Development of Vitamin D Toxicity from Overcorrection of Vitamin D Deficiency: A Review of Case Reports
Abstract
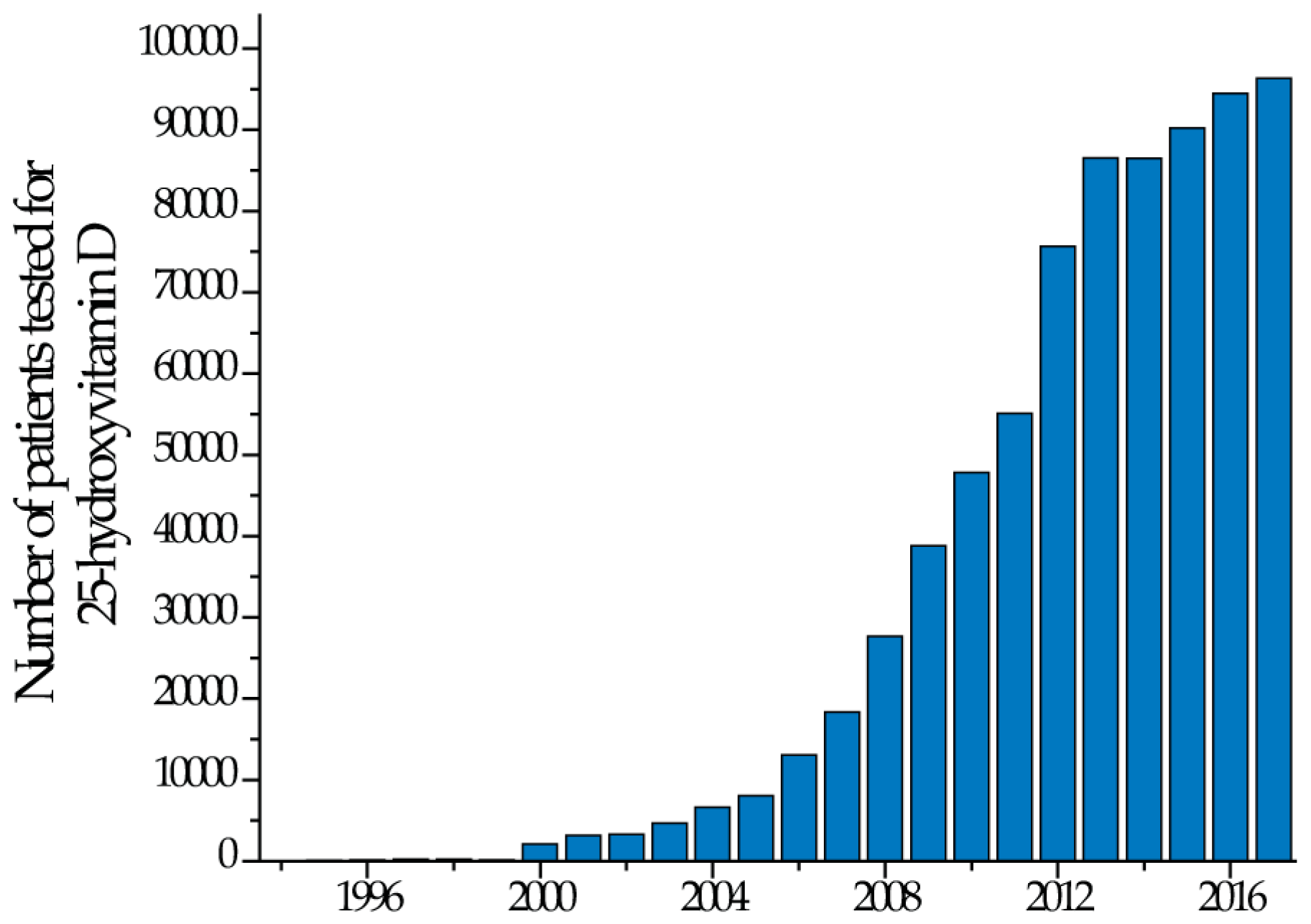
1. Introduction
2. Materials and Methods
3. Results and discussion
3.1. Manufacturing Errors
3.2. Inappropriate Administration
3.3. Incorrect Prescribing by Physicians
4. Conclusions
Author Contributions
Conflicts of Interest
References
- Delvin, E.; Souberbielle, J.C.; Viard, J.P.; Salle, B. Role of vitamin D in acquired immune and autoimmune diseases. Crit. Rev. Clin. Lab. Sci. 2014, 51, 232–247. [Google Scholar] [CrossRef] [PubMed]
- Brewer, L.C.; Michos, E.D.; Reis, J.P. Vitamin D in atherosclerosis, vascular disease, and endothelial function. Curr. Drug Targets 2011, 12, 54–60. [Google Scholar] [CrossRef] [PubMed]
- Rizzoli, R. Nutritional aspects of bone health. Best Pract. Res. Clin. Endocrinol. Metab. 2014, 28, 795–808. [Google Scholar] [CrossRef] [PubMed]
- Ross, A.C.; Manson, J.E.; Abrams, S.A.; Aloia, J.F.; Brannon, P.M.; Clinton, S.K.; Durazo-Arvizu, R.A.; Gallagher, J.C.; Gallo, R.L.; Jones, G.; et al. The 2011 report on dietary reference intakes for calcium and vitamin D from the Institute of Medicine: What clinicians need to know. J. Clin. Endocrinol. Metab. 2011, 96, 53–58. [Google Scholar] [CrossRef] [PubMed]
- Glade, M.J. A 21st century evaluation of the safety of oral vitamin D. Nutrition 2012, 28, 344–556. [Google Scholar] [CrossRef] [PubMed]
- Hathcock, J.N.; Shao, A.; Vieth, R.; Heaney, R. Risk assessment for vitamin D. Am. J. Clin. Nutr. 2007, 85, 6–18. [Google Scholar] [CrossRef] [PubMed]
- Vieth, R. Why the optimal requirement for Vitamin D3 is probably much higher than what is officially recommended for adults. J. Steroid Biochem. Mol. Biol. 2004, 8, 575–579. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F.; Binkley, N.C.; Bischoff-Ferrari, H.A.; Gordon, C.M.; Hanley, D.A.; Heaney, R.P.; Murad, M.H.; Weaver, C.M.; Endocrine, S. Evaluation, treatment, and prevention of vitamin D deficiency: An Endocrine Society clinical practice guideline. J. Clin. Endocrinol. Metab. 2011, 96, 1911–1930. [Google Scholar] [CrossRef] [PubMed]
- Chu, M.P.; Alagiakrishnan, K.; Sadowski, C. The cure of ageing: Vitamin D—magic or myth? Postgrad. Med. J. 2010, 86, 608–616. [Google Scholar] [CrossRef] [PubMed]
- Shahangian, S.; Alspach, T.D.; Astles, J.R.; Yesupriya, A.; Dettwyler, W.K. Trends in laboratory test volumes for Medicare Part B reimbursements, 2000-2010. Arch. Pathol. Lab. Med. 2014, 138, 189–203. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F. Deficiency of sunlight and vitamin D. BMJ Br. Med. J. 2008, 336, 1318. [Google Scholar] [CrossRef] [PubMed]
