. Genetic and environmental determinants of 25-hydroxyvitamin D and 1,25-dihydroxyvitamin D levels in Hispanic and African Americans. J. Clin. Endocrinol. Metab. 2008, 93, 3381–3388. [Google Scholar] [CrossRef] [PubMed]
- Velázquez-Cruz, R.; García-Ortiz, H.; Castillejos-López, M.; Quiterio, M.; Valdés-Flores, M.; Orozco, L.; Villarreal-Molina, T.; Salmerón, J. WNT3A gene polymorphisms are associated with bone mineral density variation in postmenopausal mestizo women of an urban Mexican population: Findings of a pathway-based high-density single nucleotide screening. Age 2014, 36, 9635. [Google Scholar] [CrossRef] [PubMed]
- Dudbridge, F. Power and predictive accuracy of polygenic risk scores. PLoS Genet. 2013, 9, e1003348. [Google Scholar] [CrossRef]
- Xu, W.; Sun, J.; Wang, W.; Wang, X.; Jiang, Y.; Huang, W.; Zheng, X.; Wang, Q.; Ning, Z.; Pei, Y.; et al. Association of genetic variants of vit D binding protein (DBP/GC) and of the enzyme catalyzing its 25-hydroxylation (DCYP2R1) and serum vit D in postmenopausal women. Hormones 2014, 13, 345–352. [Google Scholar] [CrossRef] [PubMed]
- StataCorp. Stata Statistical Software: Release 13. 2013. Available online: https://www.stata.com/ (accessed on 1 January 2017).
- Barrett, J.C.; Fry, B.; Maller, J.; Daly, M.J. Haploview: Analysis and visualization of LD and haplotype maps. Bioinformatics 2005, 21, 263–265. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Estrada, A.; Gignoux, C.R.; Fernández-López, J.C.; Zakharia, F.; Sikora, M.; Contreras, A.V.; Acuña-Alonzo, V.; Sandoval, K.; Eng, C.; Romero-Hidalgo, S.; et al. Human genetics. The genetics of Mexico recapitulates Native American substructure and affects biomedical traits. Science 2014, 344, 1280–1285. [Google Scholar] [CrossRef] [PubMed]
- Contreras-Manzano, A.; Villalpando, S.; Robledo-Pérez, R. Vitamin D status by sociodemographic factors and body mass index in Mexican women at reproductive age. Salud Publica Mex. 2017, 59, 518–525. [Google Scholar] [CrossRef] [PubMed]
- Forrest, K.Y.; Stuhldreher, W.L. Prevalence and correlates of vitamin D deficiency in US adults. Nutr. Res. 2011, 31, 48–54. [Google Scholar] [CrossRef] [PubMed]
- Lips, P.; Hosking, D.; Lippuner, K.; Norquist, J.M.; Wehren, L.; Maalouf, G.; Ragi-Eis, S.; Chandler, J. The prevalence of vitamin D inadequacy amongst women with osteoporosis: An international epidemiological investigation. J. Intern. Med. 2006, 260, 245–254. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.J.; Kwon, M.J.; Woo, H.Y.; Park, H. Analysis of 25-hydroxyvitamin D status according to age, gender, and seasonal variation. J. Clin. Lab. Anal. 2016, 30, 905–911. [Google Scholar] [CrossRef] [PubMed]
- Rontoyanni, V.G.; Avila, J.C.; Kaul, S.; Wong, R.; Veeranki, S.P. Association between Obesity and Serum 25(OH)D Concentrations in Older Mexican Adults. Nutrients 2017, 9, 97. [Google Scholar] [CrossRef] [PubMed]
