Nontoxic Glucomoringin-Isothiocyanate (GMG-ITC) Rich Soluble Extract Induces Apoptosis and Inhibits Proliferation of Human Prostate Adenocarcinoma Cells (PC-3)
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Preparation and Characterization
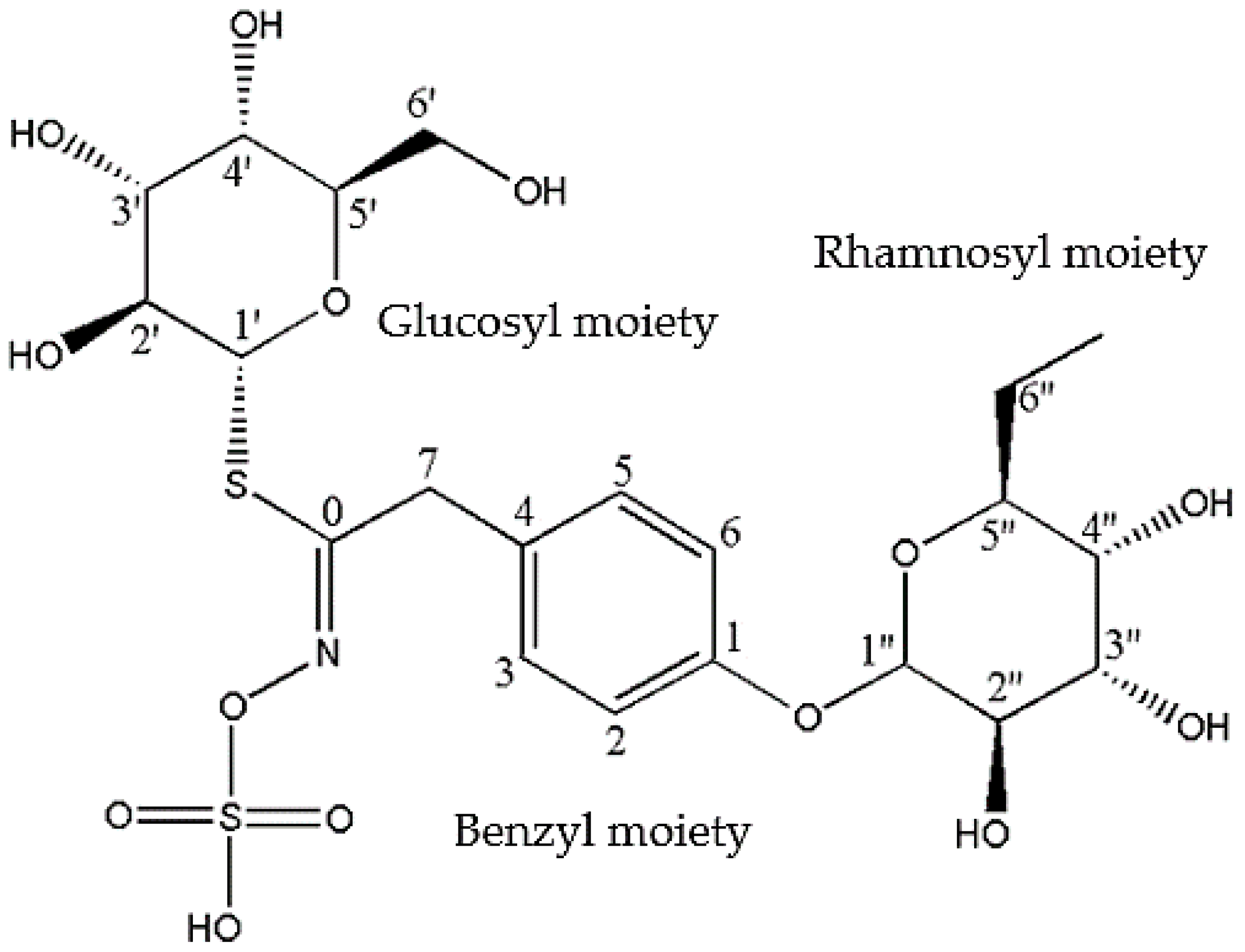
2.2. Nuclear Magnetic Resonance (NMR)
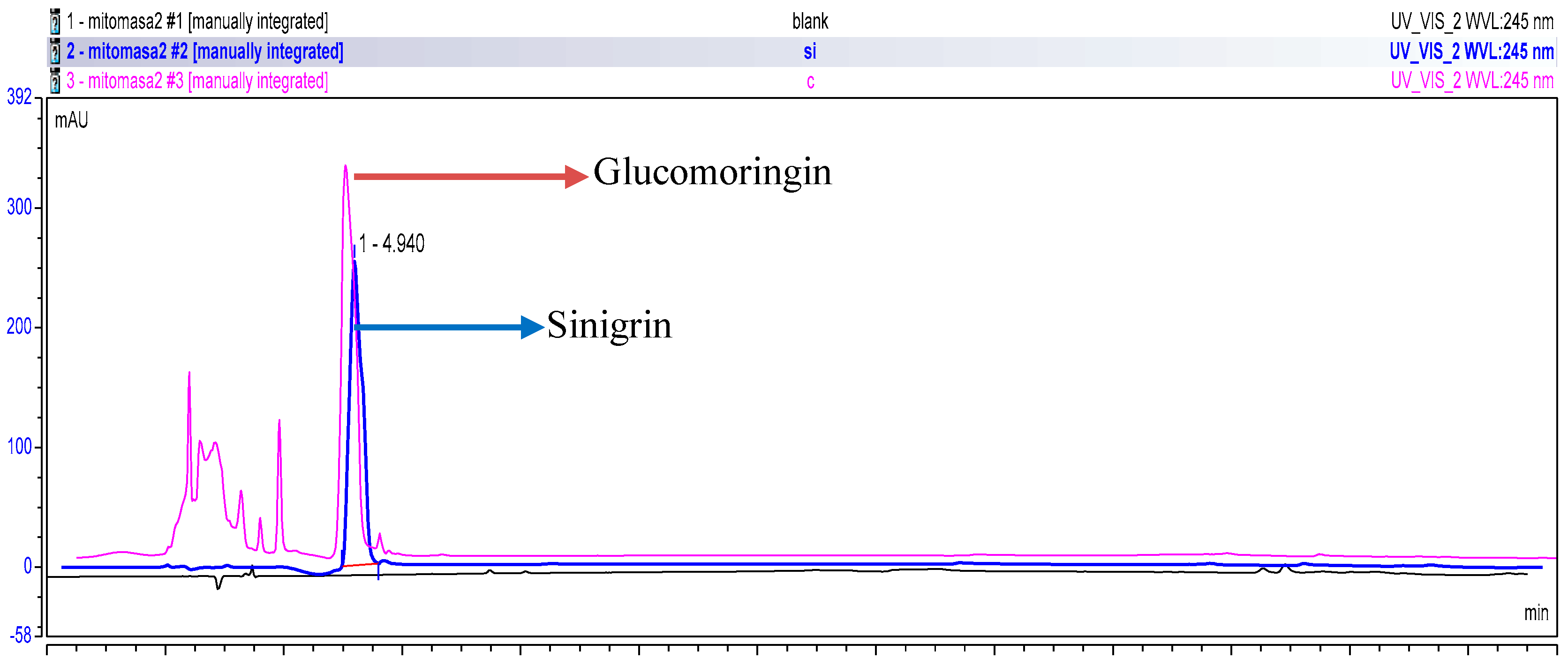
2.3. High Performance Liquid Chromatography (HPLC)
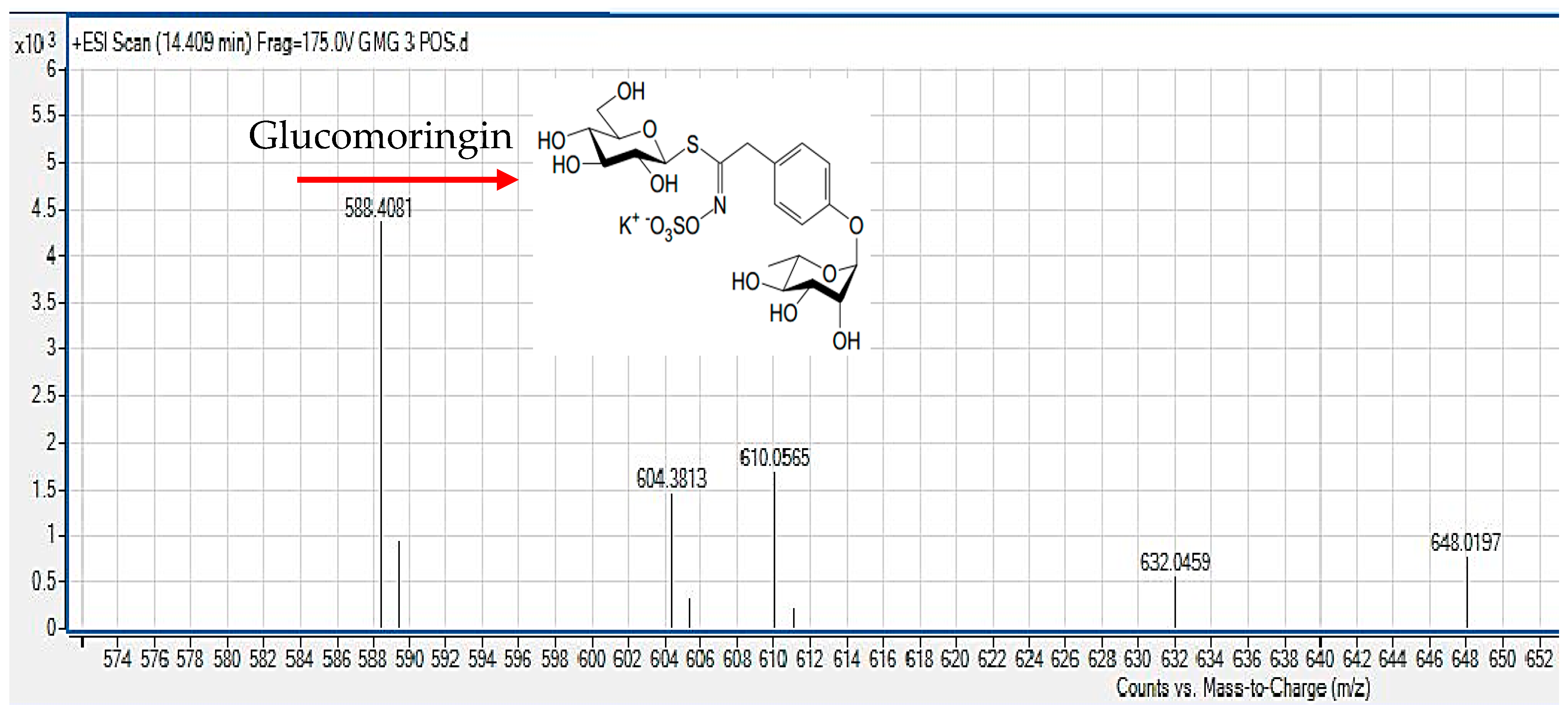
2.4. Liquid Chromatography Mass Spectrometry (LC/MS)
2.5. In Vivo Acute Toxicity
2.5.1. Ethics Statement
2.5.2. Animal Grouping and Extract Administration
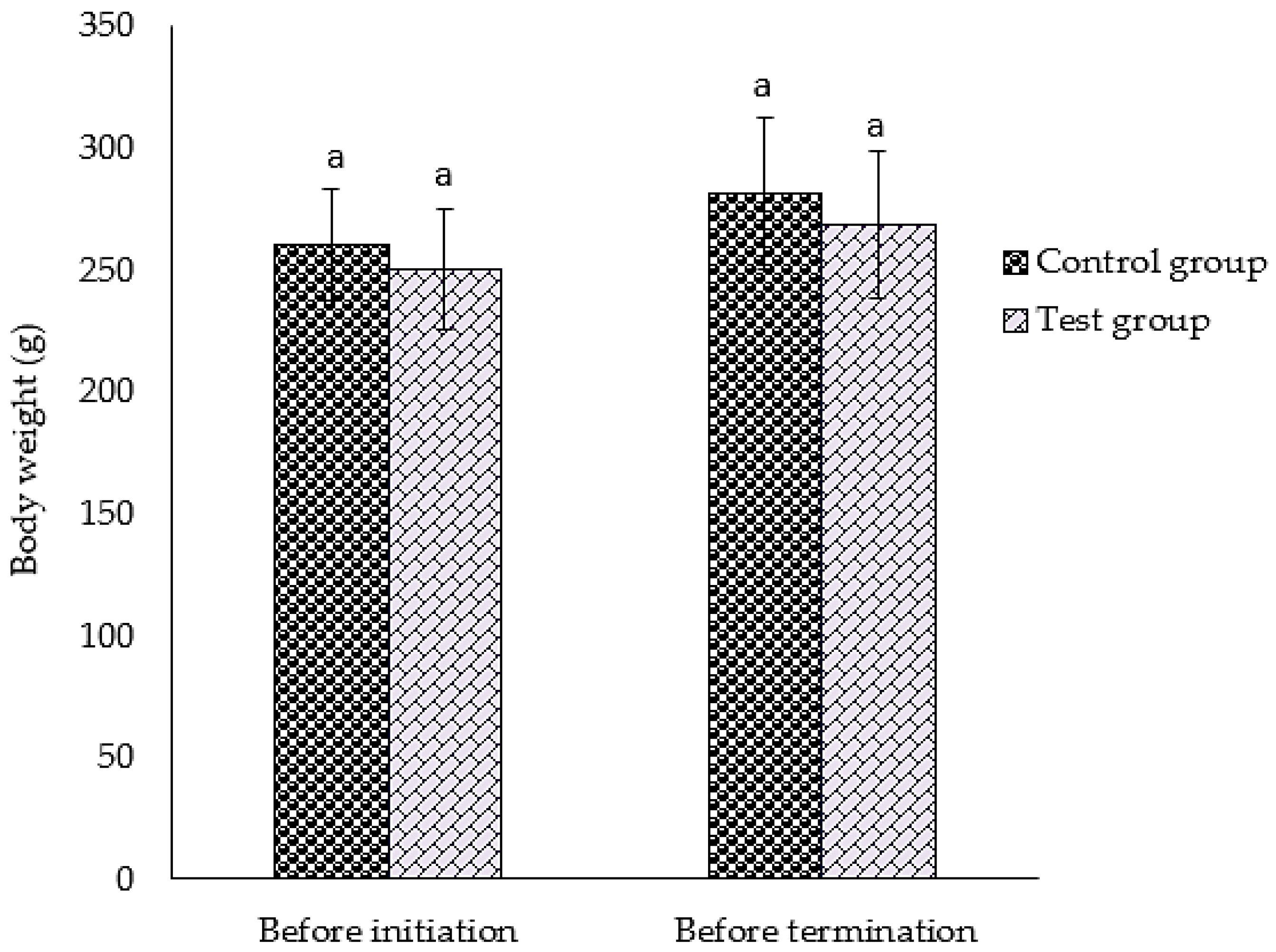
2.5.3. Body Weight Measurement and Observation of Clinical Signs
2.5.4. Necropsy
2.6. In Vitro Study
2.6.1. Cell Culture
2.6.2. Cytotoxicity and Anti-Proliferation Examination of GMG-ITC Rich Soluble Extract on PC3 and 3T3 Cells
2.6.3. Morphological Assessment of GMG-ITC Rich Soluble Extract Treated PC-3 Cells by Inverted Light Microscope
2.6.4. Morphological Assessment of Apoptotic Cells by Acridine Orange and Propidium Iodide (AO/PI) Double Staining
2.7. Statistical Analysis
3. Results
3.1. Chemical Profiling of GMG-ITC Rich Soluble Extract (GMG-ITC-RSE)
3.2. Survival, Observations, and Body Weight of Animal after GMG-ITC Rich Soluble Extract Dosage
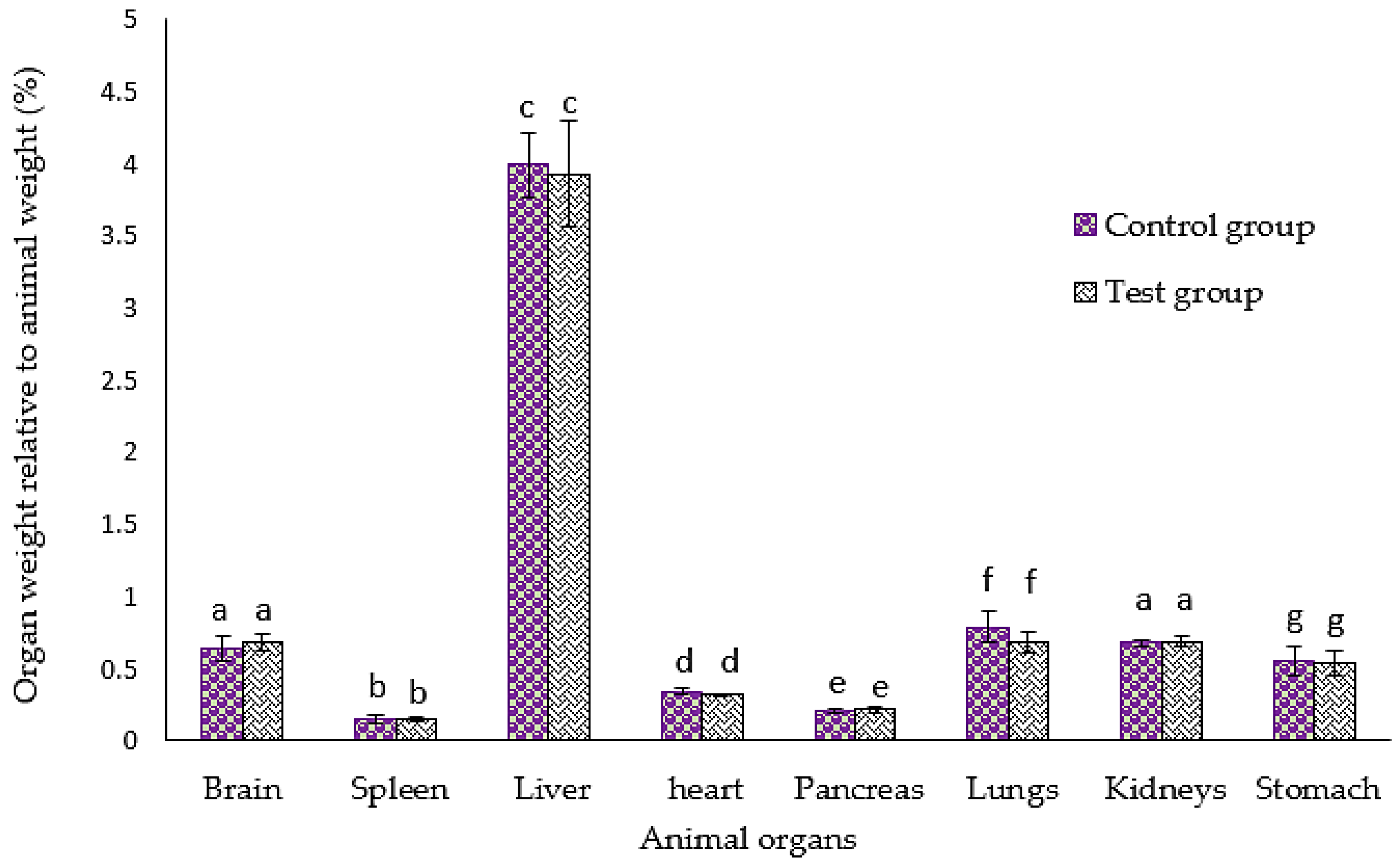
3.3. Effect of GMG-ITC Rich Soluble Extract on Animal Organs’ Weight
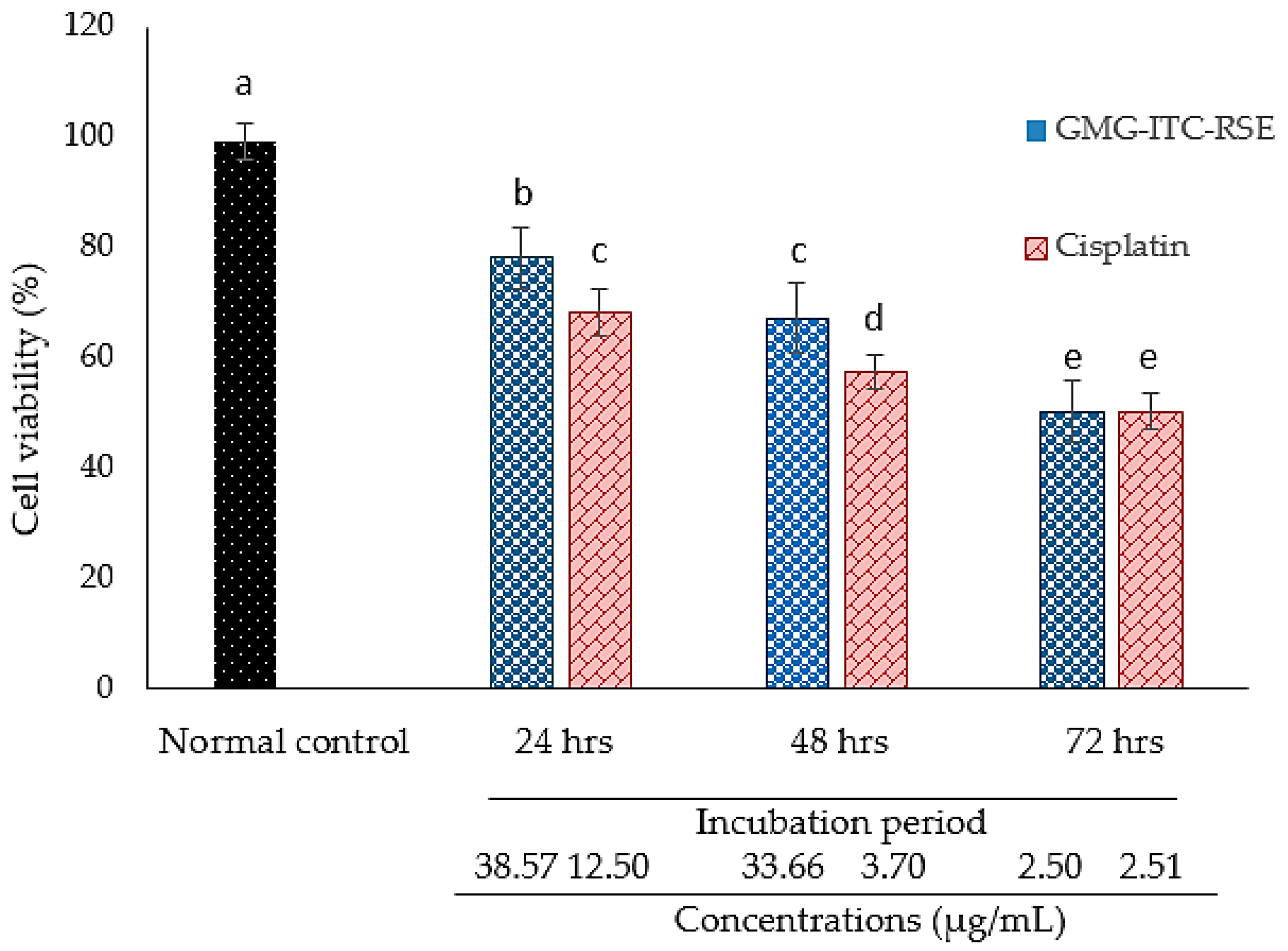
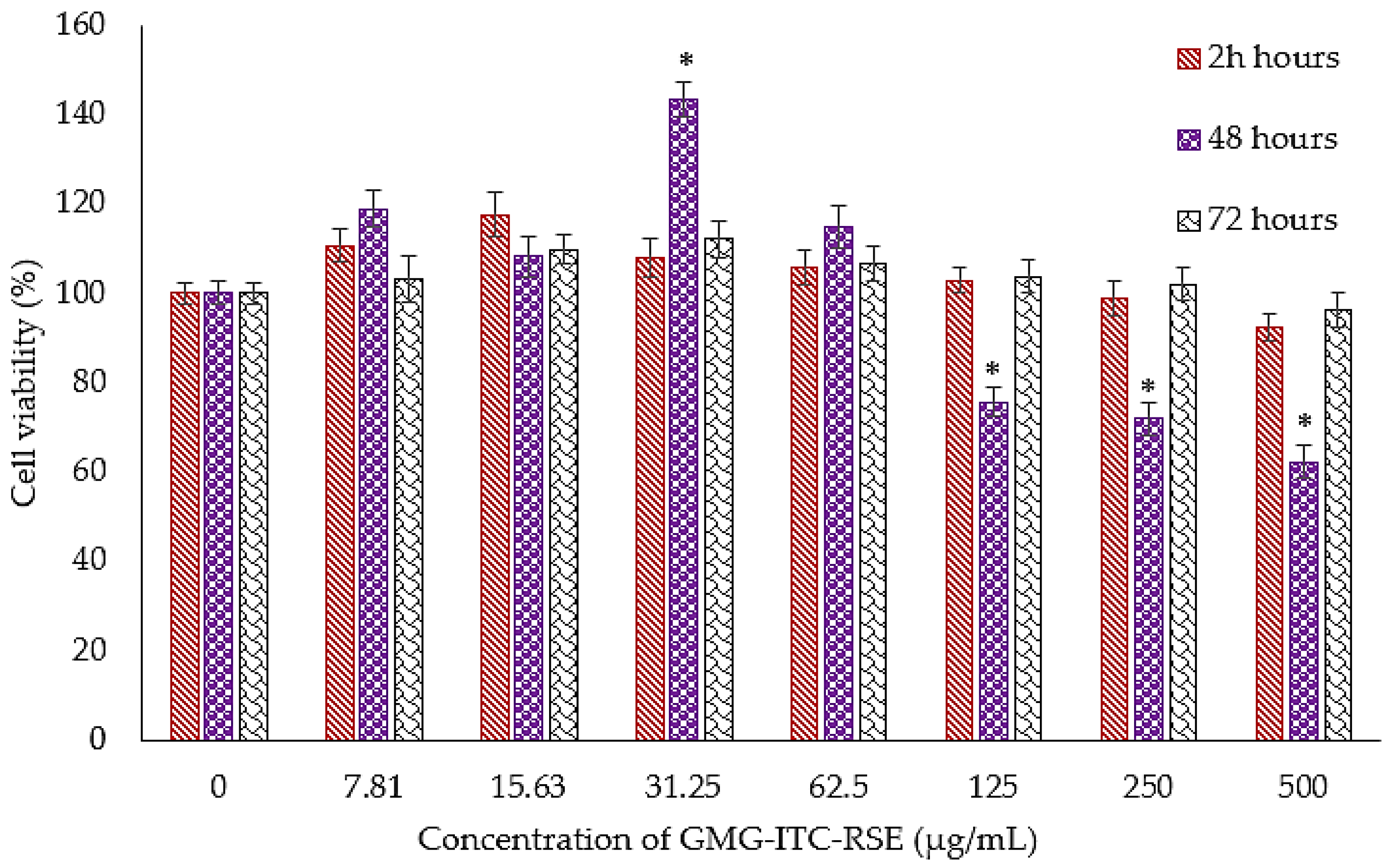
3.4. Cytotoxicity and Anti-Proliferation Evaluation of GMG-ITC Rich Soluble Extract
3.5. Morphological Assessment of PC3 Cells by Inverted Light Microscope
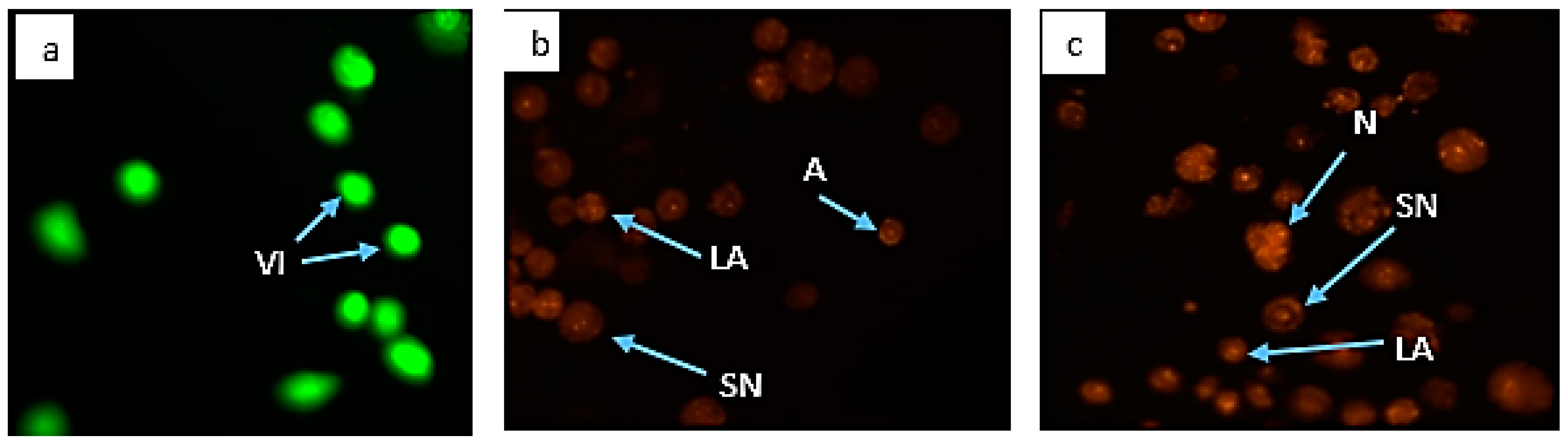
3.6. Morphological Assessment of Apoptotic Cells by AO/PI Double Staining
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Abdull, R.; Ahmad, F.; Ibrahim, M.D.; Kntayya, S.B. Health benefits of Moringa oleifera. Asian Pac. J. Cancer Prev. 2014, 15, 8571–8576. [Google Scholar] [CrossRef]
- Bako, H.Y.; Mohammad, J.S.; Wazir, P.M.; Bulus, T.; Gwarzo, M.Y.; Zubairu, M.M. Lipid profile of alloxan-induced diabetic wistar rats treated with methanolic extract of Adansonia digitata fruit pulp. Sci. World J. 2014, 9, 19–24. [Google Scholar]
- Emami, S.A.; Asili, J.; Hossein Nia, S.; Yazdian-Robati, R.; Sahranavard, M.; Tayarani-Najaran, Z. Growth Inhibition and Apoptosis Induction of Essential Oils and Extracts of Nepeta cataria L on Human Prostatic and Breast Cancer Cell Lines. Asian Pac. J. Cancer Prev. 2016, 17, 125–130. [Google Scholar] [CrossRef] [PubMed]
- Hayakawa, S.; Saito, K.; Miyoshi, N.; Ohishi, T.; Oishi, Y.; Miyoshi, M.; Nakamura, Y. Anti-cancer effects of green tea by either anti-or pro-oxidative mechanisms. Asian Pac. J. Cancer Prev. 2016, 17, 1649–1654. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Qi, Z.; Hao, Y.; Lv, C.; Jia, L.; Wang, J.; Lu, J. New Adducts of Iriflophene and Flavonoids Isolated from Sedum aizoon L. with Potential Antitumor Activity. Molecules 2017, 22, 1859. [Google Scholar]
