Impaired Hematologic Status in Relation to Clinical Outcomes among HIV-Infected Adults from Uganda: A Prospective Cohort Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population and Design
2.2. Measurements
2.2.1. Primary Determinants: Baseline Serum Ferritin, Baseline Anemia and Anemia Persistence
2.2.2. Anemia Persistence
- (i)
- Persistently anemia free: no anemia in any observation period.
- (ii)
- Completely Resolved Anemia: mild or moderate/severe anemia at enrollment only without evidence of anemia in any follow-up interval.
- (iii)
- Incompletely Resolved Anemia: baseline moderate/severe anemia that was down-modulated to mild in ≥1 follow-up interval.
- (iv)
- Incident moderate/severe or sustained mild anemia: baseline anemia that became moderate/severe in one of three follow-up intervals or mild baseline anemia that was sustained in ≥2 follow-up intervals.
- (v)
- Mild Anemia: one episode of mild anemia at baseline or developed mild anemia in one of the follow-up intervals.
- (vi)
- Progressive or Persistent moderate/severe Anemia: baseline moderate/severe anemia that was sustained in ≥2 assessment intervals or mild baseline anemia that progressed to moderate/severe in ≥1 follow-up intervals.
2.2.3. Outcomes
- (i)
- Immune function: cluster of differentiation 4 (CD4) T-helper cell count: absolute T-cell lymphocyte count in cells/micro liter was measured using a FACS Calibur flow cytometer (Becton-Dickinson, San Jose, CA, USA).
- (ii)
- Body mass index (BMI): was calculated as the ratio of weight (in kilograms) to squared height (in meters).
- (iii)
- QOL: assessed with the Medical Outcomes Study HIV Health Survey translated and culturally adapted for the study area [24]. As described previously [25], overall QOL score was the sum of scores for activities of daily living, cognitive function, emotional and physical health subscales. Overall score was linearly transformed so that the highest score would be 100.
- (iv)
- Death/hospitalization: Occurrence of hospitalization or death event was, for each participant, a composite endpoint to enhance statistical efficiency and provide a more complete picture of serious adverse clinical outcomes among PLWHA, which should become rarer in the HAART era. Censoring was at date of first hospitalization or death. Participants that experienced both events were censored at hospitalization.
2.3. Potential Confounders: Clinical, Socio-Demographic and Behavioral Characteristics
