Vitamin C to Improve Organ Dysfunction in Cardiac Surgery Patients—Review and Pragmatic Approach
Abstract
1. Introduction
1.1. Pathogenesis of Organ Dysfunction after Cardiac Surgery
1.2. Basic Metabolism and Functions of Vitamin C
1.3. The Influence of Vitamin C on Oxidative Stress and Inflammation
1.4. Current Evidence of Vitamin C in Critically Ill Patients
2. Influence of Vitamin C on Organ Systems in Cardiac Surgery Patients
2.1. Nervous System
2.1.1. Neuropsychological Dysfunction after Cardiac Surgery
2.1.2. Role of Vitamin C in the Nervous System
2.1.3. Vitamin C’s Influence on the Nervous System in Cardiac Surgery Patients
2.2. Cardiovascular System
2.2.1. Cardiovascular Dysfunction after Cardiac Surgery
2.2.2. Role of Vitamin C in the Cardiovascular System
2.2.3. Vitamin C’s Influence on the Cardiovascular System in Cardiac Surgery Patients
2.3. Respiratory System
2.3.1. Pulmonary Dysfunction after Cardiac Surgery
2.3.2. Role of Vitamin C in the Respiratory System
2.3.3. Vitamin C’s Influence on the Respiratory System in Cardiac Surgery Patients
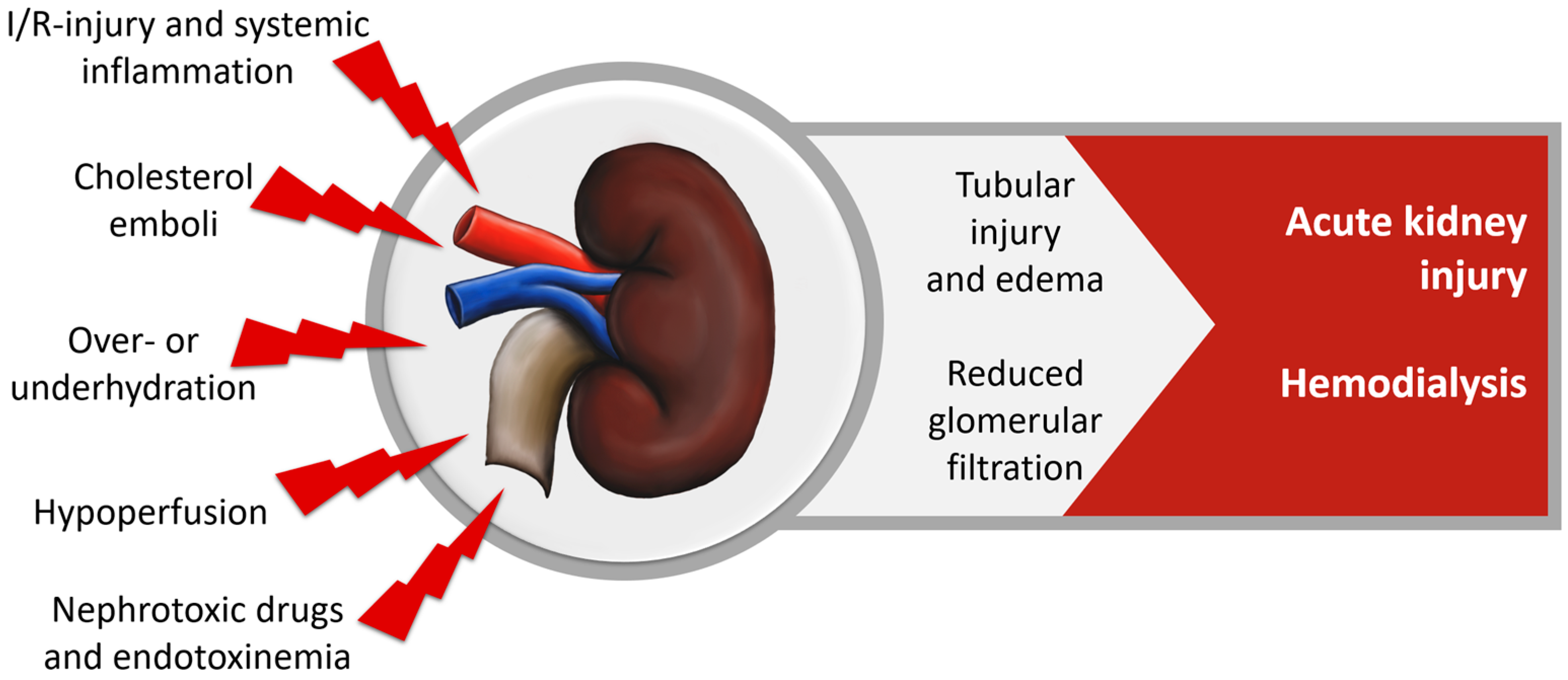
2.4. Renal System
2.4.1. Renal Dysfunction after Cardiac Surgery
2.4.2. Role of Vitamin C in the Renal System
2.4.3. Vitamin C’s Influence on the Renal System in Cardiac Surgery Patients
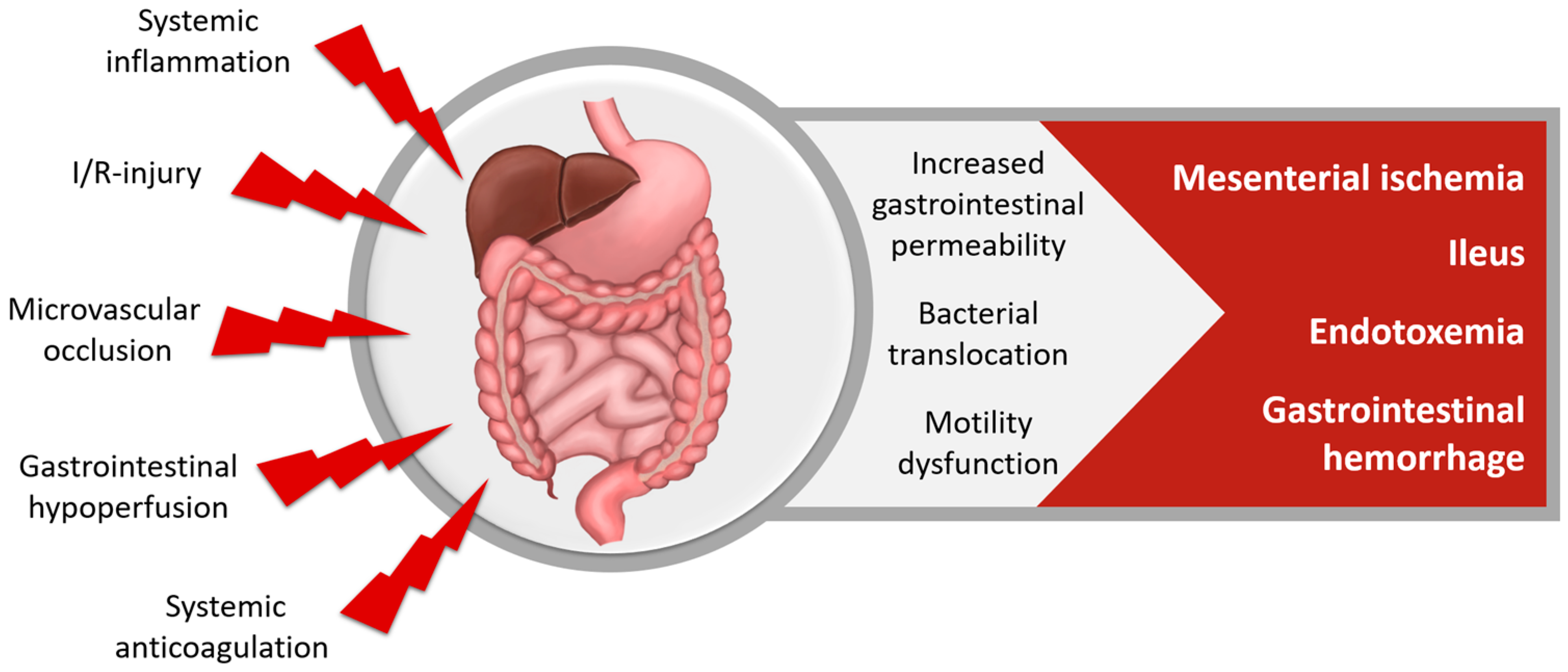
2.5. Gastrointestinal System
2.5.1. Gastrointestinal Dysfunction after Cardiac Surgery
2.5.2. Role of Vitamin C in the Gastrointestinal System
2.5.3. Vitamin C’s Influence on the Gastrointestinal System in Cardiac Surgery Patients
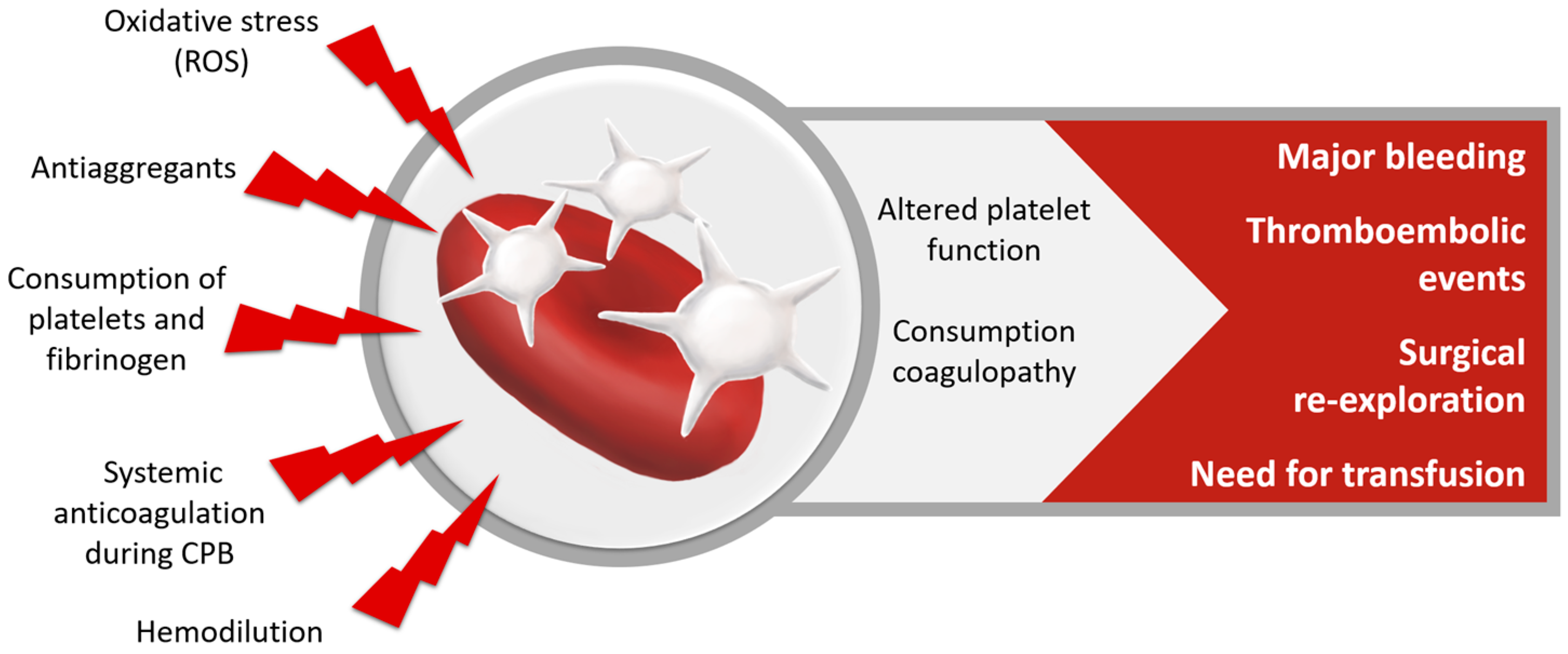
2.6. Coagulation System
2.6.1. Coagulation Disorders after Cardiac Surgery
2.6.2. Role of Vitamin C in the Coagulation System
2.6.3. Vitamin C’s Influence on the Coagulation System in Cardiac Surgery Patients
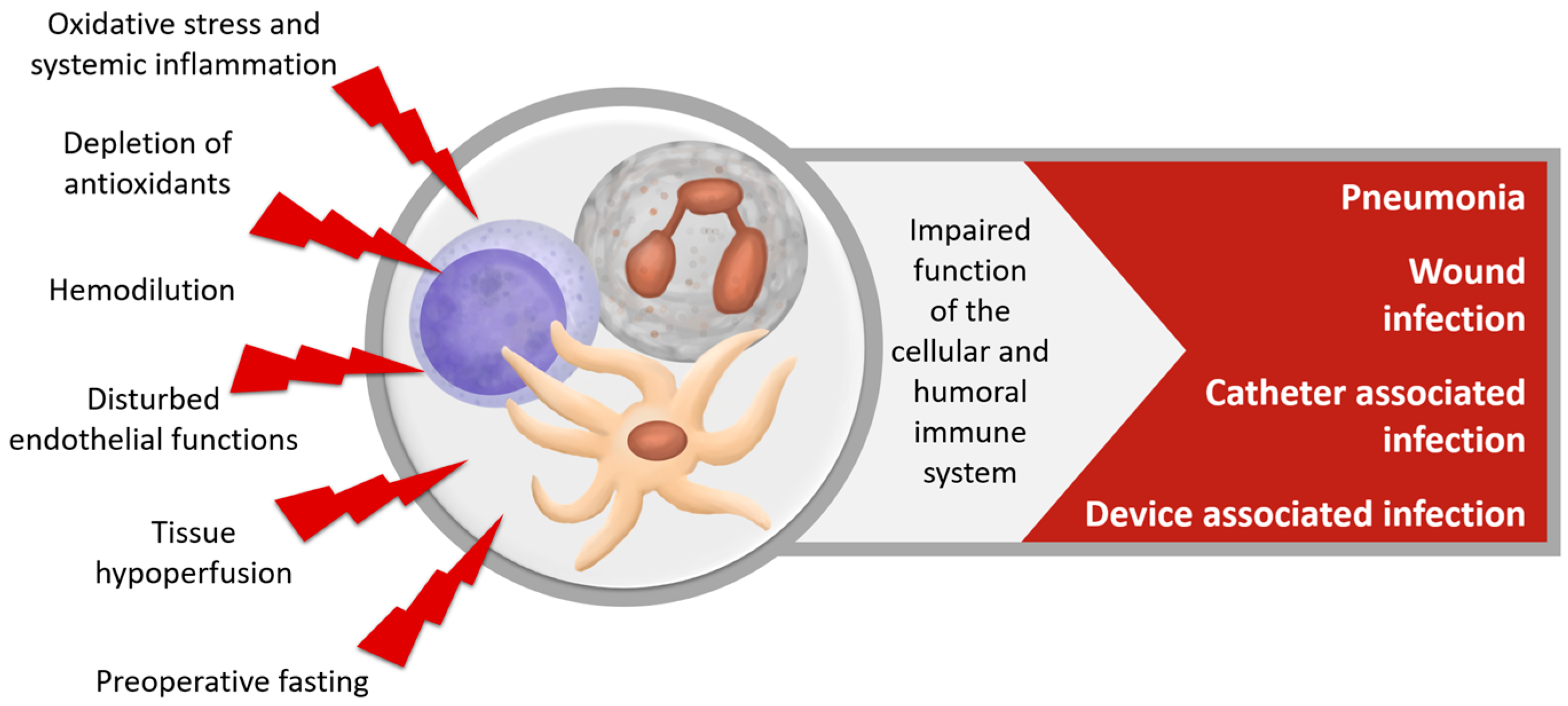
2.7. Immune System
2.7.1. Immune Dysfunction after Cardiac Surgery
2.7.2. Role of Vitamin C in the Immune System
2.7.3. Vitamin C’s Influence on the Immune System in Cardiac Surgery Patients
3. Influence of Vitamin C on the Overall Clinical Outcome of Cardiac Surgery Patients
- Reduced hospital LOS was demonstrated in a systematic 2014 review including 5 RCTs [88], as well as the meta-analyses of Geng [63] and Baker [89] and Shi et al., including 13 trials involving 1956 patients [90]. However, in the meta-analysis by Hu et al. including 8 RCTs and 1060 patients, vitamin C application was not associated with reductions in ICU or hospital-LOS [59].