- Pathak, K.; Soares, M.J.; Calton, E.K.; Zhao, Y.; Hallett, J. Vitamin D supplementation and body weight status: A systematic review and meta-analysis of randomized controlled trials. Obes. Rev. 2014, 15, 528–537. [Google Scholar] [CrossRef] [PubMed]
- Schneider, A.L.; Pankow, J.S.; Heiss, G.; Selvin, E. Validity and reliability of self-reported diabetes in the atherosclerosis risk in communities study. Am. J. Epidemiol. 2012, 176, 738–743. [Google Scholar] [CrossRef] [PubMed]
- Tint, G.S.; Irons, M.; Elias, E.R.; Batta, A.K.; Frieden, R.; Chen, T.S.; Salen, G. Defective cholesterol biosynthesis associated with the Smith-Lemli-Opitz syndrome. N. Engl. J. Med. 1994, 330, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Heaney, R.P. Vitamin D and calcium interactions: Functional outcomes. Am. J. Clin. Nutr. 2008, 88, 541S–544S. [Google Scholar] [CrossRef] [PubMed]
- Lasky-Su, J.; Lyon, H.N.; Emilsson, V.; Heid, I.M.; Molony, C.; Raby, B.A.; Lazarus, R.; Klanderman, B.; Soto-Quiros, M.E.; Avila, L.; et al. On the replication of genetic associations: Timing can be everything! Am. J. Hum. Genet. 2008, 82, 849–858. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; O′Reilly, P.F.; Aschard, H.; Hsu, Y.H.; Richards, J.B.; Dupuis, J.; Ingelsson, E.; Karasik, D.; Pilz, S.; Berry, D.; et al. Genome-wide association study in 79,366 European-ancestry individuals informs the genetic architecture of 25-hydroxyvitamin D levels. Nat. Commun. 2018, 9, 260. [Google Scholar] [CrossRef] [PubMed]
- Hong, J.; Hatchell, K.E.; Bradfield, J.P.; Andrew, B.; Alessandra, C.; Chao-Qiang, L.; Langefeld, C.D.; Lu, L.; Lu, Y.; Lutsey, P.L.; et al. Trans-ethnic evaluation identifies novel low frequency loci associated with 25-hydroxyvitamin D. J. Clin. Endocrinol. Metab. 2018, 103, 1380–1392. [Google Scholar] [CrossRef] [PubMed]
- Lauridsen, A.L.; Vestergaard, P.; Hermann, A.P.; Brot, C.; Heickendorff, L.; Mosekilde, L.; Nexo, E. Plasma concentrations of 25-hydroxy-vitamin D and 1,25-dihydroxy-vitamin D are related to the phenotype of Gc (vitamin D-binding protein): A cross-sectional study on 595 early postmenopausal women. Calcif. Tissue Int. 2005, 77, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Baca, K.M.; Govil, M.; Zmuda, J.M.; Simhan, H.N.; Marazita, M.L.; Bodnar, L.M. Vitamin D metabolic loci and vitamin D status in Black and White pregnant women. Eur. J. Obstet. Gynecol. Reprod. Biol. 2018, 220, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Barry, E.L.; Rees, J.R.; Peacock, J.L.; Mott, L.A.; Amos, C.I.; Bostick, R.M.; Figueiredo, J.C.; Ahnen, D.J.; Bresalier, R.S.; Burke, C.A.; et al. Genetic variants in CYP2R1, CYP24A1, and VDR modify the efficacy of vitamin D3 supplementation for increasing serum 25-hydroxyvitamin D levels in a randomized controlled trial. J. Clin. Endocrinol. Metab. 