2.4. Statistical Analysis
2.5. Consent Process/Ethical Approval
3. Results
3.1. Baseline Characteristics
3.2. Association between Baseline Ferritin Status and Immune Recovery, BMI and QOL Outcomes
3.3. Baseline Anemia/Anemia Severity and Change in Immune Recovery, Weight Gain, QOL
3.4. Association between Anemia Persistence, Immunologic Status and QOL
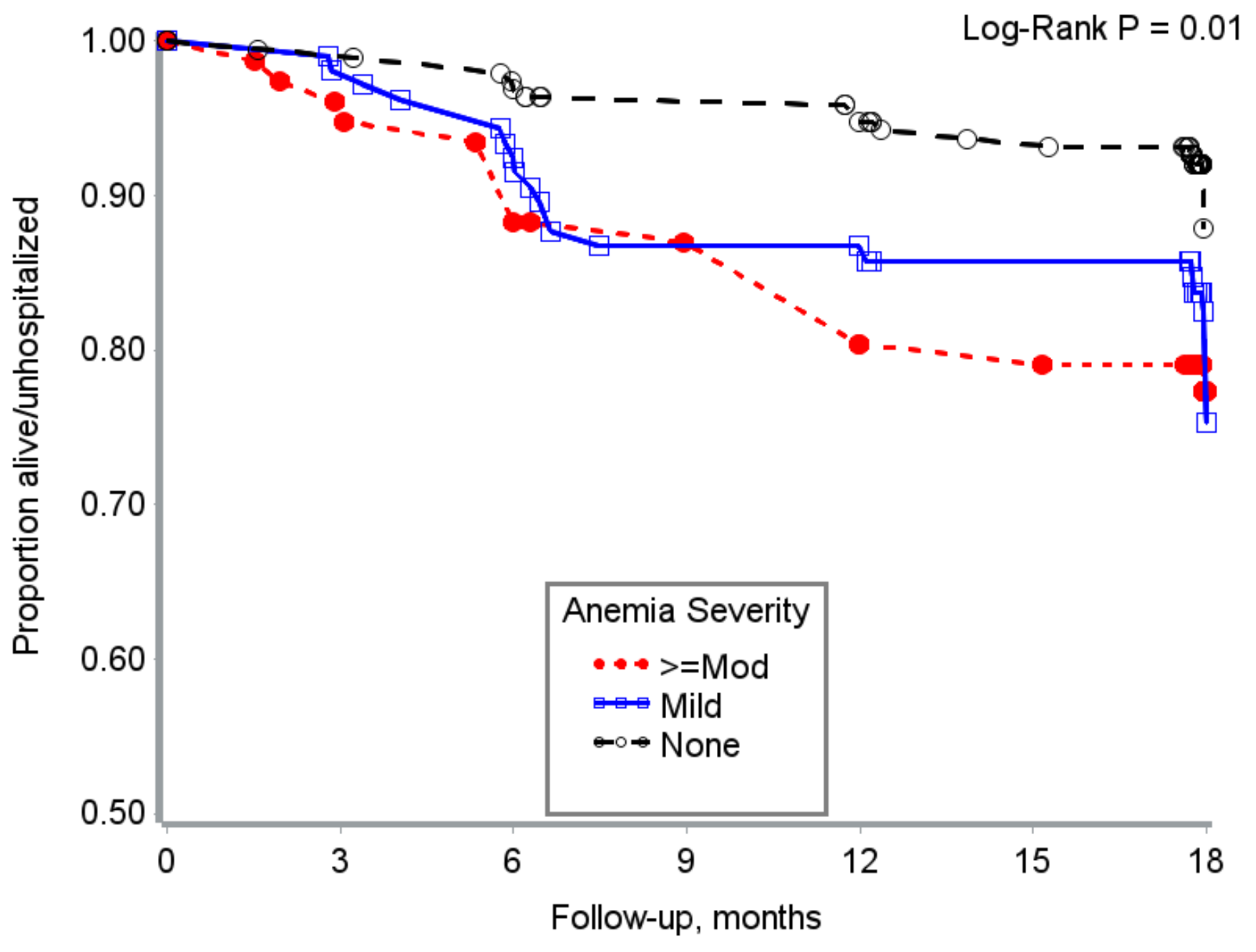
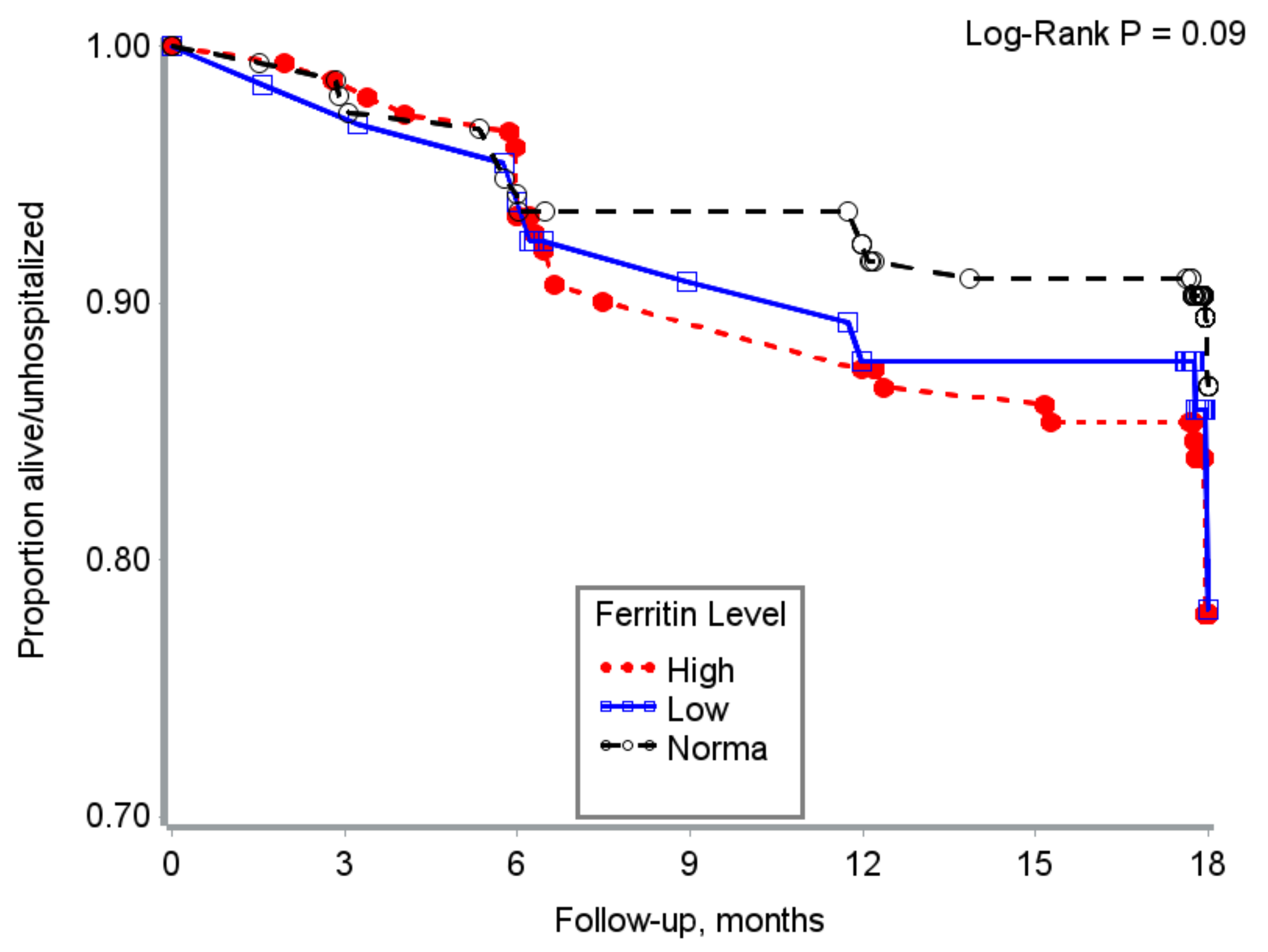
3.5. Time to Hospitalization/Death
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kerkhoff, A.D.; Wood, R.; Cobelens, F.G.; Gupta-Wright, A.; Bekker, L.G.; Lawn, S.D. Resolution of anaemia in a cohort of HIV-infected patients with a high prevalence and incidence of tuberculosis receiving antiretroviral therapy in South Africa. BMC Infect. Dis. 2014, 14, 3860. [Google Scholar] [CrossRef] [PubMed]
- Obirikorang, C.; Issahaku, R.G.; Osakunor, D.N.M.; Osei-Yeboah, J. Anaemia and iron homeostasis in a cohort of HIV-infected patients: A cross-sectional study in Ghana. AIDS Res. Treat. 2016, 2016, 1623094. [Google Scholar] [CrossRef] [PubMed]
- Belperio, P.S.; Rhew, D.C. Prevalence and outcomes of anemia in individuals with human immunodeficiency virus: A systematic review of the literature. Am. J. Med. 2004, 116 (Suppl. 7A), 27S–43S. [Google Scholar] [CrossRef] [PubMed]
- Drakesmith, H.; Chen, N.; Ledermann, H.; Screaton, G.; Townsend, A.; Xu, X.N. HIV-1 Nef down-regulates the hemochromatosis protein HFE, manipulating cellular iron homeostasis. Proc. Natl. Acad. Sci. USA 2005, 102, 11017–11022. [Google Scholar] [CrossRef] [PubMed]
- Armitage, A.E.; Stacey, A.R.; Giannoulatou, E.; Marshall, E.; Sturges, P.; Chatha, K.; Smith, N.M.; Huang, X.; Xu, X.; Pasricha, S.R.; et al. Distinct patterns of hepcidin and iron regulation during HIV-1, HBV, and HCV infections. Proc. Natl. Acad. Sci. USA 2014, 111, 12187–12192. [Google Scholar] [CrossRef] [PubMed]
- Minchella, P.A.; Armitage, A.E.; Darboe, B.; Jallow, M.W.; Drakesmith, H.; Jaye, A.; Prentice, A.M.; McDermid, J.M. Elevated hepcidin is part of a complex relation that links mortality with iron homeostasis and anemia in men and women with HIV infection. J. Nutr. 2015, 145, 1194–1201. [Google Scholar] [CrossRef] [PubMed]
- Quiros-Roldan, E.; Castelli, F.; Lanza, P.; Pezzoli, C.; Vezzoli, M.; Biasiotto, G.; Zanella, I. The impact of antiretroviral therapy on iron homeostasis and inflammation markers in HIV-infected patients with mild anemia. J. Transl. Med. 2017, 15, 256. [Google Scholar] [CrossRef] [PubMed]
