Effects of Magnesium Supplementation on Unipolar Depression: A Placebo-Controlled Study and Review of the Importance of Dosing and Magnesium Status in the Therapeutic Response
Abstract
1. Introduction
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- World Health Organization. Available online: http://www.who.int/en/news-room/fact-sheets/detail/depression (accessed on 15 May 2018).
- Deutschenbaur, L.; Beck, J.; Kiyhankhadiv, A.; Mühlhauser, M.; Borgwardt, S.; Walter, M.; Hasler, G.; Sollberger, D.; Lang, U.E. Role of calcium, glutamate and NMDA in major depression and therapeutic application. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2016, 64, 325–333. [Google Scholar] [CrossRef] [PubMed]
- Poleszak, E.; Wlaź, P.; Socała, K.; Wrobel, A.; Szewczyk, B.; Kasperek, R.; Nowak, G. Interaction of glycine/NMDA receptor ligands and antidepressant drugs in the forced swim test. Pharmacol. Rep. 2010, 62, 58. [Google Scholar] [CrossRef]
- Nowak, G.; Papp, M.; Paul, I.A. The NMDA receptor complex and the action of antidepressant drugs in the CMS model of depression. Eur. J. Pharm. Sci. 1996, 4, S53. [Google Scholar] [CrossRef]
- Wlaź, P.; Kasperek, R.; Wlaź, A.; Szumiło, M.; Wróbel, A.; Nowak, G.; Poleszak, E. NMDA and AMPA receptors are involved in the antidepressant-like activity of tianeptine in the forced swim test in mice. Pharmacol. Rep. 2011, 63, 1526–1532. [Google Scholar] [PubMed]
- Küçükibrahimoğlu, E.; Saygin, M.Z.; Calişkan, M.; Kaplan, O.K.; Unsal, C.; Gören, M.Z. The change in plasma GABA, glutamine and glutamate levels in fluoxetine- or S-citalopram-treated female patients with major depression. Eur. J. Clin. Pharmacol. 2009, 65, 571–577. [Google Scholar] [CrossRef] [PubMed]
- Frye, M.A.; Tsai, G.E.; Huggins, T.; Coyle, J.T.; Post, R.M. Low Cerebrospinal Fluid Glutamate and Glycine in Refractory Affective Disorder. Biol. Psychiatry 2007, 61, 162–166. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, K.; Sawa, A.; Iyo, M. Increased Levels of Glutamate in Brains from Patients with Mood Disorders. Biol. Psychiatry 2007, 62, 1310–1316. [Google Scholar] [CrossRef] [PubMed]
- Machado-Vieira, R.; Henter, I.D.; Zarate, C.A., Jr. New targets for rapid antidepressant action. Prog. Neurobiol. 2017, 152, 21–37. [Google Scholar] [CrossRef] [PubMed]
- Sanacora, G.; Wilkinson, S.; Schalkwyk, G.V. 201. Measuring Dissociative Effects of NMDA Receptor Antagonists in the Treatment of Depression. Biol. Psychiatry 2017, 81, S83. [Google Scholar] [CrossRef]
