Blackberry Feeding Increases Fat Oxidation and Improves Insulin Sensitivity in Overweight and Obese Males
Abstract
1. Introduction
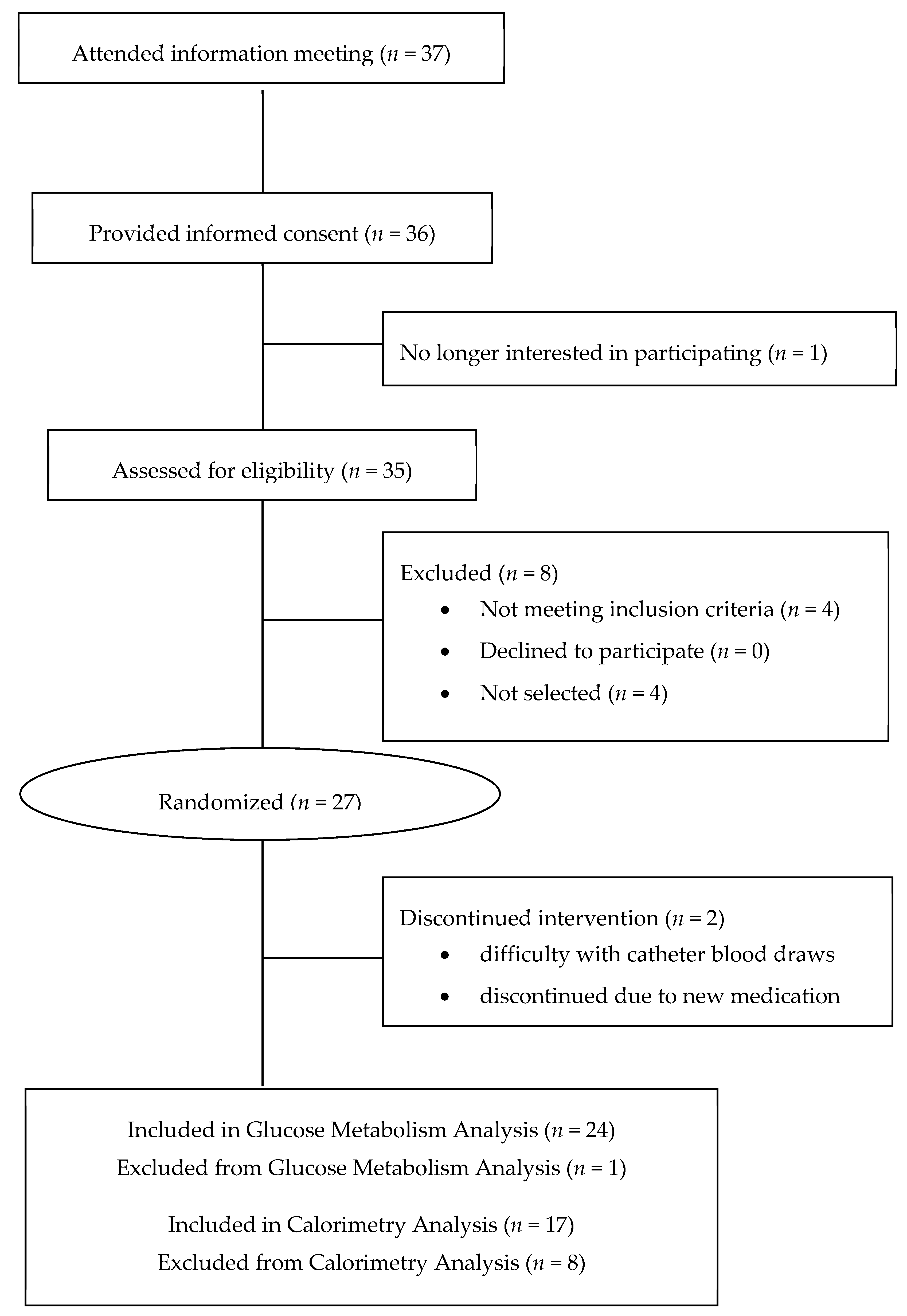
2. Materials and Methods
2.1. Study Design
2.2. Diets
2.3. Assessments Sample Collection and Analysis
2.4. Calculations and Statistics
3. Results
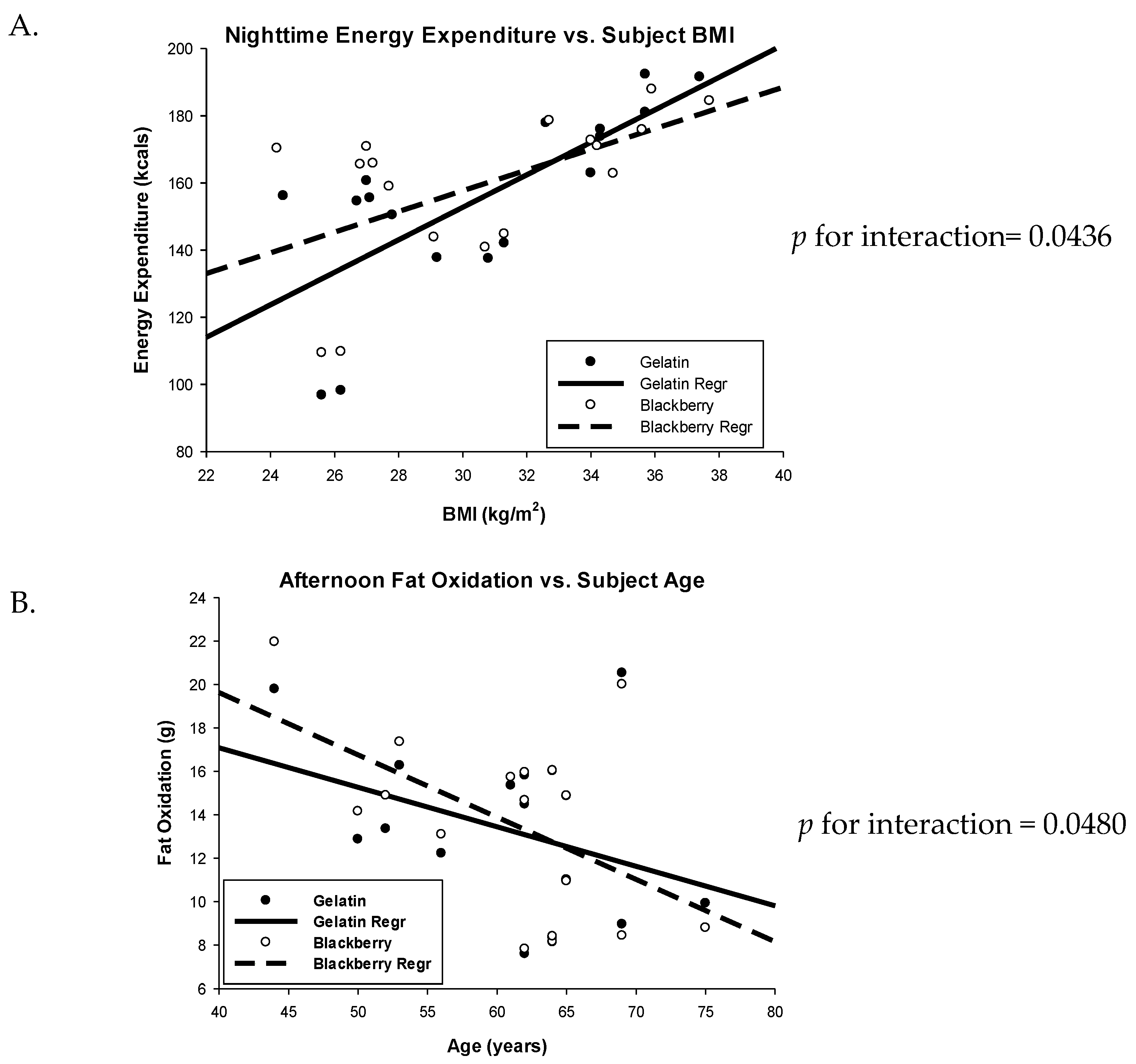