- Moghadam, S.E.; Ebrahimi, S.N.; Salehi, P.; Farimani, M.M.; Hamburger, M.; Jabbarzadeh, E. Wound Healing Potential of Chlorogenic Acid and Myricetin-3-O-β-Rhamnoside Isolated from Parrotia persica. Molecules 2017, 22, 1501. [Google Scholar] [CrossRef] [PubMed]
- Ja’afaru, M.S.; Bako, H.Y.; Waziri, P.M.; Mustapha, M.B.; Timothy, B. Ameliorative Effect of Methanolic Extract of Cassiaoccidentalis (MECO) Whole Plant, on Triton X-100-Induced Hyperlipidaemia in Albino Rats. Int. J. Sci. Res. 2016, 5, 2141–2146. [Google Scholar]
- Correa, M.G.; Couto, J.S.; Teodoro, A.J. Anticancer Properties of Psidium guajava—A Mini-Review. Asian Pac. J. Cancer Prev. 2016, 17, 4199–4204. [Google Scholar] [PubMed]
- Karim, N.A.A.; Ibrahim, M.D.; Kntayya, S.B.; Rukayadi, Y.; Hamid, H.A.; Razis, A.F.A. Moringa oleifera Lam: Targeting Chemoprevention. Asian Pac. J. Cancer Prev. 2016, 17, 3675–3686. [Google Scholar] [PubMed]
- Jaafaru, M.S.; Abd Karim, N.A.; Enas, M.E.; Rollin, P.; Mazzon, E.; Abdull Razis, A.F. Protective Effect of Glucosinolates Hydrolytic Products in Neurodegenerative Diseases (NDDs). Nutrients 2018, 10, 580. [Google Scholar] [CrossRef] [PubMed]
- Prabhu, K.; Murugan, K.; Nareshkumar, A.; Ramasubramanian, N.; Bragadeeswaran, S. Larvicidal and repellent potential of Moringa oleifera against malarial vector, Anopheles stephensi Liston (Insecta: Diptera: Culicidae). Asian Pac. J. Trop. Biomed. 2011, 1, 124–129. [Google Scholar] [CrossRef]
- Kasolo, J.N.; Bimenya, G.S.; Ojok, L.; Ochieng, J.; Ogwal-Okeng, J.W. Phytochemicals and uses of Moringa oleifera leaves in Ugandan rural communities. J. Med. Plants Res. 2010, 4, 753–757. [Google Scholar]
- Mbikay, M. Therapeutic potential of Moringa oleifera leaves in chronic hyperglycemia and dyslipidemia: A review. Front. Pharmacol. 2012, 3, 24. [Google Scholar] [CrossRef] [PubMed]
- Stohs, S.J.; Hartman, M.J. Review of the safety and efficacy of Moringa oleifera. Phytother. Res. 2015, 29, 796–804. [Google Scholar] [CrossRef] [PubMed]
- Vergara-Jimenez, M.; Almatrafi, M.M.; Fernandez, M.L. Bioactive components in Moringa Oleifera leaves protect against chronic disease. Antioxidants 2017, 6, 91. [Google Scholar] [CrossRef] [PubMed]
- Baldisserotto, A.; Buso, P.; Radice, M.; Dissette, V.; Lampronti, I.; Gambari, R.; Manfredini, S.; Vertuani, S. Moringa oleifera Leaf Extracts as Multifunctional Ingredients for “Natural and Organic” Sunscreens and Photoprotective Preparations. Molecules 2018, 23, 664. [Google Scholar] [CrossRef] [PubMed]
- Fahey, J.W. Moringa oleifera: A Review of the Medical Evidence for Its Nutritional, Therapeutic, and Prophylactic Properties. Part 1. Trees Life J. 2005, 1, 1–15. [Google Scholar]
