The Role of Lipids in Human Milk and Infant Formulae
Abstract
1. Introduction
2. Lipid Quality and Role in Human Milk and Infant Formulae
2.1. Saturated and Monounsaturated Fatty Acids
2.2. Polyunsaturated Fatty Acids
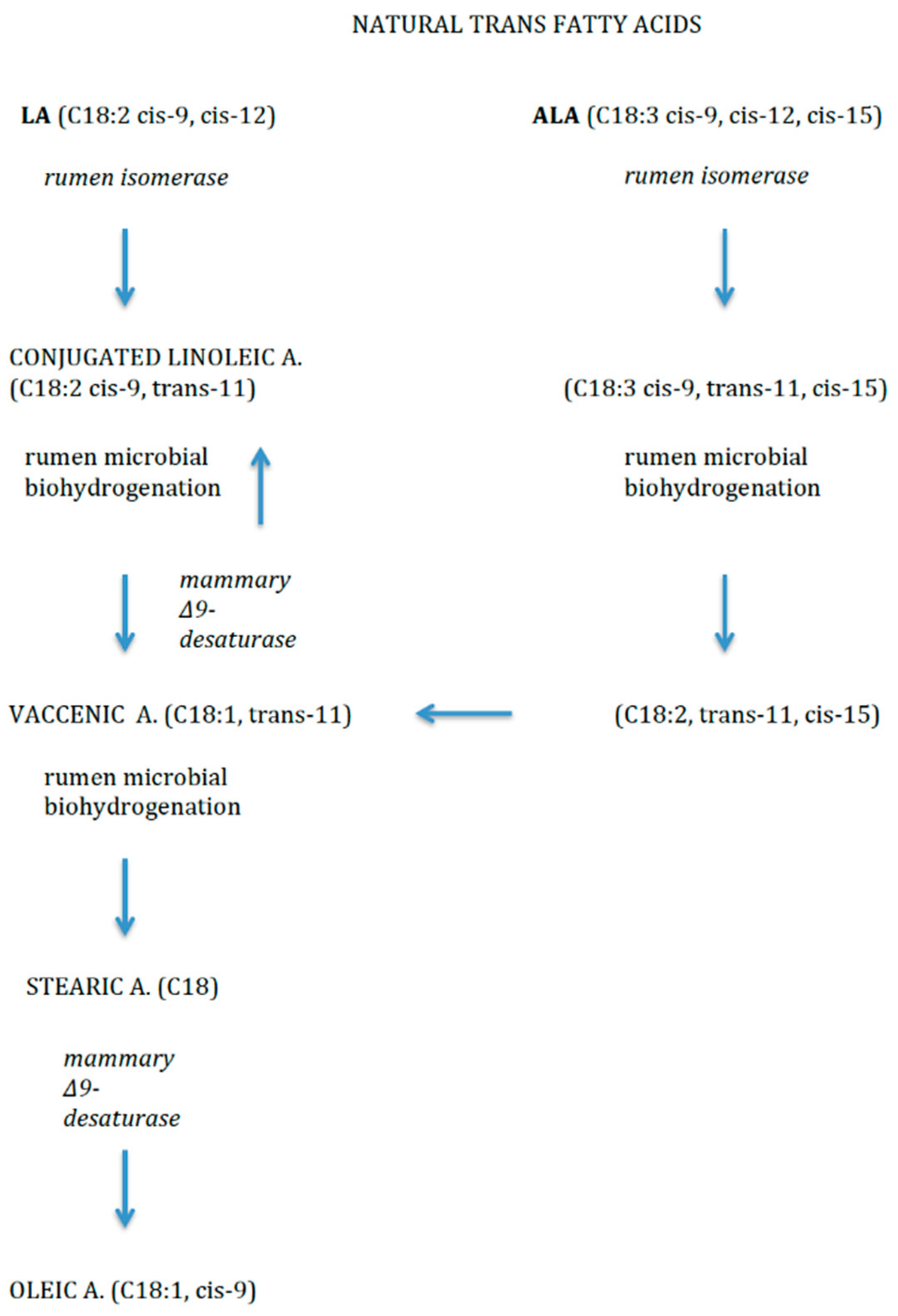
2.3. Trans Fatty Acids
3. Fats in Infant Formulae Intended for Medical Condition (According to the Regulation Eu n. 609/2013)
3.1. Preterm Formulae
3.2. Short Bowel Syndrome
3.3. Atopic Disorders
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Koletzko, B.; Agostoni, C.; Bergmann, R.; Ritzenthaler, K.; Shamir, R. Physiological aspects of human milk lipids and implications for infant feeding: A workshop report. Acta Paediatr. 2011, 100, 1405–1415. [Google Scholar] [CrossRef] [PubMed]
- Innis, S.M. Dietary triacylglycerol structure and its role in infant nutrition. Adv. Nutr. 2011, 2, 275–283. [Google Scholar] [CrossRef] [PubMed]
- Lauritzen, L.; Fewtrell, M.; Agostoni, C. Dietary arachidonic acid in perinatal nutrition: A commentary. Pediatr. Res. 2015, 77, 263–269. [Google Scholar] [CrossRef] [PubMed]
- Delplanque, B.; Gibson, R.; Koletzko, B.; Lapillonne, A.; Strandvik, B. Lipid Quality in Infant Nutrition: Current Knowledge and Future Opportunities. J. Pediatr. Gastroenterol. Nutr. 2015, 61, 8–17. [Google Scholar] [CrossRef] [PubMed]
- Gianni, M.L.; Roggero, P.; Baudry, C.; Fressange-Mazda, C.; Galli, C.; Agostoni, C.; le Ruyet, P.; Mosca, F. An infant formula containing dairy lipids increased red blood cell membrane Omega 3 fatty acids in 4 month-old healthy newborns: A randomized controlled trial. BMC Pediatr. 2018, 18, 53. [Google Scholar] [CrossRef] [PubMed]
- EFSA NDA Panel (EFSA Panel on Dietetic Products, Nutrition, and Allergies). Scientific Opinion on Dietary Reference Values for fats, including saturated fatty acids, polyunsaturated fatty acids, monounsaturated fatty acids, trans fatty acids, and cholesterol. EFSA J. 2010, 8, 1461. [Google Scholar] [CrossRef]
- EFSA NDA Panel (EFSA Panel on Dietetic Products, Nutrition, and Allergies). Scientific Opinion on the essential composition of infant and follow-on formulae. EFSA J. 2014, 12, 3760. [Google Scholar] [CrossRef]
- Goulet, O.; Olieman, J.; Ksiazyk, J.; Spolidoro, J.; Tibboe, D.; Köhler, H.; Yagci, R.V.; Falconer, J.; Grimble, G.; Beattie, R.M. Neonatal short bowel syndrome as a model of intestinal failure: Physiological background for enteral feeding. Clin. Nutr. 2013, 32, 162–171. [Google Scholar] [CrossRef] [PubMed]