3.1. Energy Expenditure
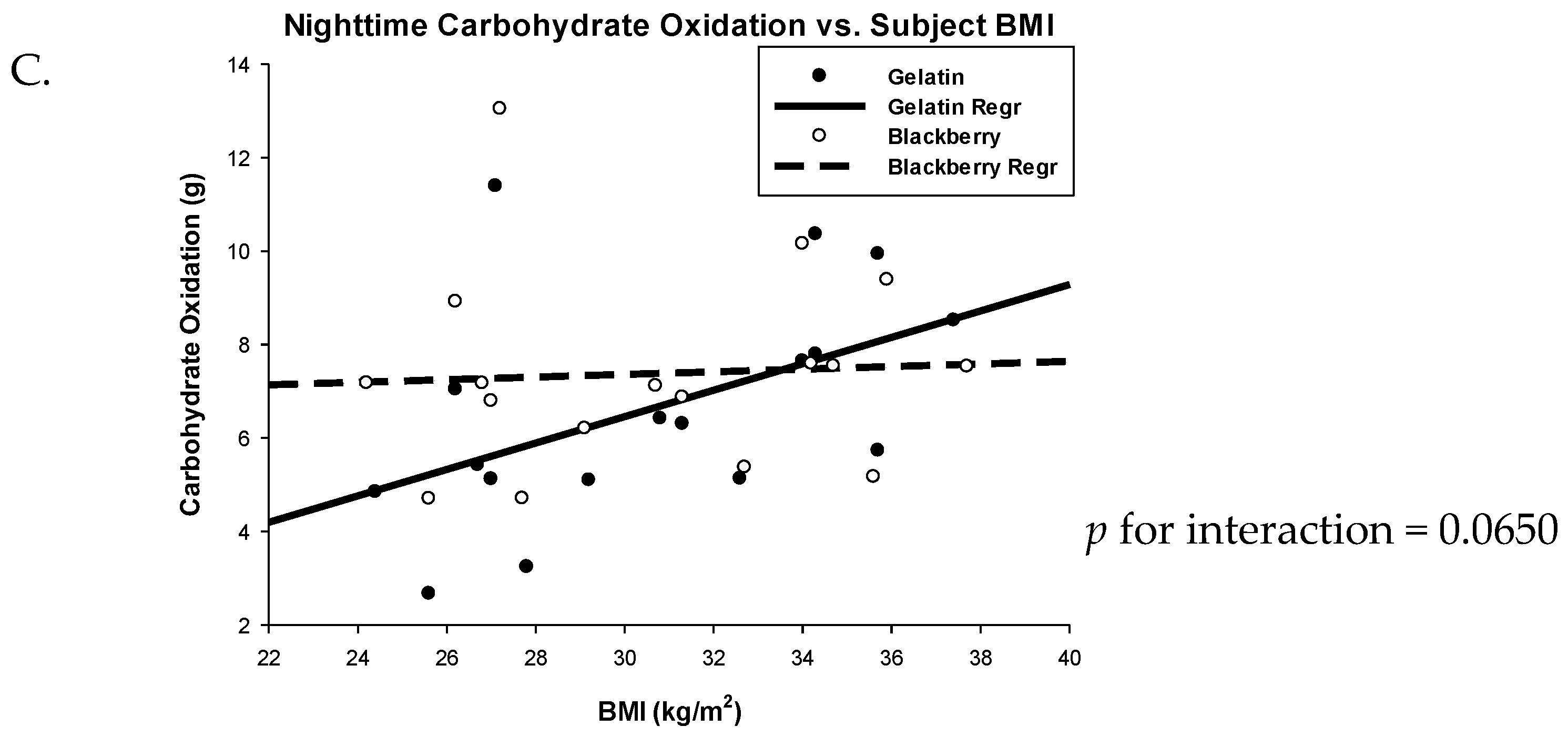
3.2. Respiratory Quotient and Fuel Oxidation
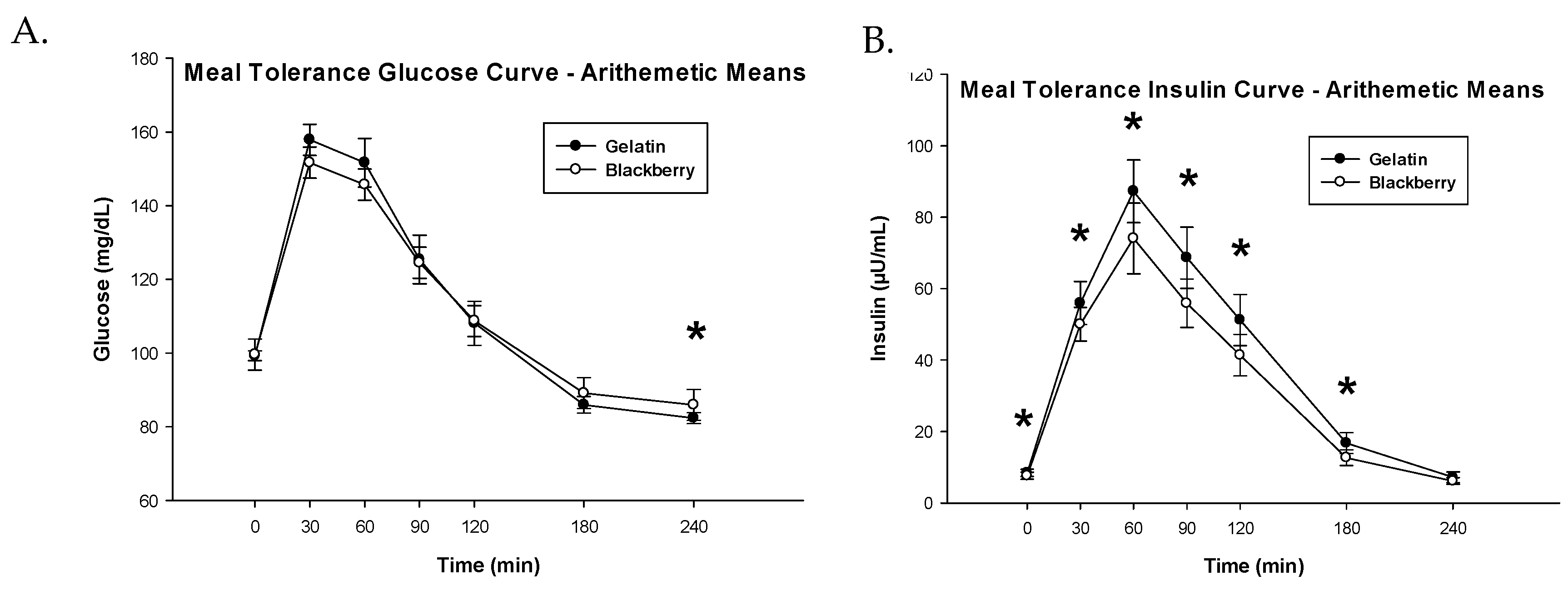
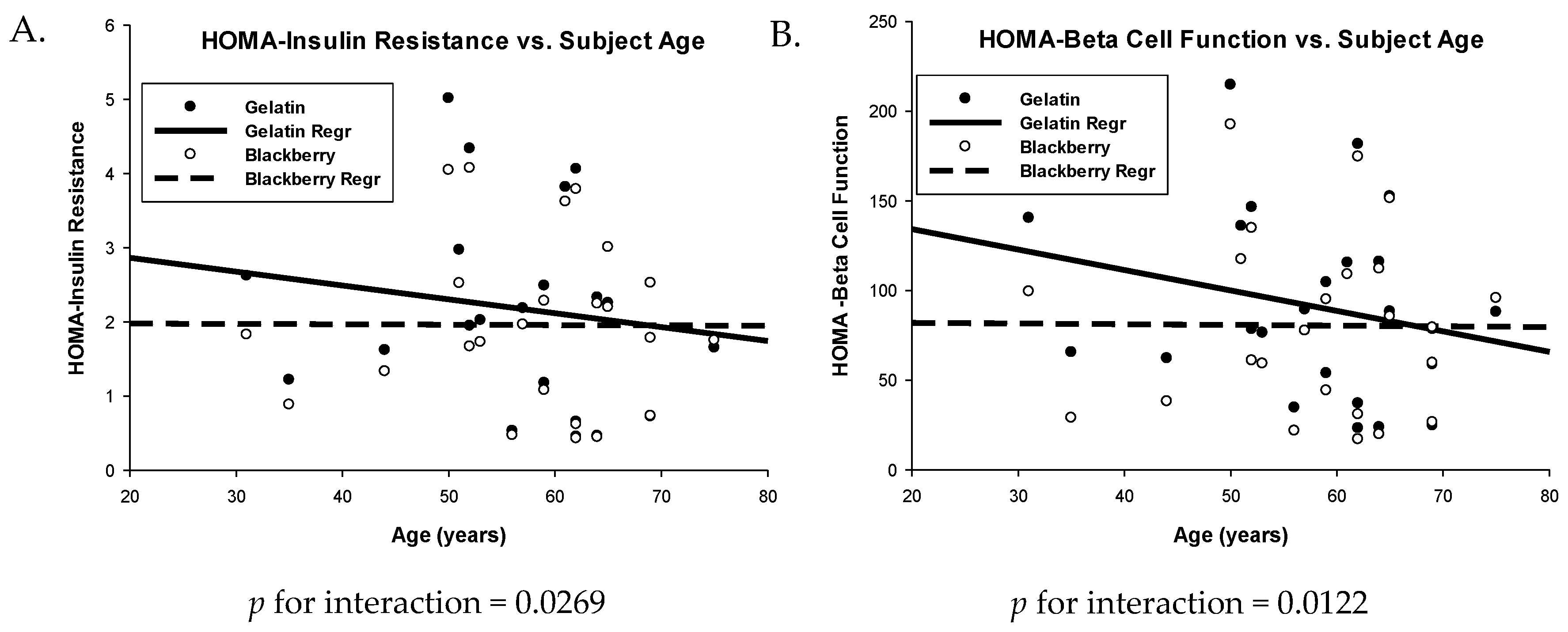
3.3. Glucose Metabolism and Insulin Sensitivity
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ogden, C.L.; Carroll, M.D.; Kit, B.K.; Flegal, K.M. Prevalence of childhood and adult obesity in the united states, 2011–2012. JAMA 2014, 311, 806–814. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control, Department of Health and Human Services. Adult Obesity Causes & Consequences. Available online: https://www.cdc.gov/obesity/adult/causes.html (accessed on 12 January 2017).
