Dietary Approaches for Japanese Patients with Diabetes: A Systematic Review
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Search
2.2. Study Selection
2.3. Validity and Quality Assessment
2.4. Data Abstraction
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- American Diabetes Association. Lifestyle management: Standards of Medical Care in Diabetes—2018. Diabetes Care 2018, 41, S38–S50. [Google Scholar] [CrossRef] [PubMed]
- Evert, A.B.; Boucher, J.L.; Cypress, M.; Dunbar, S.A.; Franz, M.J.; Mayer-Davis, E.J.; Neumiller, J.J.; Nwankwo, R.; Verdi, C.L.; Urbanski, P.; et al. Nutrition therapy recommendations for the management of adults with diabetes. Diabetes Care 2013, 36, 3821–3824. [Google Scholar] [CrossRef] [PubMed]
- Ley, S.H.; Hamdy, O.; Mohan, V.; Hu, F.B. Prevention and management of type 2 diabetes: Dietary components and nutritional strategies. Lancet 2014, 383, 1999–2007. [Google Scholar] [CrossRef]
- The Japan Diabetes Society. Diet therapy. In Practice Guideline for the Treatment for Diabetes in Japan; Nankodo: Tokyo, Japan, 2016; pp. 37–66. (In Japanese) [Google Scholar]
- The Japan Diabetes Society. Recommendation of Diet Therapy for Japanese Patients with Diabetes. Available online: http://www.jds.or.jp/modules/important/index.php?page=article&storyid=40 (accessed on 30 December 2017).
- Heianza, Y.; Arase, Y.; Kodama, S.; Tsuji, H.; Tanaka, S.; Saito, K.; Hara, S.; Sone, H. Trajectory of body mass index before the development of type2 diabetes in japanese men: Toranomon hospital health management center study 15. J. Diabetes Investig. 2015, 6, 289–294. [Google Scholar] [CrossRef] [PubMed]
- Looker, H.C.; Knowler, W.C.; Hanson, R.L. Changes in bmi and weight before and after the development of type 2 diabetes. Diabetes Care 2001, 24, 1917–1922. [Google Scholar] [CrossRef] [PubMed]
- De Fine Olivarius, N.; Richelsen, B.; Siersma, V.; Andreasen, A.H.; Beck-Nielsen, H. Weight history of patients with newly diagnosed type 2 diabetes. Diabet. Med. 2008, 25, 933–941. [Google Scholar] [CrossRef] [PubMed]