- St-Onge, M.P.; Jones, P.J. Physiological effects of medium-chain triglycerides: Potential agents in the prevention of obesity. J. Nutr. 2002, 132, 329–332. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, M.; Funke, S.; Fink, M.; Demmelmair, H.; Turini, M.; Crozier, G.; Koletzko, B. Plasma fatty acids and [13C] linoleic acid metabolism in preterm infants fed a formula with medium-chain triglycerides. J. Lipid Res. 2003, 44, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Holman, R.T. Nutritional and metabolic interrelationships between fatty acids. Fed. Proc. 1964, 23, 1062–1067. [Google Scholar] [PubMed]
- Agostoni, C. Role of long-chain polyunsaturated fatty acids in the first year of life. J. Pediatr. Gastroenterol. Nutr. 2008, 47, S41–S44. [Google Scholar] [CrossRef] [PubMed]
- Agostoni, C.; Risé, P.; Marangoni, F. Membrane composition and cellular responses to fatty acid intakes and factors explaining the variation in response. Nestle Nutr. Inst. Workshop Ser. 2013, 77, 111–120. [Google Scholar] [PubMed]
- Chisaguano, A.M.; Montes, R.; Castellote, A.I.; Morales, E.; Julvez, J.; Vioque, J.; Sunyer, J.; López-Sabater, M.C. Elaidic, vaccenic, and rumenic acid status duringpregnancy: Association with maternal plasmatic LC-PUFAs andatopic manifestations in infants. Pediatr. Res. 2014, 76, 470–476. [Google Scholar] [CrossRef] [PubMed]
- Wijga, A.H.; van Houwelingen, A.C.; Kerkhof, M.; Tabak, C.; de Jongste, J.C.; Gerritsen, J.; Boshuizen, H.; Brunekreef, B.; Smit, H.A. Breast milk fatty acids and allergic disease in preschool children: The Prevention and Incidence of Asthma and Mite Allergy birth cohort study. J. Allergy Clin. Immunol. 2006, 117, 440–447. [Google Scholar] [CrossRef] [PubMed]
- Thijs, C.; Müller, A.; Rist, L.; Kummeling, I.; Snijders, B.E.; Huber, M.; van Ree, R.; Simões-Wüst, A.P.; Dagnelie, P.C.; van den Brandt, P.A. Fatty acids in breast milk and development of atopic eczema and allergic sensitisation in infancy. Allergy 2011, 66, 58–67. [Google Scholar] [CrossRef] [PubMed]
- Agostoni, C. Docosahexaenoic acid (DHA): From the maternal-foetal dyad to the complementary feeding period. Early Hum. Dev. 2010, 86, 3–6. [Google Scholar] [CrossRef] [PubMed]
- Luxwolda, M.F.; Kuipers, R.S.; Sango, W.S.; Kwesigabo, G.; Dijck-Brouwer, D.A.; Muskiet, F.A. A maternal erythrocyte DHA content of approximately 6 g% is the DHA status at which intrauterine DHA biomagnifications turns into bioattenuation and postnatal infant DHA equilibrium is reached. Eur. J. Nutr. 2012, 51, 665–675. [Google Scholar] [CrossRef] [PubMed]
