Dose-Dependent Effects of Multispecies Probiotic Supplementation on the Lipopolysaccharide (LPS) Level and Cardiometabolic Profile in Obese Postmenopausal Women: A 12-Week Randomized Clinical Trial
Abstract
1. Introduction
2. Methods
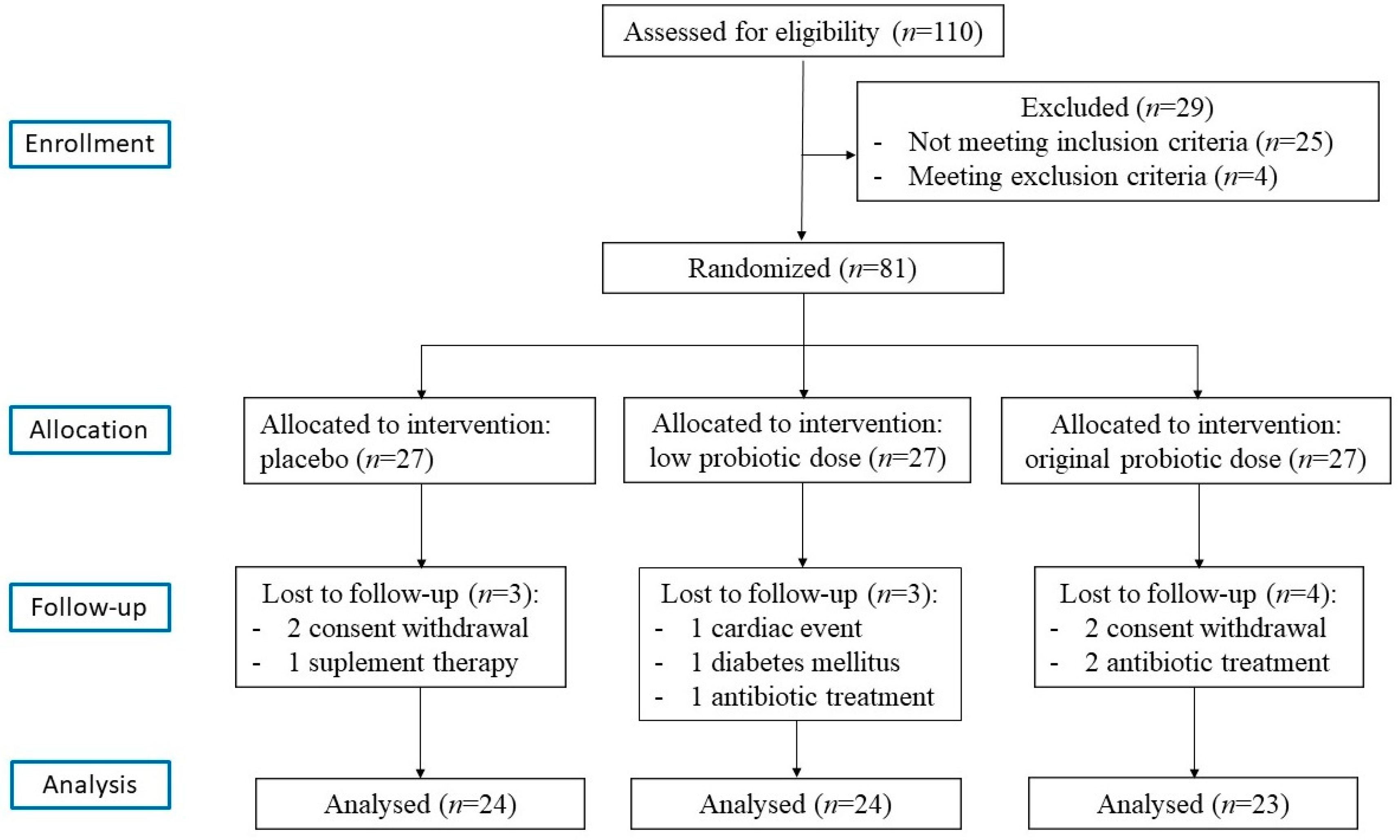
2.1. Subjects
2.2. Probiotic Supplements and Allocation
2.3. Anthropometric and Biochemical Measurement
2.4. Statistical Analysis
3. Results
4. Discussion
5. Strengths and Limitations
6. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- World Health Organization. Obesity, and Overweight. Available online: http://www.who.int/mediacentre/factsheets/fs311/en/ (accessed on 26 January 2016).
