Modulation of Intestinal Immune and Barrier Functions by Vitamin A: Implications for Current Understanding of Malnutrition and Enteric Infections in Children
Abstract
1. Introduction
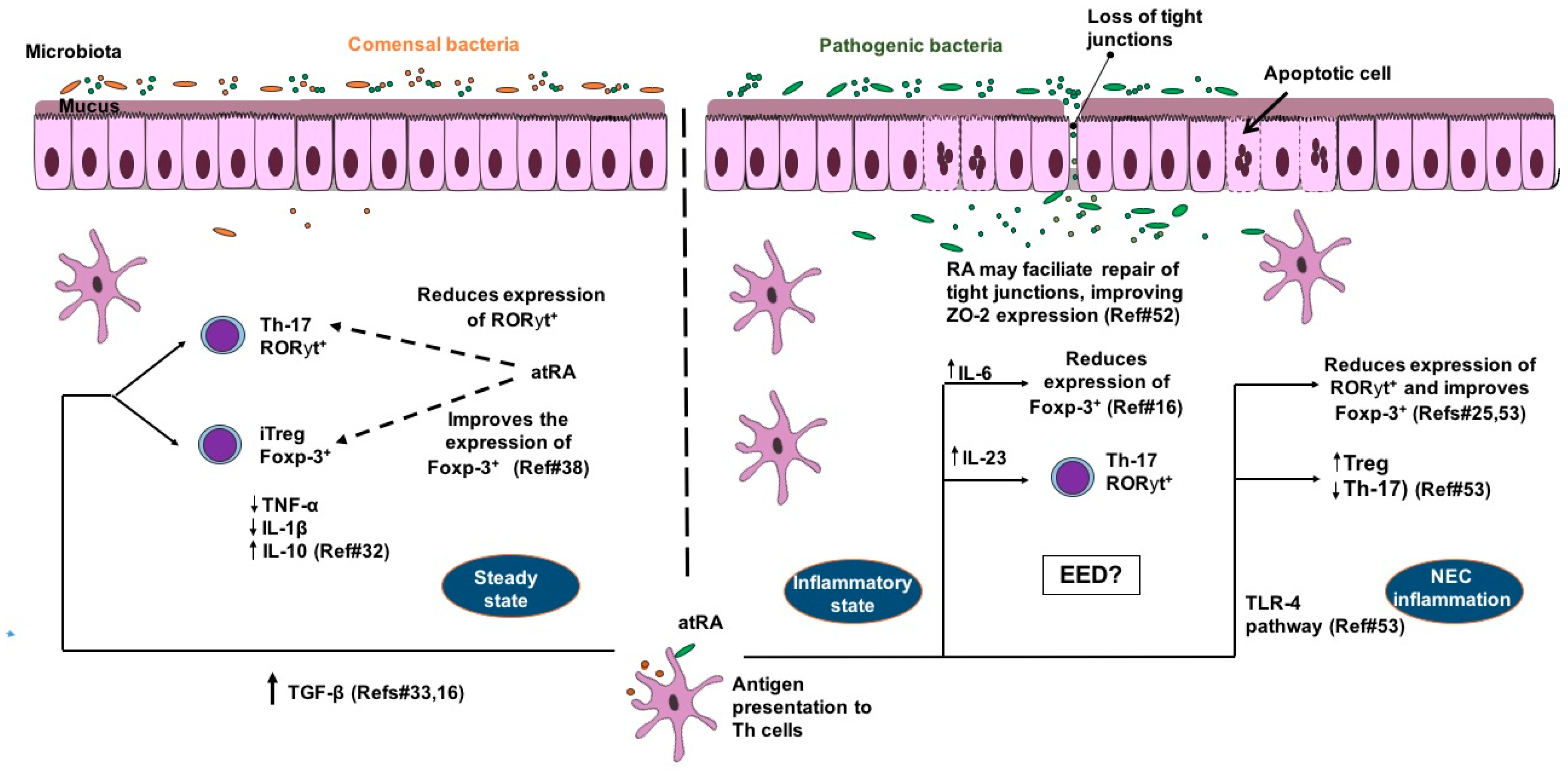
2. Cellular and Molecular Mechanisms of Vitamin A in the Gut: Crosstalk of Immune and Inflammatory Responses
3. Interactions with Intestinal Epithelial Barrier, Enteric Pathogens and Microbiota
4. Clinical Evidence of Vitamin A for Enteric Diseases in Children: Adjusting Classical Paradigms and Facing New Challenges
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Gutierrez-Mazariegos, J.; Theodosiou, M.; Campo-Paysaa, F.; Schubert, M. Vitamin A: A multifunctional tool for development. Semin. Cell. Dev. Biol. 2011, 22, 603–610. [Google Scholar] [CrossRef] [PubMed]
- Kanungo, J. Retinoic Acid Signaling in P19 Stem Cell Differentiation. Anticancer Agents Med. Chem. 2017, 17, 1184–1198. [Google Scholar] [CrossRef] [PubMed]
- Sommer, A. Preventing blindness and saving lives: The centenary of vitamin A. JAMA Ophthalmol. 2014, 132, 115–117. [Google Scholar] [CrossRef] [PubMed]
- Paganelli, A.; Gnazzo, V.; Acosta, H.; Lopez, S.L.; Carrasco, A.E. Glyphosate-based herbicides produce teratogenic effects on vertebrates by impairing retinoic acid signaling. Chem. Res. Toxicol. 2010, 23, 1586–1595. [Google Scholar] [CrossRef] [PubMed]
- Robinson, J.F.; Verhoef, A.; Pennings, J.L.; Pronk, T.E.; Piersma, A.H. A comparison of gene expression responses in rat whole embryo culture and in vivo: Time-dependent retinoic acid-induced teratogenic response. Toxicol. Sci. 2012, 126, 242–254. [Google Scholar] [CrossRef] [PubMed]
- Green, H.N.; Mellanby, E. Vitamin A as an anti-infective agent. Br. Med. J. 1928, 2, 691–696. [Google Scholar] [CrossRef] [PubMed]
- Long, K.Z.; Santos, J.I.; Rosado, J.L.; Estrada-Garcia, T.; Haas, M.; Al, M.A.; DuPont, H.L.; Nanthakumar, N.N. Vitamin A supplementation modifies the association between mucosal innate and adaptive immune responses and resolution of enteric pathogen infections. Am. J. Clin. Nutr. 2011, 93, 578–585. [Google Scholar] [CrossRef] [PubMed]
- Mitter, S.S.; Oria, R.B.; Kvalsund, M.P.; Pamplona, P.; Joventino, E.S.; Mota, R.M.; Goncalves, D.C.; Patrick, P.D.; Guerrant, R.L.; Lima, A.A. Apolipoprotein E4 influences growth and cognitive responses to micronutrient supplementation in shantytown children from northeast Brazil. Clinics (Sao Paulo) 2012, 67, 11–18. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Vitamin and Mineral Requirements in Human Nutrition, 2nd ed.; WHO: Geneva, Switzerland, 2005. [Google Scholar]
- World Health Organization (WHO). Global Prevalence of Vitamin A Deficiency in Populations at Risk 1995–2005; WHO Global Database on Vitamin A Deficiency; WHO: Geneva, Switzerland, 2009. [Google Scholar]
- Raverdeau, M.; Mills, K.H. Modulation of T cell and innate immune responses by retinoic Acid. J. Immunol. 2014, 192, 2953–2958. [Google Scholar] [CrossRef] [PubMed]
- Amengual, J.; Zhang, N.; Kemerer, M.; Maeda, T.; Palczewski, K.; Von, L.J. STRA6 is critical for cellular vitamin A uptake and homeostasis. Hum. Mol. Genet. 2014, 23, 5402–5417. [Google Scholar] [CrossRef] [PubMed]
- Blomhoff, R.; Blomhoff, H.K. Overview of retinoid metabolism and function. J. Neurobiol. 2006, 66, 606–630. [Google Scholar] [CrossRef] [PubMed]
- Rubin, L.P.; Ross, A.C.; Stephensen, C.B.; Bohn, T.; Tanumihardjo, S.A. Metabolic Effects of Inflammation on Vitamin A and Carotenoids in Humans and Animal Models. Adv. Nutr. 2017, 8, 197–212. [Google Scholar] [CrossRef] [PubMed]
