Associations between Fatty Acid Intake and Status, Desaturase Activities, and FADS Gene Polymorphism in Centrally Obese Postmenopausal Polish Women
Abstract
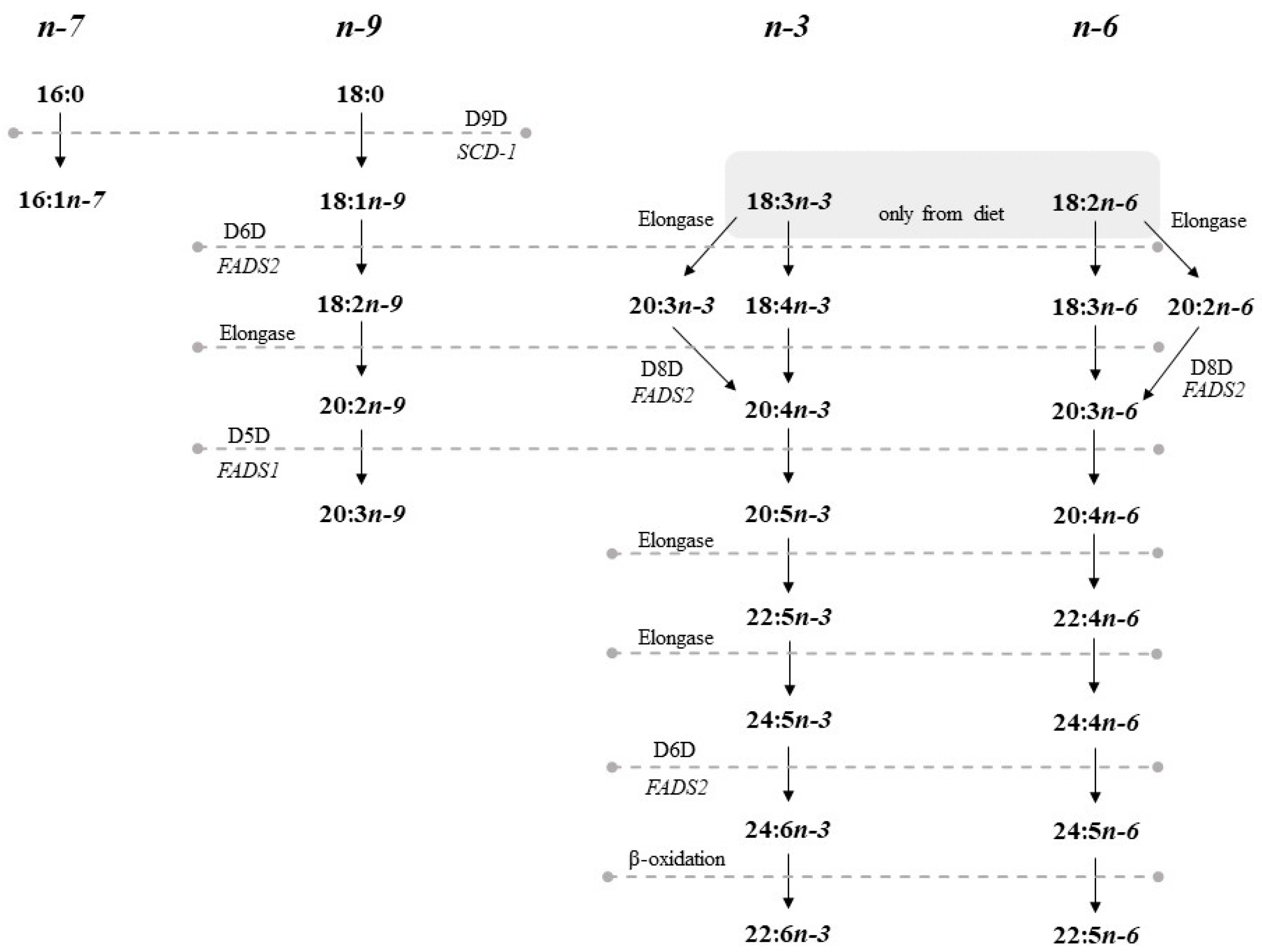
1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Dietary Assessment
2.3. Anthropometry
2.4. Physical Activity
2.5. Analysis of Fatty Acid Profile in Erythrocytes
2.6. Desaturase and Elongase Activities
2.7. Genotyping
2.8. Statistical Analysis
3. Results
3.1. Subjects Characteristics
3.2. Dietary Intake of Macronutrients and FA
3.3. Associations between Macronutrient Intake and FA Composition In Erythrocyte Membranes
3.4. Associations between FADS Genotype and FA Concentrations in Erythrocyte Membranes and Enzyme Activities
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Murff, H.J.; Edwards, T.L. Endogenous Production of Long-Chain Polyunsaturated Fatty Acids and Metabolic Disease Risk. Curr. Cardiovasc. Risk Rep. 2014, 8, 418. [Google Scholar] [CrossRef] [PubMed]
- Dubnov, G.; Brzezinski, A.; Berry, E.M. Weight control and the management of obesity after menopause: The role of physical activity. Maturitas 2003, 44, 89–101. [Google Scholar] [CrossRef]
- Lobo, R.A. Metabolic syndrome after menopause and the role of hormones. Maturitas 2008, 60, 10–18. [Google Scholar] [CrossRef] [PubMed]
- Mastorakos, G.; Valsamakis, G.; Paltoglou, G.; Creatsas, G. Management of obesity in menopause: Diet, exercise, pharmacotherapy and bariatric surgery. Maturitas 2010, 65, 219–224. [Google Scholar] [CrossRef] [PubMed]
- Vryonidou, A.; Paschou, S.A.; Muscogiuri, G.; Orio, F.; Goulis, D.G. Mechanisms in endocrinology: Metabolic syndrome through the female life cycle. Eur. J. Endocrinol. 2015, 173, R153–R163. [Google Scholar] [CrossRef] [PubMed]
- Rautiainen, S.; Wang, L.; Lee, I.-M.; Manson, J.E.; Buring, J.E.; Sesso, H.D. Higher Intake of Fruit, but Not Vegetables or Fiber, at Baseline Is Associated with Lower Risk of Becoming Overweight or Obese in Middle-Aged and Older Women of Normal BMI at Baseline. J. Nutr. 2015, 145, 960–968. [Google Scholar] [CrossRef] [PubMed]
- De Almeida Ventura, D.; de Matos Fonseca, V.; Ramos, E.G.; Marinheiro, L.P.F.; de Souza, R.A.G.; de Miranda Chaves, C.R.M.; Peixoto, M.V.M. Association between quality of the diet and cardiometabolic risk factors in postmenopausal women. Nutr. J. 2014, 13, 121. [Google Scholar] [CrossRef] [PubMed]
- Tardivo, A.P.; Nahas-Neto, J.; Nahas, E.A.P.; Maesta, N.; Rodrigues, M.A.; Orsatti, F.L. Associations between healthy eating patterns and indicators of metabolic risk in postmenopausal women. Nutr. J. 2010, 9, 64. [Google Scholar] [CrossRef] [PubMed]
- Grygiel-Górniak, B.; Marcinkowska, J.; Szczepanik, A.; Przysławski, J. Nutritional habits and oxidative stress in postmenopausal age. Pol. Arch. Med. Wewn. 2014, 124, 298–305. [Google Scholar] [CrossRef] [PubMed]
- Grygiel-Górniak, B.; Kaczmarek, E.; Mosor, M.; Przysławski, J.; Nowak, J. The gene-diet associations in postmenopausal women with newly diagnosed dyslipidemia. J. Nutr. Heal. Aging 2017, 21, 1031–1037. [Google Scholar] [CrossRef] [PubMed]
- Schaeffer, L.; Gohlke, H.; Müller, M.; Heid, I.M.; Palmer, L.J.; Kompauer, I.; Demmelmair, H.; Illig, T.; Koletzko, B.; Heinrich, J. Common genetic variants of the FADS1 FADS2 gene cluster and their reconstructed haplotypes are associated with the fatty acid composition in phospholipids. Hum. Mol. Genet. 2006, 15, 1745–1756. [Google Scholar] [CrossRef] [PubMed]
- Rzehak, P.; Heinrich, J.; Klopp, N.; Schaeffer, L.; Hoff, S.; Wolfram, G.; Illig, T.; Linseisen, J. Evidence for an association between genetic variants of the fatty acid desaturase 1 fatty acid desaturase 2 (FADS1 FADS2) gene cluster and the fatty acid composition of erythrocyte membranes. Br. J. Nutr. 2009, 101, 20. [Google Scholar] [CrossRef] [PubMed]
- Malerba, G.; Schaeffer, L.; Xumerle, L.; Klopp, N.; Trabetti, E.; Biscuola, M.; Cavallari, U.; Galavotti, R.; Martinelli, N.; Guarini, P.; et al. SNPs of the FADS Gene Cluster are Associated with Polyunsaturated Fatty Acids in a Cohort of Patients with Cardiovascular Disease. Lipids 2008, 43, 289–299. [Google Scholar] [CrossRef] [PubMed]
- Zietemann, V.; Kröger, J.; Enzenbach, C.; Jansen, E.; Fritsche, A.; Weikert, C.; Boeing, H.; Schulze, M.B. Genetic variation of the FADS1 FADS2 gene cluster and n-6 PUFA composition in erythrocyte membranes in the European Prospective Investigation into Cancer and Nutrition-Potsdam study. Br. J. Nutr. 2018, 104, 1748–1759. [Google Scholar] [CrossRef] [PubMed]
