Randomized Trial of a High Protein, Partial Meal Replacement Program with or without Alternate Day Fasting: Similar Effects on Weight Loss, Retention Status, Nutritional, Metabolic, and Behavioral Outcomes
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Study Design
2.3. Dietary Intervention Protocol
2.4. Study Outcomes
2.5. Statistical Analysis
3. Results
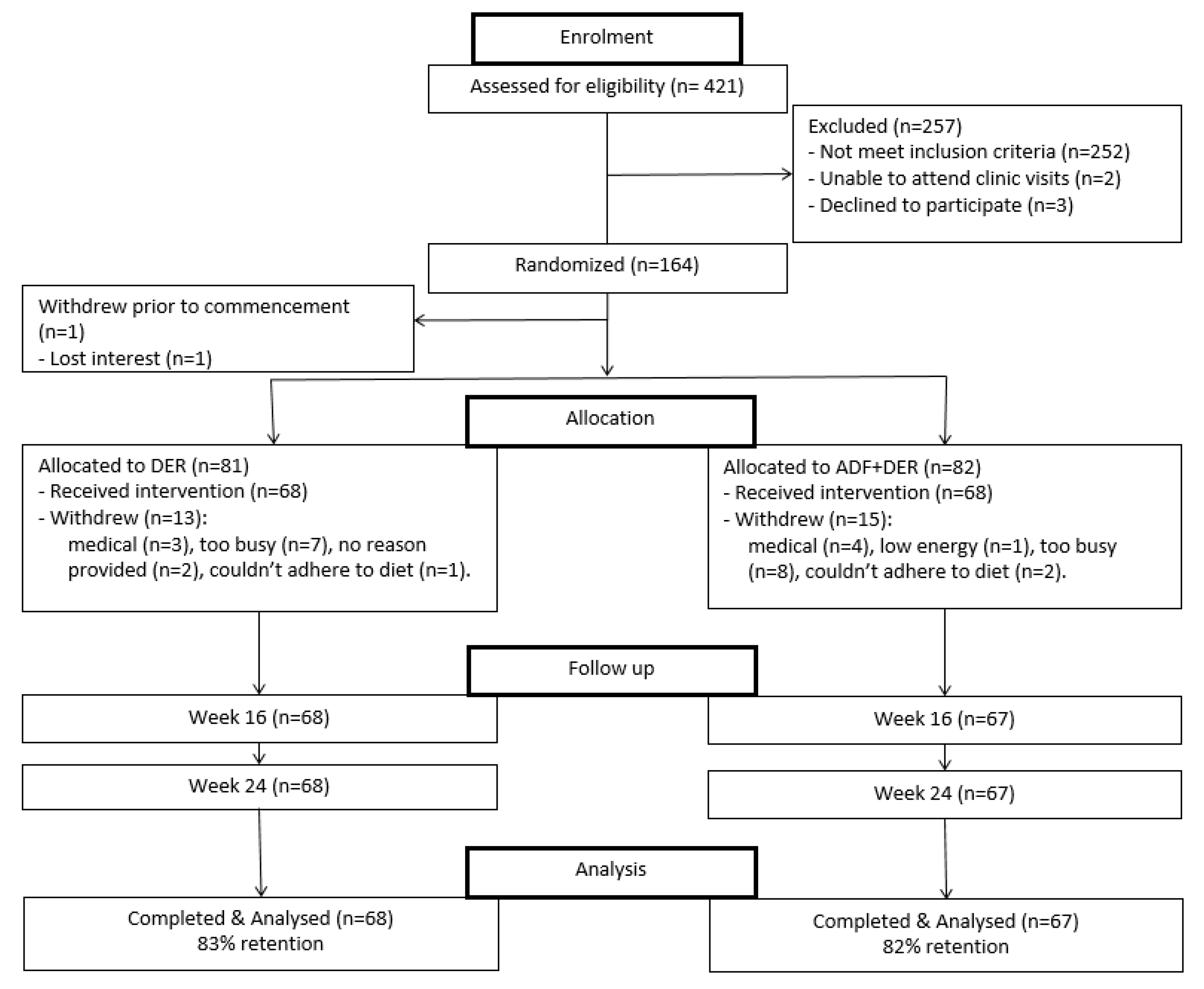
3.1. Participants and Retention
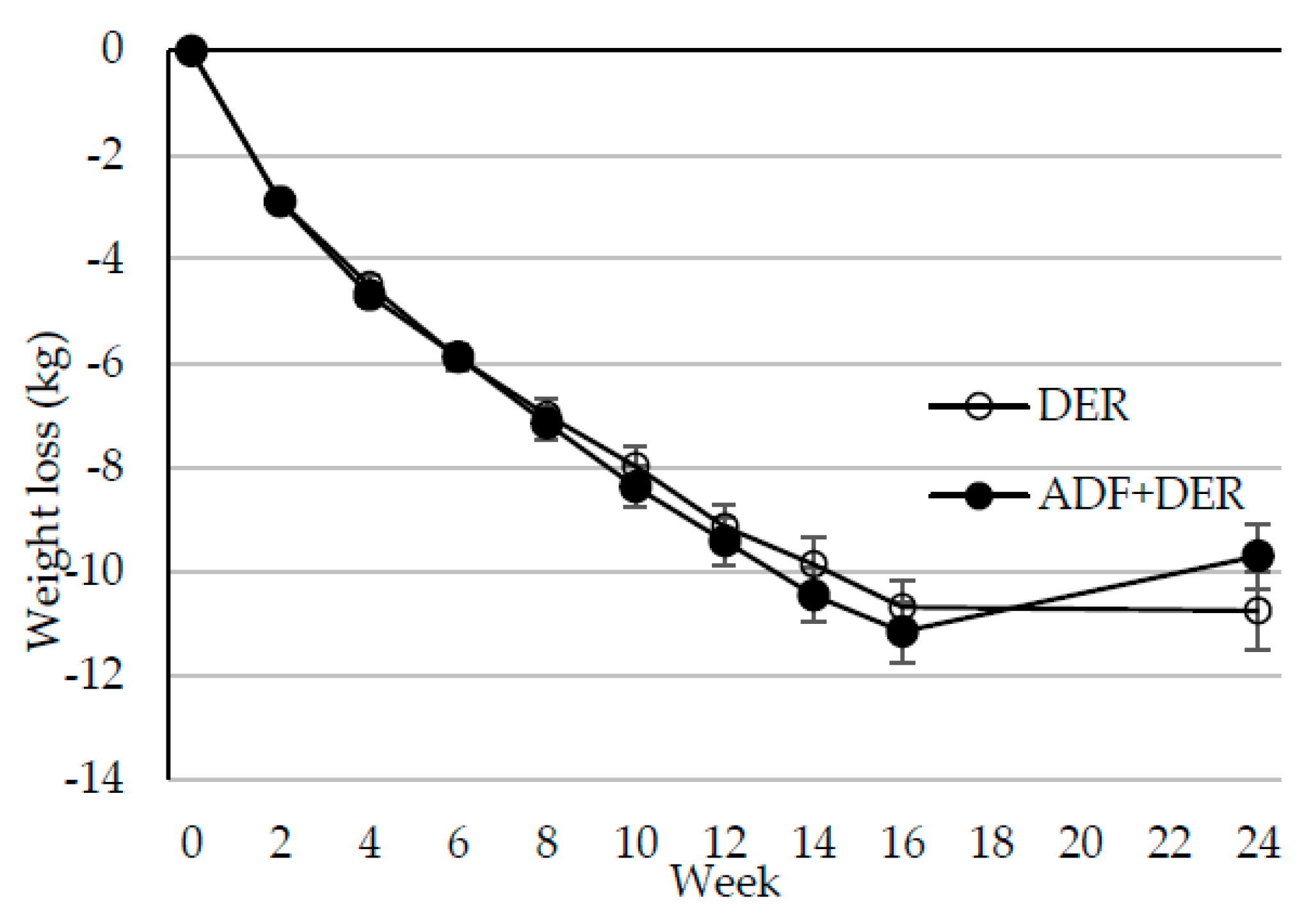
3.2. Body Weight, Composition, and Metabolic Markers
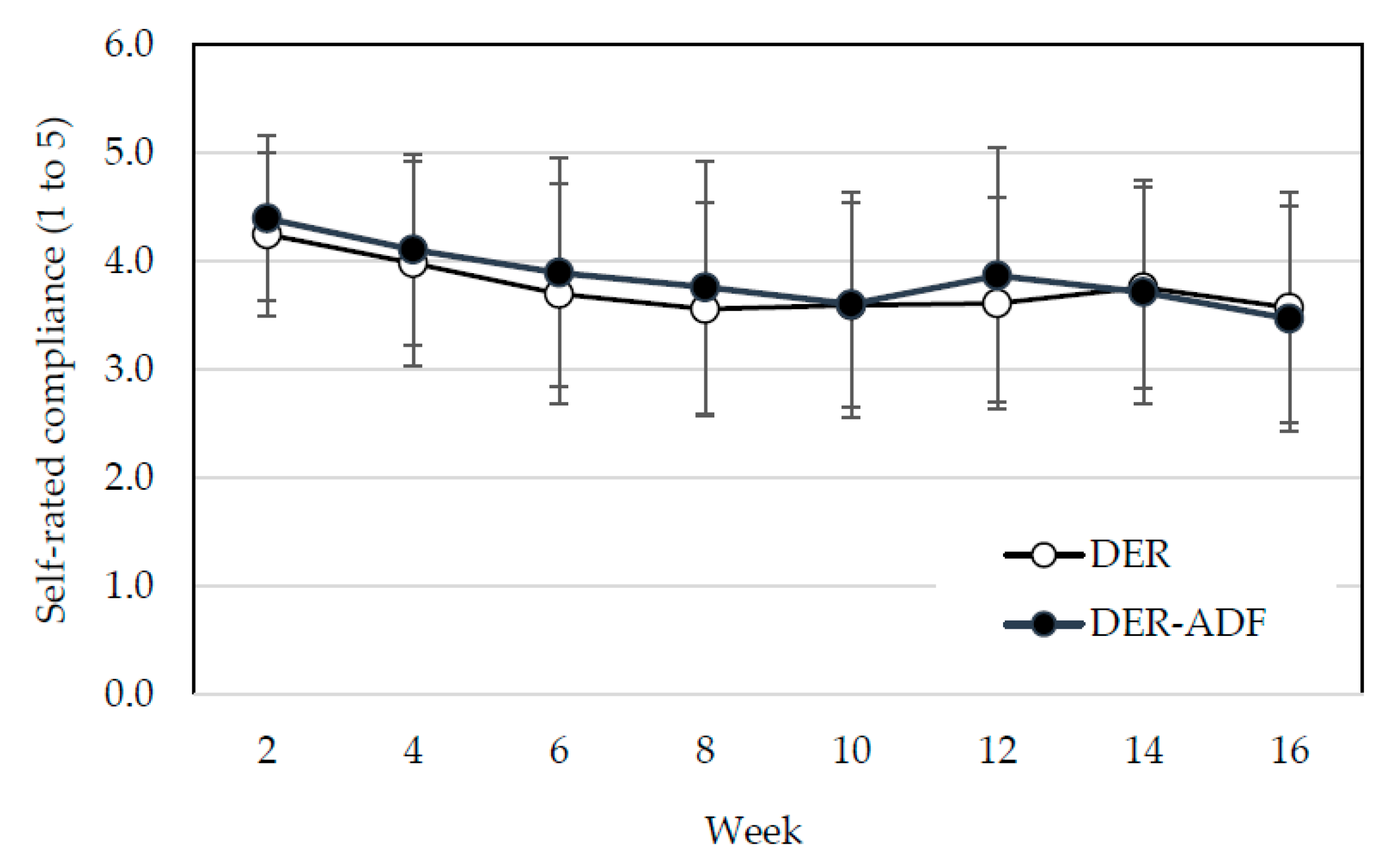
3.3. Nutritional Markers and Reported Compliance with Dietary Protocol
3.4. Behavioral Measures
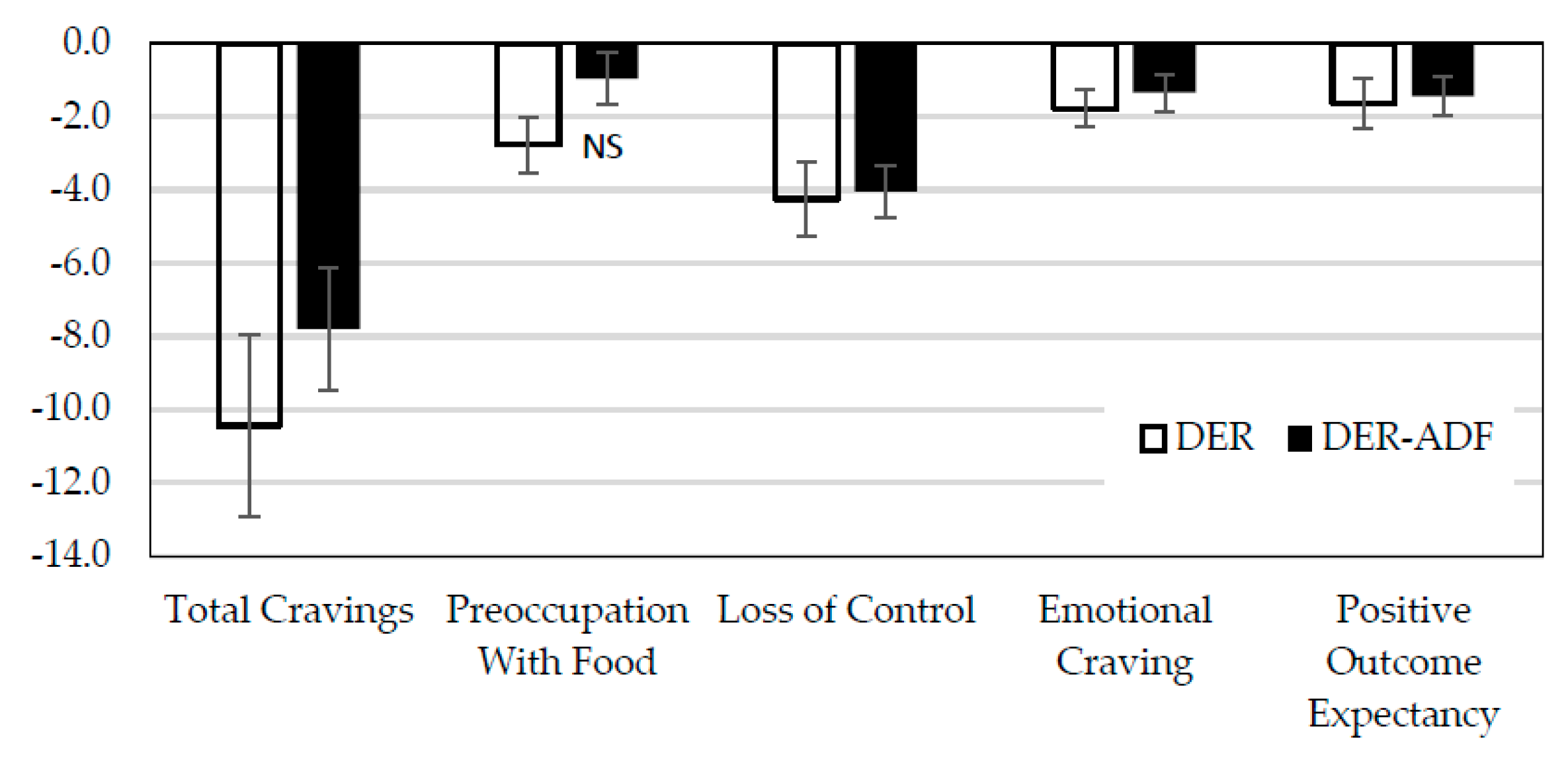
3.4.1. Food Cravings
3.4.2. Changes in Health-Related Quality of Life
3.4.3. Mood
4. Discussion
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Moroshko, I.; Brennan, L.; O’Brien, P. Predictors of dropout in weight loss interventions: A systematic review of the literature. Obes. Rev. 2011, 12, 912–934. [Google Scholar] [CrossRef] [PubMed]
- Burgess, E.; Hassmén, P.; Pumpa, K.L. Determinants of adherence to lifestyle intervention in adults with obesity: A systematic review. Clin. Obes. 2017, 7, 123–135. [Google Scholar] [CrossRef] [PubMed]