- Mowat, A.M.; Agace, W.W. Regional specialization within the intestinal immune system. Nat. Rev. Immunol. 2014, 14, 667–685. [Google Scholar] [CrossRef] [PubMed]
- Omenetti, S.; Pizarro, T.T. The Treg/Th17 axis: A dynamic balance regulated by the gut microbiome. Front. Immunol. 2015, 6, 639. [Google Scholar] [CrossRef] [PubMed]
- Scott, C.L.; Aumeunier, A.M.; Mowat, A.M. Intestinal CD103+ dendritic cells: Master regulators of tolerance? Trends Immunol. 2011, 32, 412–419. [Google Scholar] [CrossRef] [PubMed]
- Iwata, M.; Hirakiyama, A.; Eshima, Y.; Kagechika, H.; Kato, C.; Song, S.Y. Retinoic acid imprints gut-homing specificity on T cells. Immunity 2004, 21, 527–538. [Google Scholar] [CrossRef] [PubMed]
- Mwanza-Lisulo, M.; Chomba, M.S.; Chama, M.; Besa, E.C.; Funjika, E.; Zyambo, K.; Banda, R.; Imikendu, M.; Sianongo, S.; Hancock, R.E.W.; et al. Retinoic acid elicits a coordinated expression of gut homing markers on T lymphocytes of Zambian men receiving oral Vivotif, but not Rotarix, Dukoral or OPVERO vaccines. Vaccine 2018, 36, 4134–4141. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.M.; Hall, J.A.; Blank, R.B.; Bouladoux, N.; Oukka, M.; Mora, J.R.; Belkaid, Y. Small intestine lamina propria dendritic cells promote de novo generation of Foxp3 T reg cells via retinoic acid. J. Exp. Med. 2007, 204, 1775–1785. [Google Scholar] [CrossRef] [PubMed]
- Bakdash, G.; Vogelpoel, L.T.; van Capel, T.M.; Kapsenberg, M.L.; de Jong, E.C. Retinoic acid primes human dendritic cells to induce gut-homing, IL-10-producing regulatory T cells. Mucosal Immunol. 2015, 8, 265–278. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, K.; Petri, W.A., Jr. Environmental enteropathy: Elusive but significant subclinical abnormalities in developing countries. EBioMedicine 2016, 10, 25–32. [Google Scholar] [CrossRef] [PubMed]
- Guerrant, R.L.; Leite, A.M.; Pinkerton, R.; Medeiros, P.H.; Cavalcante, P.A.; DeBoer, M.; Kosek, M.; Duggan, C.; Gewirtz, A.; Kagan, J.C.; et al. Biomarkers of environmental enteropathy, inflammation, stunting, and impaired growth in children in northeast Brazil. PLoS ONE 2016, 11, e0158772. [Google Scholar] [CrossRef] [PubMed]
- Lu, L.; Lan, Q.; Li, Z.; Zhou, X.; Gu, J.; Li, Q.; Wang, J.; Chen, M.; Liu, Y.; Shen, Y.; et al. Critical role of all-trans retinoic acid in stabilizing human natural regulatory T cells under inflammatory conditions. Proc. Natl. Acad. Sci. USA 2014, 111, E3432–E3440. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Kong, N.; Wang, J.; Fan, H.; Zou, H.; Horwitz, D.; Brand, D.; Liu, Z.; Zheng, S.G. Cutting edge: All-trans retinoic acid sustains the stability and function of natural regulatory T cells in an inflammatory milieu. J. Immunol. 2010, 185, 2675–2679. [Google Scholar] [CrossRef] [PubMed]
- Ruemmele, F.M.; Garnier-Lengline, H. Transforming growth factor and intestinal inflammation: The role of nutrition. Nestle. Nutr. Inst. Workshop Ser. 2013, 77, 91–98. [Google Scholar] [PubMed]
- Cha, H.R.; Chang, S.Y.; Chang, J.H.; Kim, J.O.; Yang, J.Y.; Kim, C.H.; Kweon, M.N. Downregulation of Th17 cells in the small intestine by disruption of gut flora in the absence of retinoic acid. J. Immunol. 2010, 184, 6799–6806. [Google Scholar] [CrossRef] [PubMed]
- Elias, K.M.; Laurence, A.; Davidson, T.S.; Stephens, G.; Kanno, Y.; Shevach, E.M.; O’Shea, J.J. Retinoic acid inhibits Th17 polarization and enhances FoxP3 expression through a Stat-3/Stat-5 independent signaling pathway. Blood 2008, 111, 1013–1020. [Google Scholar] [CrossRef] [PubMed]