- Mann, J.I.; De Leeuw, I.; Hermansen, K.; Karamanos, B.; Karlström, B.; Katsilambros, N.; Riccardi, G.; Rivellese, A.A.; Rizkalla, S.; Slama, G.; et al. Diabetes and Nutrition Study Group (DNSG) of the European Association. Evidence-based nutritional approaches to the treatment and prevention of diabetes mellitus. Nutr. Metab. Cardiovasc. Dis. 2004, 14, 373–394. [Google Scholar] [CrossRef]
- Imai, S.; Matsuda, M.; Hasegawa, G.; Fukui, M.; Obahashi, H.; Ozasa, N.; Kajiyama, S. A simple meal plan of ‘eating vegetables before carbohydrate’ was more effective for achieving glycemic control than an exchange-based meal plan in Japanese patients with type 2 diabetes. Asia Pac. J. Clin. Nutr. 2011, 20, 161–168. [Google Scholar] [PubMed]
- Muramoto, A.; Yamamoto, N.; Nakamura, M.; Koike, J.; Numata, T.; Tamakoshi, A.; Tsushita, K. Effect of intensive lifestyle intervention programs on metabolic syndrome and obesity. Himan Kenkyuu 2010, 16, 182–187. [Google Scholar]
- Nanri, A.; Mizoue, T.; Kurotani, K.; Goto, A.; Oba, S.; Noda, M.; Sawada, N.; Tsugane, S. Low-carbohydrate diet and type 2 diabetes risk in japanese men and women: The japan public health center-based prospective study. PLoS ONE 2015, 10, e0118377. [Google Scholar] [CrossRef] [PubMed]
- Feinman, R.D.; Pogozelski, W.K.; Astrup, A.; Bernstein, R.K.; Fine, E.J.; Westman, E.C.; Accurso, A.; Frassetto, L.; Gower, B.A.; McFarlane, S.I.; et al. Dietary carbohydrate restriction as the first approach in diabetes management: Critical review and evidence base. Nutrition 2015, 31, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Snorgaard, O.; Poulsen, G.M.; Andersen, H.K.; Astrup, A. Systematic review and meta-analysis of dietary carbohydrate restriction in patients with type 2 diabetes. BMJ Open Diabetes Res. Care 2017, 5, e000354. [Google Scholar] [CrossRef] [PubMed]
- Fukui, T.; Yamaguchi, N. Systematic Review. In Minds Guide to Establish Clinical Guideline; Igakushoin: Tokyo, Japan, 2014; pp. 29–50. [Google Scholar]