- Traoré, H.N.; Meyer, D. The effect of iron overload on in vitro HIV-1 infection. J. Clin. Virol. 2004, 31 (Suppl. 1), 92–98. [Google Scholar] [CrossRef] [PubMed]
- Afacan, Y.E.; Hasan, M.S.; Omene, J.A. Iron deficiency anemia in HIV infection: Immunologic and virologic response. J. Natl. Med. Assoc. 2002, 94, 73–77. [Google Scholar] [PubMed]
- López-Calderón, C.; Palacios, R.; Cobo, A.; Nuño, E.; Ruiz, J.; Márquez, M.; Santos, J. Serum ferritin in HIV-positive patients is related to immune deficiency and inflammatory activity. Int. J. STD AIDS 2015, 26, 393–397. [Google Scholar] [CrossRef] [PubMed]
- Adamson, J.W. Iron deficiency and other hypoproliferative anemias. Harrisons Princ. Intern. Med. 2012, 1, 844–851. [Google Scholar]
- Wisaksana, R.; Sumantri, R.; Indrati, A.R.; Zwitser, A.; Jusuf, H.; de Mast, Q.; van Crevel, R.; Van Der Ven, A. Anemia and iron homeostasis in a cohort of HIV-infected patients in Indonesia. BMC Infect. Dis. 2011, 11, 213. [Google Scholar] [CrossRef] [PubMed]
- Makubi, A.; Okuma, J.; Spiegelman, D.; Hawkins, C.; Darling, A.M.; Jackson, E.; Mugusi, F.; Chalamilla, G.; Fawzi, W. Burden and determinants of severe anemia among HIV-infected adults: Results from a large urban HIV program in Tanzania, East Africa. J. Int. Assoc. Provid. AIDS Care 2015, 14, 148–155. [Google Scholar] [CrossRef] [PubMed]
- Bereket, D.; Bezatu, M.; Tadesse, A. Survival and determinants of mortality in adult HIV/AIDS patients initiating antiretroviral therapy in Somali region, Eastern Ethiopia. J. AIDS Clin. Res. 2014, 5, 327. [Google Scholar]
- McDermid, J.M.; Jaye, A.; van der Loeff, M.F.S.; Todd, J.; Bates, C.; Austin, S.; Jeffries, D.; Awasana, A.A.; Whittle, H.C.; Prentice, A.M. Elevated iron status strongly predicts mortality in West African adults with HIV infection. JAIDS J. Acquir. Immune Defic. Syndr. 2007, 46, 498–507. [Google Scholar] [CrossRef] [PubMed]
- Koethe, J.R.; Blevins, M.; Nyirenda, C.; Kabagambe, E.K.; Shepherd, B.E.; Wester, C.W.; Zulu, I.; Chiasera, J.M.; Mulenga, L.B.; Mwango, A.; et al. Nutrition and inflammation serum biomarkers are associated with 12-week mortality among malnourished adults initiating antiretroviral therapy in Zambia. J. Int. AIDS Soc. 2011, 14, 19. [Google Scholar] [CrossRef] [PubMed]
- Raza, M.; Amanat, S.T.; Hussain, T.; Shakoor, H.A.; Asghar, M. Latent iron deficiency and iron deficiency anaemia in hypoferritinemic patients. J. Rawalpindi Med. Coll. 2014, 18, 139–141. [Google Scholar]