- Chilton, F.; Murphy, R.; Wilson, B.; Sergeant, S.; Ainsworth, H.; Seeds, M.; Mathias, R. Diet-Gene Interactions and PUFA Metabolism: A Potential Contributor to Health Disparities and Human Diseases. Nutrients 2014, 6, 1993–2022. [Google Scholar] [CrossRef] [PubMed]
- Minihane, A.M. Impact of Genotype on EPA and DHA Status and Responsiveness to Increased Intakes. Nutrients 2016, 8, 123. [Google Scholar] [CrossRef] [PubMed]
- Chilton, F.H.; Dutta, R.; Reynolds, L.M.; Sergeant, S.; Mathias, R.A.; Seeds, M.C. Precision nutrition and omega-3 polyunsaturated fatty acids: A case for personalized supplementation approaches for the prevention and management of human diseases. Nutrients 2017, 9, 1165. [Google Scholar] [CrossRef] [PubMed]
- Mathias, R.A.; Pani, V.; Chilton, F.H. Genetic Variants in the FADS Gene: Implications for Dietary Recommendations for Fatty Acid Intake. Curr. Nutr. Rep. 2014, 3, 139–148. [Google Scholar] [CrossRef] [PubMed]
- Hallmann, J.; Kolossa, S.; Gedrich, K.; Celis-Morales, C.; Forster, H.; O’Donovan, C.B.; Woolhead, C.; Macready, A.L.; Fallaize, R.; Marsaux, C.F.M.; et al. Predicting fatty acid profiles in blood based on food intake and the FADS1 rs174546 SNP. Mol. Nutr. Food Res. 2015, 59, 2565–2573. [Google Scholar] [CrossRef] [PubMed]
- Baylin, A.; Ruiz-Narvaez, E.; Kraft, P.; Campos, H. α-Linolenic acid, Δ6-desaturase gene polymorphism, and the risk of nonfatal myocardial infarction. Am. J. Clin. Nutr. 2007, 85, 554–560. [Google Scholar] [CrossRef] [PubMed]
- Illig, T.; Gieger, C.; Zhai, G.; Römisch-Margl, W.; Wang-Sattler, R.; Prehn, C.; Altmaier, E.; Kastenmüller, G.; Kato, B.S.; Mewes, H.-W.; et al. A genome-wide perspective of genetic variation in human metabolism. Nat. Genet. 2010, 42, 137–141. [Google Scholar] [CrossRef] [PubMed]
- Moltó-Puigmartí, C.; Plat, J.; Mensink, R.P.; Müller, A.; Jansen, E.; Zeegers, M.P.; Thijs, C. FADS1 FADS2 gene variants modify the association between fish intake and the docosahexaenoic acid proportions in human milk. Am. J. Clin. Nutr. 2010, 91, 1368–1376. [Google Scholar] [CrossRef] [PubMed]
- Bokor, S.; Dumont, J.; Spinneker, A.; Gonzalez-Gross, M.; Nova, E.; Widhalm, K.; Moschonis, G.; Stehle, P.; Amouyel, P.; De Henauw, S.; et al. Single nucleotide polymorphisms in the FADS gene cluster are associated with delta-5 and delta-6 desaturase activities estimated by serum fatty acid ratios. J. Lipid Res. 2010, 51, 2325–2333. [Google Scholar] [CrossRef] [PubMed]
- Rzehak, P.; Thijs, C.; Standl, M.; Mommers, M.; Glaser, C.; Jansen, E.; Klopp, N.; Koppelman, G.H.; Singmann, P.; Postma, D.S.; et al. LISA study group Variants of the FADS1 FADS2 gene cluster, blood levels of polyunsaturated fatty acids and eczema in children within the first 2 years of life. PLoS ONE 2010, 5, e13261. [Google Scholar] [CrossRef] [PubMed]
- Koletzko, B.; Lattka, E.; Zeilinger, S.; Illig, T.; Steer, C. Genetic variants of the fatty acid desaturase gene cluster predict amounts of red blood cell docosahexaenoic and other polyunsaturated fatty acids in pregnant women: Findings from the Avon Longitudinal Study of Parents and Children. Am. J. Clin. Nutr. 2011, 93, 211–219. [Google Scholar] [CrossRef] [PubMed]
- Lattka, E.; Rzehak, P.; Szabó, É.; Jakobik, V.; Weck, M.; Weyermann, M.; Grallert, H.; Rothenbacher, D.; Heinrich, J.; Brenner, H.; et al. Genetic variants in the FADS gene cluster are associated with arachidonic acid concentrations of human breast milk at 1.5 and 6 mo postpartum and influence the course of milk dodecanoic, tetracosenoic, and trans-9-octadecenoic acid concentrations over the duration of lactation. Am. J. Clin. Nutr. 2011, 93, 382–391. [Google Scholar] [CrossRef] [PubMed]
- Sergeant, S.; Hugenschmidt, C.E.; Rudock, M.E.; Ziegler, J.T.; Ivester, P.; Ainsworth, H.C.; Vaidya, D.; Douglas Case, L.; Langefeld, C.D.; Freedman, B.I.; et al. Differences in arachidonic acid levels and fatty acid desaturase (FADS) gene variants in African Americans and European Americans with diabetes or the metabolic syndrome. Br. J. Nutr. 2012, 107, 547–555. [Google Scholar] [CrossRef] [PubMed]
- Xie, L.; Innis, S.M. Genetic Variants of the FADS1 FADS2 Gene Cluster Are Associated with Altered (n-6) and (n-3) Essential Fatty Acids in Plasma and Erythrocyte Phospholipids in Women during Pregnancy and in Breast Milk during Lactation. J. Nutr. 2008, 138, 2222–2228. [Google Scholar] [CrossRef] [PubMed]