- Rahman, T.; Hosen, I.; Islam, M.M.T.; Shekhar, H.U. Oxidative stress, and human health. Adv. Biosci. Biotechnol. 2012, 3, 997–1019. [Google Scholar] [CrossRef]
- ABriones, M.; Touyz, R.M. Oxidative stress, and hypertension: Current concepts. Curr. Hypertens. Rep. 2010, 12, 135–142. [Google Scholar] [CrossRef] [PubMed]
- GBD 2013 Mortality and Causes of Death Collaborators. Global, regional, and national age-sex specific all-cause and cause-specific mortality for 240 causes of death, 1990–2013: A systematic analysis for the Global Burden of Disease Study 2013. Lancet 2015, 385, 117–171. [Google Scholar]
- Atsma, F.; Bartelink, M.L.; Grobbee, D.E.; van der Schouw, Y.T. Postmenopausal status and early menopause as independent risk factors for cardiovascular disease: A meta-analysis. Menopause 2006, 13, 265–279. [Google Scholar] [CrossRef] [PubMed]
- Mozaffarian, D.; Benjamin, E.J.; Go, A.S.; Arnett, D.K.; Blaha, M.J.; Cushman, M.; Das, S.R.; de Ferranti, S.; Despres, J.P.; Fullerton, H.J.; et al. Heart disease and stroke statistics—2016 update: A report from the American Heart Association. Circulation 2016, 133, e38–e360. [Google Scholar] [CrossRef] [PubMed]
- Collins, P.; Webb, C.M.; de Villiers, T.J.; Stevenson, J.C.; Panay, N.; Baber, R.J. Cardiovascular risk assessment in women—An update. Climacteric 2016, 19, 329–336. [Google Scholar] [CrossRef] [PubMed]
- He, M.; Shi, B. Gut microbiota as a potential target of metabolic syndrome: The role of probiotics and prebiotics. Cell Biosci. 2017, 7, 54. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.M.; Hazen, S.L. The Gut Microbial Endocrine Organ: Bacterially-Derived Signals Driving Cardiometabolic Disease. Annu. Rev. Med. 2015, 66, 343–359. [Google Scholar] [CrossRef] [PubMed]
- Ebel, B.; Lemetis, G.; Beney, L.; Cachon, R.; Sokol, H.; Langella, P.; Gervais, P. Impact of probiotics on risk factors for cardiovascular diseases. A review. Crit. Rev. Food Sci. Nutr. 2014, 54, 175–189. [Google Scholar] [CrossRef] [PubMed]
- Yoo, J.Y.; Kim, S.S. Probiotics, and Prebiotics: Present Status and Future Perspectives on Metabolic Disorders. Nutrients 2016, 8, 173. [Google Scholar] [CrossRef] [PubMed]
- Whitman, W.B.; Coleman, D.C.; Wiebe, W.J. Prokaryotes: The unseen majority. Proc. Natl. Acad. Sci. USA 1998, 95, 6578–6583. [Google Scholar] [CrossRef] [PubMed]
- The Human Microbiome Project Consortium. Structure, function, and diversity of the healthy human microbiome. Nature 2012, 486, 207–214. [Google Scholar]
- Wang, Z.; Klipfell, E.; Bennett, B.J.; Koeth, R.; Levison, B.S.; DuGar, B.; Feldstein, A.E.; Britt, E.B.; Fu, X.; Chung, Y.M.; et al. Gut flora metabolism of phosphatidylcholine promotes cardiovascular disease. Nature 2011, 472, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Koeth, R.A.; Wang, Z.; Levinson, B.S.; Buffa, J.A.; Org, E.; Sheehy, B.T.; Britt, E.B.; Fu, X.; Wu, Y.; Li, L.; et al. Intestinal microbiota metabolism of L-carnitine, a nutrient in red meat, promotes atherosclerosis. Nat. Med. 2013, 19, 576–585. [Google Scholar] [CrossRef] [PubMed]
- Tang, W.H.; Wang, Z.; Levison, B.S.; Levison, B.S.; Koeth, R.A.; Britt, E.B.; Fu, X.; Wu, Y.; Hazen, S.L. Intestinal microbial metabolism of phosphatidylcholine and cardiovascular risk. N. Engl. J. Med. 2013, 368, 1575–1578. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Tang, W.H.; Buffa, J.A.; Fu, X.; Britt, E.B.; Koeth, R.A.; Levison, B.S.; Fan, Y.; Wu, Y.; Hazen, S.L. Prognostic value of choline and betaine depends on intestinal microbiota-generated metabolite trimethylamine-N-oxide. Eur. Heart J. 2014, 35, 904–910. [Google Scholar] [CrossRef] [PubMed]