- Aller, E.E.J.G.; Larsen, T.M.; Claus, H.; Lindroos, A.K.; Kafatos, A.; Pfeiffer, A.; Martinez, J.A.; Handjieva-Darlenska, T.; Kunesova, M.; Stender, S.; et al. Weight loss maintenance in overweight subjects on ad libitum diets with high or low protein content and glycemic index: The DIOGENES trial 12 months results. Int. J. Obes. 2014, 38, 1–33. [Google Scholar] [CrossRef] [PubMed]
- Johansson, K.; Neovius, M.; Hemmingsson, E. Effects of anti-obesity drugs, diet, and exercise on weight-loss maintenance after a very-low-calorie diet or low-calorie diet: A systematic review and meta-analysis of randomized controlled trials. Am. J. Clin. Nutr. 2014, 99, 14–23. [Google Scholar] [CrossRef] [PubMed]
- Unick, J.L.; Neiberg, R.H.; Hogan, P.E.; Cheskin, L.J.; Dutton, G.R.; Jeffery, R.; Nelson, J.A.; Pi-Sunyer, X.; West, D.S.; Wing, R.R.; et al. Weight change in the first 2 months of a lifestyle intervention predicts weight changes 8 years later. Obesity 2015, 23, 1353–1356. [Google Scholar] [CrossRef] [PubMed]