- Dunkley, A.J.; Charles, K.; Gray, L.J.; Camosso-Stefinovic, J.; Davies, M.J.; Khunti, K. Effectiveness of interventions for reducing diabetes and cardiovascular disease risk in people with metabolic syndrome: Systematic review and mixed treatment comparison meta-analysis. Diabetes Obes. Metab. 2012, 14, 616–625. [Google Scholar] [CrossRef] [PubMed]
- Dauchet, L.; Amouyel, P.; Hercberg, S.; Dallongeville, J. Fruit and vegetable consumption and risk of coronary heart disease: A meta-analysis of cohort studies. J. Nutr. 2006, 136, 2588–2593. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Ouyang, Y.Y.; Liu, J.; Zhu, M.M.; Zhao, G.; Bao, W.; Hu, F.B. Fruit and vegetable consumption and mortality from all causes, cardiovascular disease, and cancer: Systematic review and dose-response meta-analysis of prospective cohort studies. Br. Med. J. 2014, 349, g4490. [Google Scholar] [CrossRef] [PubMed]
- Tresserra-Rimbau, A.; Rimm, E.B.; Medina-Remon, A.; Martinez-Gonzalez, M.A.; Lopez-Sabater, M.C.; Covas, M.I.; Corella, D.; Salas-Salvado, J.; Gomez-Gracia, E.; Lapetra, J.; et al. Polyphenol intake and mortality risk: A re-analysis of the predimed trial. BMC Med. 2014, 12, 77. [Google Scholar] [CrossRef] [PubMed]
- Nile, S.H.; Park, S.W. Edible berries: Bioactive components and their effect on human health. Nutrition 2014, 30, 134–144. [Google Scholar] [CrossRef] [PubMed]
- Tsuda, T.; Horio, F.; Uchida, K.; Aoki, H.; Osawa, T. Dietary cyanidin 3-o-beta-d-glucoside-rich purple corn color prevents obesity and ameliorates hyperglycemia in mice. J. Nutr. 2003, 133, 2125–2130. [Google Scholar] [CrossRef] [PubMed]
- Farrell, N.J.; Norris, G.H.; Ryan, J.; Porter, C.M.; Jiang, C.; Blesso, C.N. Black elderberry extract attenuates inflammation and metabolic dysfunction in diet-induced obese mice. Br. J. Nutr. 2015, 114, 1123–1131. [Google Scholar] [CrossRef] [PubMed]
- Heyman, L.; Axling, U.; Blanco, N.; Sterner, O.; Holm, C.; Berger, K. Evaluation of beneficial metabolic effects of berries in high-fat fed c57bl/6j mice. J. Nutr. Metab. 2014, 2014. Available online: https://www.hindawi.com/journals/jnme/2014/403041/abs/ (accessed on 4 April 2014). [CrossRef] [PubMed]
- Prior, R.L.; Wilkes, S.; Rogers, T.; Khanal, R.C.; Wu, X.L.; Hager, T.J.; Hager, A.; Howard, L. Dietary black raspberry anthocyanins do not alter development of obesity in mice fed an obesogenic high-fat diet. J. Agric. Food Chem. 2010, 58, 3977–3983. [Google Scholar] [CrossRef] [PubMed]
- Prior, R.L.; Wilkes, S.E.; Rogers, T.R.; Khanal, R.C.; Wu, X.L.; Howard, L.R. Purified blueberry anthocyanins and blueberry juice alter development of obesity in mice fed an obesogenic high-fat diet. J. Agric. Food Chem. 2010, 58, 3970–3976. [Google Scholar] [CrossRef] [PubMed]
- Prior, R.L.; Wu, X.L.; Gu, L.W.; Hager, T.J.; Hager, A.; Howard, L.R. Whole berries versus berry anthocyanins: Interactions with dietary fat levels in the c57bl/6j mouse model of obesity. J. Agric. Food Chem. 2008, 56, 647–653. [Google Scholar] [CrossRef] [PubMed]
- Seymour, E.M.; Singer, A.A.M.; Kirakosyan, A.; Urcuyo-Llanes, D.E.; Kaufman, P.B.; Bolling, S.F. Altered hyperlipidemia, hepatic steatosis, and hepatic peroxisome proliferator-activated receptors in rats with intake of tart cherry. J. Med. Food 2008, 11, 252–259. [Google Scholar] [CrossRef] [PubMed]
- Seymour, E.M.; Tanone, I.I.; Urcuyo-Llanes, D.E.; Lewis, S.K.; Kirakosyan, A.; Kondoleon, M.G.; Kaufman, P.B.; Bolling, S.F. Blueberry intake alters skeletal muscle and adipose tissue peroxisome proliferator-activated receptor activity and reduces insulin resistance in obese rats. J. Med. Food 2011, 14, 1511–1518. [Google Scholar] [CrossRef] [PubMed]
- Takikawa, M.; Inoue, S.; Horio, F.; Tsuda, T. Dietary anthocyanin-rich bilberry extract ameliorates hyperglycemia and insulin sensitivity via activation of amp-activated protein kinase in diabetic mice. J. Nutr. 2010, 140, 527–533. [Google Scholar] [CrossRef] [PubMed]
- Webb, P. 24-hour energy expenditure and the menstrual cycle. Am. J. Clin. Nutr. 1986, 44, 614–619. [Google Scholar] [CrossRef] [PubMed]
- 2015–2020 Dietary Guidelines for Americans; Department of Health and Human Services and Department of Agriculture: Washington, DC, USA, 2015.
