Evaluating Whole Grain Intervention Study Designs and Reporting Practices Using Evidence Mapping Methodology
Abstract
1. Introduction
2. Materials and Methods
2.1. Search Strategy
2.2. Selection Criteria
2.3. Data Abstraction and Analysis
3. Results
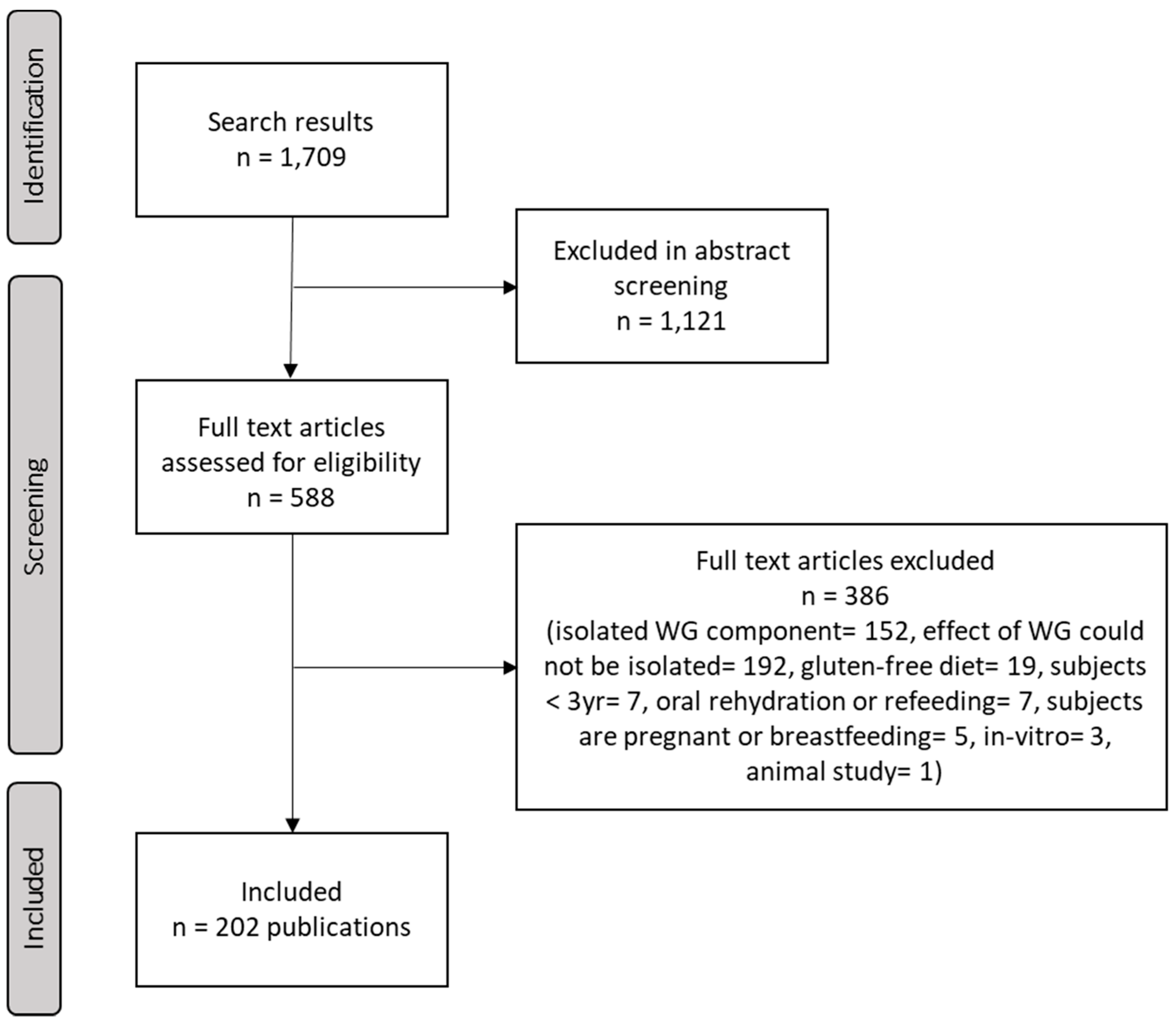
3.1. Literature Search and Screening
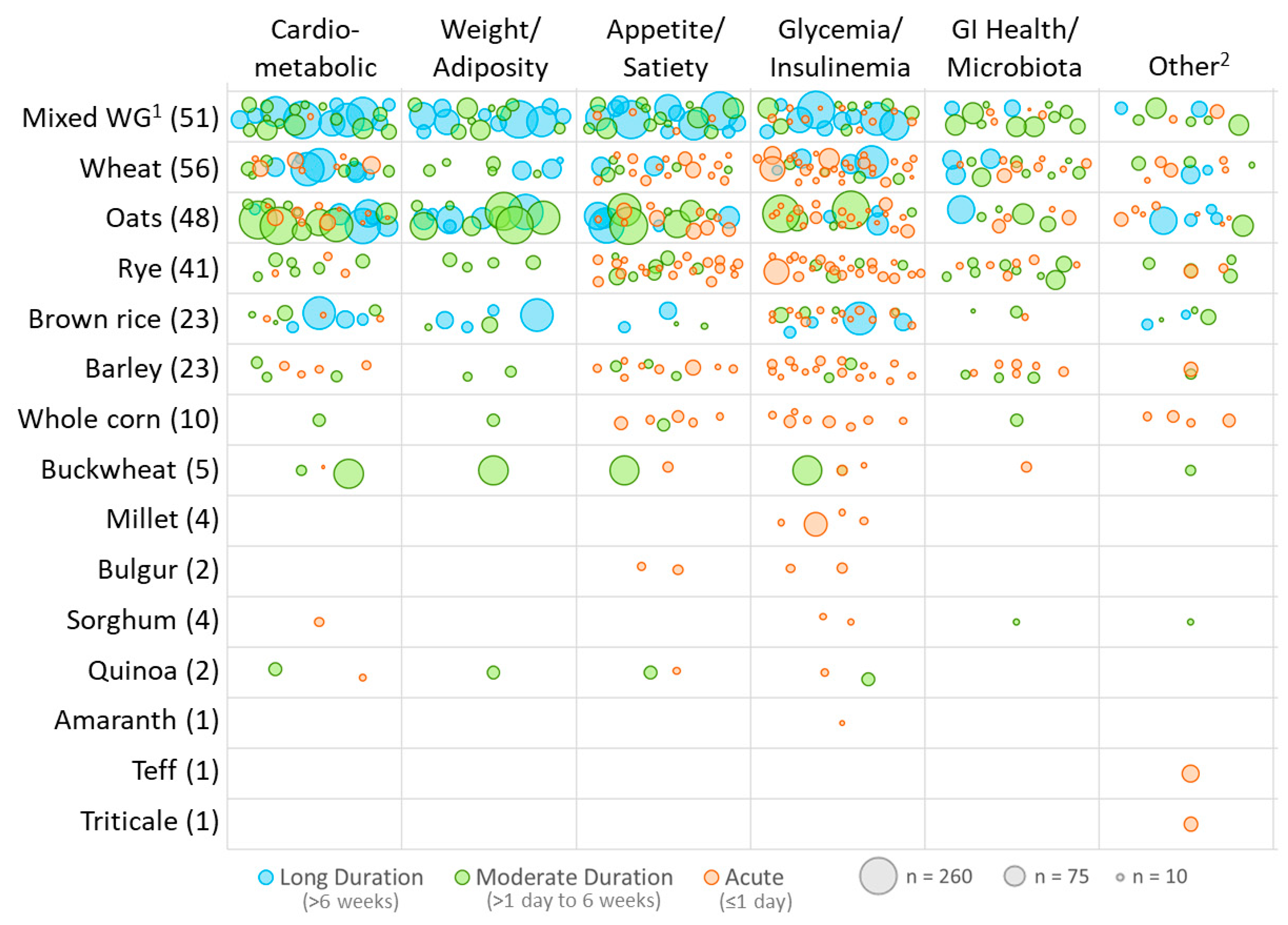
3.2. Study Design Characteristics
3.3. Reporting Practices
4. Discussion
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Data Availability
References
- USDA. Dietary Guidelines for Americans, 5th ed. Available online: https://www.cnpp.usda.gov/ (accessed on 11 January 2017).
- USDA. Dietary Guidelines for Americans, 8th ed. Available online: https://health.gov/dietaryguidelines/2015/guidelines/ (accessed on 11 January 2017).