- Horiguchi, S.; Nakayama, K.; Iwamoto, S.; Ishijima, A.; Minezaki, T.; Baba, M.; Kontai, Y.; Horikawa, C.; Kawashima, H.; Shibata, H.; et al. Associations between a fatty acid desaturase gene polymorphism and blood arachidonic acid compositions in Japanese elderly. Prostaglandins Leukot. Essent. Fat. Acids 2015, 105, 9–14. [Google Scholar] [CrossRef] [PubMed]
- Martinelli, N.; Girelli, D.; Malerba, G.; Guarini, P.; Illig, T.; Trabetti, E.; Sandri, M.; Friso, S.; Pizzolo, F.; Schaeffer, L.; et al. FADS genotypes and desaturase activity estimated by the ratio of arachidonic acid to linoleic acid are associated with inflammation and coronary artery disease. Am. J. Clin. Nutr. 2008, 88, 941–949. [Google Scholar] [CrossRef] [PubMed]
- Mathias, R.A.; Vergara, C.; Gao, L.; Rafaels, N.; Hand, T.; Campbell, M.; Bickel, C.; Ivester, P.; Sergeant, S.; Barnes, K.C.; et al. FADS genetic variants and ω-6 polyunsaturated fatty acid metabolism in a homogeneous island population. J. Lipid Res. 2010, 51, 2766. [Google Scholar] [CrossRef] [PubMed]
- Schuchardt, J.P.; Köbe, T.; Witte, V.; Willers, J.; Gingrich, A.; Tesky, V.; Pantel, J.; Rujescu, D.; Illig, T.; Flöel, A.; et al. Genetic variants of the FADS gene cluster are associated with erythrocyte membrane LC PUFA levels in patients with mild cognitive impairment. J. Nutr. Heal. Aging 2016, 20, 611–620. [Google Scholar] [CrossRef] [PubMed]
- Bajerska, J.; Chmurzynska, A.; Muzsik, A.; Krzyżanowska, P.; Mądry, E.; Malinowska, A.M.; Walkowiak, J. Weight loss and metabolic health effects from energy-restricted Mediterranean and Central-European diets in postmenopausal women: A randomized controlled trial. Sci. Rep. 2018, 8, 11170. [Google Scholar] [CrossRef] [PubMed]
- Ortega, R.M.; Pérez-Rodrigo, C.; López-Sobaler, A.M. Dietary assessment methods: Dietary records. Nutr. Hosp. 2015, 31, 38–45. [Google Scholar] [CrossRef] [PubMed]
- Jarosz, M. Nutrition Standards for the Polish Population-Amendment (Report in Polish). Warsaw. 2017. Available online: http://www.izz.waw.pl/pl/normy-zwienia (accessed on 20 January 2018).
- Zimmet, P.; Magliano, D.; Matsuzawa, Y.; Alberti, G.; Shaw, J. The metabolic syndrome: A global public health problem and a new definition. J. Atheroscler. Thromb. 2005, 12, 295–300. [Google Scholar] [CrossRef] [PubMed]
- Craig, C.L.; Marshall, A.L.; Sjöström, M.; Bauman, A.E.; Booth, M.L.; Ainsworth, B.E.; Pratt, M.; Ekelund, U.; Yngve, A.; Sallis, J.F.; et al. International Physical Activity Questionnaire: 12-Country Reliability and Validity. Med. Sci. Sport. Exerc. 2003, 35, 1381–1395. [Google Scholar] [CrossRef] [PubMed]
- Folch, J.; Lees, M.; Sloane Stanley, G.H. A simple method for the isolation and purification of total lipides from animal tissues. J. Biol. Chem. 1957, 226, 497–509. [Google Scholar] [PubMed]
- Jacobs, S.; Jäger, S.; Jansen, E.; Peter, A.; Stefan, N.; Boeing, H.; Schulze, M.B.; Kröger, J. Associations of Erythrocyte Fatty Acids in the De Novo Lipogenesis Pathway with Proxies of Liver Fat Accumulation in the EPIC-Potsdam Study. PLoS ONE 2015, 10, e0127368. [Google Scholar] [CrossRef] [PubMed]
- Paglialunga, S.; Dehn, C.A. Clinical assessment of hepatic de novo lipogenesis in non-alcoholic fatty liver disease. Lipids Health Dis. 2016, 15, 159. [Google Scholar] [CrossRef] [PubMed]
- Novak, E.M.; Innis, S.M. Dietary long chain n-3 fatty acids are more closely associated with protein than energy intakes from fat. Prostaglandins Leukot. Essent. Fat. Acids 2012, 86, 107–112. [Google Scholar] [CrossRef] [PubMed]
- Stark, K.D.; Patterson, A.C. EPA and DHA—Protein, not fat is “Where It’s at”? Prostaglandins Leukot. Essent. Fat. Acids 2012, 87, 49–51. [Google Scholar] [CrossRef] [PubMed]
- Richter, C.K.; Skulas-Ray, A.C.; Kris-Etherton, P.M. Recommended Intake of Fish and Fish Oils Worldwide. In Fish and Fish Oil in Health and Disease Prevention; Elsevier: Amsterdam, Netherlands, 2016; pp. 27–48. [Google Scholar]
- Sygnowska, E.; Waśkiewicz, A.; Głuszek, J.; Kwaśniewska, M.; Biela, U.; Kozakiewicz, K.; Zdrojewski, T.; Rywik, S. Kardiologia Polska; Polskie Towarzystwo Kardiologiczne: Warszawa, Polska, 2005. [Google Scholar]
- Niedźwiedzka, E.; Wądołowska, L. Analiza urozmaicenia spożycia żywności w kontekście statusu socjoekonomicznego polskich osób starszych Analysis of food intake variety in relation to the socio-economic status of elderly Polish citizens. Probl. Hig. Epidemiol. 2010, 91, 576–584. [Google Scholar]