- Noda, K.; Zhang, B.; Iwata, A.; Nishikawa, H.; Ogawa, M.; Nomiyama, T.; Miura, S.; Sako, H.; Matsuo, K.; Yahiro, E.; et al. Lifestyle changes through the use of delivered meals and dietary counseling in a single-blind study. STYLIST study. Circ. J. 2012, 76, 1335–1344. [Google Scholar] [CrossRef] [PubMed]
- Yamanouchi, T.; Isobe, K.; Terashima, H.; Hayashi, T.; Hiroshima, Y.; Hosaka, A. Intervention with delivered meals changes lifestyle and improves weight and glycemic control in patients with impaired glucose tolerance. J.J.C.L.A. 2017, 42, 26–30. (In Japanese) [Google Scholar]
- Enosawa, N.; Inoue, M.; Sato, T.; Suzuki, S. New trial in diet therapy for weight reduction in obese type 2 diabetic patients. J. Jap. Diab. Soc. 2004, 47, 635–641. (In Japanese) [Google Scholar]
- Miyashita, Y.; Itoh, Y.; Hashiguchi, S.; Shirai, K. Effect of low carbohydrate content of low calorie diet for obese non-insulin-dependent diabetes mellitus patients on glucose and lipid metabolism. J. Jap. Diab. Soc. 1998, 41, 885–890. (In Japanese) [Google Scholar]
- Sato, A.; Ueno, K.; Sugimoto, I. Diet therapy in diabetes mellitus. Nippon. Byouinkai Zasshi 2012, 59, 314–316. (In Japanese) [Google Scholar]
- Yamada, Y.; Uchida, J.; Izumi, H.; Tsukamoto, Y.; Inoue, G.; Watanabe, Y.; Irie, J.; Yamada, S. A non-calorie-restricted low-carbohydrate diet is effective as an alternative therapy for patients with type 2 diabetes. Intern. Med. 2014, 53, 13–19. [Google Scholar] [CrossRef] [PubMed]
- Sato, J.; Kanazawa, A.; Makita, S.; Hatae, C.; Komiya, K.; Shimizu, T.; Ikeda, F.; Tamura, Y.; Ogihara, T.; Mita, T.; et al. A randomized controlled trial of 130 g/day low-carbohydrate diet in type 2 diabetes with poor glycemic control. Clin. Nutr. 2017, 36, 992–1000. [Google Scholar] [CrossRef] [PubMed]
- Okada, K.; Ito, Y.; Kitagawa, M.; Maeda, K.; Nakamura, R. The effect of low carbohydrate diet for type 2 diabetic patients. Nippon Rinsho Eiyougakkai Zasshi 2013, 35, 103–113. (In Japanese) [Google Scholar]
- Nishimura, R.; Omiya, H.; Sugio, K.; Ubukata, M.; Sakai, S.; Samukawa, Y. Sodium-glucose cotransporter 2 inhibitor luseogliflozin improves glycaemic control, assessed by continuous glucose monitoring, even on a low-carbohydrate diet. Diabetes Obes. Metab. 2016, 18, 702–706. [Google Scholar] [CrossRef] [PubMed]
