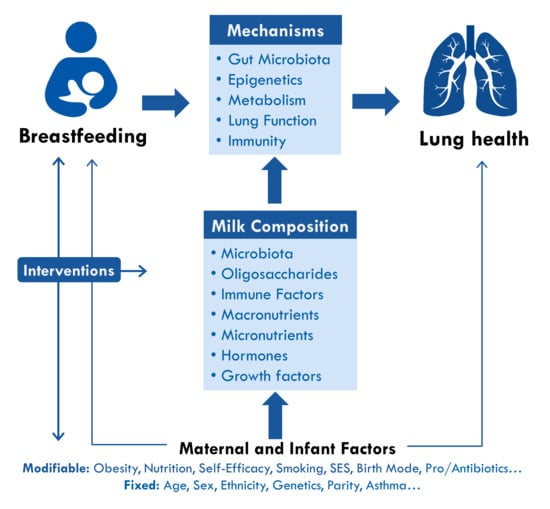
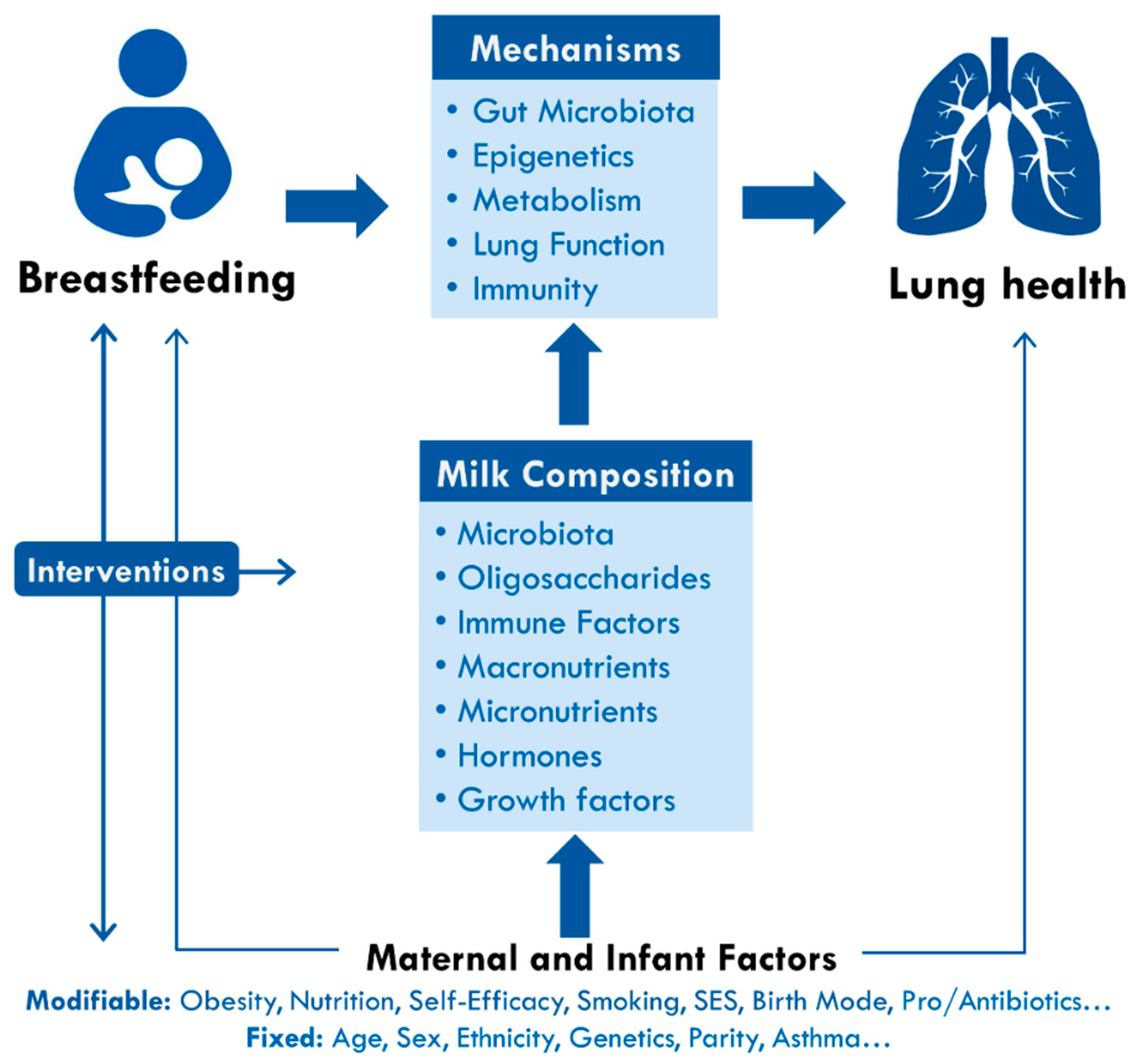
Breastfeeding and the Developmental Origins of Asthma: Current Evidence, Possible Mechanisms, and Future Research Priorities