- Frølich, W.; Åman, P.; Tetens, I. Whole grain foods and health—A Scandinavian perspective. Food Nutr. Res. 2013, 57, 18503. [Google Scholar] [CrossRef] [PubMed]
- Ye, E.Q.; Chacko, S.A.; Chou, E.L.; Kugizaki, M.; Liu, S. Greater whole-grain intake is associated with lower risk of type 2 diabetes, cardiovascular disease and weight gain. J. Nutr. 2012, 142, 1304–1313. [Google Scholar] [CrossRef] [PubMed]
- Aune, D.; Norat, T.; Romundstad, P.; Vatten, L.J. Whole grain and refined grain consumption and the risk of type 2 diabetes: A systematic review and dose-response meta-analysis of cohort studies. Eur. J. Epidemiol. 2013, 28, 845–858. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Flint, A.J.; Qi, Q.; van Dam, R.M.; Sampson, L.A.; Rimm, E.B.; Holmes, M.D.; Willett, W.C.; Hu, F.B.; Sun, Q. Association between dietary whole grain intake and risk of mortality: Two large prospective studies in US men and women. JAMA Intern. Med. 2015, 175, 373–384. [Google Scholar] [CrossRef] [PubMed]
- Huang, T.; Xu, M.; Lee, A.; Cho, S.; Qi, L. Consumption of whole grains and cereal fiber and total and cause-specific mortality: Prospective analysis of 367,442 individuals. BMC Med. 2015, 13, 59. [Google Scholar] [CrossRef]
- Ferruzzi, M.G.; Jonnalagadda, S.S.; Liu, S.; Marquart, L.; McKeown, N.; Reicks, M.; Riccardi, G.; Seal, C.; Slavin, J.; Thielecke, F.; et al. Developing a standard definition of whole-grain foods for dietary recommendations: Summary report of a multidisciplinary expert roundtable discussion12. Adv. Nutr. 2014, 5, 164–176. [Google Scholar] [CrossRef] [PubMed]
- Korczak, R.; Marquart, L.; Slavin, J.L.; Ringling, K.; Chu, Y.; O’Shea, M.; Harriman, C.; Toups, K.; de Vries, J.; Jacques, P.; et al. Thinking critically about whole-grain definitions: Summary report of an interdisciplinary roundtable discussion at the 2015 Whole Grains Summit. Am. J. Clin. Nutr. 2016, 104, 1508–1514. [Google Scholar] [CrossRef] [PubMed]
- Ross, A.B.; van der Kamp, J.W.; King, R.; Lê, K.A.; Mejborn, H.; Seal, C.J.; Thielecke, F. Forum, on behalf of the H. Perspective: A definition for whole-grain food products—Recommendations from the healthgrain forum. Adv. Nutr. 2017, 8, 525–531. [Google Scholar] [CrossRef] [PubMed]
- American Association of Cereal Chemists International. Whole Grains. Available online: http://www.aaccnet.org/initiatives/definitions/pages/wholegrain.aspx (accessed on 16 January 2017).
- American Association of Cereal Chemists International (AACCI). Whole Grains Working Group Unveils New Whole Grain Products Characterizations. Available online: http://www.aaccnet.org/about/newsreleases/Pages/WholeGrainProductCharacterization.aspx (accessed on 23 January 2017).
- Food Safety Inspection Service Use of the USDA MyPyramid Reference on Meat and Poultry Labeling and Whole Grain Claims. Available online: https://www.fsis.usda.gov/wps/wcm/connect/6ea06856-e04d-46d7-befd-5b9287c55640/Food_Guide_MYPyramid_Policy.pdf?MOD=AJPERES (accessed on 23 January 2017).
- Ross, A.B.; Kristensen, M.; Seal, C.J.; Jacques, P.; McKeown, N.M. Recommendations for reporting whole-grain intake in observational and intervention studies. Am. J. Clin. Nutr. 2015, 101, 903–907. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.D.; Shams White, M.; Bright, O.J.M.; Parrott, J.S.; Chung, M. Creating a literature database of low-calorie sweeteners and health studies: Evidence mapping. BMC Med. Res. Methodol. 2016, 16, 1. [Google Scholar] [CrossRef] [PubMed]
- Hetrick, S.E.; Parker, A.G.; Callahan, P.; Purcell, R. Evidence mapping: Illustrating an emerging methodology to improve evidence-based practice in youth mental health. J. Eval. Clin. Pract. 2010, 16, 1025–1030. [Google Scholar] [CrossRef] [PubMed]
- Bragge, P.; Clavisi, O.; Turner, T.; Tavender, E.; Collie, A.; Gruen, R.L. The Global Evidence Mapping Initiative: Scoping research in broad topic areas. BMC Med. Res. Methodol. 2011, 11, 92. [Google Scholar] [CrossRef] [PubMed]
- Evidence Gap Maps 3ie: International Initiative for Impact Evaluation | Evaluating Impact, Informing Policy, Improving Lives. Available online: http://www.3ieimpact.org/en/evaluation/evidence-gap-maps/ (accessed on 1 August 2018).