- Kuipers, R.S.; Luxwolda, M.F.; Offringa, P.J.; Boersma, E.R.; Dijck-Brouwer, D.A.; Muskiet, F.A. Gestational age dependent changes of the fetal brain, liver and adipose tissue fatty acid compositions in a population with high fish intakes. Prostaglandins Leukot. Essent. Fatty Acids 2012, 86, 189–199. [Google Scholar] [CrossRef] [PubMed]
- Lapillonne, A.; Groh-Wargo, S.; Gonzalez, C.H.; Uauy, R. Lipid needs of preterm infants: Updated recommen-dations. J. Pediatr. 2013, 162, S37–S47. [Google Scholar] [CrossRef] [PubMed]
- Lindquist, S.; Hernell, O. Lipid digestion and absorption in early life: An update. Curr. Opin. Clin. Nutr. Metab. Care 2010, 13, 314–320. [Google Scholar] [CrossRef] [PubMed]
- Klenoff-Brumberg, H.L.; Genen, L.H. High versus low medium chain triglyceride content of formula for promoting short term growth of preterm infants. Cochrane Database Syst. Rev. 2003, 1, CD002777. [Google Scholar] [CrossRef]
- Łoś-Rycharska, E.; Kieraszewicz, Z.; Czerwionka-Szaflarska, M. Medium chain triglycerides (MCT) formulas in paediatric and allergological practice. Prz. Gastroenterol. 2016, 11, 226–231. [Google Scholar] [CrossRef] [PubMed]
- Carlson, S.E.; Werkman, S.H.; Peeples, J.M.; Cooke, R.J.; Tolley, E.A. Arachidonic acid status correlates with first year growth in preterm infants. Proc. Natl. Acad. Sci. USA 1993, 90, 1073–1077. [Google Scholar] [CrossRef] [PubMed]
- Willatts, P.; Forsyth, S.; Agostoni, C.; Casaer, P.; Riva, E.; Boehm, G. Effects of long-chain PUFA supplementation in infant formula on cognitive function in later childhood. Am. J. Clin. Nutr. 2013, 98, 536S–542S. [Google Scholar] [CrossRef] [PubMed]
- Smithers, L.G.; Gibson, R.A.; McPhee, A.; Makrides, M. Higher dose of docosahexaenoic acid in the neonatal period improves visual acuity of preterm infants: Results of a randomized controlled trial. Am. J. Clin. Nutr. 2008, 88, 1049–1056. [Google Scholar] [CrossRef] [PubMed]
- Fewtrell, M.S.; Abbott, R.A.; Kennedy, K.; Singhal, A.; Morley, R.; Caine, E.; Jamieson, C.; Cockburn, F.; Lucas, A. Randomized, double-blind trial of long-chain polyunsaturated fatty acid supplementation with fish oil and borage oil in preterm infants. J. Pediatr. 2004, 144, 471–479. [Google Scholar] [CrossRef] [PubMed]
- De Curtis, M.; Gasparrini, E. Latti Formulati in Commercio in Italia, Allegato “Cangurini di Pediatria” alla Rivista Pediatria (Anno 3, Numero 6); Il Pensiero Scientifico Editore: Roma, Italy, 2013; pp. 1–16. [Google Scholar]
- Olieman, J.F.; Penning, C.; Ijsselstijn, H.; Escher, J.C.; Joosten, K.F.; Hulst, M.J.; Tibboel, D. Enteral nutrition in children with short-bowel syndrome: Current evidence and recommendations for the clinicians. J. Am. Diet. Assoc. 2010, 110, 420–426. [Google Scholar] [CrossRef] [PubMed]