4. Vitamin C in Combination with other Antioxidant Therapies
5. Practical Approach to Vitamin C Supplementation
5.1. Risks and Side Effects
- Diarrhea and abdominal bloating [27]
- False negative tests for gastrointestinal occult bleeding [27]
- Aggravation of iron overload in patients with hemochromatosis or other diseases requiring frequent blood transfusions, such as thalassemia major and sideroblastic anemia [27]
- Possible adverse pro-oxidative effect in large dosages in case of iron overload [49]
- Possible hyperuricosuria [27]
- Hemolysis in patients with hereditary glucose-6-phosphate dehydrogenase (G6DP) deficiency, when administered in high dosages of >4 g/day [27]
5.2. Application Strategies
5.2.1. Dosing
5.2.2. Timing
5.2.3. Mode of Administration
5.2.4. Monitoring
6. Discussion and Future Directions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| AF | Atrial Fibrillation |
| AKI | Acute Kidney Injury |
| CABG | Coronary Artery Bypass Graft |
| CPB | Cardiopulmonary Bypass |
| CRP | C-reactive Protein |
| DHA | Dehydroascorbate |
| GI | Gastrointestinal |
| ICU | Intensive Care Unit |
| iNOS | Inducible Nitric Oxide Synthetase |
| I/R | Ischemia/Reperfusion |
| i.v. | Intravenous |
| LOS | Length of Stay |
| N.A. | Not Available |
| NFκB | Nuclear Factor kappa-light-chain enhancer of activated B cells |
| nNOS | Neuronal Nitric Oxide Synthetase |
| NO | Nitric Oxide |
| PCA | Perchloric Acid |
| p.o. | Per Os |
| Postop | Before Surgery |
| Preop | After Surgery |
| RCT | Randomized Controlled Trial |
| ROS | Reactive Oxygen Species |
| Sign. | Significantly |
| SIRS | Systemic Inflammatory Response Syndrome |
| SOFA | Sequential Organ Failure Assessment |
| SVCT2 | Sodium-dependent Vitamin C Transporter-2 |
| TNFα | Tumor Necrosis Factor α |
| Vit C | Vitamin C |
| Vit E | Vitamin E/α-Tocopherol |
| WBC | White Blood Count |
References
- Prondzinsky, R.; Knüpfer, A.; Loppnow, H.; Redling, F.; Lehmann, D.W.; Stabenow, I.; Witthaut, R.; Unverzagt, S.; Radke, J.; Zerkowski, H.R.; et al. Surgical trauma affects the proinflammatory status after cardiac surgery to a higher degree than cardiopulmonary bypass. J. Thorac. Cardiovasc. Surg. 2005, 129, 760–766. [Google Scholar] [CrossRef] [PubMed]
- Laffey, J.G.; Boylan, J.F.; Cheng, D.C.H. The systemic inflammatory response to cardiac surgery: Implications for the anesthesiologist. Anesthesiology 2002, 97, 215–252. [Google Scholar] [PubMed]
- Bronicki, R.A.; Hall, M. Cardiopulmonary bypass-induced inflammatory response: Pathophysiology and treatment. Pediatric critical care medicine: A journal of the Society of Critical Care Medicine and the World. Pediatr. Crit. Care Med. 2016, 17, S272–S278. [Google Scholar] [CrossRef] [PubMed]
- Butler, J.; Rocker, G.M.; Westaby, S. Inflammatory Response to Cardiopulmonary Bypass. Ann. Thorac. Surg. 1993, 55, 552–559. [Google Scholar] [CrossRef]
- Chenoweth, D.E.; Cooper, S.W.; Hugli, T.E.; Stewart, R.W.; Blackstone, E.H.; Kirklin, J.W. Complement activation during cardiopulmonary bypass: Evidence for generation of c3a and c5a anaphylatoxins. N. Eng. J. Med. 1981, 304, 497–503. [Google Scholar] [CrossRef] [PubMed]
- Hall, T.S. The pathophysiology of cardiopulmonary bypass: The risks and benefits of hemodilution. Chest 1995, 107, 1125–1133. [Google Scholar] [CrossRef] [PubMed]
- Khabar, K.S.; ElBarbary, M.A.; Khouqeer, F.; Devol, E.; Al-Gain, S.; Al-Halees, Z. Circulating Endotoxin and Cytokines After Cardiopulmonary Bypass: Differential Correlation with Duration of Bypass and Systemic Inflammatory Response/multiple Organ Dysfunction Syndromes. Clin. Immunol. Immunopathol. 1997, 85, 97–103. [Google Scholar] [CrossRef] [PubMed]
- Moore, F.D.; Warner, K.G.; Assousa, S.; Valeri, C.R.; Khuri, S.F. The effects of complement activation during cardiopulmonary bypass. Attenuation by hypothermia, heparin, and hemodilution. Ann. Surg. 1998, 208, 95–103. [Google Scholar] [CrossRef]
- Raja, S.G.; Berg, G.A. Impact of off-pump coronary artery bypass surgery on systemic inflammation: Current best available evidence. J. Card. Surg. 2007, 22, 445–455. [Google Scholar] [CrossRef] [PubMed]
- Hall, R. Identification of inflammatory mediators and their modulation by strategies for the management of the systemic inflammatory response during cardiac surgery. J. Cardiothorac. Vasc. Anesth. 2013, 27, 983–1033. [Google Scholar] [CrossRef] [PubMed]
- Landis, R.C.; Brown, J.R.; Fitzgerald, D.; Likosky, D.S.; Shore-Lesserson, L.; Baker, R.A.; Hammon, J.W. Attenuating the systemic inflammatory response to adult cardiopulmonary bypass: A critical review of the evidence base. J. Extra Corpor. Technol. 2014, 46, 197–211. [Google Scholar] [PubMed]
- Rossaint, J.; Berger, C.; Aken, H.V.; Scheld, H.H.; Zahn, P.K.; Rukosujew, A.; Zarbock, A. Cardiopulmonary bypass during cardiac surgery modulates systemic inflammation by affecting different steps of the leukocyte recruitment cascade. PLoS ONE 2012, 7, e45738. [Google Scholar] [CrossRef] [PubMed]
- Paparella, D.; Yau, T.M.; Young, E. Cardiopulmonary bypass induced inflammation: Pathophysiology and treatment. an update. Eur. J. Cardiothorac. Surg. 2002, 21, 232–244. [Google Scholar] [CrossRef]
- Hickey, E.; Karamlou, T.; You, J.; Ungerleider, R.M. Effects of Circuit Miniaturization in Reducing Inflammatory Response to Infant Cardiopulmonary Bypass by Elimination of Allogeneic Blood Products. Ann. Thorac. Surg. 2006, 81, S2367–S2372. [Google Scholar] [CrossRef] [PubMed]
- Jansen, P.G.; te Velthuis, H.; Bulder, E.R.; Paulus, R.; Scheltinga, M.R.; Eijsman, L.; Wildevuur, C.R. Reduction in prime volume attenuates the hyperdynamic response after cardiopulmonary bypass. Ann. Thorac. Surg. 1995, 60, 544–549. [Google Scholar] [CrossRef]
- Magder, S. Reactive oxygen species: Toxic molecules or spark of life? Crit. Care 2006, 10, 208. [Google Scholar] [CrossRef] [PubMed]
- Roy, J.; Galano, J.-M.; Durand, T.; Le Guennec, J.Y.; Lee, J.C.-Y. Physiological role of reactive oxygen species as promoters of natural defenses. FASEB J. 2017, 31, 3729–3745. [Google Scholar] [CrossRef] [PubMed]
- Roth, E.; Manhart, N.; Wessner, B. Assessing the antioxidative status in critically ill patients. Curr. Opin. Clin. Nutr. Metab. Care 2004, 7, 161–168. [Google Scholar] [CrossRef] [PubMed]
- Koekkoek, W.A.; van Zanten, A.R. Antioxidant vitamins and trace elements in critical illness. Nutr. Clin. Pract. 2016, 31, 457–474. [Google Scholar] [CrossRef] [PubMed]
- Levy, J.H.; Tanaka, K.A. Inflammatory Response to Cardiopulmonary Bypass. Ann. Thorac. Surg. 2003, 75, S715–S720. [Google Scholar] [CrossRef]
- Seghaye, M.C.; Grabitz, R.G.; Duchateau, J.; Busse, S.; Däbritz, S.; Koch, D.; Alzen, G.; Hörnchen, H.; Messmer, B.J.; Von Bernuth, G. Inflammatory reaction and capillary leak syndrome related to cardiopulmonary bypass in neonates undergoing cardiac operations. J. Thorac. Cardiovasc. Surg. 1996, 112, 687–697. [Google Scholar] [CrossRef]
- Crimi, E.; Sica, V.; Slutsky, A.S.; Zhang, H.; Williams-Ignarro, S.; Ignarro, L.J.; Napoli, C. Role of oxidative stress in experimental sepsis and multisystem organ dysfunction. Free Radic. Res. 2006, 40, 665–672. [Google Scholar] [CrossRef] [PubMed]
- Kollef, M.H.; Wragge, T.; Pasque, C. Determinants of mortality and multiorgan dysfunction in cardiac surgery patients requiring prolonged mechanical ventilation. Chest 1995, 107, 1395–1401. [Google Scholar] [CrossRef] [PubMed]
- Suleiman, M.-S.; Zacharowski, K.; Angelini, G.D. Inflammatory response and cardioprotection during open-heart surgery: The importance of anaesthetics. Br. J. Pharmacol. 2008, 153, 21–33. [Google Scholar] [CrossRef] [PubMed]
- Stoppe, C.; McDonald, B.; Benstoem, C.; Elke, G.; Meybohm, P.; Whitlock, R.; Fremes, S.; Fowler, R.; Lamarche, R.; Jiang, X.; et al. Evaluation of persistent organ dysfunction plus death as a novel composite outcome in cardiac surgical patients. J. Cardiothorac. Vasc. Anesth. 2016, 30, 30–38. [Google Scholar] [CrossRef] [PubMed]
- Karlsen, A.; Blomhoff, R.; Gundersen, T.E. Stability of whole blood and plasma ascorbic acid. Eur. J. Clin. Nutr. 2007, 61, 1233–1236. [Google Scholar] [CrossRef] [PubMed]
- Levine, M.; Rumsey, S.C.; Daruwala, R.; Park, J.B.; Wang, Y. Criteria and recommendations for vitamin c intake. JAMA 1999, 281, 1415–1423. [Google Scholar] [CrossRef] [PubMed]