- Serefko, A.; Szopa, A.; Poleszak, E. Magnesium and depression. Magnes. Res. 2016, 29, 112–119. [Google Scholar] [CrossRef]
- Poleszak, E.; Wlaz, P.; Kedzierska, E.; Nieoczym, D.; Wrobel, A.; Fidecka, S.; Pilc, A.; Nowak, G. NMDA/glutamate mechanism of antidepressant-like action of magnesium in forced swim test in mice. Pharmacol. Biochem. Behav. 2007, 88, 158–164. [Google Scholar] [CrossRef] [PubMed]
- Murck, H. Ketamine, magnesium and major depression—From pharmacology to pathophysiology and back. J. Psychiatr. Res. 2013, 47, 955–965. [Google Scholar] [CrossRef] [PubMed]
- Gould, T.D.; Manji, H.K. Glycogen synthase kinase-3: A putative molecular target for lithium mimetic drugs. Neuropsychopharmacol. Off. Publ. Am. Coll. Neuropsychopharmacol. 2005, 30, 1223–1237. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, C.C.; Lobato, K.R.; Binfare, R.W.; Ferreira, P.K.; Rosa, A.O.; Santos, A.R.; Rodrigues, A.L. Evidence for the involvement of the monoaminergic system in the antidepressant-like effect of magnesium. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2009, 33, 235–242. [Google Scholar] [CrossRef] [PubMed]
- King, D.E.; Mainous, A.G., 3rd; Geesey, M.E.; Ellis, T. Magnesium intake and serum C-reactive protein levels in children. Magnes. Res. 2007, 20, 32–36. [Google Scholar] [PubMed]
- Decollogne, S.; Tomas, A.; Lecerf, C.; Adamowicz, E.; Seman, M. NMDA receptor complex blockade by oral administration of magnesium: Comparison with MK-801. Pharmacol. Biochem. Behav. 1997, 58, 261–268. [Google Scholar] [CrossRef]
- Poleszak, E.; Szewczyk, B.; Kedzierska, E.; Wlaz, P.; Pilc, A.; Nowak, G. Antidepressant- and anxiolytic-like activity of magnesium in mice. Pharmacol. Biochem. Behav. 2004, 78, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Pochwat, B.; Szewczyk, B.; Sowa-Kucma, M.; Siwek, A.; Doboszewska, U.; Piekoszewski, W.; Gruca, P.; Papp, M.; Nowak, G. Antidepressant-like activity of magnesium in the chronic mild stress model in rats: Alterations in the NMDA receptor subunits. Int. J. Neuropsychopharmacol. 2014, 17, 393–405. [Google Scholar] [CrossRef] [PubMed]
- Pochwat, B.; Sowa-Kucma, M.; Kotarska, K.; Misztak, P.; Nowak, G.; Szewczyk, B. Antidepressant-like activity of magnesium in the olfactory bulbectomy model is associated with the AMPA/BDNF pathway. Psychopharmacology 2015, 232, 355–367. [Google Scholar] [CrossRef] [PubMed]