2014, 99, E2133–E2137. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y.; van Meurs, J.B.J.; d’Alesio, A. Promoter and 3′-untranslated-region haplotypes in the vitamin D receptor gene predispose to osteoporotic fracture: The rotterdam study. Am. J. Hum. Genet. 2005, 77, 807–823. [Google Scholar] [CrossRef] [PubMed]
- Moon, R.J.; Harvey, N.C.; Cooper, C.; D′Angelo, S.; Curtis, E.M.; Crozier, S.R.; Barton, S.J.; Robinson, S.M.; Godfrey, K.M.; Graham, N.J.; et al. Response to antenatal cholecalciferol supplementation is associated with common vitamin d-related genetic variants. J. Clin. Endocr. Metab. 2017, 102, 2941–2949. [Google Scholar] [CrossRef] [PubMed]
- Sollid, S.T.; Hutchinson, M.Y.; Fuskevag, O.M.; Joakimsen, R.M.; Jorde, R. Large individual differences in serum 25-hydroxyvitamin D response to vitamin D supplementation: Effects of genetic factors, body mass index, and baseline concentration. Results from a randomized controlled trial. Horm. Metab. Res. 2016, 48, 27–34. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Yin, X.; Wang, J.; Xu, D.; Wang, Y.; Yang, J.; Tao, Y.; Zhang, S.; Feng, X.; Yan, C. Associations between VDR gene polymorphisms and osteoporosis risk and bone mineral density in postmenopausal women: A systematic review and meta-analysis. Sci. Rep. 2018, 8, 981. [Google Scholar] [CrossRef] [PubMed]
| Characteristics | n = 689 |
|---|---|
| Age (years) * | 62.2 (8.8) |
| 45−59, n (%) | 279 (40.9) |
| 60−74, n (%) | 340 (49.9) |
| >74, n (%) | 63 (9.2) |
| BMI (kg/m2) * | 28.1 (4.7) |
| Nutritional Status, n (%) | |
| Overweight, λ | 306 (44.9) |
| Obesity, λ | 204 (29.9) |
| Waist circumference (cm) * | 95.9 (11.3) |
| Body fat proportion * | 45.6 (6.1) |
| Leisure time physical activity (min/day) * | 20.9 (27.3) |
| Active (≥30 min/day), % | 186 (27.3) |
| Serum 25(OH)D (ng/mL) * | 21.0 (5.7) |
| 25(OH)D Deficiency (<20ng/mL), n (%) | 298 (43.8) |
| Bone Femoral neck Density (g/cm2) * | 0.87 (0.13) |
| Bone Femoral neck status, n (%) | |
| Osteopenia | 224 (32.8) |
| Osteoporosis | 32 (4.7) |
| Bone Spine Density (g/cm2) * | 1.00 (0.16) |
| Bone Spine status, n (%) | |
| Osteopenia | 324 (47.5) |
| Osteoporosis | 165 (24.2) |
| Season of blood collection, n (%) | |
| Winter | 112 (16.4) |
| Spring | 412 (60.4) |
| Summer | 113 (16.6) |
| Autumn | 45 (6.6) |
| Diet | |
| Total energy intake (kcal/day) * | 1805 (854) |
| Alcohol (g/day) * | 1.6 (4.2) |
| Vitamin D intake (IU/day) * | 199 (157) |
| Calcium intake (mg/day) * | 918 (545) |
| Calcium supplements, n (%) | 194 (28.5) |
| Medication use, n (%) | |
| Hormone replacement therapy, n (%) | 44 (6.5) |
| Non-Deficient: 25-OH-D ≥20 ng/mL n = 386 (56.2%) | Deficient: 25-OH-D <20 ng/mL n = 301 (43.8%) | p Value | |