- Camaschella, C. Iron-deficiency anemia. N. Engl. J. Med. 2015, 372, 1832–1843. [Google Scholar] [CrossRef] [PubMed]
- Al-Jafar, H.A. HWA: Hypoferritinemia without anemia a hidden hematology disorder. J. Fam. Med. Prim. Care 2017, 6, 69–72. [Google Scholar] [CrossRef] [PubMed]
- Guwatudde, D.; Ezeamama, A.E.; Bagenda, D.; Kyeyune, R.; Wabwire-Mangen, F.; Wamani, H.; Mugusi, F.; Spiegelman, D.; Wang, M.; Manabe, Y.C.; et al. Multivitamin supplementation in HIV infected adults initiating antiretroviral therapy in Uganda: The protocol for a randomized double blinded placebo controlled efficacy trial. BMC Infect. Dis. 2012, 12, 304. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). Serum Ferritin Concentrations for the Assessment of Iron Status and Iron Deficiency in Populations. Vitamin and Mineral Nutrition Information System; World Health Organization: Geneva, Switzerland, 2011. [Google Scholar]
- Erhardt, J.G.; Estes, J.E.; Pfeiffer, C.M.; Biesalski, H.K.; Craft, N.E. Combined measurement of ferritin, soluble transferrin receptor, retinol binding protein, and C-reactive protein by an inexpensive, sensitive, and simple sandwich enzyme-linked immunosorbent assay technique. J. Nutr. 2004, 134, 3127–3132. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). Haemoglobin Concentrations for the Diagnosis of Anaemia and Assessment of Severity; V.a.M.N.I.S; Department of Nutrition for Health and Development, World Health Organization: Geneva, Switzerland, 2011. [Google Scholar]
- Stangl, A.L.; Bunnell, R.; Wamai, N.; Masaba, H.; Mermin, J. Measuring quality of life in rural Uganda: Reliability and validity of summary scores from the medical outcomes study HIV health survey (MOS-HIV). Qual. Life Res. 2012, 21, 1655–1663. [Google Scholar] [CrossRef] [PubMed]
- Ezeamama, A.E.; Woolfork, M.N.; Guwatudde, D.; Bagenda, D.; Manabe, Y.C.; Fawzi, W.W.; Fawzi, M.C.S. Depressive and anxiety symptoms predict sustained quality of life deficits in HIV-Positive Ugandan adults despite antiretroviral therapy: A prospective cohort study. Medicine (Baltimore) 2016. [Google Scholar] [CrossRef] [PubMed]
- Ezeamama, A.E.; Guwatudde, D.; Wang, M.; Bagenda, D.; Brown, K.; Kyeyune, R.; Smith, E.; Wamani, H.; Manabe, Y.C.; Fawzi, W.W. High perceived social standing is associated with better health in HIV-infected Ugandan adults on highly active antiretroviral therapy. J. Behav. Med. 2016, 39, 453–464. [Google Scholar] [CrossRef] [PubMed]
