Relative Validity of a Beverage Frequency Questionnaire Used to Assess Fluid Intake in the Autosomal Dominant Polycystic Kidney Disease Population
Abstract
1. Introduction
2. Materials and Methods
2.1. Human Research Ethics Approval
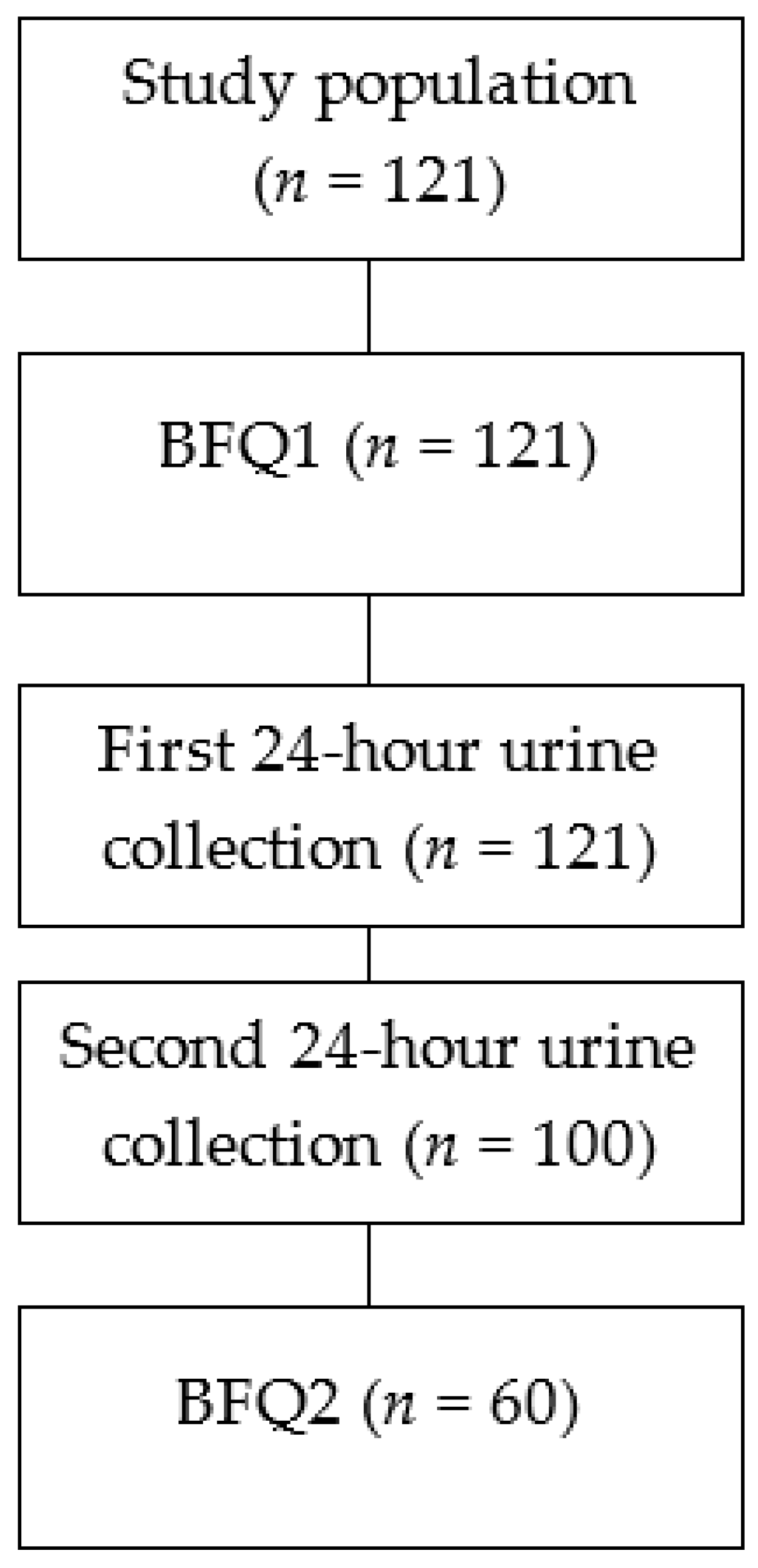
2.2. Study Population
2.3. Study Design
2.4. Development of the Beverage Frequency Questionnaire (BFQ)
2.5. Calculation of Fluid Intake Using BFQ Responses
2.6. Statistical Analysis
3. Results
3.1. Clinical Characteristics of the Study Population
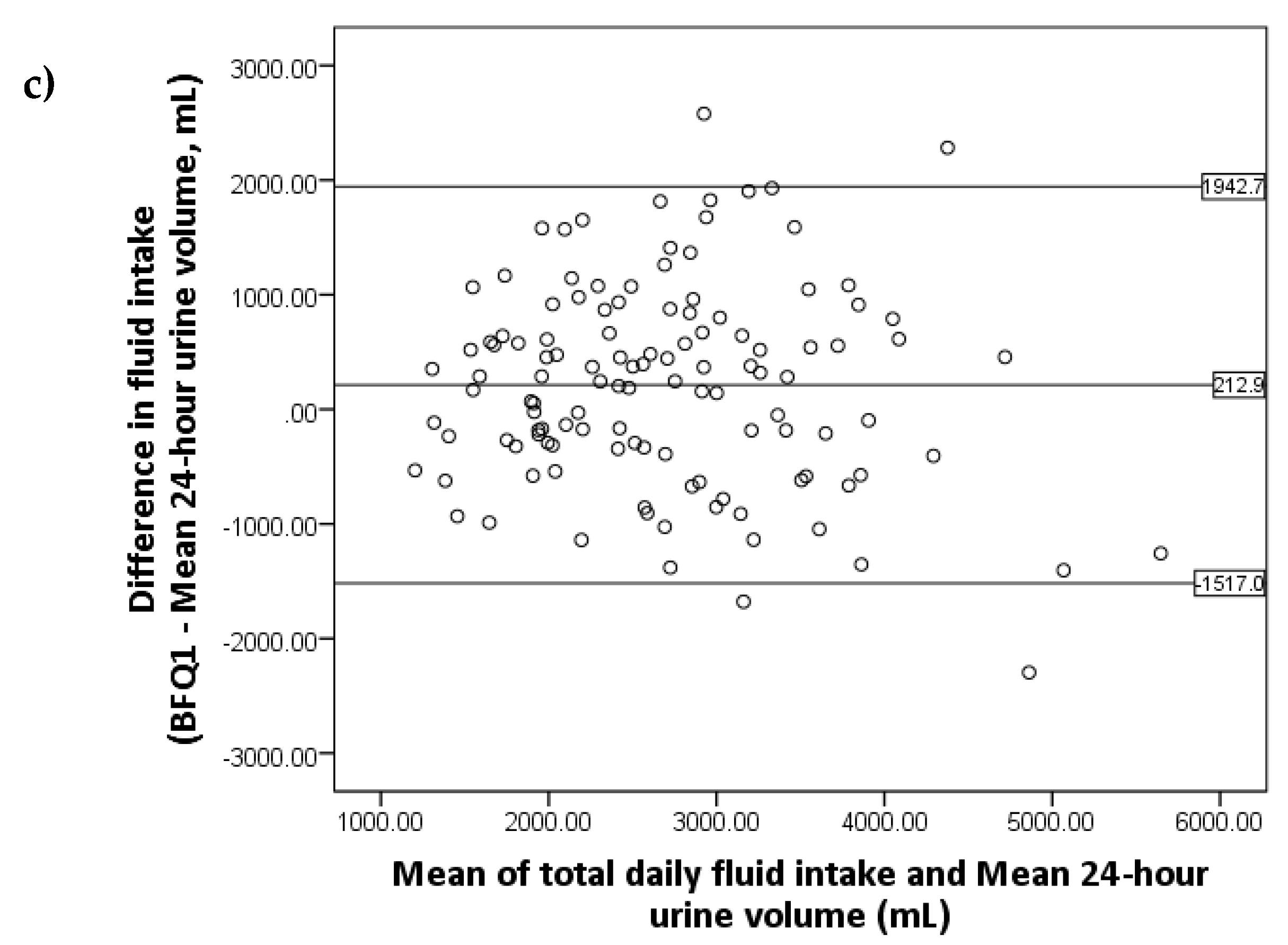
3.2. Relative Validity of the BFQ
3.3. Reliability of the BFQ
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
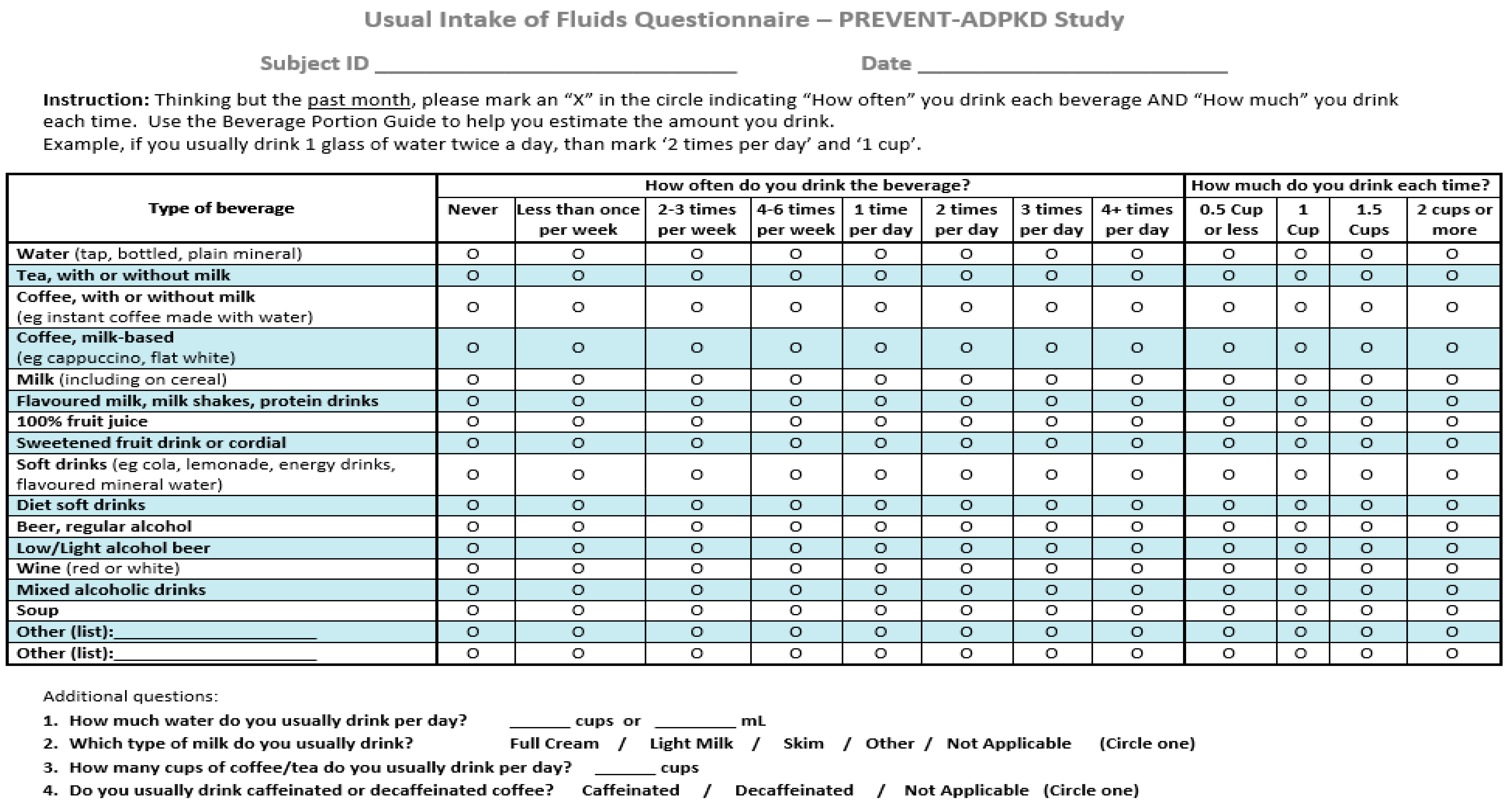
Appendix B

References
- Rangan, G.K.; Lopez-Vargas, P.; Nankivell, B.J.; Tchan, M.; Tong, A.; Tunnicliffe, D.J.; Savige, J. Autosomal dominant polycystic kidney disease: A path forward. Semin. Nephrol. 2015, 35, 524–537. [Google Scholar] [CrossRef] [PubMed]
