Intake of Dietary One-Carbon Metabolism-Related B Vitamins and the Risk of Esophageal Cancer: A Dose-Response Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Search Strategy and Selection Criteria
2.2. Data Extraction and Quality Assessment
2.3. Statistical Analysis
3. Results
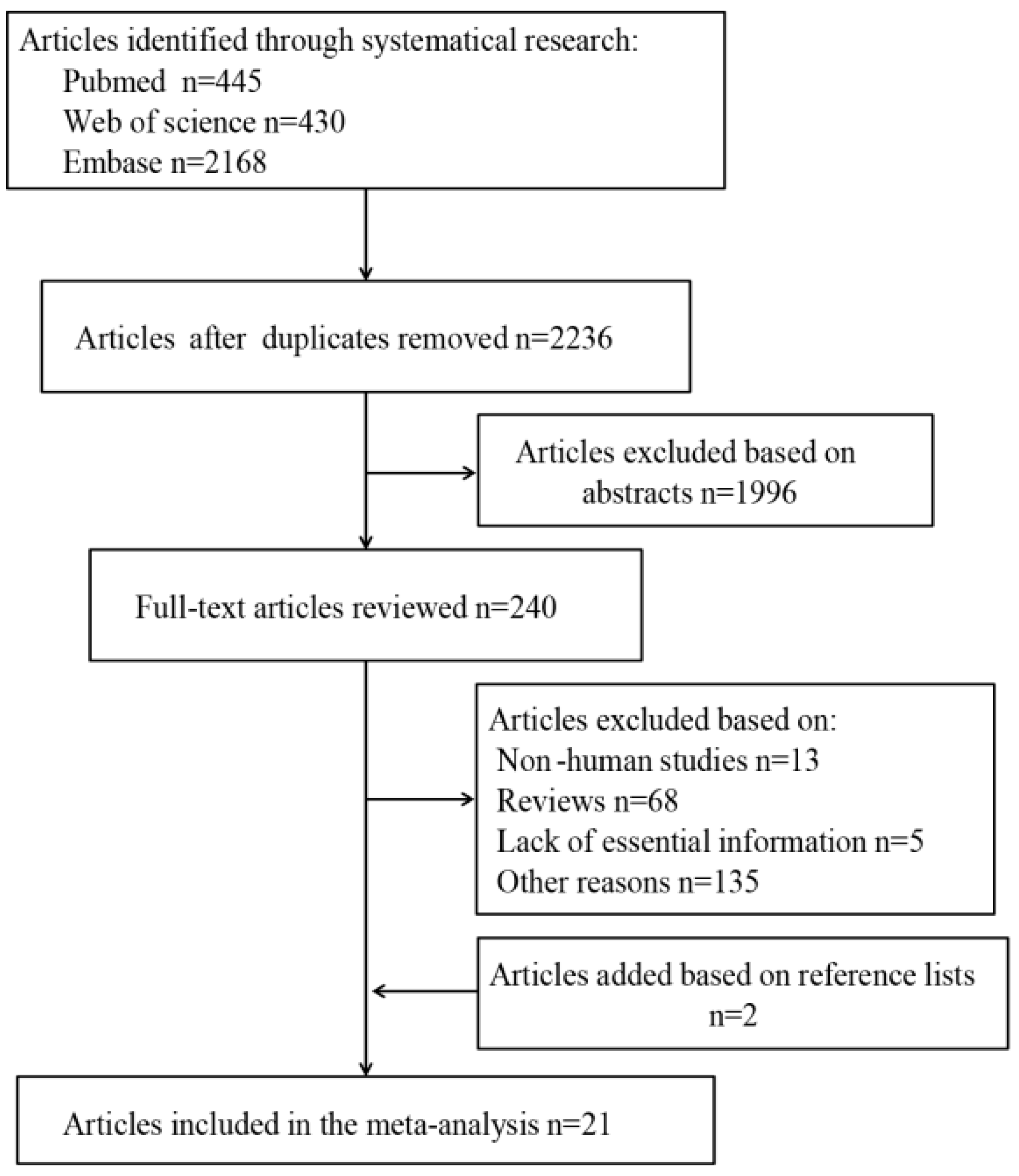
3.1. Search Results, Study Characteristics, and Quality Assessment
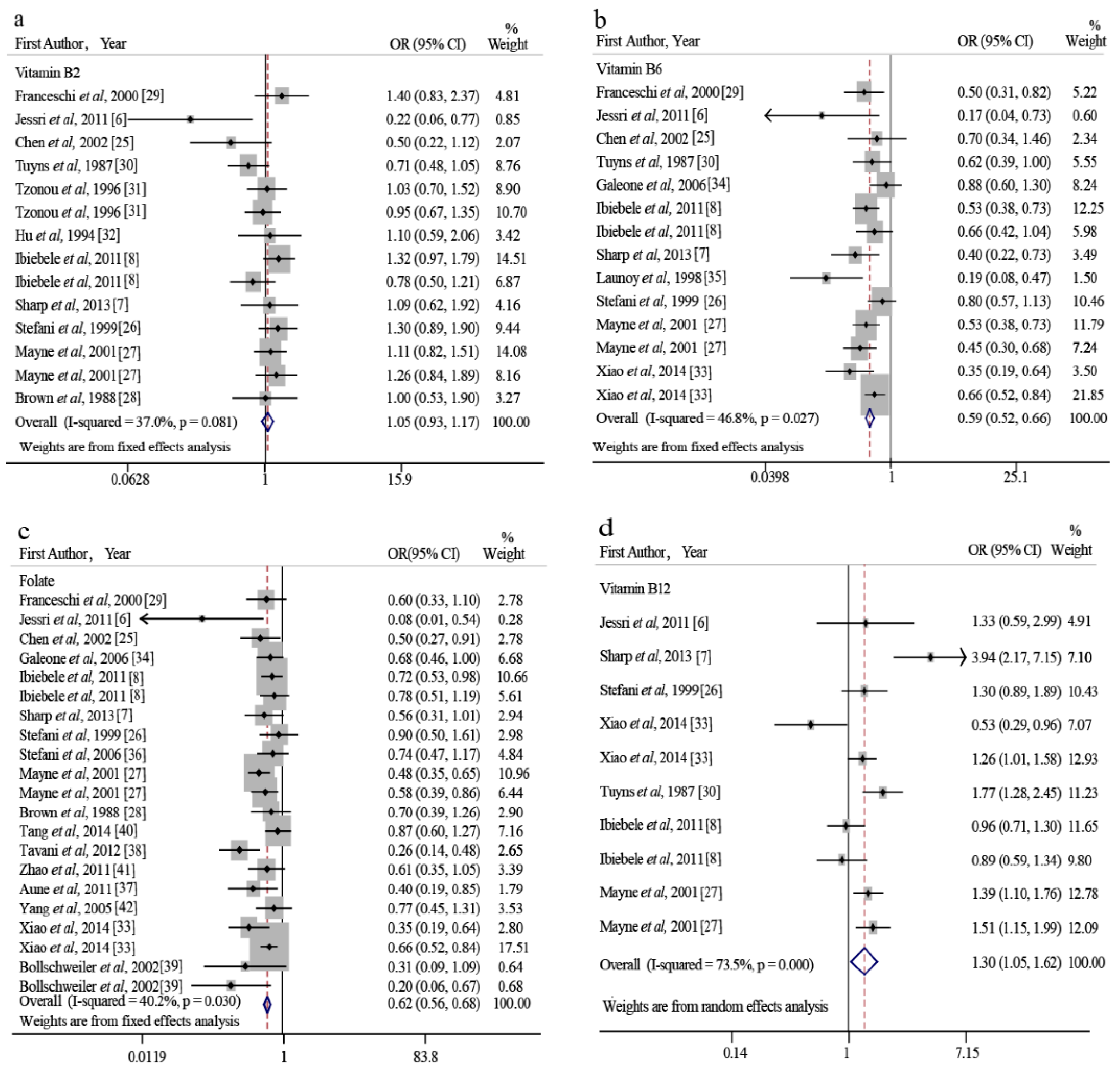
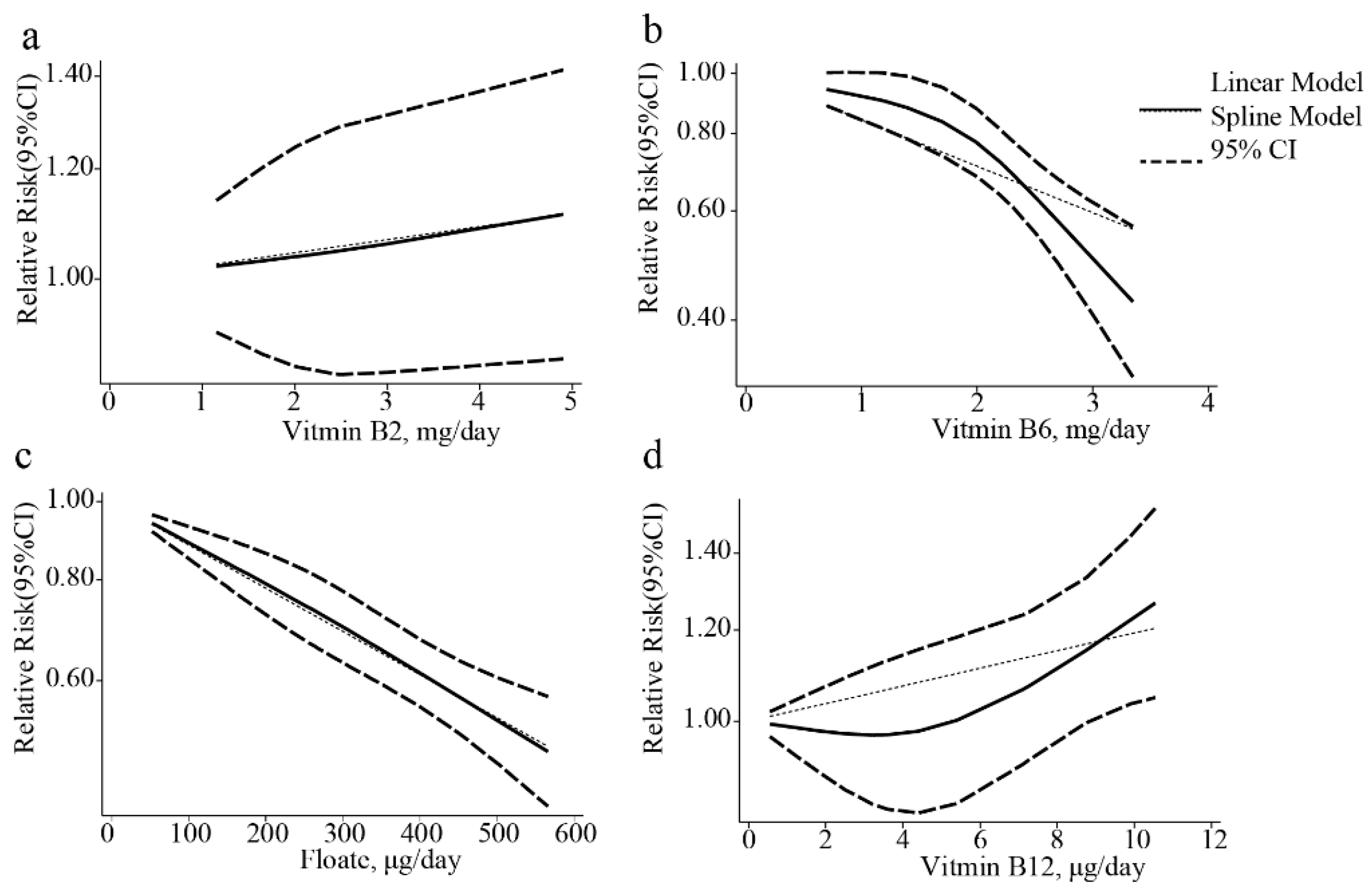
3.2. Dietary Vitamin B2 Intake and EC Risk
3.3. Dietary Vitamin B6 Intake and EC Risk
3.4. Dietary Folate Intake and EC Risk
3.5. Dietary Vitamin B12 Intake and EC Risk
3.6. Heterogeneity and Meta-Regression
3.7. Sensitivity Analysis and Publication Bias
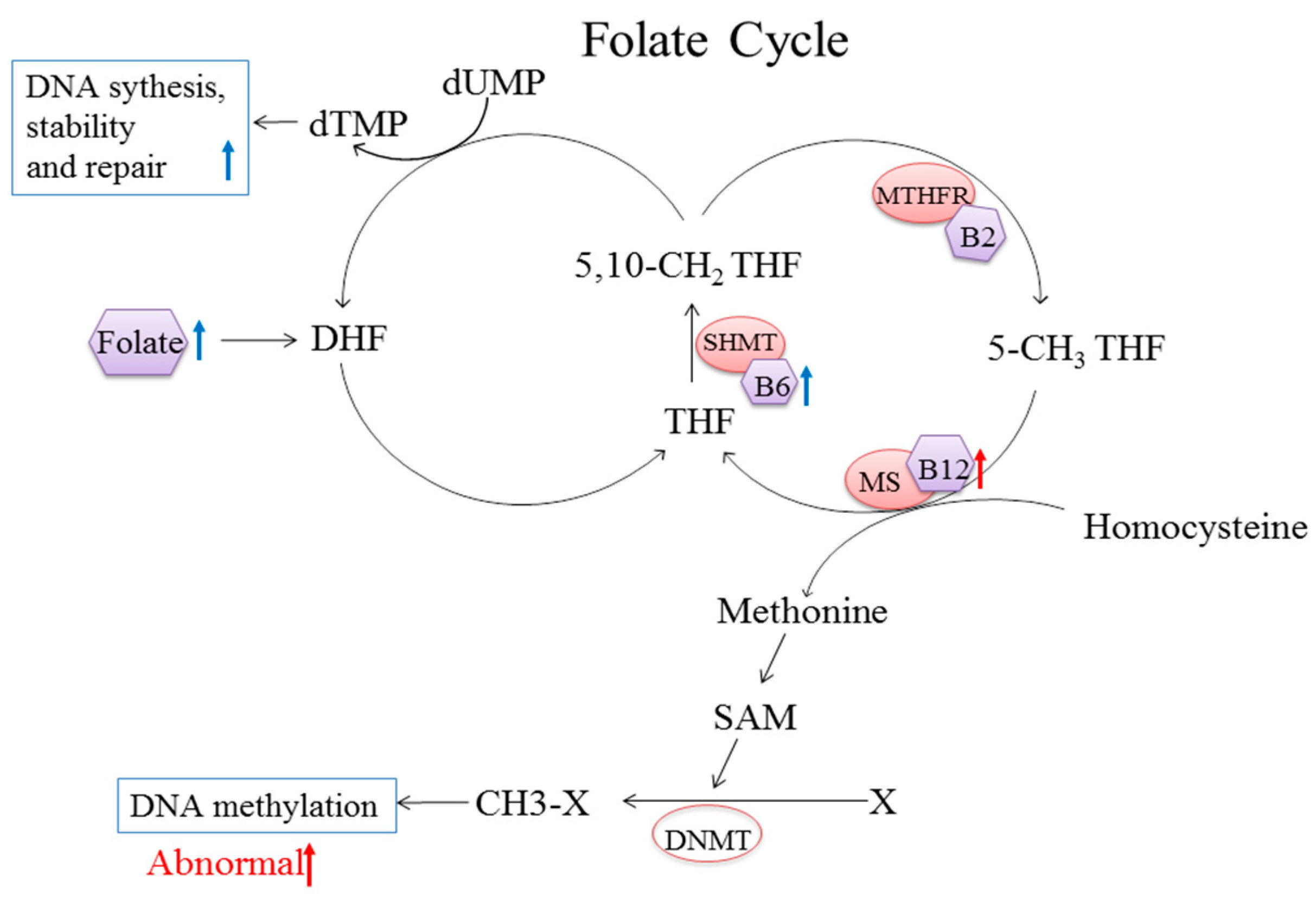
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Global Burden of Disease Cancer Collaboration; Fitzmaurice, C.; Dicker, D.; Pain, A.; Hamavid, H.; Moradi-Lakeh, M.; MacIntyre, M.F.; Allen, C.; Hansen, G.; Woodbrook, R.; et al. The global burden of cancer 2013. JAMA Oncol. 2015, 1, 505–527. [Google Scholar] [CrossRef] [PubMed]
- Palladino-Davis, A.G.; Mendez, B.M.; Fisichella, P.M.; Davis, C.S. Dietary habits and esophageal cancer. Dis. Esophagus 2015, 28, 59–67. [Google Scholar] [CrossRef] [PubMed]