- Yabe, D.; Iwasaki, M.; Kuwata, H.; Haraguchi, T.; Hamamoto, Y.; Kurose, T.; Sumita, K.; Yamazato, H.; Kanada, S.; Seino, Y. Sodium-glucose co-transporter-2 inhibitor use and dietary carbohydrate intake in japanese individuals with type 2 diabetes: A randomized, open-label, 3-arm parallel comparative, exploratory study. Diabetes Obes. Metab. 2017, 19, 739–743. [Google Scholar] [CrossRef] [PubMed]
- Villareal, D.T.; Fontana, L.; Das, S.K.; Redman, L.; Smith, S.R.; Saltzman, E.; Bales, C.; Rochon, J.; Pieper, C.; Huang, M.; et al. Effect of two-year caloric restriction on bone metabolism and bone mineral density in non-obese younger adults: A randomized clinical trial. J. Bone Miner. Res. 2016, 31, 40–51. [Google Scholar] [CrossRef] [PubMed]
- Das, S.K.; Roberts, S.B.; Bhapkar, M.V.; Villareal, D.T.; Fontana, L.; Martin, C.K.; Racette, S.B.; Fuss, P.J.; Kraus, W.E.; Wong, W.W.; et al. Body-composition changes in the comprehensive assessment of long-term effects of reducing intake of energy (calerie)-2 study: A 2-y randomized controlled trial of calorie restriction in nonobese humans. Am. J. Clin. Nutr. 2017, 105, 913–927. [Google Scholar] [CrossRef] [PubMed]
- Fung, T.T. Low-carbohydrate diets and all-cause and cause-specific mortality. Ann. Intern. Med. 2010, 153, 289. [Google Scholar] [CrossRef] [PubMed]
- Noto, H.; Goto, A.; Tsujimoto, T.; Noda, M. Low-carbohydrate diets and all-cause mortality: A systematic review and meta-analysis of observational studies. PLoS ONE 2013, 8, e55030. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, Y.; Okuda, N.; Okamura, T.; Kadota, A.; Miyagawa, N.; Hayakawa, T.; Kita, Y.; Fujiyoshi, A.; Nagai, M.; Takashima, N.; et al. Low-carbohydrate diets and cardiovascular and total mortality in japanese: A 29-year follow-up of nippon data80. Columbia J. Nutr. 2014, 112, 916–924. [Google Scholar] [CrossRef] [PubMed]