Abstract
1. Introduction
2. Breastfeeding and Asthma: What is the Evidence, and Why Is It Inconsistent?
2.1. Breastfeeding Definitions
2.2. Asthma Definitions
2.3. Study Design Issues and Limitations
2.4. Confounding
2.5. Reverse Causality
2.6. Effect Modification
2.7. Study Settings and Breastfeeding Culture
2.8. Human Milk Composition
3. Mechanisms: How Could Breastfeeding Protect Against Asthma?
3.1. Epigenetics
3.2. Microbiota
3.3. Immunity and Inflammation
3.4. Lung Growth and Pulmonary Function
4. Conclusions and Recommendations
Author Contributions
Funding
Conflicts of Interest
References
- Pearce, N.; Ait-Khaled, N.; Beasley, R.; Mallol, J.; Keil, U.; Mitchell, E.; Robertson, C. Worldwide trends in the prevalence of asthma symptoms: Phase III of the international study of asthma and allergies in childhood (ISAAC). Thorax 2007, 62, 758–766. [Google Scholar] [CrossRef] [PubMed]
- Harrison, B.D.; Pearson, M.G. Audit in acute severe asthma––Who benefits? J. R. Coll. Physicians Lond. 1993, 27, 387–390. [Google Scholar] [PubMed]
- Barnett, S.B.; Nurmagambetov, T.A. Costs of asthma in the United States: 2002–2007. J. Allergy Clin. Immunol. 2011, 127, 145–152. [Google Scholar] [CrossRef] [PubMed]
- Kozyrskyj, A.L.; Bahreinian, S.; Azad, M.B. Early life exposures: Impact on asthma and allergic disease. Curr. Opin. Allergy Clin. Immunol. 2011, 11, 400–406. [Google Scholar] [CrossRef] [PubMed]
- Barker, D.J. The fetal and infant origins of adult disease. BMJ 1990, 301, 1111. [Google Scholar] [PubMed]
- Wadhwa, P.D.; Buss, C.; Entringer, S.; Swanson, J.M. Developmental origins of health and disease: Brief history of the approach and current focus on epigenetic mechanisms. Semin. Reprod. Med. 2009, 27, 358–368. [Google Scholar] [CrossRef] [PubMed]
- Gluckman, P.D.; Hanson, M.A.; Beedle, A.S. Early life events and their consequences for later disease: A life history and evolutionary perspective. Am. J. Hum. Biol. 2007, 19, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Duijts, L.; Reiss, I.K.; Brusselle, G.; de Jongste, J.C. Early origins of chronic obstructive lung diseases across the life course. Eur. J. Epidemiol. 2014, 29, 871–885. [Google Scholar] [PubMed]
- Oddy, W.H. A review of the effects of breastfeeding on respiratory infections, atopy, and childhood asthma. J. Asthma 2004, 41, 605–621. [Google Scholar] [PubMed]
- Victora, C.G.; Bahl, R.; Barros, A.J.D.; Franca, G.V.A.; Horton, S.; Krasevec, J.; Murch, S.; Sankar, M.J.; Walker, N.; Rollins, N.C.; et al. Breastfeeding in the 21st century: Epidemiology, mechanisms, and lifelong effect. Lancet 2016, 387, 475–490. [Google Scholar] [CrossRef]
- World Health Organization. Indicators for Assessing Infant and Young Child Feeding Practices: Part 1 Definitions: Conclusions of a Consensus Meeting. Available online: http://apps.who.int/iris/bitstream/handle/10665/43895/?sequence=1 (accessed on 10 May 2018).
- Greer, F.R.; Sicherer, S.H.; Burks, A.W. Effects of early nutritional interventions on the development of atopic disease in infants and children: The role of maternal dietary restriction, breastfeeding, timing of introduction of complementary foods, and hydrolyzed formulas. Pediatrics 2008, 121, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Munblit, D.; Peroni, D.G.; Boix-Amoros, A.; Hsu, P.S.; Land, B.V.; Gay, M.C.L.; Kolotilina, A.; Skevaki, C.; Boyle, R.J.; Collado, M.C.; et al. Human milk and allergic diseases: An unsolved puzzle. Nutrients 2017, 9, 894. [Google Scholar] [CrossRef] [PubMed]
- Azad, M.B.; Vehling, L.; Lu, Z.; Dai, D.; Subbarao, P.; Becker, A.B.; Mandhane, P.J.; Turvey, S.E.; Lefebvre, D.L.; Sears, M.R. Breastfeeding, maternal asthma and wheezing in the first year of life: A longitudinal birth cohort study. Eur. Respir. J. 2017, 49, 1602019. [Google Scholar] [CrossRef] [PubMed]