- Akilimali, P.Z.; Kashala-Abotnes, E.; Musumari, P.M.; Kayembe, P.K.; Tylleskar, T.; Mapatano, M.A. Predictors of persistent anaemia in the first year of antiretroviral therapy: A retrospective cohort study from Goma, the Democratic Republic of Congo. PLoS ONE 2015, 10, e0140240. [Google Scholar] [CrossRef] [PubMed]
- Isanaka, S.; Aboud, S.; Mugusi, F.; Bosch, R.J.; Willett, W.C.; Spiegelman, D.; Duggan, C.; Fawzi, W.W. Iron status predicts treatment failure and mortality in tuberculosis patients: A prospective cohort study from Dar es Salaam, Tanzania. PLoS ONE 2012, 7, e37350. [Google Scholar] [CrossRef] [PubMed]
- Boom, J.; Kösters, E.; Duncombe, C.; Kerr, S.; Hirschel, B.; Ruxrungtham, K.; De Mast, Q.; Kosalaraksa, P.; Ulbolyam, S.; Jupimai, T.; et al. Ferritin levels during structured treatment interruption of highly active antiretroviral therapy. HIV Med. 2007, 8, 388–395. [Google Scholar] [CrossRef] [PubMed]
- Abrams, D.I.; Steinhart, C.; Frascino, R. Epoetin alfa therapy for anaemia in HIV-infected patients: Impact on quality of life. Int. J. STD AIDS 2000, 11, 659–665. [Google Scholar] [CrossRef] [PubMed]
- Brudevold, R.; Hole, T.; Hammerstrom, J. Hyperferritinemia is associated with insulin resistance and fatty liver in patients without iron overload. PLoS ONE 2008, 3, e3547. [Google Scholar] [CrossRef] [PubMed]
- Daka, D.; Lelissa, D.; Amsalu, A. Prevalence of anaemia before and after the initiation of antiretroviral therapy at ART center of Hawassa University Referral Hospital, Hawassa, South Ethiopia. Sch. J. Med. 2013, 3, 1–6. [Google Scholar]

| Characteristics | Overall | Low Ferritin n = 68 | Normal Ferritin n = 169 | High Ferritin n = 161 | p-Value |
|---|---|---|---|---|---|
| n (%) | n (%) | n (%) | n (%) | ||
| Age (years) | <0.01 | ||||
| 18–29 | 98 (24.6) | 26 (38.2%) | 50 (29.6%) | 22 (13.7%) | |
| 30–35 | 104 (26.1) | 20 (29.4%) | 41 (24.3%) | 43 (26.7%) | |
| 36–41 | 102 (25.6) | 15 (22.1%) | 39 (23.1%) | 48 (29.8%) | |
| 42+ | 94 (23.6) | 7 (10.3%) | 39 (23.1%) | 48 (29.8%) | |
| Female | 275 (69.1) | 68 (100.0%) | 134 (79.3%) | 73 (45.3%) | <0.01 |
| Clinical Measures | |||||
| High C-reactive protein (CRP) | 55 (13.9) | 4 (5.97%) | 15 (8.88%) | 36 (22.50%) | <0.01 |
| HAART naïve | 199 (50) | 36 (52.94%) | 95 (56.21%) | 68 (42.24%) | 0.03 |
| Anemia Severity | 0.03 | ||||
| None | 204 (51.3) | 44 (64.7) | 88 (52.1) | 72 (44.7) | |
| Mild | 113 (28.4) | 10 (14.7) | 48 (28.4) | 55 (34.2) | |