- Leidy, H.J.; Clifton, P.M.; Astrup, A.; Wycherley, T.P.; Westerterp-Plantenga, M.S.; Luscombe-Marsh, N.D.; Woods, S.C.; Mattes, R.D. The role of protein in weight loss and maintenance. Am. J. Clin. Nutr. 2015, 101, 1320S–1329S. [Google Scholar] [CrossRef] [PubMed]
- Wycherley, T.P.; Moran, L.J.; Clifton, P.M.; Noakes, M.; Brinkworth, G.D. Effects of energy-restricted high-protein, low-fat compared with standard-protein, low-fat diets: A meta-analysis of randomized controlled trials. Am. J. Clin. Nutr. 2012, 96, 1281–1298. [Google Scholar] [CrossRef] [PubMed]
- Noakes, M.; Foster, P.; Keogh, J.; Clifton, P. Meal replacements are as effective as structured weight-loss diets for treating obesity in adults with features of metabolic syndrome. J. Nutr. 2004, 134, 1894–1899. [Google Scholar] [CrossRef] [PubMed]
- Brindal, E.; Hendrie, G.A.; Taylor, P.; Freyne, J.; Noakes, M. Cohort analysis of a 24-week randomized controlled trial to assess the efficacy of a novel, partial meal replacement program targetingweight loss and risk factor reduction in overweight/obese adults. Nutrients 2016, 8, 265. [Google Scholar] [CrossRef] [PubMed]