- Keszei, A.P.; Goldbohm, R.A.; Schouten, L.J.; Jakszyn, P.; van den Brandt, P.A. Dietary N-nitroso compounds, endogenous nitrosation, and the risk of esophageal and gastric cancer subtypes in the netherlands cohort study. Am. J. Clin. Nutr. 2013, 97, 135–146. [Google Scholar] [CrossRef] [PubMed]
- Ciappio, E.D.; Mason, J.B.; Crott, J.W. Maternal one-carbon nutrient intake and cancer risk in offspring. Nutr. Rev. 2011, 69, 561–571. [Google Scholar] [CrossRef] [PubMed]
- Cancarini, I.; Krogh, V.; Agnoli, C.; Grioni, S.; Matullo, G.; Pala, V.; Pedraglio, S.; Contiero, P.; Riva, C.; Muti, P.; et al. Micronutrients involved in one-carbon metabolism and risk of breast cancer subtypes. PLoS ONE 2015, 10, e0138318. [Google Scholar] [CrossRef] [PubMed]
- Jessri, M.; Rashidkhani, B.; Hajizadeh, B.; Jessri, M.; Gotay, C. Macronutrients, vitamins and minerals intake and risk of esophageal squamous cell carcinoma: A case-control study in Iran. Nutr. J. 2011, 10, 137. [Google Scholar] [CrossRef] [PubMed]
- Sharp, L.; Carsin, A.E.; Cantwell, M.M.; Anderson, L.A.; Murray, L.J.; Group, F.S. Intakes of dietary folate and other B vitamins are associated with risks of esophageal adenocarcinoma, barrett’s esophagus, and reflux esophagitis. J. Nutr. 2013, 143, 1966–1973. [Google Scholar] [PubMed]
- Ibiebele, T.I.; Hughes, M.C.; Pandeya, N.; Zhao, Z.; Montgomery, G.; Hayward, N.; Green, A.C.; Whiteman, D.C.; Webb, P.M.; Study of Digestive Health; et al. High intake of folate from food sources is associated with reduced risk of esophageal cancer in an Australian population. J. Nutr. 2011, 141, 274–283. [Google Scholar] [PubMed]