- Poleszak, E.; Wlaz, P.; Szewczyk, B.; Kedzierska, E.; Wyska, E.; Librowski, T.; Szymura-Oleksiak, J.; Fidecka, S.; Pilc, A.; Nowak, G. Enhancement of antidepressant-like activity by joint administration of imipramine and magnesium in the forced swim test: Behavioral and pharmacokinetic studies in mice. Pharmacol. Biochem. Behav. 2005, 81, 524–529. [Google Scholar] [CrossRef] [PubMed]
- Serefko, A.; Szopa, A.; Wlaź, P.; Nowak, G.; Radziwoń-Zaleska, M.; Skalski, M.; Poleszak, E. Magnesium in depression. Pharmacol. Rep. 2013, 65, 547–554. [Google Scholar] [CrossRef]
- Singewald, N.; Sinner, C.; Hetzenauer, A.; Sartori, S.B.; Murck, H. Magnesium-deficient diet alters depression- and anxiety-related behavior in mice—Influence of desipramine and Hypericum perforatum extract. Neuropharmacology 2004, 47, 1189–1197. [Google Scholar] [CrossRef] [PubMed]
- Jacka, F.N.; Overland, S.; Stewart, R.; Tell, G.S.; Bjelland, I.; Mykletun, A. Association between magnesium intake and depression and anxiety in community-dwelling adults: The Hordaland Health Study. Aust. N. Z. J. Psychiatry 2009, 43, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.H.; Lu, Y.F.; Cheng, F.C.; Lee, J.N.; Tsai, L.C. Correlation of magnesium intake with metabolic parameters, depression and physical activity in elderly type 2 diabetes patients: A cross-sectional study. Nutr. J. 2012, 11, 41. [Google Scholar] [CrossRef] [PubMed]
- Tarleton, E.K.; Littenberg, B. Magnesium intake and depression in adults. J. Am. Board Fam. Med. 2015, 28, 249–256. [Google Scholar] [CrossRef] [PubMed]
- Yary, T.; Lehto, S.M.; Tolmunen, T.; Tuomainen, T.P.; Kauhanen, J.; Voutilainen, S.; Ruusunen, A. Dietary magnesium intake and the incidence of depression: A 20-year follow-up study. J. Affect Disord. 2016, 193, 94–98. [Google Scholar] [CrossRef] [PubMed]
- Linder, J.; Brismar, K.; Beck-Friis, J.; Saaf, J.; Wetterberg, L. Calcium and magnesium concentrations in affective disorder: Difference between plasma and serum in relation to symptoms. Acta Psychiatr. Scand. 1989, 80, 527–537. [Google Scholar] [CrossRef] [PubMed]
- Barragan-Rodriguez, L.; Rodriguez-Moran, M.; Guerrero-Romero, F. Depressive symptoms and hypomagnesemia in older diabetic subjects. Arch. Med. Res. 2007, 38, 752–756. [Google Scholar] [CrossRef] [PubMed]