- Althuis, M.D.; Weed, D.L. Evidence mapping: Methodologic foundations and application to intervention and observational research on sugar-sweetened beverages and health outcomes. Am. J. Clin. Nutr. 2013, 98, 755–768. [Google Scholar] [CrossRef] [PubMed]
- Livingston, K.A.; Chung, M.; Sawicki, C.M.; Lyle, B.J.; Wang, D.D.; Roberts, S.B.; McKeown, N.M. Development of a publicly available, comprehensive database of fiber and health outcomes: Rationale and methods. PLoS ONE 2016, 11, e0156961. [Google Scholar] [CrossRef] [PubMed]
- Liska, D.J.; Cook, C.M.; Wang, D.D.; Gaine, P.C.; Baer, D.J. Trans fatty acids and cholesterol levels: An evidence map of the available science. Food Chem. Toxicol. 2016, 98, 269–281. [Google Scholar] [CrossRef] [PubMed]
- Wallace, B.C.; Small, K.; Brodley, C.E.; Lau, J.; Trikalinos, T.A. Deploying an interactive machine learning system in an evidence-based practice center: Abstrackr. In Proceedings of the 2nd ACM SIGHIT International Health Informatics Symposium, Miami, FL, USA, 28–30 January 2012; pp. 819–824. [Google Scholar]
- Stringer, D.M.; Taylor, C.G.; Appah, P.; Blewett, H.; Zahradka, P. Consumption of buckwheat modulates the post-prandial response of selected gastrointestinal satiety hormones in individuals with type 2 diabetes mellitus. Metab. Clin. Exp. 2013, 62, 1021–1031. [Google Scholar] [CrossRef] [PubMed]
- Shimabukuro, M.; Higa, M.; Kinjo, R.; Yamakawa, K.; Tanaka, H.; Kozuka, C.; Yabiku, K.; Taira, S.I.; Sata, M.; Masuzaki, H. Effects of the brown rice diet on visceral obesity and endothelial function: The BRAVO study. Br. J. Nutr. 2014, 111, 310–320. [Google Scholar] [CrossRef] [PubMed]
- Seal, C.J. Whole grains and CVD risk. Proc. Nutr. Soc. 2006, 65, 24–34. [Google Scholar] [CrossRef] [PubMed]
- USDA. Dietary Guidelines for Americans, 6th ed. Available online: https://health.gov/dietaryguidelines/dga2005/document/ (accessed on 16 January 2017).
- USDA. Dietary Guidelines for Americans, 7th ed. Available online: https://health.gov/dietaryguidelines/2010/ (accessed on 17 January 2017).
- HealthGrain Forum Definition and Analysis of Whole Grain. Available online: http://www.healthgrain.org/regulatory_issues/whole_grain (accessed on 16 January 2017).
- Katcher, H.I.; Legro, R.S.; Kunselman, A.R.; Gillies, P.J.; Demers, L.M.; Bagshaw, D.M.; Kris Etherton, P.M. The effects of a whole grain-enriched hypocaloric diet on cardiovascular disease risk factors in men and women with metabolic syndrome. Am. J. Clin. Nutr. 2008, 87, 79–90. [Google Scholar] [CrossRef] [PubMed]
- Brownlee, I.A.; Moore, C.; Chatfield, M.; Richardson, D.P.; Ashby, P.; Kuznesof, S.A.; Jebb, S.A.; Seal, C.J. Markers of cardiovascular risk are not changed by increased whole-grain intake: The WHOLEheart study, a randomised, controlled dietary intervention. Br. J. Nutr. 2010, 104, 125–134. [Google Scholar] [CrossRef] [PubMed]
- Ross, A.B.; Bourgeois, A.; Macharia, H.N.; Kochhar, S.; Jebb, S.A.; Brownlee, I.A.; Seal, C.J. Plasma alkylresorcinols as a biomarker of whole-grain food consumption in a large population: Results from the WHOLEheart Intervention Study. Am. J. Clin. Nutr. 2012, 95, 204–211. [Google Scholar] [CrossRef] [PubMed]
- Langkamp Henken, B.; Nieves, C.; Culpepper, T.; Radford, A.; Girard, S.A.; Hughes, C.; Christman, M.C.; Mai, V.; Dahl, W.J.; Boileau, T.; et al. Fecal lactic acid bacteria increased in adolescents randomized to whole-grain but not refined-grain foods, whereas inflammatory cytokine production decreased equally with both interventions. J. Nutr. 2012, 142, 2025–2032. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, D.R.; Gallaher, D.D. Whole grain intake and cardiovascular disease: A review. Curr. Atheroscler. Rep. 2004, 6, 415–423. [Google Scholar] [CrossRef] [PubMed]
- Okarter, N.; Liu, R.H. Health benefits of whole grain phytochemicals. Crit. Rev. Food Sci. Nutr. 2010, 50, 193–208. [Google Scholar] [CrossRef] [PubMed]
- Fardet, A. New hypotheses for the health-protective mechanisms of whole-grain cereals: What is beyond fibre? Nutr. Res. Rev. 2010, 23, 65–134. [Google Scholar] [CrossRef] [PubMed]