| Moderate/Severe | 81 (20.3) | 14 (20.6) | 33(19.5) | 34 (21.1) | |
| Behavioral Factors | |||||
| Ever smoked cigarette | 69 (17.3) | 0 (0.0%) | 26 (15.4%) | 43 (26.7%) | <0.01 |
| Current Alcohol use | 0.61 | ||||
| Never Used | 84 (21.1) | 16 (23.5%) | 40 (23.7%) | 28 (17.4%) | |
| Former User | 230 (57.8) | 37 (54.4%) | 97 (57.4%) | 96 (59.6%) | |
| Current User | 84 (21.1) | 15 (22.1%) | 32 (18.9%) | 37 (23.0%) | |
| Baseline Multivitamin Use | 90 (22.6) | 17 (25.0%) | 34 (20.1%) | 39 (24.2%) | 0.59 |
| BMI (kg/m2) | 0.07 | ||||
| Underweight (BMI < 18.5) | 22 (5.5) | 2 (2.9%) | 8 (4.7%) | 12 (7.5%) | |
| Normal (18.5 ≤ BMI < 25) | 262 (65.8) | 42 (61.8%) | 103 (61.0%) | 117 (72.7%) | |
| Overweight (25 ≤ BMI < 30) | 72 (18.1) | 15 (22.1%) | 36 (21.3%) | 21 (13.0%) | |
| Obese (BMI ≥ 30) | 42 (10.6) | 9 (13.2%) | 22 (13.0%) | 11 (6.8%) | |
| Vitamin D Deficiency | 0.57 | ||||
| Deficient | 67 (16.9) | 12 (17.9%) | 25 (14.8%) | 30 (18.8%) | |
| Insufficient | 239 (60.4) | 42 (62.7%) | 99 (58.6%) | 98 (61.3%) | |
| Sufficient | 90 (22.7) | 13 (19.4%) | 45 (26.6%) | 32 (20.0%) | |
| Self-rated Health | 0.03 | ||||
| Good, Very good or Excellent | 180 (45.2) | 21 (30.9%) | 83 (49.1%) | 76 (47.2%) | |
| Education | 0.88 | ||||
| <Primary | 165 (41.6) | 26 (38.2%) | 73 (18.4%) | 66 (41.0%) | |
| Primary completed | 54 (13.6) | 13 (19.1%) | 21 (12.5%) | 20 (12.4%) | |
| Some O’level | 78 (19.7) | 12 (17.7%) | 32 (19.1%) | 34 (21.1%) | |
| O’level or higher | 100 (25.2) | 17 (25.0%) | 42 (25.0%) | 41 (25.5%) | |
| Unemployed/No Income | 52 (13.1) | 11 (16.18%) | 21 (12.43%) | 20 (12.4%) | 0.77 |
| Continuous Measures | Mean (standard deviation (SD)) | Mean (SD) | Mean (SD) | Mean (SD) | |
| Age (in years) | 35.8 (9.0) | 31.5 (7.1) | 35.1 (8.4) | 38.1 (9.5) | <0.01 |
| BMI (in kg/m2) | 23.8 (4.4) | 24.5 (4.2) | 24.4 (4.8) | 22.8 (3.8) | <0.01 |
| QOL Score | 91.2 (9.9) | 90.4 (10.2) | 91.3 (9.9) | 91.5 (10) | 0.77 |
| CD4 cell count (cells/µL) | 149 (96) | 161 (103) | 152.1 (94) | 140.7 (95) | 0.25 |
| Hemoglobin (mg/dL) | 12.3 (1.9) | 12.3 (1.76) | 12.3 (1.8) | 12.2 (1.9) | 0.68 |
| Vitamin D (ng/mL) | 26.7 (7.0) | 26.0 (7.5) | 26.4 (6.8) | 27.3 (7.1) | 0.37 |
| Outcome Hematologic Status Indicator | Month 0 Mean ± SE Diff (95% CI) | Month 6 Mean ± SE Diff (95% CI) | Month 12 Mean ± SE Diff (95% CI) | Month 18 Mean ± SE Diff (95% CI) | p-Value Group × Time | |
|---|---|---|---|---|---|---|