- Bennett, B.J.; de Aguiar Vallim, T.Q.; Wang, Z.; Shih, D.M.; Meng, Y.; Gregory, J.; Allayee, H.; Lee, R.; Graham, M.; Crooke, R.; et al. Trimethylamine-N-oxide, a metabolite associated with atherosclerosis, exhibits complex genetic and dietary regulation. Cell Metab. 2013, 17, 49–60. [Google Scholar] [CrossRef] [PubMed]
- Creely, S.J.; McTernan, P.G.; Kusminski, C.M.; Fisher, M.; Da Silva, N.F.; Khanolkar, M.; Evans, M.; Harte, A.L.; Kumar, S. Lipopolysaccharide activates an innate immune system response in human adipose tissue in obesity and type 2 diabetes. Am. J. Physiol. Endocrinol. Metab. 2007, 292, E740–E747. [Google Scholar] [CrossRef] [PubMed]
- Claesson, M.J.; Jeffery, I.B.; Conde, S.; Power, S.E.; O’Connor, E.M.; Cusack, S.; Harris, H.M.B.; Coakley, M.; Lakshminarayanan, B.; O’Sullivan, O.; et al. Gut microbiota composition correlates with diet and health in the elderly. Nature 2012, 488, 178–184. [Google Scholar] [CrossRef] [PubMed]
- Yatsunenko, T.; Rey, F.E.; Manary, M.J.; Trehan, I.; Dominguez-Bello, M.G.; Contreras, M.; Magris, M.; Hidalgo, G.; Baldassano, R.N.; Anokhin, A.P.; et al. Human gut microbiome viewed across age and geography. Nature 2012, 486, 222–227. [Google Scholar] [CrossRef] [PubMed]
- Muegge, B.D.; Kuczynski, J.; Knights, D.; Clemente, J.C.; González, A.; Fontana, L.; Henrissat, B.; Knight, R.; Gordon, J.I. Diet drives convergence in gut microbiome functions across mammalian phylogeny and within humans. Science 2011, 332, 970–974. [Google Scholar] [CrossRef] [PubMed]
- Ohland, C.L.; Macnaughton, W.K. Probiotic Bacteria and Intestinal Epithelial Barrier Function. Am. J. Physiol. Gastrointest. Liver Physiol. 2010, 298, G807–G819. [Google Scholar] [CrossRef] [PubMed]
- Miyauchi, E.; Hurley, G.; Melgar, S.; Tanabe, S.; Shanahan, F.; Nally, K.; O’Toole, P.W. Mechanism of Protection of Transepithelial Barrier Function by Lactobacillus Salivarius: Strain-Dependence and Attenuation by Bacteriocin Production. Am. J. Physiol. Gastrointest. Liver Physiol. 2012, 303, G1029–G1104. [Google Scholar] [CrossRef] [PubMed]
- Van Hemert, S.; Verwer, J.; Schütz, B. Clinical Studies Evaluation Effects of Probiotics on Parameters of Intestinal Barrier Function. Adv. Microbiol. 2013, 3, 212–221. [Google Scholar] [CrossRef]
- Van Hemert, S.; Ormel, G. Influence of the Multispecies Probiotic Ecologic® BARRIER on Parameters of Intestinal Barrier Function. Food Nutr. Sci. 2014, 5, 1739–1745. [Google Scholar]
- Sabico, S.; Al-Mashharawi, A.; Al-Daghri, N.; Yakout, S.; Alnaami, M.; Alokail, M.; McTernan, F. Effects of a multi-strain probiotic supplement for 12 weeks in circulating endotoxin levels and cardiometabolic profiles of medication naïve T2DM patients: A randomized clinical trial. J. Transl. Med. 2017, 15, 249. [Google Scholar] [CrossRef] [PubMed]
- Cani, P.D. Gut microbiota and obesity: Lessons from the microbiome. Brief Funct. Genom. 2013, 12, 381–387. [Google Scholar] [CrossRef] [PubMed]
- Bindels, L.B.; Dewulf, E.M.; Delzenne, N.M. GPR43/FFA2: Physiopathological relevance and therapeutic prospects. Trends Pharmacol. Sci. 2013, 34, 226–232. [Google Scholar] [CrossRef] [PubMed]
- Takemura, N.; Okubo, T.; Sonoyama, K. Lactobacillus plantarum strain No. 14 reduces adipocyte size in mice fed high-fat diet. Exp. Biol. Med. 2010, 235, 849–856. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.H.; Yun, S.I.; Park, H.O. Effects of Lactobacillus gasseri BNR17 on body weight and adipose tissue mass in diet-induced overweight rats. J. Microbiol. 2010, 48, 712–714. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.Y.; Park, J.H.; Seok, S.H.; Baek, M.W.; Kim, D.J.; Lee, K.E.; Paek, K.S.; Lee, Y.; Park, J.H. Human originated bacteria, Lactobacillus rhamnosus PL60, produce conjugated linoleic acid and show anti-obesity effects in diet-induced obese mice. Biochim. Biophys. Acta 2006, 1761, 736–744. [Google Scholar] [CrossRef] [PubMed]