- Derom, M.-L.; Sayón-Orea, C.; Martínez-Ortega, J.M.; Martínez-González, M.A. Magnesium and depression: A systematic review. Nutr. Neurosci. 2013, 16, 191–206. [Google Scholar] [CrossRef] [PubMed]
- Camardese, G.; De Risio, L.; Pizi, G.; Mattioli, B.; Buccelletti, F.; Serrani, R.; Leone, B.; Sgambato, A.; Bria, P.; Janiri, L. Plasma magnesium levels and treatment outcome in depressed patients. Nutr. Neurosci. 2012, 15, 78–84. [Google Scholar] [CrossRef] [PubMed]
- Bauer, M.; Bschor, T.; Pfennig, A.; Whybrow, P.C.; Angst, J.; Versiani, M.; Möller, H.-J. World Federation of Societies of Biological Psychiatry (WFSBP) Guidelines for Biological Treatment of Unipolar Depressive Disorders in Primary Care. World J. Biol. Psychiatry Off. J. World Fed. Soc. Biol. Psychiatry 2007, 8, 67–104. [Google Scholar] [CrossRef] [PubMed]
- Trevino, K.; McClintock, S.M.; McDonald Fischer, N.; Vora, A.; Husain, M.M. Defining treatment-resistant depression: A comprehensive review of the literature. Ann. Clin. Psychiatry Off. J. Am. Acad. Clin. Psychiatr. 2014, 26, 222–232. [Google Scholar]
- Radziwoń-Zaleska, M.; Ryszewska-Pokraśniewicz, B.; Skalski, M.; Skrzeszewski, J.; Drozdowicz, E.; Nowak, G.; Pilc, A.; Bałkowiec-Iskra, E. Therapeutic drug monitoring of depression—Amplification by magnesium ions. Pharmacol. Rep. 2015, 67, 36. [Google Scholar] [CrossRef]
- Eby, G.A.; Eby, K.L. Rapid recovery from major depression using magnesium treatment. Med. Hypotheses 2006, 67, 362–370. [Google Scholar] [CrossRef] [PubMed]
- Derom, M.L.; Martinez-Gonzalez, M.A.; Sayon-Orea Mdel, C.; Bes-Rastrollo, M.; Beunza, J.J.; Sanchez-Villegas, A. Magnesium intake is not related to depression risk in Spanish university graduates. J. Nutr. 2012, 142, 1053–1059. [Google Scholar] [CrossRef] [PubMed]
- Radziwoń-Zaleska, M.; Matsumoto, H.; Skalski, M.; Androsiuk, W.; Dziklińska, A.; Grobel, I.; Kunicki, P. Monitored therapy of depression. Farmakoterapia w Psychiatrii i Neurologii 1998, 3, 5–12. [Google Scholar]
- Hamilton, M. A rating scale for depression. J. Neurol. Neurosurg. Psychiatry 1960, 23, 56–62. [Google Scholar] [CrossRef] [PubMed]
- El-Yazigi, A.; Raines, D.A. Concurrent liquid chromatographic measurement of fluoxetine, amitryptyline, imipramine, and their active metabolites norfluoxetine, nortyptyline, and desipramine in plasma. Ther. Drug. Monit. 1993, 15, 305–309. [Google Scholar] [CrossRef] [PubMed]