- Helnæs, A.; Kyrø, C.; Andersen, I.; Lacoppidan, S.; Overvad, K.; Christensen, J.; Tjønneland, A.; Olsen, A. Intake of whole grains is associated with lower risk of myocardial infarction: The danish diet, cancer and health cohort. Am. J. Clin. Nutr. 2016, 103, 999–1007. [Google Scholar] [CrossRef] [PubMed]
- Kristensen, M.; Pelletier, X.; Ross, A.B.; Thielecke, F.A. High Rate of non-compliance confounds the study of whole grains and weight maintenance in a randomised intervention trial—The case for greater use of dietary biomarkers in nutrition intervention studies. Nutrients 2017, 9, 55. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, D.R.; Pereira, M.A.; Stumpf, K.; Pins, J.J.; Adlercreutz, H. Whole grain food intake elevates serum enterolactone. Br. J. Nutr. 2002, 88, 111–116. [Google Scholar] [CrossRef] [PubMed]
- McIntosh, G.H.; Noakes, M.; Royle, P.J.; Foster, P.R. Whole-grain rye and wheat foods and markers of bowel health in overweight middle-aged men. Am. J. Clin. Nutr. 2003, 77, 967–974. [Google Scholar] [CrossRef] [PubMed]
- Linko-Parvinen, A.M.; Landberg, R.; Tikkanen, M.J.; Adlercreutz, H.; Peñalvo, J.L. Alkylresorcinols from whole-grain wheat and rye are transported in human plasma lipoproteins. J. Nutr. 2007, 137, 1137–1142. [Google Scholar] [CrossRef] [PubMed]
- Bondia Pons, I.; Barri, T.; Hanhineva, K.; Juntunen, K.; Dragsted, L.O.; Mykkänen, H.; Poutanen, K. UPLC-QTOF/MS metabolic profiling unveils urinary changes in humans after a whole grain rye versus refined wheat bread intervention. Mol. Nutr. Food Res. 2013, 57, 412–422. [Google Scholar] [CrossRef] [PubMed]
- De Carvalho, F.G.; Ovídio, P.P.; Padovan, G.J.; Jordão Junior, A.A.; Marchini, J.S.; Navarro, A.M. Metabolic parameters of postmenopausal women after quinoa or corn flakes intake—A prospective and double-blind study. Int. J. Food Sci. Nutr. 2014, 65, 380–385. [Google Scholar] [CrossRef] [PubMed]
- Durazzo, A.; Carcea, M.; Adlercreutz, H.; Azzini, E.; Polito, A.; Olivieri, L.; Zaccaria, M.; Meneghini, C.; Maiani, F.; Bausano, G.; et al. Effects of consumption of whole grain foods rich in lignans in healthy postmenopausal women with moderate serum cholesterol: A pilot study. Int. J. Food Sci. Nutr. 2014, 65, 637–645. [Google Scholar] [CrossRef] [PubMed]
- Landberg, R.; Kamal Eldin, A.; Andersson, A.; Vessby, B.; Aman, P. Alkylresorcinols as biomarkers of whole-grain wheat and rye intake: Plasma concentration and intake estimated from dietary records. Am. J. Clin. Nutr. 2008, 87, 832–838. [Google Scholar] [CrossRef] [PubMed]
- Ross, A.B.; Bruce, S.J.; Blondel Lubrano, A.; Oguey Araymon, S.; Beaumont, M.; Bourgeois, A.; Nielsen Moennoz, C.; Vigo, M.; Fay, L.B.; Kochhar, S.; et al. A whole-grain cereal-rich diet increases plasma betaine and tends to decrease total and LDL-cholesterol compared with a refined-grain diet in healthy subjects. Br. J. Nutr. 2011, 105, 1492–1502. [Google Scholar] [CrossRef] [PubMed]
- Kristensen, M.; Toubro, S.; Jensen, M.G.; Ross, A.B.; Riboldi, G.; Petronio, M.; Bügel, S.; Tetens, I.; Astrup, A. Whole grain compared with refined wheat decreases the percentage of body fat following a 12-Week, energy-restricted dietary intervention in postmenopausal women. J. Nutr. 2012, 142, 710–716. [Google Scholar] [CrossRef] [PubMed]
- Giacco, R.; Lappi, J.; Costabile, G.; Kolehmainen, M.; Schwab, U.; Landberg, R.; Uusitupa, M.; Poutanen, K.; Pacini, G.; Rivellese, A.A.; et al. Effects of rye and whole wheat versus refined cereal foods on metabolic risk factors: A randomised controlled two-centre intervention study. Clin. Nutr. 2013, 32, 941–949. [Google Scholar] [CrossRef] [PubMed]
- Lappi, J.; Salojärvi, J.; Kolehmainen, M.; Mykkänen, H.; Poutanen, K.; de Vos, W.M.; Salonen, A. Intake of whole-grain and fiber-rich rye bread versus refined wheat bread does not differentiate intestinal microbiota composition in Finnish adults with metabolic syndrome. J. Nutr. 2013, 143, 648–655. [Google Scholar] [CrossRef] [PubMed]