- Mozaffarian, D.; Pischon, T.; Hankinson, S.E.; Rifai, N.; Joshipura, K.; Willett, W.C.; Rimm, E.B. Dietary intake of trans fatty acids and systemic inflammation in women. Am. J. Clin. Nutr. 2004, 79, 606–612. [Google Scholar] [CrossRef] [PubMed]
- Dawczynski, C.; Massey, K.A.; Ness, C.; Kiehntopf, M.; Stepanow, S.; Platzer, M.; Grün, M.; Nicolaou, A.; Jahreis, G. Randomized placebo-controlled intervention with n-3 LC-PUFA-supplemented yoghurt: Effects on circulating eicosanoids and cardiovascular risk factors. Clin. Nutr. 2013, 32, 686–696. [Google Scholar] [CrossRef] [PubMed]
- Harris, W.S.; Luo, J.; Pottala, J.V.; Margolis, K.L.; Espeland, M.A.; Robinson, J.G. Red Blood Cell Fatty Acids and Incident Diabetes Mellitus in the Women’s Health Initiative Memory Study. PLoS ONE 2016, 11, e0147894. [Google Scholar] [CrossRef] [PubMed]
- Flock, M.R.; Skulas-Ray, A.C.; Harris, W.S.; Etherton, T.D.; Fleming, J.A.; Kris-Etherton, P.M. Determinants of erythrocyte omega-3 fatty acid content in response to fish oil supplementation: A dose-response randomized controlled trial. J. Am. Heart Assoc. 2013, 2, e000513. [Google Scholar] [CrossRef] [PubMed]
- Harris, W.S.; Pottala, J.V.; Varvel, S.A.; Borowski, J.J.; Ward, J.N.; McConnell, J.P. Erythrocyte omega-3 fatty acids increase and linoleic acid decreases with age: Observations from 160,000 patients. Prostaglandins Leukot. Essent. Fat. Acids 2013, 88, 257–263. [Google Scholar] [CrossRef] [PubMed]
- Kroger, J.; Zietemann, V.; Enzenbach, C.; Weikert, C.; Jansen, E.H.; Doring, F.; Joost, H.-G.; Boeing, H.; Schulze, M.B. Erythrocyte membrane phospholipid fatty acids, desaturase activity, and dietary fatty acids in relation to risk of type 2 diabetes in the European Prospective Investigation into Cancer and Nutrition (EPIC)-Potsdam Study. Am. J. Clin. Nutr. 2011, 93, 127–142. [Google Scholar] [CrossRef] [PubMed]
- Vessby, B.; Gustafsson, I.-B.; Tengblad, S.; Berglund, L. Indices of fatty acid desaturase activity in healthy human subjects: Effects of different types of dietary fat. Br. J. Nutr. 2013, 110, 871–879. [Google Scholar] [CrossRef] [PubMed]
- Wood, K.E.; Lau, A.; Mantzioris, E.; Gibson, R.A.; Ramsden, C.E.; Muhlhausler, B.S. A low omega-6 polyunsaturated fatty acid (n-6 PUFA) diet increases omega-3 (n-3) long chain PUFA status in plasma phospholipids in humans. Prostaglandins Leukot. Essent. Fat. Acids 2014, 90, 133–138. [Google Scholar] [CrossRef] [PubMed]
- Wood, K.E.; Mantzioris, E.; Gibson, R.A.; Ramsden, C.E.; Muhlhausler, B.S. The effect of modifying dietary LA and ALA intakes on omega-3 long chain polyunsaturated fatty acid (n-3 LCPUFA) status in human adults: A systematic review and commentary. Prostaglandins Leukot. Essent. Fat. Acids 2015, 95, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Matthan, N.R.; Ooi, E.M.; Van Horn, L.; Neuhouser, M.L.; Woodman, R.; Lichtenstein, A.H. Plasma phospholipid fatty acid biomarkers of dietary fat quality and endogenous metabolism predict coronary heart disease risk: A nested case-control study within the Women’s Health Initiative observational study. J. Am. Heart Assoc. 2014, 3. [Google Scholar] [CrossRef] [PubMed]
- Mayneris-Perxachs, J.; Guerendiain, M.; Castellote, A.I.; Estruch, R.; Covas, M.I.; Fitó, M.; Salas-Salvadó, J.; Martínez-González, M.A.; Aros, F.; Lamuela-Raventós, R.M.; et al. Plasma fatty acid composition, estimated desaturase activities, and their relation with the metabolic syndrome in a population at high risk of cardiovascular disease. Clin. Nutr. 2014, 33, 90–97. [Google Scholar] [CrossRef] [PubMed]
- Warensjö, E.; Öhrvall, M.; Vessby, B. Fatty acid composition and estimated desaturase activities are associated with obesity and lifestyle variables in men and women. Nutr. Metab. Cardiovasc. Dis. 2006, 16, 128–136. [Google Scholar] [CrossRef] [PubMed]
- Warensjö, E.; Sundström, J.; Lind, L.; Vessby, B. Factor analysis of fatty acids in serum lipids as a measure of dietary fat quality in relation to the metabolic syndrome in men1–3. Am. J. Clin. Nutr. 2006, 84, 442–448. [Google Scholar] [CrossRef] [PubMed]
- Kabagambe, E.K.; Tsai, M.Y.; Hopkins, P.N.; Ordovas, J.M.; Peacock, J.M.; Borecki, I.B.; Arnett, D.K. Erythrocyte fatty acid composition and the metabolic syndrome: A National Heart, Lung, and Blood Institute GOLDN study. Clin. Chem. 2008, 54, 154–162. [Google Scholar] [CrossRef] [PubMed]