- Duester, G. Families of retinoid dehydrogenases regulating vitamin A function: Production of visual pigment and retinoic acid. Eur. J. Biochem. 2000, 267, 4315–4324. [Google Scholar] [CrossRef] [PubMed]
- Sandell, L.L.; Sanderson, B.W.; Moiseyev, G.; Johnson, T.; Mushegian, A.; Young, K.; Rey, J.P.; Ma, J.X.; Staehling-Hampton, K.; Trainor, P.A. RDH10 is essential for synthesis of embryonic retinoic acid and is required for limb, craniofacial, and organ development. Genes Dev. 2007, 21, 1113–1124. [Google Scholar] [CrossRef] [PubMed]
- Vicente-Suarez, I.; Larange, A.; Reardon, C.; Matho, M.; Feau, S.; Chodaczek, G.; Park, Y.; Obata, Y.; Gold, R.; Wang-Zhu, Y.; et al. Unique lamina propria stromal cells imprint the functional phenotype of mucosal dendritic cells. Mucosal Immunol. 2015, 8, 141–151. [Google Scholar] [CrossRef] [PubMed]
- Czarnewski, P.; Das, S.; Parigi, S.M.; Villablanca, E.J. Retinoic acid and its role in modulating intestinal innate immunity. Nutrients 2017, 9. [Google Scholar] [CrossRef] [PubMed]
- Coombes, J.L.; Siddiqui, K.R.; Arancibia-Carcamo, C.V.; Hall, J.; Sun, C.M.; Belkaid, Y.; Powrie, F. A functionally specialized population of mucosal CD103+ DCs induces Foxp3+ regulatory T cells via a TGF-beta and retinoic acid-dependent mechanism. J. Exp. Med. 2007, 204, 1757–1764. [Google Scholar] [CrossRef] [PubMed]
- Macpherson, A.J.; Slack, E. The functional interactions of commensal bacteria with intestinal secretory IgA. Curr. Opin. Gastroenterol. 2007, 23, 673–678. [Google Scholar] [CrossRef] [PubMed]
- Mora, J.R.; Iwata, M.; Eksteen, B.; Song, S.Y.; Junt, T.; Senman, B.; Otipoby, K.L.; Yokota, A.; Takeuchi, H.; Ricciardi-Castagnoli, P.; et al. Generation of gut-homing IgA-secreting B cells by intestinal dendritic cells. Science 2006, 314, 1157–1160. [Google Scholar] [CrossRef] [PubMed]
- Bono, M.R.; Tejon, G.; Flores-Santibanez, F.; Fernandez, D.; Rosemblatt, M.; Sauma, D. Retinoic acid as a modulator of T cell immunity. Nutrients 2016, 8. [Google Scholar] [CrossRef] [PubMed]
- Van de Pavert, S.A.; Ferreira, M.; Domingues, R.G.; Ribeiro, H.; Molenaar, R.; Moreira-Santos, L.; Almeida, F.F.; Ibiza, S.; Barbosa, I.; Goverse, G.; et al. Maternal retinoids control type 3 innate lymphoid cells and set the offspring immunity. Nature 2014, 508, 123–127. [Google Scholar] [CrossRef] [PubMed]
- Lu, L.; Ma, J.; Li, Z.; Lan, Q.; Chen, M.; Liu, Y.; Xia, Z.; Wang, J.; Han, Y.; Shi, W.; et al. All-trans retinoic acid promotes TGF-beta-induced Tregs via histone modification but not DNA demethylation on Foxp3 gene locus. PLoS ONE 2011, 6, e24590. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, V.; Pearson, K.; Kim, J.H.; Kamdar, K.; DePaolo, R.W. Retinoic acid can exacerbate T cell intrinsic TLR2 activation to promote tolerance. PLoS ONE 2015, 10, e0118875. [Google Scholar] [CrossRef] [PubMed]
- Reifen, R.; Levy, E.; Berkovich, Z.; Tirosh, O. Vitamin A exerts its antiinflammatory activities in colitis through preservation of mitochondrial activity. Nutrition 2015, 31, 1402–1407. [Google Scholar] [CrossRef] [PubMed]
- Okayasu, I.; Hana, K.; Nemoto, N.; Yoshida, T.; Saegusa, M.; Yokota-Nakatsuma, A.; Song, S.Y.; Iwata, M. Vitamin A inhibits development of dextran sulfate sodium-induced colitis and colon cancer in a mouse model. Biomed. Res. Int. 2016, 2016. [Google Scholar] [CrossRef] [PubMed]