- Kooltheat, N.; Sranujit, R.P.; Chumark, P.; Potup, P.; Laytragoon-Lewin, N.; Usuwanthim, K. An ethyl acetate fraction of Moringa oleifera Lam. Inhibits human macrophage cytokine production induced by cigarette smoke. Nutrients 2014, 6, 697–710. [Google Scholar] [CrossRef] [PubMed]
- Anwar, F.; Latif, S.; Ashraf, M.; Gilani, A.H. Moringa oleifera: A food plant with multiple medicinal uses. Phytother. Res. 2007, 21, 17–25. [Google Scholar] [CrossRef] [PubMed]
- Omodanisi, E.I.; Aboua, Y.G.; Oguntibeju, O.O. Assessment of the anti-hyperglycaemic, anti-inflammatory and antioxidant activities of the methanol extract of Moringa oleifera in diabetes-induced nephrotoxic male wistar rats. Molecules 2017, 22, 439. [Google Scholar] [CrossRef] [PubMed]
- Wright, R.J.; Lee, K.S.; Hyacinth, H.I.; Hibbert, J.M.; Reid, M.E.; Wheatley, A.O.; Asemota, H.N. An Investigation of the Antioxidant Capacity in Extracts from Moringa oleifera Plants Grown in Jamaica. Plants 2017, 6, 48. [Google Scholar] [CrossRef] [PubMed]
- Bones, A.M.; Rossiter, J.T. The enzymic and chemically induced decomposition of glucosinolates. Phytochemistry 2006, 67, 1053–1067. [Google Scholar] [CrossRef] [PubMed]
- Halkier, B.A.; Gershenzon, J. Biology and biochemistry of glucosinolates. Annu. Rev. Plant Biol. 2006, 57, 303–333. [Google Scholar] [CrossRef] [PubMed]
- Jaafaru, M.S.; Nordin, N.; Shaari, K.; Rosli, R.; Razis, A.F.A. Isothiocyanate from Moringa oleifera seeds mitigates hydrogen peroxide-induced cytotoxicity and preserved morphological features of human neuronal cells. PLoS ONE 2018, 13, e0196403. [Google Scholar] [CrossRef] [PubMed]
- Giacoppo, S.; Galuppo, M.; Montaut, S.; Iori, R.; Rollin, P.; Bramanti, P.; Mazzon, E. An overview on neuroprotective effects of isothiocyanates for the treatment of neurodegenerative diseases. Fitoterapia 2015, 106, 12–21. [Google Scholar] [CrossRef] [PubMed]
- Dinkova-Kostova, A.T.; Kostov, R.V. Glucosinolates and isothiocyanates in health and disease. Trends Mol. Med. 2012, 18, 337–347. [Google Scholar] [CrossRef] [PubMed]
- Abuajah, C.I.; Ogbonna, A.C.; Osuji, C.M. Functional components and medicinal properties of food: A review. Asian Pac. J. Cancer Prev. 2015, 52, 2522–2529. [Google Scholar] [CrossRef] [PubMed]
- Conaway, C.; Yang, Y.M.; Chung, F.L. Isothiocyanates as cancer chemopreventive agents: Their biological activities and metabolism in rodents and humans. Curr. Drug Metab. 2002, 3, 233–255. [Google Scholar] [CrossRef] [PubMed]
- Bianchini, F.; Vainio, H. Isothiocyanates in cancer prevention. Drug Metab. Rev. 2004, 36, 655–667. [Google Scholar] [CrossRef] [PubMed]
- Navarro, S.L.; Li, F.; Lampe, J.W. Mechanisms of action of isothiocyanates in cancer chemoprevention: An update. Food Funct. 2011, 2, 579–587. [Google Scholar] [CrossRef] [PubMed]
- Gerhauser, C. Epigenetic impact of dietary isothiocyanates in cancer chemoprevention. Curr. Opin. Clin. Nutr. Metab. Care 2013, 16, 405–410. [Google Scholar] [CrossRef] [PubMed]