- Klopp, A.; Vehling, L.; Becker, A.B.; Subbarao, P.; Mandhane, P.J.; Turvey, S.E.; Lefebvre, D.L.; Sears, M.R.; Azad, M.B. Modes of infant feeding and the risk of childhood asthma: A prospective birth cohort study. J. Pediatr. 2017, 190, 192–199. [Google Scholar] [CrossRef] [PubMed]
- Dell, S.; To, T. Breastfeeding and asthma in young children: Findings from a population-based study. Arch. Pediatr. Adolesc. Med. 2001, 155, 1261–1265. [Google Scholar] [CrossRef] [PubMed]
- Kull, I.; Almqvist, C.; Lilja, G.; Pershagen, G.; Wickman, M. Breast-feeding reduces the risk of asthma during the first 4 years of life. J. Allergy Clin. Immunol. 2004, 114, 755–760. [Google Scholar] [CrossRef] [PubMed]
- Oddy, W.H.; Holt, P.G.; Sly, P.D.; Read, A.W.; Landau, L.I.; Stanley, F.J.; Kendall, G.E.; Burton, P.R. Association between breast feeding and asthma in 6 year old children: Findings of a prospective birth cohort study. BMJ 1999, 319, 815–819. [Google Scholar] [CrossRef] [PubMed]
- Burgess, S.W.; Dakin, C.J.; O’Callaghan, M.J. Breastfeeding does not increase the risk of asthma at 14 years. Pediatrics 2006, 117, e787–e792. [Google Scholar] [CrossRef] [PubMed]
- Wright, A.L.; Holberg, C.J.; Taussig, L.M.; Martinez, F.D. Factors influencing the relation of infant feeding to asthma and recurrent wheeze in childhood. Thorax 2001, 56, 192–197. [Google Scholar] [CrossRef] [PubMed]
- Sears, M.R.; Greene, J.M.; Willan, A.R.; Taylor, D.R.; Flannery, E.M.; Cowan, J.O.; Herbison, G.P.; Poulton, R. Long-term relation between breastfeeding and development of atopy and asthma in children and young adults: A longitudinal study. Lancet 2002, 360, 901–907. [Google Scholar] [CrossRef]
- Grulee, C.G.; Sanford, H.N. The influence of breast and artificial feeding on infantile eczema. J. Pediatr. 1936, 9, 223–225. [Google Scholar] [CrossRef]
- Dogaru, C.M.; Nyffenegger, D.; Pescatore, A.M.; Spycher, B.D.; Kuehni, C.E. Breastfeeding and childhood asthma: Systematic review and meta-analysis. Am. J. Epidemiol. 2014, 179, 1153–1167. [Google Scholar] [PubMed]
- Lodge, C.J.; Tan, D.J.; Lau, M.X.; Dai, X.; Tham, R.; Lowe, A.J.; Bowatte, G.; Allen, K.J.; Dharmage, S.C. Breastfeeding and asthma and allergies: A systematic review and meta-analysis. Acta Paediatr. 2015, 104, 38–53. [Google Scholar] [PubMed]
- Brew, B.K.; Allen, C.W.; Toelle, B.G.; Marks, G.B. Systematic review and meta-analysis investigating breast feeding and childhood wheezing illness. Paediatr. Perinat. Epidemiol. 2011, 25, 507–518. [Google Scholar] [PubMed]
- Gdalevich, M.; Mimouni, D.; Mimouni, M. Breast-feeding and the risk of bronchial asthma in childhood: A systematic review with meta-analysis of prospective studies. J. Pediatr. 2001, 139, 261–266. [Google Scholar] [CrossRef] [PubMed]
- Den Dekker, H.T.; Sonnenschein-van der Voort, A.M.; Jaddoe, V.W.; Reiss, I.K.; de Jongste, J.C.; Duijts, L. Breastfeeding and asthma outcomes at the age of 6 years: The generation R study. Pediatr. Allergy Immunol. 2016, 27, 486–492. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, S.E.; Hurd, S.S.; Lemanske, R.F., Jr.; Becker, A.; Zar, H.J.; Sly, P.D.; Soto-Quiroz, M.; Wong, G.; Bateman, E.D. Global strategy for the diagnosis and management of asthma in children 5 years and younger. Pediatr. Pulmonol. 2011, 46, 1–17. [Google Scholar] [PubMed]
- Labbok, M.H.; Starling, A. Definitions of breastfeeding: Call for the development and use of consistent definitions in research and peer-reviewed literature. Breastfeed. Med. 2012, 7, 397–402. [Google Scholar] [CrossRef] [PubMed]
- Soto-Ramirez, N.; Karmaus, W.; Zhang, H.; Davis, S.; Agarwal, S.; Albergottie, A. Modes of infant feeding and the occurrence of coughing/wheezing in the first year of life. J. Hum. Lact. 2013, 29, 71–80. [Google Scholar] [CrossRef] [PubMed]
- Raoof, N.A.; Adamkin, D.H.; Radmacher, P.G.; Telang, S. Comparison of lactoferrin activity in fresh and stored human milk. J. Perinatol. 2016, 36, 207–209. [Google Scholar] [CrossRef] [PubMed]