- Penny, H.L.; Prestwood, T.R.; Bhattacharya, N.; Sun, F.; Kenkel, J.A.; Davidson, M.G.; Shen, L.; Zuniga, L.A.; Seeley, E.S.; Pai, R.; et al. Restoring retinoic acid attenuates intestinal inflammation and tumorigenesis in APCMin/+ mice. Cancer Immunol. Res. 2016, 4, 917–926. [Google Scholar] [CrossRef] [PubMed]
- Arts, R.J.; Blok, B.A.; van, C.R.; Joosten, L.A.; Aaby, P.; Benn, C.S.; Netea, M.G. Vitamin A induces inhibitory histone methylation modifications and down-regulates trained immunity in human monocytes. J. Leukoc. Biol. 2015, 98, 129–136. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Cui, T.; Li, Y.; Wang, Y.; Wang, Q.; Li, X.; Bi, Y.; Wei, X.; Liu, L.; Li, T.; et al. Vitamin A supplementation in early life enhances the intestinal immune response of rats with gestational vitamin A deficiency by increasing the number of immune cells. PLoS ONE 2014, 9, e114934. [Google Scholar] [CrossRef] [PubMed]
- Turner, J.R. Intestinal mucosal barrier function in health and disease. Nat. Rev. Immunol. 2009, 9, 799–809. [Google Scholar] [CrossRef] [PubMed]
- Yano, T.; Kanoh, H.; Tamura, A.; Tsukita, S. Apical cytoskeletons and junctional complexes as a combined system in epithelial cell sheets. Ann. N. Y. Acad. Sci. 2017, 1405, 32–43. [Google Scholar] [CrossRef] [PubMed]
- Haines, R.J.; Beard, R.S., Jr.; Eitner, R.A.; Chen, L.; Wu, M.H. TNFalpha/IFNgamma mediated intestinal epithelial barrier dysfunction is attenuated by MicroRNA-93 downregulation of PTK6 in mouse colonic epithelial cells. PLoS ONE 2016, 11, e0154351. [Google Scholar] [CrossRef] [PubMed]
- Mao, J.W.; Tang, H.Y.; Zhao, T.; Tan, X.Y.; Bi, J.; Wang, B.Y.; Wang, Y.D. Intestinal mucosal barrier dysfunction participates in the progress of nonalcoholic fatty liver disease. Int. J. Clin. Exp. Pathol. 2015, 8, 3648–3658. [Google Scholar] [PubMed]
- Maciel, A.A.; Oria, R.B.; Braga-Neto, M.B.; Braga, A.B.; Carvalho, E.B.; Lucena, H.B.; Brito, G.A.; Guerrant, R.L.; Lima, A.A. Role of retinol in protecting epithelial cell damage induced by Clostridium difficile toxin A. Toxicon 2007, 50, 1027–1040. [Google Scholar] [CrossRef] [PubMed]
- Lima, A.A.; Soares, A.M.; Lima, N.L.; Mota, R.M.; Maciel, B.L.; Kvalsund, M.P.; Barrett, L.J.; Fitzgerald, R.P.; Blaner, W.S.; Guerrant, R.L. Effects of vitamin A supplementation on intestinal barrier function, growth, total parasitic, and specific Giardia spp. infections in Brazilian children: A prospective randomized, double-blind, placebo-controlled trial. J. Pediatr Gastroenterol. Nutr. 2010, 50, 309–315. [Google Scholar] [CrossRef] [PubMed]
- Mondal, D.; Minak, J.; Alam, M.; Liu, Y.; Dai, J.; Korpe, P.; Liu, L.; Haque, R.; Petri, W.A., Jr. Contribution of enteric infection, altered intestinal barrier function, and maternal malnutrition to infant malnutrition in Bangladesh. Clin. Infect. Dis. 2012, 54, 185–192. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Gao, Y.; Cui, T.; Yang, T.; Liu, L.; Li, T.; Chen, J. Retinoic acid facilitates Toll-Like Receptor 4 expression to improve intestinal barrier function through retinoic acid receptor Beta. Cell. Physiol. Biochem. 2017, 42, 1390–1406. [Google Scholar] [CrossRef] [PubMed]
- Egan, C.E.; Sodhi, C.P.; Good, M.; Lin, J.; Jia, H.; Yamaguchi, Y.; Lu, P.; Ma, C.; Branca, M.F.; Weyandt, S.; et al. Toll-like receptor 4-mediated lymphocyte influx induces neonatal necrotizing enterocolitis. J. Clin. Investig. 2016, 126, 495–508. [Google Scholar] [CrossRef] [PubMed]