- Aymard, G.; Livi, P.; Pham, Y.T.; Diquet, B. Sensitive and rapid method for the simultaneous quantification of five antidepressants with their respective metabolites in plasma using high-performance liquid chromatography with diode-array detection. J. Chromatogr. B Biomed. Sci. Appl. 1997, 700, 183–189. [Google Scholar] [CrossRef]
- Meineke, I.; Schreeb, K.; Kress, I.; Gundert-Remy, U. Routine measurement of fluoxetine and norfluoxetine by high-performance liquid chromatography with ultraviolet detection in patients under concomitant treatment with tricyclic antidepressants. Ther. Drug Monit. 1998, 20, 14–19. [Google Scholar] [CrossRef] [PubMed]
- Komorowska, E. Walidacja i Zastosowanie Metody Oznaczania Fluoksetyny Techniką HPLC; Akademia Medyczna w Warszawie, Wydział Farmaceutyczny: Warszawa, Poland, 2002. [Google Scholar]
- Baldessarini, R.J. Drugs and treatment of psychiatric disorders: Depression and mania. W. In Goodman & Gilman’s The Pharmacological Basis of Therapeutic; McGraw-Hills Co.: New York, NY, USA, 1996; pp. 431–461. [Google Scholar]
- Hulanicki, A.; Lewenstam, A.; Maj-Żurawska, M. Magnesium; Clinical Significance and Analytical Determination; Reviews on Analytical Chemistry, Euroanalysis VIII; Littlejohn, D., Thorburn Burns, D., Eds.; RSC: Cambridge, CA, USA, 1994; pp. 317–325. [Google Scholar]
- Szelenberger, W. Farmakoelektroencefalografia: Aktualny stan wiedzy i perspektywy. Psychiatr. Polska 1990, 24, 52–57. [Google Scholar]
- Skalski, M.; Szelenberger, W.; Radziwoń-Zaleska, M.; Matsumoto, H. Zastosowanie metody farmakoelektroencefalografii (farmako—EEG) w monitorowaniu terapii depresji. Farmakoterapia w Psychiatrii i Neurologii 1995, 4, 60–70. [Google Scholar]
- Kroenke, K.; Spitzer, R.L.; Williams, J.B. The PHQ-9: Validity of a brief depression severity measure. J. Gen. Intern. Med. 2001, 16, 606–613. [Google Scholar] [CrossRef] [PubMed]
- Tarleton, E.K.; Littenberg, B.; MacLean, C.D.; Kennedy, A.G.; Daley, C. Role of magnesium supplementation in the treatment of depression: A randomized clinical trial. PLoS ONE 2017, 12, e0180067. [Google Scholar] [CrossRef] [PubMed]
- Barragan-Rodriguez, L.; Rodriguez-Moran, M.; Guerrero-Romero, F. Efficacy and safety of oral magnesium supplementation in the treatment of depression in the elderly with type 2 diabetes: A randomized, equivalent trial. Magnes. Res. 2008, 21, 218–223. [Google Scholar] [PubMed]