- Huang, T.; Bhulaidok, S.; Cai, Z.; Xu, T.; Xu, F.; Wahlqvist, M.L.; Li, D. Plasma phospholipids n-3 polyunsaturated fatty acid is associated with metabolic syndrome. Mol. Nutr. Food Res. 2010, 54, 1628–1635. [Google Scholar] [CrossRef] [PubMed]
- Cinti, D.L.; Cook, L.; Nagi, M.N.; Suneja, S.K. The fatty acid chain elongation system of mammalian endoplasmic reticulum. Prog. Lipid Res. 1992, 31, 1–51. [Google Scholar] [CrossRef]
- Obukowicz, M.G.; Welsch, D.J.; Salsgiver, W.J.; Martin-Berger, C.L.; Chinn, K.S.; Duffin, K.L.; Raz, A.; Needleman, P. Novel, selective delta6 or delta5 fatty acid desaturase inhibitors as antiinflammatory agents in mice. J. Pharmacol. Exp. Ther. 1998, 287, 157–166. [Google Scholar] [PubMed]
- Lemaitre, R.N.; Tanaka, T.; Tang, W.; Manichaikul, A.; Foy, M.; Kabagambe, E.K.; Nettleton, J.A.; King, I.B.; Weng, L.-C.; Bhattacharya, S.; et al. Genetic loci associated with plasma phospholipid n-3 fatty acids: A meta-analysis of genome-wide association studies from the CHARGE Consortium. PLoS Genet. 2011, 7, e1002193. [Google Scholar] [CrossRef] [PubMed]
- Domei, T.; Yokoi, H.; Kuramitsu, S.; Soga, Y.; Arita, T.; Ando, K.; Shirai, S.; Kondo, K.; Sakai, K.; Goya, M.; et al. Ratio of serum n-3 to n-6 polyunsaturated fatty acids and the incidence of major adverse cardiac events in patients undergoing percutaneous coronary intervention. Circ. J. 2012, 76, 423–429. [Google Scholar] [CrossRef] [PubMed]
- Warensjö, E.; Sundström, J.; Vessby, B.; Cederholm, T.; Risérus, U. Markers of dietary fat quality and fatty acid desaturation as predictors of total and cardiovascular mortality: A population-based prospective study. Am. J. Clin. Nutr. 2008, 88, 203–209. [Google Scholar] [CrossRef] [PubMed]
- Das, U.N. Essential fatty acids: Biochemistry, physiology and pathology. Biotechnol. J. 2006, 1, 420–439. [Google Scholar] [CrossRef] [PubMed]
- Do, H.J.; Chung, H.K.; Moon, J.; Shin, M.-J. Relationship between the estimates of desaturase activities and cardiometabolic phenotypes in Koreans. J. Clin. Biochem. Nutr. 2011, 49, 131–135. [Google Scholar] [CrossRef] [PubMed]
- Vessby, B.; Tengblad, S.; Lithell, H. Insulin sensitivity is related to the fatty acid composition of serum lipids and skeletal muscle phospholipids in 70-year-old men. Diabetologia 1994, 37, 1044–1050. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Medeiros, L.; Revesz, E.; O’Dorisio, T. Desaturation Function Does Not Decline after Menopause in Human Females. Horm. Metab. Res. 2000, 32, 26–32. [Google Scholar] [CrossRef] [PubMed]
- Medeiros, L.C.; Liu, Y.W.; Park, S.; Chang, P.H.; Smith, A.M. Insulin, but not estrogen, correlated with indexes of desaturase function in obese women. Horm. Metab. Res. 1995, 27, 235–238. [Google Scholar] [CrossRef] [PubMed]
- James, M.J.; Ursin, V.M.; Cleland, L.G. Metabolism of stearidonic acid in human subjects: Comparison with the metabolism of other n−3 fatty acids. Am. J. Clin. Nutr. 2003, 77, 1140–1145. [Google Scholar] [CrossRef] [PubMed]
- Sands, S.A.; Reid, K.J.; Windsor, S.L.; Harris, W.S. The impact of age, body mass index, and fish intake on the EPA and DHA content of human erythrocytes. Lipids 2005, 40, 343–347. [Google Scholar] [CrossRef] [PubMed]
- Cao, J.; Schwichtenberg, K.A.; Hanson, N.Q.; Tsai, M.Y. Incorporation and Clearance of Omega-3 Fatty Acids in Erythrocyte Membranes and Plasma Phospholipids. Clin. Chem. 2006, 52, 2265–2272. [Google Scholar] [CrossRef] [PubMed]
- Jump, D.B. Dietary polyunsaturated fatty acids and regulation of gene transcription. Curr. Opin. Lipidol. 2002, 13, 155–164. [Google Scholar] [CrossRef] [PubMed]
| Gene | SNP | Primers | Probes |
|---|---|---|---|
| FADS1 | rs174556 (C > T) | 5’ACAAGGGCCTTGTGAAGAAGT 5’GCCTGTGACCTCATGACTATGC | 5’GAGTCTAGATGGaATCACAGTCATAGT--FL |
| rs174561 (T > C) | 5’GCACCACACATACGGACCAAT 5’GGGTCAACCAGAGTGACCACTC | 5’ GCATCCCCGGCCCCA--FL | |
| rs174547 (T > C) | 5’TGGGTGACACAGATGAACCATATTC 5’GGCTAATGAGAAAATGCTGTTTGG | 5’CTACGCACCCTTTTCAATAGTTG--FL | |
| FADS2 | rs3834458 (delT) | 5’TTACTGAGACCAGGGCAAGGAC 5’CGGCAGTCGAGACTCCAGTATC | 5’TCAGACAATCTT_GAAAAGAATTGC |
| Macronutrient. | Daily Intake | |||||
|---|---|---|---|---|---|---|
| Mean ± SEM | Median | Quartile 1 | Quartile 2 | Quartile 3 | Quartile 4 | |
| Total protein (g) | 67.6 ± 1.6 | 66.5 | 45.5 ± 1.2 | 61.2 ± 0.6 | 71.0 ± 0.5 | 93.3 ± 2.6 |
| Total carbohydrates (g) a | 255.7 ± 11.1 | 229.5 | 128.6 ± 3.8 | 191.6 ± 3.3 | 265.6 ± 3.9 | 437.2 ± 20.5 |