| Absolute CD4 | Baseline Serum Ferritin | |||||
| Normal † | 152.7 ± 6.8 | 231 ± 8.0 | 251 ± 9.4 | 287 ± 10.9 | 0.78 | |
| High | −7.3 (−29.0, 14.4) | 0.3 (−16.4, 17.0) | 15.3 (−4.9, 35.4) | 16.3 (−12.8, 45.5) | ||
| Low | −7.0 (−34.7, 20.6) | −6.6 (−30.1, 16.9) | 8.1 (−19.1, 35.2) | −4.0 (−37.4, 29.5) | ||
| Baseline Anemia Severity | ||||||
| No Anemia | 148 ± 6.8 | 227 ± 8.0 | 261 ± 9.4 | 293 ± 10.9 | 0.11 | |
| Mild | 15 (−8.6, 38.4) | 20 (−8, 39) | −1 (−31, 30) | −8 (−44, 27) | ||
| Moderate/Severe | −14 (−36.4, 9.0) | −9 (−37, 9) | −7 (−41, 28) | 9 (−34, 52) | ||
| BMI | Baseline Serum Ferritin | |||||
| Normal † | 23.7 ± 0.3 | 24.3 ± 0.2 | 24.3 ± 0.2 | 24.8 ± 0.2 | 0.04 | |
| High vs. Normal | 0.0 (−0.4, 0.4) | −0.1 (−0.4, 0.2) | 0.5 (0.2, 0.8) | 0.3 (−0.1, 0.7) | ||
| Low vs. Normal | −0.1 (−0.6, 0.4) | −0.1(−0.4, 0.3) | 0.2 (−0.2, 0.6) | 0.4 (−0.2, 0.9) | ||
| Baseline Anemia Severity | ||||||
| No Anemia | 24.3 ± 0.3 | 24.5 ± 0.3 | 24.± 0.3 | 25.0 ± 0.3 | ||
| Mild | − 0.9 (−1.9, 0.1) | −0.5 (−1.5, 0.5) | −0.3 (−1.3, 0.7) | −0.3 (−1.3, 0.8) | <0.01 | |
| Moderate/Severe | −1.3 (−2.4, −0.3) | −0.8 (−1.8, 0.2) | −0.4 (−1.4, 0.6) | 0.1 (−1.0, 1.2) | ||
| QOL | Baseline Serum Ferritin | |||||
| Normal † | 91.4 ± 0.7 | 98.0 ± 0.6 | 97.6 ± 0.6 | 98.9 ± 0.6 | 0.01 | |
| High vs. normal | −0.2 (−2.2, 1.8) | −1.6 (−3.6, 0.1) | 1.0 (−0.60, 2.64) | −0.5 (−2.1, 1.2) | ||
| Low vs. normal | −0.3 (−2.96, 2.28) | −0.1 (−2.1, 1.9) | 1.6 (−0.46, 3.50) | −3.3 (−6.1, −0.6) | ||
| Baseline Anemia Severity | ||||||
| No Anemia | 91.4 ± 0.7 | 98.0 ± 0.6 | 97.6 ± 0.6 | 98.9 ± 0.6 | <0.01 | |
| Mild | −1.1 (−3.3, 1.2) | −0.6 (−2.8, 1.6) | 0.9 (−0.8, 2.6) | −1.9 (−4.2, 0.4) | ||
| Moderate/Severe | −4.5 (−3.5, −0.9) | 1.0 (−1.3, 3.3) | −0.9 (−3.9, 2.1) | 1.3 (−0.9, 3.5) | ||
| Change in Anemia from Enrolment | Absolute CD4 Cell Count | Body Mass Index * | Quality of Life |
|---|---|---|---|
| Anemia Persistence | Adjusted Mean ± SE | Adjusted Mean ± SE | Adjusted Mean ± SE |
| Mean Difference (95% CI) | Mean Difference (95% CI) | Mean Difference (95% CI) | |
| Group 1 (n = 166) † | 309 ± 11.6 | 24.7 ± 0.17 | 98.0 ± 0.67 |
| Group 2 (n = 58) | 27.4 (−16.8, 71.5) | 1.14 (0.49, 1.80) | −0.26 (−2.8, 2.3) |
| Group 3 (n = 35) | −34.6 (−80.0, 20.9) | 1.42 (0.61, 0.60) | 2.4 (−0.8, 5.7) |
| Group 4 (n = 55) | −46.2 (−91.2, −1.26) | −0.37 (−1.02, 0.28) | −0.47 (−0.31, 2.1) |
| Group 5 (n = 19) | −48.9 (−118.6, 20.9) | −0.40 (−1.40, 0.60) | 2.1 (−1.93, 6.2) |