- Carr, A.C.; Frei, B. Toward a new recommended dietary allowance for vitamin C based on antioxidant and health effects in humans. Am. J. Clin. Nutr. 1999, 69, 1086–1107. [Google Scholar] [CrossRef] [PubMed]
- Jacob, R.A.; Burri, B.J. Human metabolism and the requirement for vitamin C. In Vitamin C in Health and Disease; Packer, L., Fuchs, J., Eds.; Marcel Dekker Inc.: New York, NY, USA, 1997; pp. 341–366. [Google Scholar]
- Tsao, C.S. An overview of ascorbic acid chemistry and biochemistry. In Vitamin C in Health and Disease; Packer, L., Fuchs, J., Eds.; Marcel Dekker Inc.: New York, NY, USA, 1997; pp. 25–58. [Google Scholar]
- Huang, Y.; Tang, X.; Xie, W.; Zhou, Y.; Li, D.; Zhou, Y.; Zhu, J.; Yuan, T.; Lai, L.; Pang, D.; Ouyang, H. Vitamin C enhances in vitro and in vivo development of porcine somatic cell nuclear transfer embryos. Biochem. Biophys. Res. Commun. 2011, 411, 397–401. [Google Scholar] [CrossRef] [PubMed]
- Esteban, M.A.; Wang, T.; Qin, B.; Yang, J.; Qin, D.; Cai, J.; Li, W.; Weng, Z.; Chen, J.; Ni, S.; et al. Vitamin C enhances the generation of mouse and human induced pluripotent stem cells. Cell Stem. Cell 2010, 6, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, T.; Lord, B.; Schulze, P.C.; Fryer, R.M.; Sarang, S.S.; Gullans, S.R.; Lee, R.T. Ascorbic acid enhances differentiation of embryonic stem cells into cardiac myocytes. Circulation 2003, 107, 1912–1916. [Google Scholar] [CrossRef] [PubMed]
- Cao, N.; Liu, Z.; Chen, Z.; Wang, J.; Chen, T.; Zhao, S.; Ma, Y.; Qin, L.; Kang, J.; Wei, B.; et al. Ascorbic acid enhances the cardiac differentiation of induced pluripotent stem cells through promoting the proliferation of cardiac progenitor cells. Cell Res. 2012, 22, 219–236. [Google Scholar] [CrossRef] [PubMed]
- Yin, R.; Mao, S.-Q.; Zhao, B.; Chong, Z.; Yang, Y.; Zhao, C.; Zhang, D.; Huang, H.; Gao, J.; Li, Z.; et al. Ascorbic acid enhances tet-mediated 5-methylcytosine oxidation and promotes DNA demethylation in mammals. J. Am. Chem. Soc. 2013, 135, 10396–10403. [Google Scholar] [CrossRef] [PubMed]
- Frei, B.; England, L.; Ames, B.N. Ascorbate is an outstanding antioxidant in human blood plasma. Proc. Natl. Acad. Sci. USA 1989, 86, 6377–6381. [Google Scholar] [CrossRef] [PubMed]
- Frei, B.; Stocker, R.; England, L.; Ames, B.N. Ascorbate: The most effective antioxidant in human blood plasma. Antioxid. Ther. Prev. Med. 1990, 264, 155–163. [Google Scholar]
- Carr, A.C.; Maggini, S. Vitamin C and immune function. Nutrients 2017, 9, 1211. [Google Scholar] [CrossRef] [PubMed]
- Carr, A.C.; Shaw, G.M.; Fowler, A.A.; Natarajan, R. Ascorbate-dependent vasopressor synthesis: A rationale for vitamin C administration in severe sepsis and septic shock? Crit. Care 2015, 19, 418. [Google Scholar] [CrossRef] [PubMed]
- Oudemans-van Straaten, H.M.; Spoelstra-de Man, A.M.; de Waard, M.C. Vitamin C revisited. Crit. Care 2014, 18, 460. [Google Scholar] [CrossRef] [PubMed]
- Berger, M.M.; Oudemans-van Straaten, H.M. Vitamin C supplementation in the critically ill patient. Curr. Opin. Clin. Nutr. Metabol. Care 2015, 18, 193–201. [Google Scholar] [CrossRef] [PubMed]
- Han, M.; Pendem, S.; Teh, S.L.; Sukumaran, D.K.; Wu, F.; Wilson, J.X. Ascorbate protects endothelial barrier function during septic insult: Role of protein phosphatase type 2a. Free Radic. Biol. Med. 2010, 48, 128–135. [Google Scholar] [CrossRef] [PubMed]
- Tyml, K. Vitamin C and microvascular dysfunction in systemic inflammation. Antioxidants 2017, 6, 49. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, T.; Kinoshita, M.; Shinomiya, N.; Hiroi, S.; Sugasawa, H.; Matsushita, Y.; Majima, T.; Saitoh, D.; Seki, S. Pretreatment with ascorbic acid prevents lethal gastrointestinal syndrome in mice receiving a massive amount of radiation. J. Radiat. Res. 2010, 51, 145–156. [Google Scholar] [CrossRef] [PubMed]
- Cecconi, M.; Evans, L.; Levy, M.; Rhodes, A. Sepsis and septic shock. Lancet 2018, 392, 75–87. [Google Scholar] [CrossRef]
- Carr, A.C.; Rosengrave, P.C.; Bayer, S.; Chambers, S.; Mehrtens, J.; Shaw, G.M. Hypovitaminosis C and vitamin C deficiency in critically ill patients despite recommended enteral and parenteral intakes. Crit. Care 2017, 21, 300. [Google Scholar] [CrossRef] [PubMed]
- Wilson, J.X. Evaluation of vitamin C for adjuvant sepsis therapy. Antioxid. Redox Signal. 2013, 19, 2129–2140. [Google Scholar] [CrossRef] [PubMed]
- Long, C.L.; Maull, K.I.; Krishnan, R.S.; Laws, H.L.; Geiger, J.W.; Borghesi, L.; Franks, W.; Lawson, T.C.; Sauberlich, H.E. Ascorbic acid dynamics in the seriously ill and injured. J. Surg. Res. 2003, 109, 144–148. [Google Scholar] [CrossRef]
- Angelique, M.E.; Spoelstra-de Man, A.M.; Elbers, P.W.; Oudemans-van Straaten, H.M. Making sense of early high-dose intravenous vitamin C in ischemia/reperfusion injury. Crit. Crit. Care 2018, 22, 70. [Google Scholar]
- Borrelli, E.; Roux-Lombard, P.; Grau, G.E.; Girardin, E.; Ricou, B.; Dayer, J.M.; Suter, P.M. Plasma concentrations of cytokines, their soluble receptors, and antioxidant vitamins can predict the development of multiple organ failure in patients at risk. Crit. Care Med. 1996, 24, 392–397. [Google Scholar] [CrossRef] [PubMed]
- Fowler, A.A.; Syed, A.A.; Knowlson, S.; Sculthorpe, R.; Farthing, D.; De Wilde, C.; Farthing, C.A.; Larus, T.L.; Martin, E.; Brophy, D.F.; et al. Phase I safety trial of intravenous ascorbic acid in patients with severe sepsis. J. Transl. Med. 2014, 12, 32. [Google Scholar] [CrossRef] [PubMed]
- Zabet, M.H.; Mohammadi, M.; Ramezani, M.; Khalili, H. Effect of high-dose ascorbic acid on vasopressor’s requirement in septic shock. J. Res. Pharm. Pract. 2016, 5, 94–100. [Google Scholar] [PubMed]
- Nathens, A.B.; Neff, M.J.; Jurkovich, G.J.; Klotz, P.; Farver, K.; Ruzinski, J.T.; Radella, F.; Garcia, I.; Maier, R.V. Randomized, prospective trial of antioxidant supplementation in critically ill surgical patients. Ann. Surg. 2002, 236, 814–822. [Google Scholar] [CrossRef] [PubMed]
- Kahn, S.A.; Beers, R.J.; Lentz, C.W. Resuscitation after severe burn injury using high-dose ascorbic acid: A retrospective review. J. Burn. Care Res. 2011, 32, 110–117. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, H.; Matsuda, T.; Miyagantani, Y.; Yukioka, T.; Matsuda, H.; Shimazaki, S. Reduction of resuscitation fluid volumes in severely burned patients using ascorbic acid administration: A randomized, prospective study. Arch. Surg. 2000, 135, 326–331. [Google Scholar] [CrossRef] [PubMed]
- Bouras, E.; Chourdakis, M.; Grammatikopoulou, M.G.; Heyland, D.K. Nutrition therapy practices applied on severe burn patients: Results from the ins 2014 survey. Clin. Nutr. ESPEN 2018, 24, 182. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, A.; Shah, S.A.; Badshah, H.; Kim, M.J.; Ali, T.; Yoon, G.H.; Kim, T.H.; Abid, N.B.; Rehman, S.U.; Khan, S.; et al. Neuroprotection by Vitamin C Against Ethanol-Induced Neuroinflammation Associated Neurodegeneration in the Developing Rat Brain. CNS Neurol. Disord. Drug Targets 2016, 15, 360–370. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Agus, D.B.; Winfree, C.J.; Kiss, S.; Mack, W.J.; McTaggart, R.A.; Choudhri, T.F.; Kim, L.J.; Mocco, J.; Pinsky, D.J.; et al. Dehydroascorbic acid, a blood-brain barrier transportable form of vitamin C, mediates potent cerebroprotection in experimental stroke. Proc. Natl. Acad. Sci. USA 2001, 98, 11720–11724. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.; Yuan, L.; Wang, H.; Li, C.; Cai, J.; Hu, Y.; Ma, C. Efficacy and safety of vitamin C for atrial fibrillation after cardiac surgery: A meta-analysis with trial sequential analysis of randomized controlled trials. Int. J. Surg. 2017, 37, 58–64. [Google Scholar] [CrossRef] [PubMed]
- Polymeropoulos, E.; Bagos, P.; Papadimitriou, M.; Rizos, I.; Patsouris, E.; Τoumpoulis, I. Vitamin C for the Prevention of Postoperative Atrial Fibrillation after Cardiac Surgery: A Meta-Analysis. Adv. Pharm. Bull. 2016, 6, 243–250. [Google Scholar] [CrossRef] [PubMed]
- May, J.M.; Harrison, F.E. Role of vitamin C in the function of the vascular endothelium. Antioxid. Redox Signal. 2013, 19, 2068–2083. [Google Scholar] [CrossRef] [PubMed]
- Ashor, A.W.; Lara, J.; Mathers, J.C.; Siervo, M. Effect of vitamin C on endothelial function in health and disease: A systematic review and meta-analysis of randomised controlled trials. Atherosclerosis 2014, 235, 9–20. [Google Scholar] [CrossRef] [PubMed]