- Redan, B.W.; Albaugh, G.P.; Charron, C.S.; Novotny, J.A.; Ferruzzi, M.G. Adaptation in caco-2 human intestinal cell differentiation and phenolic transport with chronic exposure to blackberry (rubus sp.) extract. J. Agric. Food Chem. 2017, 65, 2694–2701. [Google Scholar] [CrossRef] [PubMed]
- Seale, J.L.; Rumpler, W.V.; Moe, P.W. Description of a direct-indirect room-sized calorimeter. Am. J. Physiol. 1991, 260, 306–320. [Google Scholar] [CrossRef] [PubMed]
- Gribok, A.; Hoyt, R.; Buller, M.; Rumpler, W. On the accuracy of instantaneous gas exchange rates, energy expenditure and respiratory quotient calculations obtained from indirect whole room calorimetry. Physiol. Meas. 2013, 34, 737–755. [Google Scholar] [CrossRef] [PubMed]
- Gribok, A.; Leger, J.L.; Stevens, M.; Hoyt, R.; Buller, M.; Rumpler, W. Measuring the short-term substrate utilization response to high-carbohydrate and high-fat meals in the whole-body indirect calorimeter. Physiol. Rep. 2016, 4, e12835. [Google Scholar] [CrossRef] [PubMed]
- Livesey, G.; Elia, M. Estimation of energy expenditure, net carbohydrate utilization, and net fat oxidation and synthesis by indirect calorimetry: Evaluation of errors with special reference to the detailed composition of fuels. Am. J. Clin. Nutr. 1988, 47, 608–628. [Google Scholar] [CrossRef] [PubMed]
- Wallace, T.M.; Levy, J.C.; Matthews, D.R. Use and abuse of homa modeling. Diabetes Care 2004, 27, 1487–1495. [Google Scholar] [CrossRef] [PubMed]
- Bhaswant, M.; Fanning, K.; Netzel, M.; Mathai, M.L.; Panchal, S.K.; Brown, L. Cyanidin 3-glucoside improves diet-induced metabolic syndrome in rats. Pharmacol. Res. 2015, 102, 208–217. [Google Scholar] [CrossRef] [PubMed]
- Wei, X.; Wang, D.; Yang, Y.; Xia, M.; Li, D.; Li, G.; Zhu, Y.; Xiao, Y.; Ling, W. Cyanidin-3-o-beta-glucoside improves obesity and triglyceride metabolism in kk-ay mice by regulating lipoprotein lipase activity. J. Sci. Food Agric. 2011, 91, 1006–1013. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.H.; Chen, G.Z.; Liao, D.; Zhu, Y.K.; Xue, X.Y. Effects of berries consumption on cardiovascular risk factors: A meta-analysis with trial sequential analysis of randomized controlled trials. Sci. Rep. UK 2016, 23625. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.; Yang, B.; Tan, J.; Jiang, J.; Li, D. Associations of dietary intakes of anthocyanins and berry fruits with risk of type 2 diabetes mellitus: A systematic review and meta-analysis of prospective cohort studies. Eur J. Clin. Nutr. 2016, 70, 1360–1367. [Google Scholar] [CrossRef] [PubMed]
- Qin, Y.; Xia, M.; Ma, J.; Hao, Y.; Liu, J.; Mou, H.; Cao, L.; Ling, W. Anthocyanin supplementation improves serum ldl- and hdl-cholesterol concentrations associated with the inhibition of cholesteryl ester transfer protein in dyslipidemic subjects. Am. J. Clin. Nutr. 2009, 90, 485–492. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Xia, M.; Yang, Y.; Liu, F.; Li, Z.; Hao, Y.; Mi, M.; Jin, T.; Ling, W. Purified anthocyanin supplementation improves endothelial function via no-cgmp activation in hypercholesterolemic individuals. Clin. Chem. 2011, 57, 1524–1533. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Huang, X.; Zhang, Y.; Wang, Y.; Liu, Y.; Sun, R.; Xia, M. Anthocyanin supplementation improves hdl-associated paraoxonase 1 activity and enhances cholesterol efflux capacity in subjects with hypercholesterolemia. J. Clin. Endocrinol. Metab. 2014, 99, 561–569. [Google Scholar] [CrossRef] [PubMed]
- Hoggard, N.; Cruickshank, M.; Moar, K.M.; Bestwick, C.; Holst, J.J.; Russell, W.; Horgan, G. A single supplement of a standardised bilberry (Vaccinium myrtillus L.) extract (36% wet weight anthocyanins) modifies glycaemic response in individuals with type 2 diabetes controlled by diet and lifestyle. J. Nutr. Sci. 2013, 2, e22. [Google Scholar] [CrossRef] [PubMed]