- Liu, W.; Zhou, H.; Zhu, Y.Q.; Tie, C.R. Associations between dietary folate intake and risks of esophageal, gastric and pancreatic cancers: An overall and dose-response meta-analysis. Oncotarget 2017, 8, 86828–86842. [Google Scholar] [CrossRef] [PubMed]
- Mocellin, S.; Briarava, M.; Pilati, P. Vitamin B6 and cancer risk: A field synopsis and meta-analysis. J. Natl. Cancer Inst. 2017, 109, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Guo, C.; Hu, H.; Zheng, L.; Ma, J.; Jiang, L.; Zhao, E.; Li, H. Folate intake, serum folate levels and esophageal cancer risk: An overall and dose-response meta-analysis. Oncotarget 2017, 8, 10458–10469. [Google Scholar] [CrossRef] [PubMed]
- Stroup, D.F.; Berlin, J.A.; Morton, S.C.; Olkin, I.; Williamson, G.D.; Rennie, D.; Moher, D.; Becker, B.J.; Sipe, T.A.; Thacker, S.B. Meta-analysis of observational studies in epidemiology: A proposal for reporting. Meta-analysis of observational studies in epidemiology (MOOSE) group. JAMA 2000, 283, 2008–2012. [Google Scholar] [CrossRef] [PubMed]
- Wells, G.A.; Shea, B.; O’Connell, D.; Peterson, J.; Welch, V.; Losos, M.; Tugwell, P. The Newcastle-Ottawa Scale (NOS) for Assessing the Quality of Nonrandomised Studies in Meta-Analyses. Available online: http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp (accessed on 15 March 2018).