- Harris, L.; McGarty, A.; Hutchison, L.; Ells, L.; Hankey, C. Short-term intermittent energy restriction interventions for weight management: A systematic review and meta-analysis. Obes. Rev. 2017, 19, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Seimon, R.V.; Roekenes, J.A.; Zibellini, J.; Zhu, B.; Gibson, A.A.; Hills, A.P.; Wood, R.E.; King, N.A.; Byrne, N.M.; Sainsbury, A. Do intermittent diets provide physiological benefits over continuous diets for weight loss? A systematic review of clinical trials. Mol. Cell. Endocrinol. 2015, 418, 153–172. [Google Scholar] [CrossRef] [PubMed]
- Harris, L.; Hamilton, S.; Azevedo, L.B.; Olajide, J.; De Brún, C.; Waller, G.; Whittaker, V.; Sharp, T.; Lean, M.; Hankey, C.; et al. Intermittent fasting interventions for treatment of overweight and obesity in adults: A systematic review and meta-analysis. JBI Database Syst. Rev. Implement. Rep. 2018, 16, 507–547. [Google Scholar] [CrossRef] [PubMed]
- Varady, K.A. Intermittent versus daily calorie restriction: Which diet regimen is more effective for weight loss? Obes. Rev. 2011, 12, e597–e601. [Google Scholar] [CrossRef] [PubMed]
- Kroeger, C.M.; Trepanowski, J.F.; Klempel, M.C.; Barnosky, A.; Bhutani, S.; Gabel, K.; Varady, K.A. Eating behavior traits of successful weight losers during 12 months of alternate-day fasting: An exploratory analysis of a randomized controlled trial. Nutr. Health 2018, 24, 5–10. [Google Scholar] [CrossRef] [PubMed]
- Morgan, J.F.; Reid, F.; Lacey, J.H. The SCOFF questionnaire: Assessment of a new screening tool for eating disorders. Bmj 1999, 319, 1467–1468. [Google Scholar] [CrossRef] [PubMed]
- Schofield, W. Predicting basal metabolic rate, new standards and review of previous work. Hum. Nutr. Clin. Nutr. 1895, 39, 5–41. [Google Scholar]
- The Live Well Plan, Meat and Livestock Australia 2013. Available online: https://www.dropbox.com/s/fhwmd5d7q8x7z4b/Live%20Well%20Plan_FINAL%20Brochure_27%203%2013.pdf (accessed on 22 August 2018).
- Friedewald, W.T.; Levy, R.I.; Fredrickson, D.S. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin. Chem. 1972, 18, 499–502. [Google Scholar] [PubMed]
- Nijs, I.M.T.; Franken, I.H.A.; Muris, P. The modified Trait and State Food-Cravings Questionnaires: Development and validation of a general index of food craving. Appetite 2007, 49, 38–46. [Google Scholar] [CrossRef] [PubMed]
- Health-Specific Self-Efficacy Scales. Available online: https://userpage.fu-berlin.de/health/healself.pdf (accessed on 22 August 2018).