- Geng, J.; Qian, J.; Si, W.; Cheng, H.; Ji, F.; Shen, Z. The clinical benefits of perioperative antioxidant vitamin therapy in patients undergoing cardiac surgery: A meta-analysis. Interact. Cardiovasc. Thorac. Surg. 2017, 5, 966–974. [Google Scholar] [CrossRef] [PubMed]
- Al-Asmari, A.M.; Khan, A.Q.; Al-Qasim, A.M.; Al-Yousef, Y. Ascorbic acid attenuates antineoplastic drug 5-fluorouracil induced gastrointestinal toxicity in rats by modulating the expression of inflammatory mediators. Toxicol. Rep. 2015, 2, 908–916. [Google Scholar] [CrossRef] [PubMed]
- Eagle, K.A.; Guyton, R.A.; Davidoff, R.; Edwards, F.H.; Ewy, G.A.; Gardner, T.J.; Hart, J.C.; Herrmann, H.C.; Hillis, L.D.; Hutter, A.M., Jr.; et al. ACC/AHA 2004 guideline update for coronary artery bypass graft surgery: A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Committee to Update the 1999 Guidelines for Coronary Artery Bypass Graft Surgery). Circulation 2004, 110, e340–e437. [Google Scholar] [PubMed]
- Biancari, F.; Tauriainen, T.; Perrotti, A.; Dalén, M.; Faggian, G.; Franzese, I.; Chocron, S.; Ruggieri, V.G.; Bounader, K.; Gulbins, H.; et al. Bleeding, transfusion and the risk of stroke after coronary surgery: A prospective cohort study of 2357 patients. Int. J. Surg. 2016, 32, 50–57. [Google Scholar] [CrossRef] [PubMed]
- Cropsey, C.; Kennedy, J.; Han, J.; Pandharipande, P. Cognitive dysfunction, delirium, and stroke in cardiac surgery patients. Semin. Cardiothorac. Vasc. Anesth. 2015, 19, 309–317. [Google Scholar] [CrossRef] [PubMed]
- Knipp, S.C.; Weimar, C.; Schlamann, M.; Schweter, S.; Wendt, D.; Thielmann, M.; Benedik, J.; Jakob, H. Early and long-term cognitive outcome after conventional cardiac valve surgery. Interact. Cardiovasc. Thorac. Surg. 2017, 24, 534–540. [Google Scholar] [CrossRef] [PubMed]
- Selnes, O.A.; Gottesman, R.F.; Grega, M.A.; Baumgartner, W.A.; Zeger, S.L.; McKhann, G.M. Cognitive and neurologic outcomes after coronary-artery bypass surgery. N. Engl. J. Med. 2012, 366, 250–257. [Google Scholar] [CrossRef] [PubMed]
- Van Harten, A.E.; Scheeren, T.W.; Absalom, A.R. A review of postoperative cognitive dysfunction and neuroinflammation associated with cardiac surgery and anaesthesia. Anaesthesia 2012, 67, 280–293. [Google Scholar] [CrossRef] [PubMed]
- Mangusan, R.F.; Hooper, V.; Denslow, S.A.; Travis, L. Outcomes associated with postoperative delirium after cardiac surgery. Am. J. Crit. Care 2015, 24, 156–163. [Google Scholar] [CrossRef] [PubMed]
- Gess, B.; Lohmann, C.; Halfter, H.; Young, P. Sodium-dependent vitamin C transporter 2 (SVCT2) is necessary for the uptake of l-ascorbic acid into Schwann cells. Glia 2010, 58, 287–299. [Google Scholar] [CrossRef] [PubMed]
- Harrison, F.E.; May, J.M. Vitamin C function in the brain: Vital role of the ascorbate transporter SVCT2. Free Radic. Biol. Med. 2009, 46, 719–730. [Google Scholar] [CrossRef] [PubMed]
- Eldridge, C.F.; Bunge, M.B.; Bunge, R.P.; Wood, P.M. Differentiation of axon-related schwann cells in vitro. I. ascorbic acid regulates basal lamina assembly and myelin formation. J. Cell Biol. 1987, 105, 1023–1034. [Google Scholar] [CrossRef] [PubMed]
- Henry, P.T.; Chandy, M.J. Effect of ascorbic acid on infarct size in experimental focal cerebral ischaemia and reperfusion in a primate model. Acta Neurochir. 1998, 140, 977–980. [Google Scholar] [CrossRef] [PubMed]
- Lagowska-Lenard, M.; Stelmasiak, Z.; Bartosik-Psujek, H. Influence of vitamin C on markers of oxidative stress in the earliest period of ischemic stroke. Pharmacol. Rep. 2010, 62, 751–756. [Google Scholar] [CrossRef]
- Bonnefont-Rousselot, D.; Mahmoudi, A.; Mougenot, N.; Varoquaux, O.; Le Nahour, G.; Fouret, P.; Lechat, P. Catecholamine effects on cardiac remodelling, oxidative stress and fibrosis in experimental heart failure. Redox Rep. 2002, 7, 145–151. [Google Scholar] [CrossRef] [PubMed]
- Ellenberger, C.; Sologashvili, T.; Cikirikcioglu, M.; Verdon, G.; Diaper, J.; Cassina, T.; Licker, M. Risk factors of postcardiotomy ventricular dysfunction in moderate-to-high risk patients undergoing open-heart surgery. Ann. Card. Anaesth. 2017, 20, 287–296. [Google Scholar] [PubMed]
- Lomivorotov, V.V.; Efremov, S.M.; Kirov, M.Y.; Fominskiy, E.V.; Karaskov, A.M. Low-cardiac-output syndrome after cardiac surgery. J. Cardiothorac. Vasc. Anesth. 2017, 31, 291–308. [Google Scholar] [CrossRef] [PubMed]
- Ding, W.; Ji, Q.; Shi, Y.; Ma, R. Predictors of low cardiac output syndrome after isolated coronary artery bypass grafting. Int. Heart J. 2015, 56, 144–149. [Google Scholar] [CrossRef] [PubMed]
- Epting, C.L.; McBride, M.E.; Wald, E.L.; Costello, J.M. Pathophysiology of post-operative low cardiac output syndrome. Curr. Vasc. Pharmacol. 2016, 14, 14–23. [Google Scholar] [CrossRef] [PubMed]
- Peretto, G.; Durante, A.; Limite, L.R.; Cianflone, D. Postoperative arrhythmias after cardiac surgery: Incidence, risk factors, and therapeutic management. Cardiol. Res. Pract. 2014, 2014, 615987. [Google Scholar] [CrossRef] [PubMed]
- Saran, V.; Sharma, V.; Wambolt, R.; Yuen, V.G.; Allard, M.; McNeill, J.H. Combined metoprolol and ascorbic acid treatment prevents intrinsic damage to the heart during diabetic cardiomyopathy. Can. J. Physiol. Pharmacol. 2014, 92, 827–837. [Google Scholar] [CrossRef] [PubMed]
- Hsu, C.C.; Wang, J.J. l-ascorbic acid and alpha-tocopherol attenuates liver ischemia-reperfusion induced of cardiac function impairment. Transplant. Proc. 2012, 44, 933–936. [Google Scholar] [CrossRef] [PubMed]
- Kremer, T.; Harenberg, P.; Hernekamp, F.; Riedel, K.; Gebhardt, M.M.; Germann, G.; Heitmann, C.; Walther, A. High-dose vitamin C treatment reduces capillary leakage after burn plasma transfer in rats. J. Burn Care Res. 2010, 31, 470–479. [Google Scholar] [CrossRef] [PubMed]
- Dingchao, H.; Zhiduan, Q.; Liye, H.; Xiaodong, F. The protective effects of high-dose ascorbic acid on myocardium against reperfusion injury during and after cardiopulmonary bypass. Thorac. Cardiovasc. Surg. 1994, 42, 276–278. [Google Scholar] [CrossRef] [PubMed]
- Rezk, M.E. Role of ascorbic acid in reduction of the incidence of the atrial fibrillation in patients under b-blocker and undergoing coronary artery bypass graft operation in early post-operative period. J. Egypt. Soc. Cardio Thorac. Surg. 2017, 25, 198–203. [Google Scholar]
- Ali-Hassan-Sayegh, S.; Mirhosseini, S.J.; Rezaeisadrabadi, M.; Dehghan, H.R.; Sedaghat-Hamedani, F.; Kayvanpour, E.; Popov, A.F.; Liakopoulos, O.J. Liakopoulos. Antioxidant supplementations for prevention of atrial fibrillation after cardiac surgery: An updated comprehensive systematic review and meta-analysis of 23 randomized controlled trials. Interact. Cardiovasc. Thorac. Surg. 2014, 18, 646–654. [Google Scholar] [CrossRef] [PubMed]
- Baker, W.L.; Coleman, C.I. Meta-analysis of ascorbic acid for prevention of postoperative atrial fibrillation after cardiac surgery. Am. J. Health Syst. Pharm. 2016, 73, 2056–2066. [Google Scholar] [CrossRef] [PubMed]
- Shi, R.; Li, Z.H.; Chen, D.; Wu, Q.C.; Zhou, X.L.; Tie, H.T. Sole and combined vitamin C supplementation can prevent postoperative atrial fibrillation after cardiac surgery: A systematic review and meta-analysis of randomized controlled trials. Clin. Cardiol. 2018, 41, 871–878. [Google Scholar] [CrossRef] [PubMed]
- Hemilä, H.; Suonsyrjae, T. Vitamin C for preventing atrial fibrillation in high risk patients: A systematic review and meta-analysis. BMC Cardiovasc. Dis. 2017. [Google Scholar] [CrossRef] [PubMed]
- Hemilä, H. Publication bias in meta-analysis of ascorbic acid for postoperative atrial fibrillation. Am. J. Health Syst. Pharm. 2017, 74, 372–373. [Google Scholar] [CrossRef] [PubMed]
- García-Delgado, M.; Navarrete-Sánchez, I.; Colmenero, M. Preventing and managing perioperative pulmonary complications following cardiac surgery. Curr. Opin. Anaesthesiol. 2014, 27, 146–152. [Google Scholar] [CrossRef] [PubMed]
- Bedreag, O.H.; Rogobete, A.F.; Sarandan, M.; Cradigati, A.C.; Papurica, M.; Dumbuleu, M.C.; Chira, A.M.; Rosu, O.M.; Sandesc, D. Oxidative stress in severe pulmonary trauma in critical ill patients. antioxidant therapy in patients with multiple trauma—A review. Anaesthesiol. Intensive Ther. 2015, 47, 351–359. [Google Scholar] [CrossRef] [PubMed]
- Lang, J.D.; McArdle, P.J.; O’Reilly, P.J.; Matalon, S. Oxidant-antioxidant balance in acute lung injury. Chest 2002, 122 (Suppl. 6), 314S–320S. [Google Scholar] [CrossRef] [PubMed]