- Greenland, S. Quantitative methods in the review of epidemiologic literature. Epidemiol. Rev. 1987, 9, 1–30. [Google Scholar] [CrossRef] [PubMed]
- Fang, X.; Wei, J.; He, X.; An, P.; Wang, H.; Jiang, L.; Shao, D.; Liang, H.; Li, Y.; Wang, F.; et al. Landscape of dietary factors associated with risk of gastric cancer: A systematic review and dose-response meta-analysis of prospective cohort studies. Eur. J. Cancer 2015, 51, 2820–2832. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. BMJ 2003, 327, 557–560. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.; Thompson, S.G. Controlling the risk of spurious findings from meta-regression. Stat. Med. 2004, 23, 1663–1682. [Google Scholar] [CrossRef] [PubMed]
- Greenland, S.; Longnecker, M.P. Methods for trend estimation from summarized dose-response data, with applications to meta-analysis. Am. J. Epidemiol. 1992, 135, 1301–1309. [Google Scholar] [CrossRef] [PubMed]
- Orsini, N.; Bellocco, R.; Greenland, S. Generalized least squares for trend estimation of summarized dose-response data. Stata J. 2006, 6, 40–57. [Google Scholar]
- Bekkering, G.E.; Harris, R.J.; Thomas, S.; Mayer, A.-M.B.; Beynon, R.; Ness, A.R.; Harbord, R.M.; Bain, C.; Smith, G.D.; Sterne, J.A.C. How much of the data published in observational studies of the association between diet and prostate or bladder cancer is usable for meta-analysis? Am. J. Epidemiol. 2008, 167, 1017–1026. [Google Scholar] [CrossRef] [PubMed]
- Hamling, J.; Lee, P.; Weitkunat, R.; Ambuhl, M. Facilitating meta-analyses by deriving relative effect and precision estimates for alternative comparisons from a set of estimates presented by exposure level or disease category. Stat. Med. 2008, 27, 954–970. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.; Kang, S.; Zhang, D. Association of vitamin B6, vitamin B12 and methionine with risk of breast cancer: A dose-response meta-analysis. Br. J. Cancer 2013, 109, 1926–1944. [Google Scholar] [CrossRef] [PubMed]
- Harrell, F.E., Jr.; Lee, K.L.; Pollock, B.G. Regression models in clinical studies: Determining relationships between predictors and response. J. Nat. Cancer Inst. 1988, 80, 1198–1202. [Google Scholar] [CrossRef] [PubMed]
- Egger, M.; Davey Smith, G.; Schneider, M.; Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997, 315, 629–634. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Tucker, K.L.; Graubard, B.I.; Heineman, E.F.; Markin, R.S.; Potischman, N.A.; Russell, R.M.; Weisenburger, D.D.; Ward, M.H. Nutrient intakes and adenocarcinoma of the esophagus and distal stomach. Nutr. Cancer 2002, 42, 33–40. [Google Scholar] [CrossRef] [PubMed]
- De Stefani, E.; Ronco, A.; Mendilaharsu, M.; Deneo-Pellegrini, H. Diet and risk of cancer of the upper aerodigestive tract—II. Nutrients. Oral. Oncol. 1999, 35, 22–26. [Google Scholar] [CrossRef]
- Mayne, S.T.; Risch, H.A.; Dubrow, R.; Chow, W.H.; Gammon, M.D.; Vaughan, T.L.; Farrow, D.C.; Schoenberg, J.B.; Stanford, J.L.; Ahsan, H.; et al. Nutrient intake and risk of subtypes of esophageal and gastric cancer. Cancer Epidemiol. Biomark. Prev. 2001, 10, 1055–1062. [Google Scholar] [PubMed]
- Brown, L.M.; Blot, W.J.; Schuman, S.H.; Smith, V.M.; Ershow, A.G.; Marks, R.D.; Fraumeni, J.F., Jr. Environmental factors and high risk of esophageal cancer among men in coastal south Carolina. J. Nat. Cancer Inst. 1988, 80, 1620–1625. [Google Scholar] [CrossRef] [PubMed]
- Franceschi, S.; Bidoli, E.; Negri, E.; Zambon, P.; Talamini, R.; Ruol, A.; Parpinel, M.; Levi, F.; Simonato, L.; La Vecchia, C. Role of macronutrients, vitamins and minerals in the aetiology of squamous-cell carcinoma of the oesophagus. Int. J. Cancer 2000, 86, 626–631. [Google Scholar] [CrossRef]
- Tuyns, A.J.; Riboli, E.; Doornbos, G.; Pequignot, G. Diet and esophageal cancer in Calvados (France). Nutr. Cancer 1987, 9, 81–92. [Google Scholar] [CrossRef] [PubMed]
- Tzonou, A.; Lipworth, L.; Garidou, A.; Signorello, L.B.; Lagiou, P.; Hsieh, C.; Trichopoulos, D. Diet and risk of esophageal cancer by histologic type in a low-risk population. Int. J. Cancer 1996, 68, 300–304. [Google Scholar] [CrossRef]
- Hu, J.; Nyren, O.; Wolk, A.; Bergstrom, R.; Yuen, J.; Adami, H.O.; Guo, L.; Li, H.; Huang, G.; Xu, X.; et al. Risk factors for oesophageal cancer in northeast China. Int. J. Cancer 1994, 57, 38–46. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Q.; Freedman, N.D.; Ren, J.; Hollenbeck, A.R.; Abnet, C.C.; Park, Y. Intakes of folate, methionine, vitamin B6, and vitamin B12 with risk of esophageal and gastric cancer in a large cohort study. Br. J. Cancer 2014, 110, 1328–1333. [Google Scholar] [CrossRef] [PubMed]
- Galeone, C.; Pelucchi, C.; Levi, F.; Negri, E.; Talamini, R.; Franceschi, S.; La Vecchia, C. Folate intake and squamous-cell carcinoma of the oesophagus in Italian and Swiss men. Ann. Oncol. 2006, 17, 521–525. [Google Scholar] [CrossRef] [PubMed]
- Launoy, G.; Milan, C.; Day, N.E.; Pienkowski, M.P.; Gignoux, M.; Faivre, J. Diet and squamous-cell cancer of the oesophagus: A French multicentre case-control study. Int. J. Cancer 1998, 76, 7–12. [Google Scholar] [CrossRef]