- Ahrabi, A.F.; Handa, D.; Codipilly, C.N.; Shah, S.; Williams, J.E.; McGuire, M.A.; Potak, D.; Aharon, G.G.; Schanler, R.J. Effects of extended freezer storage on the integrity of human milk. J. Pediatr. 2016, 177, 140–143. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, R.A. Storage of human milk and the influence of procedures on immunological components of human milk. Acta Paediatr. Suppl. 1999, 88, 14–18. [Google Scholar] [CrossRef] [PubMed]
- Nam, S.H.; Seo, Y.M.; Kim, M.G. Bisphenol a migration from polycarbonate baby bottle with repeated use. Chemosphere 2010, 79, 949–952. [Google Scholar] [CrossRef] [PubMed]
- Robinson, L.; Miller, R. The impact of bisphenol A and phthalates on allergy, asthma, and immune function: A review of latest findings. Curr. Environ. Health Rep. 2015, 2, 379–387. [Google Scholar] [CrossRef] [PubMed]
- Ogbuanu, I.U.; Karmaus, W.; Arshad, S.H.; Kurukulaaratchy, R.J.; Ewart, S. Effect of breastfeeding duration on lung function at age 10 years: A prospective birth cohort study. Thorax 2009, 64, 62–66. [Google Scholar] [CrossRef] [PubMed]
- Tully, M.R.; Lockhart-Borman, L.; Updegrove, K. Stories of success: The use of donor milk is increasing in North America. J. Hum. Lact. 2004, 20, 75–77. [Google Scholar] [CrossRef] [PubMed]
- Reddel, H.K.; Bateman, E.D.; Becker, A.; Boulet, L.P.; Cruz, A.A.; Drazen, J.M.; Haahtela, T.; Hurd, S.S.; Inoue, H.; de Jongste, J.C.; et al. A summary of the new GINA strategy: A roadmap to asthma control. Eur. Respir. J. 2015, 46, 622–639. [Google Scholar] [CrossRef] [PubMed]
- Miller, M.R.; Hankinson, J.; Brusasco, V.; Burgos, F.; Casaburi, R.; Coates, A.; Crapo, R.; Enright, P.; van der Grinten, C.P.; Gustafsson, P.; et al. Standardisation of spirometry. Eur. Respir. J. 2005, 26, 319–338. [Google Scholar] [CrossRef] [PubMed]
- Van Meel, E.R.; de Jong, M.; Elbert, N.J.; den Dekker, H.T.; Reiss, I.K.; de Jongste, J.C.; Jaddoe, V.W.V.; Duijts, L. Duration and exclusiveness of breastfeeding and school-age lung function and asthma. Ann. Allergy Asthma Immunol. 2017, 119, 21–26. [Google Scholar] [CrossRef] [PubMed]
- De Benedictis, F.M.; Bush, A. Infantile wheeze: Rethinking dogma. Arch. Dis. Child 2017, 102, 371–375. [Google Scholar] [PubMed]
- Wenzel, S.E. Asthma phenotypes: The evolution from clinical to molecular approaches. Nat. Med. 2012, 18, 716–725. [Google Scholar] [PubMed]
- Zedan, M.M.; Laimon, W.N.; Osman, A.M.; Zedan, M.M. Clinical asthma phenotyping: A trial for bridging gaps in asthma management. World J. Clin. Pediatr. 2015, 4, 13–18. [Google Scholar] [CrossRef] [PubMed]
- Ek, W.E.; Karlsson, T.; Hernandes, C.A.; Rask-Andersen, M.; Johansson, A. Breast-feeding and risk of asthma, hay fever, and eczema. J. Allergy Clin. Immunol. 2018, 141, 1157–1159.e1159. [Google Scholar] [CrossRef] [PubMed]
- Kramer, M.S.; Chalmers, B.; Hodnett, E.D.; Sevkovskaya, Z.; Dzikovich, I.; Shapiro, S.; Collet, J.P.; Vanilovich, I.; Mezen, I.; Ducruet, T.; et al. Promotion of breastfeeding intervention trial (PROBIT): A randomized trial in the republic of belarus. JAMA 2001, 285, 413–420. [Google Scholar] [CrossRef] [PubMed]
- Kramer, M.S.; Matush, L.; Vanilovich, I.; Platt, R.; Bogdanovich, N.; Sevkovskaya, Z.; Dzikovich, I.; Shishko, G.; Mazer, B.; Promotion of Breastfeeding Intervention Trial Study, G. Effect of prolonged and exclusive breast feeding on risk of allergy and asthma: Cluster randomised trial. BMJ 2007, 335, 815. [Google Scholar] [CrossRef] [PubMed]
- Flohr, C.; Henderson, A.J.; Kramer, M.S.; Patel, R.; Thompson, J.; Rifas-Shiman, S.L.; Yang, S.; Vilchuck, K.; Bogdanovich, N.; Hameza, M.; et al. Effect of an intervention to promote breastfeeding on asthma, lung function, and atopic eczema at age 16 years: Follow-up of the probit randomized trial. JAMA Pediatr. 2018, 172, e174064. [Google Scholar] [CrossRef] [PubMed]
- Asher, I.; Pearce, N. Global burden of asthma among children. Int. J. Tuberc. Lung Dis. 2014, 18, 1269–1278. [Google Scholar] [CrossRef] [PubMed]