- Nino, D.F.; Sodhi, C.P.; Egan, C.E.; Zhou, Q.; Lin, J.; Lu, P.; Yamaguchi, Y.; Jia, H.; Martin, L.Y.; Good, M.; et al. Retinoic Acid improves incidence and severity of necrotizing enterocolitis by lymphocyte balance restitution and repopulation of LGR5+ intestinal stem cells. Shock 2017, 47, 22–32. [Google Scholar] [CrossRef] [PubMed]
- Crane, R.J.; Jones, K.D.; Berkley, J.A. Environmental enteric dysfunction: An overview. Food Nutr. Bull. 2015, 36, S76–S87. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Mumm, J.B.; Herbst, R.; Kolbeck, R.; Wang, Y. IL-22 Increases permeability of intestinal epithelial tight junctions by enhancing Claudin-2 expression. J. Immunol. 2017, 199, 3316–3325. [Google Scholar] [CrossRef] [PubMed]
- Martin, J.C.; Beriou, G.; Heslan, M.; Chauvin, C.; Utriainen, L.; Aumeunier, A.; Scott, C.L.; Mowat, A.; Cerovic, V.; Houston, S.A.; et al. Interleukin-22 binding protein (IL-22BP) is constitutively expressed by a subset of conventional dendritic cells and is strongly induced by retinoic acid. Mucosal Immunol. 2014, 7, 101–113. [Google Scholar] [CrossRef] [PubMed]
- Mielke, L.A.; Jones, S.A.; Raverdeau, M.; Higgs, R.; Stefanska, A.; Groom, J.R.; Misiak, A.; Dungan, L.S.; Sutton, C.E.; Streubel, G.; et al. Retinoic acid expression associates with enhanced IL-22 production by gammadelta T cells and innate lymphoid cells and attenuation of intestinal inflammation. J. Exp. Med. 2013, 210, 1117–1124. [Google Scholar] [CrossRef] [PubMed]
- Cabrera, G.; Fernandez-Brando, R.J.; Abrey-Recalde, M.J.; Baschkier, A.; Pinto, A.; Goldstein, J.; Zotta, E.; Meiss, R.; Rivas, M.; Palermo, M.S. Retinoid levels influence enterohemorrhagic Escherichia coli infection and Shiga toxin 2 susceptibility in mice. Infect. Immun. 2014, 82, 3948–3957. [Google Scholar] [CrossRef] [PubMed]
- McDaniel, K.L.; Restori, K.H.; Dodds, J.W.; Kennett, M.J.; Ross, A.C.; Cantorna, M.T. Vitamin A-deficient hosts become nonsymptomatic reservoirs of Escherichia coli-like enteric infections. Infect. Immun. 2015, 83, 2984–2991. [Google Scholar] [CrossRef] [PubMed]
- Chattha, K.S.; Vlasova, A.N.; Kandasamy, S.; Esseili, M.A.; Siegismund, C.; Rajashekara, G.; Saif, L.J. Probiotics and colostrum/milk differentially affect neonatal humoral immune responses to oral rotavirus vaccine. Vaccine 2013, 31, 1916–1923. [Google Scholar] [CrossRef] [PubMed]
- Vlasova, A.N.; Chattha, K.S.; Kandasamy, S.; Siegismund, C.S.; Saif, L.J. Prenatally acquired vitamin A deficiency alters innate immune responses to human rotavirus in a gnotobiotic pig model. J. Immunol. 2013, 190, 4742–4753. [Google Scholar] [CrossRef] [PubMed]
- Guerra-Perez, N.; Frank, I.; Veglia, F.; Aravantinou, M.; Goode, D.; Blanchard, J.L.; Gettie, A.; Robbiani, M.; Martinelli, E. Retinoic acid imprints a mucosal-like phenotype on dendritic cells with an increased ability to fuel HIV-1 infection. J. Immunol. 2015, 194, 2415–2423. [Google Scholar] [CrossRef] [PubMed]
- Derebe, M.G.; Zlatkov, C.M.; Gattu, S.; Ruhn, K.A.; Vaishnava, S.; Diehl, G.E.; MacMillan, J.B.; Williams, N.S.; Hooper, L.V. Serum amyloid A is a retinol binding protein that transports retinol during bacterial infection. eLife 2014, 3, e03206. [Google Scholar] [CrossRef] [PubMed]
- Esterhazy, D.; Mucida, D. Serum amyloid A proteins take retinol for a ride. Trends Immunol. 2014, 35, 505–506. [Google Scholar] [CrossRef] [PubMed]