- Hamad, E.M.; Sato, M.; Uzu, K.; Yoshida, T.; Higashi, S.; Kawakami, H.; Kadooka, Y.; Matsuyama, H.; Abd El-Gawad, I.A.; Imaizumi, K. Milk fermented by Lactobacillus gasseri SBT2055 influences adipocyte size via inhibition of dietary fat absorption in Zucker rats. Br. J. Nutr. 2009, 101, 716–724. [Google Scholar] [CrossRef] [PubMed]
- Miyoshi, M.; Ogawa, A.; Higurashi, S.; Kadooka, Y. Anti-obesity effect of Lactobacillus gasseri SBT2055 accompanied by inhibition of pro-inflammatory gene expression in the visceral adipose tissue in diet-induced obese mice. Eur. J. Nutr. 2014, 53, 599–606. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Guo, L.; Zhang, Y.; Walzem, R.L.; Pendergast, J.S.; Printz, R.L.; Morris, L.C.; Matafonova, E.; Stien, X.; Kang, L.; et al. Incorporation of therapeutically modified bacteria into gut microbiota inhibits obesity. J. Clin. Investig. 2014, 124, 3391–3406. [Google Scholar] [CrossRef] [PubMed]
- Borgeraas, H.; Johnson, L.K.; Skattebu, J.; Hertel, J.K.; Hjelmesaeth, J. Effects of probiotics on body weight, body mass index, fat mass and fat percentage in subjects with overweight or obesity: A systematic review and meta-analysis of randomized controlled trials. Obes. Rev. 2018, 19, 219–232. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Grover, S.; Batish, V.K. Hypocholesterolemic effect of dietary inclusion of two putative probiotic bile salt hydrolase-producing Lactobacillus plantarum strains in Sprague-Dawley rats. Br. J. Nutr. 2011, 105, 561–573. [Google Scholar] [CrossRef] [PubMed]
- Ejtahed, H.S.; Mohtadi-Nia, J.; Homayouni-Rad, A.; Niafar, M. Effect of probiotic yogurt containing Lactobacillus acidophilus and Bifidobacterium lactis on lipid profile in individuals with type 2 diabetes mellitus. J. Dairy Sci. 2011, 94, 3288–3294. [Google Scholar] [CrossRef] [PubMed]
- Asemi, Z.; Zare, Z.; Shakeri, H.; Sabihi, S.; Esmaillzadeh, A. Effect of multispecies probiotic supplements on metabolic profile, hs-CRP, and oxidative stress in patients with type 2 diabetes. Ann. Nutr. Metab. 2013, 63, 631–639. [Google Scholar] [CrossRef] [PubMed]
- Fukushima, M.; Yamada, A.; Endo, T.; Nakano, M. Effects of a mixture of organisms, Lactobacillus acidophilus or Streptococcus faecalis on delta6-desaturase activity in the livers of rats fed a fat- and cholesterol-enriched diet. Nutrition 1999, 15, 373–378. [Google Scholar] [CrossRef]
- Shimizu, M.; Hashiguchi, M.; Shiga, T.; Tamura, H.O.; Mochizuki, M. Meta-Analysis: Effects of Probiotic Supplementation on Lipid Profiles in Normal to Mildly Hypercholesterolemic Individuals. PLoS ONE 2015, 10, e0139795. [Google Scholar] [CrossRef] [PubMed]
- Honda, K.; Saneyasu, T.; Hasegawa, S.; Tominaga, Y.; Yokota, S.; Kamisoyama, H. Effect of licorice flavonoid oil on cholesterol metabolism in high-fat diet rats. Biosci. Biotechnol. Biochem. 2013, 77, 1326–1328. [Google Scholar] [CrossRef] [PubMed]
- Yadav, H.; Jain, S.; Sinha, P.R. Antidiabetic effect of probiotic dahi containing Lactobacillus acidophilus and Lactobacillus casei in high fructose-fed rats. Nutrition 2007, 23, 62–68. [Google Scholar] [CrossRef] [PubMed]
- Cani, P.D.; Neyrinck, A.M.; Fava, F.; Knaus, C.; Burcelin, R.G.; Tuohy, K.M. Selective increases of bifidobacteria in gut microflora improve high-fat diet-induced diabetes in mice through the mechanism associated with endotoxemia. Diabetologia 2007, 50, 2374–2383. [Google Scholar] [CrossRef] [PubMed]