- Gupta, P.; Kim, B.; Kim, S.H.; Srivastava, S.K. Molecular targets of isothiocyanates in cancer: Recent advances. Mol. Nutr. Food Res. 2014, 58, 1685–1707. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; D’Souza, S.S.; Tickoo, S.; Salimath, B.P.; Singh, H.B. Antiangiogenic and proapoptotic activities of allyl isothiocyanate inhibit ascites tumor growth in vivo. Integr. Cancer Ther. 2009, 8, 75–87. [Google Scholar] [CrossRef] [PubMed]
- Galuppo, M.; Giacoppo, S.; De Nicola, G.R.; Iori, R.; Navarra, M.; Lombardo, G.E.; Bramanti, P.; Mazzon, E. Antiinflammatory activity of glucomoringin isothiocyanate in a mouse model of experimental autoimmune encephalomyelitis. Fitoterapia 2014, 95, 160–174. [Google Scholar] [CrossRef] [PubMed]
- Galuppo, M.; Nicola, G.R.D.; Iori, R.; Dell’Utri, P.; Bramanti, P.; Mazzon, E. Antibacterial activity of glucomoringin bioactivated with myrosinase against two important pathogens affecting the health of long-term patients in hospitals. Molecules 2013, 18, 14340–14348. [Google Scholar] [CrossRef] [PubMed]
- Brunelli, D.; Tavecchio, M.; Falcioni, C. The isothiocyanate produced from glucomoringin inhibits NF-kB and reduces myeloma growth in nude mice in vivo. Biochem. Pharmacol. 2010, 79, 1141–1148. [Google Scholar] [CrossRef] [PubMed]
- Rajan, T.S.; De Nicola, G.R.; Iori, R.; Rollin, P.; Bramanti, P.; Mazzon, E. Anticancer activity of glucomoringin isothiocyanate in human malignant astrocytoma cells. Fitoterapia 2016, 110, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Clarke, J.D.; Dashwood, R.H.; Ho, E. Multi-targeted prevention of cancer by sulforaphane. Cancer Lett. 2008, 269, 291–304. [Google Scholar] [CrossRef] [PubMed]
- Cheung, K.L.; Kong, A.N. Molecular targets of dietary phenethyl isothiocyanate and sulforaphane for cancer chemoprevention. AAPS J. 2010, 12, 87–97. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zhang, T.; Korkaya, H.; Liu, S.; Lee, H.F.; Newman, B.; Yu, Y.; Clouthier, S.G.; Schwartz, S.J.; Wicha, M.S.; et al. Sulforaphane, a dietary component of broccoli/broccoli sprouts, inhibits breast cancer stem cells. Clin. Cancer Res. 2010, 16, 2580–2590. [Google Scholar] [CrossRef] [PubMed]
- Loeb, S.; Bruinsma, S.M.; Nicholson, J.; Briganti, A.; Pickles, T.; Kakehi, Y.; Carlsson, S.V.; Roobol, M.J. Active surveillance for prostate cancer: A systematic review of clinicopathologic variables and biomarkers for risk stratification. Eur. Urol. 2015, 67, 619–626. [Google Scholar] [CrossRef] [PubMed]
- Klotz, L. Active surveillance for prostate cancer: Debate over the application, not the concept. Eur. Urol. 2015, 67, 1006–1008. [Google Scholar] [CrossRef] [PubMed]
- Jung, I.L. Soluble extract from Moringa oleifera leaves with a new anticancer activity. PLoS ONE 2014, 9, e95492. [Google Scholar] [CrossRef] [PubMed]