- Hibberd, M.C.; Wu, M.; Rodionov, D.A.; Li, X.; Cheng, J.; Griffin, N.W.; Barratt, M.J.; Giannone, R.J.; Hettich, R.L.; Osterman, A.L.; et al. The effects of micronutrient deficiencies on bacterial species from the human gut microbiota. Sci. Transl. Med. 2017, 9. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Ko, G. Antiviral effect of vitamin A on norovirus infection via modulation of the gut microbiome. Sci. Rep. 2016, 6, 25835. [Google Scholar] [CrossRef] [PubMed]
- Xiao, S.; Li, Q.; Hu, K.; He, Y.; Ai, Q.; Hu, L.; Yu, J. Vitamin A and retinoic acid exhibit protective effects on necrotizing enterocolitis by regulating intestinal flora and enhancing the intestinal epithelial barrier. Arch. Med. Res. 2018. [Google Scholar] [CrossRef] [PubMed]
- Tielsch, J.M.; Rahmathullah, L.; Thulasiraj, R.D.; Katz, J.; Coles, C.; Sheeladevi, S.; John, R.; Prakash, K. Newborn vitamin A dosing reduces the case fatality but not incidence of common childhood morbidities in South India. J. Nutr. 2007, 137, 2470–2474. [Google Scholar] [CrossRef] [PubMed]
- Black, R.E.; Victora, C.G.; Walker, S.P.; Bhutta, Z.A.; Christian, P.; de, O.M.; Ezzati, M.; Grantham-McGregor, S.; Katz, J.; Martorell, R.; et al. Maternal and child undernutrition and overweight in low-income and middle-income countries. Lancet 2013, 382, 427–451. [Google Scholar] [CrossRef]
- Tanumihardjo, S.A.; Russell, R.M.; Stephensen, C.B.; Gannon, B.M.; Craft, N.E.; Haskell, M.J.; Lietz, G.; Schulze, K.; Raiten, D.J. Biomarkers of nutrition for development (BOND)-vitamin A review. J. Nutr. 2016, 146, 1816S–1848S. [Google Scholar] [CrossRef] [PubMed]
- Imdad, A.; Mayo-Wilson, E.; Herzer, K.; Bhutta, Z.A. Vitamin A supplementation for preventing morbidity and mortality in children from six months to five years of age. Cochrane Database Syst. Rev. 2017, 3, CD008524. [Google Scholar] [CrossRef] [PubMed]
- Benn, C.S.; Aaby, P.; Arts, R.J.; Jensen, K.J.; Netea, M.G.; Fisker, A.B. An enigma: Why vitamin A supplementation does not always reduce mortality even though vitamin A deficiency is associated with increased mortality. Int. J. Epidemiol. 2015, 44, 906–918. [Google Scholar] [CrossRef] [PubMed]
- Stevens, G.A.; Bennett, J.E.; Hennocq, Q.; Lu, Y.; De-Regil, L.M.; Rogers, L.; Danaei, G.; Li, G.; White, R.A.; Flaxman, S.R.; et al. Trends and mortality effects of vitamin A deficiency in children in 138 low-income and middle-income countries between 1991 and 2013: A pooled analysis of population-based surveys. Lancet Glob. Health 2015, 3, e528–e536. [Google Scholar] [CrossRef]
- Mason, J.B.; Benn, C.S.; Sachdev, H.; West, K.P., Jr.; Palmer, A.C.; Sommer, A. Should universal distribution of high dose vitamin A to children cease? BMJ 2018, 360, k927. [Google Scholar] [CrossRef] [PubMed]
- GBD Diarrhoeal Diseases Collaborators. Estimates of global, regional, and national morbidity, mortality, and aetiologies of diarrhoeal diseases: A systematic analysis for the Global Burden of Disease Study 2015. Lancet Infect. Dis. 2017, 17, 909–948. [Google Scholar] [CrossRef]
- Mason, J.; Greiner, T.; Shrimpton, R.; Sanders, D.; Yukich, J. Vitamin A policies need rethinking. Int. J. Epidemiol. 2015, 44, 283–292. [Google Scholar] [CrossRef] [PubMed]
- Fisker, A.B.; Bale, C.; Rodrigues, A.; Balde, I.; Fernandes, M.; Jorgensen, M.J.; Danneskiold-Samsoe, N.; Hornshoj, L.; Rasmussen, J.; Christensen, E.D.; et al. High-dose vitamin A with vaccination after 6 months of age: A randomized trial. Pediatrics 2014, 134, e739–e748. [Google Scholar] [CrossRef] [PubMed]