- Everard, A.; Belzer, C.; Geurts, L.; Ouwerkerk, J.P.; Druart, C.; Bindels, L.B.; Guiot, Y.; Derrien, M.; Muccioli, G.G.; Delzenne, N.M.; et al. Cross-talk between Akkermansia muciniphila and intestinal epithelium controls diet-induced obesity. Proc. Natl. Acad. Sci. USA 2013, 110, 9066–9071. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Yang, D.; Mei, L.; Yuan, L.; Xie, A.; Yuan, J. Screening and Characterization of Purine Nucleoside Degrading Lactic Acid Bacteria Isolated from Chinese Sauerkraut and Evaluation of the Serum Uric Acid Lowering Effect in Hyperuricemic Rats. PLoS ONE 2014, 9, e105577. [Google Scholar] [CrossRef] [PubMed]
- Dehghani, H.; Heidari, F.; Mozaffari-Khosravi, H.; Nouri-Majelan, N.; Dehghani, A. Synbiotic Supplementations for Azotemia in Patients with Chronic Kidney Disease: A Randomized Controlled Trial. Iran. J. Kidney Dis. 2016, 10, 351–357. [Google Scholar] [PubMed]
- Hashem, M.A.; Mohamed, M.H. Haemato-biochemical and pathological studies on aflatoxicosis and treatment of broiler chicks in Egypt. Vet. Ital. 2009, 45, 323–337. [Google Scholar] [PubMed]
- Brun, P.; Castigaliuolo, I.; Leo, V.D.; Buda, A.; Pinzani, M.; Palu, G.; Martines, D. Increased intestinal permeability in obese mice: New evidence in the pathogenesis of nonalcoholic steatohepatitis. Am. J. Physiol. Gastrointest. Liver Physiol. 2007, 292, G518–G525. [Google Scholar] [CrossRef] [PubMed]
- Le Barz, M.; Anhe, F.F.; Varin, T.C.; Desjardins, Y.; Levy, E.; Roy, D.; Urdaci, M.C.; Marette, A. Probiotics as complementary treatment for metabolic disorders. Diabetes Metab. J. 2015, 39, 291–303. [Google Scholar] [CrossRef] [PubMed]
| Baseline | Group | Mean ± SD | SMD | p-Value |
|---|---|---|---|---|
| Body mass (kg) | High-Dose | 94.46 ± 16.61 | 0.11 * | 0.9746 |
| Low-Dose | 92.92 ± 13.66 | 0.01 † | ||
| Placebo | 92.81 ± 11.93 | 0.10 # | ||
| BMI (kg/m2) | High-Dose | 36.57 ± 5.95 | 0.09 * | 0.9365 |
| Low-Dose | 36.00 ± 5.20 | −0.02 † | ||
| Placebo | 36.10 ± 4.37 | 0.10 # | ||
| Age (years) | High-Dose | 55.16 ± 6.87 | −0.50 * | 0.2977 |
| Low-Dose | 56.38 ± 6.55 | −0.35 † | ||
| Placebo | 58.72 ± 7.25 | −0.18 # | ||
| Height (cm) | High-Dose | 160.82 ± 6.23 | 0.06 * | 0.9586 |
| Low-Dose | 160.69 ± 5.43 | 0.04 † | ||
| Placebo | 160.44 ± 6.38 | 0.02 # | ||
| Waist circumference (cm) | High-Dose | 109.84 ± 11.66 | 0.09 * | 0.9487 |
| Low-Dose | 109.65 ± 10.66 | 0.08 † | ||
| Placebo | 108.90 ± 7.31 | 0.02 # | ||
| HR (bpm) | High-Dose | 76.26 ± 10.68 | 0.44 * | 0.2466 |
| Low-Dose | 73.58 ± 10.18 | 0.13 † | ||
| Placebo | 72.48 ± 6.06 | 0.26 # | ||
| SBP (mmHg) | High-Dose | 134.80 ± 10.10 | 0.10 * | 0.7391 |
| Low-Dose | 133.50 ± 10.86 | −0.01 † | ||
| Placebo | 133.64 ± 12.20 | 0.12 # | ||
| DBP (mmHg) | High-Dose | 79.88 ± 8.05 | −0.51 * | 0.1446 |
| Low-Dose | 82.46 ± 5.53 | −0.20 † | ||
| Placebo | 83.76 ± 7.26 | −0.37 # | ||
| Fat (%) | High-Dose | 50.91 ± 6.51 | −0.29 * | 0.7040 |
| Low-Dose | 51.52 ± 5.34 | −0.21 † | ||
| Placebo | 52.85 ± 6.93 | −0.10 # | ||
| Fat (kg) | High-Dose | 48.48 ± 13.97 | −0.02 * | 0.9577 |
| Low-Dose | 48.22 ± 11.38 | −0.05 † | ||
| Placebo | 48.79 ± 11.02 | 0.02 # | ||
| FFM (%) | High-Dose | 46.88 ± 8.03 | 0.25 * | 0.8152 |
| Low-Dose | 47.06 ± 6.25 | 0.30 † | ||
| Placebo | 44.91 ± 8.09 | −0.03 # | ||
| FFM (kg) | High-Dose | 45.95 ± 5.93 | 0.44 * | 0.2934 |
| Low-Dose | 44.70 ± 4.57 | 0.27 † | ||
| Placebo | 43.03 ± 7.39 | 0.24 # | ||
| TBW (%) | High-Dose | 37.25 ± 5.19 | 0.26 * | 0.8222 |
| Low-Dose | 36.61 ± 4.04 | 0.16 † | ||