- Dehghan, M.; Mente, A.; Zhang, X.; Swaminathan, S.; Li, W.; Mohan, V.; Iqbal, R.; Kumar, R.; Wentzel-Viljoen, E.; Rosengren, A.; et al. Associations of fats and carbohydrate intake with cardiovascular disease and mortality in 18 countries from five continents (PURE): A prospective cohort study. Lancet 2017, 390, 2050–2062. [Google Scholar] [CrossRef]
- Sato, J.; Kanazawa, A.; Hatae, C.; Makita, S.; Komiya, K.; Shimizu, T.; Ikeda, F.; Tamura, Y.; Ogihara, T.; Mita, T.; et al. One year follow-up after a randomized controlled trial of a 130g/day low-carbohydrate diet in patients with type 2 diabetes mellitus and poor glycemic control. PLoS ONE 2017, 12, e0188892. [Google Scholar] [CrossRef] [PubMed]
- Haimoto, H.; Iwata, M.; Wakai, K.; Umegaki, H. Long-term effects of a diet loosely restricting carbohydrates on HbA1c levels, BMI and tapering of sulfonylureas in type 2 diabetes: A 2-year follow-up study. Diabetes Res. Clin. Pract. 2008, 79, 350–356. [Google Scholar] [CrossRef] [PubMed]
- Sanada, M.; Kabe, C.; Hata, H.; Uchida, J.; Inoue, G.; Tsukamoto, Y.; Yamada, Y.; Irie, J.; Tabata, S.; Tabata, M.; et al. Efficacy of a moderate low carbohydrate diet in a 36-month observational study of Japanese patients with type 2 diabetes. Nutrients 2018, 10, 528. [Google Scholar] [CrossRef] [PubMed]
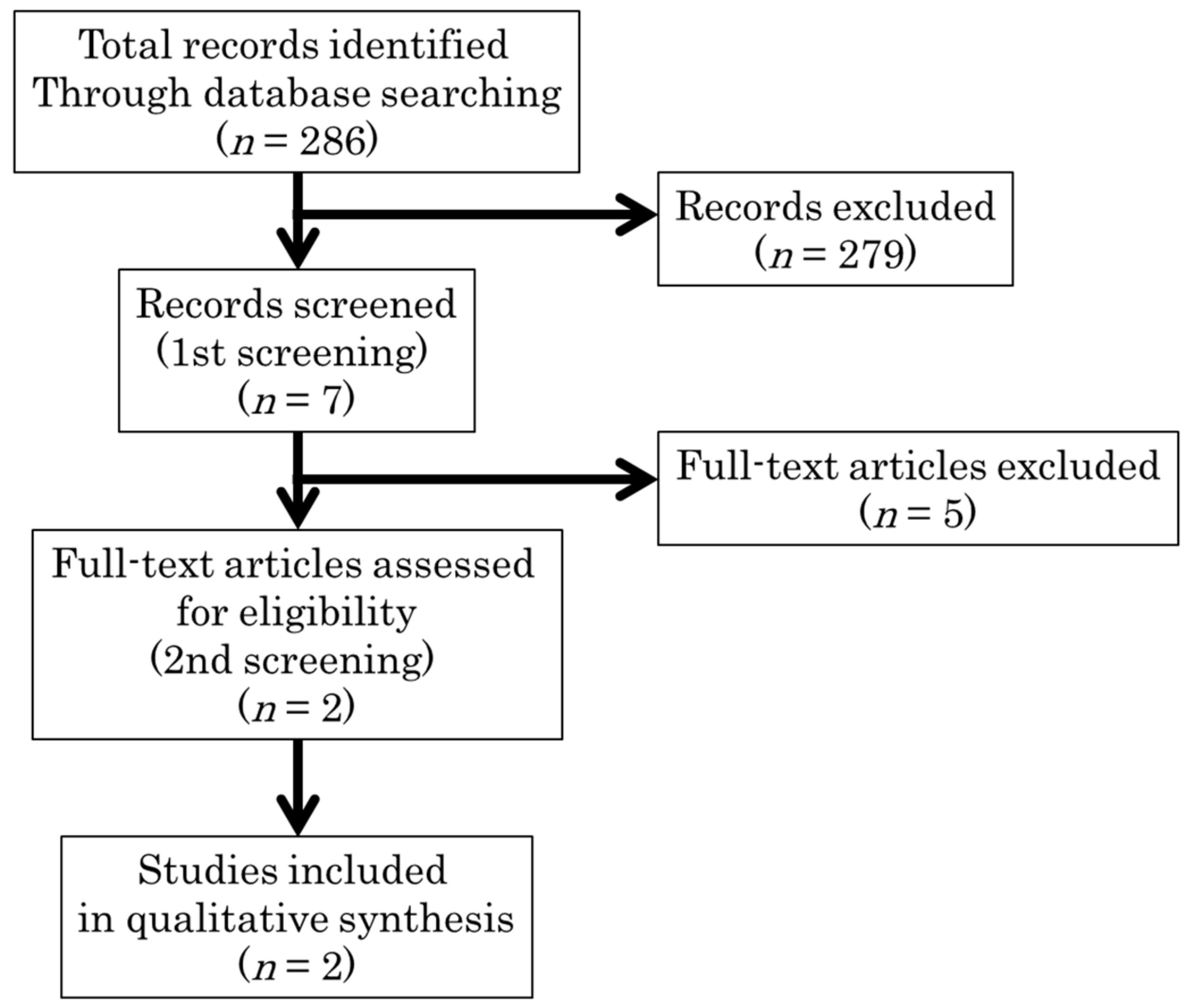
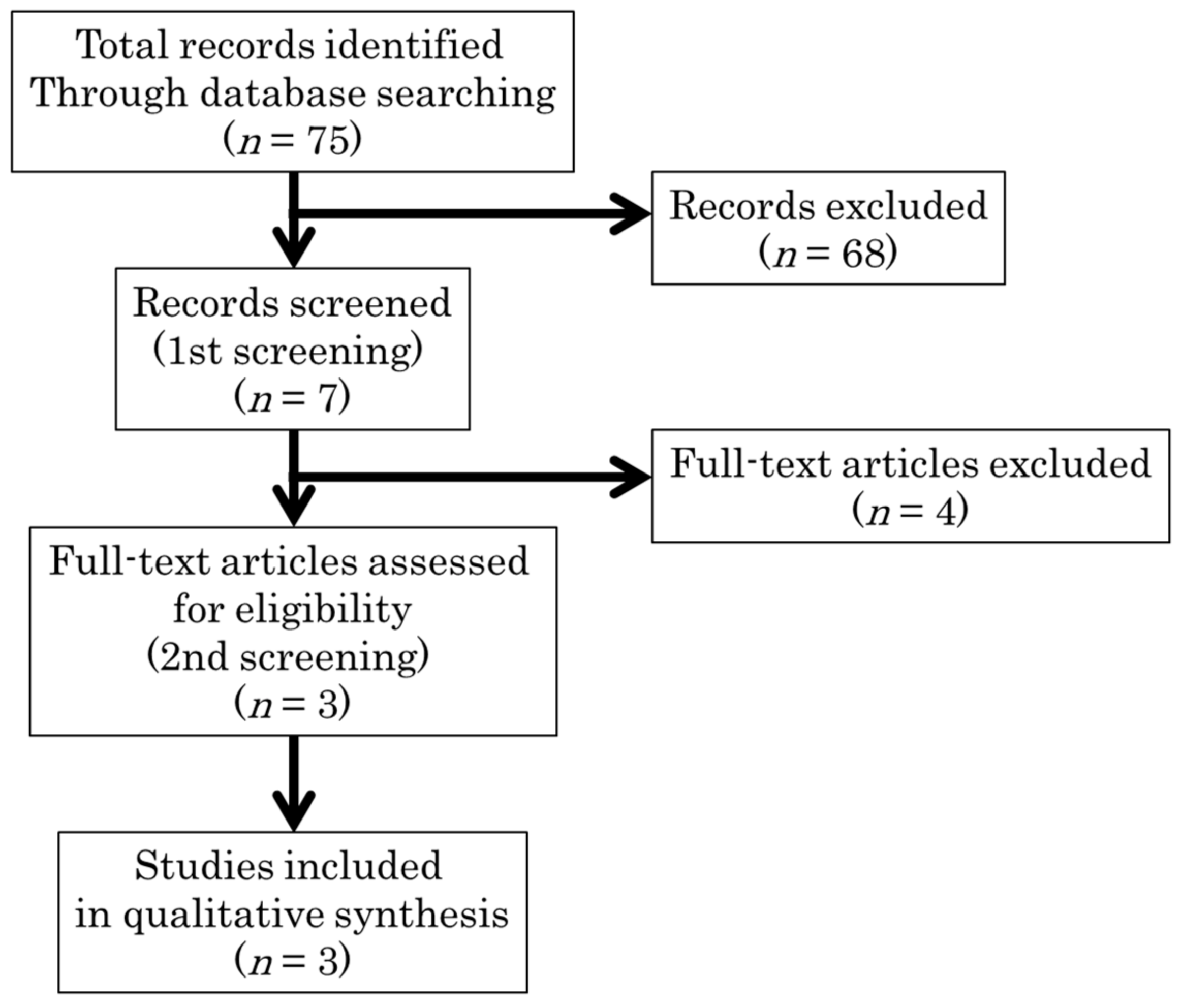
| Study ID (Reference Number) | Setting | Study Design | Patients | Intervention | Control | Primary Outcome | Results | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Number of Participants | Number of Dropouts | Age | Sex (M/F) | Duration of Diabetes (Years) | Intervention | Control | Median Difference | ||||||
| Yamada et al., 2014 [21] | Outpatient University Hospital | RCT | 24 | C: 0 E: 0 | C: 63.3 ± 13.5 E: 63.2 ± 10.2 | C: 7/5 E: 5/7 | C: 8.9 ± 3.6 E: 9.5 ± 4.8 | Carbohydrate: 70–130 g/day | IBW (kg) ×25 kcal/day | HbA1c change after 6 month | −0.6 ± 0.48 | −0.2 ± 0.68 | −0.40 |
| Sato et al., 2017 [22] | Outpatient University Hospital | RCT | 66 | C: 1 E: 3 | C: 60.5 ± 10.5 E: 58.4 ± 10.0 | C: 23/7 E: 24/8 | (median) C: 14.0 E: 13.0 | Carbohydrate 130 g/day | IBW (kg) x 28 kcal/day | HbA1c change after 6 month | (median)-0.65 | (median) 0.00 | −0.65 |