|---|---|---|---|
| Age(years) * | 61.2 (8.4) | 63.4 (9.2) | 0.0008 |
| Age categories, n (%) | |||
| 45–59 | 180 (46.6) | 101 (33.6) | 0.006 |
| 60–74 | 179 (46.4) | 163 (54.2) | 0.04 |
| >74 | 27 (7.0) | 37 (12.3) | 0.02 |
| BMI (kg/m2) * | 28.1 (4.7) | 28.2 (4.8) | 0.76 |
| Nutritional Status, n (%) | |||
| Overweight, λ | 175 (45.3) | 133 (44.2) | 0.71 |
| Obesity, λ | 114 (29.5) | 91 (30.2) | 0.84 |
| Waist circumference (cm) * | 95.5 (11.1) | 96.4 (11.6) | 0.30 |
| Body fat proportion * | 45.7 (6.2) | 45.4 (6.1) | 0.53 |
| Leisure time physical activity (min/day) * | 20.5 (27.7) | 19.4 (26.0) | 0.60 |
| Active (≥30 min/day), % | 28.5 | 25.8 | 0.43 |
| Femoral neck (g/cm2) * | 0.89 (0.13) | 0.86 (0.13) | 0.004 |
| Femoral status, n (%) | |||
| Osteopenia | 121 (31.4) | 107 (35.7) | 0.23 |
| Osteoporosis | 17 (4.4) | 16 (5.3) | 0.57 |
| Spine BMD (g/cm2) * | 1.01 (0.16) | 0.99 (0.16) | 0.004 |
| Spine status, n (%) | |||
| Osteopenia | 187 (48.6) | 139 (46.3) | 0.55 |
| Osteoporosis | 83 (21.6) | 84 (28.0) | 0.05 |
| Season of blood collection, n (%) | |||
| Winter | 54 (14.8) | 55 (18.3) | 0.22 |
| Spring | 249 (64.5) | 163 (54.1) | 0.006 |
| Summer | 58 (15.0) | 61 (20.3) | 0.07 |
| Autumn | 22 (5.7) | 22 (7.3) | 0.40 |
| Diet | |||
| Total energy intake (kcal/day) * | 1807 (826) | 1814 (895) | 0.92 |
| Alcohol intake (g/day) * | 1.5 (3.5) | 1.8 (4.9) | 0.28 |
| Vitamin D intake (IU/day) * | 209 (160) | 188 (151) | 0.02 |
| Calcium intake (mg/day) * | 941 (537) | 890 (554) | 0.22 |
| Calcium supplements, n (%) | 116 (30.1) | 79 (26.3) | 0.29 |
| Medication use | |||
| Hormone replacement therapy, n (%) | 23 (6.0) | 21 (7.0) | 0.60 |
| Calcium, n (%) | 9 (2.3) | 5 (1.7) | 0.55 |
| Gene | SNP | Genotype | Not Deficient * n (%) | Deficient * n (%) | Unadjuste d OR (CI 95%) | p Value | Adjusted Model OR (CI 95%) | p Value |
|---|---|---|---|---|---|---|---|---|
| VDR | rs10783219 | TT | 135 (35.3) | 94 (31.5) | 1.0 | 1.0 | ||
| TA | 170 (44.5) | 145 (48.7) | 1.22 (0.87–1.73) | 0.248 | 1.20 (0.84–1.72) | 0.315 | ||
| AA | 77 (20.2) | 59 (19.8) | 1.10 (0.72–1.69) | 0.662 | 1.13 (0.72–1.75) | 0.597 | ||
| Additive model | 1.07 (0.87–1.32) | 0.537 | 1.10 (0.89–1.38) | 0.377 | ||||
| VDR | rs4516035 | TT | 219 (57.5) | 146 (49.0) | 1.0 | 1.0 | ||
| TC | 141 (37.0) | 130 (43.6) | 1.38 (1.01–1.90) | 0.045 | 1.41 (1.01–1.95) | 0.041 | ||
| CC | 21 (5.5) | 22 (7.4) | 1.57 (0.83–2.96) | 0.162 | 1.75 (0.91–3.35) | 0.091 | ||
| Additive model | 1.31 (1.02–1.68) | 0.032 | 1.40 (1.08–4.43) | 0.012 | ||||
| GC | rs2282679 | TT | 247 (64.6) | 164 (55.0) | 1.0 | 1.0 | ||
| TG | 127 (33.3) | 119 (39.9) | 1.41 (1.03–1.94) | 0.034 | 1.48 (1.07–2.06) | 0.019 | ||
| GG | 8 (2.1) | 15 (2.0) | 2.82 (1.17–6.81) | 0.021 | 3.00 (1.20–7.49) | 0.019 | ||