- Christ-Crain, M.; Fenske, W. Copeptin in the diagnosis of vasopressin-dependent disorders of fluid homeostasis. Nat. Rev. Endocrinol. 2016, 12, 168–176. [Google Scholar] [CrossRef] [PubMed]
- Torres, V.E.; Harris, P.C. Strategies targeting camp signaling in the treatment of polycystic kidney disease. JASN 2014, 25, 18–32. [Google Scholar] [CrossRef] [PubMed]
- Torres, V.E.; Bankir, L.; Grantham, J.J. A case for water in the treatment of polycystic kidney disease. CJASN 2009, 4, 1140–1150. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.J.; Grantham, J.J.; Wetmore, J.B. The medicinal use of water in renal disease. Kidney Int. 2013, 84, 45–53. [Google Scholar] [CrossRef] [PubMed]
- Hedrick, V.E.; Comber, D.L.; Estabrooks, P.A.; Savla, J.; Davy, B.M. The beverage intake questionnaire: Determining initial validity and reliability. J. Am. Diet. Assoc. 2010, 110, 1227–1232. [Google Scholar] [CrossRef] [PubMed]
- Perrier, E.; Rondeau, P.; Poupin, M.; Le Bellego, L.; Armstrong, L.E.; Lang, F.; Stookey, J.; Tack, I.; Vergne, S.; Klein, A. Relation between urinary hydration biomarkers and total fluid intake in healthy adults. Eur. J. Clin. Nutr. 2013, 67, 939–943. [Google Scholar] [CrossRef] [PubMed]
- Nissensohn, M.; Ruano, C.; Serra-Majem, L. Validation of beverage intake methods vs. Hydration biomarkers; a short review. Nutr. Hosp. 2013, 28, 1815–1819. [Google Scholar] [PubMed]
- Armstrong, L.E. Challenges of linking chronic dehydration and fluid consumption to health outcomes. Nutr. Rev. 2012, 70, S121–S127. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, L.E.; Maresh, C.M.; Castellani, J.W.; Bergeron, M.F.; Kenefick, R.W.; LaGasse, K.E.; Riebe, D. Urinary indices of hydration status. Int. J. Sport Nutr. 1994, 4, 265–279. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, L.E.; Johnson, E.C.; McKenzie, A.L.; Muñoz, C.X. Interpreting common hydration biomarkers on the basis of solute and water excretion. Eur. J. Clin. Nutr. 2013, 67, 249–253. [Google Scholar] [CrossRef] [PubMed]
- Wong, A.T.Y.; Mannix, C.; Grantham, J.J.; Allman-Farinelli, M.; Badve, S.V.; Boudville, N.; Byth, K.; Chan, J.; Coulshed, S.; Edwards, M.E.; et al. Randomised controlled trial to determine the efficacy and safety of prescribed water intake to prevent kidney failure due to autosomal dominant polycystic kidney disease (PREVENT-ADPKD). BMJ Open 2018, 8, e018794. [Google Scholar] [PubMed]
- Irazabal, M.V.; Rangel, L.J.; Bergstralh, E.J.; Osborn, S.L.; Harmon, A.J.; Sundsbak, J.L.; Bae, K.T.; Chapman, A.B.; Grantham, J.J.; Mrug, M.; et al. Imaging classification of autosomal dominant polycystic kidney disease: A simple model for selecting patients for clinical trials. JASN 2015, 26, 160–172. [Google Scholar] [CrossRef] [PubMed]
- Australian Bureau of Statistics. Australian Health Survey: Nutrition First Results—Foods and Nutrients, 2011–2012; Australian Bureau of Statistics: Canberra, Australia, 2014.