- De Stefani, E.; Ronco, A.L.; Boffetta, P.; Deneo-Pellegrini, H.; Acosta, G.; Correa, P.; Mendilaharsu, M. Nutrient intake and risk of squamous cell carcinoma of the esophagus: A case-control study in Uruguay. Nutr. Cancer 2006, 56, 149–157. [Google Scholar] [CrossRef] [PubMed]
- Aune, D.; Deneo-Pellegrini, H.; Ronco, A.L.; Boffetta, P.; Acosta, G.; Mendilaharsu, M.; De Stefani, E. Dietary folate intake and the risk of 11 types of cancer: A case-control study in Uruguay. Ann. Oncol. 2011, 22, 444–451. [Google Scholar] [CrossRef] [PubMed]
- Tavani, A.; Malerba, S.; Pelucchi, C.; Dal Maso, L.; Zucchetto, A.; Serraino, D.; Levi, F.; Montella, M.; Franceschi, S.; Zambon, A.; et al. Dietary folates and cancer risk in a network of case-control studies. Ann. Oncol. 2012, 23, 2737–2742. [Google Scholar] [CrossRef] [PubMed]
- Bollschweiler, E.; Wolfgarten, E.; Nowroth, T.; Rosendahl, U.; Monig, S.P.; Holscher, A.H. Vitamin intake and risk of subtypes of esophageal cancer in Germany. J. Cancer Res. Clin. Oncol. 2002, 128, 575–580. [Google Scholar] [CrossRef] [PubMed]
- Tang, L.; Lee, A.H.; Xu, F.; Zhang, T.; Lei, J.; Binns, C.W. Fruit and vegetable consumption and risk of esophageal cancer: A case-control study in north-west China. Dis. Esophagus 2014, 27, 777–782. [Google Scholar] [CrossRef] [PubMed]
- Zhao, P.; Lin, F.; Li, Z.; Lin, B.; Lin, J.; Luo, R. Folate intake, methylenetetrahydrofolate reductase polymorphisms, and risk of esophageal cancer. Asian Pac. J. Cancer Prev. 2011, 12, 2019–2023. [Google Scholar] [PubMed]
- Yang, C.X.; Matsuo, K.; Ito, H.; Shinoda, M.; Hatooka, S.; Hirose, K.; Wakai, K.; Saito, T.; Suzuki, T.; Maeda, T.; et al. Gene-environment interactions between alcohol drinking and the MTHFR C677T polymorphism impact on esophageal cancer risk: Results of a case-control study in Japan. Carcinogenesis 2005, 26, 1285–1290. [Google Scholar] [CrossRef] [PubMed]
- Miranda, T.B.; Jones, P.A. DNA methylation: The nuts and bolts of repression. J. Cell. Physiol. 2007, 213, 384–390. [Google Scholar] [CrossRef] [PubMed]
- Winter-Vann, A.M.; Kamen, B.A.; Bergo, M.O.; Young, S.G.; Melnyk, S.; James, S.J.; Casey, P.J. Targeting ras signaling through inhibition of carboxyl methylation: An unexpected property of methotrexate. Proc. Natl. Acad. Sci. USA 2003, 100, 6529–6534. [Google Scholar] [CrossRef] [PubMed]
- Stead, L.M.; Jacobs, R.L.; Brosnan, M.E.; Brosnan, J.T. Methylation demand and homocysteine metabolism. Adv. Enzyme Regul. 2004, 44, 321–333. [Google Scholar] [CrossRef] [PubMed]
- Medicine (US) Standing Committee on the Scientific Evaluation of Dietary Reference Intakes and its Panel on Folate, Other B Vitamins, and Choline. Dietary Reference Intakes for Thiamin, Riboflavin, Niacin, Vitamin B6, Folate, Vitamin B12, Pantothenic Acid, Biotin, and Choline; National Academies Press (US): Washington, DC, USA, 1998; pp. 196–305.
- Zhang, S.M.; Cook, N.R.; Albert, C.M.; Gaziano, J.M.; Buring, J.E.; Manson, J.E. Effect of combined folic acid, vitamin B6, and vitamin B12 on cancer risk in women: A randomized trial. JAMA 2008, 300, 2012–2021. [Google Scholar] [CrossRef] [PubMed]
- Brasky, T.M.; White, E.; Chen, C.-L. Long-term, supplemental, one-carbon metabolism-related vitamin B use in relation to lung cancer risk in the vitamins and lifestyle (VITAL) cohort. J. Clin. Oncol. 2017, 35, 3440–3448. [Google Scholar] [CrossRef] [PubMed]
- Blount, B.C.; Mack, M.M.; Wehr, C.M.; MacGregor, J.T.; Hiatt, R.A.; Wang, G.; Wickramasinghe, S.N.; Everson, R.B.; Ames, B.N. Folate deficiency causes uracil misincorporation into human DNA and chromosome breakage: Implications for cancer and neuronal damage. Proc. Natl. Acad. Sci. USA 1997, 94, 3290–3295. [Google Scholar] [CrossRef] [PubMed]
- Chan, J.; Deng, L.; Mikael, L.G.; Yan, J.; Pickell, L.; Wu, Q.; Caudill, M.A.; Rozen, R. Low dietary choline and low dietary riboflavin during pregnancy influence reproductive outcomes and heart development in mice. Am. J. Clin. Nutr. 2010, 91, 1035–1043. [Google Scholar] [CrossRef] [PubMed]
- Fiorito, G.; Guarrera, S.; Valle, C.; Ricceri, F.; Russo, A.; Grioni, S.; Mattiello, A.; Di Gaetano, C.; Rosa, F.; Modica, F.; et al. B-vitamins intake, DNA-methylation of one carbon metabolism and homocysteine pathway genes and myocardial infarction risk: The EPICOR study. Nutr. Metab. Cardiovasc. Dis. 2014, 24, 483–488. [Google Scholar] [CrossRef] [PubMed]
- Wiseman, M. The second world cancer research fund/American institute for cancer research expert report. Food, nutrition, physical activity, and the prevention of cancer: A global perspective. Proc. Nutr. Soc. 2008, 67, 253–256. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.W.; Mason, J.B. Folate and carcinogenesis: An integrated scheme. J. Nutr. 2000, 130, 129–132. [Google Scholar] [CrossRef] [PubMed]
- Liu, A.Y.; Scherer, D.; Poole, E.; Potter, J.D.; Curtin, K.; Makar, K.; Slattery, M.L.; Caan, B.J.; Ulrich, C.M. Gene-diet-interactions in folate-mediated one-carbon metabolism modify colon cancer risk. Mol. Nutr. Food Res. 2013, 57, 721–734. [Google Scholar] [CrossRef] [PubMed]