| Additive model | 1.50 (1.14–1.97) | 0.003 | 1.53 (1.15–2.04) | 0.003 | ||||
| Populations | Genotypic Frequencies | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Geographical Region | Ethnic Group | rs2298850 (Proxy for rs2282679) | rs4516035 | ||||||||||
| GG | GC | CC | TT | CT | CC | ||||||||
| Count | Freq | Count | Freq | Count | Freq | Count | Freq | Count | Freq | Count | Freq | ||
| North (N) | Guarijio | 2 | 0.22 | 4 | 0.44 | 3 | 0.33 | 8 | 0.89 | 1 | 0.11 | 0 | 0 |
| Mayo | 4 | 0.80 | 1 | 0.20 | 0 | 0 | 3 | 0.60 | 2 | 0.40 | 0 | 0 | |
| Seri | 7 | 1.0 | 0 | 0 | 0 | 0 | 8 | 1.0 | 0 | 0 | 0 | 0 | |
| Tarahumara | 19 | 0.56 | 15 | 0.44 | 0 | 0 | 33 | 0.97 | 1 | 0.03 | 0 | 0 | |
| Yaqui | 4 | 1.0 | 0 | 0 | 0 | 0 | 3 | 0.75 | 1 | 0.25 | 0 | 0 | |
| Total | 36 | 0.72 | 19 | 0.22 | 3 | 0.067 | 55 | 0.84 | 5 | 0.16 | 0 | 0 | |
| Central West (CW) | Purepecha | 7 | 0.78 | 2 | 0.22 | 0 | 0 | 7 | 0.78 | 2 | 0.22 | 0 | 0 |
| Total | 7 | 0.78 | 2 | 0.22 | 0 | 0 | 7 | 0.78 | 2 | 0.22 | 0 | 0 | |
| Central East (CE) | Huasteco | 7 | 0.70 | 3 | 0.30 | 0 | 0 | 7 | 0.70 | 3 | 0.30 | 0 | 0 |
| Matlaltzinca | 8 | 0.80 | 1 | 0.10 | 1 | 0.10 | 6 | 0.60 | 4 | 0.40 | 0 | 0 | |
| Mazateco PUE | 4 | 0.44 | 5 | 0.56 | 0 | 0 | 4 | 0.44 | 5 | 0.56 | 0 | 0 | |
| Nahuatl MOR | 8 | 0.89 | 1 | 0.11 | 0 | 0 | 0 | 0 | 8 | 0.89 | 1 | 0.11 | |
| Nahuatl PUE | 5 | 0.71 | 2 | 0.29 | 0 | 0 | 4 | 0.57 | 3 | 0.43 | 0 | 0 | |
| Nahuatl SLP | 6 | 1.0 | 0 | 0 | 0 | 0 | 4 | 0.67 | 2 | 0.33 | 0 | 0 | |
| Otomi | 28 | 0.78 | 8 | 0.22 | 0 | 0 | 22 | 0.61 | 11 | 0.31 | 3 | 0.08 | |
| Pame | 8 | 0.89 | 1 | 0.11 | 0 | 0 | 7 | 0.70 | 3 | 0.30 | 0 | 0 | |
| Popoluca de la Sierra | 7 | 0.88 | 1 | 0.12 | 0 | 0 | 7 | 0.88 | 1 | 0.12 | 0 | 0 | |
| Tlahuica | 5 | 1.0 | 0 | 0 | 0 | 0 | 5 | 1.0 | 0 | 0 | 0 | 0 | |
| Totonaco PUE | 6 | 0.55 | 5 | 0.45 | 0 | 0 | 6 | 0.55 | 5 | 0.45 | 0 | 0 | |
| Totonaco VER | 7 | 0.70 | 3 | 0.30 | 0 | 0 | 6 | 0.60 | 4 | 0.40 | 0 | 0 | |
| Total | 99 | 0.79 | 30 | 0.206 | 1 | 0.009 | 78 | 0.601 | 49 | 0.381 | 4 | 0.018 | |
| South (S) | Chinanteco | 5 | 0.50 | 5 | 0.50 | 0 | 0 | 7 | 0.70 | 3 | 0.30 | 0 | 0 |
| Chontal OAX | 3 | 0.50 | 2 | 0.33 | 1 | 0.17 | 3 | 0.50 | 2 | 0.33 | 1 | 0.17 | |
| Huave | 6 | 0.86 | 1 | 0.14 | 0 | 0 | 5 | 0.72 | 1 | 0.14 | 1 | 0.14 | |
| Mazateco OAX | 4 | 0.57 | 2 | 0.29 | 1 | 0.14 | 5 | 0.71 | 2 | 0.29 | 0 | 0 | |
| Mixe | 8 | 0.89 | 1 | 0.1 | 0 | 0 | 4 | 0.44 | 5 | 0.56 | 0 | 0 | |
| Mixteco Alto | 8 | 0.73 | 3 | 0.27 | 0 | 0 | 6 | 0.55 | 5 | 0.45 | 0 | 0 | |
| Mixteco Costa | 5 | 0.50 | 4 | 0.40 | 1 | 0.10 | 7 | 0.70 | 3 | 0.30 | 0 | 0 | |
| Zapoteco | 6 | 0.75 | 2 | 0.25 | 0 | 0 | 3 | 0.38 | 2 | 0.24 | 3 | 0.38 | |
| Total | 45 | 0.66 | 20 | 0.29 | 3 | 0.05 | 40 | 0.587 | 23 | 0.328 | 5 | 0.086 | |