- Ross, A.B.; Pere Trépat, E.; Montoliu, I.; Martin, F.P.J.; Collino, S.; Moco, S.; Godin, J.P.; Cléroux, M.; Guy, P.A.; Breton, I.; et al. A whole-grain-rich diet reduces urinary excretion of markers of protein catabolism and gut microbiota metabolism in healthy men after one week. J. Nutr. 2013, 143, 766–773. [Google Scholar] [CrossRef] [PubMed]
- Giacco, R.; Costabile, G.; Della Pepa, G.; Anniballi, G.; Griffo, E.; Mangione, A.; Cipriano, P.; Viscovo, D.; Clemente, G.; Landberg, R.; et al. A whole-grain cereal-based diet lowers postprandial plasma insulin and triglyceride levels in individuals with metabolic syndrome. Nutr. Metab. Cardiovasc. Dis. 2014, 24, 837–844. [Google Scholar] [CrossRef] [PubMed]
- Harris Jackson, K.; West, S.G.; Vanden Heuvel, J.P.; Jonnalagadda, S.S.; Ross, A.B.; Hill, A.M.; Grieger, J.A.; Lemieux, S.K.; Kris Etherton, P.M. Effects of whole and refined grains in a weight-loss diet on markers of metabolic syndrome in individuals with increased waist circumference: A randomized controlled-feeding trial. Am. J. Clin. Nutr. 2014, 100, 577–586. [Google Scholar] [CrossRef] [PubMed]
- Ampatzoglou, A.; Atwal, K.K.; Maidens, C.M.; Williams, C.L.; Ross, A.B.; Thielecke, F.; Jonnalagadda, S.S.; Kennedy, O.B.; Yaqoob, P. Increased whole grain consumption does not affect blood biochemistry, body composition, or gut microbiology in healthy, low-habitual whole grain consumers. J. Nutr. 2015, 145, 215–221. [Google Scholar] [CrossRef] [PubMed]
- Horner, N.K.; Kristal, A.R.; Prunty, J.; Skor, H.E.; Potter, J.D.; Lampe, J.W. Dietary Determinants of Plasma Enterolactone. Cancer Epidemiol. Biomark. Prev. 2002, 11, 121–126. [Google Scholar]
- Kelly, S.A.M.; Summerbell, C.D.; Brynes, A.; Whittaker, V.; Frost, G. Wholegrain cereals for coronary heart disease. Cochrane Database Syst. Rev. 2007. [Google Scholar] [CrossRef]
- Hollænder, P.L.B.; Ross, A.B.; Kristensen, M. Whole-grain and blood lipid changes in apparently healthy adults: A systematic review and meta-analysis of randomized controlled studies. Am. J. Clin. Nutr. 2015, 102, 556–572. [Google Scholar] [CrossRef] [PubMed]
- Pol, K.; Christensen, R.; Bartels, E.M.; Raben, A.; Tetens, I.; Kristensen, M. Whole grain and body weight changes in apparently healthy adults: A systematic review and meta-analysis of randomized controlled studies. Am. J. Clin. Nutr. 2013, 98, 872–884. [Google Scholar] [CrossRef] [PubMed]
- Ripsin, C.M.; Keenan, J.M.; Jacobs, D.R.; Elmer, P.J.; Welch, R.R.; Horn, L.V.; Liu, K.; Turnbull, W.H.; Thye, F.W.; Kestin, M.; et al. Oat Products and Lipid Lowering: A Meta-analysis. JAMA 1992, 267, 3317–3325. [Google Scholar] [CrossRef] [PubMed]
- Bao, L.; Cai, X.; Xu, M.; Li, Y. Effect of oat intake on glycaemic control and insulin sensitivity: A meta-analysis of randomised controlled trials. Br. J. Nutr. 2014, 112, 457–466. [Google Scholar] [CrossRef] [PubMed]
- Zou, Y.; Liao, D.; Huang, H.; Li, T.; Chi, H. A systematic review and meta-analysis of beta-glucan consumption on glycemic control in hypercholesterolemic individuals. Int. J. Food Sci. Nutr. 2015, 66, 355–362. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.; Sun, X.; Wang, M.; Zhang, C.; Cao, Y.; Mo, G.; Liang, J.; Zhu, S. Quantitative assessment of the effects of beta-glucan consumption on serum lipid profile and glucose level in hypercholesterolemic subjects. Nutr. Metab. Cardiovasc. Dis. 2015, 25, 714–723. [Google Scholar] [CrossRef] [PubMed]
- Cho, S.S.; Qi, L.; Fahey, G.C.; Klurfeld, D.M. Consumption of cereal fiber, mixtures of whole grains and bran and whole grains and risk reduction in type 2 diabetes, obesity and cardiovascular disease. Am. J. Clin. Nutr. 2013, 98, 594–619. [Google Scholar] [CrossRef] [PubMed]
- Health Canada Assessment of a Health Claim about Whole Grains and Coronary Heart Disease. Available online: http://www.hc-sc.gc.ca.ezproxy.library.tufts.edu/fn-an/label-etiquet/claims-reclam/assess-evalu/grains-heart-coeur-eng.php (accessed on 27 January 2017).