- Morita, R.; Hirohashi, Y.; Suzuki, H.; Takahashi, A.; Tamura, Y.; Kanaseki, T.; Asanuma, H.; Inoda, S.; Kondo, T.; Hashino, S.; et al. DNA methyltransferase 1 is essential for initiation of the colon cancers. Exp. Mol. Pathol. 2013, 94, 322–329. [Google Scholar] [CrossRef] [PubMed]
- Pathania, R.; Ramachandran, S.; Elangovan, S.; Padia, R.; Yang, P.; Cinghu, S.; Veeranan-Karmegam, R.; Arjunan, P.; Gnana-Prakasam, J.P.; Sadanand, F.; et al. DNMT1 is essential for mammary and cancer stem cell maintenance and tumorigenesis. Nat. Commun. 2015, 6, 6910. [Google Scholar] [CrossRef] [PubMed]
- Wolf, P.; Hu, Y.C.; Doffek, K.; Sidransky, D.; Ahrendt, S.A. O(6)-methylguanine-DNA methyltransferase promoter hypermethylation shifts the p53 mutational spectrum in non-small cell lung cancer. Cancer Res. 2001, 61, 8113–8117. [Google Scholar] [PubMed]
- Siassi, F.; Ghadirian, P. Riboflavin deficiency and esophageal cancer: A case control-household study in the caspian littoral of Iran. Cancer Detect. Prev. 2005, 29, 464–469. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Ye, L.; Shan, B.; Song, G.; Meng, F.; Wang, S. Effect of riboflavin-fortified salt nutrition intervention on esophageal squamous cell carcinoma in a high incidence area, China. Asian Pac. J. Cancer Prev. 2009, 10, 619–622. [Google Scholar] [PubMed]
- Expert Group on Vitamins and Minerals (EVM). Water Soluble Vitamins. In Safe Upper Levels for Vitamins and Minerals; Expert Group on Vitamins and Minerals: London, UK, 2003; pp. 36–100. [Google Scholar]
- Guest, J.; Bilgin, A.; Hokin, B.; Mori, T.A.; Croft, K.D.; Grant, R. Novel relationships between B12, folate and markers of inflammation, oxidative stress and NAD(H) levels, systemically and in the CNS of a healthy human cohort. Nutr. Neurosci. 2015, 18, 355–364. [Google Scholar] [CrossRef] [PubMed]
- Peracchi, M.; Bamonti Catena, F.; Pomati, M.; De Franceschi, M.; Scalabrino, G. Human cobalamin deficiency: Alterations in serum tumour necrosis factor-alpha and epidermal growth factor. Eur. J. Haematol. 2001, 67, 123–127. [Google Scholar] [CrossRef] [PubMed]
- Al-Daghri, N.M.; Rahman, S.; Sabico, S.; Yakout, S.; Wani, K.; Al-Attas, O.S.; Saravanan, P.; Tripathi, G.; McTernan, P.G.; Alokail, M.S. Association of vitamin B12 with pro-inflammatory cytokines and biochemical markers related to cardiometabolic risk in Saudi subjects. Nutrients 2016, 8, 460. [Google Scholar] [CrossRef] [PubMed]
- Pace, F.; Bianchi Porro, G. Gastroesophageal reflux disease: A typical spectrum disease (a new conceptual framework is not needed). Am. J. Gastroenterol. 2004, 99, 946–949. [Google Scholar] [CrossRef] [PubMed]
- Moons, L.M.; Kusters, J.G.; Bultman, E.; Kuipers, E.J.; van Dekken, H.; Tra, W.M.; Kleinjan, A.; Kwekkeboom, J.; van Vliet, A.H.; Siersema, P.D. Barrett’s oesophagus is characterized by a predominantly humoral inflammatory response. J. Pathol. 2005, 207, 269–276. [Google Scholar] [CrossRef] [PubMed]
- Tamura, J.; Kubota, K.; Murakami, H.; Sawamura, M.; Matsushima, T.; Tamura, T.; Saitoh, T.; Kurabayshi, H.; Naruse, T. Immunomodulation by vitamin B12: Augmentation of CD8+ T lymphocytes and natural killer (NK) cell activity in vitamin B12-deficient patients by methyl-B12 treatment. Clin. Exp. Immunol. 1999, 116, 28–32. [Google Scholar] [CrossRef] [PubMed]
- Sukocheva, O.A.; Abramov, A.Y.; Levitskaya, J.O.; Gagelgans, A.I.; Carpenter, D.O. Modulation of intracellular Ca(2+) concentration by vitamin B12 in rat thymocytes. Blood Cells Mol. Dis. 2001, 27, 812–824. [Google Scholar] [CrossRef] [PubMed]
- Kok, D.E.; Dhonukshe-Rutten, R.A.; Lute, C.; Heil, S.G.; Uitterlinden, A.G.; van der Velde, N.; van Meurs, J.B.; van Schoor, N.M.; Hooiveld, G.J.; de Groot, L.C.; et al. The effects of long-term daily folic acid and vitamin B12 supplementation on genome-wide DNA methylation in elderly subjects. Clin. Epigenet. 2015, 7, 121. [Google Scholar] [CrossRef] [PubMed]
- Damayanti, D.; Jaceldo-Siegl, K.; Beeson, W.L.; Fraser, G.; Oda, K.; Haddad, E.H. Foods and supplements associated with vitamin B12 biomarkers among vegetarian and non-vegetarian participants of the adventist health study-2 (AHA-2) calibration study. Nutrients 2018, 10, 722. [Google Scholar] [CrossRef] [PubMed]
- Sicinska, E.; Cholewa, M. Evaluation of the needs and possibilities of increasing the vitamin B12 content in diet. Rocz. Panstw. Zakl. Hig. 2012, 63, 67–71. [Google Scholar] [PubMed]
- O’Doherty, M.G.; Cantwell, M.M.; Murray, L.J.; Anderson, L.A.; Abnet, C.C.; Group, F.S. Dietary fat and meat intakes and risk of reflux esophagitis, barrett’s esophagus and esophageal adenocarcinoma. Int. J. Cancer 2011, 129, 1493–1502. [Google Scholar] [CrossRef] [PubMed]
| Author, Year | Country | Histological Type | Study-Design | Age Range (Years) | Participants (Cases) | Dietary Assessment | Subclass(es) of Vitamin B | Intake Comparison (Highest vs. Lowest) | Adjustment for Covariates | Quality Score |
|---|---|---|---|---|---|---|---|---|---|---|