- Vongsak, B.; Sithisarn, P.; Mangmool, S.; Thongpraditchote, S.; Wongkrajang, Y.; Gritsanapan, W. Maximizing total phenolics, total flavonoids contents and antioxidant activity of Moringa oleifera leaf extract by the appropriate extraction method. Ind. Crops Prod. 2013, 44, 566–571. [Google Scholar] [CrossRef]
- Devi, J.R.; Thangam, E.B. Mechanisms of anticancer activity of sulforaphane from Brassica oleracea in HEp-2 human epithelial carcinoma cell line. Asian Pac. J. Cancer Prev. 2012, 13, 2095–2100. [Google Scholar] [CrossRef] [PubMed]
- Satria, D.; Silalahi, J.; Haro, G.; Ilyas, S.; Hsb, P.A. Antioxidant and Antiproliferative Activities of an Ethylacetate Fraction of Picria Fel-Terrae Lour. Herbs. Asian Pac. J. Cancer Prev. 2017, 18, 399. [Google Scholar] [PubMed]
- Popova, I.E.; Morra, M.J. Sinigrin and sinalbin quantification in mustard seed using high performance liquid chromatography–time-of-flight mass spectrometry. J. Food Compos. Anal. 2014, 35, 120–126. [Google Scholar] [CrossRef]
- De Graaf, R.M.; Krosse, S.; Swolfs, A.E.; te Brinke, E.; Prill, N.; Leimu, R.; van Galen, P.M.; Wang, Y.; Aarts, M.G.; Van Dam, N.M. Isolation and identification of 4-α-rhamnosyloxy benzyl glucosinolate in Noccaea caerulescens showing intraspecific variation. Phytochemistry 2015, 110, 166–171. [Google Scholar] [CrossRef] [PubMed]
- Jaafaru, M.S.; Kyomson, I.D.; Waziri, P.M.; Yakubu, Y.; Mustapha, M.B.; Gyutorwa, J.S. In vivo ameliorative effect of methanolic extract of Boswellia dalzielli Hutch (Mebdh) stem bark on Triton X-100 induced hyperlipidaemia. Sci. World J. 2017, 12, 34–37. [Google Scholar]
- Lui, V.W.; Wentzel, A.L.; Xiao, D.; Lew, K.L.; Singh, S.V.; Grandis, J.R. Requirement of a carbon spacer in benzyl isothiocyanate-mediated cytotoxicity and MAPK activation in head and neck squamous cell carcinoma. Carcinogenesis 2003, 24, 1705–1712. [Google Scholar] [CrossRef] [PubMed]
- Tang, L.; Zhang, Y. Mitochondria are the primary target in isothiocyanate-induced apoptosis in human bladder cancer cells. Mol. Cancer Ther. 2005, 4, 1250–1259. [Google Scholar] [CrossRef] [PubMed]
- Cavell, B.E.; Syed Alwi, S.S.; Donlevy, A.M.; Proud, C.G.; Packham, G. Natural product-derived antitumor compound phenethyl isothiocyanate inhibits mTORC1 activity via TSC2. J. Nat. Prod. 2012, 75, 1051–1057. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.H.; Kim, D.W.; Lee, H.C.; Lee, J.H.; Lee, T.H. Phenethyl isothiocyanate sensitizes glioma cells to TRAIL-induced apoptosis. Biochem. Biophys. Res. Commun. 2014, 446, 815–821. [Google Scholar] [CrossRef] [PubMed]
- Datta, D.; Aftabuddin, M.; Gupta, D.K.; Raha, S.; Sen, P. Human prostate cancer hallmarks map. Sci. Rep. 2016, 6, 30691. [Google Scholar] [CrossRef] [PubMed]
- Gold-Smith, F.; Fernandez, A.; Bishop, K. Mangiferin and cancer: Mechanisms of action. Nutrients 2016, 8, 396. [Google Scholar] [CrossRef] [PubMed]