- Herridge, M.S.; Tansey, C.M.; Matté, A.; Tomlinson, G.; Diaz-Granados, N.; Cooper, A.; Guest, C.B.; Mazer, C.D.; Mehta, S.; Stewart, T.E.; et al. Functional disability 5 years after acute respiratory distress syndrome. N. Engl. J. Med. 2011, 364, 1293–1304. [Google Scholar] [CrossRef] [PubMed]
- Kogan, A.; Segel, M.; Levin, S.; Sternik, L.; Raanani, E. Incidence of ards following cardiac surgery: Comparison between american-european consensus conference definition and berlin definition. J. Cardiothorac. Vasc. Anesth. 2017, 31, S79–S80. [Google Scholar] [CrossRef]
- Ng, C.S.; Wan, S.; Yim, A.P.; Arifi, A.A. Pulmonary dysfunction after cardiac surgery. CHEST J. 2002, 121, 1269–1277. [Google Scholar] [CrossRef]
- Stephens, R.S.; Shah, A.S.; Whitman, G.J.R. Lung injury and acute respiratory distress syndrome after cardiac surgery. Ann. Thorac. Surg. 2013, 95, 1122–1129. [Google Scholar] [CrossRef] [PubMed]
- Wynne, R.; Botti, M. Postoperative pulmonary dysfunction in adults after cardiac surgery with cardiopulmonary bypass: Clinical significance and implications for practice. Am. J. Crit. Care 2004, 13, 384–393. [Google Scholar] [PubMed]
- Fisher, B.J.; Kraskauskas, D.; Martin, E.J.; Farkas, D.; Wegelin, J.A.; Brophy, D.; Ward, K.R.; Voelkel, N.F.; Fowler, A.A.; Natarajan, R. Mechanisms of attenuation of abdominal sepsis induced acute lung injury by ascorbic acid. Am. J. Physiol. Lung Cell Mol. Physiol. 2012, 303, L20–L32. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Maeda, N.; Beck, M.A. Vitamin C deficiency increases the lung pathology of influenza virus-infected gulo-/- mice. J. Nutr. 2006, 136, 2611–2616. [Google Scholar] [CrossRef] [PubMed]
- Baltalarli, A.; Ozcan, V.; Ferda, B.; Aybek, H.; Sacar, M.; Onem, G.; Goksin, I.; Demir, S.; Zafer, T. Ascorbic Acid (Vitamin C) and Iloprost Attenuate the Lung Injury Caused by Ischemia/Reperfusion of the Lower Extremities of Rats. Ann. Vasc. Surg. 2006, 20, 49–55. [Google Scholar] [CrossRef] [PubMed]
- Jin, X.; Su, R.; Li, R.; Song, L.; Chen, M.; Cheng, L.; Li, Z. Amelioration of particulate matter-induced oxidative damage by vitamin C and quercetin in human bronchial epithelial cells. Chemosphere 2016, 144, 459–466. [Google Scholar] [CrossRef] [PubMed]
- Gupta, I.; Ganguly, S.; Rozanas, C.R.; Stuehr, D.J.; Panda, K. Ascorbate attenuates pulmonary emphysema by inhibiting tobacco smoke and Rtp801-triggered lung protein modification and proteolysis. Proc. Natl. Acad. Sci. USA 2016, 113, E4208–E4217. [Google Scholar] [CrossRef] [PubMed]
- Rice, T.W.; Wheeler, A.P.; Thompson, B.T.; de Boisblanc, B.P.; Steingrub, J.; Rock, P.; Hudson, L.; Hough, C.; Neff, M.; Sims, K. Enteral omega-3 fatty acid, gamma-linolenic acid, and antioxidant supplementation in acute lung injury. JAMA 2011, 306, 1574–1581. [Google Scholar] [CrossRef] [PubMed]
- Gadek, J.E.; DeMichele, S.J.; Karlstad, M.D.; Pacht, E.R.; Donahoe, M.; Albertson, T.E.; Van Hoozen, C.; Wennberg, A.K.; Nelson, J.L.; Noursalehi, M. Effect of enteral feeding with eicosapentaenoic acid, gamma-linolenic acid, and antioxidants in patients with acute respiratory distress syndrome. enteral nutrition in ards study group. Crit. Care Med. 1999, 27, 1409–1420. [Google Scholar] [CrossRef] [PubMed]
- Corredor, C.; Thomson, R.; Al-Subaie, N. Long-term consequences of acute kidney injury after cardiac surgery: A systematic review and meta-analysis. J. Cardiothorac. Vasc. Anesth. 2016, 30, 69–75. [Google Scholar] [CrossRef] [PubMed]
- Shi, Q.; Hong, L.; Mu, X.; Zhang, C.; Chen, X. Meta-analysis for outcomes of acute kidney injury after cardiac surgery. Medicine 2016, 95, e5558. [Google Scholar] [CrossRef] [PubMed]
- Ferreiro, A.; Lombardi, R. Acute kidney injury after cardiac surgery is associated with mid-term but not long-term mortality: A cohort-based study. PLoS ONE 2017, 12, e0181158. [Google Scholar] [CrossRef] [PubMed]
- Hobson, C.E.; Yavas, S.; Segal, M.S.; Schold, J.D.; Tribble, C.G.; Layon, A.J.; Bihorac, A. Acute kidney injury is associated with increased long-term mortality after cardiothoracic surgery. Circulation 2009, 119, 2444–2453. [Google Scholar] [CrossRef] [PubMed]
- O’Neal, J.B.; Shaw, A.D.; Billings, F.T. Acute kidney injury following cardiac surgery: Current understanding and future directions. Crit. Care 2016, 20, 187. [Google Scholar] [CrossRef] [PubMed]
- Pickering, J.W.; James, M.T.; Palmer, S.C. Acute kidney injury and prognosis after cardiopulmonary bypass: A meta-analysis of cohort studies. Am. J. Kidney Dis. 2015, 65, 283–293. [Google Scholar] [CrossRef] [PubMed]
- Provenchère, S.; Plantefève, G.; Hufnagel, G.; Vicaut, E.; De Vaumas, C.; Lecharny, J.B.; Depoix, J.P.; Vrtovsnik, F.; Desmonts, J.M.; Philip, I. Renal dysfunction after cardiac surgery with normothermic cardiopulmonary bypass: Incidence, risk factors, and effect on clinical outcome. Anesth. Anal. 2003, 96, 1258–1264. [Google Scholar] [CrossRef]
- Spargias, K.; Alexopoulos, E.; Kyrzopoulos, S.; Iokovis, P.; Greenwood, D.C.; Manginas, A.; Voudris, V.; Pavlides, G.; Buller, C.E.; Kremastinos, D.; et al. Ascorbic acid prevents contrast-mediated nephropathy in patients with renal dysfunction undergoing coronary angiography or intervention. Circulation 2004, 110, 2837–2842. [Google Scholar] [CrossRef] [PubMed]
- Sadat, U.; Usman, A.; Gillard, J.H.; Boyle, J.R. Does ascorbic acid protect against contrast-induced acute kidney injury in patients undergoing coronary angiography: A systematic review with meta-analysis of randomized, controlled trials. J. Am. Coll. Cardiol. 2013, 62, 2167–2175. [Google Scholar] [CrossRef] [PubMed]
- Gurm, H.; Sheta, M.A.; Nivera, N.; Tunkel, A. Vitamin C-induced oxalate nephropathy: A case report. J. Community Hosp. Intern. Med. Perspect. 2012, 2. [Google Scholar] [CrossRef] [PubMed]
- Sunkara, V.; Pelkowski, T.D.; Dreyfus, D.; Satoskar, A. Acute Kidney Disease Due to Excessive Vitamin C Ingestion and Remote Roux-en-Y Gastric Bypass Surgery Superimposed on CKD. Am. J. Kidney Dis. 2015, 66, 721–724. [Google Scholar] [CrossRef] [PubMed]
- Antonic, M. Effect of ascorbic acid on postoperative acute kidney injury in coronary artery bypass graft patients: A pilot study. Heart. Surg. Forum 2017, 20, E214–E218. [Google Scholar] [CrossRef] [PubMed]
- Sever, K.; Ozbek, C.; Goktas, B.; Bas, S.; Ugurlucan, M.; Mansuroglu, D. Gastrointestinal complications after open heart surgery: Incidence and determinants of risk factors. Angiology 2014, 65, 425–429. [Google Scholar] [CrossRef] [PubMed]
- Berger, M.M.; Berger-Gryllaki, M.; Wiesel, P.H.; Revelly, J.P.; Hurni, M.; Cayeux, C.; Tappy, L.; Chioléro, R. Intestinal absorption in patients after cardiac surgery. Crit. Care Med. 2000, 28, 2217–2223. [Google Scholar] [CrossRef] [PubMed]
- Goldhill, D.R.; Whelpton, R.; Winyard, J.A.; Wilkinson, K.A. Gastric emptying in patients the day after cardiac surgery. Anaesthesia 1995, 50, 122–125. [Google Scholar] [CrossRef] [PubMed]
- Chaudhry, R.; Zaki, J.; Wegner, R.; Pednekar, G.; Tse, A.; Sheinbaum, R.; Williams, G.W. Gastrointestinal complications after cardiac surgery: A nationwide population-based analysis of morbidity and mortality predictors. J. Cardiothorac. Vasc. Anesth. 2017, 31, 1268–1274. [Google Scholar] [CrossRef] [PubMed]
- Cresci, G.; Hummell, A.C.; Raheem, S.A.; Cole, D. Nutrition intervention in the critically ill cardiothoracic patient. Nutr. Clin. Pract. 2012, 27, 323–334. [Google Scholar] [CrossRef] [PubMed]
- Viana, F.F.; Chen, Y.; Almeida, A.A.; Baxter, H.D.; Cochrane, A.D.; Smith, J.A. Gastrointestinal complications after cardiac surgery: 10-year experience of a single Australian centre. ANZ J. Surg. 2013, 83, 651–656. [Google Scholar] [CrossRef] [PubMed]
- Bjelakovic, G.; Nikolova, D.; Simonetti, R.G.; Gluud, C. Antioxidant supplements for preventing gastrointestinal cancers. Cochrane Database Syst. Rev. 2008, 16, CD004183. [Google Scholar] [CrossRef] [PubMed]
- Freedman, J.E. Oxidative stress and platelets. Arterioscler. Thromb. Vasc. Biol. 2008, 28, s11–s16. [Google Scholar] [CrossRef] [PubMed]
- Levi, M. Platelets at a crossroad of pathogenic pathways in sepsis. J. Thromb. Haemost. 2004, 2, 2094–2095. [Google Scholar] [CrossRef] [PubMed]