- Hinkle, D.E.; Wiersma, W.; Jurs, S.G. Applied Statistics for the Behavioural Sciences, 5th ed.; Houghton Mifflin: Boston, MA, USA, 2003. [Google Scholar]
- Bland, J.M.; Altman, D.G. Statistical methods for assessing agreement between two methods of clinical measurement. Lancet 1986, 1, 307–310. [Google Scholar] [CrossRef]
- Ferreira-Pêgo, C.; Nissensohn, M.; Kavouras, S.A.; Babio, N.; Serra-Majem, L.; Martín Águila, A.; Mauromoustakos, A.; Álvarez Pérez, J.; Salas-Salvadó, J. Beverage intake assessment questionnaire: Relative validity and repeatability in a Spanish population with metabolic syndrome from the predimed-plus study. Nutrients 2016, 8, 475. [Google Scholar] [CrossRef] [PubMed]
- Malisova, O.; Bountziouka, V.; Panagiotakos, D.B.; Zampelas, A.; Kapsokefalou, M. The water balance questionnaire: Design, reliability and validity of a questionnaire to evaluate water balance in the general population. Int. J. Food Sci. Nutr. 2012, 63, 138–144. [Google Scholar] [CrossRef] [PubMed]
- Berl, T. Impact of solute intake on urine flow and water excretion. JASN 2008, 19, 1076–1078. [Google Scholar] [CrossRef] [PubMed]
- Cox, P. Insensible water loss and its assessment in adult patients: A review. Acta Anaesthesiol. Scand. 1987, 31, 771–776. [Google Scholar] [CrossRef] [PubMed]

| Characteristics | Mean | SD | Minimum | Maximum |
|---|---|---|---|---|
| Age (years) | 43 | 11 | 19 | 66 |
| Height (cm) | 171 | 11 | 146 | 195 |
| Mass (kg) | 80 | 19 | 51 | 137 |
| BMI (kg/m2) | 27 | 5 | 19 | 42 |
| Systolic BP (mmHg) | 135 | 14 | 98 | 172 |
| Diastolic BP (mmHg) | 83 | 10 | 62 | 111 |
| eGFR (mL/min/1.73 m2) | 73 | 19 | 25 | 90 |
| Creatinine (µmol/L) | 100 | 38 | 58 | 235 |
| Ht-TKV (mL/m) | 902 | 673 | 156 | 4504 |
| n | Mean | SD | Minimum | Maximum | p * | r | |
|---|---|---|---|---|---|---|---|
| BFQ1 (mL) | 121 | 2797 | 919 | 938 | 5518 | N/A | N/A |
| Urine volume 1 (mL) | 121 | 2637 | 1075 | 690 | 6150 | 0.065 | 0.561 † |
| Urine volume 2 (mL) | 100 | 2435 | 1006 | 770 | 6560 | 0.001 | 0.576 † |
| Mean urine volume (mL) | 2584 | 1000 | 1015 | 6275 | 0.009 | 0.580 † | |
| Urine osmolality 1 (mOsm/kg) | 120 | 421 | 183 | 123 | 923 | N/A | −0.283 ‡ |
| Urine osmolality 2 (mOsm/kg) | 100 | 434 | 174 | 122 | 961 | N/A | −0.298 ‡ |
| Mean urine osmolality (mOsm/kg) | 425 | 167 | 132 | 880 | N/A | −0.276 ‡ |
| n | Mean | SD | Minimum | Maximum | p * | R † | |
|---|---|---|---|---|---|---|---|
| BFQ1 (mL) | 121 | 2797 | 919 | 938 | 5518 | 0.598 | 0.799 † |
| BFQ2 (mL) | 60 | 2746 | 974 | 1152 | 6089 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mannix, C.; Rangan, A.; Wong, A.; Zhang, J.; Allman-Farinelli, M.; Rangan, G. Relative Validity of a Beverage Frequency Questionnaire Used to Assess Fluid Intake in the Autosomal Dominant Polycystic Kidney Disease Population. Nutrients 2018, 10, 1051. https://doi.org/10.3390/nu10081051
Mannix C, Rangan A, Wong A, Zhang J, Allman-Farinelli M, Rangan G. Relative Validity of a Beverage Frequency Questionnaire Used to Assess Fluid Intake in the Autosomal Dominant Polycystic Kidney Disease Population. Nutrients. 2018; 10(8):1051. https://doi.org/10.3390/nu10081051
Chicago/Turabian StyleMannix, Carly, Anna Rangan, Annette Wong, Jennifer Zhang, Margaret Allman-Farinelli, and Gopala Rangan. 2018. "Relative Validity of a Beverage Frequency Questionnaire Used to Assess Fluid Intake in the Autosomal Dominant Polycystic Kidney Disease Population" Nutrients 10, no. 8: 1051. https://doi.org/10.3390/nu10081051
APA StyleMannix, C., Rangan, A., Wong, A., Zhang, J., Allman-Farinelli, M., & Rangan, G. (2018). Relative Validity of a Beverage Frequency Questionnaire Used to Assess Fluid Intake in the Autosomal Dominant Polycystic Kidney Disease Population. Nutrients, 10(8), 1051. https://doi.org/10.3390/nu10081051