| Placebo | 35.81 ± 5.80 | 0.14 # | ||
| TBW (I) | High-Dose | 35.01 ± 5.11 | 0.34 * | 0.4581 |
| Low-Dose | 33.77 ± 3.56 | 0.14 † | ||
| Placebo | 33.08 ± 6.22 | 0.28 # | ||
| FFMH (%) | High-Dose | 76.35 ± 3.33 | −0.23 * | 0.5066 |
| Low-Dose | 75.70 ± 2.50 | −0.49 † | ||
| Placebo | 77.07 ± 3.05 | 0.22 # | ||
| Visceral fat (%) | High-Dose | 206.38 ± 66.91 | −0.26 * | 0.8386 |
| Low-Dose | 218.36 ± 79.02 | −0.07 † | ||
| Placebo | 223.77 ± 69.17 | −0.16 # | ||
| Subcutaneous fat (%) | High-Dose | 297.43 ± 81.90 | 0.08 * | 0.6378 |
| Low-Dose | 278.41 ± 88.98 | −0.16 † | ||
| Placebo | 291.27 ± 65.79 | 0.22 # |
| Baseline | Group | Mean ± SD | SMD | p-Value |
|---|---|---|---|---|
| Uric acid (mmol/L) | High-Dose | 6.02 ± 0.71 | 0.50 * | 0.0575 |
| Low-Dose | 5.26 ± 1.04 | −0.23 † | ||
| Placebo | 5.52 ± 1.23 | 0.85 # | ||
| TC (mg/dL) | High-Dose | 218.56 ± 32.75 | 0.44 * | 0.1377 |
| Low-Dose | 222.27 ± 43.45 | 0.47 † | ||
| Placebo | 203.60 ± 35.21 | −0.10 # | ||
| HDL (mg/dL) | High-Dose | 52.48 ± 10.71 | 0.02 * | 0.0912 |
| Low-Dose | 58.27 ± 11.96 | 0.54 † | ||
| Placebo | 52.32 ± 9.93 | −0.51 # | ||
| TG (mg/dL) | High-Dose | 165.04 ± 78.15 | 0.33 * | 0.3519 |
| Low-Dose | 134.12 ± 45.98 | −0.14 † | ||
| Placebo | 141.76 ± 62.88 | 0.48 # | ||
| LDL (mg/dL) | High-Dose | 119.40 ± 31.86 | 0.11 * | 0.2828 |
| Low-Dose | 129.38 ± 46.81 | 0.33 † | ||
| Placebo | 115.92 ± 33.47 | −0.25 # | ||
| Glucose (mg/dL) | High-Dose | 98.60 ± 5.97 | 0.21 * | 0.0620 |
| Low-Dose | 92.81 ± 9.72 | −0.29 † | ||
| Placebo | 96.32 ± 14.35 | 0.72 # | ||
| INS (IU/L) | High-Dose | 35.74 ± 12.05 | 0.59 * | 0.0521 |
| Low-Dose | 28.22 ± 10.55 | −0.10 † | ||
| Placebo | 29.28 ± 9.87 | 0.66 # | ||
| HOMA-IR | High-Dose | 8.69 ± 3.00 | 0.65 * | 0.0607 |
| Low-Dose | 6.49 ± 2.59 | −0.17 † | ||
| Placebo | 6.92 ± 2.46 | 0.79 # | ||
| LPS (ng/mL) | High-Dose | 13.01 ± 5.22 | 0.54 * | 0.0710 |
| Low-Dose | 12.28 ± 6.71 | 0.33 † | ||
| Placebo | 10.73 ± 3.32 | 0.12 # |
| Parameter | High-Dose | Low-Dose | Placebo | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Baseline | After 3 Months | SMD | p Value | Baseline | After 3 Months | SMD | p Value | Baseline | After 3 Months | SMD | p Value | |
| Body mass | 94.46 ± 16.61 | 93.46 ± 14.76 | −0.34 | 0.2173 | 92.92 ± 13.66 | 91.82 ± 13.77 | −0.39 | 0.0795 | 92.81 ± 11.93 | 92.56 ± 12.37 | −0.10 | 0.5937 |
| BMI | 36.57 ± 5.95 | 36.22 ± 5.29 | −0.31 | 0.3165 | 36.00 ± 5.20 | 35.51 ± 5.16 | −0.39 | 0.1209 | 36.10 ± 4.37 | 36.04 ± 4.32 | −0.07 | 0.9612 |
| Waist | 109.84 ± 11.66 | 107.97 ± 10.11 | −0.54 | 0.0199 | 109.65 ± 10.66 | 105.48 ± 11.97 | −1.06 | 0.0001 | 108.9 ± 0.31 | 107.27 ± 7.16 | −0.37 | 0.0888 |
| Fat% | 50.91 ± 6.51 | 49.54 ± 8.45 | −0.41 | 0.1298 | 51.52 ± 5.34 | 50.41 ± 5.60 | −0.54 | 0.0103 | 52.85 ± 6.93 | 51.38 ± 7.19 | −0.40 | 0.0544 |
| Fat (kg) | 48.48 ± 13.97 | 46.81 ± 14.26 | −0.22 | 0.0397 | 48.22 ± 11.38 | 46.63 ± 10.53 | −0.62 | 0.0099 | 48.79 ± 11.02 | 47.75 ± 11.24 | −0.29 | 0.0779 |
| FFM% | 46.88 ± 8.03 | 46.46 ± 10.41 | −0.09 | 0.6649 | 47.06 ± 6.25 | 46.66 ± 7.02 | −0.16 | 0.3948 | 44.91 ± 8.09 | 45.10 ± 9.35 | 0.06 | 0.4118 |
| FMM (kg) | 45.95 ± 5.93 | 45.60 ± 6.82 | −0.08 | 0.7361 | 44.70 ± 4.57 | 43.93 ± 4.36 | −0.30 | 0.1870 | 43.03 ± 7.39 | 43.19 ± 9.68 | 0.04 | 0.4852 |
| TBW% | 37.25 ± 5.19 | 37.83 ± 7.87 | 0.11 | 0.6389 | 36.61 ± 4.04 | 36.75 ± 4.07 | 0.08 | 0.7097 | 35.81 ± 5.80 | 36.27 ± 6.62 | 0.14 | 0.9095 |
| TBW (Itr) | 35.01 ± 5.11 | 35.24 ± 6.41 | 0.05 | 0.5901 | 33.77 ± 3.56 | 33.44 ± 3.91 | −0.15 | 0.4928 | 33.08 ± 6.22 | 34.10 ± 7.97 | 0.26 | 0.4455 |