- Dyke, C.; Aronson, S.; Dietrich, W.; Hofmann, A.; Karkouti, K.; Levi, M.; Murphy, G.J.; Sellke, F.W.; Shore-Lesserson, L.; von Heymann, C. Universal definition of perioperative bleeding in adult cardiac surgery. J. Thorac. Cardiovasc. Surg. 2013, 147, 1458–1463. [Google Scholar] [CrossRef] [PubMed]
- Kinnunen, E.M.; De Feo, M.; Reichart, D.; Tauriainen, T.; Gatti, G.; Onorati, F.; Maschietto, L.; Bancone, C.; Fiorentino, F.; Chocron, S. Incidence and prognostic impact of bleeding and transfusion after coronary surgery in low-risk patients. Transfusion 2017, 57, 178–186. [Google Scholar] [CrossRef] [PubMed]
- Ranucci, M.; Baryshnikova, E.; Castelvecchio, S.; Pelissero, G. Surgical and Clinical Outcome Research (SCORE) Group. Major bleeding, transfusions, and anemia: The deadly triad of cardiac surgery. Ann. Thorac. Surg. 2013, 96, 478–485. [Google Scholar] [CrossRef] [PubMed]
- Gielen, C.; Dekkers, O.; Stijnen, T.; Schoones, J.; Brand, A.; Klautz, R.; Eikenboom, J. The effects of pre- and postoperative fibrinogen levels on blood loss after cardiac surgery: A systematic review and meta-analysis. Interact. Cardiovasc. Thorac. Surg. 2014, 18, 292–298. [Google Scholar] [CrossRef] [PubMed]
- Dayton, P.G.; Weiner, M. Ascorbic acid and blood coagulation. Ann. N. Y. Acad. Sci. 1961, 92, 302–306. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.D.; Ye, B.Q.; Zheng, S.X.; Wang, J.T.; Wang, J.G.; Chen, M.; Liu, J.G.; Pei, X.H.; Wang, L.J.; Lin, Z.X.; et al. NF-kappaB transcription factor p50 critically regulates tissue factor in deep vein thrombosis. J. Biol. Chem. 2009, 284, 4473–4483. [Google Scholar] [CrossRef] [PubMed]
- Esmon, C.T. The interactions between inflammation and coagulation. Br. J. Haematol. 2005, 131, 417–430. [Google Scholar] [CrossRef] [PubMed]
- Parahuleva, M.S.; Jung, J.; Burgazli, M.; Erdogan, A.; Parviz, B.; Hölschermann, H. Vitamin C suppresses lipopolysaccharide-induced procoagulant response of human monocyte-derived macrophages. Eur. Rev. Med. Pharmacol. Sci. 2016, 20, 2174–2182. [Google Scholar] [PubMed]
- Tousoulis, D.; Antoniades, C.; Tountas, C.; Bosinakou, E.; Kotsopoulou, M.; Toutouzas, P.; Stefanadis, C. Vitamin C affects thrombosis/fibrinolysis system and reactive hyperemia in patients with type 2 diabetes and coronary artery disease. Diabetes Care 2003, 26, 2749–2753. [Google Scholar] [CrossRef] [PubMed]
- Kaehler, J.; Koeke, K.; Karstens, M.; Schneppenheim, R.; Meinertz, T.; Heitzer, T. Impaired capacity for acute endogenous fibrinolysis in smokers is restored by ascorbic acid. Free Radic. Biol. Med. 2008, 44, 315–321. [Google Scholar] [CrossRef] [PubMed]
- Cooke, A.R. The role of acid in the pathogenesis of aspirin-induced gastrointestinal erosions and hemorrhage. Am. J. Dig. Dis. 1973, 18, 225–237. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.; Bae, O.N.; Koh, S.H.; Kang, S.; Lim, K.M.; Noh, J.Y.; Shin, S.; Kim, I.; Chung, J.H. High-Dose Vitamin C Injection to Cancer Patients May Promote Thrombosis Through Procoagulant Activation of Erythrocytes. Toxicol. Sci. 2015, 147, 350–359. [Google Scholar] [CrossRef] [PubMed]
- Sadeghpour, A.; Alizadehasl, A.; Kyavar, M.; Sadeghi, T.; Moludi, J.; Gholizadeh, F.; Totonchi, Z.; Ghadrdoost, B. Impact of vitamin C supplementation on post-cardiac surgery ICU and hospital length of stay. Anesth. Pain Med. 2015, 5, e25337. [Google Scholar] [CrossRef] [PubMed]
- Gelijns, A.C.; Moskowitz, A.J.; Acker, M.A.; Argenziano, M.; Geller, N.L.; Puskas, J.D.; Perrault, L.P.; Smith, P.K.; Kron, I.L.; Michler, R.E.; et al. Management practices and major infections after cardiac surgery. J. Am. Coll. Cardiol. 2014, 64, 372–381. [Google Scholar] [CrossRef] [PubMed]
- Cove, M.E.; Spelman, D.W.; MacLaren, G. Infectious complications of cardiac surgery: A clinical review. J. Cardiothorac. Vasc. Anesth. 2012, 26, 1094–1100. [Google Scholar] [CrossRef] [PubMed]
- Wendler, O.; Baghai, M. Infections post-cardiac surgery: New information during challenging times. J. Am. Coll. Cardiol. 2014, 64, 382–384. [Google Scholar] [CrossRef] [PubMed]
- Benjamin, E.J.; Blaha, M.J.; Chiuve, S.E.; Cushman, M.; Das, S.R.; Deo, R.; de Ferranti, S.D.; Floyd, J.; Fornage, M.; Gillespie, C.; et al. Heart disease and stroke statistics—2017 update: A report from the American heart association. Circulation 2017, 135, e146–e603. [Google Scholar] [CrossRef] [PubMed]
- Kirklin, J.K.; Pagani, F.D.; Kormos, R.L.; Stevenson, L.W.; Blume, E.D.; Myers, S.L.; Miller, M.A.; Baldwin, J.T.; Young, J.B.; Naftel, D.C. Eighth annual INTERMACS report: Special focus on framing the impact of adverse events. J. Heart Lung Transplant. 2017, 36, 1080–1086. [Google Scholar] [CrossRef] [PubMed]
- Lampropulos, J.F.; Kim, N.; Wang, Y.; Desai, M.M.; Barreto-Filho, J.A.; Dodson, J.A.; Dries, D.L.; Mangi, A.A.; Krumholz, H.M. Trends in left ventricular assist device use and outcomes among medicare beneficiaries, 2004–2011. Open Heart 2014, 1, e000109. [Google Scholar] [CrossRef] [PubMed]
- Bouza, E.; Hortal, J.; Muñoz, P.; Pascau, J.; Pérez, M.J.; Hiesmayr, M.; European Study Group on Nosocomial Infections; European Workgroup of Cardiothoracic Intensivists. Postoperative infections after major heart surgery and prevention of ventilator-associated pneumonia: A one-day European prevalence study (ESGNI-008). J. Hosp. Infect. 2006, 64, 224–230. [Google Scholar] [CrossRef] [PubMed]
- He, S.; Chen, B.; Li, W.; Yan, J.; Chen, L.; Wang, X.; Xiao, Y. Ventilator-associated pneumonia after cardiac surgery: a meta-analysis and systematic review. J. Thorac. Cardiovasc. Surg. 2014, 148, 3148–3155.e5. [Google Scholar] [CrossRef] [PubMed]
- Hortal, J.; Muñoz, P.; Cuerpo, G.; Litvan, H.; Rosseel, P.M.; Bouza, E.; European Study Group on Nosocomial Infections; European Workgroup of Cardiothoracic Intensivists. Ventilator-associated pneumonia in patients undergoing major heart surgery: An incidence study in Europe. Crit. Care 2009, 13, R80. [Google Scholar] [CrossRef] [PubMed]
- Ibañez, J.; Riera, M.; Amezaga, R.; Herrero, J.; Colomar, A.; Campillo-Artero, C.; de Ibarra, J.I.; Bonnin, O. Long-term mortality after pneumonia in cardiac surgery patients: A propensity-matched analysis. J. Intensive Care Med. 2016, 31, 34–40. [Google Scholar] [CrossRef] [PubMed]
- Sheng, W.; Xing, Q.S.; Hou, W.M.; Sun, L.; Niu, Z.Z.; Lin, M.S.; Chi, Y.F. Independent risk factors for ventilator-associated pneumonia after cardiac surgery. J. Investig. Surg. 2014, 27, 256–261. [Google Scholar] [CrossRef] [PubMed]
- Hunt, C.; Chakravorty, N.K.; Annan, G.; Habibzadeh, N.; Schorah, C.J. The clinical effects of vitamin C supplementation in elderly hospitalised patients with acute respiratory infections. Int. J. Vitam. Nutr. Res. 1994, 64, 212–219. [Google Scholar] [PubMed]
- Hemilae, H. Vitamin C and Infections. Nutrients 2017, 9, 339. [Google Scholar] [CrossRef] [PubMed]
- Hemilä, H.; Louhiala, P. Vitamin C for preventing and treating pneumonia. Cochrane Database Syst. Rev. 2013, 8, CD005532. [Google Scholar] [CrossRef] [PubMed]
- Jouybar, R.; Kabgani, H.; Kamalipour, H.; Shahbazi, S.; Allahyary, E.; Rasouli, M.; Akhlagh, S.H.; Shafa, M.; Ghazinoor, M.; Moeinvaziri, M.T.; et al. The perioperative effect of ascorbic acid on inflammatory response in coronary artery bypass graft surgery; a randomized controlled trial coronary artery bypass graft surgery. Age (year) 2012, 56, 61–83. [Google Scholar]
- Knodell, R.G.; Tate, M.A.; Akl, B.F.; Wilson, J.W. Vitamin C prophylaxis for posttransfusion hepatitis: Lack of effect in a controlled trial. Am. J. Clin. Nutr. 1981, 34, 20–23. [Google Scholar] [CrossRef] [PubMed]
- Li, C.C. Changes of creatine phosphokinase and malondialdehyde in the serum and clinical use of large doses of vitamin C following open heart surgery. Zhonghua Wai Ke Za Zhi (Chin. J. Surg.) 1990, 28, 16–17. [Google Scholar] [PubMed]
- Carnes, C.A.; Chung, M.K.; Nakayama, T.; Nakayama, H.; Baliga, R.S.; Piao, S.; Kanderian, A.; Pavia, S.; Hamlin, R.L.; McCarthy, P.M.; et al. Ascorbate attenuates atrial pacing-induced peroxynitrite formation and electrical remodeling and decreases the incidence of postoperative atrial fibrillation. Circ. Res. 2001, 89, e32–e38. [Google Scholar] [CrossRef] [PubMed]
- Demirag, K.; Askar, F.Z.; Uyar, M.; Cevik, A.; Ozmen, D.; Mutaf, I.; Bayindir, O. The protective effects of high dose ascorbic acid and diltiazem on myocardial ischaemia-reperfusion injury. Middle East J. Anaesthesiol. 2001, 16, 67–79. [Google Scholar] [PubMed]