- Sommer, A.; Tarwotjo, I.; Djunaedi, E.; West, K.P., Jr.; Loeden, A.A.; Tilden, R.; Mele, L. Impact of vitamin A supplementation on childhood mortality. A randomised controlled community trial. Lancet 1986, 1, 1169–1173. [Google Scholar] [CrossRef]
- West, K.P., Jr.; Pokhrel, R.P.; Katz, J.; LeClerq, S.C.; Khatry, S.K.; Shrestha, S.R.; Pradhan, E.K.; Tielsch, J.M.; Pandey, M.R.; Sommer, A. Efficacy of vitamin A in reducing preschool child mortality in Nepal. Lancet 1991, 338, 67–71. [Google Scholar] [CrossRef]
- Rahmathullah, L.; Tielsch, J.M.; Thulasiraj, R.D.; Katz, J.; Coles, C.; Devi, S.; John, R.; Prakash, K.; Sadanand, A.V.; Edwin, N.; et al. Impact of supplementing newborn infants with vitamin A on early infant mortality: Community based randomised trial in southern India. BMJ 2003, 327, 254. [Google Scholar] [CrossRef] [PubMed]
- Klemm, R.D.; Labrique, A.B.; Christian, P.; Rashid, M.; Shamim, A.A.; Katz, J.; Sommer, A.; West, K.P., Jr. Newborn vitamin A supplementation reduced infant mortality in rural Bangladesh. Pediatrics 2008, 122, e242–e250. [Google Scholar] [CrossRef] [PubMed]
- Bresnahan, K.A.; Tanumihardjo, S.A. Undernutrition, the acute phase response to infection, and its effects on micronutrient status indicators. Adv. Nutr. 2014, 5, 702–711. [Google Scholar] [CrossRef] [PubMed]
- Borel, P.; Desmarchelier, C. Genetic variations associated with vitamin A status and vitamin A bioavailability. Nutrients 2017, 9. [Google Scholar] [CrossRef] [PubMed]
- Edmond, K.M.; Newton, S.; Shannon, C.; O’Leary, M.; Hurt, L.; Thomas, G.; Amenga-Etego, S.; Tawiah-Agyemang, C.; Gram, L.; Hurt, C.N.; et al. Effect of early neonatal vitamin A supplementation on mortality during infancy in Ghana (Neovita): A randomised, double-blind, placebo-controlled trial. Lancet 2015, 385, 1315–1323. [Google Scholar] [CrossRef]
- Masanja, H.; Smith, E.R.; Muhihi, A.; Briegleb, C.; Mshamu, S.; Ruben, J.; Noor, R.A.; Khudyakov, P.; Yoshida, S.; Martines, J.; et al. Effect of neonatal vitamin A supplementation on mortality in infants in Tanzania (Neovita): A randomised, double-blind, placebo-controlled trial. Lancet 2015, 385, 1324–1332. [Google Scholar] [CrossRef]
- Mazumder, S.; Taneja, S.; Bhatia, K.; Yoshida, S.; Kaur, J.; Dube, B.; Toteja, G.S.; Bahl, R.; Fontaine, O.; Martines, J.; et al. Efficacy of early neonatal supplementation with vitamin A to reduce mortality in infancy in Haryana, India (Neovita): A randomised, double-blind, placebo-controlled trial. Lancet 2015, 385, 1333–1342. [Google Scholar] [CrossRef]
- Hamer, D.H.; Keusch, G.T. Vitamin A deficiency: Slow progress towards elimination. Lancet Glob. Health 2015, 3, e502–e503. [Google Scholar] [CrossRef]
- Benn, C.S.; Diness, B.R.; Balde, I.; Rodrigues, A.; Lausch, K.R.; Martins, C.L.; Fisker, A.B.; Aaby, P. Two different doses of supplemental vitamin A did not affect mortality of normal-birth-weight neonates in Guinea-Bissau in a randomized controlled trial. J. Nutr. 2014, 144, 1474–1479. [Google Scholar] [CrossRef] [PubMed]
- Haider, B.A.; Bhutta, Z.A. Neonatal vitamin A supplementation: Time to move on. Lancet 2015, 385, 1268–1271. [Google Scholar] [CrossRef]
- Platts-Mills, J.A.; Babji, S.; Bodhidatta, L.; Gratz, J.; Haque, R.; Havt, A.; McCormick, B.J.; McGrath, M.; Olortegui, M.P.; Samie, A.; et al. Pathogen-specific burdens of community diarrhoea in developing countries: A multisite birth cohort study (MAL-ED). Lancet Glob. Health 2015, 3, e564–e575. [Google Scholar] [CrossRef]