| Yabe et al., 2017 [25] | Meal test 2 Medical Institutions | RCT | 15 | C: 0 H: 0 | C: 56.9 ± 7.3 H: 54.3 ± 5.2 | C: 7/0 E: 5/3 | C: 7.6 ± 4.3 E: 4.4 ± 3.3 | Carbohydrate 180 g/day, Energy 1800 kcal/d | Carbohydrate 247.5 g/day, Energy 1800 kcal/day | CGM data during 5−7 days | 130.32 ± 27.72 | 142.92 ± 39.6 | n.a. |
| Study ID [Reference Number] | Sample Size Calculation | Risk of Bias | Indirectness | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Random Sequence Generation | Allocation Concealment * | Blinding of Participants and Personnel * | Blinding of Outcome Assessment * | Incomplete Outcome Data | Selective Reporting | Other Source of Bias | Study free from total biases * | Subjects | Intervention | Control | Outcome | Total Indirectness | ||
| Yamada et al., 2014 [21] | Yes | Yes | Low | Unclear | Unclear | Low | Low | Unclear | Unclear | No | No | No | No | No |
| Sato et al., 2017 [22] | Yes | Yes | Low | Unclear | Unclear | Low | Low | Unclear | Unclear | Yes ** | Yes *** | No | No | No |
| Yabe et al., 2017 [25] | n.a. | Yes | Low | Unclear | Unclear | Low | Low | Unclear | Unclear | Yes ** | Yes **** | Yes **** | No | Yes |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yamada, S.; Kabeya, Y.; Noto, H. Dietary Approaches for Japanese Patients with Diabetes: A Systematic Review. Nutrients 2018, 10, 1080. https://doi.org/10.3390/nu10081080
Yamada S, Kabeya Y, Noto H. Dietary Approaches for Japanese Patients with Diabetes: A Systematic Review. Nutrients. 2018; 10(8):1080. https://doi.org/10.3390/nu10081080
Chicago/Turabian StyleYamada, Satoru, Yusuke Kabeya, and Hiroshi Noto. 2018. "Dietary Approaches for Japanese Patients with Diabetes: A Systematic Review" Nutrients 10, no. 8: 1080. https://doi.org/10.3390/nu10081080
APA StyleYamada, S., Kabeya, Y., & Noto, H. (2018). Dietary Approaches for Japanese Patients with Diabetes: A Systematic Review. Nutrients, 10(8), 1080. https://doi.org/10.3390/nu10081080