- Eslami, M.; Badkoubeh, R.S.; Mousavi, M.; Radmehr, H.; Salehi, M.; Tavakoli, N.; Avadi, M.R. Oral ascorbic acid in combination with beta-blockers is more effective than beta-blockers alone in the prevention of atrial fibrillation after coronary artery bypass grafting. Tex. Heart Inst. J. 2007, 342, 68. [Google Scholar]
- Colby, J.A.; Chen, W.T.; Baker, W.L.; Coleman, C.I.; Reinhart, K.; Kluger, J.; White, C.M. Effect of ascorbic acid on inflammatory markers after cardiothoracic surgery. Am. J. Health Syst. Pharm. 2011, 68, 1632–1639. [Google Scholar] [CrossRef] [PubMed]
- Papoulidis, P.; Ananiadou, O.; Chalvatzoulis, E.; Ampatzidou, F.; Koutsogiannidis, C.; Karaiskos, T.; Madesis, A.; Drossos, G. The role of ascorbic acid in the prevention of atrial fibrillation after elective on-pump myocardial revascularization surgery: A single-center experience–a pilot study. Interact. Cardiovasc. Thorac. Surg. 2011, 12, 121–124. [Google Scholar] [CrossRef] [PubMed]
- Bjordahl, P.M.; Helmer, S.D.; Gosnell, D.J.; Wemmer, G.E.; O’Hara, W.W.; Milfeld, D.J. Perioperative supplementation with ascorbic acid does not prevent atrial fibrillation in coronary artery bypass graft patients. Am. J. Surg. 2012, 204, 862–867. [Google Scholar] [CrossRef] [PubMed]
- Dehghani, M.R.; Majidi, N.; Rahmani, A.; Asgari, B.; Rezaei, Y. Effect of oral vitamin C on atrial fibrillation development after isolated coronary artery bypass grafting surgery: A prospective randomized clinical trial. Cardiol. J. 2014, 21, 492–499. [Google Scholar] [CrossRef] [PubMed]
- Ebade, A.; Taha, W.S.; Saleh, R.H.; Fawzy, A. Ascorbic acid versus magnesium for the prevention of atrial fibrillation after coronary artery bypass grafting surgery. Egypt. J. Cardiothorac. Anesth. 2014, 8, 59–65. [Google Scholar] [CrossRef]
- Samadikhah, J.; Golzari, S.E.J.; Sabermarouf, B.; Karimzadeh, I.; Tizro, P.; Khanli, H.M.; Ghabili, K. Efficacy of Combination Therapy of Statin and Vitamin C in Comparison with Statin in the Prevention of Post-CABG Atrial Fibrillation. Adv. Pharm. Bull. 2014, 4, 97. [Google Scholar] [PubMed]
- Das, D.; Sen, C.; Goswami, A. Effect of Vitamin C on adrenal suppression by etomidate induction in patients undergoing cardiac surgery: A randomized controlled trial. Ann. Card. Anaesth. 2016, 19, 410. [Google Scholar] [CrossRef] [PubMed]
- Antonic, M.; Lipovec, R.; Gregorcic, F.; Juric, P.; Kosir, G. Perioperative ascorbic acid supplementation does not reduce the incidence of postoperative atrial fibrillation in on-pump coronary artery bypass graft patients. J. Cardiol. 2017, 69, 98–102. [Google Scholar] [CrossRef] [PubMed]
- Crimi, E.; Sica, V.; Williams-Ignarro, S.; Zhang, H.; Slutsky, A.S.; Ignarro, L.J.; Napoli, C. The role of oxidative stress in adult critical care. Free Radic. Biol. Med. 2006, 40, 398–406. [Google Scholar] [CrossRef] [PubMed]
- Howe, K.P.; Clochesy, J.M.; Goldstein, L.S.; Owen, H. Mechanical ventilation antioxidant trial. Am. J. Crit. Care 2015, 24, 440–445. [Google Scholar] [CrossRef] [PubMed]
- Barta, E.; Pechán, I.; Cornák, V.; Luknárová, O.; Rendeková, V.; Verchovodko, P. Protective effect of alpha-tocopherol and l-ascorbic acid against the ischemic-reperfusion injury in patients during open-heart surgery. Bratisl. Lek. Listy 1991, 92, 174–183. [Google Scholar] [PubMed]
- Angdin, M.; Settergren, G.; Starkopf, J.; Zilmer, M.; Zilmer, K.; Vaage, J. Protective effect of antioxidants on pulmonary endothelial function after cardiopulmonary bypass. J. Cardiothorac. Vasc. Anesth. 2003, 17, 314–320. [Google Scholar] [CrossRef]
- Gunes, T.; Bozok, S.; Kestelli, M.; Yurekli, I.; Ilhan, G.; Ozpak, B.; Bademci, M.; Ozcem, B.; Sahin, A. α-tocopherol and ascorbic acid in early postoperative period of cardiopulmonary bypass. J. Cardiovasc. Med. 2012, 13, 691–699. [Google Scholar] [CrossRef] [PubMed]
- Westhuyzen, J.; Cochrane, A.D.; Tesar, P.J.; Mau, T.; Cross, D.B.; Frenneaux, M.P.; Khafagi, F.A.; Fleming, S.J. Effect of preoperative supplementation with alpha-tocopherol and ascorbic acid on myocardial injury in patients undergoing cardiac operations. J. Thorac. Cardiovasc. Surg. 1997, 113, 942–948. [Google Scholar] [CrossRef]
- Castillo, R.; Rodrigo, R.; Perez, F.; Cereceda, M.; Asenjo, R.; Zamorano, J.; Navarrete, R.; Villalabeitia, E.; Sanz, J.; Baeza, C. Antioxidant therapy reduces oxidative and inflammatory tissue damage in patients subjected to cardiac surgery with extracorporeal circulation. Basic Clin. Pharmacol. Toxicol. 2011, 108, 256–262. [Google Scholar] [CrossRef] [PubMed]
- Rodrigo, R.; Prieto, J.C.; Castillo, R. Cardioprotection against ischaemia/reperfusion by vitamins C and E plus n-3 fatty acids: Molecular mechanisms and potential clinical applications. Clin. Sci. 2013, 124, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Stanger, O.; Aigner, I.; Schimetta, W.; Wonisch, W. Antioxidant supplementation attenuates oxidative stress in patients undergoing coronary artery bypass graft surgery. Tohoku J. Exp. Med. 2014, 232, 145–154. [Google Scholar] [CrossRef] [PubMed]
- Sartor, Z.; Kesey, J.; Dissanaike, S. The effects of intravenous vitamin C on point-of-care glucose monitoring. J. Burn Care Res. 2015, 36, 50–56. [Google Scholar] [CrossRef] [PubMed]
- Vasudevan, S.; Hirsch, I.B. Interference of intravenous vitamin C with blood glucose testing. Diabetes Care 2014, 37, e93–e94. [Google Scholar] [CrossRef] [PubMed]
- Halliwell, B. Vitamin C: Antioxidant or pro-oxidant in vivo? Free Radic. Res. 1996, 25, 439–454. [Google Scholar] [CrossRef] [PubMed]
- Gey, K.F. Vitamins E plus C and interacting conutrients required for optimal health. A critical and constructive review of epidemiology and supplementation data regarding cardiovascular disease and cancer. BioFactors 1998, 7, 113–174. [Google Scholar] [PubMed]
- Bucala, R. Lipid and lipoprotein oxidation: Basic mechanisms and unresolved questions in vivo. Redox Rep. 1996, 2, 291–307. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Espey, M.G.; Krishna, M.C.; Mitchell, J.B.; Corpe, C.P.; Buettner, G.R.; Shacter, E.; Levine, M. Pharmacologic ascorbic acid concentrations selectively kill cancer cells: Action as a pro-drug to deliver hydrogen peroxide to tissues. Proc. Natl. Acad. Sci. USA 2005, 102, 13604–13609. [Google Scholar] [CrossRef] [PubMed]
- Collins, A.; Cadet, J.; Epe, B.; Gedik, C. Problems in the measurement of 8-oxoguanine in human DNA. Report of a workshop, DNA oxidation, held in Aberdeen, UK, 19–21 January, 1997. Carcinogenesis 1997, 18, 1833–1836. [Google Scholar] [CrossRef] [PubMed]
- Rümelin, A.; Jaehde, U.; Kerz, T.; Roth, W.; Krämer, M.; Fauth, U. Early postoperative substitution procedure of the antioxidant ascorbic acid. J. Nutr. Biochem. 2005, 16, 104–108. [Google Scholar] [CrossRef] [PubMed]
- Rümelin, A.; Humbert, T.; Lühker, O.; Drescher, A.; Fauth, U. Metabolic clearance of the antioxidant ascorbic acid in surgical patients. J. Surg. Res. 2005, 129, 46–51. [Google Scholar] [CrossRef] [PubMed]
- Bjugstad, K.B.; Rael, L.T.; Stewart Levy, S.; Carrick, M.; Mains, C.W.; Slone, D.S.; Bar-Or, D. Oxidation-reduction potential as a biomarker for severity and acute outcome in traumatic brain injury. Oxid. Med. Cell. Longev. 2016. [Google Scholar] [CrossRef] [PubMed]
- Rael, L.T.; Bar-Or, R.; Salottolo, K.; Mains, C.W.; Slone, D.S.; Offner, P.J.; Bar-Or, D. Injury severity and serum amyloid a correlate with plasma oxidation-reduction potential in multi-trauma patients: A retrospective analysis. Scand. J. Trauma Resusc. Emerg. Med. 2009. [Google Scholar] [CrossRef] [PubMed]
- Robitaille, L.; Hoffer, L.J. A simple method for plasma total vitamin C analysis suitable for routine clinical laboratory use. Nutr. J. 2015, 15, 40. [Google Scholar] [CrossRef] [PubMed]
- Pullar, J.M.; Bayer, S.; Carr, A.C. Appropriate handling, processing and analysis of blood samples is essential to avoid oxidation of vitamin C to dehydroascorbic acid. Antioxidants 2018, 7, 29. [Google Scholar] [CrossRef] [PubMed]
- Key, T.; Oakes, S.; Davey, G.; Moore, J.; Edmond, L.M.; McLoone, U.J.; Thurnham, D.I. Stability of vitamins a, c, and e, carotenoids, lipids, and testosterone in whole blood stored at 4 degrees c for 6 and 24 h before separation of serum and plasma. Cancer Epidemiol. Biomark. Prev. 1996, 5, 811–814. [Google Scholar]