- Thornton, K.A.; Mora-Plazas, M.; Marin, C.; Villamor, E. Vitamin A deficiency is associated with gastrointestinal and respiratory morbidity in school-age children. J. Nutr. 2014, 144, 496–503. [Google Scholar] [CrossRef] [PubMed]
- Al-Mekhlafi, H.M.; Anuar, T.S.; Al-Zabedi, E.M.; Al-Maktari, M.T.; Mahdy, M.A.; Ahmed, A.; Sallam, A.A.; Abdullah, W.A.; Moktar, N.; Surin, J. Does vitamin A supplementation protect schoolchildren from acquiring soil-transmitted helminthiasis? A randomized controlled trial. Parasit. Vectors 2014, 7, 367. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, A.M.; Soares Magalhaes, R.J.; Long, K.Z.; Ahmed, T.; Alam, M.A.; Hossain, M.I.; Islam, M.M.; Mahfuz, M.; Mondal, D.; Haque, R.; et al. Association of vitamin D status with incidence of enterotoxigenic, enteropathogenic and enteroaggregative Escherichia coli diarrhoea in children of urban Bangladesh. Trop. Med. Int. Health 2016, 21, 973–984. [Google Scholar] [CrossRef] [PubMed]
- Kosek, M.; Oberhelman, R.A. Unraveling the contradictions of vitamin A and infectious diseases in children. J. Infect. Dis. 2007, 196, 965–967. [Google Scholar] [CrossRef] [PubMed]
- Hossain, M.I.; Haque, R.; Mondal, D.; Mahfuz, M.; Ahmed, A.S.; Islam, M.M.; Guerrant, R.L.; Petri, W.A., Jr.; Ahmed, T. Undernutrition, Vitamin A and Iron Deficiency Are Associated with Impaired Intestinal Mucosal Permeability in Young Bangladeshi Children Assessed by Lactulose/Mannitol Test. PLoS ONE 2016, 11, e0164447. [Google Scholar] [CrossRef] [PubMed]
- Vieira, M.M.; Paik, J.; Blaner, W.S.; Soares, A.M.; Mota, R.M.; Guerrant, R.L.; Lima, A.A. Carotenoids, retinol, and intestinal barrier function in children from northeastern Brazil. J. Pediatr Gastroenterol. Nutr. 2008, 47, 652–659. [Google Scholar] [CrossRef] [PubMed]
- Guerrant, R.L.; DeBoer, M.D.; Moore, S.R.; Scharf, R.J.; Lima, A.A. The impoverished gut—A triple burden of diarrhoea, stunting and chronic disease. Nat. Rev. Gastroenterol. Hepatol. 2013, 10, 220–229. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
De Medeiros, P.H.Q.S.; Pinto, D.V.; De Almeida, J.Z.; Rêgo, J.M.C.; Rodrigues, F.A.P.; Lima, A.Â.M.; Bolick, D.T.; Guerrant, R.L.; Oriá, R.B. Modulation of Intestinal Immune and Barrier Functions by Vitamin A: Implications for Current Understanding of Malnutrition and Enteric Infections in Children. Nutrients 2018, 10, 1128. https://doi.org/10.3390/nu10091128
De Medeiros PHQS, Pinto DV, De Almeida JZ, Rêgo JMC, Rodrigues FAP, Lima AÂM, Bolick DT, Guerrant RL, Oriá RB. Modulation of Intestinal Immune and Barrier Functions by Vitamin A: Implications for Current Understanding of Malnutrition and Enteric Infections in Children. Nutrients. 2018; 10(9):1128. https://doi.org/10.3390/nu10091128
Chicago/Turabian StyleDe Medeiros, Pedro Henrique Q. S., Daniel V. Pinto, Juliana Zani De Almeida, Juliana M. C. Rêgo, Francisco A. P. Rodrigues, Aldo Ângelo M. Lima, David T. Bolick, Richard L. Guerrant, and Reinaldo B. Oriá. 2018. "Modulation of Intestinal Immune and Barrier Functions by Vitamin A: Implications for Current Understanding of Malnutrition and Enteric Infections in Children" Nutrients 10, no. 9: 1128. https://doi.org/10.3390/nu10091128
APA StyleDe Medeiros, P. H. Q. S., Pinto, D. V., De Almeida, J. Z., Rêgo, J. M. C., Rodrigues, F. A. P., Lima, A. Â. M., Bolick, D. T., Guerrant, R. L., & Oriá, R. B. (2018). Modulation of Intestinal Immune and Barrier Functions by Vitamin A: Implications for Current Understanding of Malnutrition and Enteric Infections in Children. Nutrients, 10(9), 1128. https://doi.org/10.3390/nu10091128