| Characteristic | Acute | Moderate (>1 Day––6 Weeks) | Long (>6 Weeks) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Total | RCT | Other 1 | Total | RTC | Other 2 | Total | RTC | Other 3 | |
| Number of trials, n | 118 | 101 | 17 | 63 | 57 | 6 | 32 | 31 | 1 |
| Parallel, n (%) | - | 1 (1) | - | - | 20 (35) | - | - | 26 (84) | - |
| Crossover, n (%) | - | 100 (99) | - | - | 37 (65) | - | - | 1 (3) | - |
| Duration, n (%) | |||||||||
| ≤ 1 day | 118 (100) | 101 (100) | 17 (100) | - | - | - | - | - | - |
| < 1 week | - | - | - | 10 (16) | 7 (12) | 3 (50) | - | - | - |
| 1 to 6 weeks | - | - | - | 53 (84) | 50 (88) | 3 (50) | - | - | - |
| 6 to 12 weeks | - | - | - | - | - | - | 25 (78) | 24 (77) | 1 (100) |
| >12 weeks | - | - | - | - | - | - | 7 (22) | 7 (23) | 0 (0) |
| Sample size, n (%) | |||||||||
| ≤10 | 39 (33) | 29 (29) | 10 (59) | 4 (6) | 2 (4) | 2 (33) | 0 (0) | 0 (0) | 0 (0) |
| 11–25 | 58 (49) | 54 (53) | 4 (24) | 27 (43) | 23 (40) | 4 (67) | 4 (13) | 4 (13) | 0 (0) |
| 26–50 | 16 (14) | 15 (15) | 1 (6) | 18 (29) | 18 (32) | 0 (0) | 9 (28) | 8 (26) | 1 (100) |
| >50 | 5 (4) | 3 (3) | 2 (12) | 14 (22) | 14 (25) | 0 (0) | 19 (59) | 19 (61) | 0 (0) |
| Age group, n (%) | |||||||||
| Adults (≥18 year) | 115 (97) | 98 (97) | 17 (100) | 60 (95) | 54 (95) | 6 (100) | 31 (97) | 31 (100) | 0 (0) |
| Adolescents (12–17) | 0 (0) | 0 (0) | 0 (0) | 2 (3) | 2 (4) | 0 (0) | 0 (0) | 0 (0) | 0 (0) |
| Children (3–11 year) | 2 (2) | 2 (2) | 0 (0) | 1 (2) | 1 (2) | 0 (0) | 0 (0) | 0 (0) | 0 (0) |
| NR | 1 (1) | 1 (1) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 1 (3) | 0 (0) | 1 (100) |
| Sex, mean % male ± SD | 54 ± 27 | 55 ± 26 | 50 ± 32 | 44 ± 25 | 43 ± 23 | 49 ± 42 | 42 ± 21 | 40 ± 18 | 100 ± 0 |
| Baseline health, n (%) | |||||||||
| Healthy | 79 (67) | 67 (66) | 12 (71) | 30 (48) | 26 (46) | 4 (67) | 4 (13) | 4 (13) | 0 (0) |
| Overweight/Obese | 19 (16) | 18 (18) | 1 (6) | 19 (30) | 18 (32) | 1 (17) | 19 (59) | 18 (58) | 1 (100) |
| T1DM | 2 (2) | 1 (1) | 1 (6) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) |
| T2DM | 18 (15) | 15 (15) | 3 (18) | 5 (8) | 3 (5) | 2 (33) | 4 (12) | 4 (13) | 0 (0) |
| MetS (≥1 criteria) 4 | 8 (7) | 7 (7) | 1 (6) | 20 (32) | 19 (33) | 1 (17) | 15 (47) | 14 (45) | 1 (100) |
| Digestive Issues | 3 (3) | 2 (2) | 1 (6) | 3 (5) | 3 (5) | 0 (0) | 1 (3) | 1 (3) | 0 (0) |
| Region, n (%) | |||||||||
| Europe | 61 (52) | 54 (53) | 7 (41) | 28 (44) | 24 (42) | 4 (67) | 17 (53) | 17 (55) | 0 (0) |
| North America | 36 (31) | 33 (33) | 3 (18) | 21 (33) | 20 (35) | 1 (17) | 8 (25) | 8 (26) | 0 (0) |
| Asia | 12 (10) | 5 (5) | 7 (41) | 10 (16) | 9 (16) | 1 (17) | 6 (19) | 5 (16) | 1 (100) |