| FFMH% | 76.35 ± 3.33 | 77.86 ± 3.68 | 0.39 | 0.0129 | 75.70 ± 2.50 | 76.03 ± 3.94 | 0.19 | 0.4556 | 77.07 ± 3.05 | 77.54 ± 3.39 | 0.30 | 0.1492 |
| Visceral fat (%) | 206.38 ± 66.91 | 208.71 ± 66.91 | 0.03 | 0.8176 | 218.36 ± 79.02 | 192.86 ± 62.38 | −0.58 | 0.0336 | 223.77 ± 69.17 | 212.14 ± 56.93 | −0.23 | 0.2514 |
| Subcutaneous fat (%) | 297.43 ± 81.90 | 229.29 ± 65.30 | −0.83 | 0.0002 | 278.41 ± 88.98 | 225.50 ± 59.93 | −0.99 | 0.0022 | 291.27 ± 65.79 | 241.77 ± 67.28 | −0.34 | 0.0700 |
| Uric acid (mmo/L) | 6.02 ± 0.71 | 5.35 ± 0.91 | −0.87 | 0.0001 | 5.26 ± 1.04 | 5.28 ± 1.09 | 0.04 | 0.8401 | 5.52 ± 1.23 | 5.40 ± 1.02 | −0.19 | 0.4004 |
| TC (mg/dL) | 218.56 ± 32.75 | 202.56 ± 30.76 | −0.57 | 0.0019 | 222.27 ± 43.45 | 211.50 ± 41.39 | −0.49 | 0.0124 | 203.60 ± 35.21 | 198.08 ± 37.86 | −0.18 | 0.3259 |
| HDL-C (mg/dL) | 52.48 ± 10.71 | 54.68 ± 8.63 | 0.22 | 0.1295 | 58.27 ± 11.96 | 58.50 ± 11.34 | 0.02 | 0.8639 | 52.32 ± 9.93 | 55.48 ± 10.76 | 0.40 | 0.0511 |
| TG (mg/dL) | 165.04 ± 78.15 | 153.40 ± 55.63 | −0.43 | 0.0140 | 134.12 ± 45.98 | 123.88 ± 39.51 | −0.37 | 0.0959 | 141.76 ± 62.88 | 135.72 ± 69.01 | −0.19 | 0.3002 |
| LDL-C (mg/dL) | 119.40 ± 31.86 | 114.64 ± 37.16 | −0.41 | 0.0149 | 129.38 ± 46.81 | 121.15 ± 40.62 | −0.59 | 0.0168 | 113.28 ± 35.25 | 115.92 ± 33.47 | 0.19 | 0.3732 |
| Glucose (mg/dL) | 98.60 ± 5.97 | 90.79 ± 8.82 | −0.94 | 0.0001 | 92.81 ± 9.72 | 92.38 ± 12.29 | −0.04 | 0.8484 | 96.32 ± 14.35 | 94.92 ± 8.24 | −0.18 | 0.6373 |
| INS (IU/L) | 35.74 ± 12.05 | 27.73 ± 9.23 | −0.72 | 0.0002 | 28.22 ± 10.55 | 23.93 ± 8.97 | −0.76 | 0.0007 | 29.28 ± 9.87 | 29.58 ± 8.39 | 0.05 | 0.8119 |
| HOMA-IR | 8,69 ± 3.00 | 6.32 ± 2.47 | −0.82 | 0.0001 | 6.49 ± 2.59 | 5.50 ± 2.27 | −0.54 | 0.0194 | 6.92 ± 2.46 | 6.94 ± 2.15 | 0.01 | 0.9406 |
| LPS (ng/mL) | 13.01 ± 5.22 | 10.39 ± 5.54 | −0.77 | 0.0008 | 12.28 ± 6.71 | 11.95 ± 6.84 | −0.09 | 0.2414 | 10.73 ± 3.32 | 11.0 ± 3.49 | 0.17 | 0.5104 |
| Variable | Group | Mean ± SD | p-Value | SMD | p-Value Post-Hoc |
|---|---|---|---|---|---|
| Δ Body mass (kg) | High-Dose | −0.99 ± 3.37 | 0.8611 | −0.26 * | ns |
| Low-Dose | −1.10 ± 3.07 | −0.31 † | |||
| Placebo | −0.25 ± 2.28 | 0.03 # | |||
| Δ BMI (kg/m2) | High-Dose | −0.35 ± 1.29 | 0.6960 | −0.26 * | ns |
| Low-Dose | −0.49 ± 1.29 | −0.38 † | |||
| Placebo | −0.06 ± 0.87 | 0.11 # | |||
| Δ Waist circumference (cm) | High-Dose | −1.90 ± 3.81 | 0.1777 | −0.06 * | ns |
| Low-Dose | −4.17 ± 4.05 | −0.58 † | |||
| Placebo | −1.67 ± 4.27 | 0.55 # | |||
| Δ Fat (%) | High-Dose | −1.37 ± 4.36 | 0.9704 | −0.68 * | ns |
| Low-Dose | −1.10 ± 2.03 | −0.84 † | |||
| Placebo | 1.46 ± 3.37 | −0.08 # | |||
| Δ Fat (kg) | High-Dose | −1.67 ± 4.49 | 0.7858 | −0.17 * | ns |
| Low-Dose | −1.59 ± 2.65 | −0.21 † | |||
| Placebo | −1.03 ± 2.62 | −0.02 # | |||
| Δ FFM (%) | High-Dose | −0.42 ± 4.79 | 0.5319 | −0.15 * | ns |
| Low-Dose | −0.40 ± 2.37 | −0.20 † | |||
| Placebo | 0.19 ± 3.33 | −0.01 # | |||
| Δ FMM (kg) | High-Dose | −0.35 ± 4.67 | 0.9471 | −0.11 * | ns |
| Low-Dose | −0.76 ± 2.63 | −0.25 † | |||
| Placebo | 0.15 ± 4.28 | 0.11 # | |||
| Δ TBW (%) | High-Dose | 0.58 ± 5.72 | 0.5381 | 0.03 * | ns |
| Low-Dose | 0.14 ± 1.78 | −0.12 † | |||
| Placebo | 0.46 ± 3.19 | 0.10 # | |||
| Δ TBW (Itr) | High-Dose | 0.23 ± 5.06 | 0.5598 | −0.17 * | ns |
| Low-Dose | −0.32 ± 2.17 | −0.41 † | |||
| Placebo | 1.02 ± 4.03 | 0.14 # | |||
| Δ FFMH (%) | High-Dose | 1.51 ± 3.52 | 0.2750 | 0.38 * | ns |
| Low-Dose | 0.33 ± 2.06 | −0.08 † | |||
| Placebo | 0.47 ± 1.54 | 0.40 # | |||
| Δ Visceral fat (%) | High-dose | 2.33 ± 45.76 | 0.2281 | 0.26 * | ns |
| Low-Dose | −25.50 ± 52.61 | −0.25 † | |||