- Hattner, J.A.T. Infant formula update. Nutr. Focus 2006, 20, 1–12. [Google Scholar]
- Miller, M.; Burjonrappa, S. A review of enteral strategies in infant short bowel syndrome: Evidence-based or NICU culture? J. Pediatr. Surg. 2013, 48, 1099–1112. [Google Scholar] [CrossRef] [PubMed]
- Serrano, M.S.; Schmidt-Sommerfeld, E. Nutrition support of infants with short bowel syndrome. Nutrition 2002, 18, 966–970. [Google Scholar] [CrossRef]
- Jeppesen, P.B.; Mortensen, P.B. Colonic digestion and absorption of energy from carbohydrates and medium-chain fat in small bowel failure. J. Parenter. Enteral. Nutr. 1999, 23, 101–105. [Google Scholar] [CrossRef] [PubMed]
- Agostoni, C.; Terracciano, L.; Varin, E.; Fiocchi, A. The Nutritional Value of Proteinhydrolyzed Formulae. Crit. Rev. Food Sci. Nutr. 2016, 56, 65–69. [Google Scholar] [CrossRef] [PubMed]
- Mazzocchi, A.; Venter, C.; Maslin, K.; Agostoni, C. The Role of Nutritional Aspects in Food Allergy: Prevention and Management. Nutrients 2017, 9, 850. [Google Scholar] [CrossRef] [PubMed]
- Glaser, C.; Heinrich, J.; Koletzko, B. Role of FADS1 and FADS2 polymorphisms in polyunsaturated fatty acid metabolism. Metabolism 2010, 59, 993–999. [Google Scholar] [CrossRef] [PubMed]
- Kaila, M.; Salo, M.K.; Isolauri, E. Fatty acids in substitute formulas for cow’s milk allergy. Allergy 1999, 54, 174–177. [Google Scholar] [CrossRef] [PubMed]
- Munblit, D.; Peroni, D.; Boix-Amorós, A.; Hsu, P.S.; Van’t Land, B.; Gay, M.C.L.; Kolotilina, A.; Skevaki, C.; Boyle, R.J.; Collado, M.C.; et al. Human Milk and Allergic Diseases: An Unsolved Puzzle. Nutrients 2017, 9, 894. [Google Scholar] [CrossRef] [PubMed]
- Kumari, M.; Kozyrskyj, A.L. Gut microbial metabolism defines host metabolism: An emerging perspective in obesity and allergic inflammation. Obes. Rev. 2017, 18, 18–31. [Google Scholar] [CrossRef] [PubMed]
- Del Giudice, M.M.; D’Auria, E.; Peroni, D.; Palazzo, S.; Radaelli, G.; Comberiati, P.; Galdo, F.; Maiello, N.; Riva, E. Flavor, relative palatability and components of cow’s milk hydrolysed formulas and amino acid-based formula. Ital. J. Pediatr. 2015, 41, 42. [Google Scholar] [CrossRef] [PubMed]


| Formula | Fat (% En tot) | TFA (g) | Saturated (g) | MUFAs (g) | PUFAs (g) | MCTs (g) | LA (mg) | ALA (mg) | EPA (mg) | ARA (mg) | DHA (mg) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Pre Nidina ospedale (100 mL) | 44.4 | 4 | 1.7 | 1.6 | 560 | 80 | 16 | 16 | |||