- Kramer, M.S. Breastfeeding and allergy: The evidence. Ann. Nutr. Metab. 2011, 59, 20–26. [Google Scholar] [CrossRef] [PubMed]
- Kramer, M.S. Methodological challenges in studying long-term effects of breast-feeding. Adv. Exp. Med. Biol. 2009, 639, 121–133. [Google Scholar] [PubMed]
- Leung, J.Y.; Kwok, M.K.; Leung, G.M.; Schooling, C.M. Breastfeeding and childhood hospitalizations for asthma and other wheezing disorders. Ann. Epidemiol. 2016, 26, 21–27. [Google Scholar] [CrossRef] [PubMed]
- Da Costa Lima, R.; Victora, C.G.; Menezes, A.M.; Barros, F.C. Do risk factors for childhood infections and malnutrition protect against asthma? A study of Brazilian male adolescents. Am. J. Public Health 2003, 93, 1858–1864. [Google Scholar] [CrossRef] [PubMed]
- Vehling, L.; Chan, D.; McGavock, J.; Becker, A.B.; Subbarao, P.; Moraes, T.J.; Mandhane, P.J.; Turvey, S.E.; Lefebvre, D.L.; Sears, M.R.; et al. Exclusive breastfeeding in hospital predicts longer breastfeeding duration in Canada: Implications for health equity. Birth 2018. [Google Scholar] [CrossRef] [PubMed]
- Miliku, K.; Voortman, T.; Bakker, H.; Hofman, A.; Franco, O.H.; Jaddoe, V.W. Infant breastfeeding and kidney function in school-aged children. Am. J. Kidney Dis. 2015, 66, 421–428. [Google Scholar] [CrossRef] [PubMed]
- Thulier, D.; Mercer, J. Variables associated with breastfeeding duration. J. Obstet. Gynecol. Neonatal Nurs. 2009, 38, 259–268. [Google Scholar] [CrossRef] [PubMed]
- Wen, X.; Kong, K.L.; Eiden, R.D.; Sharma, N.N.; Xie, C. Sociodemographic differences and infant dietary patterns. Pediatrics 2014, 134, e1387–e1398. [Google Scholar] [CrossRef] [PubMed]
- Frisell, T.; Oberg, S.; Kuja-Halkola, R.; Sjolander, A. Sibling comparison designs: Bias from non-shared confounders and measurement error. Epidemiology 2012, 23, 713–720. [Google Scholar] [CrossRef] [PubMed]
- Smith, G.D.; Ebrahim, S. Mendelian randomization: Prospects, potentials, and limitations. Int. J. Epidemiol. 2004, 33, 30–42. [Google Scholar] [CrossRef] [PubMed]
- Lowe, A.J.; Carlin, J.B.; Bennett, C.M.; Abramson, M.J.; Hosking, C.S.; Hill, D.J.; Dharmage, S.C. Atopic disease and breast-feeding––Cause or consequence? J. Allergy Clin. Immunol. 2006, 117, 682–687. [Google Scholar] [CrossRef] [PubMed]
- Dogaru, C.M.; Strippoli, M.P.; Spycher, B.D.; Frey, U.; Beardsmore, C.S.; Silverman, M.; Kuehni, C.E. Breastfeeding and lung function at school age: Does maternal asthma modify the effect? Am. J. Respir. Crit. Care Med. 2012, 185, 874–880. [Google Scholar] [CrossRef] [PubMed]
- Dong, G.H.; Qian, Z.M.; Liu, M.M.; Wang, D.; Ren, W.H.; Bawa, S.; Fu, J.; Wang, J.; Lewis, R.; Zelicoff, A.; et al. Breastfeeding as a modifier of the respiratory effects of air pollution in children. Epidemiology 2013, 24, 387–394. [Google Scholar] [CrossRef] [PubMed]
- Woodward, A.; Douglas, R.M.; Graham, N.M.; Miles, H. Acute respiratory illness in Adelaide children: Breast feeding modifies the effect of passive smoking. J. Epidemiol. Community Health 1990, 44, 224–230. [Google Scholar] [CrossRef] [PubMed]
- Montgomery, S.M.; Ehlin, A.; Sacker, A. Breast feeding and resilience against psychosocial stress. Arch. Dis. Child 2006, 91, 990–994. [Google Scholar] [CrossRef] [PubMed]
- Beasley, R.; Crane, J.; Lai, C.K.; Pearce, N. Prevalence and etiology of asthma. J. Allergy Clin. Immunol. 2000, 105, S466–S472. [Google Scholar] [CrossRef]
- Beasley, R. Worldwide variation in prevalence of symptoms of asthma, allergic rhinoconjunctivitis, and atopic eczema: ISAAC. Lancet 1998, 351, 1225–1232. [Google Scholar] [CrossRef]
- Kelly, Y.J.; Watt, R.G.; Nazroo, J.Y. Racial/ethnic differences in breastfeeding initiation and continuation in the United Kingdom and comparison with findings in the United States. Pediatrics 2006, 118, e1428–e1435. [Google Scholar] [CrossRef] [PubMed]
- Albernaz, E.; Araujo, C.L.; Tomasi, E.; Mintem, G.; Giugliani, E.; Matijasevich, A.; Onis, M.; Barros, F.C.; Victora, C.G. Influence of breastfeeding support on the tendencies of breastfeeding rates in the city of Pelotas (RS), Brazil, from 1982 to 2004. J. Pediatr. 2008, 84, 560–564. [Google Scholar] [CrossRef]