| Placebo | −11.64 ± 57.93 | 0.54 # | |||
| Δ Subcutaneous fat (%) | High-Dose | −68.14 ± 67.54 | 0.3664 | −0.25 * | ns |
| Low-Dose | −52.91 ± 71.20 | −0.05 † | |||
| Placebo | −49.50 ± 77.63 | −0.22 # | |||
| Δ Uric acid (mmol/L) | High-Dose | −0.68 ± 0.71 | 0.0009 | −0.73 * | * 0.0109 # 0.0016 |
| Low-Dose | −0.02 ± 0.55 | 0.16 † | |||
| Placebo | −0.12 ± 0.72 | −0.92 # | |||
| Δ TC (mgl/dL) | High-Dose | −16.00 ± 29.24 | 0.1164 | −0.36 * | ns |
| Low-Dose | −10.77 ± 22.63 | −0.21 † | |||
| Placebo | −5.52 ± 27.52 | −0.20 # | |||
| Δ HDL (mg/dL) | High-Dose | 2.20 ± 7.01 | 0.4023 | −0.13 * | ns |
| Low-Dose | 0.23 ± 13.79 | −0.26 † | |||
| Placebo | 3.16 ± 7.70 | 0.18 # | |||
| Δ TG (mg/dL) | High-Dose | −11.64 ± 39.43 | 0.7958 | −0.16 * | ns |
| Low-Dose | −10.23 ± 30.15 | −0.14 † | |||
| Placebo | −6.04 ± 31.46 | −0.04 # | |||
| Δ LDL (mg/dL) | High-Dose | −4.76 ± 12.21 | 0.6503 | −0.16 * | ns |
| Low-Dose | −8.23 ± 16.37 | −0.36 † | |||
| Placebo | −2.64 ± 14.55 | 0.24 # | |||
| Δ Glucose (mg/dL) | High-Dose | −7.67 ± 6.88 | 0.0033 | −0.61 * | * 0.0272 # 0.0043 |
| Low-Dose | −0.42 ± 11.17 | 0.08 † | |||
| Placebo | −1.40 ± 11.86 | −0.72 # | |||
| Δ INS (UI/L) | High-Dose | −8.01 ± 11.30 | 0.0001 | −0.83 * | * 0.0002 # 0.0155 |
| Low-Dose | −4.29 ± 6.40 | −0.68 † | |||
| Placebo | 0.30 ± 6.32 | −0.40 # | |||
| Δ HOMA-IR | High-Dose | −2.35 ± 2.77 | 0.0001 | −0.90 * | * 0.0005 # 0.0127 |
| Low-Dose | −0.99 ± 2.01 | −0.51 † | |||
| Placebo | 0.03 ± 1.86 | −0.54 # | |||
| Δ LPS (ng/mL) | High-Dose | −2.62 ± 3.26 | 0.0002 | −0.99 * | * 0.001 |
| Low-Dose | −0.33 ± 3.74 | −0.21 † | |||
| Placebo | 0.27 ± 1.60 | −0.62 # |
| Group | ||||||
|---|---|---|---|---|---|---|
| Correlation n = 25 | HD | LD | Placebo | |||
| r | p-Value | r | p-Value | r | p-Value | |
| Δ LPS and Δ waist circumference | 0.407 | 0.0436 | −0.229 | 0.261 | 0.107 | 0.6101 |
| Δ LPS and Δ INS | 0.272 | 0.1884 | 0.306 | 0.1284 | 0.220 | 0.2906 |
| Δ LPS and Δ HOMA-IR | 0.301 | 0.1434 | 0.400 | 0.0427 | 0.148 | 0.4811 |
| Δ LPS and Δ LDL | −0.100 | 0.6346 | −0.125 | 0.5433 | 0.237 | 0.2536 |
| Δ LPS and Δ uric acid | 0.296 | 0.1506 | −0.189 | 0.3543 | −0.486 | 0.0738 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Szulińska, M.; Łoniewski, I.; Van Hemert, S.; Sobieska, M.; Bogdański, P. Dose-Dependent Effects of Multispecies Probiotic Supplementation on the Lipopolysaccharide (LPS) Level and Cardiometabolic Profile in Obese Postmenopausal Women: A 12-Week Randomized Clinical Trial. Nutrients 2018, 10, 773. https://doi.org/10.3390/nu10060773
Szulińska M, Łoniewski I, Van Hemert S, Sobieska M, Bogdański P. Dose-Dependent Effects of Multispecies Probiotic Supplementation on the Lipopolysaccharide (LPS) Level and Cardiometabolic Profile in Obese Postmenopausal Women: A 12-Week Randomized Clinical Trial. Nutrients. 2018; 10(6):773. https://doi.org/10.3390/nu10060773
Chicago/Turabian StyleSzulińska, Monika, Igor Łoniewski, Saskia Van Hemert, Magdalena Sobieska, and Paweł Bogdański. 2018. "Dose-Dependent Effects of Multispecies Probiotic Supplementation on the Lipopolysaccharide (LPS) Level and Cardiometabolic Profile in Obese Postmenopausal Women: A 12-Week Randomized Clinical Trial" Nutrients 10, no. 6: 773. https://doi.org/10.3390/nu10060773
APA StyleSzulińska, M., Łoniewski, I., Van Hemert, S., Sobieska, M., & Bogdański, P. (2018). Dose-Dependent Effects of Multispecies Probiotic Supplementation on the Lipopolysaccharide (LPS) Level and Cardiometabolic Profile in Obese Postmenopausal Women: A 12-Week Randomized Clinical Trial. Nutrients, 10(6), 773. https://doi.org/10.3390/nu10060773