- Watson, D.; Clark, L.A.; Tellegen, A. Development and validation of brief measures of positive and negative affect: The PANAS scales. J. Pers. Soc. Psychol. 1988, 54, 1063–1070. [Google Scholar] [CrossRef] [PubMed]
- Hays, R.D.; Morales, L.S. The RAND-36 measure of health-related quality of life. Ann. Med. 2001, 33, 350–357. [Google Scholar] [CrossRef] [PubMed]
- Neiberg, R.H.; Wing, R.R.; Bray, G.A.; Reboussin, D.M.; Rickman, A.D.; Johnson, K.C.; Kitabchi, A.E.; Faulconbridge, L.F.; Kitzman, D.W.; Espeland, M.A.; et al. Patterns of weight change associated with long-term weight change and cardiovascular disease risk factors in the look AHEAD study. Obesity 2012, 20, 2048–2056. [Google Scholar] [CrossRef] [PubMed]
- Wing, R.R.; Espeland, M.A.; Clark, J.M.; Hazuda, H.P.; Knowler, W.C.; Pownall, H.J.; Unick, J.; Wadden, T.; Wagenknecht, L.; the Action for Health in Diabetes (Look AHEAD) Study Group. Association of weight loss maintenance and weight regain on 4-year changes in CVD risk factors: The action for health in diabetes (Look AHEAD) clinical trial. Diabetes Care 2016, 39, 1345–1355. [Google Scholar] [CrossRef] [PubMed]
- Clayton, D.J.; Creese, M.; Skidmore, N.; Stensel, D.J.; James, L.J. No Effect of 24 h severe energy restriction on appetite regulation and ad libitum energy intake in overweight and obese males. Int. J. Obes. 2016, 40, 1–31. [Google Scholar] [CrossRef] [PubMed]
- Alhussain, M.H.; Macdonald, I.A.; Taylor, M.A. Irregular meal-pattern effects on energy expenditure, metabolism, and appetite regulation: A randomized controlled trial in healthy normal-weight women. Am. J. Clin. Nutr. 2016, 104, 21–32. [Google Scholar] [CrossRef] [PubMed]
- Westerterp, K.R. Diet induced thermogenesis. Nutr. Metab. 2004. [Google Scholar] [CrossRef] [PubMed]
- Raben, A.; Tagliabue, A.; Christensen, N.J.; Madsen, J.; Holst, J.J.; Astrup, A. Meals with similar energy densities but rich in protein, fat, effects on energy expenditure not on appetite and energy intake. Am. J. Clin. Nutr. 1994, 60, 544–551. [Google Scholar] [CrossRef] [PubMed]
- Tentolouris, N.; Pavlatos, S.; Kokkinos, A.; Perrea, D.; Pagoni, S.; Katsilambros, N. Diet-induced thermogenesis and substrate oxidation are not different between lean and obese women after two different isocaloric meals, one rich in protein and one rich in fat. Metabolism 2008, 57, 313–320. [Google Scholar] [CrossRef] [PubMed]
- Johnston, C.S.; Day, C.S.; Swan, P.D. Postprandial Thermogenesis Is Increased 100% on a High-Protein, Low-Fat Diet versus a High-Carbohydrate, Low-Fat Diet in Healthy, Young Women. J. Am. Coll. Nutr. 2002, 21, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Catenacci, V.A.; Pan, Z.; Ostendorf, D.; Brannon, S.; Gozansky, W.S.; Mattson, M.P.; Martin, B.; MacLean, P.S.; Melanson, E.L.; Troy Donahoo, W. A randomized pilot study comparing zero-calorie alternate-day fasting to daily caloric restriction in adults with obesity. Obesity 2016, 24, 1874–1883. [Google Scholar] [CrossRef] [PubMed]
- Hoddy, K.K.; Kroeger, C.M.; Trepanowski, J.F.; Barnosky, A.; Bhutani, S.; Varady, K.A. Meal timing during alternate day fasting: Impact on body weight and cardiovascular disease risk in obese adults. Obesity 2014, 22, 2524–2531. [Google Scholar] [CrossRef] [PubMed]
- Alhamdan, B.A.; Garcia-Alvarez, A.; Alzahrnai, A.H.; Karanxha, J.; Stretchberry, D.R.; Contrera, K.J.; Utria, A.F.; Cheskin, L.J. Alternate-day versus daily energy restriction diets: Which is more effective for weight loss? A systematic review and meta-analysis. Obes. Sci. Pract. 2016, 2, 293–302. [Google Scholar] [CrossRef] [PubMed]