| Pre Nidina casa (100 mL 14.4% w/v) | 46.8 | 3.8 | 1 | 373.1 | 75.2 | 14.1 | 14.4 | ||||
| Pre-Humana (100 mL 13.5% w /v) | 43.6 | 3.2 | 1.1 | 1.5 | 0.7 | ||||||
| Humana 0 (100 mL 15.4% w/v) | 46.7 | 4 | 1.4 | 1.8 | 0.9 | 739 | 80 | 17.6 | 17.6 | ||
| Humana 0-VLB (100 mL 15.4% w/v) | 41.7 | 3.9 | 1.3 | 1.9 | 0.7 | 0.6 | 549 | 69 | 21.5 | 21.5 | |
| Plasmon Pre-0 (100 mL) | 46 | 4.2 | 0.8 | 25.1 | 14.7 | ||||||
| Plasmon 0 (100 mL) | 44.4 | 3.9 | 1.6 | 1.4 | 0.9 | 0.6 | 770 | 100 | 23.2 | 13.5 | |
| Mellin 0 | 43.8 | 3.9 | 0.2 | 2.9 | 17.7 | 13.6 | |||||
| Mellin 0 POST (100 mL al 15.5% w/v) | 48 | 4 | 1.7 | 1.7 | 0.7 | 0.8 | 513 | 71 | 2.5 | 17 | 13 |
| Formulat Pre-0 (100 ml) | 44.5 | 4.1 | 1.25 | 1.7 | 0.8 | 670 | 97 | 28 | 21.5 | ||
| Formulat 0 (100 mL) | 45 | 4.1 | 1.4 | 1.7 | 0.8 | 670 | 97 | 24.6 | 21.3 | ||
| Pre- Aptamil (100 mL 14.4% w/v) | 48 | 4 | 1.5 | 1.8 | 0.6 | 0,8 | 490 | 76.7 | 2.3 | 17.4 | 13.2 |
| Similac 24 (100 mL) | 48.8 | 4.4 | 568 | 8 | 8 | ||||||
| Miltina 0 (100 mL) | 48 | 4 | 1.1 | 8 | 8 | ||||||
| Miltina 0 post (100 mL) | 48 | 4 | 0.8 | 2.5 | 17 | 13 | |||||
| Blemil Plus (100 mL 15.9% w/v) | 45.5 | 4.1 | 0.5 | 611 | 49 | 24.6 | 16.4 | ||||
| Eveil formula 0 (100 mL) | 45 | 4.1 | 0.6 | 24 | 12 | ||||||
| Nutribén Pre (100 mL) | 48.3 | 4.3 | 0.4 | 20 | 20 |
| Formula | Fat (% En tot) | TFA Total (g) | Saturated (g) | MUFAs (g) | PUFAs (g) | LCTs (g) | MCTs (g) | Omega-3 (g) | LA (mg) | ALA (mg) | ɣ-LA (mg) | ARA (mg) | DHA (mg) | EPA (mg) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Elemental 028 extra * | 36 | 3.5 | 1.35 | 1.6 | 0.45 | 2.275 | 1.225 | |||||||
| Monogen (100 mL 16.8% w/v) | 26.5 | 2.2 | 1.9 | 0.4 | 1.3 | 0.35 | 1.85 | |||||||
| Peptamen Junior PHGG (100 mL) | 32 | 3.6 | 2 | 0.5 | 1.1 | 1.5 | 0.16 | |||||||
| Alfaré (100 mL 13.5% w/v) | 43 | 3.4 | 2.1 | 1.3 | 480 | 60 | 17 | 5.7 | ||||||
| Neocate LCP (100 mL 13.8% w/v) | 46 | 3.4 | 1.2 | 1.3 | 0.66 | 3.26 | 0.136 | 579 | 57.8 | 11.3 | 11.3 | |||
| Infatrini Peptisorb (100 mL 14% w/v) | 49 | 5.4 | 3.4 | 2.7 | 421 | 83.9 | 15.8 | 15.7 | ||||||
| Nutrini Peptisorb (100 mL 15% w/v) | 35 | 3.9 | 2.2 | 0.5 | 1.2 | 1.8 | 1010 | 92.7 | ||||||
| Humana Disanal (100 mL 14.5% w/v) | 29.5 | 2.1 | 0.8 | 0.9 | 0.4 | 374 | 53 | |||||||
| Aptamil- pepti Junior (100 mL 12.8% w/v) | 47.7 | 3.5 | 1.9 | 0.81 | 0.6 | 0.02 | 1.71 | 486.5 | 94.85 | 0.35 | 7 | 7 | 1.4 | |
| Pantolac 1 (100 mL 13.7% w/v) | 46.4 | 3.4 | 1.5 | 1.3 | 0.5 | 418 | 84 | 11 | 6.4 | 1.4 | ||||
| Pantolac 2 (100 mL 13.8% w/v) | 40.3 | 3 | 1.4 | 0.517 | 0.05 | 7.9 | 4.4 | |||||||