- Brown, A. Breastfeeding as a public health responsibility: A review of the evidence. J. Hum. Nutr. Diet. 2017, 30, 759–770. [Google Scholar] [PubMed]
- Gross, T.T.; Powell, R.; Anderson, A.K.; Hall, J.; Davis, M.; Hilyard, K. WIC peer counselors’ perceptions of breastfeeding in African American women with lower incomes. J. Hum. Lact 2015, 31, 99–110. [Google Scholar] [CrossRef] [PubMed]
- Kent, G. Global infant formula: Monitoring and regulating the impacts to protect human health. Int. Breastfeed. J. 2015, 10, 6. [Google Scholar] [CrossRef] [PubMed]
- Dagher, R.K.; McGovern, P.M.; Schold, J.D.; Randall, X.J. Determinants of breastfeeding initiation and cessation among employed mothers: A prospective cohort study. BMC Pregnancy Childbirth 2016, 16, 194. [Google Scholar] [CrossRef] [PubMed]
- Mirkovic, K.R.; Perrine, C.G.; Scanlon, K.S. Paid maternity leave and breastfeeding outcomes. Birth 2016, 43, 233–239. [Google Scholar] [CrossRef] [PubMed]
- Geraghty, S.R.; Khoury, J.C.; Kalkwarf, H.J. Human milk pumping rates of mothers of singletons and mothers of multiples. J. Hum. Lact. 2005, 21, 413–420. [Google Scholar] [CrossRef] [PubMed]
- D’Alessandro, A.; Scaloni, A.; Zolla, L. Human milk proteins: An interactomics and updated functional overview. J. Proteome Res. 2010, 9, 3339–3373. [Google Scholar] [CrossRef] [PubMed]
- Arrieta, M.C.; Stiemsma, L.T.; Dimitriu, P.A.; Thorson, L.; Russell, S.; Yurist-Doutsch, S.; Kuzeljevic, B.; Gold, M.J.; Britton, H.M.; Lefebvre, D.L.; et al. Early infancy microbial and metabolic alterations affect risk of childhood asthma. Sci. Transl. Med. 2015, 7, 307ra152. [Google Scholar] [PubMed]
- Waidyatillake, N.T.; Dharmage, S.C.; Allen, K.J.; Lodge, C.J.; Simpson, J.A.; Bowatte, G.; Abramson, M.J.; Lowe, A.J. Association of breast milk fatty acids with allergic disease outcomes—A systematic review. Allergy 2018, 73, 295–312. [Google Scholar] [CrossRef] [PubMed]
- Doherty, A.M.; Lodge, C.J.; Dharmage, S.C.; Dai, X.; Bode, L.; Lowe, A.J. Human Milk Oligosaccharides and Associations with Immune-Mediated Disease and Infection in Childhood: A Systematic Review. Front. Pediatr. 2018, 20, 6. [Google Scholar]
- Oddy, W.H.; Rosales, F. A systematic review of the importance of milk TGF-beta on immunological outcomes in the infant and young child. Pediatr. Allergy Immunol. 2010, 21, 47–59. [Google Scholar] [CrossRef] [PubMed]
- Smilowitz, J.T.; Lebrilla, C.B.; Mills, D.A.; German, J.B.; Freeman, S.L. Breast milk oligosaccharides: Structure-function relationships in the neonate. Annu. Rev. Nutr. 2014, 34, 143–169. [Google Scholar] [CrossRef] [PubMed]
- Ballard, O.; Morrow, A.L. Human milk composition: Nutrients and bioactive factors. Pediatr. Clin. N. Am. 2013, 60, 49–74. [Google Scholar] [CrossRef] [PubMed]
- Peroni, D.G.; Pescollderungg, L.; Piacentini, G.L.; Rigotti, E.; Maselli, M.; Watschinger, K.; Piazza, M.; Pigozzi, R.; Boner, A.L. Immune regulatory cytokines in the milk of lactating women from farming and urban environments. Pediatr. Allergy Immunol. 2010, 21, 977–982. [Google Scholar] [CrossRef] [PubMed]
- Munblit, D.; Boyle, R.J.; Warner, J.O. Factors affecting breast milk composition and potential consequences for development of the allergic phenotype. Clin. Exp. Allergy 2015, 45, 583–601. [Google Scholar] [PubMed]
- Miliku, K.; Goruk, S.; Becker, A.B.; Padmaja, S.; Mandhane, P.; Turvey, S.E.; Lefebvre, D.L.; Sears, M.R.; Field, C.J.; Azad, M.B. Human milk fatty acids: Associations with maternal characteristics and infant body composition in the child study. In Proceedings of the Canadian National Perinatal Research Meeting, Banff, AB, Canada, 14–17 February 2018; Available online: http://www.cnprm.org/wp-content/uploads/2013/09/CNPRM-2018-full-program-rev-08-02-18.pdf (accessed on 10 May 2018).