| Franceschi et al., 2000 [29] | Italy | ESCC | HBCC | <79 | 1047 (304) | FFQ-78 items, validated | Vitamin B2 | Q5 vs. Q1 | Age, gender, area of residence, education, physical activity, BMI, tobacco smoking, alcohol drinking and non-alcohol energy. | 7 |
| Vitamin B6 | ||||||||||
| Folate | ||||||||||
| Jessri et al., 2011 [6] | Iran | ESCC | HBCC | 40–75 | 143 (47) | FFQ-125 items, validated | Vitamin B2 | T3 vs. T1 | Age (years), sex (male/female), gastroesophageal reflux disease symptoms, BMI, smoking status, smoking intensity, smoking duration, physical activity, and education level. | 8 |
| Vitamin B6 | ||||||||||
| Folate | ||||||||||
| Vitamin B12 | ||||||||||
| Chen et al., 2002 [25] | US | EAC | PBCC | >21 | 573 (124) | DHQ, validated | Vitamin B2 | Q4 vs. Q1 | Age, age squared, gender, respondent type, BMI, alcohol use, tobacco use, education level, family history of respective cancers, and vitamin supplement use. | 7 |
| Vitamin B6 | ||||||||||
| Folate | ||||||||||
| Tuyns et al., 1987 [30] | France | Mix-type | PBCC | N/A | 2718 (743) | DHQ-40 items, validated | Vitamin B2 | >2.0 mg/day vs. <1.5 mg/day | Age, alcohol consumption, and tobacco smoking. | 6 |
| Vitamin B6 | >3.0 mg/day vs. <2.0 mg/day | |||||||||
| Vitamin B12 | >10.0 μg/day vs. <5.0 μg/day | |||||||||
| Tzonou et al., 1996 [31] | Greece | ESCC/EAC | HBCC | N/A | 243 (43)/256 (56) | FFQ-115 items, validated | Vitamin B2 | Q5 vs. Q1 | Gender, age, birthplace, schooling, height, analgesic use, coffee drinking, alcohol intake, tobacco smoking, and energy intake (though not mutually analyzed). | 6 |
| Hu et al., 1994 [32] | China | Mix-type | HBCC | N/A | 588 (196) | FFQ-32 items, N/A | Vitamin B2 | Q4 vs. Q1 | Alcohol intake, smoking, household income, and occupation. | 7 |
| Launoy et al., 1998 [35] | France | ESCC | HBCC | <85 | 607 (208) | DHQ-39 items, N/A | Vitamin B6 | >2.5 mg/day vs. <1.5 mg/day | Interviewer, age, smoking, beer intake, aniseed aperitifs, hot Calvados, whisky, total alcohol intake, and total energy intake. | 7 |
| Galeone et al., 2006 [34] | Italy and Swiss | ESCC | HBCC | <80 | 1226 (351) | FFQ-78 items, validated | Vitamin B6 | >2.249 mg/day vs. <1.722 mg/day | Education, BMI, tobacco smoking, and alcohol drinking. | 8 |
| Folate | >305.1 μg/day vs. <228.1 μg/day | |||||||||
| Ibiebele et al., 2011 [8] | Australia | ESCC/EAC | PBCC | 18–79 | 1732 (225)/2120 (613) | FFQ-135 items, N/A | Vitamin B2 | 4.9 mg/d vs. 1.65 mg/day | Age, gender, education, BMI 1 year previously, frequency of heartburn or acid reflux 10 year prior to diagnosis, lifetime alcohol intake, pack-years of smoking, NSAID use, and total energy intake. | 7 |
| Vitamin B6 | 2.3 mg/day vs. 0.7 mg/day | |||||||||
| Folate | 504.5 μg/day vs. 136.0 μg/day | |||||||||
| Vitamin B12 | 4.95 μg/day vs. 0.55 μg/day | |||||||||
| Sharp et al., 2013 [7] | Ireland | EAC | PBCC | ≤85 | 479 (223) | FFQ-101 items, validated | Vitamin B2 | ≥2.8 mg/day vs. ≤1.8 mg/day | Age, sex, total energy, years of full-time education, BMI, and alcohol intake. | 8 |
| Vitamin B6 | ≥3.2 mg/day vs. ≤2.3 mg/day | |||||||||
| Folate | ≥421 μg/day vs. ≤318 μg/day | |||||||||
| Vitamin B12 | ≥9.7 μg/day vs. ≤6.4 μg/day | |||||||||
| Stefani et al., 2006 [36] | Uruguay | ESCC | HBCC | N/A | 1266 (234) | FFQ-64 items, validated | Folate | Q4 vs. Q1 | Age, sex, residence, urban/rural status, birthplace, education, BMI, smoking status, years since quitting smoking, number of cigarettes smoked per day, alcohol drinking, mate consumption, and total energy intake. | 7 |
| Mayne et al., 2001 [27] | US | ESCC/EAC | PBCC | 30–79 | 893 (206)/969 (282) | FFQ-104 items, validated | Vitamin B2 | 75th vs. 25th | Sex, location, age, race, proxy status, income, education, usual BMI, cigarettes smoked/day, years of consuming beer, wine, and/or hard liquor, and energy intake. | 8 |
| Vitamin B6 | ||||||||||
| Folate | ||||||||||
| Vitamin B12 | ||||||||||
| Tang et al., 2014 [40] | China | Mix-type | HBCC | N/A | 739 (359) | FFQ-137 items, validated | Folate | >204.5 μg/day vs. <104.5 μg/day | Age, gender, education level, BMI, total energy intake, smoking status, alcohol drinking, and family history of cancer in first-degree relatives. | 6 |
| Tavani et al., 2012 [38] | Italy | Mix-type | HBCC | N/A | 1767 (505) | FFQ-78 items, validated | Folate | ≥312.5 μg/day vs. ≤257.3 μg/day | Sex, age, study center, year of interview, education, alcohol drinking, tobacco smoking, BMI, total energy intake, and physical activity at work. | 7 |
| Zhao et al., 2011 [41] | China | ESCC | HBCC | 37–75 | 465 (155) | FFQ-45 items, N/A | Folate | >300 μg/day vs. <230 μg/day | Age, sex, smoking, and drinking. | 7 |
| Brown et al., 1988 [28] | US | Mix-type | PBCC | ≤79 | 629 (207) | DHQ, N/A | Vitamin B2 | high vs. low | Use of cigarettes and/or alcohol. | 6 |
| Folate | ||||||||||
| Aune et al., 2011 [37] | Uruguay | Mix-type | HBCC | <90 | 2266 (234) | FFQ-64 items, N/A | Folate | 275.31 μg/day vs. 123.83 μg/day | Age, sex, residence, education, income, interviewer, smoking status, cigarettes per day, duration of smoking, age at starting smoking, years since quitting smoking, calcium, dietary fiber, and iron intake, mate drinking, BMI, and energy intake. | 7 |