- Galior, K.; Ketha, H.; Grebe, S.; Singh, R.J. 10 years of 25-hydroxyvitamin-D testing by LC-MS/MS-trends in vitamin-D deficiency and sufficiency. Bone Rep. 2018, 8, 268–273. [Google Scholar] [CrossRef] [PubMed]
- Lehmann, U.; Gjessing, H.R.; Hirche, F.; Mueller-Belecke, A.; Gudbrandsen, O.A.; Ueland, P.M.; Mellgren, G.; Lauritzen, L.; Lindqvist, H.; Hansen, A.L.; et al. Efficacy of fish intake on vitamin D status: A meta-analysis of randomized controlled trials. Am. J. Clin. Nutr. 2015, 102, 837–847. [Google Scholar] [CrossRef] [PubMed]
- Keegan, R.J.; Lu, Z.; Bogusz, J.M.; Williams, J.E.; Holick, M.F. Photobiology of vitamin D in mushrooms and its bioavailability in humans. Dermatoendocrinol 2013, 5, 165–176. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.P.; Tansey, M.; Jetton, J.G.; Krasowski, M.D. Vitamin D Toxicity: A 16-Year Retrospective Study at an Academic Medical Center. Lab. Med. 2018, 49, 123–129. [Google Scholar] [CrossRef] [PubMed]
- Spiller, H.A.; Good, T.F.; Spiller, N.E.; Aleguas, A. Vitamin D exposures reported to US poison centers 2000-2014: Temporal trends and outcomes. Hum. Exp. Toxicol. 2016, 35, 457–461. [Google Scholar] [CrossRef] [PubMed]
- Taylor, P.N.; Davies, J.S. A review of the growing risk of vitamin D toxicity from inappropriate practice. Br. J. Clin. Pharmacol. 2018, 84, 1121–1127. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, R.; Hashiba, K. Reliability of QT intervals as indicators of clinical hypercalcemia. Clin. Cardiol. 1988, 11, 395–400. [Google Scholar] [CrossRef] [PubMed]
- Minisola, S.; Pepe, J.; Piemonte, S.; Cipriani, C. The diagnosis and management of hypercalcaemia. BMJ 2015, 350, h2723. [Google Scholar] [CrossRef] [PubMed]
- Alshahrani, F.; Aljohani, N. Vitamin D: Deficiency, sufficiency and toxicity. Nutrients 2013, 5, 3605–3616. [Google Scholar] [CrossRef] [PubMed]
- Bischoff-Ferrari, H.A. Optimal serum 25-hydroxyvitamin D levels for multiple health outcomes. Adv. Exp. Med. Biol. 2014, 810, 500–525. [Google Scholar] [PubMed]
- Anik, A.; Catli, G.; Abaci, A.; Dizdarer, C.; Bober, E. Acute vitamin D intoxication possibly due to faulty production of a multivitamin preparation. J. Clin. Res. Pediatr. Endocrinol. 2013, 5, 136–139. [Google Scholar] [PubMed]
- Kara, C.; Gunindi, F.; Ustyol, A.; Aydin, M. Vitamin D intoxication due to an erroneously manufactured dietary supplement in seven children. Pediatrics 2014, 133, e240–e244. [Google Scholar] [CrossRef] [PubMed]
- Araki, T.; Holick, M.F.; Alfonso, B.D.; Charlap, E.; Romero, C.M.; Rizk, D.; Newman, L.G. Vitamin D intoxication with severe hypercalcemia due to manufacturing and labeling errors of two dietary supplements made in the United States. J. Clin. Endocrinol. Metab. 2011, 96, 3603–3608. [Google Scholar] [CrossRef] [PubMed]