- Stull, A.J.; Cash, K.C.; Johnson, W.D.; Champagne, C.M.; Cefalu, W.T. Bioactives in blueberries improve insulin sensitivity in obese, insulin-resistant men and women. J. Nutr. 2010, 140, 1764–1768. [Google Scholar] [CrossRef] [PubMed]
- Stull, A.J.; Cash, K.C.; Champagne, C.M.; Gupta, A.K.; Boston, R.; Beyl, R.A.; Johnson, W.D.; Cefalu, W.T. Blueberries improve endothelial function, but not blood pressure, in adults with metabolic syndrome: A randomized, double-blind, placebo-controlled clinical trial. Nutrients 2015, 7, 4107–4123. [Google Scholar] [CrossRef] [PubMed]
- Basu, A.; Fu, D.X.; Wilkinson, M.; Simmons, B.; Wu, M.Y.; Betts, N.M.; Du, M.; Lyons, T.J. Strawberries decrease atherosclerotic markers in subjects with metabolic syndrome. Nutr. Res. 2010, 30, 462–469. [Google Scholar] [CrossRef] [PubMed]
- Basu, A.; Du, M.; Leyva, M.J.; Sanchez, K.; Betts, N.M.; Wu, M.; Aston, C.E.; Lyons, T.J. Blueberries decrease cardiovascular risk factors in obese men and women with metabolic syndrome. J. Nutr. 2010, 140, 1582–1587. [Google Scholar] [CrossRef] [PubMed]
- Cook, M.D.; Myers, S.D.; Blacker, S.D.; Willems, M.E. New zealand blackcurrant extract improves cycling performance and fat oxidation in cyclists. Eur. J. Appl. Physiol. 2015, 115, 2357–2365. [Google Scholar] [CrossRef] [PubMed]
- Rumpler, W.; Seale, J.; Clevidence, B.; Judd, J.; Wiley, E.; Yamamoto, S.; Komatsu, T.; Sawaki, T.; Ishikura, Y.; Hosoda, K. Oolong tea increases metabolic rate and fat oxidation in men. J. Nutr. 2001, 131, 2848–2852. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Yang, Z.; Hosoda, K.; Chen, L.; Lin, B.; Kimura, J.; Matsui, Y.; Matsui, K. Clinical efficacy of oolong tea on anti-simple obesity. Jpn. J. Clin. Nutr. 1998, 20, 83–90. [Google Scholar]
- Dulloo, A.G.; Duret, C.; Rohrer, D.; Girardier, L.; Mensi, N.; Fathi, M.; Chantre, P.; Vandermander, J. Efficacy of a green tea extract rich in catechin polyphenols and caffeine in increasing 24-h energy expenditure and fat oxidation in humans. Am. J. Clin. Nutr. 1999, 70, 1040–1045. [Google Scholar] [CrossRef] [PubMed]
- Rudelle, S.; Ferruzzi, M.G.; Cristiani, I.; Moulin, J.; Mace, K.; Acheson, K.J.; Tappy, L. Effect of a thermogenic beverage on 24-hour energy metabolism in humans. Obesity 2007, 15, 349–355. [Google Scholar] [CrossRef] [PubMed]
- Gahreman, D.; Wang, R.; Boutcher, Y.; Boutcher, S. Green tea, intermittent sprinting exercise, and fat oxidation. Nutrients 2015, 7, 5646–5663. [Google Scholar] [CrossRef] [PubMed]
- Thielecke, F.; Rahn, G.; Bohnke, J.; Adams, F.; Birkenfeld, A.L.; Jordan, J.; Boschmann, M. Epigallocatechin-3-gallate and postprandial fat oxidation in overweight/obese male volunteers: A pilot study. Eur. J. Clin. Nutr. 2010, 64, 704–713. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Li, D.; Zhang, Y.; Sun, R.; Xia, M. Anthocyanin increases adiponectin secretion and protects against diabetes-related endothelial dysfunction. Am. J. Physiol. Endocrinol. Metab. 2014, 306, 975–988. [Google Scholar] [CrossRef] [PubMed]
- Park, E.; Edirisinghe, I.; Wei, H.; Vijayakumar, L.P.; Banaszewski, K.; Cappozzo, J.C.; Burton-Freeman, B. A dose-response evaluation of freeze-dried strawberries independent of fiber content on metabolic indices in abdominally obese individuals with insulin resistance in a randomized, single-blinded, diet-controlled crossover trial. Mol. Nutr. Food Res. 2016, 60, 1099–1109. [Google Scholar] [CrossRef] [PubMed]
- Moazen, S.; Amani, R.; Homayouni Rad, A.; Shahbazian, H.; Ahmadi, K.; Taha Jalali, M. Effects of freeze-dried strawberry supplementation on metabolic biomarkers of atherosclerosis in subjects with type 2 diabetes: A randomized double-blind controlled trial. Ann. Nutr. Metab. 2013, 63, 256–264. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.Y.; Fang, J.C.; Gao, Z.H.; Zhang, C.; Xie, S.Y. Higher intake of fruits, vegetables or their fiber reduces the risk of type 2 diabetes: A meta-analysis. J. Diabetes Investig. 2016, 7, 56–69. [Google Scholar] [CrossRef] [PubMed]