| Vivonex Pediatric (100 mL 22% w/v) | 30.4 | 2.7 | 1.8 | 382 |
| Formula | Fat (% En tot) | TFA (g) | Saturated (g) | MUFAs (g) | PUFAs (g) | LCTs (g) | MCTs (g) | LA (mg) | ALA (mg) | ɣ-LA (mg) | ARA (mg) | EPA (mg) | DHA (mg) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mellin Polilat 1 (100 mL 13.6% w/v) | 47 | 3.5 | 1.6 | 1.3 | 0.6 | 455 | 83 | 12 | 2.3 | 11 | |||
| Mellin Polilat 2 (100 mL 14.4% w/v) | 41 | 3.1 | 1.4 | 1.2 | 0.5 | 411 | 75 | 9 | 1.9 | 9 | |||
| Nidina H.A 1 (100 mL 13.1% w/v) | 45.7 | 3.4 | 1 | 536 | 65 | 7.8 | 7.8 | ||||||
| Nidina H.A 2 (100 mL 13.6% p/v) | 41.6 | 3.1 | 0.8 | 0.6 | 466.6 | 57 | 5.7 | 5.7 | |||||
| Althéra (100 mL 13.2% w/v) | 45.6 | 3.4 | 1.3 | 500 | 50 | 7.4 | 7.4 | ||||||
| Alfaré (100 mL 13.5% w/v) | 43 | 3.4 | 1.3 | 480 | 60 | 5.7 | |||||||
| Pregestemil Lipil (100 mL) | 50.3 | 3.8 | 2.1 | 23 | 11.5 | ||||||||
| Neocate Spoon (100 g powder) | 36 | 18.8 | 6.9 | 7.5 | 3.5 | 18 | 0.8 | 3222 | 322 | ||||
| Neocate LCP (100 mL 13.8% w/v) | 46 | 3.4 | 1.2 | 1.3 | 0.66 | 3.3 | 0.1 | 579 | 57.8 | 11.3 | 11.3 | ||
| Neocate Junior (100 mL 21.1% w/v) | 42 | 4.6 | 2 | 1.6 | 0.84 | 1.61 | |||||||
| Humana AT 1 (100 mL 14% w/v) | 46.9 | 3.6 | 1.4 | 1.4 | 0.7 | 595 | 52 | 13 | 13 | ||||
| Humana AT 2 (100 mL 13.4% w/v) | 46.4 | 3.4 | 1.4 | 1.3 | 0.6 | 569 | 50 | 0.7 | 13 | 13 | |||
| Aptamil pepti 1 (100 mL 13.6% w/v) | 47 | 3.5 | 1.9 | 1.29 | 0.62 | 2.73 | 0.13 | 479.5 | 87.85 | 16.8 | 12 | 2.45 | 11 |
| Aptamil pepti 2 (100 mL 14.4% w/v) | 41 | 3.1 | 1.4 | 1.14 | 4.36 | 2.41 | 0.11 | 479.5 | 87.85 | 0.7 | 9 | 2.1 | 9 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mazzocchi, A.; D’Oria, V.; Cosmi, V.D.; Bettocchi, S.; Milani, G.P.; Silano, M.; Agostoni, C. The Role of Lipids in Human Milk and Infant Formulae. Nutrients 2018, 10, 567. https://doi.org/10.3390/nu10050567
Mazzocchi A, D’Oria V, Cosmi VD, Bettocchi S, Milani GP, Silano M, Agostoni C. The Role of Lipids in Human Milk and Infant Formulae. Nutrients. 2018; 10(5):567. https://doi.org/10.3390/nu10050567
Chicago/Turabian StyleMazzocchi, Alessandra, Veronica D’Oria, Valentina De Cosmi, Silvia Bettocchi, Gregorio Paolo Milani, Marco Silano, and Carlo Agostoni. 2018. "The Role of Lipids in Human Milk and Infant Formulae" Nutrients 10, no. 5: 567. https://doi.org/10.3390/nu10050567
APA StyleMazzocchi, A., D’Oria, V., Cosmi, V. D., Bettocchi, S., Milani, G. P., Silano, M., & Agostoni, C. (2018). The Role of Lipids in Human Milk and Infant Formulae. Nutrients, 10(5), 567. https://doi.org/10.3390/nu10050567