- Koutkia, P.; Chen, T.C.; Holick, M.F. Vitamin D intoxication associated with an over-the-counter supplement. N. Engl. J. Med. 2001, 345, 66–77. [Google Scholar] [CrossRef] [PubMed]
- Ketha, H.; Wadams, H.; Lteif, A.; Singh, R.J. Iatrogenic vitamin D toxicity in an infant--a case report and review of literature. J. Steroid. Biochem. Mol. Biol. 2015, 148, 14–18. [Google Scholar] [CrossRef] [PubMed]
- Bilbao, N.A. Vitamin D Toxicity in Young Breastfed Infants: Report of 2 Cases. Glob. Pediatr. Health 2017, 4, 2333794X17731695. [Google Scholar] [CrossRef] [PubMed]
- Rocha, P.N.; Santos, C.S.; Avila, M.O.; Neves, C.L.; Bahiense-Oliveira, M. Hypercalcemia and acute kidney injury caused by abuse of a parenteral veterinary compound containing vitamins A, D, and E. J. Bras. Nefrol. 2011, 33, 467–471. [Google Scholar] [CrossRef] [PubMed]
- Kaur, P.; Mishra, S.K.; Mithal, A. Vitamin D toxicity resulting from overzealous correction of vitamin D deficiency. Clin. Endocrinol. 2015, 83, 327–331. [Google Scholar] [CrossRef] [PubMed]
- Koul, P.A.; Ahmad, S.H.; Ahmad, F.; Jan, R.A.; Shah, S.U.; Khan, U.H. Vitamin d toxicity in adults: A case series from an area with endemic hypovitaminosis d. Oman Med. J. 2011, 26, 201–204. [Google Scholar] [CrossRef] [PubMed]
- Bansal, R.K.; Tyagi, P.; Sharma, P.; Singla, V.; Arora, V.; Bansal, N.; Kumar, A.; Arora, A. Iatrogenic hypervitaminosis D as an unusual cause of persistent vomiting: A case report. J. Med. Case Rep. 2014, 8, 74. [Google Scholar] [CrossRef] [PubMed]
- Pandita, K.K.; Razdan, S.; Kudyar, R.P.; Beigh, A.; Kuchay, S.; Banday, T. “Excess gooD can be Dangerous”. A case series of iatrogenic symptomatic hypercalcemia due to hypervitaminosis D. Clin. Cases. Miner. Bone Metab. 2012, 9, 118–120. [Google Scholar] [PubMed]
- Chowdry, A.M.; Azad, H.; Najar, M.S.; Mir, I. Acute kidney injury due to overcorrection of hypovitaminosis D: A tertiary center experience in the Kashmir Valley of India. Saudi J. Kidney Dis. Transpl. 2017, 28, 1321–1329. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Stephens, L.D.; Fitzgerald, R.L. How much is too much? Two contrasting cases of excessive vitamin D supplementation. Clin. Chim. Acta 2017, 473, 35–38. [Google Scholar] [CrossRef] [PubMed]
| Age (years) | Vitamin D dose | Form of Intake | Reason | Vitamin D, Serum (ng/m) | Total Ca, Serum (mg/dL) | Symptoms | Ref. |
|---|---|---|---|---|---|---|---|
| 0.4–4.2 (n = 7) | 260,000–800,000 IU/day | Fish oil supplements | Labeling errors | 340–962 | 13.4–18.8 | Weakness, loss of appetite, vomiting | [23] |
| 1–2 (n = 2) 1 | 200 IU/day (2–4weeks) 200 IU/day (1 month) | Oral preparation Oral preparation | Labeling errors Labeling errors | >160 760 | 13.7–19.3 19.4 | Abdominal pain, vomiting Poor appetite, vomiting | [22] |