- Tsuda, T. Recent progress in anti-obesity and anti-diabetes effect of berries. Antioxidants 2016, 5, 13. [Google Scholar] [CrossRef] [PubMed]
- Matsukawa, T.; Villareal, M.O.; Motojima, H.; Isoda, H. Increasing camp levels of preadipocytes by cyanidin-3-glucoside treatment induces the formation of beige phenotypes in 3t3-l1 adipocytes. J. Nutr. Biochem. 2016, 40, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Matsui, T.; Ueda, T.; Oki, T.; Sugita, K.; Terahara, N.; Matsumoto, K. Alpha-glucosidase inhibitory action of natural acylated anthocyanins. 1. Survey of natural pigments with potent inhibitory activity. J. Agric. Food Chem. 2001, 49, 1948–1951. [Google Scholar] [CrossRef] [PubMed]
- Stefan, N.; Stumvoll, M.; Bogardus, C.; Tataranni, P.A. Elevated plasma nonesterified fatty acids are associated with deterioration of acute insulin response in igt but not ngt. Am. J. Physiol. Endocrinol. Metab. 2003, 284, 1156–1161. [Google Scholar] [CrossRef] [PubMed]
- Walker, M.; Agius, L.; Orskov, H.; Alberti, K.G. Peripheral and hepatic insulin sensitivity in non-insulin-dependent diabetes mellitus: Effect of nonesterified fatty acids. Metabolism 1993, 42, 601–608. [Google Scholar] [CrossRef]
- Atlas of Atherosclerosis and Metabolic Syndrome; Grundy, S.M., Ed.; Springer: New York, NY, USA, 2011. [Google Scholar]
- Ramos-Roman, M.A.; Lapidot, S.A.; Phair, R.D.; Parks, E.J. Insulin activation of plasma nonesterified fatty acid uptake in metabolic syndrome. Arterioscler. Thromb. Vasc. Biol. 2012, 32, 1799–1808. [Google Scholar] [CrossRef] [PubMed]
- Lambert, J.E.; Parks, E.J. Postprandial metabolism of meal triglyceride in humans. Biochim. Biophys. Acta. 2012, 1821, 721–726. [Google Scholar] [CrossRef] [PubMed]
| Flavonoid | Per 300 g Serving | Per 600 g Daily Dose |
|---|---|---|
| Cyanidin 3-glucoside (mg) | 180.7 | 361.3 |
| Catechin (mg) | 32.9 | 65.8 |
| Epicatechin (mg) | 200.9 | 401.7 |
| Kaempferol glucoside (mg) | 283.7 | 567.3 |
| Quercetin glucoside (mg) | 39.7 | 79.4 |
| Total measured flavonoids (mg) | 737.9 | 1475.8 |
| Quantity (g) | Protein (g) | CHO 1 (g) | Sugar (g) | Fat (g) | Energy (kcal) | |
|---|---|---|---|---|---|---|
| Blackberry treatment | ||||||
| Waffles | 56.5 | 3.3 | 20.5 | 2.4 | 3.7 | 128.5 |
| Pancake Syrup | 60 | 0 | 40.2 | 36.3 | 0 | 160.8 |
| Blackberries, unsweetened | 302 | 3.6 | 47.3 | 32.2 | 1.3 | 215.3 |
| Total | 418.5 | 6.9 | 108 | 70.9 | 5 | 504.6 |
| Gelatin treatment | ||||||
| Waffles | 56.5 | 3.3 | 20.5 | 2.4 | 3.7 | 128.5 |
| Pancake Syrup | 60 | 0 | 40.2 | 36.3 | 0 | 160.8 |
| Gelatin, strawberry | 273 | 2.8 | 45.5 | 43.2 | 0 | 193.2 |
| Total | 389.5 | 6.1 | 106.2 | 81.9 | 3.7 | 482.5 |
| Glucose Tolerance (means ± SE, n = 24) | Calorimetry (means ± SE, n = 17) | p1 | |
|---|---|---|---|
| Age (years) | 57.8 ± 2.1 | 61 ± 1.9 | 0.2868 |
| Weight (kg) | 95.7 ± 2.6 | 94.2 ± 3.3 | 0.7319 |
| BMI (kg/m2) | 30.6 ± 0.8 | 30.7 ± 1 | 0.9463 |
| Total Cholesterol (mg/dL) | 186.9 ± 8.7 | 175.8 ± 11.7 | 0.4426 |
| LDL Cholesterol (mg/dL) | 116 ± 7.6 | 104.7 ± 9.8 | 0.3589 |
| HDL Cholesterol (mg/dL) | 46.1 ± 2.2 | 47.5 ± 3 | 0.7007 |
| Triglycerides (mg/dL) | 124.1 ± 11.6 | 118.2 ± 12 | 0.7338 |
| Systolic BP (mm Hg) | 123.3 ± 3 | 123.1 ± 3.5 | 0.9629 |
| Diastolic BP (mm Hg) | 75.6 ± 1.7 | 76.2 ± 1.9 | 0.8202 |
| Glucose (mg/dL) | 98.6 ± 1.4 | 97.6 ± 1.4 | 0.6403 |
| Gelatin | Blackberry | |||
|---|---|---|---|---|
| LSmean (95% CI) | LSmean (95% CI) | Difference (95% CI) | p1 | |
| RQ (CO2/O2) | ||||
| 24 h | 0.8171 (0.8079, 0.8262) | 0.8101 (0.801, 0.8192) | 0.0069 (0.0004, 0.0135) | 0.0402 |
| Evening | 0.8358 (0.8229, 0.8486) | 0.8178 (0.805, 0.8306) | 0.018 (0.00594, 0.0301) | 0.0063 |
| Nighttime | 0.7928 (0.779, 0.8066) | 0.796 (0.7822, 0.8098) | −0.0031 (−0.0135, 0.0072) | 0.5289 |
| Morning | 0.8512 (0.8403, 0.862) | 0.8416 (0.8308, 0.8525) | 0.0095 (0.0036, 0.0155) | 0.0036 |