- Headland, M.; Clifton, P.M.; Carter, S.; Keogh, J.B. Weight-loss outcomes: A systematic review and meta-analysis of intermittent energy restriction trials lasting a minimum of 6 months. Nutrients 2016, 8, 354. [Google Scholar] [CrossRef] [PubMed]
- Zuo, L.; He, F.; Tinsley, G.M.; Pannell, B.K.; Ward, E.; Arciero, P.J. Comparison of high-protein, intermittent fasting low-calorie diet and heart healthy diet for vascular health of the obese. Front. Physiol. 2016. [Google Scholar] [CrossRef] [PubMed]
- Raynor, H.A.; Anderson, A.M.; Miller, G.D.; Reeves, R.; Delahanty, L.M.; Vitolins, M.Z.; Harper, P.; Mobley, C.; Konersman, K.; Mayer-Davis, E. Partial Meal Replacement Plan and Quality of the Diet at 1 Year: Action for Health in Diabetes (Look AHEAD) Trial. J. Acad. Nutr. Diet. 2015, 115, 731–742. [Google Scholar] [CrossRef] [PubMed]
- Keogh, J.B.; Cleanthous, X.; Wycherley, T.P.; Brinkworth, G.D.; Noakes, M.; Clifton, P.M. Increased thiamine intake may be required to maintain thiamine status during weight loss in patients with type 2 diabetes. Diabetes Res. Clin. Pract. 2012, 98, e40–e42. [Google Scholar] [CrossRef] [PubMed]
- Carrodeguas, L.; Kaidar-Person, O.; Szomstein, S.; Antozzi, P.; Rosenthal, R. Preoperative thiamine deficiency in obese population undergoing laparoscopic bariatric surgery. Surg. Obes. Relat. Dis. 2005, 1, 517–522. [Google Scholar] [CrossRef] [PubMed]
- Nath, A.; Tran, T.; Shope, T.R.; Koch, T.R. Prevalence of clinical thiamine deficiency in individuals with medically complicated obesity. Nutr. Res. 2017, 37, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Kahathuduwa, C.N.; Binks, M.; Martin, C.K.; Dawson, J.A. Extended calorie restriction suppresses overall and specific food cravings: A systematic review and a meta-analysis. Obes. Rev. 2017, 18, 1122–1135. [Google Scholar] [CrossRef] [PubMed]
- Martin, C.K.; O’Neil, P.M.; Pawlow, L. Changes in food cravings during low-calorie and very-low-calorie diets. Obesity 2006, 14, 115–121. [Google Scholar] [CrossRef] [PubMed]
- McHorney, C.A.; Ware, J.E.; Raczek, A.E. The MOS 36-item short-form health survey (Sf-36): II. Psychometric and clinical tests of validity in measuring physical and mental health constructs. Med. Care 1993. [Google Scholar] [CrossRef]
- Maciejewski, M.L.; Patrick, D.L.; Williamson, D.F. A structured review of randomized controlled trials of weight loss showed little improvement in health-related quality of life. J. Clin. Epidemiol. 2005, 58, 568–578. [Google Scholar] [CrossRef] [PubMed]
- Kolotkin, R.L.; Andersen, J.R. A systematic review of reviews: Exploring the relationship between obesity, weight loss and health-related quality of life. Clin. Obes. 2017. [Google Scholar] [CrossRef] [PubMed]


| DER | ADF + DER | |||
|---|---|---|---|---|
| Mon-Sun | M,W,F | Tu,Th,Su | Sa | |
| Breakfast | MR b | MR b | MR b | ad libitum |
| Snack c | 1 snack | 1 snack | - | |
| Lunch | MR b | MR b | MR b | |
| Snack c | 1 snack | 1 snack | - | |
| Dinner | 200 g lean protein + low energy vegetables + 2 teaspoons oil | 200 g lean protein + low energy vegetables + 2 teaspoons oil | Low energy vegetables | |
| Energy (kJ) | 5000 | 5000 | 2400 | 10,000 (estimate) |
| Protein (g) | 102 | 102 | 55 | NA |
| DER (n = 81) | ADF + DER (n = 82) | |
|---|---|---|
| Age (years) | 40.6 ± 8.8 | 40.0 ± 8.3 |
| Sex, Female n (%) | 65 (80) | 67 (82) |
| Male n (%) | 16 (20) | 15 (18) |
| Body weight (kg) | 99.6 ± 15.6 | 100.6 ± 19.6 |
| BMI (kg/m2) | 35.5 ± 5.5 | 35.7 ± 5.8 |