- Daniyal, M.; Siddiqui, Z.A.; Akram, M.; Asif, H.M.; Sultana, S.; Khan, A. Epidemiology, etiology, diagnosis and treatment of prostate cancer. Asian Pac. J. Cancer Prev. 2014, 15, 9575–9578. [Google Scholar] [CrossRef] [PubMed]
- Ochiai, S.; Nomoto, Y.; Kobayashi, S.; Yamashita, Y.; Watanabe, Y.; Toyomasu, Y.; Kawamura, T.; Takada, A.; Ii, N.; Sakuma, H. Multimodality Treatment for Patients with Node-Positive Prostate Cancer: The Role of Radiation Therapy. Asian Pac. J. Cancer Prev. 2016, 17, 1625–1630. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Yu, Q.Y.; Liu, Y.; Zhu, Z.L.; Huang, Y.W.; Li, K. Efficacy and Safety of Laser Surgery and Transurethral Resection of the Prostate for Treating Benign Prostate Hyperplasia: A Network Meta-analysis. Asian Pac. J. Cancer Prev. 2016, 17, 4281–4288. [Google Scholar] [PubMed]
- Waziri, P.M.; Abdullah, R.; Rosli, R.; Omar, A.R.; Abdul, A.B.; Kassim, N.K.; Malami, I.; Etti, I.C.; Sani, J.M.; Mohd Lila, M.A.; et al. Clausenidin Induces Caspase 8-Dependent Apoptosis and Suppresses Production of VEGF in Liver Cancer Cells. Asian Pac. J. Cancer Prev. 2018, 19, 917–922. [Google Scholar] [PubMed]
- Khalafalla, M.M.; Abdellatef, E.; Dafalla, H.M.; Nassrallah, A.A.; Aboul-Enein, K.M.; Lightfoot, D.A.; El-Deeb, F.E.; El-Shemy, H.A. Active principle from Moringa oleifera Lam leaves effective against two leukemias and a hepatocarcinoma. Afr. J. Biotechnol. 2010, 9, 8467–8471. [Google Scholar]
- Sreelatha, S.; Jeyachitra, A.; Padma, P.R. Antiproliferation and induction of apoptosis by Moringa oleifera leaf extract on human cancer cells. Food Chem. Toxicol. 2011, 49, 1270–1275. [Google Scholar] [CrossRef] [PubMed]
- Berkovich, L.; Earon, G.; Ron, I. Moringa Oleifera aqueous leaf extract down-regulates nuclear factor-kappaB and increases cytotoxic effect of chemotherapy in pancreatic cancer cells. BMC Complement. Altern. Med. 2013, 13, 212. [Google Scholar] [CrossRef] [PubMed]
- William, F.; Lakshminarayanan, S.; Chegu, H. Effect of some Indian vegetables on the glucose and insulin response in diabetic subjects. Int. J. Food Sci. Nutr. 1993, 44, 191–195. [Google Scholar] [CrossRef]
- Kumari, D.J. Hypoglycaemic effect of Moringa oleifera and Azadirachta indica in type 2 diabetes mellitus. Bioscan 2010, 5, 211–214. [Google Scholar]
- Asiedu-Gyekye, I.J.; Frimpong-Manso, S.A.M.U.E.L.; Awortwe, C.; Antwi, D.A.; Nyarko, A.K. Micro-and macroelemental composition and safety evaluation of the nutraceutical Moringa oleifera leaves. J. Toxicol. 2014, 2014, 786979. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, S.K.; Singh, S.V. Cell cycle arrest, apoptosis induction and inhibition of nuclear factor kappa B activation in anti-proliferative activity of benzyl isothiocyanate against human pancreatic cancer cells. Carcinogenesis 2004, 25, 1701–1709. [Google Scholar] [CrossRef] [PubMed]