- McGuire, M.K.; Meehan, C.L.; McGuire, M.A.; Williams, J.E.; Foster, J.; Sellen, D.W.; Kamau-Mbuthia, E.W.; Kamundia, E.W.; Mbugua, S.; Moore, S.E.; et al. What’s normal? Oligosaccharide concentrations and profiles in milk produced by healthy women vary geographically. Am. J. Clin. Nutr. 2017, 105, 1086–1100. [Google Scholar] [PubMed]
- Sinanoglou, V.J.; Cavouras, D.; Boutsikou, T.; Briana, D.D.; Lantzouraki, D.Z.; Paliatsiou, S.; Volaki, P.; Bratakos, S.; Malamitsi-Puchner, A.; Zoumpoulakis, P. Factors affecting human colostrum fatty acid profile: A case study. PLoS ONE 2017, 12, e0175817. [Google Scholar] [CrossRef] [PubMed]
- Su, L.L.; S, K.T.C.; Lim, S.L.; Chen, Y.; Tan, E.A.; Pai, N.N.; Gong, Y.H.; Foo, J.; Rauff, M.; Chong, Y.S. The influence of maternal ethnic group and diet on breast milk fatty acid composition. Ann. Acad. Med. Singap. 2010, 39, 675. [Google Scholar] [PubMed]
- Brenna, J.T.; Varamini, B.; Jensen, R.G.; Diersen-Schade, D.A.; Boettcher, J.A.; Arterburn, L.M. Docosahexaenoic and arachidonic acid concentrations in human breast milk worldwide. Am. J. Clin. Nutr. 2007, 85, 1457–1464. [Google Scholar] [PubMed]
- Urwin, H.J.; Miles, E.A.; Noakes, P.S.; Kremmyda, L.S.; Vlachava, M.; Diaper, N.D.; Perez-Cano, F.J.; Godfrey, K.M.; Calder, P.C.; Yaqoob, P. Salmon consumption during pregnancy alters fatty acid composition and secretory iga concentration in human breast milk. J. Nutr. 2012, 142, 1603–1610. [Google Scholar] [CrossRef] [PubMed]
- Van Elten, T.M.; van Rossem, L.; Wijga, A.H.; Brunekreef, B.; de Jongste, J.C.; Koppelman, G.H.; Smit, H.A. Breast milk fatty acid composition has a long-term effect on the risk of asthma, eczema, and sensitization. Allergy 2015, 70, 1468–1476. [Google Scholar] [PubMed]
- Hoppu, U.; Isolauri, E.; Laakso, P.; Matomaki, J.; Laitinen, K. Probiotics and dietary counselling targeting maternal dietary fat intake modifies breast milk fatty acids and cytokines. Eur. J. Nutr. 2012, 51, 211–219. [Google Scholar] [CrossRef] [PubMed]
- Bianco-Miotto, T.; Craig, J.M.; Gasser, Y.P.; van Dijk, S.J.; Ozanne, S.E. Epigenetics and DOHaD: From basics to birth and beyond. J. Dev. Orig. Health Dis. 2017, 8, 513–519. [Google Scholar] [PubMed]
- Martino, D.; Prescott, S. Epigenetics and prenatal influences on asthma and allergic airways disease. Chest 2011, 139, 640–647. [Google Scholar] [CrossRef] [PubMed]
- Hartwig, F.P.; Loret de Mola, C.; Davies, N.M.; Victora, C.G.; Relton, C.L. Breastfeeding effects on DNA methylation in the offspring: A systematic literature review. PLoS ONE 2017, 12, e0173070. [Google Scholar]
- Tao, M.H.; Marian, C.; Shields, P.G.; Potischman, N.; Nie, J.; Krishnan, S.S.; Berry, D.L.; Kallakury, B.V.; Ambrosone, C.; Edge, S.B.; et al. Exposures in early life: Associations with DNA promoter methylation in breast tumors. J. Dev. Orig. Health Dis. 2013, 4, 182–190. [Google Scholar] [PubMed]
- Rossnerova, A.; Tulupova, E.; Tabashidze, N.; Schmuczerova, J.; Dostal, M.; Rossner, P., Jr.; Gmuender, H.; Sram, R.J. Factors affecting the 27k DNA methylation pattern in asthmatic and healthy children from locations with various environments. Mutat. Res. 2013, 74, 18–26. [Google Scholar] [CrossRef] [PubMed]
- Karlsson, O.; Rodosthenous, R.S.; Jara, C.; Brennan, K.J.; Wright, R.O.; Baccarelli, A.A.; Wright, R.J. Detection of long non-coding RNAs in human breastmilk extracellular vesicles: Implications for early child development. Epigenetics 2016, 11, 721–729. [Google Scholar] [CrossRef] [PubMed]
- Alsaweed, M.; Hartmann, P.E.; Geddes, D.T.; Kakulas, F. Micrornas in breastmilk and the lactating breast: Potential immunoprotectors and developmental regulators for the infant and the mother. Int. J. Environ. Res. Public Health 2015, 12, 13981–14020. [Google Scholar] [PubMed]
- Azad, M.B.; Konya, T.; Persaud, R.R.; Guttman, D.S.; Chari, R.S.; Field, C.J.; Sears, M.R.; Mandhane, P.J.; Turvey, S.E.; Subbarao, P.; et al. Impact of maternal intrapartum antibiotics, method of birth and breastfeeding on gut microbiota during the first year of life: A prospective cohort study. BJOG 2016, 123, 983–993. [Google Scholar] [CrossRef] [PubMed]
- Forbes, J.; Konya, T.; Guttman, D.S.; Field, C.J.; Sears, M.R.; Becker, A.B.; Scott, J.A.; the CHILD Study Investigators; Kozyrskyj, A.L.; Azad, M.B. Formula exposure in hospital and subsequent infant feeding practices: Associations with gut microbiota and overweight risk in the first year of life. JAMA Pediatr. 2018, 172, e181161. [Google Scholar] [CrossRef] [PubMed]