- Mehdi, S.M.; Atlas, S.E.; Qadir, S.; Musselman, D.; Goldberg, S.; Woolger, J.M.; Corredor, R.; Abbas, M.H.; Arosemena, L.; Caccamo, S.; et al. Double-blind, randomized crossover study of intravenous infusion of magnesium sulfate versus 5% dextrose on depressive symptoms in adults with treatment-resistant depression. Psychiatry Clin. Neurosci. 2017, 71, 204–211. [Google Scholar] [CrossRef] [PubMed]
- Fard, F.E.; Mirghafourvand, M.; Mohammad-Alizadeh Charandabi, S.; Farshbaf-Khalili, A.; Javadzadeh, Y.; Asgharian, H. Effects of zinc and magnesium supplements on postpartum depression and anxiety: A randomized controlled clinical trial. Women Health 2017, 57, 1115–1128. [Google Scholar] [CrossRef] [PubMed]
- Martínez-González, M.Á.; Sánchez-Villegas, A. Magnesium intake and depression: The SUN cohort. Magnes. Res. 2016, 29, 102–111. [Google Scholar] [PubMed]
- Nechifor, M. Magnesium in major depression. Magnes. Res. 2009, 22, 163s–166s. [Google Scholar] [PubMed]
- Poleszak, E. Modulation of antidepressant-like activity of magnesium by serotonergic system. J. Neural Transm. 2007, 114, 1129–1134. [Google Scholar] [CrossRef] [PubMed]
- George, M.S.; Rosenstein, D.; Rubinow, D.R.; Kling, M.A.; Post, R.M. CSF magnesium in affective disorder: Lack of correlation with clinical course of treatment. Psychiatry Res. 1994, 51, 139–146. [Google Scholar] [CrossRef]
- Herzberg, L.; Bold, A.M. Sex difference in mean serum-magnesium levels in depression. Lancet 1972, 1, 1128–1129. [Google Scholar] [CrossRef]
- Stanton, M.F.; Lowenstein, F.W. Serum magnesium in women during pregnancy, while taking contraceptives, and after menopause. J. Am. Coll. Nutr. 1987, 6, 313–319. [Google Scholar] [CrossRef] [PubMed]
- Brown, R.A.; Abrantes, A.M.; Strong, D.R.; Niaura, R.; Kahler, C.W.; Miller, I.W.; Price, L.H. Efficacy of sequential use of fluoxetine for smoking cessation in elevated depressive symptom smokers. Nicotine Tob. Res. Off. J. Soc. Res. Nicotine Tob. 2014, 16, 197–207. [Google Scholar] [CrossRef] [PubMed]
- Kenney, B.A.; Holahan, C.J.; North, R.J.; Holahan, C.K. Depressive symptoms and cigarette smoking in American workers. Am. J. Health Promot. AJHP 2006, 20, 179–182. [Google Scholar] [CrossRef] [PubMed]
- Vázquez-Palacios, G.; Bonilla-Jaime, H.; Velázquez-Moctezuma, J. Antidepressant effects of nicotine and fluoxetine in an animal model of depression induced by neonatal treatment with clomipramine. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2005, 29, 39–46. [Google Scholar] [CrossRef] [PubMed]