| South East (SE) | Chuj | 5 | 0.83 | 1 | 0.17 | 0 | 0 | 2 | 0.33 | 2 | 0.33 | 2 | 0.33 |
| Jakalteco | 3 | 0.60 | 2 | 0.40 | 0 | 0 | 2 | 0.40 | 3 | 0.60 | 0 | 0 | |
| Kanjobal | 9 | 0.90 | 1 | 0.10 | 0 | 0 | 2 | 0.20 | 7 | 0.70 | 1 | 0.10 | |
| Kaqchikel | 6 | 0.67 | 3 | 0.33 | 0 | 0 | 5 | 0.56 | 3 | 0.33 | 1 | 0.11 | |
| Mam | 4 | 0.44 | 4 | 0.44 | 1 | 0.12 | 7 | 0.78 | 2 | 0.22 | 0 | 0 | |
| Maya | 12 | 0.80 | 3 | 0.20 | 0 | 0 | 9 | 0.60 | 4 | 0.27 | 2 | 0.13 | |
| Mocho | 5 | 0.83 | 1 | 0.17 | 0 | 0 | 5 | 0.83 | 0 | 0 | 1 | 0.17 | |
| Tojolabal | 5 | 0.63 | 3 | 0.37 | 0 | 0 | 5 | 0.63 | 3 | 0.37 | 0 | 0 | |
| Tseltal | 3 | 0.60 | 2 | 0.40 | 0 | 0 | 1 | 0.20 | 3 | 0.60 | 1 | 0.20 | |
| Tsotsil | 5 | 0.63 | 3 | 0.37 | 0 | 0 | 6 | 0.75 | 2 | 0.25 | 0 | 0 | |
| Zoque | 3 | 0.67 | 2 | 0.33 | 0 | 0 | 4 | 0.80 | 1 | 0.20 | 0 | 0 | |
| Total | 60 | 0.68 | 25 | 0.31 | 1 | 0.01 | 48 | 0.552 | 30 | 0.353 | 8 | 0.095 | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rivera-Paredez, B.; Macías, N.; Martínez-Aguilar, M.M.; Hidalgo-Bravo, A.; Flores, M.; Quezada-Sánchez, A.D.; Denova-Gutiérrez, E.; Cid, M.; Martínez-Hernández, A.; Orozco, L.; et al. Association between Vitamin D Deficiency and Single Nucleotide Polymorphisms in the Vitamin D Receptor and GC Genes and Analysis of Their Distribution in Mexican Postmenopausal Women. Nutrients 2018, 10, 1175. https://doi.org/10.3390/nu10091175
Rivera-Paredez B, Macías N, Martínez-Aguilar MM, Hidalgo-Bravo A, Flores M, Quezada-Sánchez AD, Denova-Gutiérrez E, Cid M, Martínez-Hernández A, Orozco L, et al. Association between Vitamin D Deficiency and Single Nucleotide Polymorphisms in the Vitamin D Receptor and GC Genes and Analysis of Their Distribution in Mexican Postmenopausal Women. Nutrients. 2018; 10(9):1175. https://doi.org/10.3390/nu10091175
Chicago/Turabian StyleRivera-Paredez, Berenice, Nayeli Macías, Mayeli M. Martínez-Aguilar, Alberto Hidalgo-Bravo, Mario Flores, Amado D. Quezada-Sánchez, Edgar Denova-Gutiérrez, Miguel Cid, Angelica Martínez-Hernández, Lorena Orozco, and et al. 2018. "Association between Vitamin D Deficiency and Single Nucleotide Polymorphisms in the Vitamin D Receptor and GC Genes and Analysis of Their Distribution in Mexican Postmenopausal Women" Nutrients 10, no. 9: 1175. https://doi.org/10.3390/nu10091175
APA StyleRivera-Paredez, B., Macías, N., Martínez-Aguilar, M. M., Hidalgo-Bravo, A., Flores, M., Quezada-Sánchez, A. D., Denova-Gutiérrez, E., Cid, M., Martínez-Hernández, A., Orozco, L., Quiterio, M., Flores, Y. N., Salmerón, J., & Velázquez-Cruz, R. (2018). Association between Vitamin D Deficiency and Single Nucleotide Polymorphisms in the Vitamin D Receptor and GC Genes and Analysis of Their Distribution in Mexican Postmenopausal Women. Nutrients, 10(9), 1175. https://doi.org/10.3390/nu10091175