- Bode, L.; McGuire, M.; Rodriguez, J.M.; Geddes, D.T.; Hassiotou, F.; Hartmann, P.E.; McGuire, M.K. It’s alive: Microbes and cells in human milk and their potential benefits to mother and infant. Adv. Nutr. 2014, 5, 571–573. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, J.L.; Shi, H.N.; Walker, W.A. The role of microbes in developmental immunologic programming. Pediatr. Res. 2011, 69, 465–472. [Google Scholar] [PubMed]
- Moossavi, S.; Miliku, K.; Sepehri, S.; Khafipour, E.; Azad, M.B. The prebiotic and probiotic properties of human milk: Implications for infant immune development and pediatric asthma. Front. Pediatr. 2018, 6, 197. [Google Scholar] [CrossRef]
- Dethlefsen, L.; McFall-Ngai, M.; Relman, D.A. An ecological and evolutionary perspective on human-microbe mutualism and disease. Nature 2007, 449, 811–818. [Google Scholar] [CrossRef] [PubMed]
- Abrahamsson, T.R.; Jakobsson, H.E.; Andersson, A.F.; Bjorksten, B.; Engstrand, L.; Jenmalm, M.C. Low gut microbiota diversity in early infancy precedes asthma at school age. Clin. Exp. Allergy 2014, 44, 842–850. [Google Scholar] [CrossRef] [PubMed]
- Pabst, H.F.; Spady, D.W.; Pilarski, L.M.; Carson, M.M.; Beeler, J.A.; Krezolek, M.P. Differential modulation of the immune response by breast- or formula-feeding of infants. Acta Paediatr. 1997, 86, 1291–1297. [Google Scholar] [CrossRef] [PubMed]
- Beijers, R.; Riksen-Walraven, J.M.; de Weerth, C. Cortisol regulation in 12-month-old human infants: Associations with the infants’ early history of breastfeeding and co-sleeping. Stress 2013, 16, 267–277. [Google Scholar] [CrossRef] [PubMed]
- Wright, R.J. Stress and childhood asthma risk: Overlapping evidence from animal studies and epidemiologic research. Allergy Asthma Clin. Immunol. 2008, 4, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Waidyatillake, N.T.; Allen, K.J.; Lodge, C.J.; Dharmage, S.C.; Abramson, M.J.; Simpson, J.A.; Lowe, A.J. The impact of breastfeeding on lung development and function: A systematic review. Expert. Rev. Clin. Immunol. 2013, 9, 1253–1265. [Google Scholar] [CrossRef] [PubMed]
- Hill, A.B. The environment and disease: Association or causation? 1965. J. R. Soc. Med. 2015, 108, 32–37. [Google Scholar] [CrossRef] [PubMed]
- Asher, M.I.; Keil, U.; Anderson, H.R.; Beasley, R.; Crane, J.; Martinez, F.; Mitchell, E.A.; Pearce, N.; Sibbald, B.; Stewart, A.W.; et al. International study of asthma and allergies in childhood (ISAAC): Rationale and methods. Eur. Respir. J. 1995, 8, 483–491. [Google Scholar] [CrossRef] [PubMed]
| Domain | Recommendations |
|---|---|
| Study design | Ideally, recruit prenatally and follow prospectively |
| Breastfeeding exposures | Essential: capture exclusivity and duration, and evaluate dose effects:
|
| Asthma outcomes | Define using international criteria [38,110]. Ideally, use objective physician diagnosis and capture pulmonary function. Also consider:
|
| Confounding | Adjust for essential confounders
|
| Effect Modifiers | Test for interactions or conduct stratified analyses by:
|
| Reverse causality | Conduct sensitivity analyses excluding infants with asthma symptoms prior to weaning. |
| Mechanistic studies | Use biological specimens or pre-clinical models to study causal pathways identified in clinical studies
|
| Generalizability | Consider and comment on generalizability of results based on study population and setting (e.g., breastfeeding rates, policies and culture; asthma prevalence) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Miliku, K.; Azad, M.B. Breastfeeding and the Developmental Origins of Asthma: Current Evidence, Possible Mechanisms, and Future Research Priorities. Nutrients 2018, 10, 995. https://doi.org/10.3390/nu10080995
Miliku K, Azad MB. Breastfeeding and the Developmental Origins of Asthma: Current Evidence, Possible Mechanisms, and Future Research Priorities. Nutrients. 2018; 10(8):995. https://doi.org/10.3390/nu10080995
Chicago/Turabian StyleMiliku, Kozeta, and Meghan B. Azad. 2018. "Breastfeeding and the Developmental Origins of Asthma: Current Evidence, Possible Mechanisms, and Future Research Priorities" Nutrients 10, no. 8: 995. https://doi.org/10.3390/nu10080995
APA StyleMiliku, K., & Azad, M. B. (2018). Breastfeeding and the Developmental Origins of Asthma: Current Evidence, Possible Mechanisms, and Future Research Priorities. Nutrients, 10(8), 995. https://doi.org/10.3390/nu10080995