- Glassman, A.H.; Covey, L.S.; Stetner, F.; Rivelli, S. Smoking cessation and the course of major depression: A follow-up study. Lancet 2001, 357, 1929–1932. [Google Scholar] [CrossRef]
- Salín-Pascual, R.J.; Galicia-Polo, L.; Drucker-Colín, R.; de la Fuente, J.R.; Drucker-Colín, R.; Salín-Pascual, R.J. Effects of transderman nicotine on mood and sleep in nonsmoking major depresssed patients. Psychopharmacology 1995, 121, 476–479. [Google Scholar] [CrossRef] [PubMed]
- Cosci, F.; Nardi, A.E.; Griez, E.J. Nicotine effects on human affective functions: A systematic review of the literature on a controversial issue. CNS Neurol. Disord. Drug Targets 2014, 13, 981–991. [Google Scholar] [CrossRef] [PubMed]
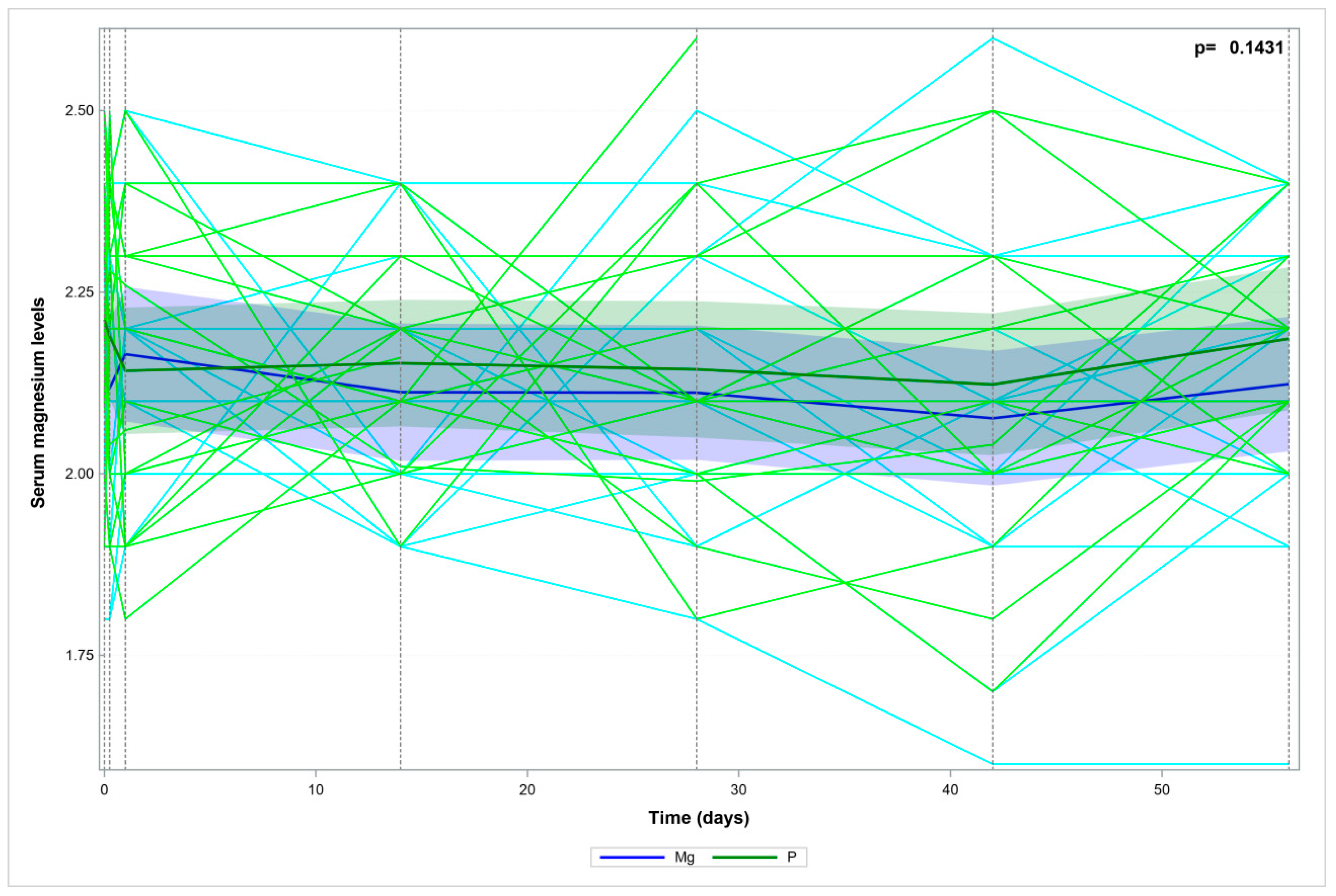
| Group I: Magnesium | Group II: Placebo | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Variable | N | Mean | SD | Med | Min | Max | N | Mean | SD | Med | Min | Max | p-Value |
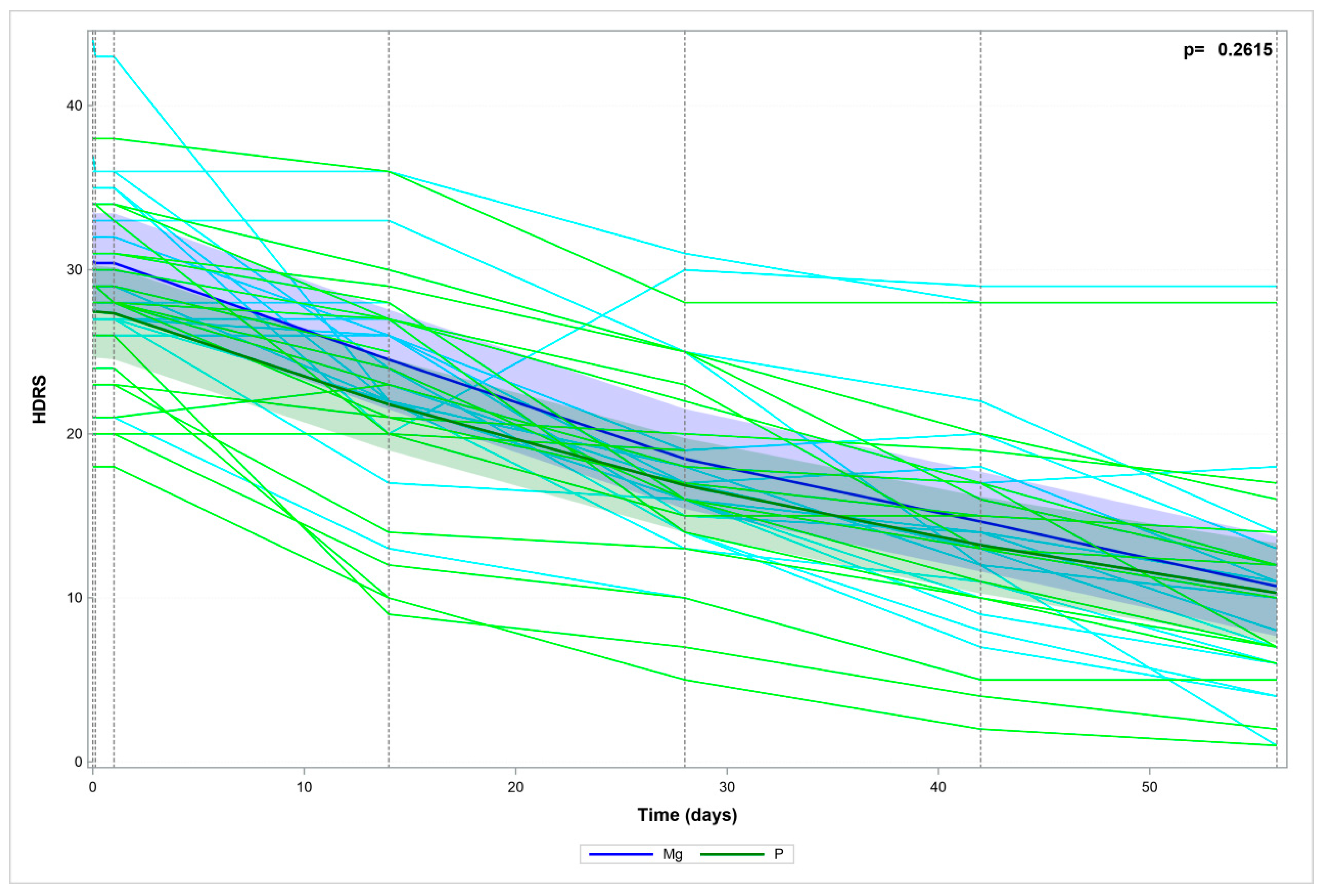
| HDRS_0 | 17 | 30.5 | 6 | 29 | 21 | 44 | 20 | 27.5 | 5.5 | 28 | 18 | 38 | 0.1120 |
| HDRS_6H | 17 | 30.4 | 5.8 | 29 | 21 | 43 | 20 | 27.5 | 5.5 | 28 | 18 | 38 | 0.1197 |
| HDRS_24H | 17 | 30.4 | 5.8 | 29 | 21 | 43 | 20 | 27.4 | 5.4 | 28 | 18 | 38 | 0.1059 |
| HDRS_2W | 17 | 24.5 | 5.6 | 24 | 13 | 36 | 20 | 21.8 | 7.5 | 23 | 9 | 36 | 0.2237 |
| HDRS_4W | 17 | 18.5 | 5.9 | 17 | 10 | 31 | 17 | 17.2 | 6.4 | 17 | 5 | 28 | 0.5605 |
| HDRS_6W | 17 | 14.6 | 6.9 | 13 | 5 | 29 | 16 | 13.4 | 6.6 | 14 | 2 | 28 | 0.6100 |
| HDRS_8W | 17 | 10.7 | 7.9 | 8 | 1 | 29 | 15 | 10.4 | 6.8 | 10 | 1 | 28 | 0.9080 |
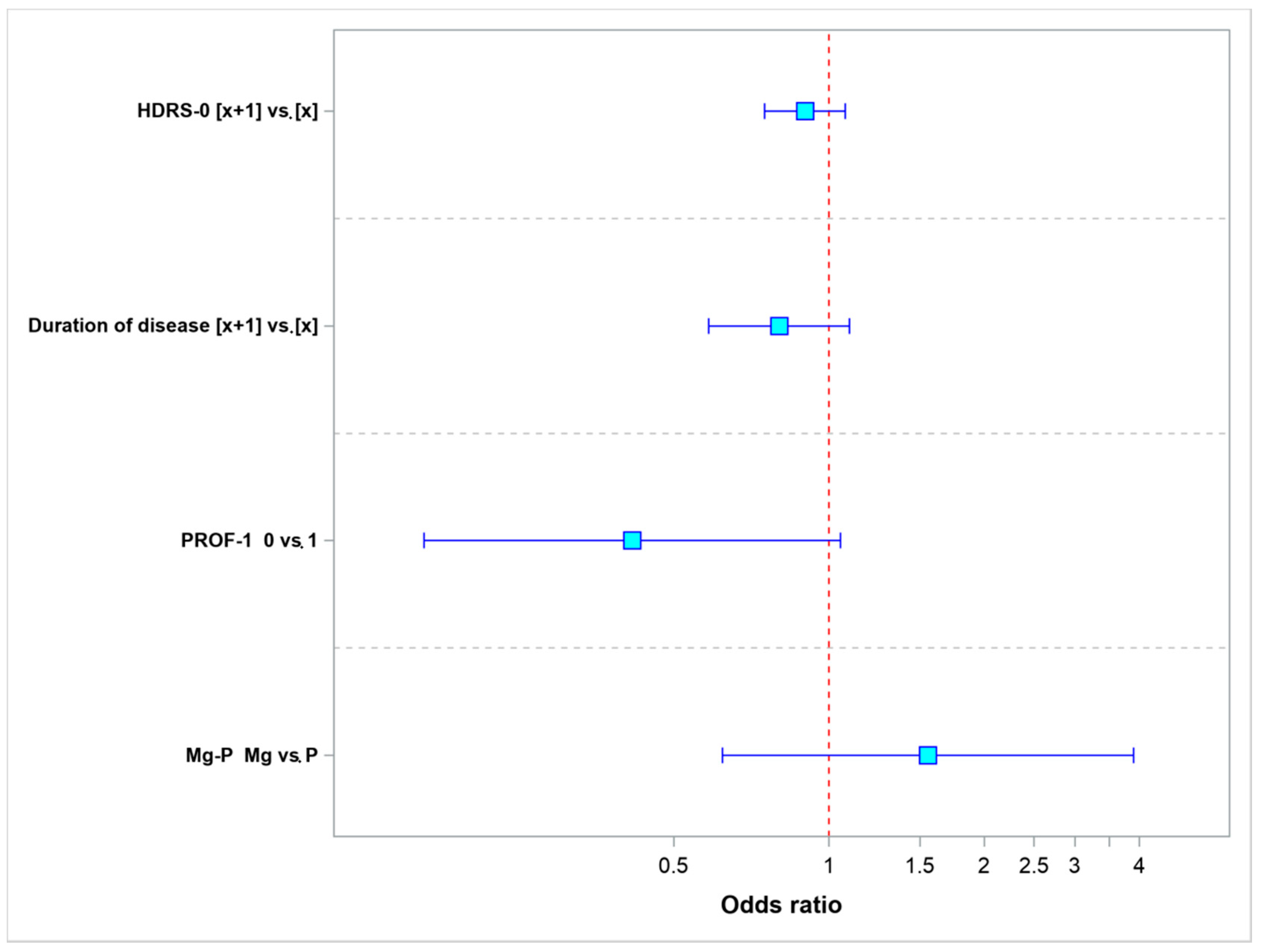
| Parameter | Values | Odds Ratio | 95% LCL | 95% UCL | p-Value |
|---|---|---|---|---|---|
| HDRS-0 | [x + 1] vs. [x] | 0.8982 | 0.7503 | 1.0752 | 0.2422 |
| Duration of disease | [x + 1] vs. [x] | 0.8001 | 0.5842 | 1.0956 | 0.1643 |
| PROF-1 | 0 vs. 1 | 0.4151 | 0.1637 | 1.0527 | 0.0641 |
| Mg-P | Mg vs. P | 1.5545 | 0.6206 | 3.8938 | 0.3464 |
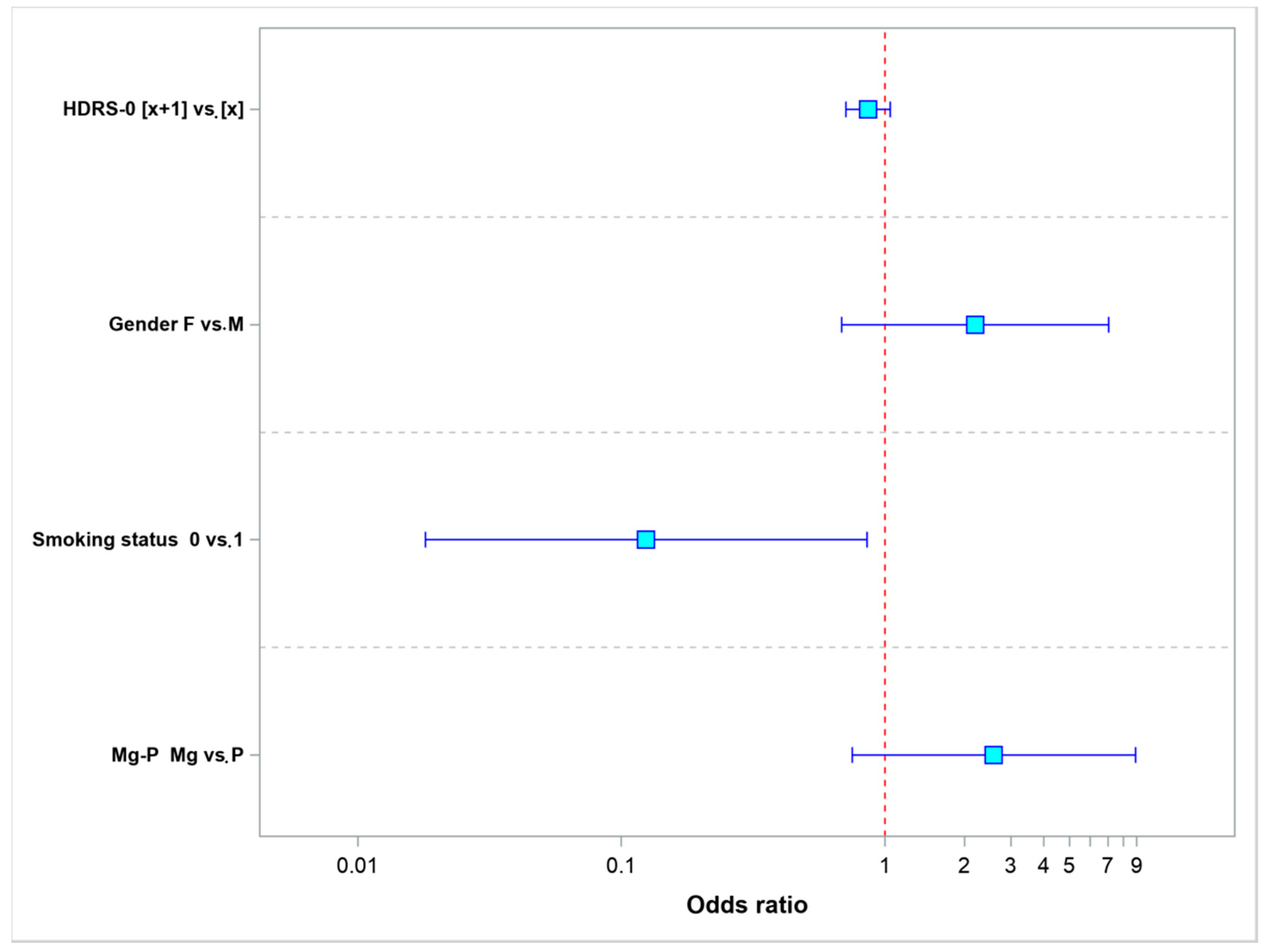
| Parameter | Values | Odds Ratio | 95% LCL | 95% UCL | p-Value |
|---|---|---|---|---|---|
| HDRS-0 | [x + 1] vs. [x] | 0.8614 | 0.7093 | 1.0461 | 0.1321 |
| Gender | F vs. M | 2.1942 | 0.6840 | 7.0392 | 0.1864 |
| Smoking status | 0 vs. 1 | 0.1242 | 0.0181 | 0.8530 | 0.0339 |
| Mg-P | Mg vs. P | 2.5869 | 0.7510 | 8.9111 | 0.1320 |