| Total lipids (g) | 52.4 ± 2.0 | 47.4 | 27.4 ± 0.8 | 41.0 ± 0.7 | 57.4 ± 0.9 | 83.8 ± 3.6 |
| Total SFAs (g) | 20.1 ± 0.9 | 18.4 | 9.9 ± 0.3 | 15.2 ± 0.3 | 21.7 ± 0.3 | 33.7 ± 1.8 |
| 4:0 (mg) | 315.4 ± 26.7 | 258.3 | 90.7 ± 6.3 | 201.1 ± 4.8 | 315.8 ± 5.6 | 654.2 ± 79.3 |
| 6:0 (mg) | 211.6 ± 16.8 | 171.6 | 71.7 ± 4.4 | 135.0 ± 2.9 | 210.8 ± 3.7 | 428.7 ± 49.6 |
| 8:0 (mg) | 153.2 ± 10.7 | 129.9 | 56.7 ± 3.0 | 104.5 ± 2.4 | 156.4 ± 2.7 | 295.3 ± 30.2 |
| 10:0 (mg) | 378.6 ± 26.1 | 318.6 | 136.0 ± 6.4 | 258.0 ± 5.9 | 379.9 ± 6.6 | 740.5 ± 70.7 |
| 12:0 (mg) | 558.3 ± 35.6 | 468.3 | 209.5 ± 10.6 | 379.0 ± 8.1 | 578.4 ± 8.8 | 1067.0 ± 91.6 |
| 14:0 (g) | 2.2 ± 0.1 | 1.9 | 0.9 ± 0.03 | 1.6 ± 0.03 | 2.2 ± 0.04 | 3.9 ± 0.3 |
| 15:0 (mg) | 252.4 ± 14.4 | 215.9 | 95.1 ± 5.0 | 178.6 ± 3.9 | 264.4 ± 5.0 | 471.9 ± 32.0 |
| 16:0 (g) | 11.2 ± 0.5 | 10.3 | 5.7 ± 0.2 | 8.7 ± 0.2 | 12.1 ± 0.2 | 18.3 ± 0.8 |
| 17:0 (mg) | 173.8 ± 10.6 | 151.5 | 66.7 ± 3.4 | 121.0 ± 3.0 | 179.5 ± 2.9 | 328.0 ± 26.4 |
| 18:0 (g) | 4.6 ± 0.2 | 4.1 | 2.0 ± 0.1 | 3.3 ± 0.1 | 5.0 ± 0.1 | 8.1 ± 0.4 |
| 20:0 (mg) | 75.7 ± 4.8 | 62.9 | 20.0 ± 1.2 | 47.9 ± 1.2 | 81.3 ± 2.0 | 153.9 ± 9.0 |
| Total MUFA (g) | 20.4 ± 08 | 19.2 | 9.7 ± 0.3 | 15.8 ± 0.4 | 22.3 ± 0.4 | 33.7 ± 1.3 |
| 14:1 (mg) | 193.0 ± 13.3 | 164.7 | 74.0 ± 3.8 | 132.7 ± 2.8 | 196.5 ± 2.9 | 370.4 ± 37.4 |
| 15:1 (mg) | 67.1 ± 5.9 | 48.3 | 21.8 ± 1.4 | 40.6 ± 0.9 | 65.4 ± 1.6 | 140.8 ± 18.0 |
| 16:1 (g) | 1.2 ± 0.1 | 1.1 | 0.5 ± 0.03 | 1.0 ± 0.02 | 1.4 ± 0.02 | 2.1 ± 0.1 |
| 17:1 (mg) | 111.6 ± 9.2 | 90.1 | 36.6 ± 2.1 | 69.7 ± 1.8 | 111.1 ± 1.9 | 228.7 ± 27.3 |
| 18:1 (g) | 18.3 ± 0.8 | 16.8 | 8.7 ± 0.3 | 14.1 ± 0.3 | 19.9 ± 0.3 | 30.6 ± 1.1 |
| 20:1 (mg) | 239.1 ± 15.9 | 191.3 | 73.6 ± 4.8 | 155.0 ± 3.0 | 239.5 ± 6.2 | 488.5 ± 35.2 |
| 22:1 (mg) | 156.2 ± 21.3 | 51.8 | 1.1 ± 0.3 | 23.7 ± 2.3 | 115.6 ± 8.1 | 483.2 ± 54.2 |
| PUFA total (g) | 7.4 ± 0.4 | 6.1 | 3.8 ± 0.1 | 5.5 ± 0.1 | 7.3 ± 0.1 | 13.0 ± 1.1 |
| 18:2 (g) | 5.8 ± 0.3 | 4.9 | 3.0 ± 0.1 | 4.5 ± 0.1 | 5.8 ± 0.1 | 9.8 ± 0.5 |
| 18:3 (g) | 1.3 ± 0.2 | 0.8 | 0.5 ± 0.01 | 0.7 ± 0.01 | 1.0 ± 0.02 | 3.1 ± 0.7 |
| 18:4 (mg) | 9.7 ± 2.2 | 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 2.8 ± 0.3 | 37.5 ± 6.9 |
| 20:3 (mg) | 0.6 ± 0.3 | 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 9.4 ± 4.4 |
| 20:4 (mg) | 119.5 ± 8.7 | 91.0 | 30.7 ± 2.3 | 68.9 ± 2.0 | 123.1 ± 4.1 | 255.2 ± 19.1 |
| 20:5 (mg) | 59.3 ± 10.4 | 11.3 | 0.2 ± 0.1 | 5.6 ± 0.4 | 27.0 ± 2.1 | 203.7 ± 30.3 |
| 22:5 (mg) | 23.8 ± 3.9 | 5.9 | 0.1 ± 0.1 | 3.7 ± 0.2 | 11.4 ± 0.5 | 79.8 ± 10.8 |
| 22:6 (mg) | 139.4 ± 21.6 | 50.3 | 9.6 ± 1.0 | 34.6 ± 1.4 | 82.4 ± 3.5 | 429.4 ± 64.8 |
| Concentrations of FA in RBC (µg/mL) | Low Protein a | High Protein a | p-Value | Low Carbohydrates a | High Carbohydrates a | p-Value | Low Fat a | High Fat a | p-Value |
|---|---|---|---|---|---|---|---|---|---|
| 14:0 | 10.23 ± 9.48 | 13.39 ± 12.88 | 0.213 | 12.01 ± 11.49 | 11.58 ± 11.31 | 0.924 | 11.28 ± 10.86 | 12.32 ± 11.91 | 0.812 |
| 15:0 | 12.08 ± 5.66 | 12.15 ± 5.03 | 0.893 | 12.20 ± 4.81 | 12.02 ± 5.86 | 0.976 | 12.35 ± 5.77 | 11.88 ± 4.89 | 0.425 |
| 16:0 | 417.76 ± 148.18 | 456.41 ± 189.46 | 0.316 | 440.62 ± 175.05 | 433.19 ± 166.75 | 0.902 | 426.90 ± 157.45 | 447.13 ± 183.22 | 0.694 |
| 17:0 | 13.01 ± 10.82 | 15.66 ± 9.53 | 0.185 | 15.41 ± 9.48 | 13.23 ± 10.94 | 0.330 | 13.65 ± 10.66 | 15.01 ± 9.84 | 0.637 |
| 18:0 | 273.15 ± 55.14 | 275.71 ± 62.72 | 0.973 | 273.11 ± 59.65 | 275.75 ± 58.37 | 0.640 | 272.05 ± 58.44 | 276.83 ± 59.53 | 0.896 |
| 22:0 | 6.44 ± 3.71 | 6.45 ± 3.51 | 0.877 | 6.32 ± 3.59 | 6.57 ± 3.63 | 0.661 | 6.46 ± 3.49 | 6.42 ± 3.73 | 0.861 |
| 14:1n-9 | 4.58 ± 2.71 | 5.29 ± 2.59 | 0.254 | 5.02 ± 2.50 | 4.84 ± 2.84 | 0.909 | 4.94 ± 2.91 | 4.92 ± 2.41 | 0.762 |
| 14:1n-5 | 2.81 ± 2.68 | 2.32 ± 1.72 | 0.120 | 2.73 ± 2.52 | 2.40 ± 1.96 | 0.533 | 2.34 ± 1.97 | 2.79 ± 2.51 | 0.420 |
| 15:1 | 27.66 ± 11.10 | 27.78 ± 8.94 | 0.895 | 26.74 ± 8.24 | 28.71 ± 11.58 | 0.213 | 27.95 ± 11.04 | 27.48 ± 9.02 | 0.887 |
| 16:1n-7 | 17.47 ± 18.29 | 27.17 ± 26.81 | 0.035 | 24.16 ± 25.01 | 20.37 ± 21.54 | 0.424 | 20.03 ± 20.65 | 24.57 ± 25.74 | 0.387 |
| 17:1 | 34.47 ± 19.87 | 32.03 ± 16.66 | 0.567 | 29.61 ± 14.93 | 36.97 ± 20.68 | 0.020 | 33.97 ± 19.49 | 32.54 ± 17.17 | 0.914 |
| 18:1n-9t | 2.64 ± 1.80 | 2.67 ± 1.73 | 0.959 | 2.56 ± 1.73 | 2.75 ± 1.79 | 0.493 | 2.65 ± 1.69 | 2.66 ± 1.84 | 0.898 |