| Afternoon | 0.8135 (0.8003, 0.8267) | 0.8098 (0.7966, 0.823) | 0.0037 (−0.0049, 0.0123) | 0.3806 |
| Exercise | 0.8708 (0.8615, 0.8801) | 0.8553 (0.846, 0.8646) | 0.0155 (0.0057, 0.0253) | 0.0041 |
| Fat Oxidation (g) | ||||
| 24 h | 132.38 (117.22, 147.53) | 140.84 (125.69, 156) | −8.47 (−16.59, −0.35) | 0.042 |
| Evening | 24.6 (21.34, 27.85) | 27.98 (24.73, 31.24) | −3.39 (−5.70, −1.07) | 0.0069 |
| Nighttime | 9.49 (8.34, 10.63) | 9.48 (8.34, 10.63) | 0.01 (−1.61, 1.62) | 0.9935 |
| Morning | 19.35 (16.58, 22.12) | 21.30 (18.53, 24.07) | −1.95 (−3.43, −0.47) | 0.0129 |
| Afternoon | 13.30 (11.37, 15.23) | 13.63 (11.70, 15.56) | −0.33 (−1.08, 0.42) | 0.3671 |
| Exercise | 8.32 (7.06, 9.59) | 9.35 (8.08, 10.62) | −1.03 (−1.68, −0.37) | 0.0044 |
| Carbohydrate Oxidation (g) | ||||
| 24 h | 182.71 (166.19, 199.24) | 172.95 (156.42, 189.47) | 9.77 (−0.80, 20.33) | 0.0678 |
| Evening | 41.56 (37.14, 45.98) | 35.87 (31.45, 40.29) | 5.69 (1.87, 9.51) | 0.0061 |
| Nighttime | 6.6 (5.11, 8.09) | 7.35 (5.86, 8.83) | −0.75 (−1.86, 0.37) | 0.1727 |
| Morning | 43.11 (39.80, 46.42) | 41.13 (37.82, 44.44) | 1.98 (−0.85, 4.81) | 0.1567 |
| Afternoon | 15.02 (12.1, 17.94) | 15.45 (12.53, 18.37) | −0.43 (−2.91, 2.05) | 0.7185 |
| Exercise | 25.71 (23.07, 28.35) | 23.46 (20.82, 26.10) | 2.24 (0.00, 4.47) | 0.0497 |
| Energy Expenditure (kcals) | ||||
| 24 h | 2438.5 (2254.16, 2622.83) | 2485.4 (2301.06, 2669.75) | −46.91 (−104.86, 11.05) | 0.1055 |
| Evening | 483.38 (449.84, 516.91) | 493.29 (459.75, 526.82) | −9.91 (−27.96, 8.15) | 0.2616 |
| Nighttime | 155.71 (143.59, 167.83) | 159.63 (147.51, 171.75) | −3.92 (−10.72, 2.88) | 0.2384 |
| Morning | 439.99 (406.11, 473.88) | 451.67 (417.79, 485.56) | −11.68 (−30.32, 6.96) | 0.2028 |
| Afternoon | 224.94 (203.72, 246.15) | 230.4 (209.18, 251.61) | −5.46 (−17.94, 7.02) | 0.3674 |
| Exercise | 196.37 (175.68, 217.06) | 197.36 (176.67, 218.05) | −1.00 (−7.98, 5.99) | 0.7667 |
| Gelatin | Blackberry | |||
|---|---|---|---|---|
| LSmean (95% CI) | LSmean (95% CI) | Difference (95% CI) | p3 | |
| Glucose iAUC (mg·min/dL) 1 | 8.31 (8.05, 8.57) | 8.16 (7.90, 8.41) | 0.15 (−0.04, 0.35) | 0.1151 |
| Insulin iAUC (µU·min/mL) 1 | 9.02 (8.77, 9.26) | 8.78 (8.53, 9.02) | 0.24 (0.13, 0.35) | 0.0002 |
| HOMA-IR 1 | 0.57 (0.34, 0.80) | 0.47 (0.24, 0.70) | 0.10 (0.01, 0.19) | 0.0318 |
| HOMA-β | 91.24 (72.22, 110.26) | 80.32 (61.30, 99.34) | 10.93 (2.11, 19.75) | 0.0175 |
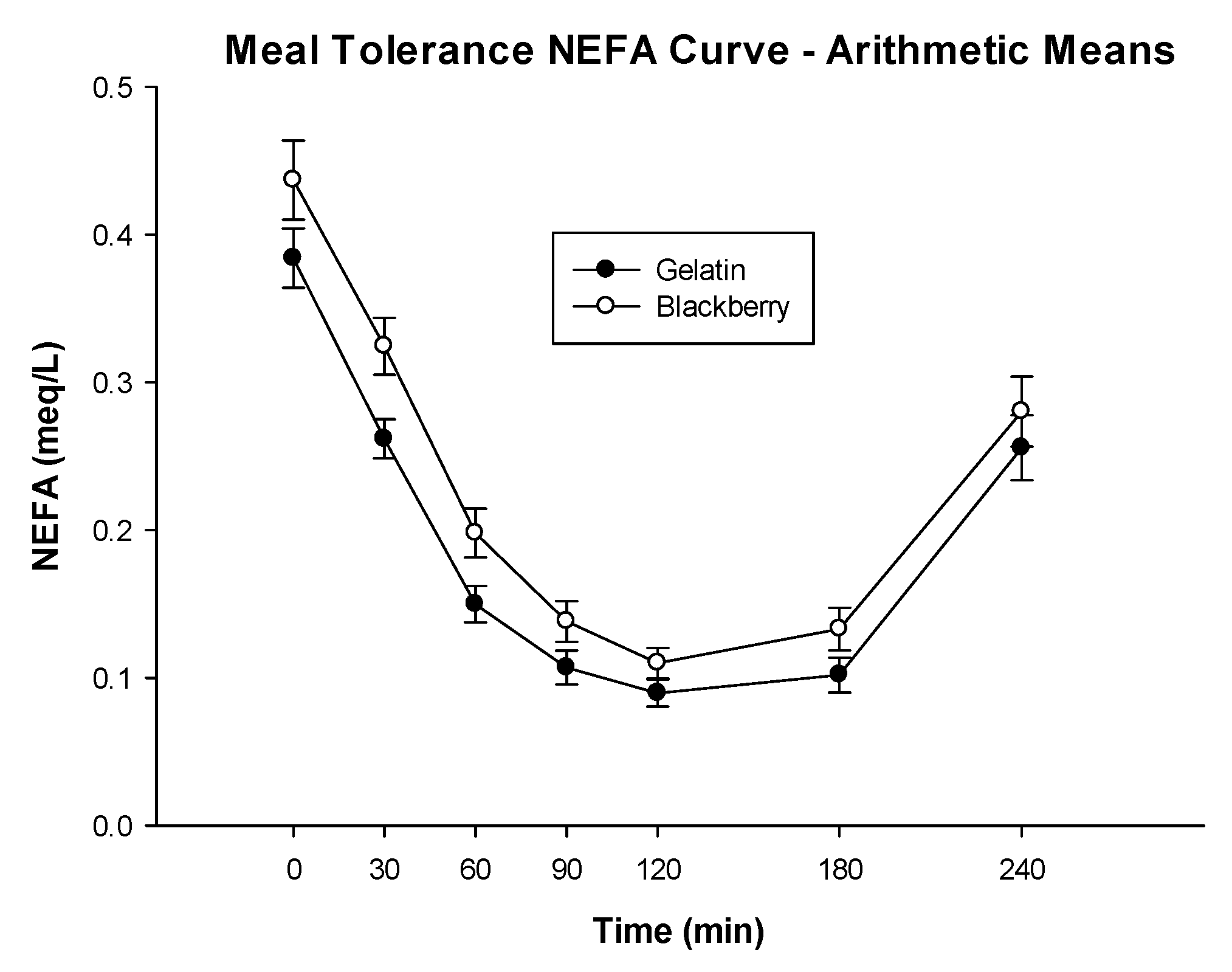
| NEFA AUC (meq·min/L) | 39.13 (34.09, 44.17) | 47.67 (42.63, 52.71) | −8.54 (−13.15, −3.92) | 0.0009 |
| Fasting Triglycerides(mg/dL) 2 | 0.0107 (0.009, 0.0123) | 0.0111 (0.0094, 0.0127) | −0.0004 (−0.0011, 0.0003) | 0.2412 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Solverson, P.M.; Rumpler, W.V.; Leger, J.L.; Redan, B.W.; Ferruzzi, M.G.; Baer, D.J.; Castonguay, T.W.; Novotny, J.A. Blackberry Feeding Increases Fat Oxidation and Improves Insulin Sensitivity in Overweight and Obese Males. Nutrients 2018, 10, 1048. https://doi.org/10.3390/nu10081048
Solverson PM, Rumpler WV, Leger JL, Redan BW, Ferruzzi MG, Baer DJ, Castonguay TW, Novotny JA. Blackberry Feeding Increases Fat Oxidation and Improves Insulin Sensitivity in Overweight and Obese Males. Nutrients. 2018; 10(8):1048. https://doi.org/10.3390/nu10081048
Chicago/Turabian StyleSolverson, Patrick M., William V. Rumpler, Jayme L. Leger, Benjamin W. Redan, Mario G. Ferruzzi, David J. Baer, Thomas W. Castonguay, and Janet A. Novotny. 2018. "Blackberry Feeding Increases Fat Oxidation and Improves Insulin Sensitivity in Overweight and Obese Males" Nutrients 10, no. 8: 1048. https://doi.org/10.3390/nu10081048
APA StyleSolverson, P. M., Rumpler, W. V., Leger, J. L., Redan, B. W., Ferruzzi, M. G., Baer, D. J., Castonguay, T. W., & Novotny, J. A. (2018). Blackberry Feeding Increases Fat Oxidation and Improves Insulin Sensitivity in Overweight and Obese Males. Nutrients, 10(8), 1048. https://doi.org/10.3390/nu10081048