| Depression Type | Type of Study | N | Dose of Mg+2 mg/day p.o. | Salt | Effect | References |
|---|---|---|---|---|---|---|
| Major depression | Case | 4 | 125–300 | Glycinate taurinate | + | [28] |
| Depression (early, type 2 diabetes, hypomagnesemia) | Randomized Mg vs. IMI| | 23 | 450 | Chloride | + | [42] |
| Depression Gitelman’s syndrome, (hypomagnesemia) | Case | 1 | 300–900 plus intravenous 1200 | Oxide sulfate | + | [46] |
| Major depression (hypomagnesemia) | Placebo | 60 | 300 | Oxide | + | [47] |
| Major depression | Placebo cross-over | 112 | 248 | Chloride | + | [41] |
| Postpartum depression | Placebo | 66 | 87 | Sulfate | − | [44] |
| Major depression | Placebo | 37 | 120 | Aspartate | − | present study |
| Major depression (TRD) | Monotherapy | 12 | Intravenous 1000 | Sulfate | − | [43] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ryszewska-Pokraśniewicz, B.; Mach, A.; Skalski, M.; Januszko, P.; Wawrzyniak, Z.M.; Poleszak, E.; Nowak, G.; Pilc, A.; Radziwoń-Zaleska, M. Effects of Magnesium Supplementation on Unipolar Depression: A Placebo-Controlled Study and Review of the Importance of Dosing and Magnesium Status in the Therapeutic Response. Nutrients 2018, 10, 1014. https://doi.org/10.3390/nu10081014
Ryszewska-Pokraśniewicz B, Mach A, Skalski M, Januszko P, Wawrzyniak ZM, Poleszak E, Nowak G, Pilc A, Radziwoń-Zaleska M. Effects of Magnesium Supplementation on Unipolar Depression: A Placebo-Controlled Study and Review of the Importance of Dosing and Magnesium Status in the Therapeutic Response. Nutrients. 2018; 10(8):1014. https://doi.org/10.3390/nu10081014
Chicago/Turabian StyleRyszewska-Pokraśniewicz, Beata, Anna Mach, Michał Skalski, Piotr Januszko, Zbigniew M. Wawrzyniak, Ewa Poleszak, Gabriel Nowak, Andrzej Pilc, and Maria Radziwoń-Zaleska. 2018. "Effects of Magnesium Supplementation on Unipolar Depression: A Placebo-Controlled Study and Review of the Importance of Dosing and Magnesium Status in the Therapeutic Response" Nutrients 10, no. 8: 1014. https://doi.org/10.3390/nu10081014
APA StyleRyszewska-Pokraśniewicz, B., Mach, A., Skalski, M., Januszko, P., Wawrzyniak, Z. M., Poleszak, E., Nowak, G., Pilc, A., & Radziwoń-Zaleska, M. (2018). Effects of Magnesium Supplementation on Unipolar Depression: A Placebo-Controlled Study and Review of the Importance of Dosing and Magnesium Status in the Therapeutic Response. Nutrients, 10(8), 1014. https://doi.org/10.3390/nu10081014