| 18:1n-7t | 4.79 ± 3.14 | 4.42 ± 2.40 | 0.448 | 4.62 ± 2.73 | 4.59 ± 2.88 | 0.950 | 4.53 ± 3.07 | 4.68 ± 2.50 | 0.799 |
| 18:1n-9c | 354.50 ± 162.53 | 403.01 ± 209.02 | 0.176 | 383.59 ± 202.80 | 373.47 ± 172.90 | 0.744 | 369.89 ± 166.72 | 387.38 ± 208.17 | 0.665 |
| 18:1n-7c | 27.54 ± 10.52 | 33.46 ± 15.87 | 0.022 | 31.53 ± 14.88 | 29.40 ± 12.45 | 0.457 | 29.27 ± 12.79 | 31.70 ± 14.60 | 0.403 |
| 18:1n-5c | 3.41 ± 2.42 | 5.26 ± 4.28 | 0.007 | 4.90 ± 4.12 | 3.75 ± 2.85 | 0.112 | 3.95 ± 3.20 | 4.72 ± 3.91 | 0.338 |
| 20:1n-9 | 7.09 ± 2.88 | 7.32 ± 2.63 | 0.621 | 7.33 ± 2.80 | 7.07 ± 2.72 | 0.674 | 7.26 ± 2.71 | 7.15 ± 2.81 | 0.723 |
| 22:1n-9 | 12.36 ± 8.93 | 10.98 ± 8.03 | 0.426 | 10.52 ± 7.38 | 12.86 ± 9.39 | 0.139 | 12.15 ± 9.03 | 11.20 ± 7.94 | 0.765 |
| 24:1n-9 | 13.89 ± 12.59 | 16.34 ± 11.06 | 0.237 | 15.34 ± 10.97 | 14.88 ± 12.81 | 0.946 | 14.09 ± 12.33 | 16.14 ± 11.40 | 0.312 |
| 18:2n-6t | 7.59 ± 4.36 | 10.47 ± 4.55 | 0.001 | 9.89 ± 4.62 | 8.14 ± 4.58 | 0.051 | 7.99 ± 4.53 | 10.07 ± 4.60 | 0.018 |
| 18:2n-6 | 263.82 ± 161.35 | 326.61 ± 217.22 | 0.092 | 312.01 ± 207.61 | 277.65 ± 176.79 | 0.333 | 276.75 ± 175.44 | 313.46 ± 209.02 | 0.381 |
| 18:3n-6 | 4.75 ± 3.71 | 6.45 ± 5.19 | 0.052 | 6.07 ± 5.05 | 5.11 ± 4.00 | 0.289 | 5.06 ± 3.98 | 6.13 ± 5.07 | 0.216 |
| 20:2n-6 | 4.69 ± 8.70 | 5.25 ± 8.89 | 0.641 | 5.29 ± 8.85 | 4.65 ± 8.74 | 0.634 | 5.67 ± 10.16 | 4.27 ± 7.09 | 0.405 |
| 20:3n-6 | 30.84 ± 9.37 | 33.49 ± 12.71 | 0.316 | 31.94 ± 11.10 | 32.37 ± 11.37 | 0.702 | 32.12 ± 11.44 | 32.19 ± 11.02 | 0.783 |
| 20:4n-6 | 278.58 ± 55.85 | 283.64 ± 58.84 | 0.939 | 282.06 ± 59.92 | 280.10 ± 54.72 | 0.804 | 280.99 ± 57.55 | 281.19 ± 57.27 | 0.534 |
| 22:2n-6 | 2.91 ± 1.54 | 3.00 ± 1.71 | 0.787 | 3.05 ± 1.71 | 2.86 ± 1.53 | 0.434 | 2.91 ± 1.66 | 3.00 ± 1.60 | 0.729 |
| 22:4n-6 | 47.91 ± 12.16 | 44.41 ± 11.58 | 0.068 | 44.45 ± 11.80 | 47.92 ± 11.96 | 0.060 | 48.33 ± 12.03 | 43.98 ± 11.57 | 0.019 |
| 22:5n-6 | 10.69 ± 3.52 | 10.81 ± 3.34 | 0.907 | 10.27 ± 3.46 | 11.24 ± 3.33 | 0.070 | 11.07 ± 3.45 | 10.44 ± 3.39 | 0.223 |
| 18:3n-3t | 2.33 ± 2.33 | 2.61 ± 2.95 | 0.672 | 2.41 ± 2.35 | 2.54 ± 2.94 | 0.695 | 2.16 ± 2.24 | 2.79 ± 2.99 | 0.177 |
| 18:3n-3 | 13.17 ± 9.30 | 14.16 ± 8.18 | 0.578 | 13.46 ± 8.31 | 13.86 ± 9.21 | 0.589 | 13.17 ± 8.96 | 14.15 ± 8.55 | 0.640 |
| 20:3n-3 | 9.06 ± 8.32 | 12.08 ± 9.53 | 0.072 | 11.46 ± 9.71 | 9.63 ± 8.26 | 0.353 | 9.95 ± 8.61 | 11.16 ± 9.48 | 0.539 |
| 20:5n-3 | 25.45 ± 11.14 | 27.74 ± 14.05 | 0.320 | 27.58 ± 14.08 | 25.57 ± 11.09 | 0.467 | 24.66 ± 9.54 | 28.54 ± 15.04 | 0.123 |
| 22:5n-3 | 134.26 ± 61.66 | 136.29 ± 45.00 | 0.990 | 142.04 ± 50.02 | 128.38 ± 57.03 | 0.193 | 133.53 ± 53.37 | 137.03 ± 54.68 | 0.986 |
| 22:6n-3 | 104.81 ± 29.13 | 97.72 ± 26.87 | 0.164 | 99.78 ± 25.44 | 102.83 ± 30.79 | 0.477 | 100.25 ± 29.90 | 102.35 ± 26.44 | 0.745 |
| 20:3n-9 | 5.50 ± 2.18 | 5.65 ± 2.72 | 0.682 | 5.55 ± 2.69 | 5.60 ± 2.21 | 0.928 | 5.38 ± 2.03 | 5.77 ± 2.83 | 0.254 |
| Variable | Polymorphisms | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| rs174556 FADS1 | rs174547 FADS1 | rs174561 FADS1 | rs3834458 FADS2 | |||||||||
| CT + TT N = 53 | CC N = 72 | p-Value | TC + CC N = 54 | TT N = 72 | p-Value | TC + CC N = 58 | TT N = 67 | p-Value | T/- + -/- N = 59 | TT N = 66 | p-Value | |
| n-6 PUFA in RBC (µg/mL) | ||||||||||||
| 18:2n-6t | 9.19 ± 4.54 | 8.85 ± 4.84 | NS | 9.24 ± 4.51 | 8.85 ± 4.84 | NS | 8.90 ± 4.38 | 9.08 ± 4.99 | NS | 8.90 ± 4.58 | 9.08 ± 4.83 | NS |
| 18:2n-6 | 312.16 ± 202.00 | 286.07 ± 188.05 | NS | 309.59 ± 200.98 | 286.07 ± 188.05 | NS | 293.42 ± 192.49 | 300.35 ± 196.16 | NS | 304.90 ± 193.68 | 290.19 ± 194.97 | NS |
| 18:3n-6 | 5.60 ± 3.80 | 5.67 ± 5.12 | NS | 5.54 ± 3.79 | 5.67 ± 5.12 | NS | 5.55 ± 3.77 | 5.72 ± 5.22 | NS | 5.61 ± 3.87 | 5.67 ± 5.18 | NS |
| 20:2n-6 | 4.40 ± 6.85 | 5.53 ± 10.04 | NS | 4.32 ± 6.81 | 5.53 ± 10.04 | NS | 4.39 ± 6.61 | 5.62 ± 10.37 | NS | 4.19 ± 6.54 | 5.81 ± 10.44 | NS |
| 20:3n-6 | 34.06 ± 11.04 | 30.81 ± 11.32 | NS | 34.05 ± 10.94 | 30.81 ± 11.32 | NS | 32.97 ± 10.85 | 31.50 ± 11.66 | NS | 33.63 ± 11.06 | 30.90 ± 11.39 | NS |
| 20:4n-6 | 267.92 ± 55.96 | 289.97 ± 57.23 | NS | 268.81 ± 55.81 | 289.97 ± 57.23 | NS | 265.27 ± 54.84 | 293.90 ± 56.85 | <0.05 | 270.18 ± 54.81 | 289.95 ± 58.67 | NS |
| 22:2n-6 | 2.96 ± 1.87 | 2.96 ± 1.45 | NS | 2.96 ± 1.86 | 2.96 ± 1.45 | NS | 2.90 ± 1.80 | 3.02 ± 1.49 | NS | 3.02 ± 1.91 | 2.91 ± 1.36 | NS |