| 58 40 | 1,864,000 IU (2 months) 970,000 IU (1 month) | Oral supplements Oral supplements | Labeling errors Labeling errors | 1220 645 | 15 13.2 | Fatigue, thirst, polyuria Nausea, vomiting, thirst, polyuria, muscle aches | [24] |
| 42 | 156,000–2,604,000 IU/day (2 years) | Oral supplements | Labeling errors | 487.3 | 15 | Dehydration, fatigue, loss of apetite | [25] |
| 0.3 | 50,000 IU/day (2 months) | Oral supplements | Inappropriate administration | 294 | 18.7 | Vomiting, diarrhea, dehydration | [26] |
| 0.3–0.2 (n = 2) | 20,000 IU/day (1.5 weeks) | Oral supplements | Inappropriate administration | 644 680 | 15 L 21 L | Poor appetite, lethargy, crying | [27] |
| 19 | 15,000,000 IU (1 year) | Injection | Inappropriate administration | 150 | 14.8 | Anorexia, nausea, vomiting | [28] |
| 42–86 (n = 16) | 2,220,000–6,360,000 IU (1–3 months) | Injection or oral sachets | Iatrogenic (body aches and fatigue) | 175–1161 | 11.1–15.7 | Nausea, vomiting, constipation | [29] |
| 48–75 (n = 0) | 3,000,000–60,000,000 IU (1–4 months) | Injection or oral sachets | Iatrogenic (various indications) | 164–306 | 12–13.98 | Vomiting, polyuria, anorexia | [30] |
| 45 | 6,000,000 IU (2 weeks) | Injection | Iatrogenic (knee surgery) | 150 | 23.1 | Anorexia, vomiting, abdominal pain | [31] |
| 42–85 (n = 15) | 600,000 IU (1 month–3 years) | Oral supplements + injections | Iatrogenic (improve health) | 103–164 | 10.9–15.2 | Altered sensorium, dehydration, vomiting, anorexia | [32] |
| 45–89 (n = 19) | 4,200,000–9,000,000 IU (1–5 months) | Oral tablets or injections | Iatrogenic (bone pain, aches, fatigue) | 190–988 | 11.9–15.2 | Vomiting, altered sensorium, AKI, constipation, | [33] |
| 75 | 50,000 IU/day (1 year) | Oral supplements | Iatrogenic (hypoparathyroidism) | 243 | 15.3 | Altered mental status | [34] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Galior, K.; Grebe, S.; Singh, R. Development of Vitamin D Toxicity from Overcorrection of Vitamin D Deficiency: A Review of Case Reports. Nutrients 2018, 10, 953. https://doi.org/10.3390/nu10080953
Galior K, Grebe S, Singh R. Development of Vitamin D Toxicity from Overcorrection of Vitamin D Deficiency: A Review of Case Reports. Nutrients. 2018; 10(8):953. https://doi.org/10.3390/nu10080953
Chicago/Turabian StyleGalior, Kornelia, Stefan Grebe, and Ravinder Singh. 2018. "Development of Vitamin D Toxicity from Overcorrection of Vitamin D Deficiency: A Review of Case Reports" Nutrients 10, no. 8: 953. https://doi.org/10.3390/nu10080953
APA StyleGalior, K., Grebe, S., & Singh, R. (2018). Development of Vitamin D Toxicity from Overcorrection of Vitamin D Deficiency: A Review of Case Reports. Nutrients, 10(8), 953. https://doi.org/10.3390/nu10080953