| Stefani et al., 1999 [26] | Uruguay | Mix-Type | HBCC | N/A | 459 (66) | FFQ-64 items, N/A | Vitamin B2 | T3 vs. T1 | Age, sex, residence, urban/rural status, education, BMI, tobacco smoking (in pack-years), alcohol drinking, and total energy intake. | 6 |
| Vitamin B6 | ||||||||||
| Folate | ||||||||||
| Vitamin B12 | ||||||||||
| Yang et al., 2005 [42] | Japan | Mix-type | HBCC | 18–80 | 660 (165) | FFQ-47 items, validated | Folate | >400 μg/day vs. <300 μg/day | Smoking, drinking, and total energy, | 6 |
| Bollschweiler et al., 2002 [39] | Germany | ESCC/EAC | PBCC | >40 | 102 (52)/97 (47) | FFQ-110 items, validated | Folate | >164 μg/day vs. 0–120 μg/day | N/A | 6 |
| Xiao et al., 2014 [33] | US | ESCC/EAC | Cohort | 50–71 | 490,780 (185)/491,169 (574) | FFQ-124 items, validated | Vitamin B6 | 2.7 mg/day vs. 1.4 mg/day | N/A | 7 |
| Folate | 566 μg/day vs. 288 μg/day | |||||||||
| Vitamin B12 | 7.3 μg/day vs. 2.5 μg/day |
| Vitamin B6 | Folate | Vitamin B12 | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| N | Cases/controls | OR (95%CI) | I2 | p-Value | N | Cases/Controls | OR (95%CI) | I2 | p-Value | N | Cases/Controls | OR (95%CI) | I2 | p-Value | |||
| Overall | 14 | 4151/497,974 | 0.59 (0.52–0.66) | 46.8% | 0.027 | 21 | 5158/501,583 | 0.62 (0.56–0.68) | 40.2% | 0.030 | 10 | 3164/495,508 | 1.30 (1.05–1.62) | 73.5% | 0.000 | ||
| Geographic Location | |||||||||||||||||
| Asia | - | - | - | - | - | 3 | 679/1185 | 0.77 (0.59–1.01) | 0.0% | 0.575 | - | - | - | - | - | ||
| America | 7 | 1484/492,219 | 0.59 (0.51–0.69) | 49.1% | 0.067 | 10 | 2159/496,392 | 0.58 (0.51–0.67) | 37.3% | 0.110 | 6 | 1360/492,457 | 1.26 (1.03–1.53) | 50.7% | 0.071 | ||
| Europe | 5 | 1829/4253 | 0.51 (0.34–0.78) | 66.6% | 0.017 | 6 | 1482/3186 | 0.51 (0.40–0.65) | 49.8% | 0.076 | 2 | 966/2231 | 2.54 (1.16–5.53) | 81.2% | 0.021 | ||
| Australia | 2 | 838/1507 | 0.57 (0.44–0.74) | 0.0% | 0.442 | 2 | 838/1507 | 0.74 (0.58–0.95) | 0.0% | 0.764 | 2 | 838/1507 | 0.93 (0.73–1.19) | 0.0% | 0.771 | ||
| Histological Type | |||||||||||||||||
| EAC | 5 | 1816/493–493 | 0.58 (0.49–0.68) | 0.0% | 0.487 | 6 | 1863/493,544 | 0.60 (0.51–0.69) | 34.1% | 0.181 | 4 | 1692/493,245 | 1.47 (1.02–2.11) | 82.9% | 0.001 | ||
| ESCC | 7 | 1526/494–901 | 0.47 (0.33–0.67) | 64.7% | 0.009 | 9 | 1759/495,894 | 0.61 (0.51–0.73) | 28.2% | 0.194 | 4 | 663/492,884 | 1.00 (0.63–1.61) | 74.55% | 0.008 | ||
| Dietary Assessment (FFQ/DHQ) | |||||||||||||||||
| Validated | 10 | 3039/495–675 | 0.58 (0.50–0.66) | 39.4% | 0.095 | 15 | 3658/497,556 | 0.59 (0.52–0.66) | 49.1% | 0.017 | 7 | 2260/493,608 | 1.45 (1.10–1.91) | 75.8% | 0.000 | ||
| N/A | 4 | 1112/2299 | 0.56 (0.38–0.83) | 68.2% | 0.024 | 6 | 1500/4664 | 0.70 (0.58–0.85) | 0.0% | 0.641 | 3 | 3164/1900 | 1.03 (0.84–1.26) | 8.4% | 0.335 | ||
| Study Design | |||||||||||||||||
| HBCC | 4 | 910/2113 | 0.41 (0.20–0.84) | 77.2% | 0.004 | 9 | 2354/7225 | 0.59 (0.44–0.77) | 55.3% | 0.022 | 1 | 47/380 | 1.33 (0.59–2.99) | - | - | ||
| PBCC | 8 | 2482/5267 | 0.58 (0.50–0.67) | 5.8% | 0.385 | 10 | 2045/3764 | 0.61 (0.52–0.70) | 22.8% | 0.233 | 7 | 2358/5505 | 1.42 (1.10–1.84) | 75.6% | 0.000 | ||
| Cohort | 2 | 758/490,594 | 0.51 (0.28–0.94) | 73.0% | 0.054 | 2 | 759/490,594 | 0.51 (0.28–0.94) | 73.0% | 0.054 | 2 | 759/490,594 | 0.86 (0.37–1.99) | 85.5% | 0.008 | ||
| Samples | |||||||||||||||||
| ≥500 | 11 | 3815/497–229 | 0.58 (0.51–0.65) | 40.9% | 0.076 | 15 | 4568/500,478 | 0.62 (0.42–0.76) | 38.7% | 0.063 | 7 | 2828/494,763 | 1.19 (0.96–1.47) | 70.5% | 0.002 | ||
| <500 | 3 | 336/745 | 0.47 (0.23–0.98) | 72.1% | 0.028 | 6 | 590/1105 | 0.48 (0.30–0.78) | 51.4% | 0.068 | 3 | 336/745 | 1.89 (0.90–3.98) | 80.0% | 0.007 | ||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qiang, Y.; Li, Q.; Xin, Y.; Fang, X.; Tian, Y.; Ma, J.; Wang, J.; Wang, Q.; Zhang, R.; Wang, J.; et al. Intake of Dietary One-Carbon Metabolism-Related B Vitamins and the Risk of Esophageal Cancer: A Dose-Response Meta-Analysis. Nutrients 2018, 10, 835. https://doi.org/10.3390/nu10070835
Qiang Y, Li Q, Xin Y, Fang X, Tian Y, Ma J, Wang J, Wang Q, Zhang R, Wang J, et al. Intake of Dietary One-Carbon Metabolism-Related B Vitamins and the Risk of Esophageal Cancer: A Dose-Response Meta-Analysis. Nutrients. 2018; 10(7):835. https://doi.org/10.3390/nu10070835
Chicago/Turabian StyleQiang, Yuzhen, Qianwen Li, Yongjuan Xin, Xuexian Fang, Yongmei Tian, Jifei Ma, Jianyao Wang, Qingqing Wang, Ruochen Zhang, Junhao Wang, and et al. 2018. "Intake of Dietary One-Carbon Metabolism-Related B Vitamins and the Risk of Esophageal Cancer: A Dose-Response Meta-Analysis" Nutrients 10, no. 7: 835. https://doi.org/10.3390/nu10070835
APA StyleQiang, Y., Li, Q., Xin, Y., Fang, X., Tian, Y., Ma, J., Wang, J., Wang, Q., Zhang, R., Wang, J., & Wang, F. (2018). Intake of Dietary One-Carbon Metabolism-Related B Vitamins and the Risk of Esophageal Cancer: A Dose-Response Meta-Analysis. Nutrients, 10(7), 835. https://doi.org/10.3390/nu10070835