| DER | ADF + DER | p | |||
|---|---|---|---|---|---|
| Baseline (n = 81) | Week 16 (n = 68) | Baseline (n = 82) | Week 16 (n = 67) | ||
| Body weight 2, kg | 99.6 ± 15.6 | 87.1 ± 14.0 | 100.6 ± 19.6 | 90.6 ± 18.7 | 0.803 |
| BMI 2, kg/m2 | 35.5 ± 5.5 | 31.1 ± 5.1 | 35.7 ± 5.8 | 31.9 ± 5.5 | 0.913 |
| Total FFM 2, kg | 50.7 ± 7.9 | 48.8 ± 7.6 | 51.1 ± 9.1 | 49.7 ± 9.2 | 0.842 |
| Total FM 2, kg | 45.1 ± 11.2 | 34.8 ± 10.2 | 45.7 ± 13.1 | 37.3 ± 12.8 | 0.674 |
| TC 2, mmol/L | 5.3 ± 1.0 | 4.7 ± 0.9 | 5.2 ± 1.0 | 4.7 ± 1.0 | 0.754 |
| Triglycerides 2, mmol/L | 1.5 ± 0.8 | 1.1 ± 0.5 | 1.4 ± 0.6 | 1.2 ± 0.6 | 0.029 3 |
| HDL-C 4, mmol/L | 1.3 ± 0.3 | 1.3 ±0.3 | 1.3 ± 0.3 | 1.2 ± 0.2 | 0.154 |
| LDL-C 2, mmol/L | 3.3 ± 0.9 | 2.9 ± 0.7 | 3.3 ± 0.9 | 2.9 ± 0.8 | 0.987 |
| hsCRP 2, 5, mg/L | 3.2 ± 2.6 | 2.9 ± 2.2 | 3.7 ± 2.5 | 3.0 ± 2.2 | 0.450 |
| Glucose 2, mmol/L | 5.5 ± 0.6 | 5.3 ± 0.4 | 5.4 ± 0.4 | 5.3 ± 0.4 | 0.341 |
| Insulin 2, mU/L | 12.4 ± 7.3 | 8.01 ± 4.2 | 13.4 ± 8.4 | 9.1 ± 4.5 | 0.334 |
| SBP 2, mm Hg | 120.6 ± 12.8 | 112.2 ± 9.7 | 119.4 ±12.9 | 112.2 ± 9.9 | 0.893 |
| DBP 2, mm Hg | 74.7 ± 9.0 | 71.1 ± 8.7 | 75.6 ± 9.4 | 71.5 ± 7.8 | 0.394 |
| DER | ADF + DER | p | |||
|---|---|---|---|---|---|
| Baseline (n = 81) | Week 16 (n = 68) | Baseline (n = 82) | Week 16 (n = 67) | ||
| Iron, umol/L | 16.2 ± 5.6 | 16.8 ± 5.5 | 15.1 ± 5.7 | 15.6 ± 5.5 | 0.921 |
| Ferritin 3, µg/L | 83.7 ± 66.9 | 90.8 ± 72.6 | 92.6 ± 96.5 | 114.6 ± 101.5 | 0.332 |
| Transferrin 2, g/L | 2.85 ± 0.4 | 2.7 ± 0.4 | 2.8 ± 0.4 | 2.7 ± 0.4 | 0.507 |
| Transferrin Saturation, % | 23.0 ± 8.8 | 25.1 ± 8.9 | 21.9 ± 8.7 | 23.7 ± 9.0 | 0.109 |
| Serum Zinc 4, µmol/L | 12.6 ± 1.5 | 13.0 ± 1.6 | 12.5 ± 1.5 | 12.8 ± 1.6 | 0.887 |
| Folate 2, nmol/L packed red cells | 1375.7 ± 290.2 | 1543.0 ± 318.8 | 1344.7 ± 274.7 | 1537 ± 286.8 | 0.533 |
| Serum Vitamin B12 2, pmol/L | 315.2 ± 118.7 | 384.2 ± 145.1 | 287.8 ± 82.5 | 352.0 ± 101.4 | 0.664 |
| Erythrocyte Thiamine Pyrophosphate 2,*, nmol/L | 134.0 ± 22.6 | 121.9 ± 22.5 | 127.2 ± 20.7 | 114.1 ± 21.7 | 0.824 |
| 25 Hydroxy-Vitamin D 3, nmol/L | 73.6 ± 20.4 | 71.7 ± 18.7 | 70.7 ± 17.4 | 70.7 ± 17.4 | 0.075 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bowen, J.; Brindal, E.; James-Martin, G.; Noakes, M. Randomized Trial of a High Protein, Partial Meal Replacement Program with or without Alternate Day Fasting: Similar Effects on Weight Loss, Retention Status, Nutritional, Metabolic, and Behavioral Outcomes. Nutrients 2018, 10, 1145. https://doi.org/10.3390/nu10091145
Bowen J, Brindal E, James-Martin G, Noakes M. Randomized Trial of a High Protein, Partial Meal Replacement Program with or without Alternate Day Fasting: Similar Effects on Weight Loss, Retention Status, Nutritional, Metabolic, and Behavioral Outcomes. Nutrients. 2018; 10(9):1145. https://doi.org/10.3390/nu10091145
Chicago/Turabian StyleBowen, Jane, Emily Brindal, Genevieve James-Martin, and Manny Noakes. 2018. "Randomized Trial of a High Protein, Partial Meal Replacement Program with or without Alternate Day Fasting: Similar Effects on Weight Loss, Retention Status, Nutritional, Metabolic, and Behavioral Outcomes" Nutrients 10, no. 9: 1145. https://doi.org/10.3390/nu10091145
APA StyleBowen, J., Brindal, E., James-Martin, G., & Noakes, M. (2018). Randomized Trial of a High Protein, Partial Meal Replacement Program with or without Alternate Day Fasting: Similar Effects on Weight Loss, Retention Status, Nutritional, Metabolic, and Behavioral Outcomes. Nutrients, 10(9), 1145. https://doi.org/10.3390/nu10091145