| 22:4n-6 | 44.97 ± 13.65 | 47.10 ± 10.70 | NS | 45.07 ± 13.54 | 47.10 ± 10.70 | NS | 45.01 ± 12.96 | 47.22 ± 11.17 | NS | 45.35 ± 13.67 | 46.95 ± 10.41 | NS |
| 22:5n-6 | 11.06 ± 3.67 | 10.57 ± 3.27 | NS | 11.03 ± 3.65 | 10.57 ± 3.27 | NS | 10.91 ± 3.41 | 10.67 ± 3.49 | NS | 11.01 ± 3.57 | 10.58 ± 3.34 | NS |
| n-3 PUFA in RBC (µg/mL) | ||||||||||||
| 18:3n-3t | 2.22 ± 1.93 | 2.70 ± 3.09 | NS | 2.20 ± 1.92 | 2.70 ± 3.09 | NS | 2.13 ± 1.44 | 2.82 ± 3.37 | NS | 2.23 ± 1.89 | 2.74 ± 3.20 | NS |
| 18:3n-3 | 12.96 ± 7.10 | 14.33 ± 9.84 | NS | 12.84 ± 7.09 | 14.33 ± 9.84 | NS | 12.40 ± 6.65 | 14.92 ± 10.18 | NS | 12.72 ± 7.17 | 14.67 ± 9.97 | NS |
| 20:3n-3 | 10.62 ± 8.29 | 10.74 ± 9.62 | NS | 10.47 ± 8.28 | 10.74 ± 9.62 | NS | 10.03 ± 7.73 | 11.26 ± 10.07 | NS | 11.02 ± 9.30 | 10.40 ± 8.87 | NS |
| 20:5n-3 | 24.89 ± 12.80 | 27.97 ± 12.64 | NS | 24.75 ± 12.72 | 27.97 ± 12.64 | NS | 24.42 ± 12.79 | 28.61 ± 12.48 | NS | 24.73 ± 12.56 | 28.40 ± 12.76 | NS |
| 22:5n-3 | 129.50 ± 54.13 | 138.00 ± 53.68 | NS | 130.53 ± 54.16 | 138.00 ± 53.68 | NS | 128.93 ± 54.39 | 139.13 ± 53.27 | NS | 132.32 ± 53.54 | 136.26 ± 54.40 | NS |
| 22:6n-3 | 101.03 ± 29.62 | 101.13 ± 27.15 | NS | 100.76 ± 29.40 | 101.13 ± 27.15 | NS | 100.21 ± 29.35 | 101.85 ± 27.18 | NS | 101.01 ± 28.77 | 101.16 ± 27.72 | NS |
| Parameter | Mean ± SD | Minimum | Maximum |
|---|---|---|---|
| Desaturase activity ratios | |||
| D5D | 9.40 ± 0.21 | 4.95 | 15.37 |
| D6D | 0.021 ± 0.001 | 0.003 | 0.057 |
| Combined effect of desaturase and elongase activity ratios | |||
| 20:4n-6/18:2n-6 | 1.24 ± 0.05 | 0.32 | 2.34 |
| 22:6n-3/20:5n-3 | 4.27 ± 0.12 | 1.22 | 8.98 |
| 20:5n-3/18:3n-3 | 2.41 ± 0.12 | 0.48 | 8.40 |
| 22:6n-3/18:3n-3 | 10.16 ± 0.52 | 2.12 | 27.38 |
| 22:4n-6/18:2n-6 | 0.22 ± 0.01 | 0.03 | 0.50 |
| Variable | Polymorphisms | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| rs174556 FADS1 | rs174547 FADS1 | rs174561 FADS1 | rs3834458 FADS2 | |||||||||
| CT + TT N = 53 | CC N = 72 | p-Value | TC + CC N = 54 | TT N = 72 | p-Value | TC + CCN N = 58 | TT N = 67 | p-Value | T/- + -/- N = 59 | TT N= 66 | p-Value | |
| Desaturase activity ratios | ||||||||||||
| D5D | 8.29 ± 1.97 | 10.10 ± 2.37 | <0.0001 | 8.31 ± 1.96 | 10.10 ± 2.37 | <0.0001 | 8.51 ± 2.12 | 10.04 ± 2.37 | < 0.001 | 8.52 ± 2.14 | 10.06 ± 2.36 | <0.001 |
| D6D | 0.02 ± 0.01 | 0.02 ± 0.01 | NS | 0.02 ± 0.01 | 0.02 ± 0.01 | NS | 0.02 ± 0.01 | 0.02 ± 0.01 | NS | 0.02 ± 0.01 | 0.02 ± 0.01 | NS |
| Combined effect of desaturase and elongase activity ratios | ||||||||||||
| 20:4n-6/18:2n-6 | 1.14 ± 0.52 | 1.30 ± 0.50 | <0.05 | 1.15 ± 0.52 | 1.30 ± 0.50 | NS | 1.18 ± 0.50 | 1.27 ± 0.52 | NS | 1.16 ± 0.50 | 1.30 ± 0.51 | NS |
| 22:4n-6/18:2n-6 | 0.21 ± 0.12 | 0.23 ± 0.12 | NS | 0.21 ± 0.12 | 0.23 ± 0.12 | NS | 0.22 ± 0.12 | 0.22 ± 0.12 | NS | 0.21 ± 0.12 | 0.23 ± 0.12 | NS |
| 20:5n-3/18:3n-3 | 2.29 ± 1.28 | 2.52 ± 1.43 | NS | 2.29 ± 1.27 | 2.52 ± 1.43 | NS | 2.30 ± 1.25 | 2.52 ± 1.47 | NS | 2.33 ± 1.26 | 2.50 ± 1.47 | NS |
| 22:6n-3/18:3n-3 | 10.01 ± 5.61 | 10.22 ± 6.39 | NS | 10.06 ± 5.57 | 10.22 ± 6.39 | NS | 10.33 ± 5.80 | 9.95 ± 6.29 | NS | 10.37 ± 5.84 | 9.91 ± 6.26 | NS |
| 22:6n-3/20:5n-3 | 4.49 ± 1.34 | 4.04 ± 1.28 | NS | 4.50 ± 1.33 | 4.04 ± 1.28 | NS | 4.57 ± 1.36 | 3.95 ± 1.23 | <0.05 | 4.53 ± 1.36 | 3.97 ± 1.24 | <0.05 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Muzsik, A.; Bajerska, J.; Jeleń, H.H.; Gaca, A.; Chmurzynska, A. Associations between Fatty Acid Intake and Status, Desaturase Activities, and FADS Gene Polymorphism in Centrally Obese Postmenopausal Polish Women. Nutrients 2018, 10, 1068. https://doi.org/10.3390/nu10081068
Muzsik A, Bajerska J, Jeleń HH, Gaca A, Chmurzynska A. Associations between Fatty Acid Intake and Status, Desaturase Activities, and FADS Gene Polymorphism in Centrally Obese Postmenopausal Polish Women. Nutrients. 2018; 10(8):1068. https://doi.org/10.3390/nu10081068
Chicago/Turabian StyleMuzsik, Agata, Joanna Bajerska, Henryk H. Jeleń, Anna Gaca, and Agata Chmurzynska. 2018. "Associations between Fatty Acid Intake and Status, Desaturase Activities, and FADS Gene Polymorphism in Centrally Obese Postmenopausal Polish Women" Nutrients 10, no. 8: 1068. https://doi.org/10.3390/nu10081068
APA StyleMuzsik, A., Bajerska, J., Jeleń, H. H., Gaca, A., & Chmurzynska, A. (2018). Associations between Fatty Acid Intake and Status, Desaturase Activities, and FADS Gene Polymorphism in Centrally Obese Postmenopausal Polish Women. Nutrients, 10(8), 1068. https://doi.org/10.3390/nu10081068