| Australia | 6 (5) | 6 (6) | 0 (0) | 3 (5) | 3 (5) | 0 (0) | 0 (0) | 0 (0) | 0 (0) |
| Africa | 2 (2) | 2 (2) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) |
| South America | 0 (0) | 0 (0) | 0 (0) | 1 (2) | 1 (2) | 0 (0) | 0 (0) | 0 (0) | 0 (0) |
| Multiple | 1 (1) | 1 (1) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 1 (3) | 1 (3) | 0 (0) |
| Controls 5, n (%) | |||||||||
| Refined grain | 98 (74) | 86 (75) | 12 (67) | 47 (71) | 44 (75) | 3 (43) | 22 (69) | 21 (68) | 1 (100) |
| Usual diet | 0 (0) | 0 (0) | 0 (0) | 2 (3) | 1 (2) | 1 (14) | 5 (16) | 5 (16) | 0 (0) |
| Matched, no WG 6 | 4 (3) | 3 (3) | 1 (6) | 4 (6) | 4 (7) | 0 (0) | 2 (6) | 2 (6) | 0 (0) |
| Other 7/unspecified | 21 (16) | 17 (15) | 4 (22) | 6 (9) | 6 (10) | 0 (0) | 3 (9) | 3 (10) | 0 (0) |
| Crossover of WG only 8 | 10 (8) | 9 (8) | 1 (6) | 4 (6) | 4 (7) | 0 (0) | 0 (0) | 0 (0) | 0 (0) |
| No control arm | 0 (0) | 0 (0) | 0 (0) | 3 (5) | 0 (0) | 3 (43) | 0 (0) | 0 (0) | 0 (0) |
| Reporting Criteria | n (%) |
|---|---|
| Reporting of dose | |
| Amount of grain | 52 (55) |
| Amount of food/product | 34 (36) |
| Not reported 1 | 9 (9) |
| Reported a definition of whole grain | |
| Referenced established definition 2 | 10 (12) |
| Described WG or WG food | 12 (14) |
| Not reported | 61 (73) |
| Reported distribution of grain types3 | 18 (46) |
| Control | |
| Refined grain | 69 (70) |
| Usual diet | 7 (7) |
| Other 4 | 22 (22) |
| Adherence5 | |
| Questionnaires/Records | 69 (73) |
| Food weighing or observation | 14 (15) |
| Biomarkers | 26 (22) |
| Not reported | 19 (20) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sawicki, C.M.; Livingston, K.A.; Ross, A.B.; Jacques, P.F.; Koecher, K.; McKeown, N.M. Evaluating Whole Grain Intervention Study Designs and Reporting Practices Using Evidence Mapping Methodology. Nutrients 2018, 10, 1052. https://doi.org/10.3390/nu10081052
Sawicki CM, Livingston KA, Ross AB, Jacques PF, Koecher K, McKeown NM. Evaluating Whole Grain Intervention Study Designs and Reporting Practices Using Evidence Mapping Methodology. Nutrients. 2018; 10(8):1052. https://doi.org/10.3390/nu10081052
Chicago/Turabian StyleSawicki, Caleigh M., Kara A. Livingston, Alastair B. Ross, Paul F. Jacques, Katie Koecher, and Nicola M. McKeown. 2018. "Evaluating Whole Grain Intervention Study Designs and Reporting Practices Using Evidence Mapping Methodology" Nutrients 10, no. 8: 1052. https://doi.org/10.3390/nu10081052
APA StyleSawicki, C. M., Livingston, K. A., Ross, A. B., Jacques, P. F., Koecher, K., & McKeown, N. M. (2018). Evaluating Whole Grain Intervention Study Designs and Reporting Practices Using Evidence Mapping Methodology. Nutrients, 10(8), 1052. https://doi.org/10.3390/nu10081052