| Group 6 (n = 39) | −72.8 (−125.7, −19.8) | 0.23 (−0.54, 1.01) | −2.1 (−5.2, 1.04) |
| p-value (Log Rank test) | 0.01 | <0.0001 | 0.24 |
| Hematologic Status Indicator | Number of Events/Person-Months at Risk | Unadjusted Association Hazard Ratio (95% CI) | Adjusted Association ** Hazard Ratio (95% CI) |
|---|---|---|---|
| Serum Ferritin | |||
| High | 32/2477 | 1.83 (1.04, 3.27) | 1.75 (0.92, 3.3) |
| Low | 11/1073 | 1.56 (0.73, 3.30) | 1.86 (0.85, 4.1) |
| Normal Ferritin | 18/2608 | Ref | Ref |
| Baseline Anemia | |||
| Present | 40/2906 | 2.09 (1.23, 3.55) | 2.03 (1.18, 3.56) |
| Absent | 21/3388 | Ref | Ref |
| Baseline Anemia Severity | |||
| No Anemia | 21/3388 | Ref | Ref |
| Mild Anemia | 22/1840 | 3.4 (1.89, 6.23) | 3.9 (2.1, 7.2) |
| Moderate/Severe | 18/1146 | 6.3 (3.34, 11.7) | 6.7 (3.6, 12.7) |
| Anemia Persistence *** | |||
| Group 1 | 15/2767 | Ref | Ref |
| Group 2 | 6/882 | 1.2 (0.5, 3.2) | 1.11 (0.4, 2.9) |
| Group 3 | 6/538 | 2.2 (0.8, 5.6) | 2.1 (0.8, 5.5) |
| Group 4 | 11/862 | 2.27 (1.04, 4.96) | 2.27 (1.01, 5.1) |
| Group 5 | 10/393 | 4.74 (2.1,10.6) | 4.70 (2.0,10.9) |
| Group 6 | 13/753 | 3.0 (1.4, 6.4) | 3.1 (1.4, 6.5) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ezeamama, A.E.; Guwatudde, D.; Sikorskii, A.; Kabagambe, E.K.; Spelts, R.; Vahey, G.; Fenton, J.I.; Fawzi, W.W. Impaired Hematologic Status in Relation to Clinical Outcomes among HIV-Infected Adults from Uganda: A Prospective Cohort Study. Nutrients 2018, 10, 475. https://doi.org/10.3390/nu10040475
Ezeamama AE, Guwatudde D, Sikorskii A, Kabagambe EK, Spelts R, Vahey G, Fenton JI, Fawzi WW. Impaired Hematologic Status in Relation to Clinical Outcomes among HIV-Infected Adults from Uganda: A Prospective Cohort Study. Nutrients. 2018; 10(4):475. https://doi.org/10.3390/nu10040475
Chicago/Turabian StyleEzeamama, Amara E., David Guwatudde, Alla Sikorskii, Edmond K. Kabagambe, Raybun Spelts, Grace Vahey, Jenifer I. Fenton, and Wafaie W. Fawzi. 2018. "Impaired Hematologic Status in Relation to Clinical Outcomes among HIV-Infected Adults from Uganda: A Prospective Cohort Study" Nutrients 10, no. 4: 475. https://doi.org/10.3390/nu10040475
APA StyleEzeamama, A. E., Guwatudde, D., Sikorskii, A., Kabagambe, E. K., Spelts, R., Vahey, G., Fenton, J. I., & Fawzi, W. W. (2018). Impaired Hematologic Status in Relation to Clinical Outcomes among HIV-Infected Adults from Uganda: A Prospective Cohort Study. Nutrients, 10(4), 475. https://doi.org/10.3390/nu10040475