- Wang, Y.; Liu, X.J.; Robitaille, L.; Eintracht, S.; MacNamara, E.; Hoffer, L.J. Effects of vitamin C and vitamin d administration on mood and distress in acutely hospitalized patients. Am. J. Clin. Nutr. 2013, 98, 705–711. [Google Scholar] [CrossRef] [PubMed]
- Margolis, S.A.; Duewer, D.L. Measurement of ascorbic acid in human plasma and serum: Stability, intralaboratory repeatability, and interlaboratory reproducibility. Clin. Chem. 1996, 42, 1257–1262. [Google Scholar] [PubMed]
- Kirkham, J.J.; Dwan, K.M.; Altman, D.G.; Gamble, C.; Dodd, S.; Smyth, R.; Williamson, P.R. The impact of outcome reporting bias in randomised controlled trials on a cohort of systematic reviews. BMJ 2010, 340, c365. [Google Scholar] [CrossRef] [PubMed]
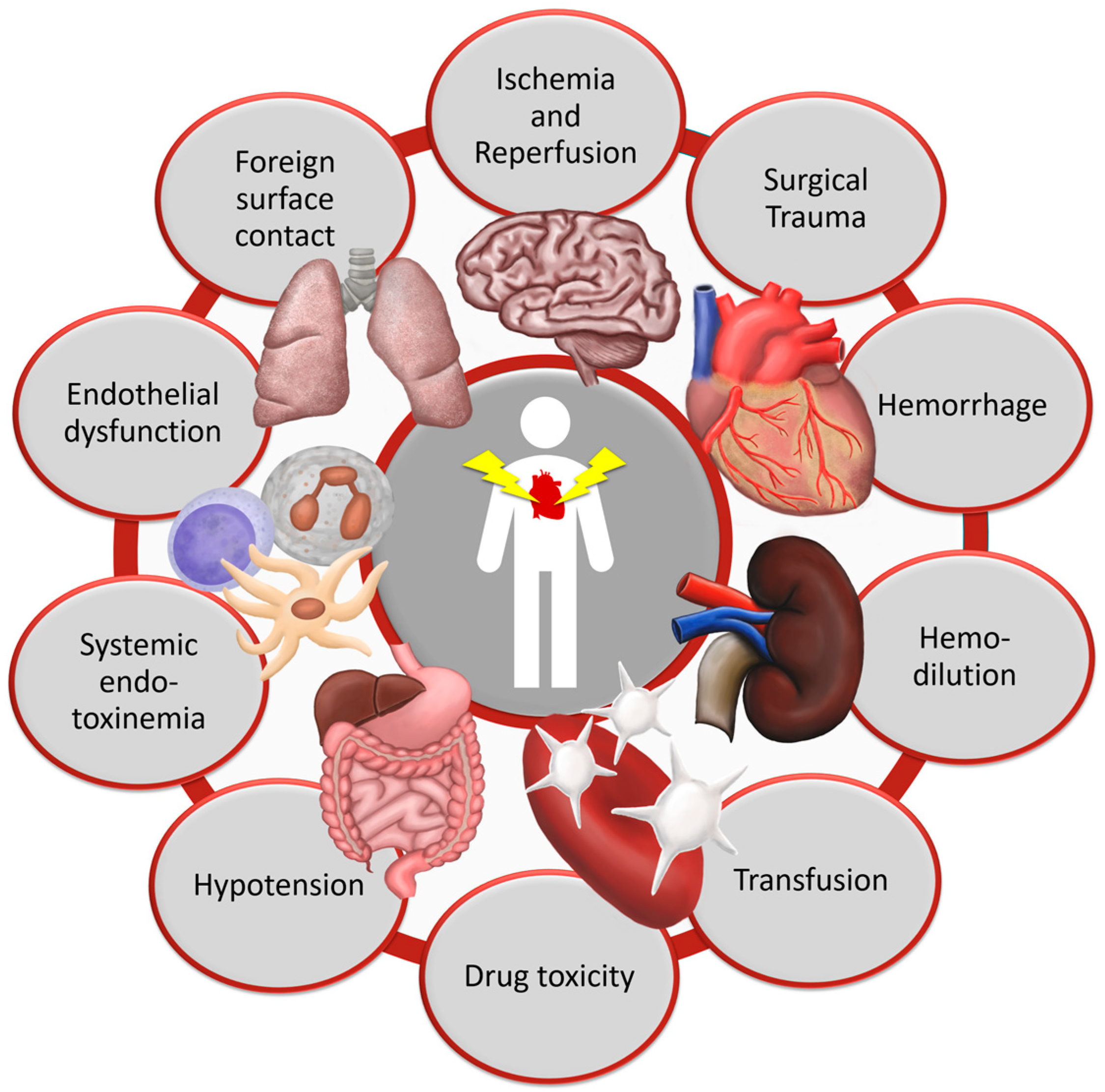
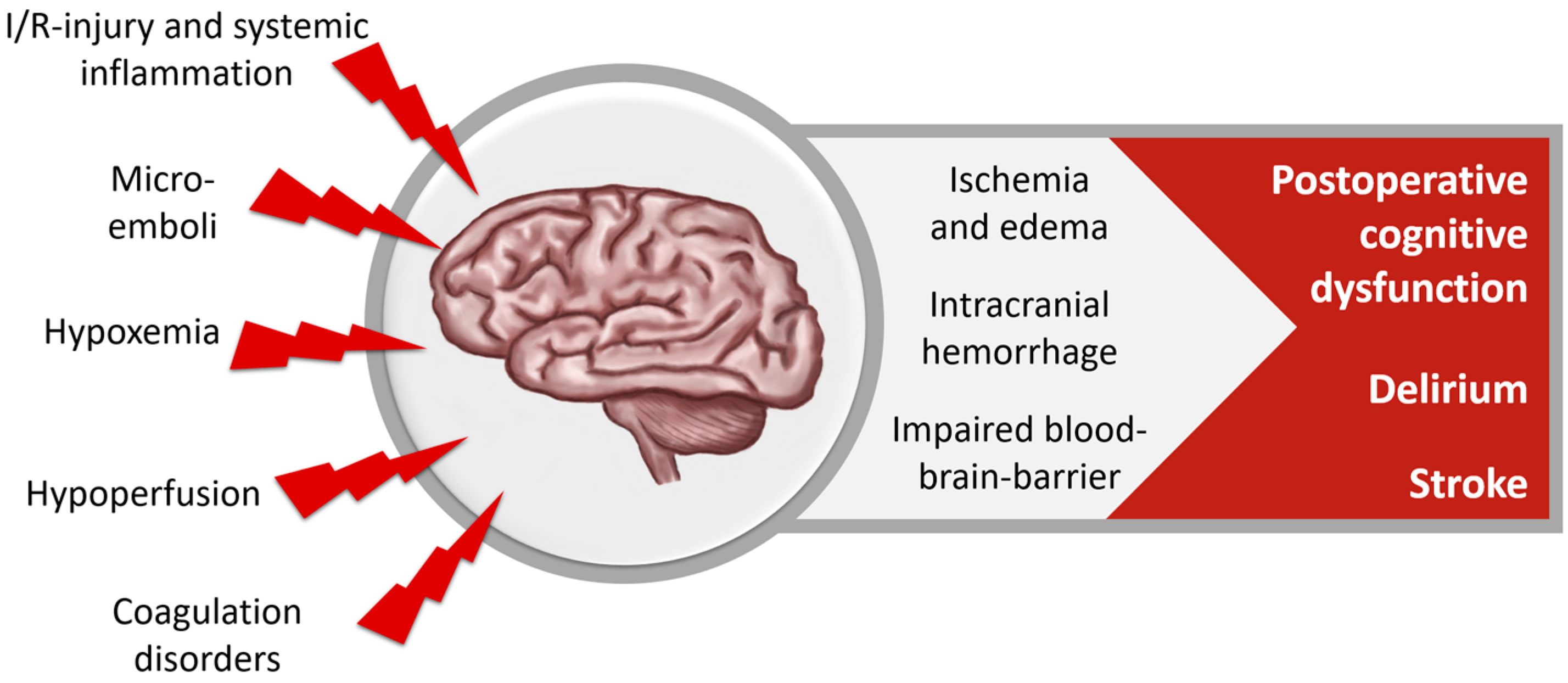
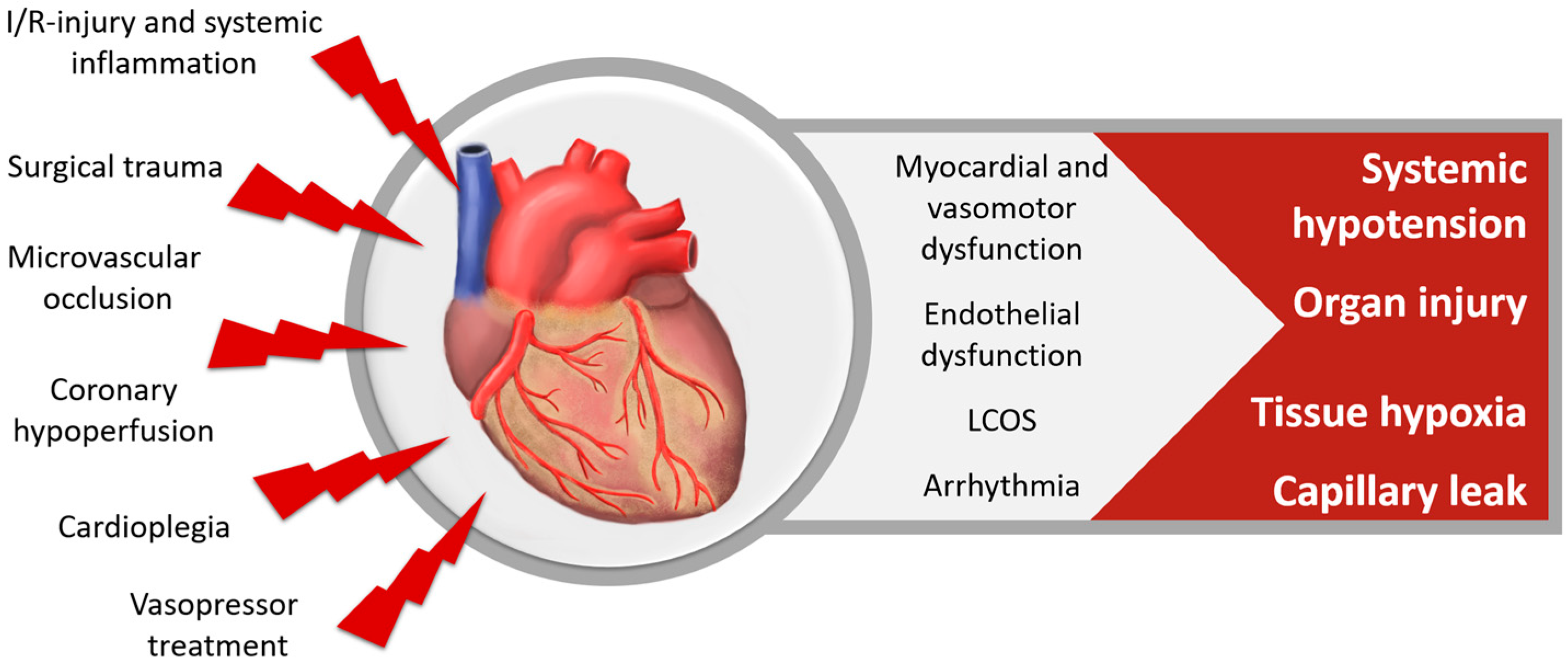
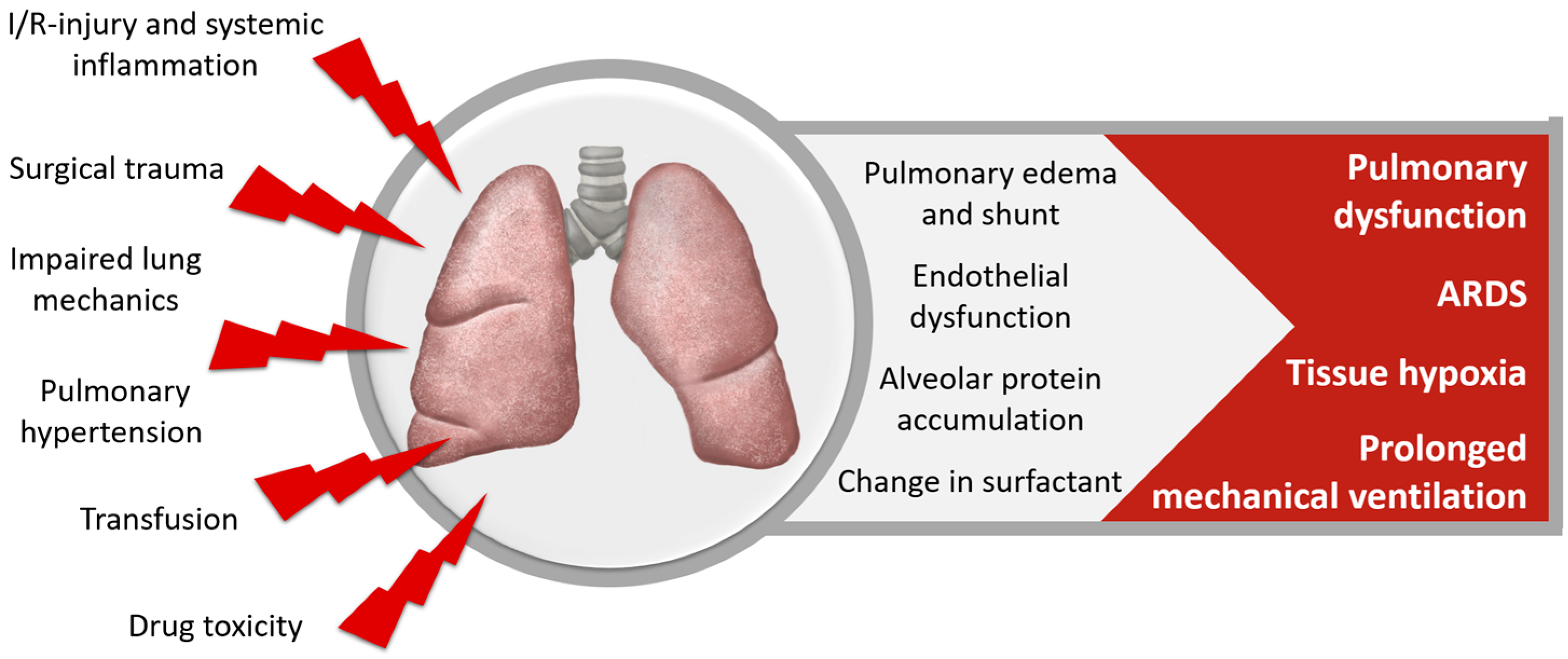
| Organ System | Influence of Vitamin C |
|---|---|
| Nervous system | Elevated levels protect neurons from oxidative damage [49,57] |
| Reduces the infarct volume after ischemia [58] | |
| Cardiovascular System | Attenuates myocardial damage and improves myocardial stunning [49] |
| Reduces vasopressor demand [52] | |
| Reduces rate of atrial fibrillation [59,60] | |
| Improves endothelial function [61,62] | |
| Respiratory System | Reduces intubation time [63] |
| Decreases risk of pneumonia and alveolar inflammation [53] | |
| Renal System | Reduces fluid demand and increases urine production [54,55] |
| Gastrointestinal System | Attenuates drug toxicity, decreases inflammatory reaction [64] |
| Lowers infiltration of neutrophils [64] | |
| Reduces the expression of apoptosis related genes [44] | |
| Coagulation System | Restores platelet function and decreases capillary plugging [43] |
| Attenuates a sepsis-induced drop of thrombocytes [43] | |
| Immune System | Inhibits bacterial growth [47], enhances microbial killing [38] |
| Supports endothelial barrier function and promotes antioxidant scavenging [38] |
| Author and Year | Patients | Dosage of Vitamin C | p.o./i.v. | Results |
|---|---|---|---|---|
| Knodell 1981 [157] | 175+ hepatitis | Preop: 4 × 800 mg/day for 2 days | p.o. | Elevations of plasma vitamin C, no influence on the hepatitis |
| Postop: 4 × 800 mg/day for 2 weeks | ||||
| Li 1990 [158] | 20 | Preop: 250 mg/kg before the start of extracorporeal circulation | N.A. | Sign. reduction in lipid peroxidation |
| Dingchao 1994 [86] | 85 CPB | 125 mg/kg 30 min before surgery and at the end of CPB | i.v. | Decreased CK/CKMB, LDH, & rate of defibrillation, ICU- and hospital LOS, improved CI |
| Carnes 2001 [159] | 86 CABG | Preop: 1 × 2 g the night before | N.A. | Lower rate of AF |
| Postop: 2 × 0.5 g/day for 5 days | ||||
| Demirag 2001 [160] | 30 elective | Group 1: 2 × 50 mg/kg vitamin C at induction and end of CPB | i.v. | Prevention of lipid peroxidation no difference in myocardial I/R-injury |
| Group 2: vitamin C + diltiazem: bolus and 2 µg/kg/min until end of CPB | ||||
| Eslami 2007 [161] | 100 CABG | Preop: 1 × 2 g night before | p.o. | Lower rate of AF |
| Postop: 2 × 1 g/day for 5 days | ||||
| Colby 2011 [162] | 24 CABG and/or valve | Preop: 1 × 2 g night before | p.o. | No difference in CRP, WBC, fibrinogen, Trend: decreased AF, hospital- and ICU-LOS |
| Postop: 2 × 0.5 g/day for 4 days | ||||
| Papoulidis 2011 [163] | 170 CABG | Preop: 1 × 2 g 3 h prior to surgery | i.v. | Sign. lower rate of AF, hospital- and ICU-LOS |
| Postop: 2 × 0.5 mg/day for 5 days | ||||
| Bjordahl 2012 [164] | 185 CABG | Preop: 1 × 2 g night before surgery | p.o. | No difference in postoperative complications, mortality or AF |
| Postop: 2 × 1 g/day for 5 days | ||||
| Jouybar 2012 [156] | 40 CABG | Preop: 2 × 3 g 12–18 h before surgery and during CPB initiation | i.v. | No difference in inflammatory cytokines, hemodynamics, blood gases, urea nitrogen, creatinine, WBC, platelet counts & outcomes |
| Dehghani 2014 [165] | 100 CABG | Preop: 1 × 2 g | p.o. | Sign. lower rate of AF, hospital- and ICU-LOS |
| Postop: 2 × 0.5 g/day for 5 days | ||||
| Ebade 2014 [166] | 40 | Preop: 1 × 2 g | i.v. | Lower incidence of AF |
| Postop: 1 × 1 g 12 h after surgery, 3 × 1 g for 6 days after surgery | Shortened ICU- and hospital-LOS | |||
| Sama-dikhah 2014 [167] | 120 CABG | Preop: 1 × 2 g | p.o. | Sign. lower rate of AF |
| Postop: 1 × 1 g/day for 5 days | ||||
| Plus atorvastatin 40 mg | ||||
| Sadegh-pour 2015 [141] | 290 CABG, valve | Preop: 1 × 2 g before surgery | Preop: i.v. Postop: p.o. | Sign. reductions in AF, hospital-LOS, intubation time, complications (death, renal function, infection) and drainage, unchanged ICU-LOS |
| Postop: 1 × 1 g/day for 4 days | ||||
| Das 2016 [168] | 70 elective low risk CABG | Preop: 2 × 0.5 g for 7 days prior to surgery | p.o. | Lower vasopressors-demand, no difference in time to extubation, ICU- and hospital-LOS, mortality or complications |
| Antonic 2016 [169] | 105 CABG | Preop: 2 × 2 g: 24 and 2 h before surgery | i.v. | Trend: decreased rate of AF, no difference in complications |
| Postop: 2 × 1 g/day for 4 days | ||||
| Antonic 2017 [119] | 100 CABG | Preop: 2 × 2 g: 24 and 2 h | i.v. | No sign. protective effect of ascorbic acid on the incidence of postoperative AKI |
| Postop: 2 × 1 g/day for 5 days |
| Author and Year | N | Treatment | Outcomes |
|---|---|---|---|
| Barta 1991 [172] | 20 | Preop: 2000 IU Vit E: 12 h before surgery; 2 g vitamin C in the morning on the day of surgery | Inhibition of the decrease of catalase Lower lipid oxidation and lysosomal enzymes in intervention group |
| Westhuyzen 1997 [175] | 76 | Preoperative (7–10 days): 1 g vitamin C and 750 IU Vit E | Supplementation of the vitamins prevented depletion, but provided no clinical advantage |
| Angdin 2003 [173] | 22 | Preop: 900 mg Vit E for 10–14 days plus 1 × 2 g vitamin C and 600 mg allopurinol the evening before surgery, and acetylcysteine during surgery | Reduction of pulmonary vascular endothelial dysfunction in the group treated with ntioxidants |
| Castillo 2011 [176] | 95 | Preop: for 7 days n-3 PUFA 2 g/day Plus, for 2 days preop until discharge vitamin C 1 g/day and Vit E 400 IU/day | Decrease in oxidative stress-related biomarkers in atrial tissue |
| Gunes 2012 [174] | 59 | Preop: vitamin C 500 mg and Vit E 300 mg Postop: vitamin C 500 mg/day and Vit E 300 mg/day for 4 days | Significant reduction of CRP |
| Rodrigo 2013 [177] | 203 | Preop: 1 g/day vitamin C plus PUFA and Vit E for 2 days preop until discharge | Decrease in oxidative stress-related biomarkers in atrial tissue |
| Stanger 2014 [178] | 75 | 4 subgroups: control, vitamins, n-3 PUFAs, and a combination of vitamins and n-3 PUFAs | Attenuation of postop oxidative stress, Oxidative stress associated with consumption of antioxidants and onset of AF |
| Vitamin group: 500 mg vitamin C + 45 IE Vit E 30 min before reperfusion, postop and 120 min after reperfusion | |||
| Rezk 2017 [87] | 100 | 3 days preoperatively | Significantly lower incidence in vitamin C group, ICU-LOS, need for inotropes and mechanical ventilation |
| Group 1: β-blocker: 5 mg bisoprolol and 2 g/day vitamin C | |||
| Group 2: β-blocker only |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hill, A.; Wendt, S.; Benstoem, C.; Neubauer, C.; Meybohm, P.; Langlois, P.; Adhikari, N.K.; Heyland, D.K.; Stoppe, C. Vitamin C to Improve Organ Dysfunction in Cardiac Surgery Patients—Review and Pragmatic Approach. Nutrients 2018, 10, 974. https://doi.org/10.3390/nu10080974
Hill A, Wendt S, Benstoem C, Neubauer C, Meybohm P, Langlois P, Adhikari NK, Heyland DK, Stoppe C. Vitamin C to Improve Organ Dysfunction in Cardiac Surgery Patients—Review and Pragmatic Approach. Nutrients. 2018; 10(8):974. https://doi.org/10.3390/nu10080974
Chicago/Turabian StyleHill, Aileen, Sebastian Wendt, Carina Benstoem, Christina Neubauer, Patrick Meybohm, Pascal Langlois, Neill KJ Adhikari, Daren K. Heyland, and Christian Stoppe. 2018. "Vitamin C to Improve Organ Dysfunction in Cardiac Surgery Patients—Review and Pragmatic Approach" Nutrients 10, no. 8: 974. https://doi.org/10.3390/nu10080974
APA StyleHill, A., Wendt, S., Benstoem, C., Neubauer, C., Meybohm, P., Langlois, P., Adhikari, N. K., Heyland, D. K., & Stoppe, C. (2018). Vitamin C to Improve Organ Dysfunction in Cardiac Surgery Patients—Review and Pragmatic Approach. Nutrients, 10(8), 974. https://doi.org/10.3390/nu10080974