- Waterman, C.; Cheng, D.M.; Rojas-Silva, P.; Poulev, A.; Dreifus, J.; Lila, M.A.; Raskin, I. Stable, water extractable isothiocyanates from Moringa oleifera leaves attenuate inflammation in vitro. Phytochemistry 2014, 103, 114–122. [Google Scholar] [CrossRef] [PubMed]
- Kou, X.; Li, B.; Olayanju, J.B.; Drake, J.M.; Chen, N. Nutraceutical or Pharmacological Potential of Moringa oleifera Lam. Nutrients 2018, 10, 343. [Google Scholar] [CrossRef] [PubMed]
- Guon, T.E.; Chung, H.S. Moringa oleifera fruit induce apoptosis via reactive oxygen species-dependent activation of mitogen-activated protein kinases in human melanoma A2058 cells. Oncol. Lett. 2017, 14, 1703–1710. [Google Scholar] [CrossRef] [PubMed]
- Leelawat, S.; Leelawat, K. Molecular mechanisms of cholangiocarcinoma cell inhibition by medicinal plants. Oncol. Lett. 2017, 13, 961–966. [Google Scholar] [CrossRef] [PubMed]
- Shang, H.S.; Shih, Y.L.; Lu, T.J.; Lee, C.H.; Hsueh, S.C.; Chou, Y.C.; Lu, H.F.; Liao, N.C.; Chung, J.G. Benzyl isothiocyanate (BITC) induces apoptosis of GBM 8401 human brain glioblastoma multiforms cells via activation of caspase-8/Bid and the reactive oxygen species-dependent mitochondrial pathway. Environ. Toxicol. 2016, 31, 1751–1760. [Google Scholar] [CrossRef] [PubMed]
- Hait, W.N. Anticancer drug development: The grand challenges. Nat. Rev. Drug Discov. 2016, 9, 253–254. [Google Scholar] [CrossRef] [PubMed]
- Begley, C.G.; Ellis, L.M. Drug development: Raise standards for preclinical cancer research. Nature 2012, 483, 531–533. [Google Scholar] [CrossRef] [PubMed]
- Abdull Razis, A.F.; De Nicola, G.R.; Pagnotta, E.; Iori, R.; Ioannides, C. A glucosinolate-rich extract of Japanese Daikon perturbs carcinogen-metabolizing enzyme systems in rat, being a potent inducer of hepatic glutathione S-transferase. Eur. J. Nutr. 2013, 52, 1279–1285. [Google Scholar] [CrossRef] [PubMed]

| Reaction | Score | Severity |
|---|---|---|
| Fur and skin changes | A | 0 |
| Eyes changes | B | 0 |
| Mucous membrane | C | 0 |
| Pale | D | 0 |
| Congested | a | 0 |
| Motor activity | E | 0 |
| Increased motor activity-speed of movement increases | a | 0 |
| Decreased motor activity—lethargy, does not respond to external stimuli | b | 0 |
| Tremor (continuous repetitive twitching of skeletal muscles, usually palpable and visible) | F | 0 |
| Convulsion (Involuntary contraction of the voluntary muscle) | G | 0 |
| Tonic convulsion—sustained spasm with head arched backward | a | 0 |
| Clonic convulsion—short choppy spasm with head arched toward stomach | b | 0 |
| Mixed convulsions—combination of tonic and clonic convulsions | c | 0 |
| Walking backwards and/or ataxia, inability to coordinate bodily movements (gross wobbling) | H | 0 |
| Diarrhea | I | 0 |
| Death—self-explanatory | J | 0 |
| Others | K | 0 |
| Severity | Score |
|---|---|
| Normal/No changes | 0 |
| Mild | 1 |
| Moderate | 2 |
| Severe | 3 |
| Individual Moieties | NMR Chemical Shifts de Graaf et al. [45] | NMR Chemical Shifts (Present Study) | ||
|---|---|---|---|---|
| δC | δH (J in Hz) | δC | δH (J in Hz) | |
| Glucose Moiety | ||||
| 1′ | 80.9 | 4.57, d, (9.0) | 80.4 | 4.57, d |
| 2′ | 71.9 | 3.17–3.30, m | 71.8 | 3.17–3.36, m |
| 3′ | 77.0 | 3.17–3.30, m | 76.9 | 3.17–3.36, m |
| 4′ | 68.8 | 3.17–3.30, m | 68.6 | 3.17–3.36, m |
| 5′ | 79.7 | 3.09, m | 79.7 | 3.09, m |
| 6′ | 60.2 | 3.50, m | 60.2 | 3.49, m |
| Benzyl Moiety | ||||
| 1 | 154.4 | — | 154.5 | — |
| 2 | 117.4 | 7.00, d, (8.7) | 117.5 | 7.00, d (8.7) |
| 3 | 129.3 | 7.18, d, (8.7) | 129.4 | 7.21, d, (8.7) |
| 4 | 130.4 | — | 130.7 | — |
| 5 | 129.3 | 7.18, d, (8.7) | 129.4 | 7.21, d (8.7) |
| 6 | 117.4 | 7.00, d, (8.7) | 117.5 | 7.00, d, (8.7) |
| 7 | 37.2 | 3.86, d, (17.0) | 37.4 | 3.86, d, (17.0) |
| Others | ||||
| 0 | 154.6 | 154.5 | ||
| Rhamnose Moiety | ||||
| 1″ | 98.1 | 5.41, d, (1.7) | 98.0 | 5.41, d, (1.7) |
| 2″ | 69.9 | 4.03, d, (1.7–3.5) | 69.6 | 4.02, d (1.7–3.5) |
| 3″ | 70.0 | 3.86, d, (3.5, 7.0) | 69.9 | 3.86, d, (3.5–7.0) |
| 4″ | 70.2 | 3.38, t, (9.7) | 70.4 | 3.36, t, (9.7) |
| 5″ | 69.3 | 3.67, m | 69.3 | 3.65, m |
| 6″ | 16.6 | 1.09, d, (6.3) | 16.6 | 1.08, d (63) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jaafaru, M.S.; Abd Karim, N.A.; Mohamed Eliaser, E.; Maitalata Waziri, P.; Ahmed, H.; Mustapha Barau, M.; Kong, L.; Abdull Razis, A.F. Nontoxic Glucomoringin-Isothiocyanate (GMG-ITC) Rich Soluble Extract Induces Apoptosis and Inhibits Proliferation of Human Prostate Adenocarcinoma Cells (PC-3). Nutrients 2018, 10, 1174. https://doi.org/10.3390/nu10091174
Jaafaru MS, Abd Karim NA, Mohamed Eliaser E, Maitalata Waziri P, Ahmed H, Mustapha Barau M, Kong L, Abdull Razis AF. Nontoxic Glucomoringin-Isothiocyanate (GMG-ITC) Rich Soluble Extract Induces Apoptosis and Inhibits Proliferation of Human Prostate Adenocarcinoma Cells (PC-3). Nutrients. 2018; 10(9):1174. https://doi.org/10.3390/nu10091174
Chicago/Turabian StyleJaafaru, Mohammed Sani, Nurul Ashikin Abd Karim, Enas Mohamed Eliaser, Peter Maitalata Waziri, Hamidu Ahmed, Mohammed Mustapha Barau, Liliya Kong, and Ahmad Faizal Abdull Razis. 2018. "Nontoxic Glucomoringin-Isothiocyanate (GMG-ITC) Rich Soluble Extract Induces Apoptosis and Inhibits Proliferation of Human Prostate Adenocarcinoma Cells (PC-3)" Nutrients 10, no. 9: 1174. https://doi.org/10.3390/nu10091174
APA StyleJaafaru, M. S., Abd Karim, N. A., Mohamed Eliaser, E., Maitalata Waziri, P., Ahmed, H., Mustapha Barau, M., Kong, L., & Abdull Razis, A. F. (2018). Nontoxic Glucomoringin-Isothiocyanate (GMG-ITC) Rich Soluble Extract Induces Apoptosis and Inhibits Proliferation of Human Prostate Adenocarcinoma Cells (PC-3). Nutrients, 10(9), 1174. https://doi.org/10.3390/nu10091174








